Dietary Cholesterol, Serum Lipids, and Heart Disease: Are Eggs Working for or Against You?
Abstract
1. Introduction
2. Cholesterol, Eggs, and Heart Disease
2.1. Relationship between Dietary Cholesterol and/or Egg Intake on Risk for CVD in Observational Studies
2.2. Serum Cholesterol Responses and Adaptations to Cholesterol Intake
3. Dietary Cholesterol from Egg Intake and Lipoprotein Metabolism
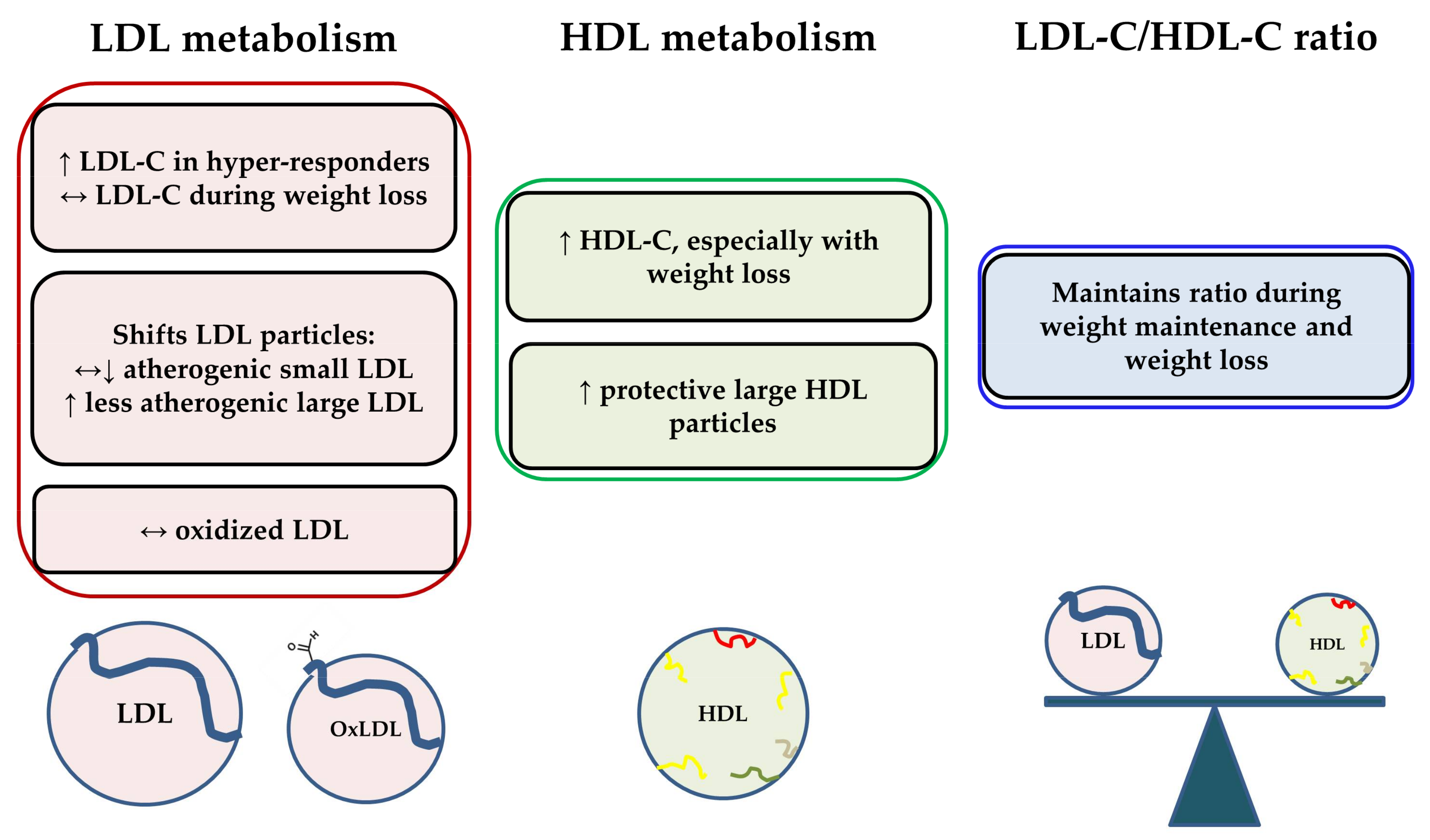
3.1. Effects of Dietary Cholesterol from Egg Intake on LDL-C, HDL-C, and the LDL-C/HDL-C Ratio
3.2. Effects of Dietary Cholesterol from Egg Intake on Lipoprotein Particle Characteristics
3.3. Effects of Dietary Cholesterol and/or Eggs on HDL Metabolism and Functionality
3.4. The Phospholipid Component of Eggs May Influence the Response to Dietary Cholesterol
4. Conclusions
Author Contributions
Conflicts of Interest
References
- Organization, W.H. Global Status Report on Noncommunicable Diseases 2014; World Health Organization: Geneva, Switzerland, 2014. [Google Scholar]
- Ross, R. Atherosclerosis—An inflammatory disease. N. Engl. J. Med. 1999, 340, 115–126. [Google Scholar] [CrossRef] [PubMed]
- Lewington, S.; Whitlock, G.; Clarke, R.; Sherliker, P.; Emberson, J.; Halsey, J.; Qizilbash, N.; Peto, R.; Collins, R. Blood cholesterol and vascular mortality by age, sex, and blood pressure: A meta-analysis of individual data from 61 prospective studies with 55,000 vascular deaths. Lancet 2007, 370, 1829–1839. [Google Scholar] [PubMed]
- Gordon, D.J.; Probstfield, J.L.; Garrison, R.J.; Neaton, J.D.; Castelli, W.P.; Knoke, J.D.; Jacobs, D.R., Jr.; Bangdiwala, S.; Tyroler, H.A. High-density lipoprotein cholesterol and cardiovascular disease. Four prospective american studies. Circulation 1989, 79, 8–15. [Google Scholar] [CrossRef] [PubMed]
- Law, M.R.; Wald, N.J.; Rudnicka, A.R. Quantifying effect of statins on low density lipoprotein cholesterol, ischaemic heart disease, and stroke: Systematic review and meta-analysis. BMJ 2003, 326, 1423. [Google Scholar] [CrossRef] [PubMed]
- Dawber, T.R.; Moore, F.E.; Mann, G.V. Coronary heart disease in the framingham study. Am. J. Public Health Nations Health 1957, 47, 4–24. [Google Scholar] [CrossRef] [PubMed]
- Mann, G.V.; Pearson, G.; Gordon, T.; Dawber, T.R. Diet and cardiovascular disease in the framingham study. I. Measurement of dietary intake. Am. J. Clin. Nutr. 1962, 11, 200–225. [Google Scholar] [CrossRef] [PubMed]
- Classics in arteriosclerosis research: On experimental cholesterin steatosis and its significance in the origin of some pathological processes by N. Anitschkow and S. Chalatow, translated by Mary Z. Pelias, 1913. Arteriosclerosis 1983, 3, 178–182.
- U.S. Department of Agriculture, Agricultural Research Service. Nutrient Intakes from Food and Beverages: Mean Amounts Consumed Per Individual, by Gender and Age, What We Eat in America. NHANES 2013-2014. Available online: www.ars.usda.gov/ba/bhnrc/fsrg (accessed on 22 January 2018).
- Keast, D.R.; Fulgoni, V.L., 3rd; Nicklas, T.A.; O’Neil, C.E. Food sources of energy and nutrients among children in the united states: National health and nutrition examination survey 2003–2006. Nutrients 2013, 5, 283–301. [Google Scholar] [CrossRef] [PubMed]
- O’Neil, C.E.; Keast, D.R.; Fulgoni, V.L.; Nicklas, T.A. Food sources of energy and nutrients among adults in the us: Nhanes 2003-2006. Nutrients 2012, 4, 2097–2120. [Google Scholar] [CrossRef] [PubMed]
- Shekelle, R.B.; Stamler, J. Dietary cholesterol and ischaemic heart disease. Lancet 1989, 1, 1177–1179. [Google Scholar] [CrossRef]
- Kushi, L.H.; Lew, R.A.; Stare, F.J.; Ellison, C.R.; el Lozy, M.; Bourke, G.; Daly, L.; Graham, I.; Hickey, N.; Mulcahy, R.; et al. Diet and 20-year mortality from coronary heart disease. The ireland-boston diet-heart study. N. Engl. J. Med. 1985, 312, 811–818. [Google Scholar] [CrossRef] [PubMed]
- Rong, Y.; Chen, L.; Zhu, T.; Song, Y.; Yu, M.; Shan, Z.; Sands, A.; Hu, F.B.; Liu, L. Egg consumption and risk of coronary heart disease and stroke: Dose-response meta-analysis of prospective cohort studies. BMJ 2013, 346, e8539. [Google Scholar] [CrossRef] [PubMed]
- Shin, J.Y.; Xun, P.; Nakamura, Y.; He, K. Egg consumption in relation to risk of cardiovascular disease and diabetes: A systematic review and meta-analysis. Am. J. Clin. Nutr. 2013, 98, 146–159. [Google Scholar] [CrossRef] [PubMed]
- Virtanen, J.K.; Mursu, J.; Virtanen, H.E.; Fogelholm, M.; Salonen, J.T.; Koskinen, T.T.; Voutilainen, S.; Tuomainen, T.P. Associations of egg and cholesterol intakes with carotid intima-media thickness and risk of incident coronary artery disease according to apolipoprotein e phenotype in men: The kuopio ischaemic heart disease risk factor study. Am. J. Clin. Nutr. 2016, 103, 895–901. [Google Scholar] [CrossRef] [PubMed]
- Larsson, S.C.; Akesson, A.; Wolk, A. Egg consumption and risk of heart failure, myocardial infarction, and stroke: Results from 2 prospective cohorts. Am. J. Clin. Nutr. 2015, 102, 1007–1013. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Y.; Li, Y.; Rimm, E.B.; Hu, F.B.; Albert, C.M.; Rexrode, K.M.; Manson, J.E.; Qi, L. Dietary phosphatidylcholine and risk of all-cause and cardiovascular-specific mortality among us women and men. Am. J. Clin. Nutr. 2016, 104, 173–180. [Google Scholar] [CrossRef] [PubMed]
- Pihlajamaki, J.; Gylling, H.; Miettinen, T.A.; Laakso, M. Insulin resistance is associated with increased cholesterol synthesis and decreased cholesterol absorption in normoglycemic men. J. Lipid Res. 2004, 45, 507–512. [Google Scholar] [CrossRef] [PubMed]
- Simonen, P.; Gylling, H.; Howard, A.N.; Miettinen, T.A. Introducing a new component of the metabolic syndrome: Low cholesterol absorption. Am. J. Clin. Nutr. 2000, 72, 82–88. [Google Scholar] [CrossRef] [PubMed]
- Miettinen, T.A.; Gylling, H. Cholesterol absorption efficiency and sterol metabolism in obesity. Atherosclerosis 2000, 153, 241–248. [Google Scholar] [CrossRef]
- Wang, Z.; Klipfell, E.; Bennett, B.J.; Koeth, R.; Levison, B.S.; Dugar, B.; Feldstein, A.E.; Britt, E.B.; Fu, X.; Chung, Y.M.; et al. Gut flora metabolism of phosphatidylcholine promotes cardiovascular disease. Nature 2011, 472, 57–63. [Google Scholar] [CrossRef] [PubMed]
- Tang, W.H.; Wang, Z.; Levison, B.S.; Koeth, R.A.; Britt, E.B.; Fu, X.; Wu, Y.; Hazen, S.L. Intestinal microbial metabolism of phosphatidylcholine and cardiovascular risk. N. Engl. J. Med. 2013, 368, 1575–1584. [Google Scholar] [CrossRef] [PubMed]
- DiMarco, D.M.; Missimer, A.; Murillo, A.G.; Lemos, B.S.; Malysheva, O.V.; Caudill, M.A.; Blesso, C.N.; Fernandez, M.L. Intake of up to 3 eggs/day increases hdl cholesterol and plasma choline while plasma trimethylamine-n-oxide is unchanged in a healthy population. Lipids 2017, 52, 255–263. [Google Scholar] [CrossRef] [PubMed]
- Missimer, A.; Fernandez, M.L.; DiMarco, D.M.; Norris, G.H.; Blesso, C.N.; Murillo, A.G.; Vergara-Jimenez, M.; Lemos, B.S.; Medina-Vera, I.; Malysheva, O.V.; et al. Compared to an oatmeal breakfast, two eggs/day increased plasma carotenoids and choline without increasing trimethyl amine n-oxide concentrations. J. Am. Coll. Nutr. 2018, 37, 140–148. [Google Scholar] [CrossRef] [PubMed]
- Cho, C.E.; Taesuwan, S.; Malysheva, O.V.; Bender, E.; Tulchinsky, N.F.; Yan, J.; Sutter, J.L.; Caudill, M.A. Trimethylamine-n-oxide (tmao) response to animal source foods varies among healthy young men and is influenced by their gut microbiota composition: A randomized controlled trial. Mol. Nutr. Food Res. 2017, 61. [Google Scholar] [CrossRef] [PubMed]
- Blesso, C.N. Egg phospholipids and cardiovascular health. Nutrients 2015, 7, 2731–2747. [Google Scholar] [CrossRef] [PubMed]
- Cho, C.E.; Caudill, M.A. Trimethylamine-n-oxide: Friend, foe, or simply caught in the cross-fire? Trends Endocrinol. Metab. 2017, 28, 121–130. [Google Scholar] [CrossRef] [PubMed]
- Berger, S.; Raman, G.; Vishwanathan, R.; Jacques, P.F.; Johnson, E.J. Dietary cholesterol and cardiovascular disease: A systematic review and meta-analysis. Am. J. Clin. Nutr. 2015, 102, 276–294. [Google Scholar] [CrossRef] [PubMed]
- Howell, W.H.; McNamara, D.J.; Tosca, M.A.; Smith, B.T.; Gaines, J.A. Plasma lipid and lipoprotein responses to dietary fat and cholesterol: A meta-analysis. Am. J. Clin. Nutr. 1997, 65, 1747–1764. [Google Scholar] [CrossRef] [PubMed]
- McNamara, D.J.; Kolb, R.; Parker, T.S.; Batwin, H.; Samuel, P.; Brown, C.D.; Ahrens, E.H., Jr. Heterogeneity of cholesterol homeostasis in man. Response to changes in dietary fat quality and cholesterol quantity. J. Clin. Investig. 1987, 79, 1729–1739. [Google Scholar] [CrossRef] [PubMed]
- Bosner, M.S.; Lange, L.G.; Stenson, W.F.; Ostlund, R.E., Jr. Percent cholesterol absorption in normal women and men quantified with dual stable isotopic tracers and negative ion mass spectrometry. J. Lipid Res. 1999, 40, 302–308. [Google Scholar] [PubMed]
- Lin, D.S.; Connor, W.E. The long term effects of dietary cholesterol upon the plasma lipids, lipoproteins, cholesterol absorption, and the sterol balance in man: The demonstration of feedback inhibition of cholesterol biosynthesis and increased bile acid excretion. J. Lipid Res. 1980, 21, 1042–1052. [Google Scholar] [PubMed]
- Ikonen, E. Cellular cholesterol trafficking and compartmentalization. Nat. Rev. Mol. Cell Biol. 2008, 9, 125–138. [Google Scholar] [CrossRef] [PubMed]
- McNamara, D.J. The impact of egg limitations on coronary heart disease risk: Do the numbers add up? J. Am. Coll. Nutr. 2000, 19, 540S–548S. [Google Scholar] [CrossRef] [PubMed]
- Mensink, R.P.; Zock, P.L.; Kester, A.D.; Katan, M.B. Effects of dietary fatty acids and carbohydrates on the ratio of serum total to hdl cholesterol and on serum lipids and apolipoproteins: A meta-analysis of 60 controlled trials. Am. J. Clin. Nutr. 2003, 77, 1146–1155. [Google Scholar] [CrossRef] [PubMed]
- Mensink, R.P.; Katan, M.B. Effect of dietary fatty acids on serum lipids and lipoproteins. A meta-analysis of 27 trials. Arterioscler. Thromb. 1992, 12, 911–919. [Google Scholar] [CrossRef] [PubMed]
- Keys, A.; Anderson, J.T.; Grande, F. Serum cholesterol response to changes in the diet: Ii. The effect of cholesterol in the diet. Metabolism 1965, 14, 759–765. [Google Scholar] [CrossRef]
- Katan, M.B.; Beynen, A.C. Characteristics of human hypo- and hyperresponders to dietary cholesterol. Am. J. Epidemiol. 1987, 125, 387–399. [Google Scholar] [CrossRef] [PubMed]
- Kern, F., Jr. Normal plasma cholesterol in an 88-year-old man who eats 25 eggs a day. Mechanisms of adaptation. N. Engl. J. Med. 1991, 324, 896–899. [Google Scholar] [CrossRef] [PubMed]
- Knopp, R.H.; Retzlaff, B.; Fish, B.; Walden, C.; Wallick, S.; Anderson, M.; Aikawa, K.; Kahn, S.E. Effects of insulin resistance and obesity on lipoproteins and sensitivity to egg feeding. Arterioscler. Thromb. Vasc. Biol. 2003, 23, 1437–1443. [Google Scholar] [CrossRef] [PubMed]
- Herron, K.L.; McGrane, M.M.; Waters, D.; Lofgren, I.E.; Clark, R.M.; Ordovas, J.M.; Fernandez, M.L. The abcg5 polymorphism contributes to individual responses to dietary cholesterol and carotenoids in eggs. J. Nutr. 2006, 136, 1161–1165. [Google Scholar] [CrossRef] [PubMed]
- Ballesteros, M.N.; Cabrera, R.M.; Saucedo Mdel, S.; Fernandez, M.L. Dietary cholesterol does not increase biomarkers for chronic disease in a pediatric population from northern mexico. Am. J. Clin. Nutr. 2004, 80, 855–861. [Google Scholar] [CrossRef] [PubMed]
- Herron, K.L.; Vega-Lopez, S.; Conde, K.; Ramjiganesh, T.; Roy, S.; Shachter, N.S.; Fernandez, M.L. Pre-menopausal women, classified as hypo- or hyperresponders, do not alter their ldl/hdl ratio following a high dietary cholesterol challenge. J. Am. Coll. Nutr. 2002, 21, 250–258. [Google Scholar] [CrossRef] [PubMed]
- Herron, K.L.; Vega-Lopez, S.; Conde, K.; Ramjiganesh, T.; Shachter, N.S.; Fernandez, M.L. Men classified as hypo- or hyperresponders to dietary cholesterol feeding exhibit differences in lipoprotein metabolism. J. Nutr. 2003, 133, 1036–1042. [Google Scholar] [CrossRef] [PubMed]
- Greene, C.M.; Zern, T.L.; Wood, R.J.; Shrestha, S.; Aggarwal, D.; Sharman, M.J.; Volek, J.S.; Fernandez, M.L. Maintenance of the ldl cholesterol:Hdl cholesterol ratio in an elderly population given a dietary cholesterol challenge. J. Nutr. 2005, 135, 2793–2798. [Google Scholar] [CrossRef] [PubMed]
- Fernandez, M.L.; Webb, D. The ldl to hdl cholesterol ratio as a valuable tool to evaluate coronary heart disease risk. J. Am. Coll. Nutr. 2008, 27, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Cullen, P.; Schulte, H.; Assmann, G. The munster heart study (procam): Total mortality in middle-aged men is increased at low total and ldl cholesterol concentrations in smokers but not in nonsmokers. Circulation 1997, 96, 2128–2136. [Google Scholar] [CrossRef] [PubMed]
- Knopp, R.H.; Retzlaff, B.M.; Walden, C.E.; Dowdy, A.A.; Tsunehara, C.H.; Austin, M.A.; Nguyen, T. A double-blind, randomized, controlled trial of the effects of two eggs per day in moderately hypercholesterolemic and combined hyperlipidemic subjects taught the ncep step i diet. J. Am. Coll. Nutr. 1997, 16, 551–561. [Google Scholar] [PubMed]
- Vishwanathan, R.; Goodrow-Kotyla, E.F.; Wooten, B.R.; Wilson, T.A.; Nicolosi, R.J. Consumption of 2 and 4 egg yolks/d for 5 wk increases macular pigment concentrations in older adults with low macular pigment taking cholesterol-lowering statins. Am. J. Clin. Nutr. 2009, 90, 1272–1279. [Google Scholar] [CrossRef] [PubMed]
- Ballesteros, M.N.; Valenzuela, F.; Robles, A.E.; Artalejo, E.; Aguilar, D.; Andersen, C.J.; Valdez, H.; Fernandez, M.L. One egg per day improves inflammation when compared to an oatmeal-based breakfast without increasing other cardiometabolic risk factors in diabetic patients. Nutrients 2015, 7, 3449–3463. [Google Scholar] [CrossRef] [PubMed]
- Fuller, N.R.; Caterson, I.D.; Sainsbury, A.; Denyer, G.; Fong, M.; Gerofi, J.; Baqleh, K.; Williams, K.H.; Lau, N.S.; Markovic, T.P. The effect of a high-egg diet on cardiovascular risk factors in people with type 2 diabetes: The diabetes and egg (diabegg) study-a 3-mo randomized controlled trial. Am. J. Clin. Nutr. 2015, 101, 705–713. [Google Scholar] [CrossRef] [PubMed]
- Harman, N.L.; Leeds, A.R.; Griffin, B.A. Increased dietary cholesterol does not increase plasma low density lipoprotein when accompanied by an energy-restricted diet and weight loss. Eur. J. Nutr. 2008, 47, 287–293. [Google Scholar] [CrossRef] [PubMed]
- Mutungi, G.; Ratliff, J.; Puglisi, M.; Torres-Gonzalez, M.; Vaishnav, U.; Leite, J.O.; Quann, E.; Volek, J.S.; Fernandez, M.L. Dietary cholesterol from eggs increases plasma hdl cholesterol in overweight men consuming a carbohydrate-restricted diet. J. Nutr. 2008, 138, 272–276. [Google Scholar] [CrossRef] [PubMed]
- Blesso, C.N.; Andersen, C.J.; Barona, J.; Volek, J.S.; Fernandez, M.L. Whole egg consumption improves lipoprotein profiles and insulin sensitivity to a greater extent than yolk-free egg substitute in individuals with metabolic syndrome. Metabolism 2013, 62, 400–410. [Google Scholar] [CrossRef] [PubMed]
- Pearce, K.L.; Clifton, P.M.; Noakes, M. Egg consumption as part of an energy-restricted high-protein diet improves blood lipid and blood glucose profiles in individuals with type 2 diabetes. Br. J. Nutr. 2011, 105, 584–592. [Google Scholar] [CrossRef] [PubMed]
- Freedman, D.S.; Otvos, J.D.; Jeyarajah, E.J.; Barboriak, J.J.; Anderson, A.J.; Walker, J.A. Relation of lipoprotein subclasses as measured by proton nuclear magnetic resonance spectroscopy to coronary artery disease. Arterioscler. Thromb. Vasc. Biol. 1998, 18, 1046–1053. [Google Scholar] [CrossRef] [PubMed]
- Mora, S.; Otvos, J.D.; Rifai, N.; Rosenson, R.S.; Buring, J.E.; Ridker, P.M. Lipoprotein particle profiles by nuclear magnetic resonance compared with standard lipids and apolipoproteins in predicting incident cardiovascular disease in women. Circulation 2009, 119, 931–939. [Google Scholar] [CrossRef] [PubMed]
- Tribble, D.L.; Rizzo, M.; Chait, A.; Lewis, D.M.; Blanche, P.J.; Krauss, R.M. Enhanced oxidative susceptibility and reduced antioxidant content of metabolic precursors of small, dense low-density lipoproteins. Am. J. Med. 2001, 110, 103–110. [Google Scholar] [CrossRef]
- Witztum, J.L.; Steinberg, D. Role of oxidized low density lipoprotein in atherogenesis. J. Clin. Investig. 1991, 88, 1785–1792. [Google Scholar] [CrossRef] [PubMed]
- Herron, K.L.; Lofgren, I.E.; Sharman, M.; Volek, J.S.; Fernandez, M.L. High intake of cholesterol results in less atherogenic low-density lipoprotein particles in men and women independent of response classification. Metabolism 2004, 53, 823–830. [Google Scholar] [CrossRef] [PubMed]
- Greene, C.M.; Waters, D.; Clark, R.M.; Contois, J.H.; Fernandez, M.L. Plasma ldl and hdl characteristics and carotenoid content are positively influenced by egg consumption in an elderly population. Nutr. Metab. 2006, 3, 6. [Google Scholar] [CrossRef] [PubMed]
- Mutungi, G.; Waters, D.; Ratliff, J.; Puglisi, M.; Clark, R.M.; Volek, J.S.; Fernandez, M.L. Eggs distinctly modulate plasma carotenoid and lipoprotein subclasses in adult men following a carbohydrate-restricted diet. J. Nutr. Biochem. 2010, 21, 261–267. [Google Scholar] [CrossRef] [PubMed]
- Rye, K.A.; Barter, P.J. Cardioprotective functions of hdls. J. Lipid Res. 2014, 55, 168–179. [Google Scholar] [CrossRef] [PubMed]
- Escola-Gil, J.C.; Llaverias, G.; Julve, J.; Jauhiainen, M.; Mendez-Gonzalez, J.; Blanco-Vaca, F. The cholesterol content of western diets plays a major role in the paradoxical increase in high-density lipoprotein cholesterol and upregulates the macrophage reverse cholesterol transport pathway. Arterioscler. Thromb. Vasc. Biol. 2011, 31, 2493–2499. [Google Scholar] [CrossRef] [PubMed]
- Kingwell, B.A.; Chapman, M.J.; Kontush, A.; Miller, N.E. Hdl-targeted therapies: Progress, failures and future. Nat. Rev. Drug Discov. 2014, 13, 445–464. [Google Scholar] [CrossRef] [PubMed]
- Toth, P.P.; Barylski, M.; Nikolic, D.; Rizzo, M.; Montalto, G.; Banach, M. Should low high-density lipoprotein cholesterol (hdl-c) be treated? Best Pract. Res. Clin. Endocrinol. Metab. 2014, 28, 353–368. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.S.; Burt, A.A.; Ranchalis, J.E.; Richter, R.J.; Marshall, J.K.; Nakayama, K.S.; Jarvik, E.R.; Eintracht, J.F.; Rosenthal, E.A.; Furlong, C.E.; et al. Dietary cholesterol increases paraoxonase 1 enzyme activity. J. Lipid Res. 2012, 53, 2450–2458. [Google Scholar] [CrossRef] [PubMed]
- Shih, D.M.; Xia, Y.R.; Wang, X.P.; Miller, E.; Castellani, L.W.; Subbanagounder, G.; Cheroutre, H.; Faull, K.F.; Berliner, J.A.; Witztum, J.L.; et al. Combined serum paraoxonase knockout/apolipoprotein e knockout mice exhibit increased lipoprotein oxidation and atherosclerosis. J. Biol. Chem. 2000, 275, 17527–17535. [Google Scholar] [CrossRef] [PubMed]
- Aharoni, S.; Aviram, M.; Fuhrman, B. Paraoxonase 1 (pon1) reduces macrophage inflammatory responses. Atherosclerosis 2013, 228, 353–361. [Google Scholar] [CrossRef] [PubMed]
- Rosenblat, M.; Vaya, J.; Shih, D.; Aviram, M. Paraoxonase 1 (pon1) enhances hdl-mediated macrophage cholesterol efflux via the abca1 transporter in association with increased hdl binding to the cells: A possible role for lysophosphatidylcholine. Atherosclerosis 2005, 179, 69–77. [Google Scholar] [CrossRef] [PubMed]
- DiMarco, D.M.; Norris, G.H.; Millar, C.L.; Blesso, C.N.; Fernandez, M.L. Intake of up to 3 eggs per day is associated with changes in hdl function and increased plasma antioxidants in healthy, young adults. J. Nutr. 2017, 147, 323–329. [Google Scholar] [PubMed]
- Rousset, X.; Shamburek, R.; Vaisman, B.; Amar, M.; Remaley, A.T. Lecithin cholesterol acyltransferase: An anti- or pro-atherogenic factor? Curr. Atheroscler. Rep. 2011, 13, 249–256. [Google Scholar] [CrossRef] [PubMed]
- Khera, A.V.; Cuchel, M.; de la Llera-Moya, M.; Rodrigues, A.; Burke, M.F.; Jafri, K.; French, B.C.; Phillips, J.A.; Mucksavage, M.L.; Wilensky, R.L.; et al. Cholesterol efflux capacity, high-density lipoprotein function, and atherosclerosis. N. Engl. J. Med. 2011, 364, 127–135. [Google Scholar] [CrossRef] [PubMed]
- Rohatgi, A.; Khera, A.; Berry, J.D.; Givens, E.G.; Ayers, C.R.; Wedin, K.E.; Neeland, I.J.; Yuhanna, I.S.; Rader, D.R.; de Lemos, J.A.; et al. Hdl cholesterol efflux capacity and incident cardiovascular events. N. Engl. J. Med. 2014, 371, 2383–2393. [Google Scholar] [CrossRef] [PubMed]
- Andersen, C.J.; Blesso, C.N.; Lee, J.; Barona, J.; Shah, D.; Thomas, M.J.; Fernandez, M.L. Egg consumption modulates hdl lipid composition and increases the cholesterol-accepting capacity of serum in metabolic syndrome. Lipids 2013, 48, 557–567. [Google Scholar] [CrossRef] [PubMed]
- Cohn, J.S.; Kamili, A.; Wat, E.; Chung, R.W.; Tandy, S. Dietary phospholipids and intestinal cholesterol absorption. Nutrients 2010, 2, 116–127. [Google Scholar] [CrossRef] [PubMed]
- Noh, S.K.; Koo, S.I. Egg sphingomyelin lowers the lymphatic absorption of cholesterol and alpha-tocopherol in rats. J. Nutr. 2003, 133, 3571–3576. [Google Scholar] [CrossRef] [PubMed]
- Noh, S.K.; Koo, S.I. Milk sphingomyelin is more effective than egg sphingomyelin in inhibiting intestinal absorption of cholesterol and fat in rats. J. Nutr. 2004, 134, 2611–2616. [Google Scholar] [CrossRef] [PubMed]
- Feng, D.; Ohlsson, L.; Ling, W.; Nilsson, A.; Duan, R.D. Generating ceramide from sphingomyelin by alkaline sphingomyelinase in the gut enhances sphingomyelin-induced inhibition of cholesterol uptake in caco-2 cells. Dig. Dis. Sci. 2010, 55, 3377–3383. [Google Scholar] [CrossRef] [PubMed]
- Norris, G.H.; Blesso, C.N. Dietary sphingolipids: Potential for management of dyslipidemia and nonalcoholic fatty liver disease. Nutr. Rev. 2017, 75, 274–285. [Google Scholar] [CrossRef] [PubMed]
- Norris, G.H.; Porter, C.M.; Jiang, C.; Millar, C.L.; Blesso, C.N. Dietary sphingomyelin attenuates hepatic steatosis and adipose tissue inflammation in high-fat-diet-induced obese mice. J. Nutr. Biochem. 2017, 40, 36–43. [Google Scholar] [CrossRef] [PubMed]
- Chung, R.W.S.; Wang, Z.; Bursill, C.A.; Wu, B.J.; Barter, P.J.; Rye, K.A. Effect of long-term dietary sphingomyelin supplementation on atherosclerosis in mice. PLoS ONE 2017, 12, e0189523. [Google Scholar] [CrossRef] [PubMed]
- Norris, G.H.; Jiang, C.; Ryan, J.; Porter, C.M.; Blesso, C.N. Milk sphingomyelin improves lipid metabolism and alters gut microbiota in high fat diet-fed mice. J. Nutr. Biochem. 2016, 30, 93–101. [Google Scholar] [CrossRef] [PubMed]
- Norris, G.H.; Blesso, C.N. Dietary and endogenous sphingolipid metabolism in chronic inflammation. Nutrients 2017, 9, 1180. [Google Scholar] [CrossRef] [PubMed]
| Study/Population | Design | # Days | LDL-C | HDL-C | LDL-C/HDL-C |
|---|---|---|---|---|---|
| Children | |||||
| Ballesteros et al. 2004 [43]; Healthy boys and girls | Crossover (n = 54): 2 eggs per day (518 mg cholesterol) vs. egg substitute | 30 | Hyper-: +25% Hypo-: ↔ | Hyper-: +10% Hypo-: ↔ | ↔ |
| Adults | |||||
| Herron et al. 2002 [44]; Healthy women | Crossover (n = 51): 3 eggs per day (640 mg cholesterol) vs. egg substitute | 30 | Hyper-: +20% Hypo-: ↔ | Hyper-: +12% Hypo-: ↔ | ↔ |
| Herron et al. 2003 [45]; Healthy men | Crossover (n = 40): 3 eggs per day (640 mg cholesterol) vs. egg substitute | 30 | Hyper-: +30% Hypo-: ↔ | Hyper-: +8% Hypo-: ↔ | Hyper-: + 22% Hypo-: ↔ |
| Greene et al. 2005 [46]; Healthy older adults | Crossover (n = 42): 3 eggs per day (640 mg cholesterol) vs. egg substitute | 30 | Women: +10% Men: +2% | Women: +3% Men: +10% | ↔ |
| Knopp et al. 2003 [41]; Insulin-sensitive | Crossover (n = 65): 4 eggs per day (850 mg cholesterol) vs. egg substitute | 28 | +7% | +7% | ND * |
| Hyperlipidemic | |||||
| Knopp et al. 1997 [49]; Hypercholesterolemic (HC) and combined hyperlipidemic (CHL) men/women | Parallel: 2 eggs per day (425 mg cholesterol) (HC: n = 44; CHL: n = 31) vs. egg substitute (HC: n = 35; CHL: n = 21) | 84 | HC: ↔ CHL: +8% from baseline | HC: +8% from baseline CHL: +7% from baseline | ND |
| Vishwanathan et al. 2009 [50]; Statin-taking older adults | Crossover (n = 52): 2 or 4 eggs per day (~400–800 mg cholesterol) vs. egg exclusion | 35 | 2 eggs: ↔ 4 eggs: ↔ | 2 eggs: +5% 4 eggs: +5% | ND |
| Study/Population | Design | # Days | LDL-C | HDL-C | LDL-C/HDL-C |
|---|---|---|---|---|---|
| Insulin-resistant | |||||
| Knopp et al. 2003 [41]; Insulin-resistant (IR) and obese insulin-resistant (OIR) | Crossover (IR: n = 75; OIR: n = 57): 4 eggs per day (850 mg cholesterol) vs. egg substitute | 28 | IR: +6% OIR: ↔ | IR: +6% OIR: +6% | ND * |
| Diabetic | |||||
| Ballesteros et al. 2015 [51]; Diabetic patients | Crossover (n = 29): 1 egg per day (250 mg cholesterol) vs. oatmeal breakfast | 35 | ↔ | ↔ | ↔ |
| Fuller et al. 2015 [52]; Diabetic patients | Parallel: High egg (12 eggs/week; ~300–350 mg cholesterol/day) (n = 72) vs. low egg (<2 eggs/week) (n = 68) | 42 | ↔ | ↔ | ND |
| Study/Population | Design | # Days | LDL-C | HDL-C | LDL-C/HDL-C |
|---|---|---|---|---|---|
| Harman et al. 2008 [53]; Men/women | Parallel: 2 eggs per day (~400 mg cholesterol) (n = 24) vs. egg exclusion (n = 21) | 84 | ↔ | ↔ | ND * |
| Mutungi et al. 2008 [54]; Overweight/obese men | Parallel: 3 eggs per day (640 mg cholesterol) (n = 15) vs. egg substitute (n = 13) | 84 | ↔ | +25% from baseline | ↔ |
| Pearce et al. 2011 [56]; Diabetic patients | Parallel: 2 eggs per day (590 mg cholesterol/day) (n = 31) vs. egg exclusion (213 mg cholesterol/day) (n = 34) | 84 | ↔ | Eggs +2% from baseline, egg exclusion −6% from baseline | ↔ |
| Blesso et al. 2013 [55]; Metabolic syndrome men/women | Parallel: 3 eggs per day (640 mg cholesterol) (n = 20) vs. egg substitute (n = 17) | 84 | ↔ | +17% from baseline | ↓ |
| Study/Population | Design | # Days | LDL Particles | Oxidized LDL | HDL Particles |
|---|---|---|---|---|---|
| Weight maintenance | |||||
| Ballesteros et al. 2004 [43]; Healthy children | Crossover (n = 54): 2 eggs per day (518 mg cholesterol) vs. egg substitute | 30 | ↑Large LDL (+31% LDL-1 in hyper-) ↓Small LDL (−38% LDL-3 in hyper-) ↑LDL size | ND * | ND |
| Herron et al. 2004 [61]; Healthy men/women | Crossover (n = 52): 3 eggs per day (640 mg cholesterol) vs. egg substitute | 30 | ↑Large LDL (+13% LDL-1, +30% LDL-2 in women hyper-) | ↔ | ND |
| Greene et al. 2006 [62]; Healthy elderly men/women | Crossover (n = 42): 3 eggs per day (640 mg cholesterol) vs. egg substitute | 30 | ↑Large LDL (+30% from baseline in hyper-) | ND | ↑Large HDL (+23% from baseline in hyper-) ↑HDL size |
| Ballesteros et al. 2015 [51]; Diabetic patients | Crossover (n = 29): 1 egg per day (250 mg cholesterol) vs. oatmeal breakfast | 35 | ↔ | ↔ | ↔ |
| Weight loss | |||||
| Mutungi et al. 2010 [63]; Overweight/obese men | Parallel: 3 eggs per day (640 mg cholesterol) (n = 15) vs. egg substitute (n = 13) | 84 | ↑Large LDL (+42% from baseline) | ND | ↑Large HDL (+52% from baseline) ↑HDL size |
| Blesso et al. 2013 [55]; Metabolic syndrome men/women | Parallel: 3 eggs per day (640 mg cholesterol) (n = 20) vs. egg substitute (n = 17) | 84 | ↑Large LDL (+22% from baseline) | ↔ | ↑Large HDL (+30% from baseline) ↑HDL size |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Blesso, C.N.; Fernandez, M.L. Dietary Cholesterol, Serum Lipids, and Heart Disease: Are Eggs Working for or Against You? Nutrients 2018, 10, 426. https://doi.org/10.3390/nu10040426
Blesso CN, Fernandez ML. Dietary Cholesterol, Serum Lipids, and Heart Disease: Are Eggs Working for or Against You? Nutrients. 2018; 10(4):426. https://doi.org/10.3390/nu10040426
Chicago/Turabian StyleBlesso, Christopher N., and Maria Luz Fernandez. 2018. "Dietary Cholesterol, Serum Lipids, and Heart Disease: Are Eggs Working for or Against You?" Nutrients 10, no. 4: 426. https://doi.org/10.3390/nu10040426
APA StyleBlesso, C. N., & Fernandez, M. L. (2018). Dietary Cholesterol, Serum Lipids, and Heart Disease: Are Eggs Working for or Against You? Nutrients, 10(4), 426. https://doi.org/10.3390/nu10040426






