Rebelling against the (Insulin) Resistance: A Review of the Proposed Insulin-Sensitizing Actions of Soybeans, Chickpeas, and Their Bioactive Compounds
Abstract
:1. Introduction
2. The Search Strategy Used for This Review
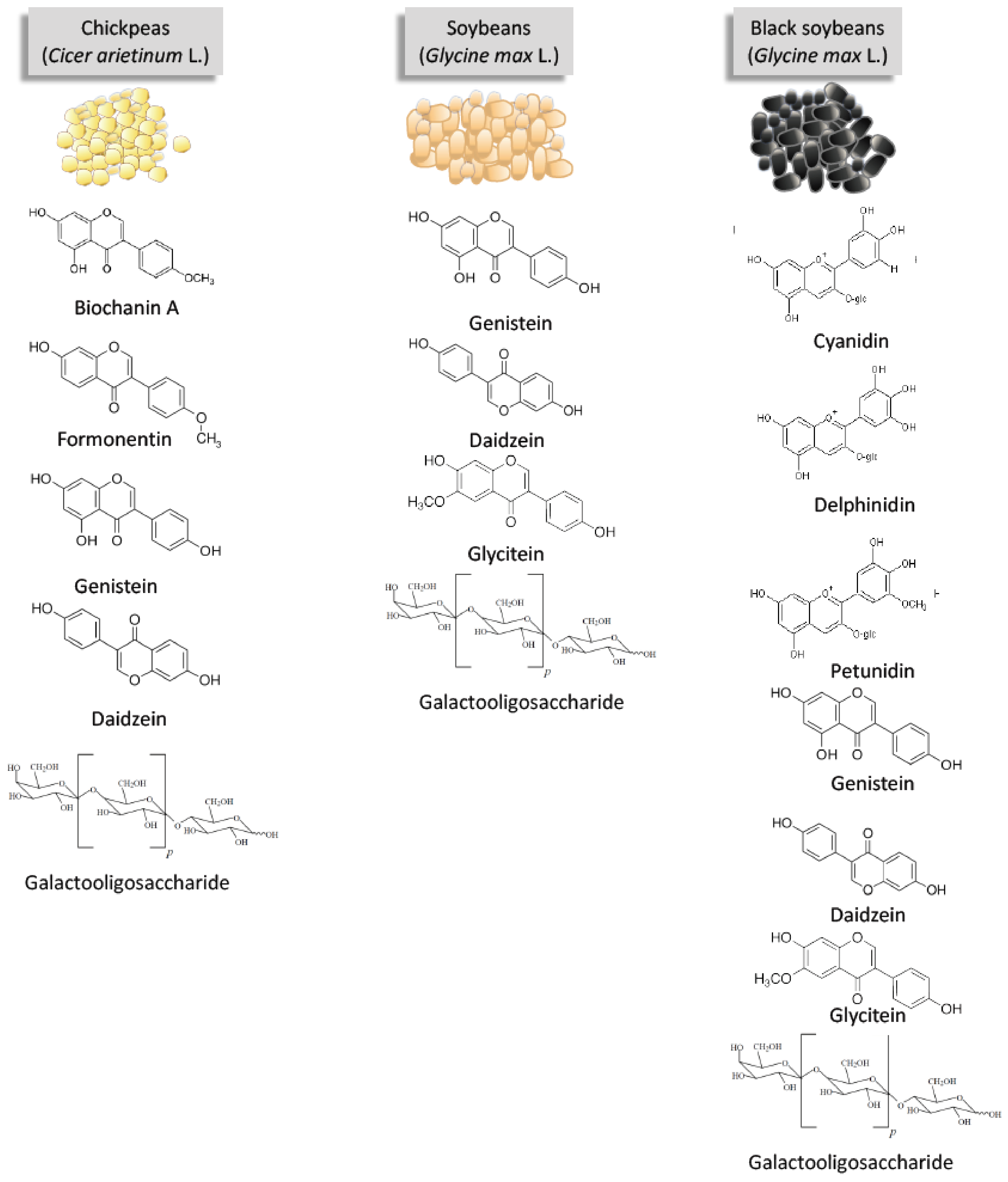
3. The Bioactive Compounds of Soybeans and Pulses Discussed in This Review
3.1. Isoflavones
3.2. Anthocyanins
3.3. Galactooligosaccharides
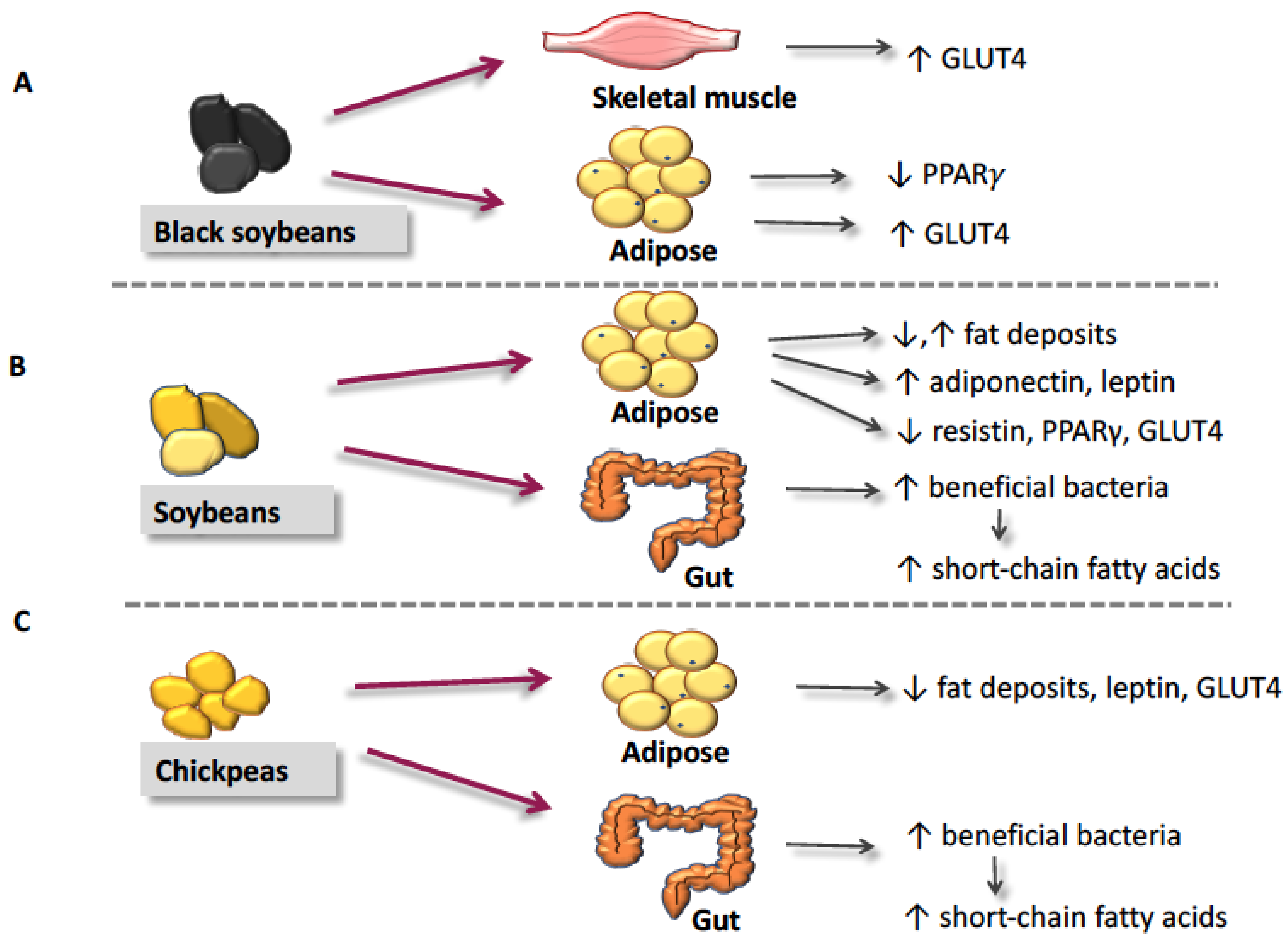
4. The Proposed Mechanisms of Action for Reduction of Insulin Resistance by Soybeans and Pulses
4.1. Glucose Transporter-4 and Glucose Utilization
4.2. Peroxisome Proliferator-Activated Receptor-γ
4.3. Fat Deposition & Metabolism
4.4. Adipokines
4.4.1. Adiponectin
4.4.2. Leptin
4.4.3. Resistin
4.5. Short-Chain Fatty Acids and Gut Microflora
5. General Summary and Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Zhang, H.M.; Chen, S.W.; Zhang, L.S.; Feng, X.F. The effects of soy isoflavone on insulin sensitivity and adipocytokines in insulin resistant rats administered with high-fat diet. Nat. Prod. Res. 2008, 22, 1637–1649. [Google Scholar] [CrossRef] [PubMed]
- Artunc, F.; Schleicher, E.; Weigert, C.; Fritsche, A.; Stefan, N.; Häring, H.U. The impact of insulin resistance on the kidney and vasculature. Nat. Rev. Nephrol. 2016, 12, 721–737. [Google Scholar] [CrossRef] [PubMed]
- Park, S.E.; Park, C.Y.; Sweeney, G. Biomarkers of insulin sensitivity and insulin resistance: Past, present and future. Crit. Rev. Clin. Lab. Sci. 2014, 52, 180–190. [Google Scholar] [CrossRef]
- National Institute of Diabetes and Digestive and Kidney Disorders. Available online: https://www.niddk.nih.gov/health-information/diabetes/overview/what-is-diabetes/prediabetes-insulin-resistance (accessed on 7 December 2017).
- Llaneza, P.; González, C.; Fernández-Iñarrea, J.; Alonso, A.; Díaz, F.; Péréz-López, F.R. Soy isoflavones improve insulin sensitivity without changing serum leptin among postmenopausal women. Climacteric 2012, 15, 611–620. [Google Scholar] [CrossRef] [PubMed]
- Medscape. Available online: https://emedicine.medscape.com/article/122501-medication (accessed on 29 November 2017).
- Gandhi, G.R.; Jothi, G.; Antony, P.J.; Balakrishna, K.; Paulraj, M.G.; Ignacimuthu, S.; Stalin, A.; Al-Dhabi, N.A. Gallic acid attenuates high-fat diet fed-streptozotocin-induced insulin resistance via partial agonism of PPARγ in experimental type 2 diabetic rats and enhances glucose uptake through translocation and activation of GLUT4 in PI3K/p-Akt signaling pathway. Eur. J. Pharmacol. 2014, 745, 201–216. [Google Scholar] [CrossRef] [PubMed]
- Afshin, A.; Micha, R.; Khatibzadeh, S.; Mozaffarian, D. Consumption of nuts and legumes and risk of incident ischemic heart disease, stroke, and diabetes: A systematic review and meta-analysis. Am. J. Clin. Nutr. 2014, 100, 278–288. [Google Scholar] [CrossRef] [PubMed]
- Polak, R.; Phillips, E.M.; Campbell, A. Legumes: Health benefits and culinary approaches to increase intake. Clin. Diabetes 2015, 33, 198–205. [Google Scholar] [CrossRef] [PubMed]
- Rebello, C.J.; Greenway, F.L.; Finley, J.W. A review of the nutritional value of legumes and their effects on obesity and its related co-morbidities. Obes. Rev. 2014, 15, 392–407. [Google Scholar] [CrossRef] [PubMed]
- Blair, R.M.; Henley, E.C.; Tabor, A. Soy foods have low glycemic and insulin response indices in normal weight subjects. Nutr. J. 2006, 5, 35. [Google Scholar] [CrossRef]
- Mudryj, A.N.; Yu, N.; Aukema, H.M. Nutritional and health benefits of pulses. Appl. Physiol. Nutr. Metab. 2014, 39, 1197–1204. [Google Scholar] [CrossRef] [PubMed]
- Thompson, S.V.; Winham, D.M.; Hutchins, A.M. Bean and rice meals reduce postprandial glycemic response in adults with type 2 diabetes: A cross-over study. Nutr. J. 2012, 11, 23. [Google Scholar] [CrossRef] [PubMed]
- Pulse Canada. Available online: http://www.pulsecanada.com/about-pulse-canada/what-is-a-pulse/ (accessed on 29 January 2018).
- Anderson, J.W.; Smith, B.M.; Washnock, C.S. Cardiovascular and renal benefits of dry bean and soybean intake. Am. J. Clin. Nutr. 1999, 70, 464s–474s. [Google Scholar] [CrossRef] [PubMed]
- Brar, G.S.; Carter, T.E., Jr. Soybean: Glycine max (L.) Merrill. In Genetic Improvements of Vegetable Crops; Kalloo, G., Bergh, B.O., Eds.; Pergamon Press: Oxford, UK, 1993; Chapter 30; pp. 427–463. [Google Scholar]
- Choi, M.S.; Ryu, R.; Seo, Y.R.; Jeong, T.S.; Shin, D.H.; Park, Y.B.; Kim, S.R.; Jung, U.J. The beneficial effects of soybean (Glycine max (L.) Merr.) leaf extracts in adults with prediabetes: A randomized placebo controlled trial. Food Funct. 2014, 5, 1621–1630. [Google Scholar] [CrossRef] [PubMed]
- Choquette, S.; Riesco, E.; Cormier, E.; Dion, T.; Aubertin-Leheudre, M.; Dionne, I.J. Effects of soya isoflavones and exercise on body composition and clinical risk factors of cardiovascular diseases in overweight postmenopausal women: A 6-month double-blind controlled trial. Br. J. Nutr. 2011, 105, 1199–1209. [Google Scholar] [CrossRef] [PubMed]
- Fei, B.B.; Ling, L.; Hua, C.; Ren, S.Y. Effects of soybean oligosaccharides on antioxidant enzyme activities and insulin resistance in pregnant women with gestational diabetes mellitus. Food Chem. 2014, 158, 429–432. [Google Scholar] [CrossRef] [PubMed]
- Jamilian, M.; Asemi, Z. The effects of soy isoflavones on metabolic status of patients with polycystic ovary syndrome. J. Clin. Endocrinol. Metab. 2016, 101, 3386–3394. [Google Scholar] [CrossRef] [PubMed]
- Nilsson, A.; Johnsson, E.; Ekström, L.; Björck, I. Effects of a brown beans evening meal on metabolic risk markers and appetite regulating hormones at a subsequent standardized breakfast: A randomized cross-over study. PLoS ONE 2013, 8, e59985. [Google Scholar] [CrossRef] [PubMed]
- Reverri, E.J.; Randolph, J.M.; Steinberg, F.M.; Kappagoda, C.T.; Edirisinghe, I.; Burton-Freeman, B.M. Black beans, fiber, and antioxidant capacity pilot study: Examination of whole foods vs. functional components on postprandial metabolic oxidative stress, and inflammation in adults with metabolic syndrome. Nutrients 2015, 7, 6139–6154. [Google Scholar] [CrossRef] [PubMed]
- Mariangeli, C.P.; Jones, P.J. Whole and fractionated yellow pea flours reduce fasting insulin and insulin resistance in hypercholesterolaemic and overweight human subjects. Br. J. Nutr. 2011, 105, 110–117. [Google Scholar] [CrossRef] [PubMed]
- Johnson, S.K.; Thomas, S.J.; Hall, R.S. Palatability and glucose, insulin and satiety responses of chickpea floru and extruded chickpea flour bread eaten as part of a breakfast. Eur. J. Clin. Nutr. 2005, 59, 169–176. [Google Scholar] [CrossRef] [PubMed]
- Nestel, P.; Cehun, M.; Chronopoulos, A. Effects of long-term consumption and single meals of chickpeas on plasma glucose, insulin, and triacylglycerol concentrations. Am. J. Clin. Nutr. 2004, 79, 390–395. [Google Scholar] [CrossRef] [PubMed]
- Pittaway, J.K.; Robertson, I.K.; Ball, M.J. Chickpeas may influence fatty acid and fiber intake in an ad libitum diet, leading to small improvments in serum lipid profile and glycemic control. J. Am. Diet. Assoc. 2008, 108, 1009–1013. [Google Scholar] [CrossRef] [PubMed]
- Ye, Y.B.; Chen, A.L.; Lu, W.; Zhuo, S.Y.; Liu, J.; Guan, J.H.; Deng, W.P.; Fang, S.; Li, Y.B.; Chen, Y.M. Daidzein and genistein fail to improve glycemic control and insulin sensitivity in Chinese women with impaired glucose regulation: A double-blind, randomized, placebo-controlled trial. Mol. Nutr. Food Res. 2015, 59, 240–249. [Google Scholar] [CrossRef] [PubMed]
- Bourdon, I.; Olson, B.; Backus, R.; Richter, B.D.; Davis, P.A.; Schrieeman, B.O. Beans, as a source of dietary fibre, increase cholecystokinin and apolipoprotein B48 response to test meals in men. J. Nutr. 2001, 131, 1485–1490. [Google Scholar] [CrossRef] [PubMed]
- Winham, D.M.; Hutchins, A.M.; Johnston, C.S. Pinto bean consumption reduces biomarkers for heart disease risk. J. Am. Coll. Nutr. 2007, 26, 243–249. [Google Scholar] [CrossRef] [PubMed]
- Clark, J.L.; Zahradka, P.; Taylor, C.G. Efficacy of flavonoids in the management of high blood pressure. Nutr. Rev. 2015, 73, 799–822. [Google Scholar] [CrossRef] [PubMed]
- Ko, K.P. Isoflavones: Chemistry, analysis, functions and effects on health and cancer. Asian Pac. J. Cancer Prev. 2014, 15, 7001–7010. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.; Yao, Y.; Zhu, Y.; Ren, G. Isoflavones in chickpeas inhibit adipocyte differentiation and prevent insulin resistance in 3T3-L1 cells. J. Agric. Food Chem. 2015, 63, 9696–9703. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.; Bi, X.; Chen, B. Isoflavones: Anti-inflammatory benefit and possible caveats. Nutrients 2016, 10, 361. [Google Scholar] [CrossRef] [PubMed]
- Kim, U.H.; Yoon, J.H.; Li, H.; Kang, J.H.; Ji, H.S.; Park, K.H.; Shin, D.H.; Park, H.Y.; Jeong, T.S. Pterocarpan-enriched soy leaf extract ameliorates insulin sensitivity and pancreatic β-cell proliferation in type 2 diabetic mice. Molecules 2014, 19, 18493–18510. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Morató, J.; Farré, M.; Pérez-Mañá, C.; Papaseit, E.; Martínez-Riera, R.; de la Torre, R.; Pizarro, N. Pharmacokinetic comparision of soy isoflavone extracts in human plasma. J. Agric. Food Chem. 2015, 63, 6946–6953. [Google Scholar] [CrossRef] [PubMed]
- Belwal, T.; Nabavi, S.F.; Nabavi, S.M.; Habtemariam, S. Dietary anthocyanins and insulin resistance: When food becomes a medicine. Nutrients 2017, 9, 1111. [Google Scholar] [CrossRef] [PubMed]
- Hu, J.; Chen, G.; Zhang, Y.; Cui, B.; Yin, W.; Yu, X.; Zhu, Z.; Hu, Z. Anthocyanin composition and expression analysis of anthocyanin biosynthetic genes in kidney bean pod. Plant Phys. Biochem. 2015, 97, 304–312. [Google Scholar] [CrossRef] [PubMed]
- Nizamutdinova, I.T.; Jin, Y.C.; Chung, J.I.; Shin, S.C.; Lee, S.J.; Seo, H.G.; Lee, J.H.; Chang, K.C.; Kim, H.J. The anti-diabetic effect of anthocyanins in streptozotocin-induced diabetic rats through glucose transporter 4 regulation and prevention of insulin resistance and pancreatic apoptosis. Mol. Nutr. Food Res. 2009, 53, 1419–1429. [Google Scholar] [CrossRef] [PubMed]
- Koh, K.; Youn, J.E.; Kim, H.S. Identification of anthocyanins in black soybean (Glycine max (L.) Merr.) varieties. J. Food Sci. Technol. 2014, 51, 377–381. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.H.; Kang, N.S.; Shin, S.O.; Shin, S.H.; Lim, S.G.; Suh, D.Y.; Baek, I.Y.; Park, K.Y.; Ha, T.J. Characteristics of anthocyanins in the black soybean (Glycine max L.) by HPLC-DAD-ESI/MS analysis. Food Chem. 2009, 112, 226–231. [Google Scholar] [CrossRef]
- Dai, Z.; Lyu, W.; Xie, M.; Yuan, Q.; Ye, H.; Hu, B.; Zhou, L.; Zeng, X. Effects of α-galactooligosaccharides from chickpeas on high-fat-diet-induced metabolic syndrome in mice. J. Agric. Food Chem. 2017, 65, 3160–3166. [Google Scholar] [CrossRef] [PubMed]
- Niittynen, L.; Kajander, K.; Korpeia, R. Galacto-oligosaccharides and bowel function. Scand. J. Food Nutr. 2007, 51, 62–66. [Google Scholar] [CrossRef]
- U.S. Food and Drug Administration. Available online: https://www.fda.gov/downloads/Food/IngredientsPackagingLabeling/GRAS/NoticeInventory/ucm381400.pdf (accessed on 8 December 2017).
- Sangwan, V.; Tomar, S.K.; Ali, B.; Singh, R.R.B.; Singh, A.K. Hypoglycaemic effect of galactooligosaccharides in alloxan-induced diabetic rats. J. Diary Res. 2015, 82, 70–77. [Google Scholar] [CrossRef] [PubMed]
- Fu, Z.; Gilbert, E.R.; Liu, D. Regulation of insulin synthesis and secretion and pancreatic Beta-cell dysfunction in diabetes. Curr. Diabetes Rev. 2013, 9, 25–53. [Google Scholar] [CrossRef] [PubMed]
- Shan, W.F.; Chen, B.Q.; Zhu, S.J.; Jiang, L.; Zhou, Y.F. Effects of GLUT4 expression on insulin resistance in patients with advanced liver cirrhosis. J. Zheijang Univ. Sci. B 2011, 12, 677–682. [Google Scholar] [CrossRef] [PubMed]
- Mueckler, M. Insulin resistance and the disruption of GLUT4 trafficking in skeletal muscle. J. Clin. Investig. 2001, 107, 1211–1213. [Google Scholar] [CrossRef] [PubMed]
- Gannon, N.P.; Conn, C.A.; Vaughan, R.A. Dietary stimulators of GLUT4 expression and translocation in skeletal muscle: A mini-review. Mol. Nutr. Food Res. 2015, 59, 48–64. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.C.; Huang, W.C.; Hou, C.W.; Chi, Y.W.; Huang, H.Y. Effect of black soybean koji extract on glucose utilization and adipocyte differentiation in 3T3-L1 cells. Int. J. Mol. Sci. 2014, 15, 8280–8292. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Solana, B.; Laborda, J.; Baladrón, V. Mouse resistin modulates adipogenesis and glucose uptake in 3T3-L1 preadipocytes through the ROR1 receptor. Mol. Endocrinol. 2012, 26, 110–127. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Liu, B.L.; Liu, K.; Tang, N.; Huang, J.; An, Y.; Li, L. Establishment of the insulin-resistance induced by inflammatory response in 3T3-L1 preadipocytes cell line. Inflammation 2008, 31, 355–364. [Google Scholar] [CrossRef] [PubMed]
- Lo, K.A.; Labadorf, A.; Kennedy, N.J.; Han, M.S.; Yap, Y.S.; Matthews, B.; Xin, X.; Sun, L.; Davis, R.J.; Lodish, H.F.; et al. Analysis of in vitro insulin resistance models and their physiological relevance to in vivo diet-induced adipose insulin resistance. Cell Rep. 2013, 5, 259–270. [Google Scholar] [CrossRef] [PubMed]
- Inaguma, T.; Han, J.; Isoda, H. Improvement of insulin resistance by cyanidin 3-glucoside, anthocyanin from black beans through the up-regulation of GLUT4 gene expression. BMC Proc. 2011, 5, 21. [Google Scholar] [CrossRef] [PubMed]
- Luna-Vital, D.; Weiss, M.; Gonzalez de Mejia, E. Anthocyanins from purple corn ameliorated tumor necrosis factor-α-induced inflammation and insulin resistance in 3T3-L1 adipocytes via activation of insulin signaling and enhanced GLUT4 translocation. Mol. Nutr. Food Res. 2017, 61, 1700362. [Google Scholar] [CrossRef] [PubMed]
- Halzuík, M.M.; Halzuík, M. PPAR-α and insulin sensitivity. Physiol. Rev. 2006, 55, 115–122. [Google Scholar]
- Ferré, P. The biology of peroxisome proliferator-activated receptors: Relationship with lipid metabolism and insulin sensitivity. Diabetes 2004, 53, S43–S50. [Google Scholar] [CrossRef] [PubMed]
- Leonardini, A.; Laviola, L.; Perrini, S.; Natalicchio, A.; Giorgino, F. Cross-talk between PPARγ and insulin signaling and modulation of insulin sensitivity. PPAR Res. 2009, 2009, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Olefsky, J.M.; Saltiel, A.R. PPARγ and the treatment of insulin resistance. Trends Endocrinol. Metab. 2000, 11, 362–368. [Google Scholar] [CrossRef]
- Wagner, J.D.; Zhang, L.; Shadoan, M.K.; Kavanagh, K.; Chen, H.; Trenasari, K.; Kaplan, J.R.; Adams, M.R. Effects of soy protein and isoflavones on insulin resistance and adiponectin in male monkeys. Metabolism 2008, 57, 524–531. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.K.; Kim, J.N.; Han, S.N.; Nam, J.H.; Na, H.N.; Ha, T.J. Black soybean anthocyanins inhibit adipocyte differentiation in 3T3-L1 cells. Nutr. Res. 2012, 32, 770–777. [Google Scholar] [CrossRef] [PubMed]
- Kadowaki, T.; Hara, K.; Kubota, N.; Tobe, K.; Terauchi, Y.; Yamauchi, T.; Eto, K.; Kadowaki, H.; Noda, M.; Hagura, R.; et al. The role of PPARγ in high-fat diet-induced obesity and insulin resistance. J. Diabetes Complicat. 2002, 16, 41–45. [Google Scholar] [CrossRef]
- Metzger, D.; Imai, T.; Jiang, M.; Takukawa, R.; Desvergne, B.; Wahil, W.; Chambon, P. Functional role of RXRs and PPARγ in mature adipocytes. Prostagland. Leukot. Essent. Fat. Acids 2005, 73, 51–58. [Google Scholar] [CrossRef] [PubMed]
- Kubota, N.; Terauchi, Y.; Miki, H.; Tamemoto, H.; Yamauchi, T.; Komeda, K.; Saton, S.; Nakano, R.; Ishii, C.; Sugiyama, T.; et al. PPARγ mediates high-fat diet-induced adipocyte hypertrophy and insulin resistance. Mol. Cell 1999, 4, 597–609. [Google Scholar] [CrossRef]
- Ali, A.A.; Velasquez, M.T.; Hansen, C.T.; Mohamed, A.I.; Bhathena, S.J. Effects of soybean isoflavones, probiotics, and their interactions on lipid metabolism and endocrine system in an animal model of obesity and diabetes. J. Nutr. Biochem. 2004, 15, 583–590. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.S.; Koh, I.U.; Song, J. Genistein reduced insulin resistance index through modulating lipid metabolism in ovariectomized rats. Nutr. Res. 2012, 32, 844–855. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Zhou, L.; Gu, Y.; Zhang, Y.; Tang, J.; Li, F.; Shang, W.; Jiang, B.; Yue, W.; Chen, M. Dietary chickpeas reverse visceral adiposity, dyslipidaemia and insulin resistance in rats induced by a chronic high-fat diet. Br. J. Nutr. 2007, 98, 720–726. [Google Scholar] [CrossRef] [PubMed]
- El-Adawy, T.A. Nutritional composition and anti-nutritional factors of chickpeas (Cicer arietinum L.) undergoing different cooking methods and germination. Plant Foods Hum. Nutr. 2002, 57, 83–92. [Google Scholar] [CrossRef] [PubMed]
- Pulse Canada. Available online: http://www.pulsecanada.com/wp-content/uploads/2017/12/Cooking-With-Pulses.pdf (accessed on 29 January 2018).
- Hernandez, C.M.; Høifødt, H.; Terry, A.V., Jr. Spontaneously hypertensive rats: Further evaluation of age-related memory performance and cholinergic marker expression. J. Psychiatry Neurosci. 2003, 28, 197–209. [Google Scholar] [PubMed]
- Zanella, I.; Marrazzo, E.; Biasiotto, G.; Penza, M.; Romani, A.; Vignolini, P.; Caimi, L.; Di Lorenzo, D. Soy and the soy isoflavone genistein promote adipose tissue development in male mice on a low-fat diet. Eur. J. Nutr. 2015, 54, 1095–1107. [Google Scholar] [CrossRef] [PubMed]
- Vidal-Puigi, A.; O’Rahilly, S. Resistin: A new link between obesity and insulin resistance. Clin. Endocrinol. 2001, 55, 437–438. [Google Scholar] [CrossRef]
- Kwon, H.; Pessin, J.E. Adipokines mediate inflammation and insulin resistance. Front. Endocrinol. (Lausanne) 2013, 4, 71. [Google Scholar] [CrossRef] [PubMed]
- Rabe, K.; Lehrke, M.; Parhofer, K.G.; Broedl, U.C. Adipokines and insulin resistance. Mol. Med. 2008, 14, 741–751. [Google Scholar] [CrossRef] [PubMed]
- Guilherme, A.; Virbasius, J.V.; Puri, V.; Czech, M.P. Adipocyte dysfunctions linking obesity to insulin resistance and type 2 diabetes. Nat. Rev. Mol. Cell Biol. 2008, 9, 367–377. [Google Scholar] [CrossRef] [PubMed]
- Jung, U.J.; Choi, M.S. Obesity and its metabolic complications: The role of adipokines and the relationship between obesity, inflammation, insulin resistance, dyslipidemia, and nonalcoholic fatty liver disease. Int. J. Mol. Sci. 2014, 15, 6184–6223. [Google Scholar] [CrossRef] [PubMed]
- Kashino, I.; Nanri, A.; Kurotani, K.; Akter, S.; Yasuda, K.; Sato, M.; Hayabuchi, H.; Mizoue, T. Association of dietary patterns with serum adipokines among Japanese: A cross-sectional study. Nutr. J. 2015, 14, 58. [Google Scholar] [CrossRef] [PubMed]
- Clark, J.L.; Taylor, C.G.; Zahradka, P. Exploring the cardio-metabolic relevance of T-cadherin: A pleiotropic adiponectin receptor. Endocrinol. Metab. Immune Disord. Drug. Targets 2017, 17, 200–206. [Google Scholar] [CrossRef] [PubMed]
- Balsan, G.A.; da Costa Vieira, J.L.; de Oliveira, A.M.; Portal, V.L. Relationship between adiponectin, obesity, and insulin resistance. Rev. Assoc. Med. Bras. 2015, 61, 72–80. [Google Scholar] [CrossRef] [PubMed]
- Han, J.; Inaguma, T.; Isoda, H. Cyanidin-3-glucoside anthocyanin from black beans has potential to protect insulin resistance on 3T3-L1 adipocytes by inhibiting TNF-α release. Br. J. Nutr. 2018, in press. [Google Scholar]
- Kavanagh, K.; Jones, K.L.; Zhang, L.; Flynn, D.M.; Shadoan, M.K.; Wagner, J.D. High isoflavone soy diet increases insulin secretion without decreasing insulin sensitivity in premenopausal nonhuman primates. Nutr. Res. 2008, 28, 368–376. [Google Scholar] [CrossRef] [PubMed]
- Antuna-Puente, B.; Feve, B.; Fellahi, S.; Bastard, J.P. Adipokines: The missing link between insulin resistance and obesity. Diabetes Metab. 2008, 34, 2–11. [Google Scholar] [CrossRef] [PubMed]
- Paz-Filho, G.; Mastronardi, C.; Wang, M.L.; Licinio, J. Leptin therapy, insulin sensitivity, and glucose homeostasis. Indian Endocrinol. Metab. 2012, 16, S549–S555. [Google Scholar] [CrossRef] [PubMed]
- O’Connor, S.; Chouinard-Castonguay, S.; Gagnon, C.; Rudkowska, I. Prebiotics in the management of components of the metabolic syndrome. Maturitas 2017, 104, 11–18. [Google Scholar] [CrossRef] [PubMed]
- Weitkunat, K.; Stuhlmann, C.; Postel, A.; Rumberger, S.; Fankhänel, M.; Woting, A.; Petzke, K.J.; Gohlke, S.; Schulz, T.J.; Blaut, M.; et al. Short-chain fatty acids and inulin, but not guar gum, prevent diet-induced obesity and insulin resistance through differential mechanisms in mice. Sci. Rep. 2017, 7, 6109. [Google Scholar] [CrossRef] [PubMed]
- Murphy, E.A.; Velazquez, K.T.; Herbert, K.M. Influence of high-fat-diet on gut microbiota: A driving force for chronic disease risk. Curr. Opin. Clin. Nutr. Metab. Care 2015, 18, 515–520. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.L.; Kong, X.F.; Lian, G.Q.; Blachier, F.; Geng, M.M.; Yin, Y.L. Dietary supplementation with soybean oligosaccharides increases short-chain fatty acids but decreases protein-derived catabolites in the intestinal luminal content of weaned Huangjiang mini-piglets. Nutr. Res. 2014, 34, 780–788. [Google Scholar] [CrossRef] [PubMed]
- Fu, D.; Yang, H.S.; Kong, X.; Blachier, F.; Wang, W.; Yin, Y. Molecular cloning and expression profiling of excitatory amino acid carrier 1 in suckling Huangjiang mini-piglets with large or small body weight at birth. Mol. Biol. Rep. 2012, 40, 3341–3350. [Google Scholar] [CrossRef] [PubMed]
- Charles River. Available online: https://www.criver.com/products-services/find-model/cd-1-igs-mouse?region=3611 (accessed on 5 December 2017).
- Kavadi, P.K.; Pothuraj, R.; Chagalamarri, J.; Bhakri, G.; Mallepogu, A.; Sharma, R.K. Dietary incorporation of whey protein isolate and galactooligosaccharides exhibits improvement in glucose homeostasis and insulin resistance in high fat diet fed mice. J. Intercult. Ethnopharmacol. 2017, 6, 326–332. [Google Scholar] [CrossRef] [PubMed]
- Research Diets. Available online: http://www.researchdiets.com/resource-center-page/typical-food-intake (accessed on 29 January 2018).
- Stahel, P.; Kim, J.J.; Xiao, C.; Cant, J.P. Of the milk sugars, galactose, but not prebiotic galacto-oligosaccharide, improves insulin sensitivity in male Sprague Dawley rats. PLoS ONE 2017, 12, e0172260. [Google Scholar] [CrossRef] [PubMed]
- Canfora, E.E.; van der Beek, C.M.; Hermes, G.D.A.; Goossens, G.H.; Jocken, J.W.E.; Holst, J.J.; van Ejik, H.M.; Venema, K.; Smidt, H.; Zoetendal, E.G.; et al. Supplementation of diet with galacto-oligosaccharides increases Bifidobacteria, but not insulin sensitivity, in obese prediabetic individuals. Gastroenterology 2017, 153, 87–97. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Ge, X.; Tian, X.; Zhang, Y.; Zhang, J.; Zhang, P. Soy isoflavone: The multipurpose phytochemical (review). Biomed. Res. 2013, 1, 697–701. [Google Scholar] [CrossRef] [PubMed]
- Yoon, G.E.; Park, S. Antioxidant action of soy isoflavones on oxidative stress and antioxidant enzyme activities in exercised rats. Nutr. Res. Pract. 2014, 8, 618–624. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.F.; Zheng, Y.L.; Wu, D.M.; Hu, B.; Shan, Q.; Cheng, W.; Li, M.Q.; Sun, Y.Y. Purple sweet potato color attenuates hepatic insulin resistance via blocking oxidative stress in high-fat-diet-treated mice. J. Nutr. Biochem. 2013, 24, 1008–1018. [Google Scholar] [CrossRef] [PubMed]
- Medjakovic, S.; Mueller, M.; Jungbauer, A. Potential health-modulating effects of isoflavones and metabolites via activation of PPAR and AhR. Nutrients 2010, 2, 241–279. [Google Scholar] [CrossRef] [PubMed]
- Feng, S.; Reuss, L.; Wang, Y. Potential of natural products in the inhibition of adipogenesis through regulation of PPARγ expression and/or its transcriptional activity. Molecules 2016, 21, 1278. [Google Scholar] [CrossRef]
- Hossain, M.K.; Dayem, A.A.; Han, J.; Yin, Y.; Kim, K.; Saha, S.K.; Yang, G.M.; Choi, H.Y.; Cho, S.G. Molecular mechanisms of the anti-obesity and anti-diabetic properties of flavonoids. Int. J. Mol. Sci. 2016, 17, 569. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Wong, P.; Zuo, Y.; Zhao, M.; Chen, F. Health benefits of anthocyanins and molecular mechanisms: Update from recent decade. Crit. Rev. Food Sci. Nutr. 2017, 57, 1729–1741. [Google Scholar] [CrossRef] [PubMed]
- Tsuda, T.; Ueno, Y.; Aoki, H.; Koda, T.; Horio, F.; Takahashi, N.; Kawada, T.; Osawa, T. Anthocyanin enhances adipocytokine secretion and adipocyte-specific gene expression in isolated rat adipocytes. Biochem. Biophys. Res. Commun. 2004, 316, 148–157. [Google Scholar] [CrossRef] [PubMed]
- Huang, B.; Wong, Z.; Park, J.H.; Ryu, O.H.; Choi, M.K.; Lee, J.Y.; Kang, Y.H.; Lim, S.S. Anti-diabetic effect of purple corn extract on c57BL/KsJ db/db mice. Nutr. Res. Pract. 2015, 9, 22–29. [Google Scholar] [CrossRef] [PubMed]
- Chun, O.K.; Chung, S.J.; Sang, W.O. Urinary isoflavones and their metabolites validate the dietary isoflavone intakes in US adults. J. Am. Diet. Assoc. 2009, 109, 245–254. [Google Scholar] [CrossRef] [PubMed]
- Filiberto, A.C.; Mumford, S.L.; Pollack, A.Z.; Zhang, C.; Yeung, E.H.; Perkins, N.J.; Wactawski-Wende, J.; Schisterman, E.F. Habitual dietary isoflavone intake is associated with decreased C-reative protein concentrations among healthy premenopausal women. J. Nutr. 2013, 143, 900–906. [Google Scholar] [CrossRef] [PubMed]
- Pop, E.A.; Fischer, L.M.; Coan, A.D.; Gitzinger, M.; Nakamura, J.; Zeisel, S.H. Effects of a high daily dose of soy isoflavones on DNA damage, apoptosis and estrogenic outcomes in healthy, postmenopausal women–A Phase I clinical trial. Menopause 2008, 15, 684–692. [Google Scholar] [CrossRef] [PubMed]
- Van der Velpen, V.; Hollman, P.C.; van Nielen, M.; Schouten, E.G.; Mensink, M.; van’t Veer, P.; Geelen, P. Large inter-individual variation in isoflavone plasma concentration limits use of isoflavone intake data for risk assessment. Eur. J. Clin. Nutr. 2014, 68, 1141–1147. [Google Scholar] [CrossRef] [PubMed]
- Wallace, T.C. Anthocyanins. Adv. Nutr. 2015, 6, 620–622. [Google Scholar] [CrossRef] [PubMed]
- Grosso, G.; Stepaniak, U.; Topor-Mądry, R.; Szafraniec, K.; Pająk, A. Estimated dietary intake and major food sources of polyphenols in the Polish arm of the HAPIEE study. Nutrition 2014, 30, 1398–1403. [Google Scholar] [CrossRef] [PubMed]
- Khoo, H.E.; Azlan, A.; Tang, S.T.; Lim, S.M. Anthocyanidins and anthocyanins: Colored pigments as food, pharmaceutical ingredients, and the potential health benefits. Food Nutr. Res. 2017, 61, 1361779. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.; Beecher, G.R.; Holden, J.M.; Haytowitz, D.B.; Gebhardt, S.E.; Prior, R.L. Concentrations of anthocyanins in common foods in the United States and estimation of normal consumption. J. Agric. Food Chem. 2006, 54, 4069–4075. [Google Scholar] [CrossRef] [PubMed]
- Laurentin, A.; Edwards, C.A. Carbohydrates: Resistance starch and oligosaccharides. In Guide to Nutritional Supplements; Caballero, E., Ed.; Elsevier Ltd.: Oxford, UK, 2009; pp. 84–92. [Google Scholar]
- Han, I.H.; Baik, B.Y. Oligosaccharide content and composition of legumes and their reduction by soaking, cooking, ultrasound, and high hydrostatic pressure. Cereal Chem. 2006, 83, 428–433. [Google Scholar] [CrossRef]
- Leon, B.M.; Maddox, T.M. Diabetes and cardiovascular disease: Epidemiology, biological mechanisms, treatment recommendations and future research. World J. Diabetes 2015, 6, 1246–1258. [Google Scholar] [CrossRef] [PubMed]
- Beavers, D.P.; Beavers, K.M.; Miller, M.; Stamey, J.; Messina, M.J. Exposure to isoflavone-containing soy products and endothelial function: A Bayesian meta-analysis of randomized controlled trials. Nutr. Metab. Cardiovasc. Dis. 2012, 22, 182–191. [Google Scholar] [CrossRef] [PubMed]
- Hanson, M.G.; Zahradka, P.; Taylor, C.G. Lentil-based diets attenuate hypertension and large-artery remodelling in spontaneously hypertensive rats. Br. J. Nutr. 2014, 111, 690–698. [Google Scholar] [CrossRef] [PubMed]
- Hanson, M.G.; Taylor, C.G.; Wu, Y.; Anderson, H.D.; Zahradka, P. Lentil consumption reduces resistance artery remodeling and restores arterial compliance in the spontaneously hypertensive rat. J. Nutr. Biochem. 2016, 37, 30–38. [Google Scholar] [CrossRef] [PubMed]
- Zahradka, P.; Wright, B.; Weighell, W.; Blewett, H.; Baldwin, A.O.K.; Guzman, R.P.; Taylor, C.G. Daily non-soy legume consumption reverses vascular impairment due to peripheral artery disease. Atherosclerosis 2013, 230, 310–314. [Google Scholar] [CrossRef] [PubMed]
| Legume | Author | Study Design | Participants and Disease State/Condition | Intervention | Results |
|---|---|---|---|---|---|
| Soy (Glycine max. L.) | Llaneza et al. [5] |
|
|
|
|
| Choi et al. [17] |
|
|
|
| |
| Choquette et al. [18] |
|
|
|
| |
| Fei et al. [19] |
|
|
|
| |
| Jamilian et al. [20] |
|
|
|
| |
| Ye et al. [27] |
|
|
|
| |
| Beans (Phaseolus vulgaris L.) | Bourdon et al. [28] |
|
|
|
|
| Nilsson et al. [21] |
|
|
|
| |
| Reverri et al. [22] |
|
|
|
| |
| Beans (Phaseolus vulgaris L.) and Peas (Pisum sativum L.) | Winham et al. [29] |
|
|
|
|
| Peas (Pisum sativum L.) | Marinangeli & Jones [23] |
|
|
|
|
| Chickpeas (Cicer arietinum L.) | Johnson et al. [24] |
|
|
|
|
| Nestel et al. [25] |
|
|
|
| |
| Pittaway et al. [26] |
|
|
|
|
| Legume Source | Reference | Study Design | Parameters Measured | Key Findings |
|---|---|---|---|---|
| Soybean (Glycine max. L.) | Zhang et al. [1] |
|
|
|
| Nizamutdinova et al. [38] |
|
|
| |
| Ali et al. [64] |
|
|
| |
| Zanella et al. [70] |
|
|
| |
| Kavanagh et al. [80] |
|
|
| |
| Zhou et al. [86] |
|
|
| |
| Chickpea (Cicer arietinum L.) | Dai et al. [41] |
|
|
|
| Yang et al. [66] |
|
|
| |
| N/A | Choi et al. [65] |
|
|
|
| N/A | Weitkunat et al. [84] |
|
|
|
| N/A | Stahel et al. [91] |
|
|
|
| Legume Source | Reference | Cell Line and Treatment | Experimental Methods | Key Findings |
|---|---|---|---|---|
| Chickpea (Cicer arietinum L.) | Gao et al. [32] |
|
|
|
| Soybean (Glycine max. L.) | Huang et al. [49] |
|
|
|
| Inaguma et al. [53] |
|
|
| |
| Kim et al. [60] |
|
|
| |
| N/A | Luna-Vital et al. [54] |
|
|
|
| Legume | Compound | Model | Proposed Mechanisms | ||||||
|---|---|---|---|---|---|---|---|---|---|
| GLUT4 | PPAR-γ | Adiposity | Adiponectin | Leptin | Resistin | Gut Microbiota (SCFA-Producing Bacteria) | |||
| Black soybean | Isoflavones | 3T3-L1 cells | ↑ [49] | ↓ [49] | ↑ [49] | ↑ [49] | |||
| Anthocyanins | 3T3-L1 cells | ↑ [53] | ↓ [60] | ||||||
| Diabetic rats | ↑ [38] | ||||||||
| Soybean | Isoflavones | Insulin resistant rats | ↓ [1] | ↑ [1] | ↑ [1] | ↓ [1] | |||
| Lean & obese rats | ↓ [64] | ||||||||
| C57BL/6 mice | ↑ [70] | ||||||||
| Insulin resistant, menopausal monkeys | X [80] | ||||||||
| Galactooligosaccharides | Mini-piglets | ↑ [86] | |||||||
| Chickpea | Isoflavones | 3T3-L1 cells | ↓ [32] | ↓ [32] | ↓ [32] | ||||
| n/a | SD rats | ↓ [66] | ↓ [66] | ||||||
| α-galactooligosaccharides | CD-1 mice | ↑ [41] | |||||||
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Clark, J.L.; Taylor, C.G.; Zahradka, P. Rebelling against the (Insulin) Resistance: A Review of the Proposed Insulin-Sensitizing Actions of Soybeans, Chickpeas, and Their Bioactive Compounds. Nutrients 2018, 10, 434. https://doi.org/10.3390/nu10040434
Clark JL, Taylor CG, Zahradka P. Rebelling against the (Insulin) Resistance: A Review of the Proposed Insulin-Sensitizing Actions of Soybeans, Chickpeas, and Their Bioactive Compounds. Nutrients. 2018; 10(4):434. https://doi.org/10.3390/nu10040434
Chicago/Turabian StyleClark, Jaime L., Carla G. Taylor, and Peter Zahradka. 2018. "Rebelling against the (Insulin) Resistance: A Review of the Proposed Insulin-Sensitizing Actions of Soybeans, Chickpeas, and Their Bioactive Compounds" Nutrients 10, no. 4: 434. https://doi.org/10.3390/nu10040434
APA StyleClark, J. L., Taylor, C. G., & Zahradka, P. (2018). Rebelling against the (Insulin) Resistance: A Review of the Proposed Insulin-Sensitizing Actions of Soybeans, Chickpeas, and Their Bioactive Compounds. Nutrients, 10(4), 434. https://doi.org/10.3390/nu10040434







