Dietary Magnesium and Cardiovascular Disease: A Review with Emphasis in Epidemiological Studies
Abstract
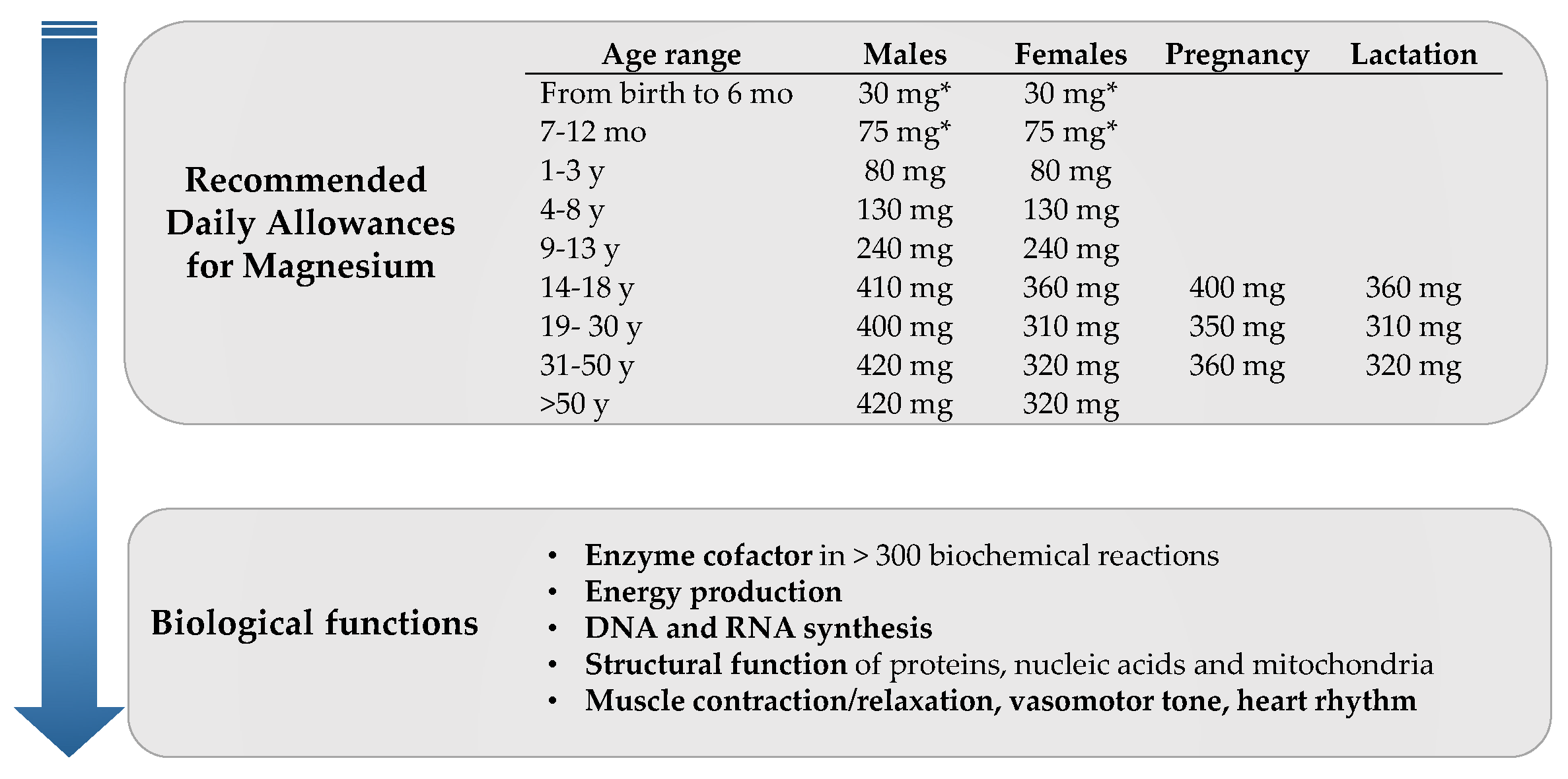
1. Introduction
2. Dietary Magnesium and Cardiovascular Disease Risk Factors
2.1. Type 2 Diabetes and Metabolic Syndrome
2.2. Hypertension and Endothelial Function
2.3. Lipid Profile
3. Dietary Magnesium and Cardiovascular Disease
3.1. Stroke
3.2. Coronary Heart Disease
3.3. Heart Failure
3.4. Atrial Fibrillation
3.5. Cardiovascular Death
4. Circulating Magnesium and Cardiovascular Disease
4.1. Cardiovascular Disease, Coronary Disease and Stroke
4.2. Atrial Fibrillation and Sudden Death
4.3. Left Ventricular Hypertrophy and Heart Failure
4.4. Atherosclerosis and Coronary Artery Calcification
4.5. Cardiovascular Death
5. Plausible Mechanisms Connecting Magnesium and Cardiovascular Disease
5.1. Oxidative and Inflammatory Stress
5.2. Lipid Profile and Peroxidation
5.3. Glucose Homeostasis/Type 2 Diabetes
5.4. Endothelial Function, Blood Pressure and Hypertension
5.5. Cardiovascular Disease Events: Arrhythmia and Acute Myocardial Infarction
5.6. A Focus on Magnesium Receptors
6. Final Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| ARIC | Atherosclerosis Risk in Communities |
| ATP | adenosine triphosphate |
| BMI | body mass index |
| BP | blood pressure |
| Ca2+ | calcium |
| CHD | coronary heart disease |
| CV | cardiovascular |
| CVD | cardiovascular disease |
| DNA | deoxyribonucleic acid |
| GLUT 4 | glucose transporter protein activity 4 |
| HDL | high-density lipoprotein |
| HF | heart failure |
| HR | hazard ratio |
| HOMA-IR | homeostatic model assessment of insulin resistance |
| IHD | ischemic heart disease |
| IL | interleukin |
| IR | insulin resistance |
| K+ | potassium |
| LDL | low-density lipoprotein |
| MetS | metabolic syndrome |
| Mg | magnesium |
| Mg-D | Mg-deficiency |
| [Mg2+] | magnesium concentrations |
| Mg2+ | intracellular magnesium |
| Na+ | sodium |
| NO | nitric oxide |
| O2 | oxygen |
| T2D | type 2 diabetes |
| TGRLP | triglyceride-rich lipoproteins |
| TRPM | transient receptor potential melastatin |
| RCT | randomized control trial |
| RDA | recommended dietary allowance |
| RR | Relative Risk |
| sICAM-1 | soluble intercellular adhesion molecule 1 |
| SLC41A1 | solute carrier family 41 member 1 |
| SHR | spontaneously hypertensive rats |
| UVEC | umbilical vein endothelial cells |
| VCAM | vascular cell adhesion protein 1 |
| VSMC | vascular smooth muscle cells |
| WKY | Wistar-Kyoto rats |
References
- Gröber, U.; Schmidt, J.; Kisters, K. Magnesium in Prevention and Therapy. Nutrients 2015, 8199–8226. [Google Scholar] [CrossRef] [PubMed]
- Standing Committee on the Scientific Evaluation of Dietary Reference Intakes; Food and Nutrition Board; Institute of Medicine (IOM). Dietary Reference Intakes for Calcium, Phosphorus, Magnesium, Vitamin D, and Fluoride; National Academies Press: Washington, DC, USA, 1997. [Google Scholar]
- Fang, X.; Wang, K.; Han, D.; He, X.; Wei, J.; Zhao, L.; Imam, M.U.; Ping, Z.; Li, Y.; Xu, Y.; et al. Dietary magnesium intake and the risk of cardiovascular disease, type 2 diabetes, and all-cause mortality: A dose—Response meta- analysis of prospective cohort studies. BMC Med. 2016. [Google Scholar] [CrossRef] [PubMed]
- Del Gobbo, L.C.; Imamura, F.; Wu, J.H.Y.; Otto, M.C.D.O.; Chiuve, S.E. Circulating and dietary magnesium and risk of cardiovascular disease: A systematic review and meta-analysis of prospective studies. Am. J. Clin. Nutr. 2013, 160–173. [Google Scholar] [CrossRef] [PubMed]
- Qu, X.; Jin, F.; Hao, Y.; Li, H.; Tang, T.; Wang, H.; Yan, W.; Dai, K. Magnesium and the Risk of Cardiovascular Events: A Meta-Analysis of Prospective Cohort Studies. PLoS ONE 2013, 8. [Google Scholar] [CrossRef] [PubMed]
- Chiuve, S.E.; Sun, Q.; Curhan, G.C.; Taylor, E.N.; Spiegelman, D.; Willett, W.C.; Manson, J.E.; Rexrode, K.M.; Albert, C.M. Dietary and plasma magnesium and risk of coronary heart disease among women. J. Am. Heart Assoc. 2013, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Meyer, T.E.; Verwoert, G.C.; Hwang, S.J.; Glazer, N.L.; Smith, A.V.; van Rooij, F.J.A.; Ehret, G.B.; Boerwinkle, E.; Felix, J.F.; Leak, T.S.; et al. Genome-wide association studies of serum magnesium, potassium, and sodium concentrations identify six loci influencing serum magnesium levels. PLoS Genet. 2010, 6. [Google Scholar] [CrossRef] [PubMed]
- Saris, N.-E.L.E.A. Magnesium An update on phsyiological, clinical and analytical aspects. Clin. Chim. Acta 2000, 294, 1–26. [Google Scholar] [CrossRef]
- Jahnen-Dechent, W.; Ketteler, M. Magnesium basics. CKJ Clin. Kidney J. 2012, 5. [Google Scholar] [CrossRef] [PubMed]
- US Department of Agriculture, Agricultural Research Service, Nutrient Data Laboratory. USDA National Nutrient Database for Standard Reference, Release 28 (Revised). Available online: www.ars.usda.gov/ba/bhnrc/ndl (accessed on 24 January 2018).
- Shechter, M. Magnesium and cardiovascular system. Magnes. Res. 2010, 23, 60–72. [Google Scholar] [CrossRef] [PubMed]
- Gommers, L.M.M.; Hoenderop, J.G.J.; Bindels, R.J.M.; de Baaij, J.H.F. Hypomagnesemia in Type 2 Diabetes: A Vicious Circle? Diabetes 2016, 65, 3–13. [Google Scholar] [CrossRef] [PubMed]
- Fang, X.; Han, H.; Li, M.; Liang, C.; Fan, Z.; Aaseth, J.; He, J.; Montgomery, S.; Cao, Y. Dose-Response Relationship between Dietary Magnesium Intake and Risk of Type 2 Diabetes Mellitus: A Systematic Review and Meta-Regression Analysis of Prospective Cohort Studies. Nutrients 2016, 8, 739. [Google Scholar] [CrossRef] [PubMed]
- Dong, J.-Y.; Xun, P.; He, K.; Qin, L.-Q. Magnesium Intake and Risk of Type 2 Diabetes: Meta-analysis of prospective cohort studies. Diabetes Care 2011, 34, 2116–2122. [Google Scholar] [CrossRef] [PubMed]
- Schulze, M.B.; Schulz, M.; Heidemann, C.; Schienkiewitz, A.; Hoffmann, K.; Boeing, H. Fiber and magnesium intake and incidence of type 2 diabetes: A prospective study and meta-analysis. Arch. Intern. Med. 2007, 167, 956–965. [Google Scholar] [CrossRef] [PubMed]
- Hruby, A.; Guasch-Ferré, M.; Bhupathiraju, S.N.; Manson, J.E.; Willett, W.C.; McKeown, N.M.; Hu, F.B. Magnesium Intake, Quality of Carbohydrates, and Risk of Type 2 Diabetes: Results from Three U.S. Cohorts. Diabetes Care 2017, 40, 1695–1702. [Google Scholar] [CrossRef] [PubMed]
- Simental-Mendía, L.E.; Sahebkar, A.; Rodríguez-Morán, M.; Guerrero-Romero, F. A systematic review and meta-analysis of randomized controlled trials on the effects of magnesium supplementation on insulin sensitivity and glucose control. Pharmacol. Res. 2016, 111, 272–282. [Google Scholar] [CrossRef] [PubMed]
- Guerrero-Romero, F.; Rodríguez-Morán, M. Magnesium improves the beta-cell function to compensate variation of insulin sensitivity: Double-blind, randomized clinical trial. Eur. J. Clin. Investig. 2011, 41, 405–410. [Google Scholar] [CrossRef] [PubMed]
- Song, Y.; He, K.; Levitan, E.B.; Manson, J.E.; Liu, S. Effects of oral magnesium supplementation on glycaemic control in Type 2 diabetes: A meta-analysis of randomized double-blind controlled trials. Diabetes Med. 2006, 23, 1050–1056. [Google Scholar] [CrossRef] [PubMed]
- Guerrero-Romero, F.; Tamez-Perez, H.E.; González-González, G.; Salinas-Martínez, A.M.; Montes-Villarreal, J.; Treviño-Ortiz, J.H.; Rodríguez-Morán, M. Oral magnesium supplementation improves insulin sensitivity in non-diabetic subjects with insulin resistance. A double-blind placebo-controlled randomized trial. Diabetes Metab. 2004, 30, 253–258. [Google Scholar] [CrossRef]
- He, K.; Liu, K.; Daviglus, M.L.; Morris, S.J.; Loria, C.M.; Van Horn, L.; Jacobs, D.R.; Savage, P.J. Magnesium intake and incidence of metabolic syndrome among young adults. Circulation 2006, 113, 1675–1682. [Google Scholar] [CrossRef] [PubMed]
- Sarrafzadegan, N.; Khosravi-Boroujeni, H.; Lotfizadeh, M.; Pourmogaddas, A.; Salehi-Abargouei, A. Magnesium status and the metabolic syndrome: A systematic review and meta-analysis. Nutrition 2016, 32, 409–417. [Google Scholar] [CrossRef] [PubMed]
- McKeown, N.M.; Jacques, P.F.; Zhang, X.L.; Juan, W.; Sahyoun, N.R. Dietary magnesium intake is related to metabolic syndrome in older Americans. Eur. J. Nutr. 2008, 47, 210–216. [Google Scholar] [CrossRef] [PubMed]
- Ju, S.-Y.; Choi, W.-S.; Ock, S.-M.; Kim, C.-M.; Kim, D.-H. Dietary magnesium intake and metabolic syndrome in the adult population: Dose-response meta-analysis and meta-regression. Nutrients 2014, 6, 6005–6019. [Google Scholar] [CrossRef] [PubMed]
- Dibaba, D.T.; Xun, P.; Fly, A.D.; Yokota, K.; He, K. Dietary magnesium intake and risk of metabolic syndrome: A meta-analysis. Diabet. Med. 2014, 31, 1301–1309. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, H.; Odelola, O.A.; Rangaswami, J.; Amanullah, A. A Review of Nutritional Factors in Hypertension Management. Int. J. Hypertens. 2013, 2013, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Touyz, R.M.; Milne, F.J.; Reinach, S.G. Intracellular Mg2+, Ca2+, Na2+ and K+ in platelets and erythrocytes of essential hypertension patients: Relation to blood pressure. Clin. Exp. Hypertens. A 1992, 14, 1189–1209. [Google Scholar] [CrossRef] [PubMed]
- Dibaba, D.T.; Xun, P.; Song, Y.; Rosanoff, A.; Shechter, M.; He, K. The effect of magnesium supplementation on blood pressure in individuals with insulin resistance, prediabetes, or noncommunicable chronic diseases: A meta-analysis of randomized controlled trials. Am. J. Clin. Nutr. 2017, 106, 921–929. [Google Scholar] [CrossRef] [PubMed]
- Jee, S.H.; Miller, E.R.; Guallar, E.; Singh, V.K.; Appel, L.J.; Klag, M.J. The effect of magnesium supplementation on blood pressure: A meta-analysis of randomized clinical trials. Am. J. Hypertens. 2002, 15, 691–696. [Google Scholar] [CrossRef]
- Kass, L.; Weekes, J.; Carpenter, L. Effect of magnesium supplementation on blood pressure: A meta-analysis. Eur. J. Clin. Nutr. 2012, 66, 411–418. [Google Scholar] [CrossRef] [PubMed]
- Kolte, D.; Vijayaraghavan, K.; Khera, S.; Sica, D.A.; Frishman, W.H. Role of magnesium in cardiovascular diseases. Cardiol. Rev. 2014, 22, 182–192. [Google Scholar] [CrossRef] [PubMed]
- Song, Y.; Li, T.Y.; van Dam, R.M.; Manson, J.E.; Hu, F.B. Magnesium intake and plasma concentrations of markers of systemic inflammation and endothelial dysfunction in women. Am. J. Clin. Nutr. 2007, 85, 1068–1074. [Google Scholar] [PubMed]
- Chacko, S.A.; Song, Y.; Nathan, L.; Tinker, L.; de Boer, I.H.; Tylavsky, F.; Wallace, R.; Liu, S. Relations of dietary magnesium intake to biomarkers of inflammation and endothelial dysfunction in an ethnically diverse cohort of postmenopausal women. Diabetes Care 2010, 33, 304–310. [Google Scholar] [CrossRef] [PubMed]
- Shechter, M.; Sharir, M.; Labrador, M.J.; Forrester, J.; Silver, B.; Bairey Merz, C.N. Oral magnesium therapy improves endothelial function in patients with coronary artery disease. Circulation 2000, 102, 2353–2358. [Google Scholar] [CrossRef] [PubMed]
- Carr, M.C.; Brunzell, J.D. Abdominal obesity and dyslipidemia in the metabolic syndrome: Importance of type 2 diabetes and familial combined hyperlipidemia in coronary artery disease risk. J. Clin. Endocrinol. Metab. 2004, 89, 2601–2607. [Google Scholar] [CrossRef] [PubMed]
- Singh, R.B.; Rastogi, S.S.; Sharma, V.K.; Saharia, R.B.; Kulshretha, S.K. Can dietary magnesium modulate lipoprotein metabolism? Magnes. Trace Elem. 1990, 9, 255–264. [Google Scholar] [PubMed]
- Geiger, H.; Wanner, C. Magnesium in disease. Clin. Kidney J. 2012, 5, i25–i38. [Google Scholar] [CrossRef] [PubMed]
- Guerrero-Romero, F.; Rodríguez-Morán, M. Low serum magnesium levels and metabolic syndrome. Acta Diabetol. 2002, 39, 209–213. [Google Scholar] [CrossRef] [PubMed]
- Mirmiran, P.; Shab-Bidar, S.; Hosseini-Esfahani, F.; Asghari, G.; Hosseinpour-Niazi, S.; Azizi, F. Magnesium intake and prevalence of metabolic syndrome in adults: Tehran Lipid and Glucose Study. Public Health Nutr. 2012, 15, 693–701. [Google Scholar] [CrossRef] [PubMed]
- Bain, L.K.M.; Myint, P.K.; Jennings, A.; Lentjes, M.A.H.; Luben, R.N.; Khaw, K.-T.; Wareham, N.J.; Welch, A.A. The relationship between dietary magnesium intake, stroke and its major risk factors, blood pressure and cholesterol, in the EPIC-Norfolk cohort. Int. J. Cardiol. 2015, 196, 108–114. [Google Scholar] [CrossRef] [PubMed]
- Simental-Mendía, L.E.; Simental-Mendía, M.; Sahebkar, A.; Rodríguez-Morán, M.; Guerrero-Romero, F. Effect of magnesium supplementation on lipid profile: A systematic review and meta-analysis of randomized controlled trials. Eur. J. Clin. Pharmacol. 2017, 73, 525–536. [Google Scholar] [CrossRef] [PubMed]
- Larsson, S.C.; Orsini, N.; Wolk, A. Dietary magnesium intake and risk of stroke: A meta-analysis of prospective studies. Am. J. Clin. Nutr. 2012, 95, 362–366. [Google Scholar] [CrossRef] [PubMed]
- Nie, Z.-L.; Wang, Z.-M.; Zhou, B.; Tang, Z.-P.; Wang, S.-K. Magnesium intake and incidence of stroke: Meta-analysis of cohort studies. Nutr. Metab. Cardiovasc. Dis. 2013, 23, 169–176. [Google Scholar] [CrossRef] [PubMed]
- Adebamowo, S.N.; Spiegelman, D.; Willett, W.C.; Rexrode, K.M. Association between intakes of magnesium, potassium, and calcium and risk of stroke: 2 Cohorts of US women and updated meta-analyses. Am. J. Clin. Nutr. 2015, 101, 1269–1277. [Google Scholar] [CrossRef] [PubMed]
- Liao, F.; Folsom, A.R.; Brancati, F.L. Is low magnesium concentration a risk factor for coronary heart disease? The Atherosclerosis Risk in Communities (ARIC) Study. Am. Heart J. 1998, 136, 480–490. [Google Scholar] [CrossRef]
- Abbott, R.D.; Ando, F.; Masaki, K.H.; Tung, K.H.; Rodriguez, B.L.; Petrovitch, H.; Yano, K.; Curb, J.D. Dietary magnesium intake and the future risk of coronary heart disease (The Honolulu heart program). Am. J. Cardiol. 2003, 92, 665–669. [Google Scholar] [CrossRef]
- Song, Y.; Manson, J.E.; Cook, N.R.; Albert, C.M.; Buring, J.E.; Liu, S. Dietary magnesium intake and risk of cardiovascular disease among women. Am. J. Cardiol. 2005, 96, 1135–1141. [Google Scholar] [CrossRef] [PubMed]
- Dai, Q.; Shu, X.-O.; Deng, X.; Xiang, Y.-B.; Li, H.; Yang, G.; Shrubsole, M.J.; Ji, B.; Cai, H.; Chow, W.-H.; et al. Modifying effect of calcium/magnesium intake ratio and mortality: A population-based cohort study. Br. Med. J. 2013, 3, e002111. [Google Scholar] [CrossRef] [PubMed]
- Kokubo, Y.; Saito, I.; Iso, H.; Yamagishi, K.; Yatsuya, H.; Ishihara, J.; Maruyama, K.; Inoue, M.; Sawada, N.; Tsugane, S. Dietary magnesium intake and risk of incident coronary heart disease in men: A prospective cohort study. Clin. Nutr. 2017, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Taveira, T.H.; Ouellette, D.; Gulum, A.; Choudhary, G.; Eaton, C.B.; Liu, S.; Wu, W.C. Relation of magnesium intake with cardiac function and heart failure hospitalizations in black adults. Circ. Heart Fail. 2016, 9. [Google Scholar] [CrossRef]
- Zhang, W.; Iso, H.; Ohira, T.; Date, C.; Tamakoshi, A. Associations of dietary magnesium intake with mortality from cardiovascular disease: The JACC study. Atherosclerosis 2012, 221, 587–595. [Google Scholar] [CrossRef] [PubMed]
- Misialek, J.R.; Lopez, F.L.; Lutsey, P.L.; Huxley, R.R.; Peacock, J.M.; Chen, L.Y.; Soliman, E.Z.; Agarwal, S.K.; Alonso, A. Serum and Dietary Magnesium and Incidence of Atrial Fibrillation in Whites and in African Americans—Atherosclerosis Risk in Communities (ARIC) Study. Circ. J. 2013, 77, 323–329. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, F.H.; Milne, D.B.; Klevay, L.M.; Gallagher, S.; Johnson, L.A. Dietary magnesium deficiency induces heart rhythm changes, impairs glucose tolerance, and decreases serum cholesterol in post menopausal women. J. Am. Coll. Nutr. 2007, 26, 121–132. [Google Scholar] [CrossRef] [PubMed]
- Deng, X.; Song, Y.; Manson, J.E.; Signorello, L.B.; Zhang, S.M.; Shrubsole, M.J.; Ness, R.M.; Seidner, D.L.; Dai, Q. Magnesium, vitamin D status and mortality: Results from US National Health and Nutrition Examination Survey (NHANES) 2001 to 2006 and NHANES III. BMC Med. 2013, 11, 187. [Google Scholar] [CrossRef] [PubMed]
- Levitan, E.B.; Shikany, J.M.; Ahmed, A.; Snetselaar, L.G.; Martin, L.W.; Curb, J.D.; Lewis, C.E. Calcium, magnesium and potassium intake and mortality in women with heart failure: The Women’s Health Initiative. Br. J. Nutr. 2013, 110, 179–185. [Google Scholar] [CrossRef] [PubMed]
- Chiuve, S.E.; Korngold, E.C.; Januzzi, J.L., Jr.; Gantzer, M.L.; Albert, C.M. Plasma and dietary magnesium and risk of sudden cardiac death. Am. J. Clin. Nutr. 2011, 253–260. [Google Scholar] [CrossRef] [PubMed]
- Guasch-Ferré, M.; Bulló, M.; Estruch, R.; Corella, D.; Martínez-González, M.A.; Ros, E.; Covas, M.; Arós, F.; Gómez-Gracia, E.; Fiol, M.; et al. Dietary magnesium intake is inversely associated with mortality in adults at high cardiovascular disease risk. J. Nutr. 2014, 144, 55–60. [Google Scholar] [CrossRef] [PubMed]
- Xu, T.; Sun, Y.; Xu, T.; Zhang, Y. Magnesium intake and cardiovascular disease mortality: A meta-analysis of prospective cohort studies. Int. J. Cardiol. 2013, 167, 3044–3047. [Google Scholar] [CrossRef] [PubMed]
- Fang, X.; Liang, C.; Li, M.; Montgomery, S.; Fall, K.; Aaseth, J.; Cao, Y. Dose-response relationship between dietary magnesium intake and cardiovascular mortality: A systematic review and dose-based meta-regression analysis of prospective studies. J. Trace Elem. Med. Biol. 2016, 38, 64–73. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Del Gobbo, L.C.; Hruby, A.; Rosanoff, A.; He, K.; Dai, Q.; Costello, R.B.; Zhang, W.; Song, Y. The Circulating Concentration and 24-h Urine Excretion of Magnesium Dose- and Time-Dependently Respond to Oral Magnesium Supplementation in a Meta-Analysis of Randomized Controlled Trials. J. Nutr. 2016, 146, 595–602. [Google Scholar] [CrossRef] [PubMed]
- Joosten, M.M.; Gansevoort, R.T.; Mukamal, K.J.; Van Der Harst, P.; Geleijnse, J.M.; Feskens, E.J.M. Urinary and plasma magnesium and risk of ischemic heart disease. Am. J. Clin. Nutr. 2013, 97, 1299–1306. [Google Scholar] [CrossRef] [PubMed]
- Ohira, T.; Peacock, J.M.; Iso, H.; Chambless, L.E.; Rosamond, W.D.; Folsom, A.R. Serum and dietary magnesium and risk of ischemic stroke. Am. J. Epidemiol. 2009, 169, 1437–1444. [Google Scholar] [CrossRef] [PubMed]
- Akarolo-Anthony, S.N.; Jiménez, M.C.; Chiuve, S.E.; Spiegelman, D.; Willett, W.C.; Rexrode, K.M. Plasma magnesium and risk of ischemic stroke among women. Stroke 2014, 45, 2881–2886. [Google Scholar] [CrossRef]
- Khan, A.M.; Lubitz, S.A.; Sullivan, L.M.; Sun, J.X.; Levy, D.; Vasan, R.S.; Magnani, J.W.; Ellinor, P.T.; Benjamin, E.J.; Wang, T.J. Low serum magnesium and the development of atrial fibrillation in the community: The framingham heart study. Circulation 2013, 127, 33–38. [Google Scholar] [CrossRef] [PubMed]
- Reffelmann, T.; Dörr, M.; Ittermann, T.; Schwahn, C.; Völzke, H.; Ruppert, J.; Robinson, D.; Felix, S.B. Low serum magnesium concentrations predict increase in left ventricular mass over 5 years independently of common cardiovascular risk factors. Atherosclerosis 2010, 213, 563–569. [Google Scholar] [CrossRef] [PubMed]
- Lutsey, P.L.; Alonso, A.; Michos, E.D.; Loehr, L.R.; Astor, B.C.; Coresh, J.; Folsom, A.R. Serum magnesium, phosphorus, and calcium are associated with risk of incident heart failure: The Atherosclerosis Risk in Communities (ARIC). Am. J. Clin. Nutr. 2014, 756–764. [Google Scholar] [CrossRef] [PubMed]
- Kunutsor, S.K.; Khan, H.; Laukkanen, J.A. Serum magnesium and risk of new onset heart failure in men: The Kuopio Ischemic Heart Disease Study. Eur. J. Epidemiol. 2016, 31, 1035–1043. [Google Scholar] [CrossRef] [PubMed]
- Ma, J.; Folsom, A.R.; Melnick, S.L.; Eckfeldt, J.H.; Sharrett, A.R.; Nabulsi, A.A.; Hutchinson, R.G.; Metcalf, P.A. Associations of serum and dietary magnesium with cardiovascular disease, hypertension, diabetes, insulin, and carotid arterial wall thickness: The aric study. J. Clin. Epidemiol. 1995, 48. [Google Scholar] [CrossRef]
- Hashimoto, T.; Hara, A.; Ohkubo, T.; Kikuya, M.; Shintani, Y.; Metoki, H.; Inoue, R.; Asayama, K.; Kanno, A.; Nakashita, M.; et al. Serum magnesium, ambulatory blood pressure, and carotid artery alteration: The ohasama study. Am. J. Hypertens. 2010, 23, 1292–1298. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.Y.; Hyun, Y.Y.; Lee, K.B.; Kim, H. Low serum magnesium is associated with coronary artery calcification in a Korean population at low risk for cardiovascular disease. Nutr. Metab. Cardiovasc. Dis. 2015, 25, 1056–1061. [Google Scholar] [CrossRef] [PubMed]
- Posadas-Sánchez, R.; Posadas-Romero, C.; Cardoso-Saldaña, G.; Vargas-Alarcón, G.; Villarreal-Molina, M.T.; Pérez-Hernández, N.; Rodríguez-Pérez, J.M.; Medina-Urrutia, A.; Jorge-Galarza, E.; Juárez-Rojas, J.G.; et al. Serum magnesium is inversely associated with coronary artery calcification in the Genetics of Atherosclerotic Disease (GEA) study. Nutr. J. 2015, 15, 22. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Xia, J.; Del Gobbo, L.C.; Hruby, A.; Dai, Q.; Song, Y. Serum magnesium concentrations and all-cause, cardiovascular, and cancer mortality among U.S. adults: Results from the NHANES I Epidemiologic Follow-up Study. Clin. Nutr. 2017. [Google Scholar] [CrossRef] [PubMed]
- Kieboom, B.C.T.; Niemeijer, M.N.; Leening, M.J.G.; van den Berg, M.E.; Franco, O.H.; Deckers, J.W.; Hofman, A.; Zietse, R.; Stricker, B.H.; Hoorn, E.J. Serum Magnesium and the Risk of Death From Coronary Heart Disease and Sudden Cardiac Death. J. Am. Heart Assoc. 2016, 5, e002707. [Google Scholar] [CrossRef] [PubMed]
- Angkananard, T.; Anothaisintawee, T.; Eursiriwan, S.; Gorelik, O.; McEvoy, M.; Attia, J.; Thakkinstian, A. The association of serum magnesium and mortality outcomes in heart failure patients. Medicine (Baltimore) 2016, 95, e5406. [Google Scholar] [CrossRef] [PubMed]
- Barbagallo, M.; Dominguez, L.J.; Galioto, A.; Ferlisi, A.; Cani, C.; Malfa, L.; Pineo, A.; Busardo’, A.; Paolisso, G. Role of magnesium in insulin action, diabetes and cardio-metabolic syndrome X. Mol. Asp. Med. 2003, 24, 39–52. [Google Scholar] [CrossRef]
- Mazur, A.; Maier, J.A.M.; Rock, E.; Gueux, E.; Nowacki, W.; Rayssiguier, Y. Magnesium and the inflammatory response: Potential physiopathological implications. Arch. Biochem. Biophys. 2007, 458, 48–56. [Google Scholar] [CrossRef] [PubMed]
- Blache, D.; Devaux, S.; Joubert, O.; Loreau, N.; Schneider, M.; Durand, P.; Prost, M.; Gaume, V.; Adrian, M.; Laurant, P.; et al. Long-term moderate magnesium-deficient diet shows relationships between blood pressure, inflammation and oxidant stress defense in aging rats. Free Radic. Biol. Med. 2006, 41, 277–284. [Google Scholar] [CrossRef] [PubMed]
- Freedman, A.M.; Atrakchi, A.H.; Cassidy, M.M.; Weglicki, W.B. Magnesium deficiency-induced cardiomyopathy: Protection by vitamin E. Biochem. Biophys. Res. Commun. 1990, 170, 1102–1106. [Google Scholar] [CrossRef]
- Freedman, A.M.; Cassidy, M.M.; Weglicki, W.B. Magnesium-deficient myocardium demonstrates an increased susceptibility to an in vivo oxidative stress. Magnes. Res. 1991, 4, 185–189. [Google Scholar] [PubMed]
- King, D.E. Inflammation and elevation of C-reactive protein: Does magnesium play a key role? Magnes. Res. 2009, 22, 57–59. [Google Scholar] [PubMed]
- Olatunji, L.A.; Soladoye, A.O. Increased magnesium intake prevents hyperlipidemia and insulin resistance and reduces lipid peroxidation in fructose-fed rats. Pathophysiology 2007, 14, 11–15. [Google Scholar] [CrossRef] [PubMed]
- Garcia, L.A.; Dejong, S.C.; Martin, S.M.; Smith, R.S.; Buettner, G.R.; Kerber, R.E. Magnesium reduces free radicals in an in vivo coronary occlusion- reperfusion model. J. Am. Coll. Cardiol. 1998, 32, 536–539. [Google Scholar] [CrossRef]
- Rock, E.; Astier, C.; Lab, C.; Malpuech, C.; Nowacki, W.; Gueux, E.; Mazur, A.; Rayssiguier, Y. Magnesium deficiency in rats induces a rise in plasma nitric oxide. Magnes. Res. 1995, 8, 237–242. [Google Scholar] [PubMed]
- Dickens, B.F.; Weglicki, W.B.; Li, Y.S.; Mak, I.T. Magnesium deficiency in vitro enhances free radical-induced intracellular oxidation and cytotoxicity in endothelial cells. FEBS Lett. 1992, 311, 187–191. [Google Scholar] [CrossRef]
- Wiles, M.E.; Wagner, T.L.; Weglicki, W.B. Effect of acute magnesium deficiency (MgD) on aortic endothelial cell (EC) oxidant production. Life Sci. 1997, 60, 221–236. [Google Scholar] [CrossRef]
- Kramer, J.H.; Mišík, V.; Weglicki, W.B. Magnesium-deficiency potentiates free radical production associated with postischemic injury to rat hearts: Vitamin E affords protection. Free Radic. Biol. Med. 1994, 16, 713–723. [Google Scholar] [CrossRef]
- Kramer, J.H.; Mak, I.T.; Phillips, T.M.; Weglicki, W.B. Dietary magnesium intake influences circulating pro-inflammatory neuropeptide levels and loss of myocardial tolerance to postischemic stress. Exp. Biol. Med. (Maywood) 2003, 228, 665–673. [Google Scholar] [CrossRef] [PubMed]
- Bernardini, D.; Nasulewic, A.; Mazur, A.; Maier, J.A.M. Magnesium and microvascular endothelial cells: A role in inflammation and angiogenesis. Front. Biosci. 2005, 10, 1177–1182. [Google Scholar] [CrossRef] [PubMed]
- Rayssiguier, Y.; Gueux, E.; Bussière, L.; Durlach, J.; Mazur, A. Dietary magnesium affects susceptibility of lipoproteins and tissues to peroxidation in rats. J. Am. Coll. Nutr. 1993, 12, 133–137. [Google Scholar] [CrossRef] [PubMed]
- Mylonas, C.; Kouretas, D. Lipid peroxidation and tissue damage. In Vivo 1999, 13, 295–309. [Google Scholar] [PubMed]
- Morrill, G.A.; Gupta, R.K.; Kostellow, A.B.; Ma, G.Y.; Zhang, A.; Altura, B.T.; Altura, B.M. Mg2+ modulates membrane lipids in vascular smooth muscle: A link to atherogenesis. FEBS Lett. 1997, 408, 191–194. [Google Scholar] [CrossRef]
- Bussière, L.; Mazur, A.; Gueux, E.; Nowacki, W.; Rayssiguier, Y. Triglyceride-rich lipoproteins from magnesium-deficient rats are more susceptible to oxidation by cells and promote proliferation of cultured vascular smooth muscle cells. Magnes. Res. 1995, 8, 151–157. [Google Scholar] [PubMed]
- Gueux, E.; Azais-Braesco, V.; Bussière, L.; Grolier, P.; Mazur, A.; Rayssiguier, Y. Effect of magnesium deficiency on triacylglycerol-rich lipoprotein and tissue susceptibility to peroxidation in relation to vitamin E content. Br. J. Nutr. 1995, 74, 849–856. [Google Scholar] [PubMed]
- Altura, B.M.; Gebrewold, A.; Altura, B.T.; Brautbar, N. Magnesium depletion impairs myocardial carbohydrate and lipid metabolism and cardiac bioenergetics and raises myocardial calcium content in-vivo: Relationship to etiology of cardiac diseases. Biochem. Mol. Biol. Int. 1996, 40, 1183–1190. [Google Scholar] [CrossRef] [PubMed]
- Takaya, J.; Higashino, H.; Kobayashi, Y. Intracellular magnesium and insulin resistance. Magnes. Res. 2004, 17, 126–136. [Google Scholar] [PubMed]
- Pham, P.-C.T.; Pham, P.-M.T.; Pham, S.V.; Miller, J.M.; Pham, P.-T.T. Hypomagnesemia in Patients with Type 2 Diabetes. Clin. J. Am. Soc. Nephrol. 2007, 2, 366–373. [Google Scholar] [CrossRef] [PubMed]
- Sontia, B.; Touyz, R.M. Magnesium transport in hypertension. Pathophysiol. Off. J. Int. Soc. Pathophysiol. 2007, 14, 205–211. [Google Scholar] [CrossRef] [PubMed]
- Barbagallo, M.; Dominguez, L.J.; Resnick, L.M. Magnesium metabolism in hypertension and type 2 diabetes mellitus. Am. J. Ther. 2007, 14, 375–385. [Google Scholar] [CrossRef] [PubMed]
- Maier, J.A.M.; Bernardini, D.; Rayssiguier, Y.; Mazur, A. High concentrations of magnesium modulate vascular endothelial cell behaviour in vitro. Biochim. Biophys. Acta Mol. Basis Dis. 2004, 1689, 6–12. [Google Scholar] [CrossRef] [PubMed]
- Laurant, P.; Kantelip, J.P.; Berthelot, A. Dietary magnesium supplementation modifies blood pressure and cardiovascular function in mineralocorticoid-salt hypertensive rats but not in normotensive rats. J. Nutr. 1995, 125, 830–841. [Google Scholar] [PubMed]
- Touyz, R.M.; Milne, F.J. Magnesium supplementation attenuates, but does not prevent, development of hypertension in spontaneously hypertensive rats. Am. J. Hypertens. 1999, 12, 757–765. [Google Scholar] [CrossRef]
- Agus, M.S.D.; Agus, Z.S. Cardiovascular actions of magnesium. Crit. Care Clin. 2001, 17, 175–186. [Google Scholar] [CrossRef]
- Mubagwa, K.; Gwanyanya, A.; Zakharov, S.; Macianskiene, R. Regulation of cation channels in cardiac and smooth muscle cells by intracellular magnesium. Arch. Biochem. Biophys. 2007, 458, 73–89. [Google Scholar] [CrossRef] [PubMed]
- Johnson, C.J.; Peterson, D.R.; Smith, E.K. Myocardial tissue concentrations of magnesium and potassium in men dying suddenly from ischemic heart disease. Am. J. Clin. Nutr. 1979, 32, 967–970. [Google Scholar] [CrossRef] [PubMed]
- Paravicini, T.M.; Chubanov, V.; Gudermann, T. TRPM7: A unique channel involved in magnesium homeostasis. Int. J. Biochem. Cell Biol. 2012, 44, 1381–1384. [Google Scholar] [CrossRef] [PubMed]
- Touyz, R.M. Transient receptor potential melastatin 6 and 7 channels, magnesium transport, and vascular biology: Implications in hypertension. Am. J. Physiol. Heart Circ. Physiol. 2008, 294, H1103–H1118. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Ramírez, M.; Rodríguez-Morán, M.; Reyes-Romero, M.A.; Guerrero-Romero, F. Effect of oral magnesium supplementation on the transcription of TRPM6, TRPM7, and SLC41A1 in individuals newly diagnosed of pre-hypertension. A randomized, double-blind, placebo-controlled trial. Magnes. Res. 2017, 30, 80–87. [Google Scholar] [PubMed]
- Kappeler, D.; Heimbeck, I.; Herpich, C.; Naue, N.; Höfler, J.; Timmer, W.; Michalke, B. Higher bioavailability of magnesium citrate as compared to magnesium oxide shown by evaluation of urinary excretion and serum levels after single-dose administration in a randomized cross-over study. BMC Nutr. 2017, 3, 7. [Google Scholar] [CrossRef]
- Schuchardt, J.P.; Hahn, A. Intestinal Absorption and Factors Influencing Bioavailability of Magnesium—An Update. Curr. Nutr. Food Sci. 2017, 13, 260–278. [Google Scholar] [CrossRef] [PubMed]

| Food from Plant Origin | Food from Animal Origin | ||
|---|---|---|---|
| Nuts and Seeds | Mg mg/100 g | Dairy and Eggs | Mg mg/100 g |
| Pumpkin seeds, dried | 592 | Parmesan cheese | 44 |
| Flaxseed | 392 | Feta cheese | 19 |
| Sesame seeds, roasted | 356 | Whole-fat milk | 13 |
| Almonds, raw | 270 | Plain whole-fat yogurt | 12 |
| Cashew nuts, roasted | 260 | Whole fresh egg | 12 |
| Walnuts | 158 | ||
| Pistachio nuts, roasted | 109 | ||
| Legumes | Fish and seafood | ||
| Peanuts, roasted | 178 | Cod, cooked | 133 |
| Soybeans, cooked | 86 | Salmon, cooked | 122 |
| Chickpeas, cooked | 48 | Canned anchovies | 69 |
| Kidney beans, cooked | 45 | Shrimps, cooked | 37 |
| Lentils, cooked | 36 | ||
| Vegetables and fruits | Meat and meat products | ||
| Sun-dried tomatoes | 194 | Chicken breast, cooked | 34 |
| Spinach, cooked | 87 | Turkey, cooked | 32 |
| Kale, cooked | 57 | Veal, cooked | 34 |
| Dates | 54 | Rabbit, cooked | 21 |
| Fresh parsley | 50 | ||
| Baked potatoes with skin | 43 | ||
| Whole grains | |||
| Buckwheat flour | 251 | ||
| Amaranth grain | 248 | ||
| Quinoa grain | 197 | ||
| Oats | 177 | ||
| Spelt | 136 | ||
| Barley | 133 | ||
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rosique-Esteban, N.; Guasch-Ferré, M.; Hernández-Alonso, P.; Salas-Salvadó, J. Dietary Magnesium and Cardiovascular Disease: A Review with Emphasis in Epidemiological Studies. Nutrients 2018, 10, 168. https://doi.org/10.3390/nu10020168
Rosique-Esteban N, Guasch-Ferré M, Hernández-Alonso P, Salas-Salvadó J. Dietary Magnesium and Cardiovascular Disease: A Review with Emphasis in Epidemiological Studies. Nutrients. 2018; 10(2):168. https://doi.org/10.3390/nu10020168
Chicago/Turabian StyleRosique-Esteban, Nuria, Marta Guasch-Ferré, Pablo Hernández-Alonso, and Jordi Salas-Salvadó. 2018. "Dietary Magnesium and Cardiovascular Disease: A Review with Emphasis in Epidemiological Studies" Nutrients 10, no. 2: 168. https://doi.org/10.3390/nu10020168
APA StyleRosique-Esteban, N., Guasch-Ferré, M., Hernández-Alonso, P., & Salas-Salvadó, J. (2018). Dietary Magnesium and Cardiovascular Disease: A Review with Emphasis in Epidemiological Studies. Nutrients, 10(2), 168. https://doi.org/10.3390/nu10020168







