Aged Oolong Tea Reduces High-Fat Diet-Induced Fat Accumulation and Dyslipidemia by Regulating the AMPK/ACC Signaling Pathway
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation of EAOTs
2.3. Determination of Ingredients in EAOTs
2.4. Animals
2.5. Diet-Induced Obesity
2.6. EAOT Treatment
2.7. Lee’s Index
2.8. Serum Chemistry Analysis
2.9. Histological Examination
2.10. Western Blotting Analysis
2.11. Immunohistochemistry (IHC)
2.12. Statistical Analysis
3. Results
3.1. The Ingredients of EAOTs
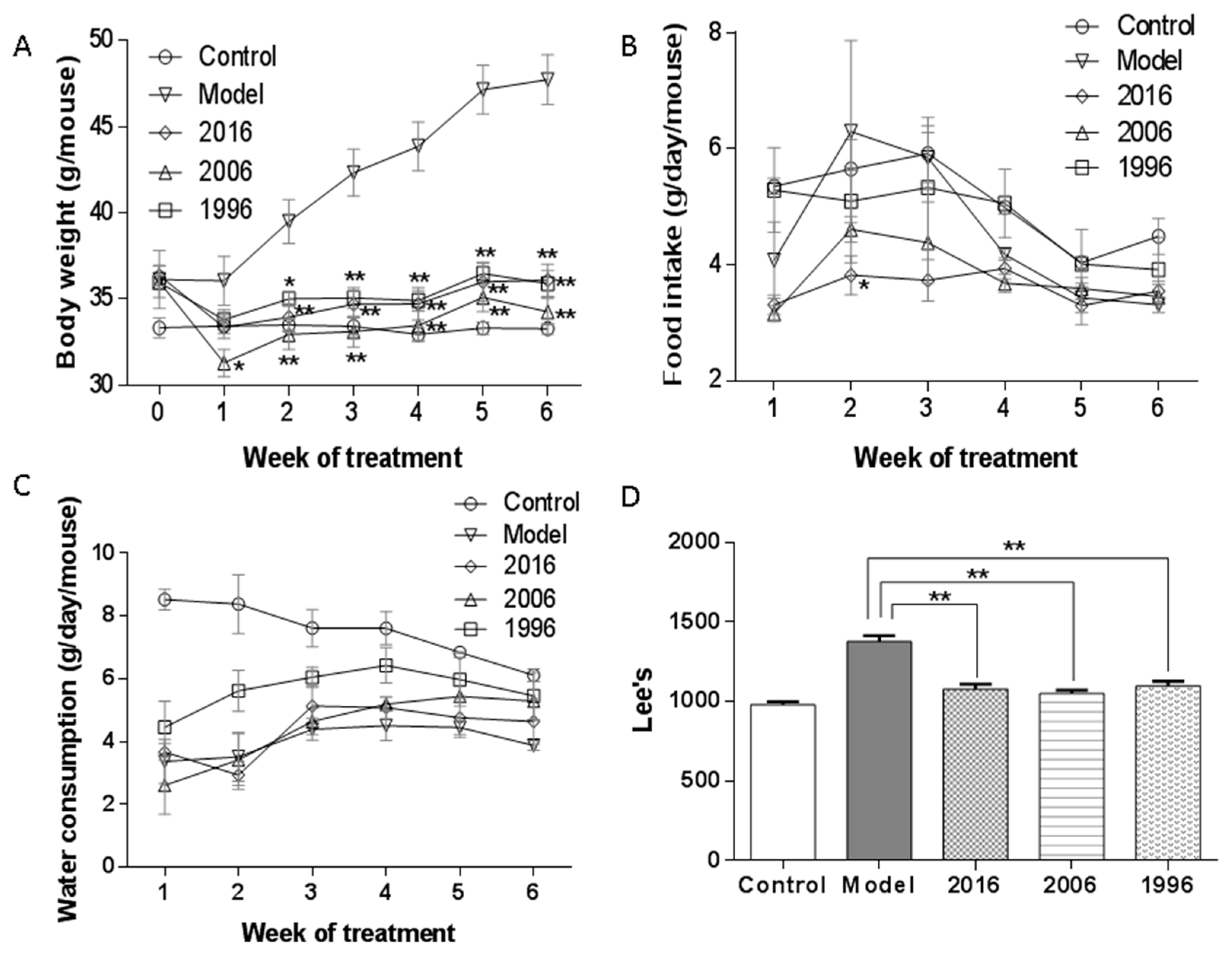
3.2. Body Weight, Food and Water Intake, and Lee’s Index
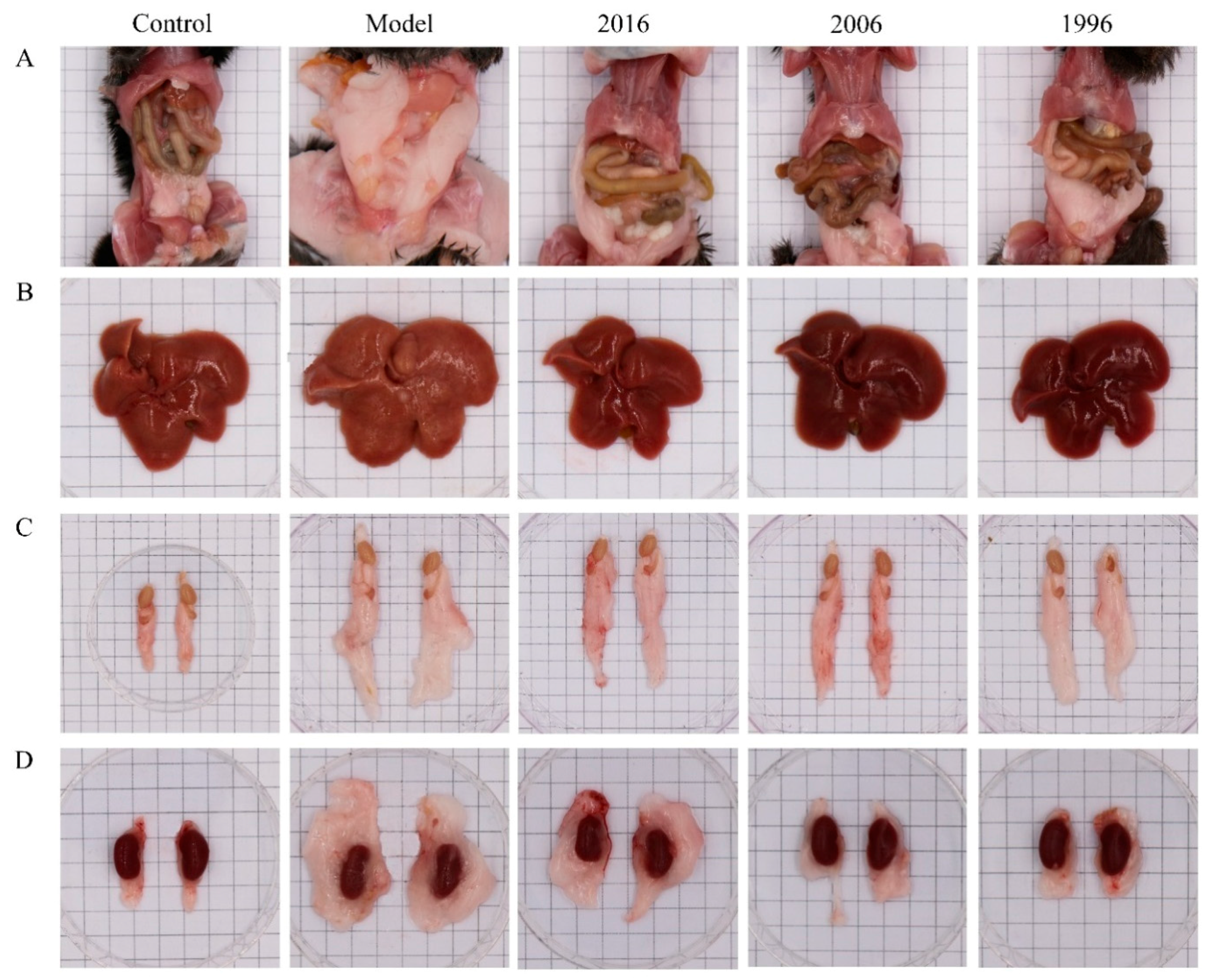
3.3. EAOTs Attenuate Fatty Liver and Adiposity in HFD-Induced Obese Mice
3.4. Effects of EAOTs on Serum Levels in HFD-Induced Obesity Mice
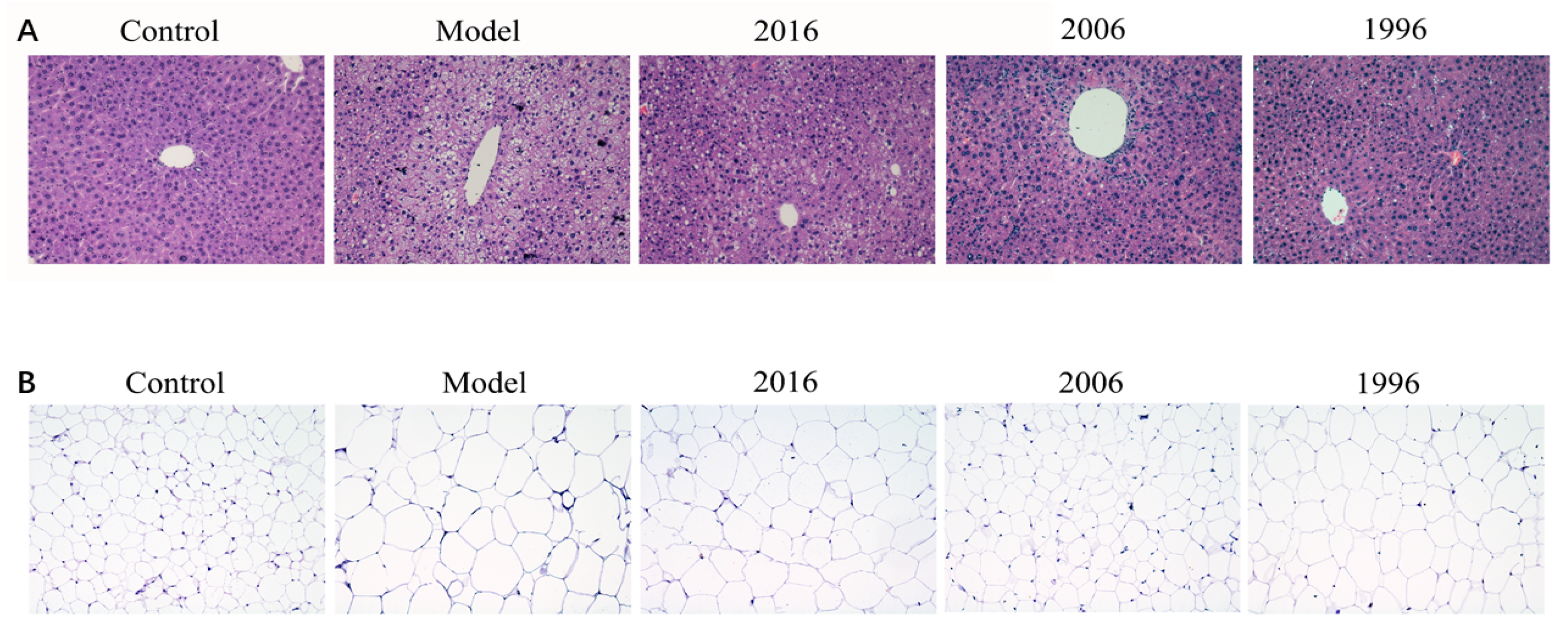
3.5. Effect of EAOTs on Accumulation of Lipid Droplets in Liver and Epididymal Fat
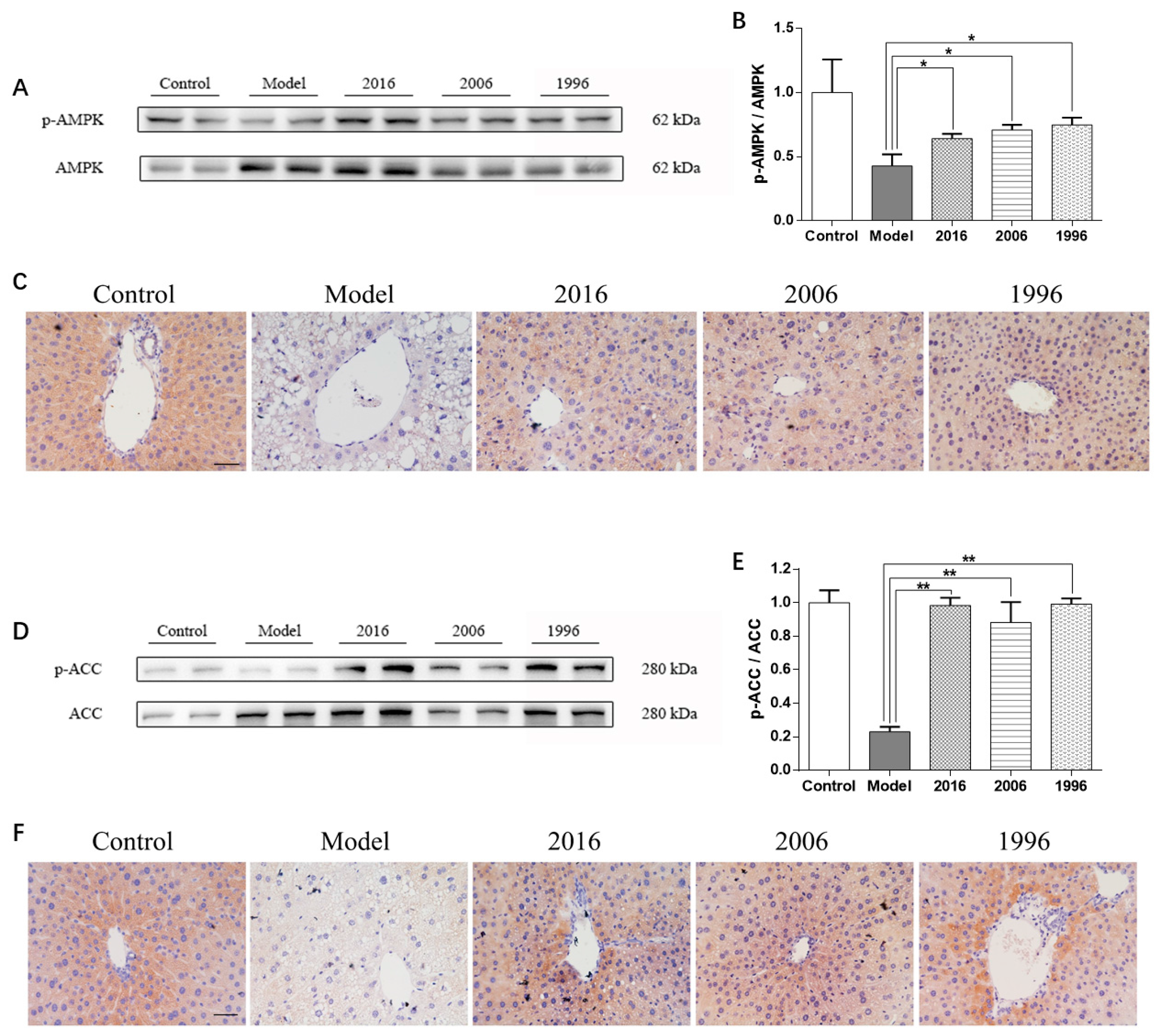
3.6. EAOTs Activate AMPK and ACC Phosphorylation
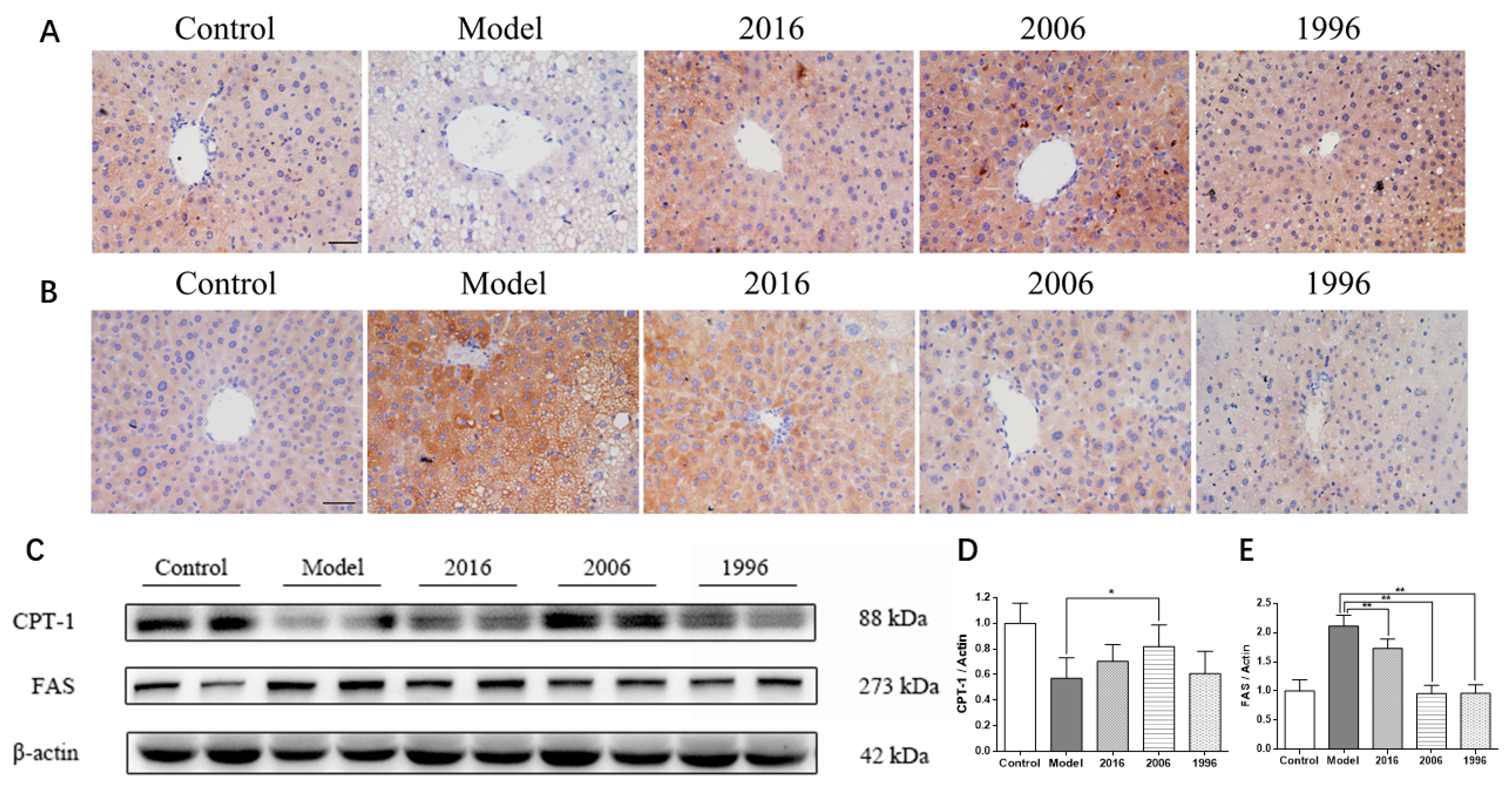
3.7. EAOTs Activate CPT-1 Expression and Inhibit FAS Expression
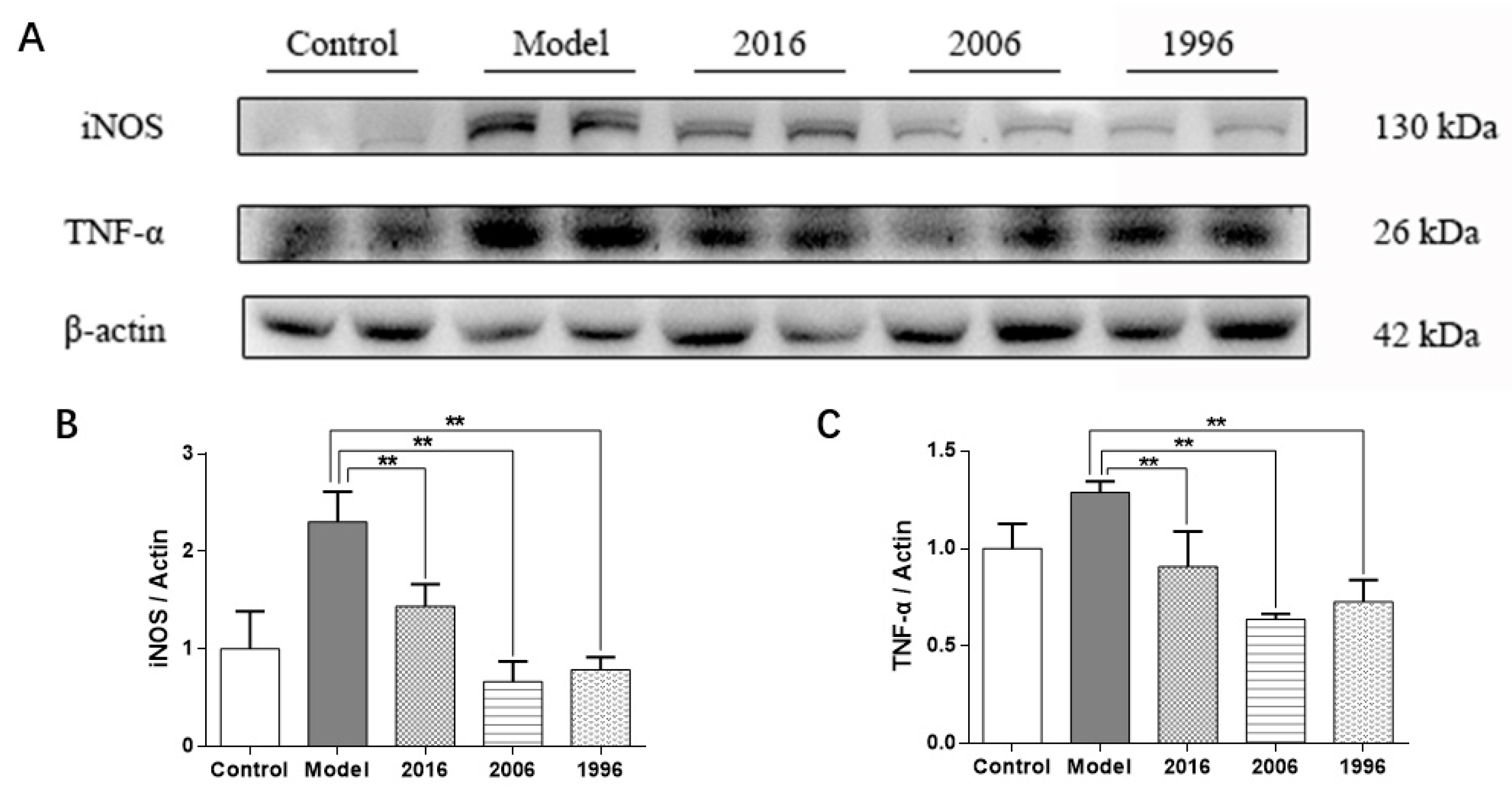
3.8. EAOTs Inhibit iNOS and TNF-α Expression
4. Discussion
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Keith, S.W.; Redden, D.T.; Katzmarzyk, P.T.; Boggiano, M.M.; Hanlon, E.C.; Benca, R.M.; Ruden, D.; Pietrobelli, A.; Barger, J.L.; Fontaine, K.; et al. Putative contributors to the secular increase in obesity: Exploring the roads less traveled. Int. J. Obes. 2006, 30, 1585–1594. [Google Scholar] [CrossRef] [PubMed]
- McAllister, E.J.; Dhurandhar, N.V.; Keith, S.W.; Aronne, L.J.; Barger, J.; Baskin, M.; Benca, R.M.; Biggio, J.; Boggiano, M.M.; Eisenmann, J.C.; et al. Ten putative contributors to the obesity epidemic. Crit. Rev. Food Sci. Nutr. 2009, 49, 868–913. [Google Scholar] [CrossRef] [PubMed]
- Gauvreau, D.; Villeneuve, N.; Deshaies, Y.; Cianflone, K. Novel adipokines: Links between obesity and atherosclerosis. Ann. Endocrinol. 2011, 72, 224–231. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Lewis, J.R.; Baur, L.A.; Macaskill, P.; Craig, J.C. Obesity and hypertension in Australian young people: Results from the Australian Health Survey 2011–2012. Intern. Med. J. 2017, 47, 162–170. [Google Scholar] [CrossRef] [PubMed]
- Colditz, G.A.; Peterson, L.L. Obesity and cancer: Evidence, impact, and future directions. Clin. Chem. 2017, 64, 154–162. [Google Scholar] [CrossRef] [PubMed]
- Domingo, K.B.; Chertow, G.M.; Coxson, P.G.; Moran, A.; Lightwood, J.M.; Pletcher, M.J.; Goldman, L. Projected effect of dietary salt reductions on future cardiovascular disease. N. Engl. J. Med. 2010, 362, 590–599. [Google Scholar] [CrossRef] [PubMed]
- Zare, H.; Sarvestani, F.S. Combined effects of physical exercise and green tea on obese people. Int. J. Ayurvedic. Herb. Med. 2012, 2, 435–440. [Google Scholar]
- Heber, D.; Zhang, Y.; Yang, J.; Ma, J.E.; Henning, S.M.; Li, Z. Green tea, black tea and oolong tea polyphenols reduce visceral fat and inflammation in mice fed high-fat, high-sucrose obesogenic diets. J. Nutr. 2014, 144, 1385–1393. [Google Scholar] [CrossRef] [PubMed]
- Kuo, K.L.; Weng, M.S.; Chiang, C.T.; Tsai, Y.J.; Lin-Shiau, S.Y.; Lin, J.K. Comparative studies on the hypolipidemic and growth suppressive effects of oolong, black, pu-erh, and green tea leaves in rats. J. Agric. Food Chem. 2005, 53, 480–489. [Google Scholar] [CrossRef] [PubMed]
- Ku, K.M.; Kim, J.; Park, H.J.; Liu, K.H.; Lee, C.H. Application of metabolomics in the analysis of manufacturing type of pu-erh tea and composition changes with different postfermentation year. J. Agric. Food Chem. 2009, 58, 345–352. [Google Scholar] [CrossRef] [PubMed]
- Hariri, N.; Thibault, L. High-fat diet-induced obesity in animal models. Nutr. Res. Rev. 2010, 23, 270–299. [Google Scholar] [CrossRef] [PubMed]
- Friedman, M.; Levin, C.E.; Lee, S.U.; Kozukue, N. Stability of green tea catechins in commercial tea leaves during storage for 6 months. J. Food Sci. 2009, 74, H47–H51. [Google Scholar] [CrossRef] [PubMed]
- Hawley, S.A.; Davison, M.; Woods, A.; Davies, S.P.; Beri, R.K.; Carling, D.; Hardie, D.G. Characterization of the AMP-activated protein kinase kinase from rat liver and identification of threonine 172 as the major site at which it phosphorylates AMP-activated protein kinase. J. Biol. Chem. 1996, 271, 27879–27887. [Google Scholar] [CrossRef] [PubMed]
- Boone, A.N.; Rodrigues, B.; Brownsey, R.W. Multiple-site phosphorylation of the 280 kDa isoform of acetyl-CoA carboxylase in rat cardiac myocytes: Evidence that cAMP-dependent protein kinase mediates effects of beta-adrenergic stimulation. Biochem. J. 1999, 341, 347–354. [Google Scholar] [CrossRef] [PubMed]
- Tiegs, G. Cellular and cytokine-mediated mechanisms of inflammation and its modulation in immune-mediated liver injury. Z. Gastroenterol. 2007, 45, 63–70. [Google Scholar] [CrossRef] [PubMed]
- Scherer, P.E. Adipose tissue: From lipid storage compartment to endocrine organ. Diabetes 2006, 55, 1537–1545. [Google Scholar] [CrossRef] [PubMed]
- Shepherd, P.R.; Gnudi, L.; Tozzo, E.; Kahn, B.B. Adipose cell hyperplasia and enhanced glucose disposal in transgenic mice overexpressing glut4 selectively in adipose tissue. J. Biol. Chem. 1993, 268, 22243–22246. [Google Scholar] [PubMed]
- Kopelman, P.G. Obesity as a medical problem. Nature 2000, 404, 635–643. [Google Scholar] [CrossRef] [PubMed]
- Haslam, D.W.; James, W.P. Obesity. Lancet 2005, 366, 1197–1209. [Google Scholar] [CrossRef]
- Zhang, B.B.; Zhou, G.; Li, C. AMPK: An emerging drug target for diabetes and the metabolic syndrome. Cell Metab. 2009, 9, 407–416. [Google Scholar] [CrossRef] [PubMed]
- Kjøbsted, R.; Pedersen, A.J.T.; Hingst, J.R.; Sabaratnam, R.; Birk, J.B.; Kristensen, J.M.; Højlund, K.; Wojtaszewski, J.F. Intact regulation of the AMPK signaling network in response to exercise and insulin in skeletal muscle of male patients with type 2 diabetes—Illumination of AMPK activation in recovery from exercise. Diabetes 2016, 65, 1219–1230. [Google Scholar] [CrossRef] [PubMed]
- Viollet, B.; Guigas, B.; Leclerc, J.; Hebrard, S.; Lantier, L.; Mounier, R.; Andreelli, F.; Foretz, M. AMP-activated protein kinase in the regulation of hepatic energy metabolism: From physiology to therapeutic perspectives. Acta Physiol. 2009, 196, 81–98. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Liu, X.; Han, L.; Gao, X.; Liu, E.; Wang, T. Regulation of lipid and glucose homeostasis by mango tree leaf extract is mediated by AMPK and PI3K/AKT signaling pathways. Food Chem. 2013, 141, 2896–2905. [Google Scholar] [CrossRef] [PubMed]
- Lin, W.S.; Lo, J.H.; Yang, J.H.; Wang, H.W.; Fan, S.Z.; Yen, J.H.; Wang, P.Y. Ludwigiaoctovalvis extract improves glycemic control and memory performance in diabetic mice. J. Ethnopharmacol. 2017, 207, 211–219. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.J.; Bang, C.Y.; Guo, Y.R.; Choung, S.Y. Anti-obesity effects of Aster spathulifolius extract in high-fat diet-induced obese rats. J. Med. Food 2016, 19, 353–364. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Chen, G.; Li, N.; Dai, M.; Chen, C.; Wang, P.; Tang, H.; Hoopes, S.L.; Zeldin, D.C.; Wang, D.W.; et al. CYP2J2 overexpression ameliorates hyperlipidemia via increased fatty acid oxidation mediated by the AMPK pathway. Obesity 2015, 23, 1401–1413. [Google Scholar] [CrossRef] [PubMed]
- Brownsey, R.W.; Boone, A.N.; Elliott, J.E.; Kulpa, J.E.; Lee, W.M. Regulation of acetyl-CoA carboxylase. Biochem. Soc. Trans. 2006, 34, 223–227. [Google Scholar] [CrossRef] [PubMed]
- Wolfgang, M.J.; Lane, M.D. Hypothalamic malonyl-CoA and CPT1c in the treatment of obesity. FEBS J. 2011, 278, 552–558. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.K.; Kim, S.H.; Yu, H.S.; Park, H.G.; Kang, H.G.; Ahn, Y.M.; Kim, Y.S. The effect of clozapine on the AMPK-ACC-CPT-1 pathway in the rat frontal cortex. Int. J. Neuropsychopharmacol. 2012, 15, 907–917. [Google Scholar] [CrossRef] [PubMed]
- Boizard, M.; Le, L.X.; Lemarchand, P.; Foufelle, F.; Ferre, P.; Dugail, I. Obesity-related overexpression of fatty-acid synthase gene in adipose tissue involves sterol regulatory element-binding protein transcription factors. J. Biol. Chem. 1998, 273, 29164–29171. [Google Scholar] [CrossRef] [PubMed]
- Moon, Y.S.; Latasa, M.J.; Griffin, M.J.; Sul, H.S. Suppression of fatty acid synthase promoter by polyunsaturated fatty acids. J. Lipid Res. 2002, 43, 691–698. [Google Scholar] [PubMed]
- Jeong, H.W.; Hsu, K.C.; Lee, J.W.; Ham, M.; Huh, J.Y.; Shin, H.J.; Kim, W.S.; Kim, J.B. Berberine suppresses proinflammatory responses through AMPK activation in macrophages. Am. J. Physiol.-Endocrinol. Metab. 2009, 296, E955–E964. [Google Scholar] [CrossRef] [PubMed]
| Sample | Water (%) | Free Amino Acids (%) | Total Soluble Sugar (%) | Tea Polyphenols (%) |
|---|---|---|---|---|
| 2016 | 0.07 ± 0.01 a | 5.13 ± 0.03 a | 7.08 ± 0.25 a | 39.7 ± 0.3 a |
| 2006 | 0.08 ± 0.01 a | 3.99 ± 0.19 b | 8.18 ± 0.08 b | 46.5 ± 0.6 b |
| 1996 | 0.06 ± 0.01 a | 3.81 ± 0.04 b | 8.39 ± 0.08 b | 34.0 ± 0.5 c |
| Group | Liver/Weight (%) | Kidney/Weight (%) | Epididymal Fat/Weight (%) | Intestinal Fat/Weight (%) | Pararenal Fat/Wright (%) | White Fat/Weight (%) |
|---|---|---|---|---|---|---|
| Control | 44.5 ± 0.1 a | 13.0 ± 1.0 a | 1.30 ± 0.46 a | 5.14 ± 2.43 a | 4.15 ± 1.98 a | 9.31 ± 4.32 a |
| Model | 45.2 ± 0.5 a | 9.39 ± 0.88 b | 5.35 ± 0.29 b | 23.9 ± 5.3 b | 30.6 ± 3.3 b | 56.3 ± 6.1 b |
| 2016 | 36.4 ± 0.1 b | 11.9 ± 0.9 a | 3.02 ± 1.16 c | 10.4 ± 5.4 a | 16.4 ± 8.0 c | 24.7 ± 16.3 c |
| 2006 | 37.0 ± 0.1 b | 12.4 ± 1.5 a | 2.77 ± 0.75 c | 7.73 ± 3.76 a | 11.8 ± 6.1 a,c | 16.4 ± 5.6 a,c |
| 1996 | 37.0 ± 0.1 b | 12.4 ± 1.6 a | 2.91 ± 1.07 c | 9.20 ± 4.65 a | 12.8 ± 6.5 c | 19.7 ± 9.3 a,c |
| Group | TG (mmol/L) | TC (mmol/L) | HDL-C (mmol/L) | LDL-C (mmol/L) |
|---|---|---|---|---|
| Control | 1.41 ± 0.30 a | 4.18 ± 0.58 a | 2.78 ± 0.37 a | 0.55 ± 0.12 a |
| Model | 1.95 ± 0.34 b | 9.25 ± 1.30 b | 2.26 ± 0.24 a | 1.46 ± 0.33 b |
| 2016 | 1.45 ± 0.22 a | 6.20 ± 0.59 c | 4.03 ± 0.44 b | 1.00 ± 0.11 c |
| 2006 | 1.13 ± 0.26 a | 5.74 ± 0.46 c | 3.80 ± 0.36 b | 0.86 ± 0.10 c |
| 1996 | 1.19 ± 0.24 a | 6.42 ± 0.84 c | 4.21 ± 0.58 b | 0.97 ± 0.24 c |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yuan, E.; Duan, X.; Xiang, L.; Ren, J.; Lai, X.; Li, Q.; Sun, L.; Sun, S. Aged Oolong Tea Reduces High-Fat Diet-Induced Fat Accumulation and Dyslipidemia by Regulating the AMPK/ACC Signaling Pathway. Nutrients 2018, 10, 187. https://doi.org/10.3390/nu10020187
Yuan E, Duan X, Xiang L, Ren J, Lai X, Li Q, Sun L, Sun S. Aged Oolong Tea Reduces High-Fat Diet-Induced Fat Accumulation and Dyslipidemia by Regulating the AMPK/ACC Signaling Pathway. Nutrients. 2018; 10(2):187. https://doi.org/10.3390/nu10020187
Chicago/Turabian StyleYuan, Erdong, Xuefei Duan, Limin Xiang, Jiaoyan Ren, Xingfei Lai, Qiuhua Li, Lingli Sun, and Shili Sun. 2018. "Aged Oolong Tea Reduces High-Fat Diet-Induced Fat Accumulation and Dyslipidemia by Regulating the AMPK/ACC Signaling Pathway" Nutrients 10, no. 2: 187. https://doi.org/10.3390/nu10020187
APA StyleYuan, E., Duan, X., Xiang, L., Ren, J., Lai, X., Li, Q., Sun, L., & Sun, S. (2018). Aged Oolong Tea Reduces High-Fat Diet-Induced Fat Accumulation and Dyslipidemia by Regulating the AMPK/ACC Signaling Pathway. Nutrients, 10(2), 187. https://doi.org/10.3390/nu10020187





