Galacto-Oligosaccharide/Polidextrose Enriched Formula Protects against Respiratory Infections in Infants at High Risk of Atopy: A Randomized Clinical Trial
Abstract
:1. Introduction
2. Materials and Methods
2.1. Clinical Trials.Gov Identifier
2.2. Patient Selection Criteria
2.3. Randomisation
2.4. Study Product
2.5. Patient Follow-up
2.6. Outcomes
2.7. Microbiological and Faecal Markers
2.8. Statistical Methods
3. Results
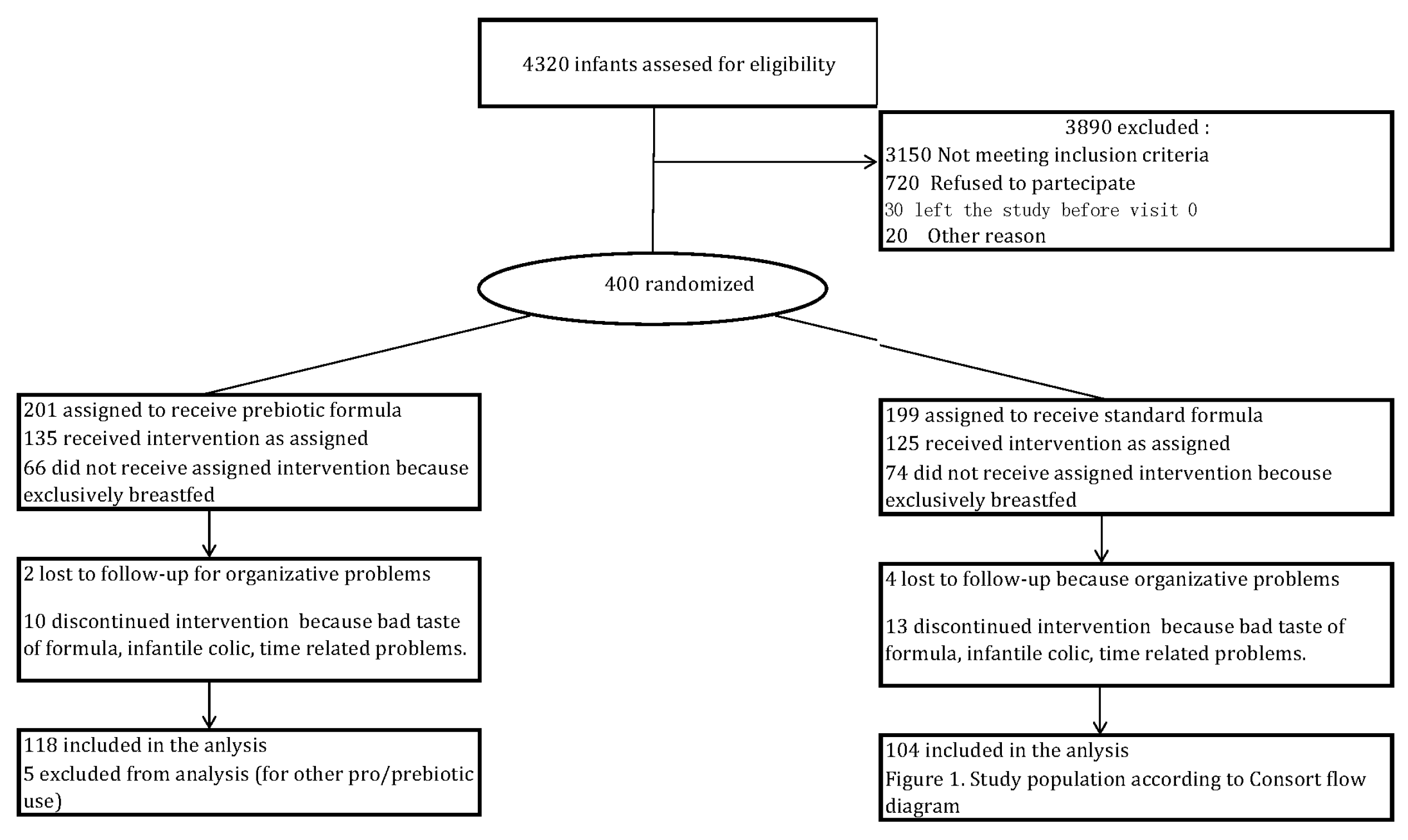
3.1. Patients
3.2. Clinical Outcomes
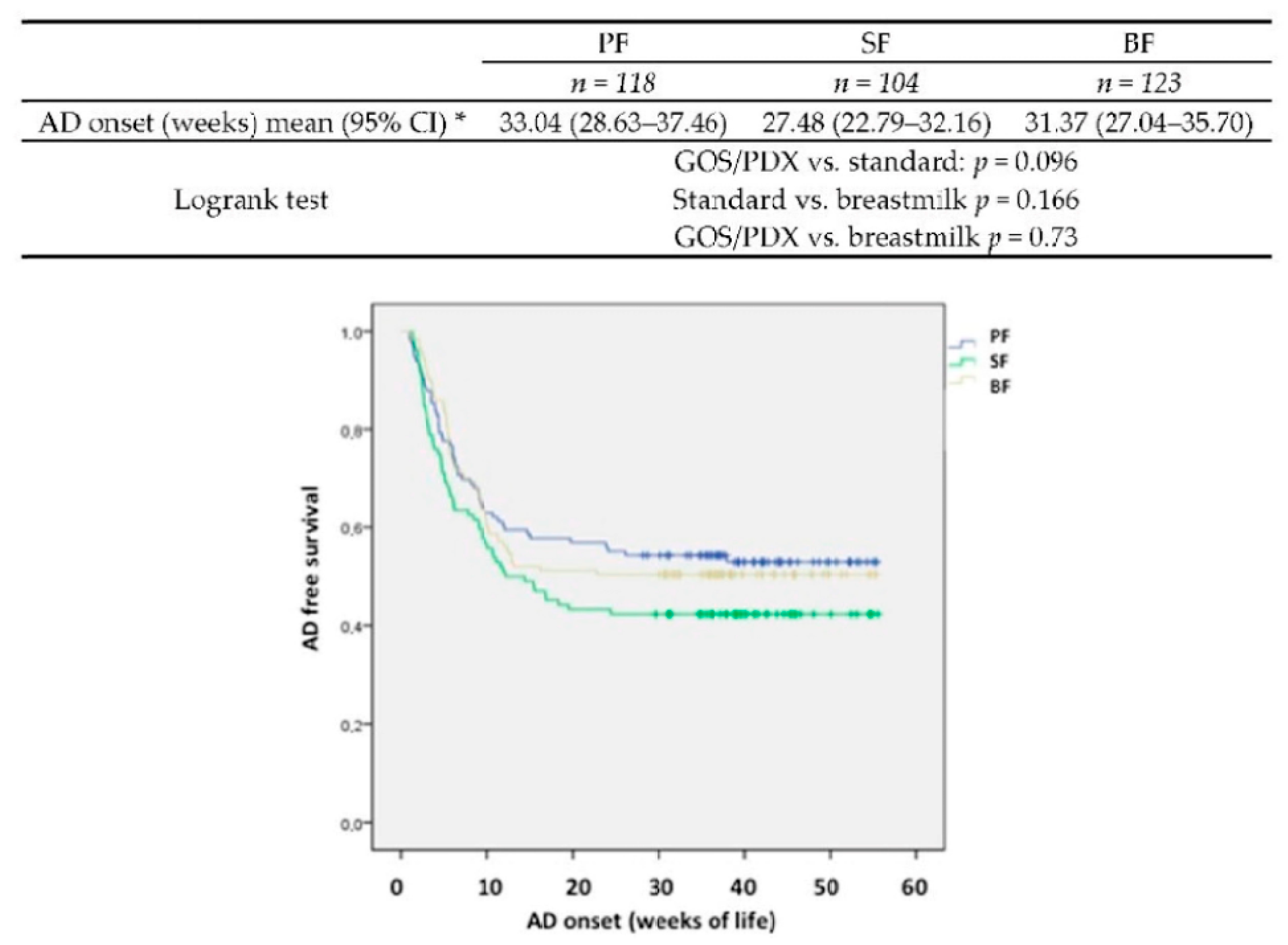
3.2.1. Atopic Dermatitis
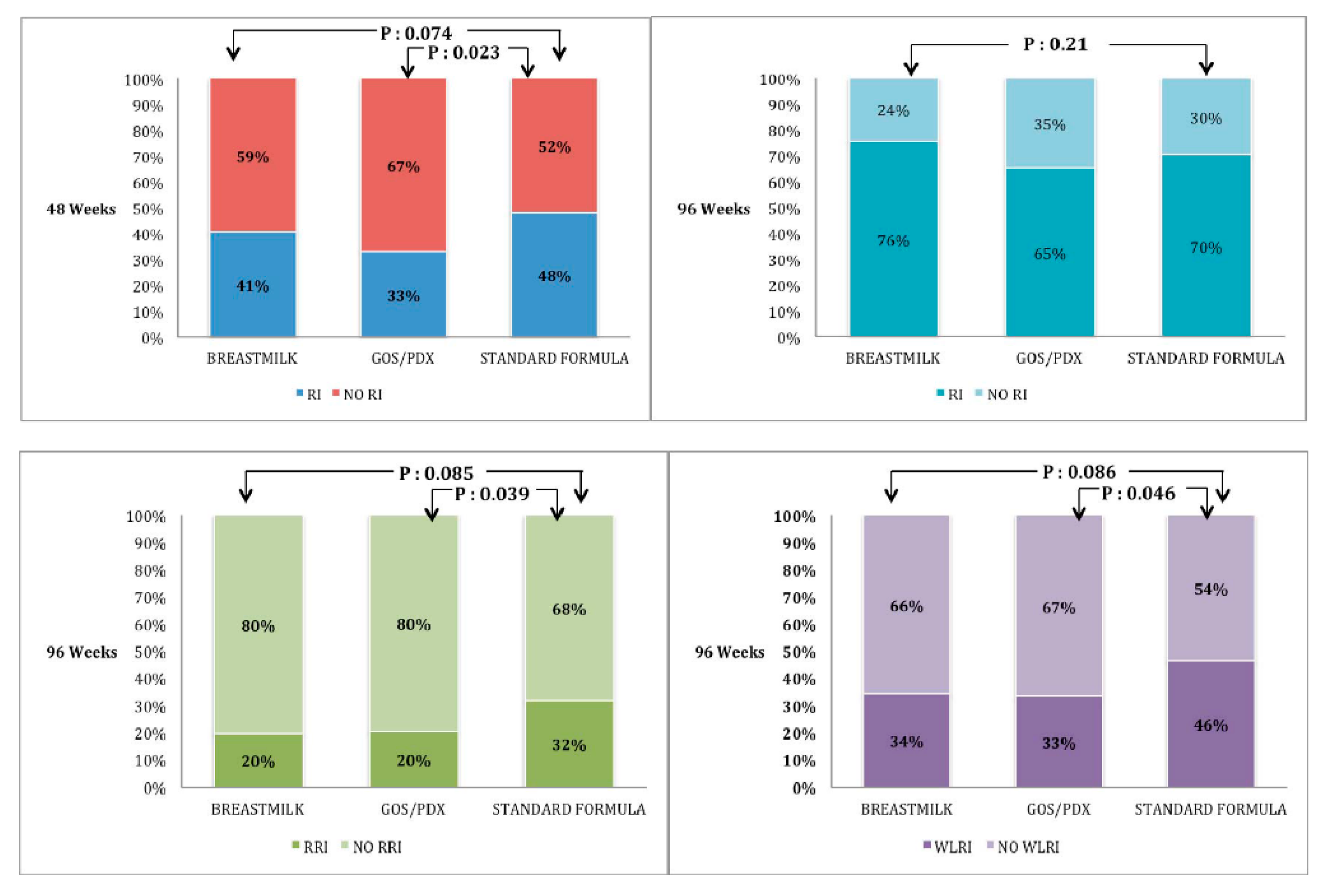
3.2.2. Respiratory Infections
3.2.3. Acute Gastroenteritis
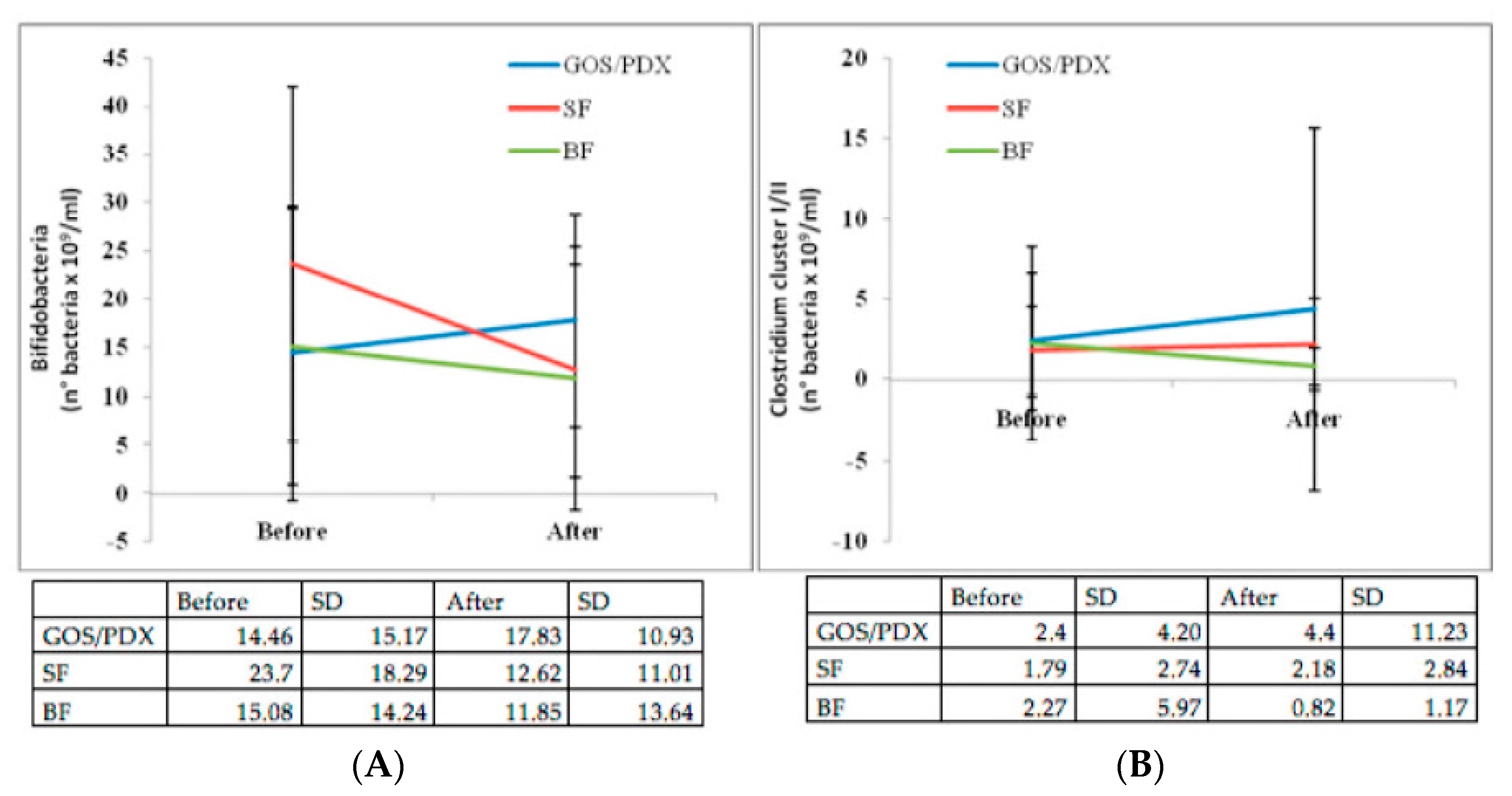
3.3. Intestinal Microbiology Substudy
3.3.1. Intestinal Microbiology According to Type of Feeding
3.3.2. Intestinal Microbiology According to the Development of Atopic Dermatitis and Common Infections
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| Prebiotics in the Prevention of Atopy | PIPA |
| Respiratory infection | RI |
| Fructooligosaccharide | FOS |
| Galactooligosaccharide | GOS |
| Polidextrose | PDX |
| Atopic Dermatitis | AD |
| Prebiotic (GOS/PDX)-enriched formula | PF |
| Breastfeeding | BF |
| Human nondigestible Milk Oligosaccharides | HMO |
| Randomized Controlled Trial | RCT |
| CONsolidated Standards of Reporting Trials | CONSORT |
| Recurrent RIs | RRIs |
| SCORing Atopic Dermatitis | SCORAD |
| Intention-To-Treat analysis | ITT |
| Per Protocol | PP |
| Standard Deviation | SD |
| Wheezing Lower espiratory-tract Illnesses | WLRIs |
| Acute Gastroenteritis | AG |
| Cystic Fibrosis | CF |
| Standard Formula | SF |
References
- Austin, S.; De Castro, C.A.; Bénet, T.; Hou, Y.; Sun, H.; Thakkar, S.K.; Vinyes-Pares, G.; Zhang, Y.; Wang, P. Temporal change of the content of 10 oligosaccharides in the milk of Chinese urban mothers. Nutrients 2016, 8. [Google Scholar] [CrossRef] [PubMed]
- Xu, G.; Davis, J.C.; Goonatilleke, E.; Smilowitz, J.T.; German, J.B.; Lebrilla, C.B. Absolute quantitation of human milk oligosaccharides reveals phenotypic variations during lactation. J. Nutr. 2017, 147, 117–124. [Google Scholar] [CrossRef] [PubMed]
- Harmsen, H.J.; Wildeboer-Veloo, A.C.; Raangs, G.C.; Wagendorp, A.A.; Klijn, N.; Bindels, J.G.; Welling, G.W. Analysis of intestinal flora development in breast-fed and formula-fed infants by using molecular identification and detection methods. J. Pediatr. Gastroenterol. Nutr. 2000, 30, 61–67. [Google Scholar] [CrossRef] [PubMed]
- Roberfroid, M.; Gibson, G.R.; Hoyles, L.; McCartney, A.L.; Rastall, R.; Rowland, I.; Wolvers, D.; Watzl, B.; Szajewska, H.; Stahl, B.; et al. Prebiotic effects: Metabolic and health benefits. Br. J. Nutr. 2010, 104, S1–S63. [Google Scholar] [CrossRef] [PubMed]
- Kusel, M.M.; de Klerk, N.; Holt, P.G.; Landau, L.I.; Sly, P.D. Occurrence and management of acute respiratory illnesses in early childhood. J. Paediatr. Child Health 2007, 43, 139–146. [Google Scholar] [CrossRef] [PubMed]
- Braegger, C.; Chmielewska, A.; Decsi, T.; Kolacek, S.; Mihatsch, W.; Moreno, L.; Pieścik, M.; Puntis, J.; Shamir, R.; Szajewska, H.; et al. ESPGHAN Committee on Nutrition. Supplementation of infant formula with probiotics and/or prebiotics: A systematic review and comment by the ESPGHAN committee on nutrition. J. Pediatr. Gastroenterol. Nutr. 2011, 52, 238–250. [Google Scholar] [CrossRef] [PubMed]
- Osborn, D.A.; Sinn, J.K. Prebiotics in infants for prevention of allergy. Cochrane Database Syst. Rev. 2013, 3. [Google Scholar] [CrossRef] [PubMed]
- Bruzzese, E.; Volpicelli, M.; Squeglia, V.; Bruzzese, D.; Salvini, F.; Bisceglia, M.; Lionetti, P.; Cinquetti, M.; Iacono, G.; Amarri, S.; et al. A formula containing galacto- and fructo-oligosaccharides prevents intestinal and extra-intestinal infections: an observational study. Clin. Nutr. 2009, 28, 156–161. [Google Scholar] [CrossRef] [PubMed]
- Arslanoglu, S.; Moro, G.E.; Boehm, G. Early supplementation of prebiotic oligosaccharides protects formula-fed infants against infections during the first 6 months of life. J. Nutr. 2007, 137, 2420–2424. [Google Scholar] [CrossRef] [PubMed]
- Arslanoglu, S.; Moro, G.E.; Schmitt, J.; Tandoi, L.; Rizzardi, S.; Boehm, G. Early dietary intervention with a mixture of prebiotic oligosaccharides reduces the incidence of allergic manifestations and infections during the first two years of life. J. Nutr. 2008, 138, 1091–1095. [Google Scholar] [CrossRef] [PubMed]
- Grüber, C.; van Stuijvenberg, M.; Mosca, F.; Moro, G.; Chirico, G.; Braegger, C.P.; Riedler, J.; Boehm, G.; Wahn, U. MIPS 1 Working Group. Reduced occurrence of early atopic dermatitis because of immunoactive prebiotics among low-atopy-risk infants. J. Allergy Clin. Immunol. 2010, 126, 791–797. [Google Scholar] [CrossRef] [PubMed]
- Grüber, C.; van Stuivenberg, M.; Mosca, F.; Moro, G.; Chirico, G.; Braegger, C.P.; Riedler, J.; Yavuz, Y.; Boehm, G.; Wahn, U. MIPS II Working Group. Immunoactive prebiotics transiently prevent occurrence of early atopic dermatitis among low-atopy-risk infants. J. Allergy Clin. Immunol. 2015, 136, 1696–1698. [Google Scholar] [CrossRef] [PubMed]
- Sierra, C.; Bernal, M.J.; Blasco, J.; Martínez, R.; Dalmau, J.; Ortuño, I.; Espín, B.; Vasallo, M.I.; Gil, D.; Vidal, M.L.; et al. Prebiotic effect during the first year of life in healthy infants fed formula containing GOS as the only prebiotic: A multicentre, randomised, double-blind and placebo-controlled trial. Eur. J. Nutr. 2015, 54, 89–99. [Google Scholar] [CrossRef] [PubMed]
- Herfel, T.M.; Jacobi, S.K.; Lin, X.; Fellner, V.; Walker, D.C.; Jouni, Z.E.; Odle, J. Polydextrose enrichment of infant formula demonstrates prebiotic characteristics by altering intestinal microbiota, organic acid concentrations, and cytokine expression in suckling piglets. J. Nutr. 2011, 141, 2139–2145. [Google Scholar] [CrossRef] [PubMed]
- Food and Drug Administration (FDA). GRAS Notification; GRN No. 236; FDA: Silver Spring, MD, USA, 2007.
- Kummeling, I.; Thijs, C.; Penders, J.; Snijders, B.E.; Stelma, F.; Reimerink, J.; Koopmans, M.; Dagnelie, P.C.; Huber, M.; Jansen, M.C.; et al. Etiology of atopy in infancy: The KOALA Birth Cohort Study. Pediatr. Allergy Immunol. 2005, 16, 679–684. [Google Scholar] [CrossRef] [PubMed]
- Halken, S.; Host, A. The lessons of noninterventional and interventional prospective studies on the development of atopic disease during childhood. Allergy 2000, 55, 793–802. [Google Scholar] [CrossRef] [PubMed]
- Muraro, A.; Dreborg, S.; Halken, S.; Host, A.; Niggemann, B.; Aalberse, R.; Arshad, S.H.; von Berg, A.; Karlsen, K.H.; Duschen, K.; et al. Dietary prevention of allergic diseases in infants and small children. Part II: evaluation of methods in allergy prevention studies and sensitization markers. Definitions and diagnostic criteria for allergic diseases. Pediatr. Allergy Immunol. 2004, 15, 196–205. [Google Scholar] [CrossRef] [PubMed]
- Guarino, A.; Bruzzese, E.; Lo Vecchio, A.; Dagan, R.; Tsolia, M. Definitions and outcomes of nutritional interventions in children with respiratory infections: the approach of the COMMENT initiative. Ann. Nutr. Metab. 2013, 63, 248–255. [Google Scholar] [CrossRef] [PubMed]
- Penders, J.; Stobberingh, E.E.; van den Brandt, P.A.; Thijs, C. The role of the intestinal microbiota in the development of atopic disorders. Allergy 2007, 62, 1223–1236. [Google Scholar] [CrossRef] [PubMed]
- Bruzzese, E.; Callegari, M.L.; Raia, V.; Viscovo, S.; Scotto, R.; Ferrari, S.; Morelli, L.; Buccigrossi, V.; Lo Vecchio, A.; Ruberto, E.; et al. Disrupted intestinal microbiota and intestinal inflammation in children with cystic fibrosis and its restoration with Lactobacillus GG: A randomised clinical trial. PLoS ONE 2014, 9. [Google Scholar] [CrossRef] [PubMed]
- Weichert, S.; Schroten, H.; Adam, R. The role of prebiotics and probiotics in prevention and treatment of childhood infectious diseases. Pediatr. Infect. Dis. J. 2012, 31, 859–862. [Google Scholar] [CrossRef] [PubMed]
- Kusel, M.M.; Kebadze, T.; Johnston, S.L.; Holt, P.G.; Sly, P.D. Febrile respiratory illnesses in infancy and atopy are risk factors for persistent asthma and wheeze. Eur. Respir. J. 2012, 39, 876–882. [Google Scholar] [CrossRef] [PubMed]
- Gidaris, D.; Urquhart, D.; Anthracopoulos, M.B. They said it was bronchiolitis: Is it going to turn into asthma doctor? Respirology 2014, 19, 1158–1164. [Google Scholar] [CrossRef] [PubMed]
- Kalliomaki, M.; Kirjavainen, P.; Eerola, E.; Kero, P.; Salminen, S.; Isolauri, E. Distinct patterns of neonatal gut microflora in infants in whom atopy was and was not developing. J. Allergy Clin. Immunol. 2001, 107, 129–134. [Google Scholar] [CrossRef] [PubMed]
- Mah, K.W.; Bjorkstemn, B.; Lee, B.W.; van Bever, H.P.; Shek, L.P.; Tan, T.N.; Lee, Y.K.; Chua, K.Y. Distinct pattern of commensal gut microbiota in toddlers with eczema. Int. Arch. Allergy Immunol. 2006, 140, 157–163. [Google Scholar] [CrossRef] [PubMed]
- Nakayama, J.; Kobayashi, T.; Tanaka, S.; Korenori, Y.; Tateyama, A.; Sakamoto, N.; Kiyohara, C.; Shirakawa, T.; Sonomoto, K. Aberrant structures of fecal bacterial community in allergic infants profiled by 16S rRNA gene pyrosequencing. FEMS Immunol. Med. Microbiol. 2011, 63, 397–406. [Google Scholar] [CrossRef] [PubMed]
- Penders, J.; Gerhold, K.; Stobberingh, E.E.; Thijs, C.; Zimmermann, K.; Lau, S.; Hamelmann, E. Establishment of the intestinal microbiota and its role for atopic dermatitis in early childhood. J. Allergy Clin. Immunol. 2013, 132, 601–607. [Google Scholar] [CrossRef] [PubMed]
- Kanai, T.; Mikami, Y.; Hayashi, A. A breakthrough in probiotics: Clostridium butyricum regulates gut homeostasis and anti-inflammatory response in inflammatory bowel disease. J. Gastroenterol. 2015, 50, 928–939. [Google Scholar] [CrossRef] [PubMed]
- Liao, H.Y.; Tao, L.; Zhao, J.; Qin, J.; Zeng, G.C.; Cai, S.W.; Li, Y.; Zhang, J.; Chen, H.G. Clostridium butyricum in combination with specific immunotherapy converts antigen-specific B cells to regulatory B cells in asthmatic patients. Sci. Rep. 2016, 6. [Google Scholar] [CrossRef] [PubMed]
- Bin, L.; Yang, F.; Lu, D.; Lin, Z. Specific immunotherapy plus Clostridium butyricum alleviates ulcerative colitis in patients with food allergy. Sci. Rep. 2016, 6. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.F.; Tang, Z.S.; Tong, L.; Tao, X.X.; Suo, Q.F.; Xu, X.M. Effects of clostridium butyricum and bifidobacterium on BTLA expression on CD4+ T cells and lymphocyte differentiation in late preterm infants. Microb. Pathog. 2016, 100, 112–118. [Google Scholar] [CrossRef] [PubMed]
- Moro, G.; Arslanoglu, S.; Stahl, B.; Jelinek, J.; Wahn, U.; Boehm, G. A mixture of prebiotic oligosaccharides reduces the incidence of atopic dermatitis during the first six months of age. Arch. Dis. Child. 2006, 91, 814–819. [Google Scholar] [CrossRef] [PubMed]
| PF | SF | BF | TOTAL | P | |
|---|---|---|---|---|---|
| (n = 118) | (n = 104) | (n = 123) | (n = 345) | ||
| Delivery | 0.085 | ||||
| Natural: n (%) | 74 (62.7%) | 69 (66.3%) | 93 (75.6%) | 236 (68.4%) | |
| Caesarean: n (%) | 44 (37.3%) | 35 (33.7%) | 30 (24.4%) | 109 (31.6%) | |
| Weight at birth (g) | 0.33 | ||||
| Mean ± SD | 3387.3 ± 378.3 | 3388.0 ± 424.1 | 3453.2 ± 370.7 | 3411.0 ± 390.2 | |
| Median | 3375 | 3352.5 | 3470 | 3390 | |
| (25–75 percentiles) | (3209–3629) | (3036.2–3651.2) | (3195–3670) | (3170–3645) | |
| Length at birth (cm) | 0.65 | ||||
| Mean ± SD | 49.37 ± 1.72 | 49.57 ± 1.93 | 49.55 ± 1.81 | 49.49 ± 1.81 | |
| Median (25–75 percentiles) | 49 (48–51) | 50 (48–51) | 50 (48–51) | 50 (48–51) | |
| Gestational age (weeks) | 0.91 | ||||
| Mean ± SD | 40.25 ± 2.29 | 40.34 ± 2.21 | 40.37 ± 2.06 | 40.32 ± 2.18 | |
| Median (25–75 percentiles) | 40 (39–42) | 40 (39–41.7) | 40 (39–42) | 40 (39–42) | |
| First born: n (%) | 56 (47.5%) | 60 (57.7%) | 58 (47.2%) | 174 (50.4%) | 0.21 |
| Number of siblings | 1 (0–1) | 1 (0–1) | 1 (0–1) | 1 (0–1) | 0.78 |
| Median (25–75 percentiles) | |||||
| Mother’s age (years) | 0.7 | ||||
| Mean ± SD | 34.03 ± 4.53 | 34.35 ± 4.60 | 34.5 ± 3.96 | 34.29 ± 4.35 | |
| Median (25–75 percentiles) | 34 (32–37) | 34 (31–38) | 35 (31–38) | 35 (31–37.5) | |
| Smoke in pregnancy: n (%) | 5 (4.2%) | 9 (8.7%) | 5 (4.1%) | 19 (5.5%) | 0.24 |
| Pets at home: n (%) | 25 (21.2%) | 35 (33.7%) | 31 (25.2%) | 91 (26.4%) | 0.1 |
| Mother’s education: n (%) | 0.52 | ||||
| Lower secondary | 9 (7.7%) | 5 (4.8%) | 5 (4.1%) | 19 (5.5%) | |
| Upper secondary | 52 (44.4%) | 46 (44.2%) | 47 (38.5%) | 145 (42.3%) | |
| Degree | 56 (47.9%) | 53 (51.0%) | 70 (57.4%) | 179 (52.2%) | |
| Father’s education: n (%) | 0.033 * | ||||
| Lower secondary | 23 (19.8%) | 16 (15.4%) | 8 (6.5%) | 47 (13.7%) | |
| Upper secondary | 53 (45.7%) | 46 (44.2%) | 57 (46.3%) | 156 (45.5%) | |
| Degree | 40 (34.5%) | 42 (40.4%) | 58 (47.2%) | 140 (40.8%) |
| PF | SF | BF | P | |
|---|---|---|---|---|
| n = 118 | n = 104 | n = 123 | ||
| AD episodes 36 weeks | 44 (37%) | 45 (43%) | 48 (39%) | 0.65 |
| AD episodes 48 weeks | 49 (41%) | 50 (48%) | 55 (45%) | 0.62 |
| AD episodes 96 weeks | 56 (47%) | 60 (58%) | 61 (50%) | 0.28 |
| AD drug use 48 weeks * | 33 (67%) | 28 (56%) | 31 (56%) | 0.42 |
| AD drug use 96 weeks * | 39 (70%) | 33(55%) | 36 (59%) | 0.25 |
| AD drug use > 3 times (median) 48 weeks * | 13 (33%) | 13 (32%) | 7 (13%) | 0.025 (1) |
| AD drug use > 3 times (median) 96 weeks * | 16 (29%) | 18 (30%) | 11 (18%) | 0.26 |
| Mean number of AD episodes 96 weeks | ||||
| Mean ± SD | 5.02 ± 8.44 | 5.56 ± 6.64 | 3.14 ± 2.87 | 0.11 |
| Median (25–75 percentiles) | 2 (1–5.25) | 4 (1–7) | 2 (1–4) | |
| Duration of AD (days) | ||||
| Mean ± SD | 9.74 ± 7.44 | 8.39 ± 8.04 | 10.07 ± 8.83 | 0.51 |
| Median (25–75 percentiles) | 7.33 (4.5–12.5) | 5.64 (3.25–11) | 7.48 (4.42–14) |
| Variable | Hazard Ratio (95% CI) | p |
|---|---|---|
| Prebiotic formula (versus standard formula) | 0.646 (0.369–1.129) | 0.09 |
| Breastfeeding (versus standard formula) | 0.512 (0.309–0.850) | 0.01 * |
| Breastifeeding (versus prebiotic formula) | 1.02 (0.543-1.214) | 0.71 |
| Female sex (vs. male) | 1.052 (0.684–1.619) | 0.82 |
| Pets at home (vs. no pets) | 0.947 (0.575–1.561) | 0.83 |
| Postsecondary education of father | 1.452 (0.922–2.286) | 0.11 |
| (vs. secondary) | ||
| Smoke in pregnancy | 0.84 (0.71–1.561) | 0.92 |
| Age of introduction of solid foods | 1.422 (1.034–1.957) | 0.03 * |
| No. of sibilings in household | 0.895 (0.717–1.118) | 0.33 |
| PF | SF | BF | P | |
|---|---|---|---|---|
| n = 118 | n = 104 | n = 123 | ||
| Infants with RI (at least one) until 48 weeks | 39 (33%) | 50 (48%) | 50 (41%) | 0.074 (1) |
| Infants with RI (at least one) until 96 weeks | 77 (65%) | 73 (70%) | 93 (76%) | 0.21 |
| Infants with RRI until 96 weeks | 24 (20%) | 33 (31%) | 24 (20%) | 0.085 (2) |
| Antibiotic use 48 weeks * | 38 (72%) | 43 (72%) | 57 (85%) | 0.12 |
| Antibiotic use 96 weeks * | 58 (75%) | 58 (79%) | 75 (81%) | 0.69 |
| Antibiotic use > 3 times (median) 48 weeks * | 8 (15%) | 8 (13%) | 8 (12%) | 0.88 |
| Antibiotic use > 3 times (median) 96 weeks * | 22 (29%) | 26 (36%) | 14 (15%) | 0.008 (3) |
| Mean number of RI episodes 48 weeks | ||||
| Mean ± standard deviation | 1.9 ± 1.49 | 2.93 ± 1.11 | 2.2 ± 1.7 | 0.013 (4) |
| Median | 1 (1–2) | 2 (2–4) | 1 (1–4) | |
| Mean number of RI episodes/96 weeks | 0.06 (5) | |||
| Mean ± SD | 4.21 ± 1.89 | 4.93 ± 2.11 | 3.8 ± 2.16 | |
| Median (25th–75th percentiles) | 4 (2–6) | 4 (2–6) | 3 (2–5) | |
| Duration of RI (days) | ||||
| Mean ± SD | 5.85 ± 2.33 | 5.90 ± 2.68 | 5.99 ± 2.29 | 0.93 |
| Median (25th–75th percentiles) | 5.67 (4–7) | 5.5 (4.32–7) | 6 (4.9–7) | |
| Wheezy lower RI 48 weeks | 33 (28%) | 38 (36%) | 36 (29%) | 0.34 |
| Wheezy lower RI 96 weeks | 39 (33%) | 48 (46%) | 42 (34%) | 0.086 (6) |
| Number of parent workdays lost within 96 weeks | 36 (30%) | 28 (27%) | 26 (21%) | 0.25 |
| Number of days lost | 7.03 ± 7.56 | 6.57 ± 10.21 | 9.85 ± 23.03 | 0.66 |
| Mean ± SD | 5 (2–9) | 3 (1–9) | 3 (2–5.25) | |
| Median (25th–75th percentiles) |
| PF | SF | BF | P | |
|---|---|---|---|---|
| n = 118 | n = 104 | n = 123 | ||
| Infants with acute diarrhea (at least once) until 48 weeks | 50 (42%) | 50 (48%) | 33 (27%) | 0.003 (1) |
| Infants with acute diarrhea (at least once) until 96 weeks | 62 (52%) | 65 (62%) | 50 (41%) | 0.004 (2) |
| Mean number of diarrhea episodes at 96 weeks | 0.16 | |||
| Mean ± SD | 2.52 ± 3.04 | 2.66 ± 2.77 | 1.78 ± 1.28 | |
| Median (25–75 percentiles) | 2 (1–3) | 2 (1–3) | 1 (1–2) |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ranucci, G.; Buccigrossi, V.; Borgia, E.; Piacentini, D.; Visentin, F.; Cantarutti, L.; Baiardi, P.; Felisi, M.; Spagnuolo, M.I.; Zanconato, S.; et al. Galacto-Oligosaccharide/Polidextrose Enriched Formula Protects against Respiratory Infections in Infants at High Risk of Atopy: A Randomized Clinical Trial. Nutrients 2018, 10, 286. https://doi.org/10.3390/nu10030286
Ranucci G, Buccigrossi V, Borgia E, Piacentini D, Visentin F, Cantarutti L, Baiardi P, Felisi M, Spagnuolo MI, Zanconato S, et al. Galacto-Oligosaccharide/Polidextrose Enriched Formula Protects against Respiratory Infections in Infants at High Risk of Atopy: A Randomized Clinical Trial. Nutrients. 2018; 10(3):286. https://doi.org/10.3390/nu10030286
Chicago/Turabian StyleRanucci, Giusy, Vittoria Buccigrossi, Eleonora Borgia, Daniela Piacentini, Federica Visentin, Luigi Cantarutti, Paola Baiardi, Mariagrazia Felisi, Maria Immacolata Spagnuolo, Stefania Zanconato, and et al. 2018. "Galacto-Oligosaccharide/Polidextrose Enriched Formula Protects against Respiratory Infections in Infants at High Risk of Atopy: A Randomized Clinical Trial" Nutrients 10, no. 3: 286. https://doi.org/10.3390/nu10030286
APA StyleRanucci, G., Buccigrossi, V., Borgia, E., Piacentini, D., Visentin, F., Cantarutti, L., Baiardi, P., Felisi, M., Spagnuolo, M. I., Zanconato, S., Baraldi, E., Giaquinto, C., & Guarino, A. (2018). Galacto-Oligosaccharide/Polidextrose Enriched Formula Protects against Respiratory Infections in Infants at High Risk of Atopy: A Randomized Clinical Trial. Nutrients, 10(3), 286. https://doi.org/10.3390/nu10030286






