Protein Supplementation During or Following a Marathon Run Influences Post-Exercise Recovery
Abstract
1. Introduction
2. Materials and Methods
2.1. Study A—Protein Supplementation During Exercise
2.1.1. Subjects
2.1.2. Experimental Design
2.1.3. Experimental Treatments
2.1.4. Dietary Controls
2.1.5. Dependent Measurements
2.1.6. Statistical Analyses
2.2. Study B—Protein Supplementation Following Exercise
2.2.1. Subjects
2.2.2. Experimental Design & Nutritional Intervention
3. Results
3.1. Study A—Protein Supplementation During Exercise
3.1.1. Subjects
3.1.2. Marathon Outcomes and Nutrient Intake
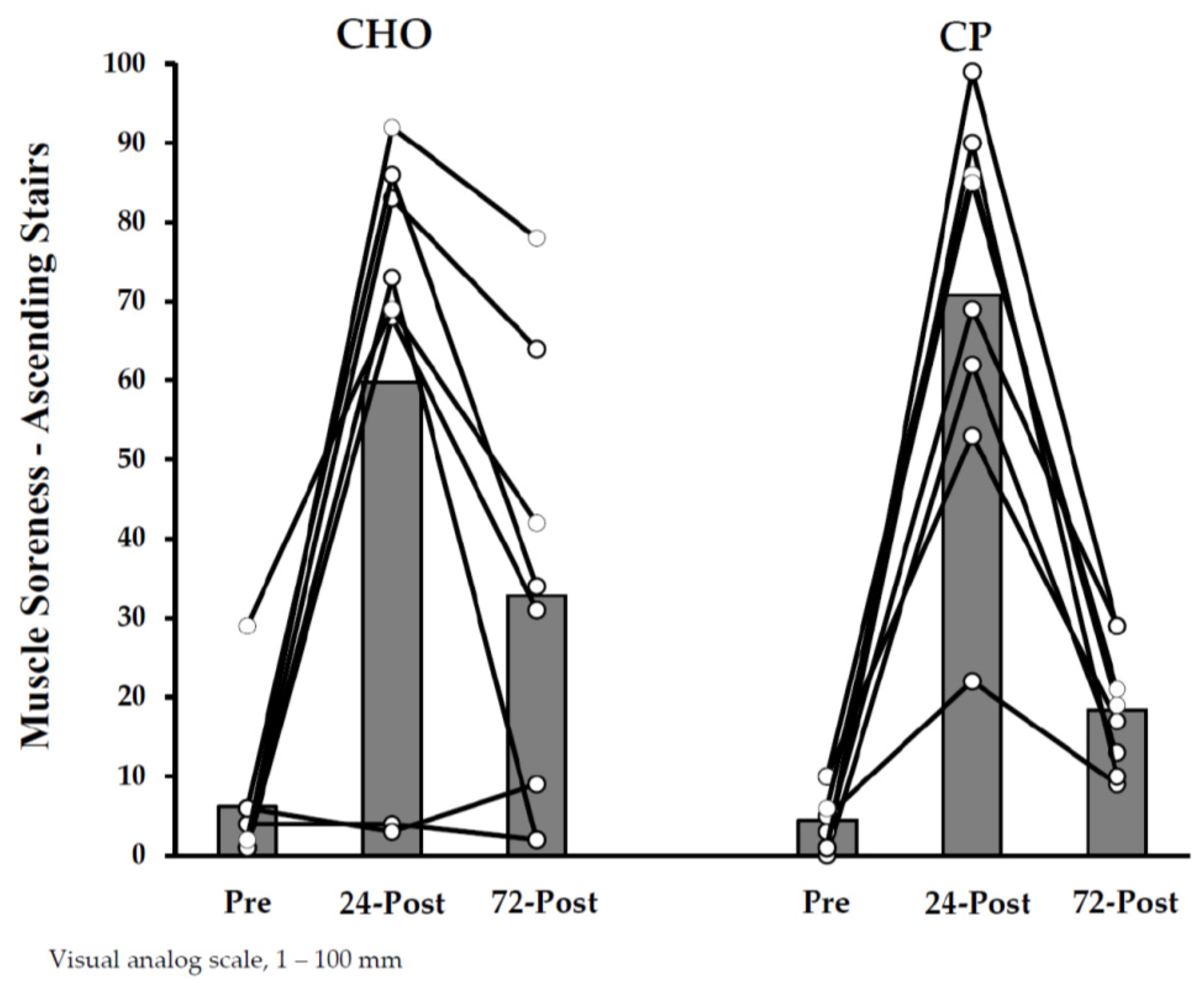
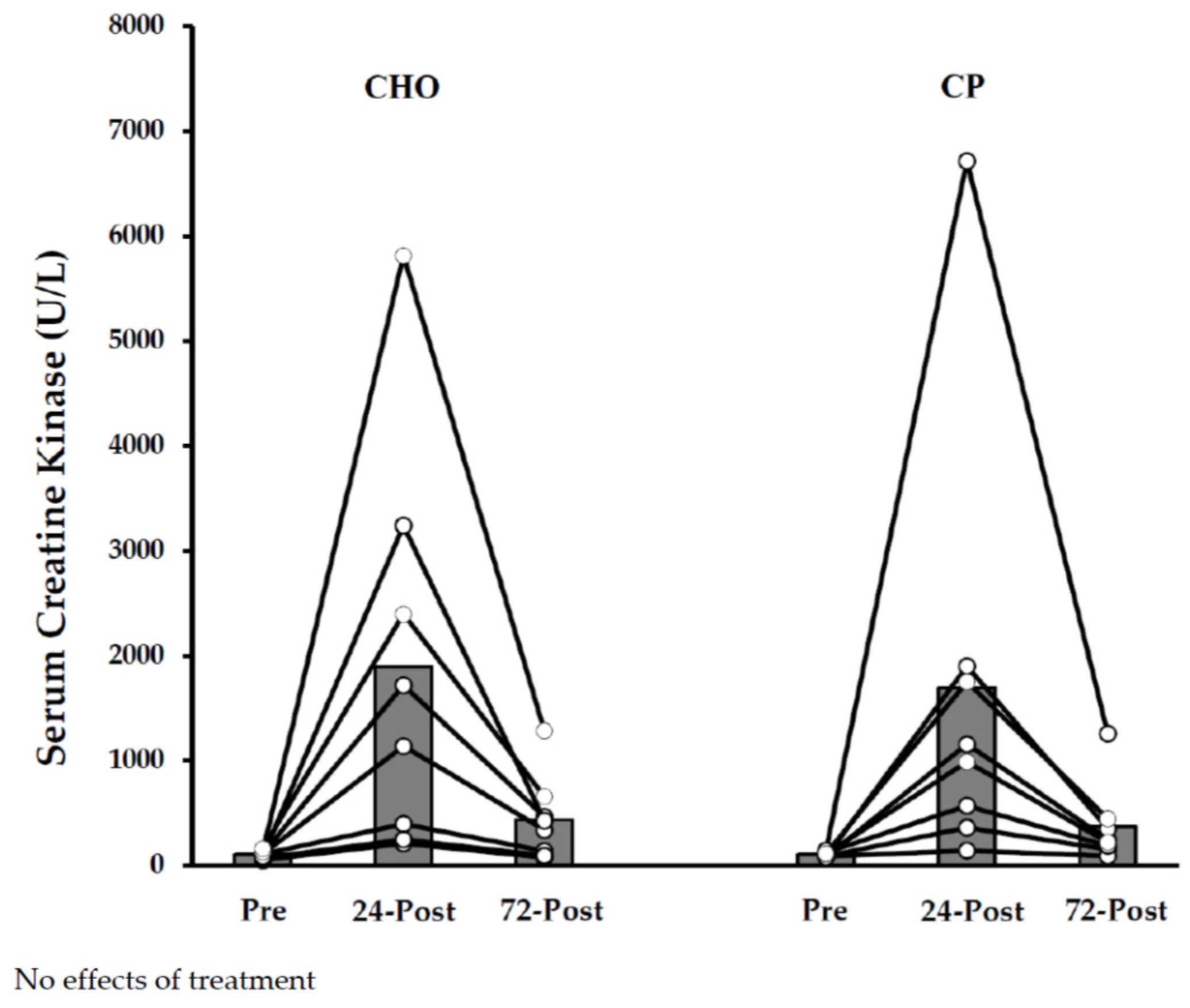
3.1.3. Dependent Measurements
3.2. Study B—Protein Supplementation Following Exercise
3.2.1. Subjects
3.2.2. Marathon Outcomes and Nutrient Intake
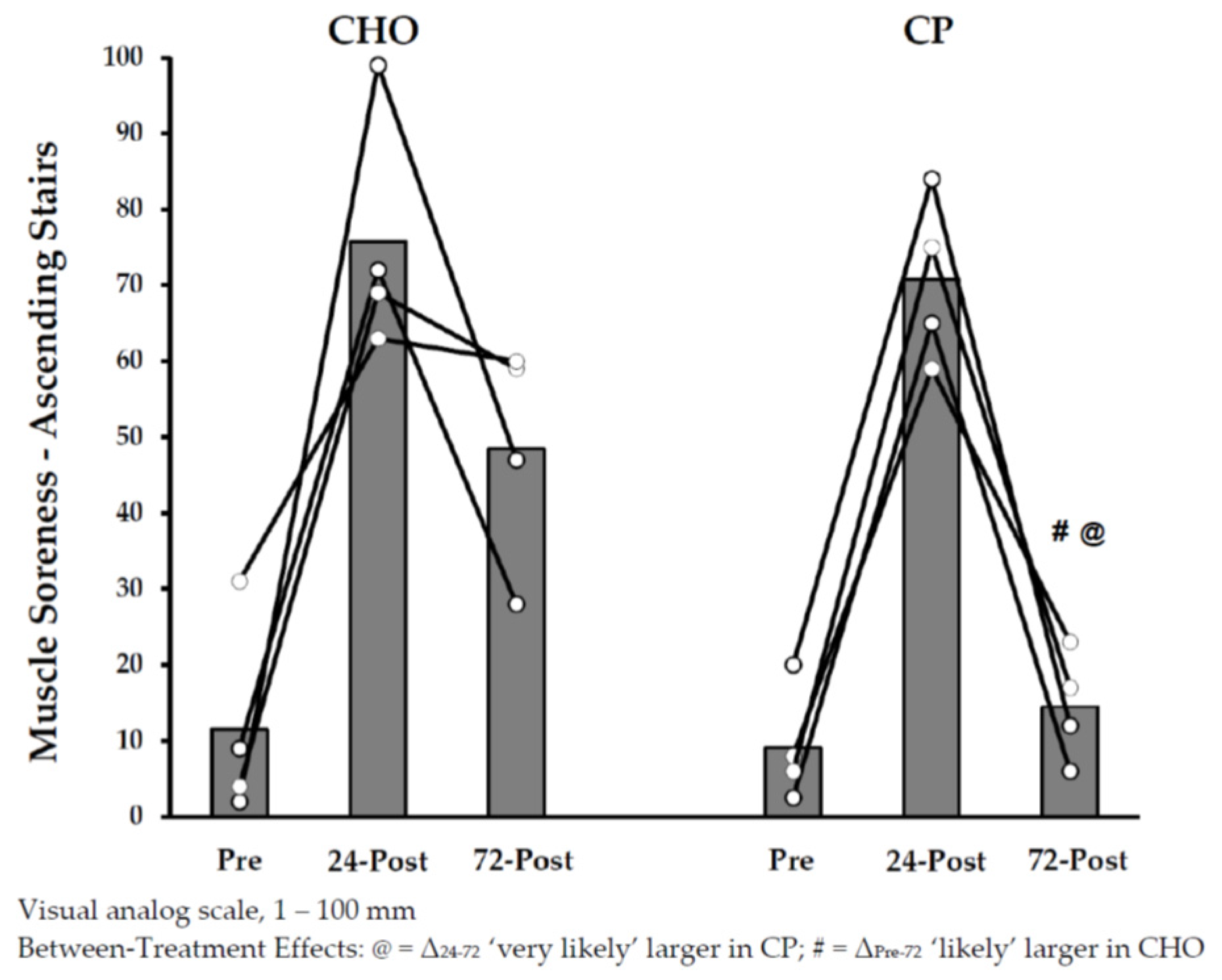
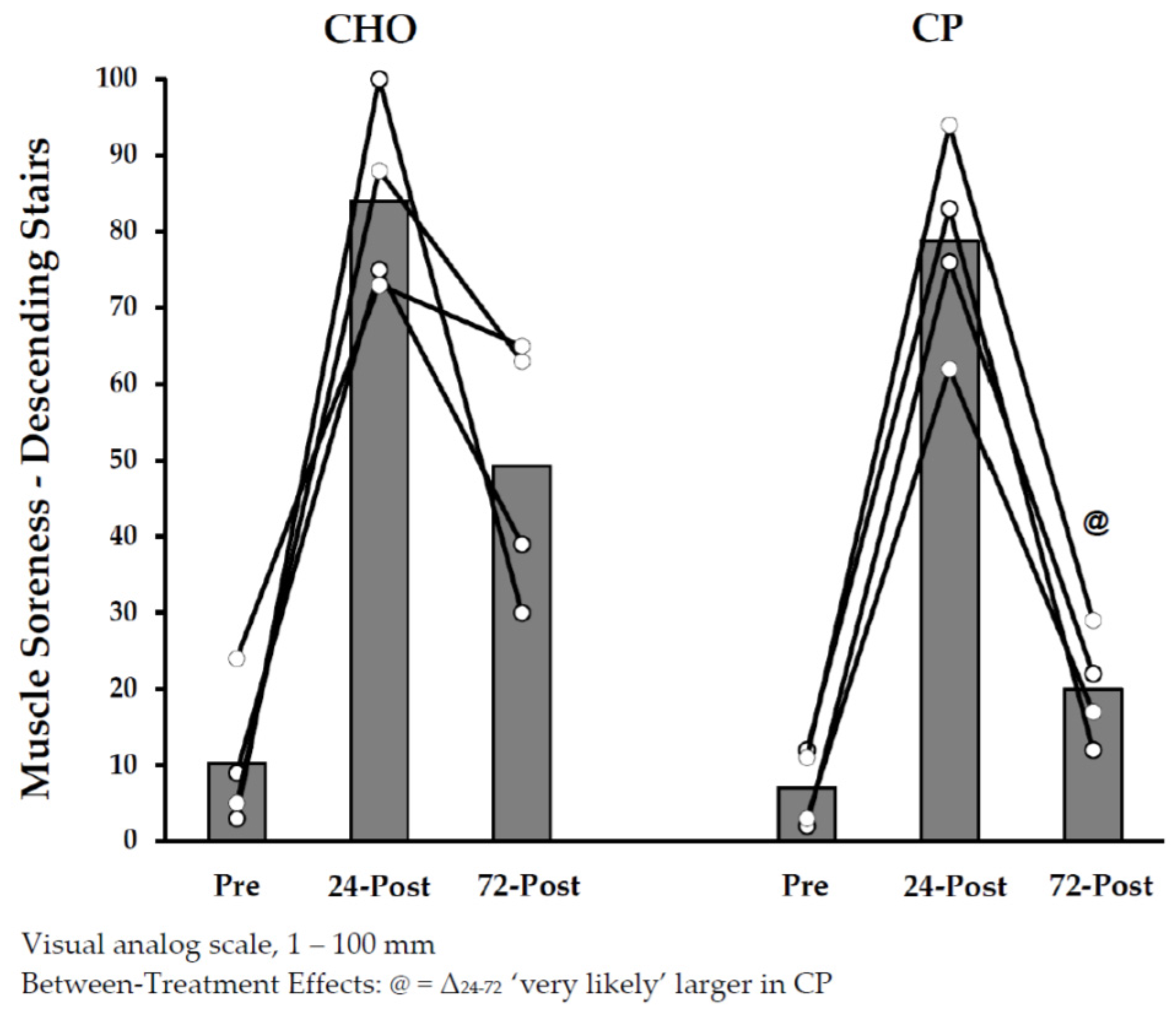
3.2.3. Dependent Measurements
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Millard-Stafford, M.; Warren, G.L.; Thomas, L.M.; Doyle, J.A.; Snow, T.; Hitchcock, K. Recovery from Run Training : Efficacy of a Carbohydrate-Protein Beverage ? Int. J. Sport Nutr. 2005, 15, 610–624. [Google Scholar] [CrossRef]
- Romano-Ely, B.C.; Todd, M.K.; Saunders, M.J.; St., Laurent, T. Effect of an isocaloric carbohydrate-protein-antioxidant drink on cycling performance. Med. Sci. Sports Exerc. 2006, 38, 1608–1616. [Google Scholar] [CrossRef] [PubMed]
- Papacosta, E.; Nassis, G.P.; Gleeson, M. Effects of acute postexercise chocolate milk consumption during intensive judo training on the recovery of salivary hormones, salivary SIgA, mood state, muscle soreness, and judo-related performance. Appl. Physiol. Nutr. Metab. 2015, 40, 1116–1122. [Google Scholar] [CrossRef] [PubMed]
- Luden, N.; Saunders, M.; Todd, M. Postexercise carbohydrate-protein-antioxidant ingestion decreases plasma creatine kinase and muscle soreness. Int. J. Sport Nutr. 2007, 17, 109–123. [Google Scholar] [CrossRef]
- Greer, B.K.; Woodard, J.L.; White, J.P.; Arguello, E.M.; Haymes, E.M. Branched-chain amino acid supplementation and indicators of muscle damage after endurance exercise. Int. J. Sport Nutr. Exerc. Metab. 2007, 17, 595–607. [Google Scholar] [CrossRef] [PubMed]
- Rowlands, D.S.; Rössler, K.; Thorp, R.M.; Graham, D.F.; Timmons, B.W.; Stannard, S.R.; Tarnopolsky, M.A. Effect of dietary protein content during recovery from high-intensity cycling on subsequent performance and markers of stress, inflammation, and muscle damage in well-trained men. Appl. Physiol. Nutr. Metab. 2008, 33, 39–51. [Google Scholar] [CrossRef] [PubMed]
- Cockburn, E.; Robson-Ansley, P.; Hayes, P.R.; Stevenson, E. Effect of volume of milk consumed on the attenuation of exercise-induced muscle damage. Eur. J. Appl. Physiol. 2012, 112, 3187–3194. [Google Scholar] [CrossRef] [PubMed]
- Gilson, S.; Saunders, M.; Moran, C.; Moore, R.; Womack, C.; Todd, M. Effects of chocolate milk consumption on markers of muscle recovery following soccer training: A randomized cross-over study. J. Int. Soc. Sports Nutr. 2010, 7, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Valentine, R.J.; Saunders, M.J.; Todd, M.K.; Laurent, T.G.S. Influence of Carbohydrate-Protein Beverage on Cycling Endurance and Indices of Muscle Disruption. Int. J. Sport Nutr. Exerc. Metab. 2008, 18, 363–378. [Google Scholar] [CrossRef] [PubMed]
- Hall, A.H.; Leveritt, M.D.; Ahuja, K.D.K.; Shing, C.M. Coingestion of carbohydrate and protein during training reduces training stress and enhances subsequent exercise performance. Appl. Physiol. Nutr. Metab. 2013, 38, 597–604. [Google Scholar] [CrossRef] [PubMed]
- Witard, O.C.; Jackman, S.R.; Kies, A.K.; Jeukendrup, A.E.; Tipton, K.D. Effect of Increased Dietary Protein on Tolerance to Intensified Training. Med. Sci. Sports 2011, 43, 598–607. [Google Scholar] [CrossRef] [PubMed]
- Rankin, P.; Lawlor, M.J.; Hills, F.A.; Bell, P.G.; Stevenson, E.J.; Cockburn, E. The effect of milk on recovery from repeat-sprint cycling in female team-sport athletes. Appl. Physiol. Nutr. Metab. 2017, 10, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Cockburn, E.; Bell, P.G.; Stevenson, E. Effect of milk on team sport performance after exercise-induced muscle damage. Med. Sci. Sports Exerc. 2013, 45, 1585–1592. [Google Scholar] [CrossRef] [PubMed]
- Betts, J.A.; Toone, R.J.; Stokes, K.A.; Thompson, D. Systemic indices of skeletal muscle damage and recovery of muscle function after exercise: Effect of combined carbohydrate–protein ingestion. Appl. Physiol. Nutr. Metab. 2009, 34, 773–784. [Google Scholar] [CrossRef] [PubMed]
- Goh, Q.; Boop, C.; Luden, N.; Smith, A. Recovery from cycling exercise: Effects of carbohydrate and protein beverages. Nutrients 2012, 4, 568–584. [Google Scholar] [CrossRef] [PubMed]
- Breen, L.; Tipton, K.D.; Jeukendrup, A.E. No Effect of Carbohydrate–Protein on Cycling Performance and Indices of Recovery. Med. Sci. Sports Exerc. 2010, 1140–1148. [Google Scholar] [CrossRef] [PubMed]
- Saunders, M.J. Carbohydrate-protein intake and recovery from endurance exercise: Is chocolate milk the answer? Curr. Sports Med. Rep. 2011, 10, 203–210. [Google Scholar] [CrossRef] [PubMed]
- Sousa, M.; Teixeira, V.H.; Soares, J. Dietary strategies to recover from exercise-induced muscle damage. Int. J. Food Sci. Nutr. 2014, 65, 151–163. [Google Scholar] [CrossRef] [PubMed]
- Running USA. Available online: http://www.runningusa.org/marathon-report-2017 (accessed on 6 January 2018).
- Overgaard, K.; Fredsted, A.; Hyldal, A.; Ingemann-Hansen, T.; Gissel, H.; Clausen, T. Effects of Running Distance and Training on Ca2+ Content and Damage in Human Muscle. Med. Sci. Sports Exerc. 2004, 36, 821–829. [Google Scholar] [CrossRef] [PubMed]
- Howatson, G.; McHugh, M.P.; Hill, J.A.; Brouner, J.; Jewell, A.P.; Van Someren, K.A.; Shave, R.E.; Howatson, S.A. Influence of tart cherry juice on indices of recovery following marathon running. Scand. J. Med. Sci. Sports 2010, 20, 843–852. [Google Scholar] [CrossRef] [PubMed]
- Siegel, A.J.; Silverman, L.M.; Lopez, R.E. Creatine kinase elevations in marathon runners: Relationship to training and competition. Yale J. Biol. Med. 1980, 53, 275–279. [Google Scholar] [PubMed]
- Burke, L.M.; Hawley, J.A.; Wong, S.H.; Jeukendrup, A.E. Carbohydrates for training and competition. J. Sports Sci. 2011, 29, S17–S27. [Google Scholar] [CrossRef] [PubMed]
- Trappe, S. Single muscle fiber adaptations with marathon training. J. Appl. Physiol. 2006, 101, 721–727. [Google Scholar] [CrossRef] [PubMed]
- Hopkins, W.; Marshall, S.; Batterham, A.M.; Hanin, J. Progressive statistics for studies in sports medicine and exercise science. Med. Sci. 2009, 41, 3–12. [Google Scholar] [CrossRef] [PubMed]
- Hopkins, W.G. A spreadsheet for deriving a confidence interval, mechanistic inference, and clinical inference from a p value. Sport Sci. 2007, 11, 16–19. [Google Scholar]
- Hopkins, W.G. Estimating Sample Size for Magnitude-Based Inferences. Sportscience 2006, 10, 63–70. [Google Scholar]
- Beelen, M.; Burke, L.M.; Gibala, M.J.; van Loon L, J.C. Nutritional Strategies to promote postexercise recovery.pdf. Int. J. Sport Nutr. Exerc. Metab. 2010, 20, 515–532. [Google Scholar] [CrossRef] [PubMed]
- Rankin, P.; Stevenson, E.; Cockburn, E. The effect of milk on the attenuation of exercise-induced muscle damage in males and females. Eur. J. Appl. Physiol. 2015, 115, 1245–1261. [Google Scholar] [CrossRef] [PubMed]
- Clarkson, P.M.; Hubal, M.J. Exercise-induced muscle damage in humans. Am. J. Phys. Med. Rehabil. 2002, 81, S52–S69. [Google Scholar] [CrossRef] [PubMed]
- Moore, D.R.D.; Robinson, M.J.M.; Fry, J.L.J.; Tang, J.E.; Glover, E.I.; Wilkinson, S.B.; Prior, T.; Tarnopolsky, M.A.; Phillips, S.M. Ingested protein dose response of muscle and albumin protein synthesis after resistance exercise in young men. Am. J. Clin. Nutr. 2009, 89, 161–168. [Google Scholar] [CrossRef] [PubMed]

| Week | Monday | Tuesday | Wednesday | Thursday | Friday | Saturday | Sunday | Total |
|---|---|---|---|---|---|---|---|---|
| 1 | 5 | 6 | - | 5 | - | 8 | - | 24 |
| 2 | 5 | 6 | - | 5 | - | 10 | - | 26 |
| 3 | 5 | 6 | - | 5 | - | 11 | - | 27 |
| 4 | 5 | 8 | - | 5 | - | 13 | - | 31 |
| 5 | 5 | 8 | - | 5 | - | 16 | - | 34 |
| 6 | 6 | 8 | - | 6 | - | 17.5 | - | 37.5 |
| 7 | 6 | 10 | - | 6 | - | 19 | - | 41 |
| 8 | 6 | 10 | - | 6 | - | 22.5 | - | 44.5 |
| 9 | 6 | 11 | - | 6 | - | 25.5 | - | 48.5 |
| 10 | 8 | 13 | - | 8 | - | 25.5 | - | 54.5 |
| 11 | 8 | 13 | - | 8 | - | 29 | - | 58 |
| 12 | 8 | 13 | - | 8 | - | 29 | - | 58 |
| 13 | 8 | 13 | - | 8 | - | 14.5 | - | 43.5 |
| 14 | 5 | 8 | - | 5 | - | 13 | - | 31 |
| 15 | 5 | 5 | - | 5 (walk) | - | 42.2 | 57.2 |
| Within-Treatment Effects (Mean ± SD) | Between-Treatment Differences Mean ± 90% CI % Likelihoods*, Inference | |||||||
|---|---|---|---|---|---|---|---|---|
| Variable | Treatment | Pre | 24-Post | 72-Post | Pre-24 | 24–72 | Pre-72 | |
| Physical Energy | CHO | 240 ± 40 | 108 ± 64**** | 166 ± 77 | 45 ± 79 | 43 ± 87 | 88 ± 88 | |
| 76/4/20 | 75/4/21 | 94/1/5 | ||||||
| Unclear | Unclear | Likely positive | ||||||
| CP | 200 ± 88 | 113 ± 63 | 214 ± 64### | |||||
| Physical Fatigue | CHO | 51 ± 40 | 191 ± 75**** | 106 ± 70**,## | −7 ± 90 32/20/48 Unclear | −64 ± 74 5/10/85 Unclear | −71 ± 74 8/9/84 Unclear | |
| CP | 84 ± 101 | 217 ± 69**** | 67 ± 48#### | |||||
| Mental Energy | CHO | 230 ± 56 | 162 ± 71*** | 206 ± 76 | 57 ± 79 72/6/22 Unclear | 5 ± 63 52/14/34 Unclear | 62 ± 62 76/5/19 Unclear | |
| CP | 149 ± 94 | 137 ± 80 | 186 ± 92## | |||||
| Mental Fatigue | CHO | 75 ± 73 | 135 ± 77*** | 75 ± 72### | −40 ± 93 13/21/66 Unclear | −13 ± 70 21/29/50 Unclear | −53 ± 60 1/10/89 Likely negative | |
| CP | 147 ± 101 | 166 ± 78 | 94 ± 88***,## | |||||
| Within-Treatment Effects Mean ± SD | Between-Treatment Differences Mean ± 90% CI% Likelihoods*, Inference | ||||||
|---|---|---|---|---|---|---|---|
| Variable | Treat | Pre | 24-Post | 72-Post | Pre-24 | 24–72 | Pre-72 |
| Physical Energy | CHO | 233 ± 58 | 94 ± 63 | 137 ±38 | 33 ± 133 76/1/22 Unclear | 69 ± 86 29/2/69 Unclear | 102 ± 111 93/2/5 Unclear |
| CP | 202 ± 52 | 96 ± 35** | 207 ± 39### | ||||
| Physical Fatigue | CHO | 54 ± 68 | 208 ± 66** | 154 ± 27**,## | −49 ± 174 13/6/82 Unclear | −102 ±96 3/2/94 Likely negative | −152 ± 107 2/1/97 Very likely negative |
| CP | 118 ± 87 | 223 ± 43 | 66 ± 61### | ||||
| Mental Energy | CHO | 216 ± 54 | 131 ± 60 | 155 ± 27 | 56 ± 134 75/5/20 Unclear | 42 ± 92 56/10/34 Unclear | 98 ± 84 97/1/2 Very likely positive |
| CP | 184 ± 62 | 154 ± 57 | 220 ± 46** | ||||
| Mental Fatigue | CHO | 91 ± 74 | 175 ± 40 | 148 ± 31# | −59 ± 150 20/6/74 Unclear | −35 ± 93 9/9/82 Unclear | −93 ± 100 10/3/87 Unclear |
| CP | 106 ± 84 | 131 ± 72 | 69 ± 59 | ||||
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Saunders, M.J.; Luden, N.D.; DeWitt, C.R.; Gross, M.C.; Dillon Rios, A. Protein Supplementation During or Following a Marathon Run Influences Post-Exercise Recovery. Nutrients 2018, 10, 333. https://doi.org/10.3390/nu10030333
Saunders MJ, Luden ND, DeWitt CR, Gross MC, Dillon Rios A. Protein Supplementation During or Following a Marathon Run Influences Post-Exercise Recovery. Nutrients. 2018; 10(3):333. https://doi.org/10.3390/nu10030333
Chicago/Turabian StyleSaunders, Michael J., Nicholas D. Luden, Cash R. DeWitt, Melinda C. Gross, and Amanda Dillon Rios. 2018. "Protein Supplementation During or Following a Marathon Run Influences Post-Exercise Recovery" Nutrients 10, no. 3: 333. https://doi.org/10.3390/nu10030333
APA StyleSaunders, M. J., Luden, N. D., DeWitt, C. R., Gross, M. C., & Dillon Rios, A. (2018). Protein Supplementation During or Following a Marathon Run Influences Post-Exercise Recovery. Nutrients, 10(3), 333. https://doi.org/10.3390/nu10030333




