Colour Counts: Sunlight and Skin Type as Drivers of Vitamin D Deficiency at UK Latitudes
Abstract
:1. Introduction
2. Materials and Methods
3. Results
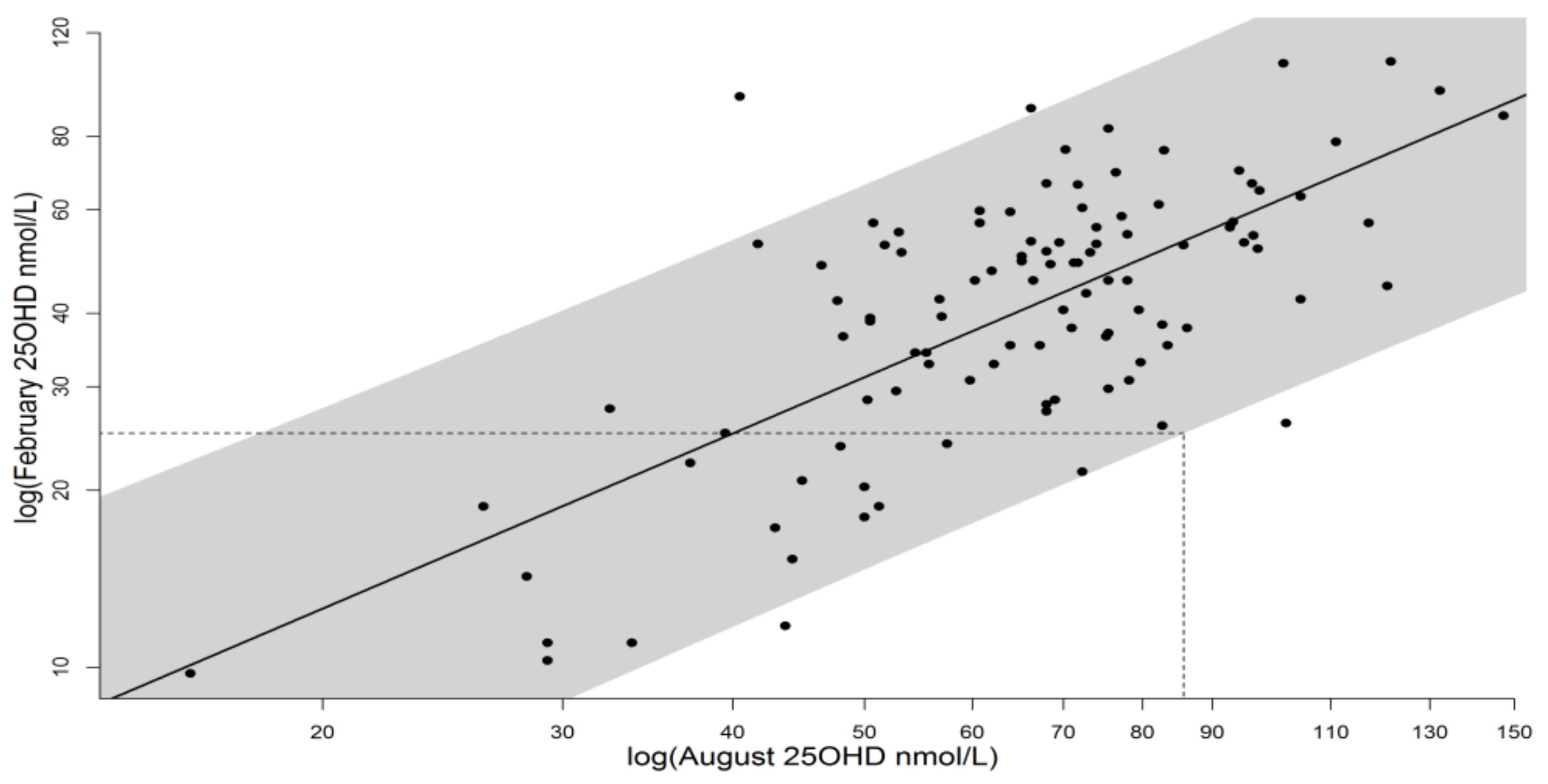
3.1. Input to the Vitamin D Synthesis Calculations
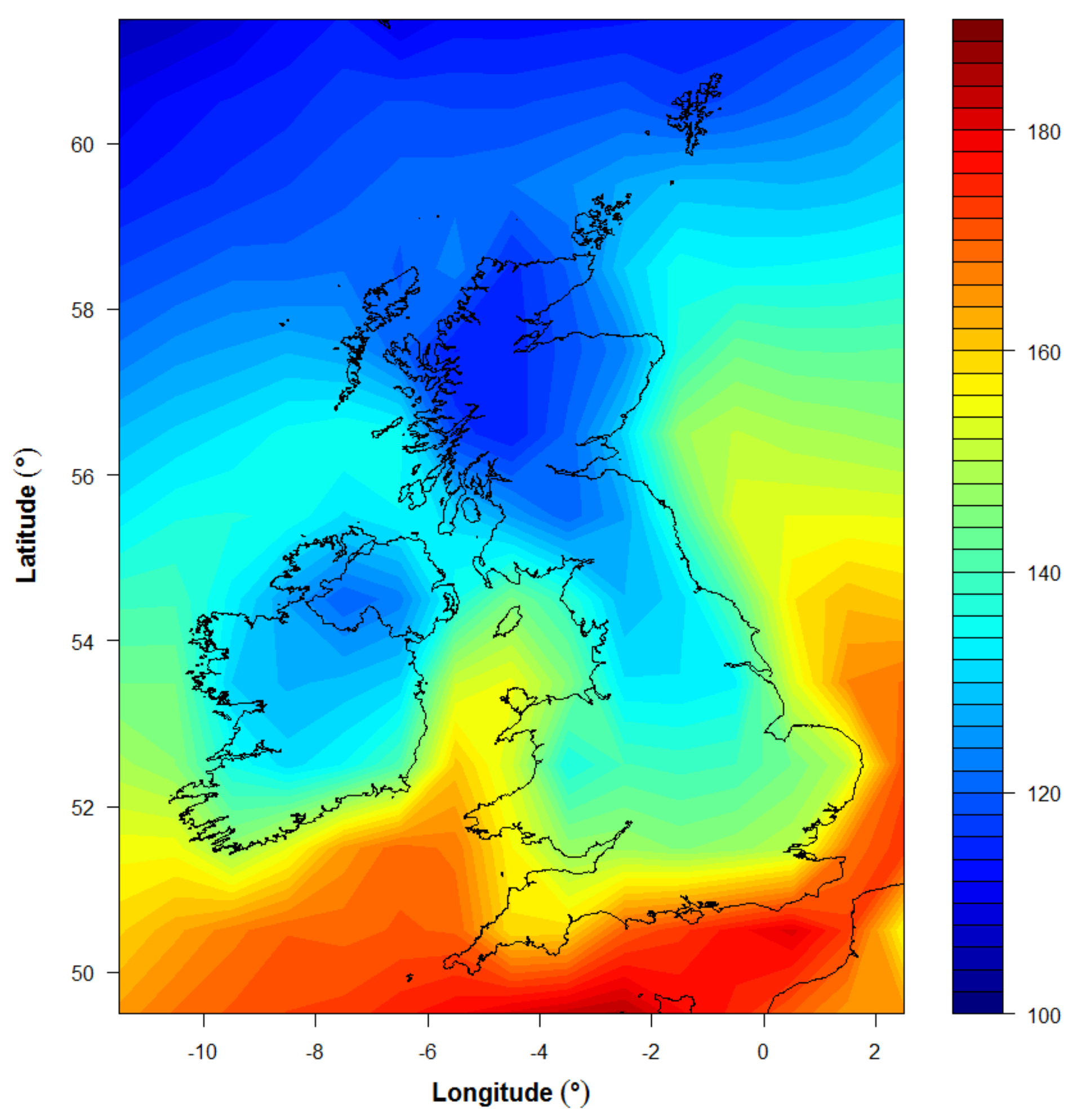
3.2. Application for UK Climatology
4. Discussion
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Pfotenhauer, K.M.; Shubrook, J.H. Vitamin D deficiency, Its Role in Health and Disease, and Current Supplementation Recommendations. Am. Osteopath. Assoc. 2017, 117, 301–305. [Google Scholar] [CrossRef] [PubMed]
- Webb, A.R.; Holick, M.F. The role of sunlight in the cutaneous production of vitamin D3. In Annual Review of Nutrition; Olson, R.E., Ed.; Annual Reviews Inc.: Palo Alto, CA, USA, 1988; Volume 8, pp. 375–399. ISBN 0-8243-2808-6. [Google Scholar]
- Cashman, K.; Kazantzidis, A.; Webb, A.R.; Keily, M. An integrated predictive model of population serum 25-hydroxycholecalciferol for application in strategy development for vitamin D deficiency prevention. J. Nutr. 2015, 145, 2419–2425. [Google Scholar] [CrossRef] [PubMed]
- Institute of Medicine. Dietary Reference Intakes for Calcium and Vitamin D; The National Academies Press: Washington, DC, USA, 2011; ISBN 978-0-309-16394-1. [Google Scholar]
- SACN Vitamin D and Health. 2016. Available online: https://www.gov.uk/government/publications/sacn-vitamin-d-and-health-report (accessed on 21 February 2018).
- Webb, A.R. Who, What, Where and When? Influences on cutaneous vitamin D synthesis. Prog. Biophys. Mol. Biol. 2006, 92, 17–25. [Google Scholar] [CrossRef] [PubMed]
- Farrar, M.D.; Kift, R.; Felton, S.J.; Berry, J.L.; Durkin, M.T.; Allan, D.; Vail, A.; Webb, A.R.; Rhodes, L.E. Recommended summer sunlight exposure levels fail to produce sufficient vitamin D status in UK adults of South Asian origin. Am. J. Clin. Nutr. 2011, 94, 1219–1224. [Google Scholar] [CrossRef] [PubMed]
- O’Neill, C.M.; Kazantzidis, A.; Ryan, M.J.; Barber, N.; Sempos, C.T.; Durazo-Arvizu, R.A.; Jorde, R.; Grimnes, G.; Eiriksdottir, G.; Gudnason, V.; et al. Seasonal Changes in vitamin D-effective UVB Availability in Europe and Associations with Population Serum 25-Hydroxyvitamin D. Nutrients 2016, 8, 533. [Google Scholar] [CrossRef] [PubMed]
- NICE Guideline. Sunlight Exposure: Risks and Benefits. 2016. Available online: https://www.nice.org.uk/guidance/ng34/resources/sunlight-exposure-risks-and-benefits-1837392363205 (accessed on 20 February 2018).
- Kift, R.; Berry, J.L.; Vail, A.; Durkin, M.T.; Rhodes, L.E.; Webb, A.R. Lifestyle factors including less cutaneous sun exposure contribute to starkly lower vitamin D status in UK South Asians compared to the white Caucasian population. Br. J. Dermatol. 2013, 169, 1272–1278. [Google Scholar] [CrossRef] [PubMed]
- Farrar, M.D.; Webb, A.R.; Kift, R.; Durkin, M.T.; Allan, D.; Herbert, A.; Berry, J.L.; Rhodes, L.E. Efficacy of a dose-range of simulated sunlight exposures in raising vitamin D status in South Asian adults: Implications for targeted guidance on sun exposure. Am. J. Clin. Nutr. 2013, 97, 1210–1216. [Google Scholar] [CrossRef] [PubMed]
- Kazantzidis, A.; Smedley, A.R.D.; Kift, R.C.; Rimmer, J.S.; Berry, J.L.; Rhodes, L.E.; Jorde, R.; Grimnes, G.; Eiriksdottir, G.; Gudnason, V.; et al. Modeling approach to determine how much UV radiation is available across the UK and Ireland for health risk and benefit studies. Photochem. Photobiol. Sci. 2015, 14, 1073–1081. [Google Scholar] [CrossRef] [PubMed]
- Webb, A.R.; Kazantzidis, A.; Kift, R.C.; Farrar, M.D.; Wilkinson, J.; Rhodes, L.E. Meeting vitamin D requirements at UK latitudes: Providing a choice. Nutrients 2018. in review. [Google Scholar]
- Webb, A.R.; Kift, R.; Durkin, M.T.; O’brien, S.J.; Vail, A.; Berry, J.L.; Rhodes, L.E. The role of sunlight exposure in determining the vitamin D status of the UK white Caucasian adult population. Br. J. Dermatol. 2010, 163, 1050–1055. [Google Scholar] [CrossRef] [PubMed]
- Webb, A.R.; Kift, R.; Berry, J.L.; Rhodes, L.E. The vitamin D debate: Translating controlled experiments into reality for human sun exposure times. Photochem. Photobiol. 2011, 87, 741–745. [Google Scholar] [CrossRef] [PubMed]
- Rhodes, L.E.; Webb, A.R.; Fraser, H.; Kift, R.; Durkin, M.; Vail, A. Recommended Summer Sunlight Exposure Levels Can Produce Sufficient (>20 ng mL−1) but Not the Proposed Optimal (>32 ng mL−1) 25(OH)D Levels at UK Latitudes. J. Investig. Dermatol. 2010, 130, 1411–1418. [Google Scholar] [CrossRef] [PubMed]
- Fitzpatrick, T.B. The validity and practicality of sun-reactive skin type I through VI. Arch. Dermatol. 1988, 124, 869–871. [Google Scholar] [CrossRef] [PubMed]
- Webb, A.R.; Aseem, S.; Kift, R.; Rhodes, L.E.; Farrar, M.D. Target the message: A qualitative study exploring knowledge and cultural attitudes to sunlight and vitamin D in Greater Manchester, UK. Br. J. Dermatol. 2016, 175, 1401–1403. [Google Scholar] [CrossRef] [PubMed]
- Springbett, P.; Buglass, S.; Young, A.R. Photoprotection and vitamin D status. J. Photochem. Photobiol. B. 2010, 101, 160–168. [Google Scholar] [CrossRef] [PubMed]
- European Food Safety Authority (EFSA). Panel on Dietetic Products. Dietary reference values for vitamin D. EFSA J. 2016, 14, 4547. [Google Scholar]
| Model Parameters and Summary Results | |
|---|---|
| End summer month | August |
| End summer 25(OH)D target + (nmol/L) | 85.8 |
| Monthly 25(OH)D spend (nmol/L/month) | 6.25 |
| Summer dose required (SED) | 89.6 * |
| Acceptable daily dose (SED) | 2.75 |
| Time for fixed daily dose (S1–3), (minutes) | 25 |
| Time range (S4) for daily dose of 2.75 SED at noon in June. Time (minutes) varies with latitude from southern England to northern Scotland | 25–40 |
| S1: 35% skin area March–September, maintains 25(OH)D status | Y |
| S2: 10% skin area March–May + September plus 35% skin area June–August, maintains 25(OH)D status | Y ** |
| S3: 10% skin area all summer, maintains 25(OH)D status | N |
| S4: 35% skin area, June–August, D adjusted for latitude to give 2.75 SED, maintains 25(OH)D status | Y |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Webb, A.R.; Kazantzidis, A.; Kift, R.C.; Farrar, M.D.; Wilkinson, J.; Rhodes, L.E. Colour Counts: Sunlight and Skin Type as Drivers of Vitamin D Deficiency at UK Latitudes. Nutrients 2018, 10, 457. https://doi.org/10.3390/nu10040457
Webb AR, Kazantzidis A, Kift RC, Farrar MD, Wilkinson J, Rhodes LE. Colour Counts: Sunlight and Skin Type as Drivers of Vitamin D Deficiency at UK Latitudes. Nutrients. 2018; 10(4):457. https://doi.org/10.3390/nu10040457
Chicago/Turabian StyleWebb, Ann R., Andreas Kazantzidis, Richard C. Kift, Mark D. Farrar, Jack Wilkinson, and Lesley E. Rhodes. 2018. "Colour Counts: Sunlight and Skin Type as Drivers of Vitamin D Deficiency at UK Latitudes" Nutrients 10, no. 4: 457. https://doi.org/10.3390/nu10040457
APA StyleWebb, A. R., Kazantzidis, A., Kift, R. C., Farrar, M. D., Wilkinson, J., & Rhodes, L. E. (2018). Colour Counts: Sunlight and Skin Type as Drivers of Vitamin D Deficiency at UK Latitudes. Nutrients, 10(4), 457. https://doi.org/10.3390/nu10040457






