Viewpoint: A Contributory Role of Shell Ginger (Alpinia zerumbet) for Human Longevity in Okinawa, Japan?
Abstract
:1. Introduction
2. Search Strategy
2.1. Search and Identification Terms
2.2. Data Analysis
3. Alpinia Species
3.1. The Alpinia Genus and the Zingiberaceae (Ginger) Family
3.2. Alpinia Zerumbet as A Plant
4. Phytochemicals of Alpinia Zerumbet
4.1. Kavalactones
4.2. Non-Kavalactones
4.3. Antioxidative Phytochemicals
5. Longevity-Specific Bioactivities of Alpinia Zerumbet
5.1. Animal Longevity
5.2. Anti-Obesity
5.2.1. Anti-Lipocytes
5.2.2. Anti-Pancreatic Lipase
5.2.3. Anti-Dyslipidaemia
5.2.4. Anti-Atherosclerosis
5.2.5. Anti-Diabetes
5.2.6. Anti-Hypertension
5.3. Anti-Tumor
6. Okinawa Life-Longevity
7. Genetics and Traditional Lifestyle in Okinawa
7.1. Genetic Background
7.2. Physical and Social Activities
7.3. Caloric and Dietary Restriction
7.4. Westernized Lifestyle
8. Traditional Okinawa Cuisine
9. Challenges and Perspectives
9.1. Current Issues
9.2. Future Research
10. New Encouraging Conceptual Approaches
11. Conclusions
Author contributions
Conflicts of Interest
References
- Life Expectancy Increased by 5 Years Since 2000, but Health Inequalities Persist. Available online: http://www.who.int/mediacentre/news/releases/2016/health-inequalities-persist/en/ (accessed on 19 November 2017).
- Global Health Observatory Country Views. Available online: http://apps.who.int/gho/data/node.country (accessed on 19 November 2017).
- Shao, A.; Drewnowsk, A.; Willcox, D.C.; Krämer, L.; Lausted, C.; Eggersdorfer, M. Optimal nutrition and ever-changing dietary landscape: A conference report. Eur. J. Nutr. 2017, 56, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Miyagi, S.; Iwama, N.; Kawabata, T.; Hasegawa, K. Longevity and diet in Okinawa, Japan: The past, present and future. Asia-Pac. J. Public Health 2003, 15 (Suppl. 1), S3–S9. [Google Scholar] [CrossRef] [PubMed]
- Hokama, T.; Binns, C. Declining longevity advantage and low birthweight in Okinawa. Asia-Pac. J. Public Health 2008, 20, 95–101. [Google Scholar] [PubMed]
- Willcox, B.J.; Willcox, D.C. Caloric restriction, CR mimetics, and healthy aging in Okinawa: Controversies and clinical implications. Curr. Opin. Clin. Nutr. Metab. Care 2014, 17, 51–58. [Google Scholar] [PubMed]
- Gavrilova, N.S.; Gavrilov, L.A. Comments on dietary restriction, Okinawa diet and longevity. Gerontology 2012, 58, 221–223. [Google Scholar] [CrossRef] [PubMed]
- Willcox, D.C.; Willcox, B.J.; Todoriki, H.; Suzuki, M. The Okinawan diet: Health implications of a low-calorie, nutrient-dense, antioxidant-rich dietary pattern low in glycemic load. J. Am. Coll. Nutr. 2009, 28 (Suppl. 4), 500S–516S. [Google Scholar] [CrossRef] [PubMed]
- Takata, H.; Suzuki, M.; Ishii, T.; Sekiguschi, S.; Iri, H. Influence of major histocompatibility complex region genes on human longevity among Okinawan-Japanese centenarians and nonagenarians. Lancet 1987, 2, 824–826. [Google Scholar] [CrossRef]
- Tu, P.T.B.; Tawata, S. Anti-obesity effects of hispidin and Alpinia zerumbet bioactives in 3T3-Li adipocytes. Molecules 2014, 19, 16656–16671. [Google Scholar] [CrossRef] [PubMed]
- Tu, P.T.B.; Chompoo, J.; Tawata, S. Hispidin and related herbal compounds from Alpinia zerumbet inhibit both PAK1-dependent melanogenesis and reactive oxygen species (ROS) production in adipocytes. Drug Discov. Ther. 2015, 9, 197–204. [Google Scholar]
- Chompoo, J.; Upadhyay, A.; Gima, S.; Fukuta, M.; Tawata, S. Antiatherogenetic properties of acetone extract of Alpinia zerumbet seeds. Molecules 2012, 17, 6237–6248. [Google Scholar] [CrossRef] [PubMed]
- Chompoo, J.; Upadhyay, A.; Fukuta, M.; Tawata, S. Effect of Alpinia zerumbet components on antioxidant and skin diseases-related enzymes. BMC Complementary. Alternative Med. 2012, 12, 106. [Google Scholar] [CrossRef] [PubMed]
- Elzaawely, A.A.; Xuan, T.D.; Koyama, H.; Tawata, S. Antioxidant activity and contents of essential oil and phenolic compounds in flowers and seeds of Alpinia zerumbet (Pers.) B.L. Burtt & R.M. Sm. Food Chem. 2007, 104, 1648–1653. [Google Scholar]
- Elzaawely, A.A.; Xuan, T.D.; Tawata, S. Changes in essential oils, kava pyrones and total phenolics of Alpinia zerumbet (Pers.) B. L. Burtt. & R.M. Sm. leaves exposed to copper sulphate. Environ. Exp. Bot. 2007, 59, 347–353. [Google Scholar]
- Elzaawely, A.A.; Xuan, T.D.; Tawata, S. Essential oils, kava pyrones and phenolic compounds from leaves and rhizomes of Alpinia zerumbet (Pers.) B. L. Burtt. & R.M. Sm. and their antioxidant activity. Food Chem. 2007, 103, 486–494. [Google Scholar]
- Tawata, S.; Fukuta, M.; Xuan, T.D.; Deba, F. Total utilization of tropical plants Leucaena leucocephala and Alpinia zerumbet. J. Pestic. Sci. 2008, 33, 40–43. [Google Scholar] [CrossRef]
- Tawata, S.; Taira, S.; Kobamoto, N.; Ishihara, M.; Toyama, S. Syntheses and biological activities of dihydro-5,6-dehydrokawain derivatives. Biosci. Biotechnol. Biochem. 1996, 60, 1643–1645. [Google Scholar] [CrossRef] [PubMed]
- Xuan, T.D.; Teschke, R. Dihydro-5,6-dehydrokavain (DDK) from Alpinia zerumbet: Its isolation, synthesis, and characterization. Molecules 2015, 20, 16306–16319. [Google Scholar] [CrossRef] [PubMed]
- Upadhyay, A.; Chompoo, J.; Taira, N.; Fukuta, M.; Tawata, S. Significant longevity-extending effects of Alpinia zerumbet leaf extracts on the life span of Caenorhabditis elegans. Biosci. Biotechnol. Biochem. 2013, 77, 217–223. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, S.; Rangan, L. Alpinia: The gold mine of future therapeutics. 3 Biotech 2013, 3, 173–185. [Google Scholar] [CrossRef] [PubMed]
- Ma, X.N.; Xie, C.L.; Miao, Z.; Yang, Q.; Yang, X.W. An overview of chemical constituents from Alpinia species in the last six decades. RSC Adv. 2017, 7, 14114–14144. [Google Scholar] [CrossRef]
- Lin, L.Y.; Peng, C.C.; Liang, Y.J.; Yeh, W.T.; Wang, H.E.; Yu, T.H.; Peng, R.Y. Alpinia zerumbet potentially elevates high-density lipoprotein cholesterol level in hamsters. J. Agric. Food Chem. 2008, 56, 4435–4443. Available online: http://pubs.acs.org/author/Lin%2C+Li-Yun (accessed on 19 November 2017). [CrossRef] [PubMed]
- Cunha, G.H.; Moraes, M.O.; Fechine, F.V.; Bezerra, F.A.F.; Silveira, E.R.; Canuto, K.M.; de Moraes, M.E.A. Vasorelaxant and antihypertensive effects of methanolic fraction of the essential oil of Alpinia zerumbet. Vasc. Pharmacol. 2013, 58, 337–345. [Google Scholar] [CrossRef] [PubMed]
- Junior, W.A.R.; Gomes, D.B.; Zanchet, B.; Schönell, A.P.; Diel, K.A.P.; Banzato, T.P.; Ruiz, A.L.T.G.; Carvalho, J.E.; Neppel, A.; Barison, A.; et al. Antiproliferative effects of pinostrobin and 5,6-dehydrokavain isolated from leaves of Alpinia zerumbet. Rev. Bras. Farmacogn. 2017, 27, 592–598. [Google Scholar] [CrossRef]
- Taira, N.; Nguyen, B.C.Q.; Tawata, S. Hair growth promoting and anticancer effects of p21-activated kinase 1 (PAK1) inhibitors isolated from different parts of Alpinia zerumbet. Molecules 2017, 22, 132. [Google Scholar] [CrossRef] [PubMed]
- Xuan, T.D.; Khanh, T.D.; Khang, D.T.; Quan, N.T.; Elzaawely, A.A. Changes in chemical composition, total phenolics and antioxidant activity of Alpinia (Alpinia zerumbet) leaves exposed to UV. Int. Let. Nat. Sci. 2016, 55, 25–34. [Google Scholar] [CrossRef]
- Mpalantinos, M.A.; Moura, R.S.; Parente, J.P.; Kuster, R.M. Biologically active flavonoids and kava pyrones from the aqueous extract of Alpinia zerumbet. Phytother. Res. 1998, 12, 442–444. [Google Scholar] [CrossRef]
- Rao, Y.K.; Shih, H.N.; Lee, Y.C.; Cheng, W.T.; Hung, H.C.; Wang, H.C. Purification of kavalactones from Alpinia zerumbet and their protective actions against hydrogen peroxide-induced cytotoxicity in PC12 cells. J. Biosci. Bioeng. 2014, 118, 679–688. [Google Scholar] [CrossRef] [PubMed]
- Leading Causes of Death Worldwide in 2015 (in Millions). Available online: https://www.statista.com/statistics/288839/leading-causes-of-death-worldwide/ (accessed on 19 November 2017).
- Araújo, P.F.; Coelho-de-Souza, A.N.; Morais, S.M.; Ferreira, S.C.; Leal-Cardoso, J.H. Antinociceptive effects of the essential oil of Alpinia zerumbet on mice. Phytomedicine 2005, 12, 482–486. [Google Scholar] [PubMed]
- Araújo, F.Y.; Silva, M.I.; Moura, B.A.; Oliveira, G.V.; Leal, L.K.; Vasconcelos, S.M.; Viana, G.S.; Moraes, M.O.; Sousa, F.C.; Macêdo, D.S. Central nervous system effects of the essential oil of the leaves of Alpinia zerumbet in mice. J. Pharm. Pharmacol. 2009, 61, 1521–1527. [Google Scholar] [CrossRef] [PubMed]
- Araújo, F.Y.; Oliveira, G.V.; Gomes, P.X.L.; Soares, M.A.; Silva, M.I.G.; Carvalho, A.F.; Moraes, M.O.; Moraes, M.E.A.; Vasconcelos, S.M.; Viana, G.S.; et al. Inhibition of ketamine-induced hyperlocomotion in mice by the essential oil of Alpinia zerumbet: Possible involvement of an antioxidant effect. J. Pharm. Pharmacol. 2011, 63, 1103–1110. [Google Scholar] [CrossRef] [PubMed]
- Cavalcanti, B.C.; Ferreira, J.R.; Cabral, I.O.; Magalhães, H.I.F.; Oliveira, C.C.; Rodrigues, F.A.; Rocha, D.D.; Barros, F.W.; Silva, C.R.; Júnior, H.V.; et al. Genetic toxicology evaluation of essential oil of Alpinia zerumbet and its chemoprotective effects against H(2)O(2)-induced DNA damage in cultured human leukocytes. Food Chem. Toxicol. 2012, 50, 4051–4061. [Google Scholar] [CrossRef] [PubMed]
- Victorio, C.P.; Riehl, C.A.; Lage, C.L. Simultaneous distillation-extraction, hydrodistillation and static headspace methods for the analysis of volatile secondary metabolites of Alpinia zerumbet (Per.) Burtt et Smith. from Southeast Brazil. J. Essent. Oil Bear. Plants 2009, 12, 137–143. [Google Scholar] [CrossRef]
- Victorio, C.P.; Leitao, S.G.; Lage, C.L. Chemical composition of the leaf oils of Alpinia zerumbet (Pers.) Burtt et Smith and A. purpurata (Vieill) K. Schum. from Rio de Janeiro, Brazil. J. Essent. Oil Res. 2010, 22, 52–54. [Google Scholar] [CrossRef]
- Victorio, C.P.; Arruda, R.C.; Riehl, C.A.; Lage, C.L. Leaf volatiles and secretory cells of Alpinia zerumbet (Pers.) Burtt et Smith (Zingiberaceae). Nat. Prod. Res. 2011, 25, 939–948. [Google Scholar] [CrossRef] [PubMed]
- Mendes, F.R.S.; Silva, F.G.E.; Sousa, E.O.; Rodrigues, F.F.G.; Costa, J.G.M.; Monte, F.J.Q.; Lemos, T.L.; Assunção, J.C. Essential oil of Alpinia zerumbet (Pers.) BL Burtt. & RM Sm. (Zingiberaceae): Chemical composition and modulation of the activity of aminoglycoside antibiotics. J. Essent. Oil Res. 2015, 27, 259–263. [Google Scholar]
- Murakami, S.; Li, W.; Matsuura, M.; Satou, T.; Hayashi, S.; Koike, K. Composition and seasonal variation of essential oil in Alpinia zerumbet from Okinawa Island. J. Nat. Med. 2009, 63, 204–208. [Google Scholar] [CrossRef] [PubMed]
- Murakami, S.; Matsuura, M.; Satou, T.; Hayashi, S.; Koike, K. Effects of the essential oil from leaves of Alpinia zerumbet on behavioral alterations in mice. Nat. Prod. Commun. 2009, 4, 129–132. [Google Scholar] [PubMed]
- Lahlou, S.; Interaminense, L.F.; Leal-Cardoso, J.H.; Duarte, G.P. Antihypertensive effects of the essential oil of Alpinia zerumbet and its main constituent, terpinen-4-ol, in DOCA-salt hypertensive conscious rats. Fundam. Clin. Pharmacol. 2003, 17, 323–330. [Google Scholar] [CrossRef] [PubMed]
- Lahlou, S.; Galindo, C.A.B.; Leal-Cardoso, J.H.; Fonteles, M.C.; Duarte, G.P. Cardiovascular effects of the essential oil of Alpinia zerumbet leaves and its main constituent, terpinen-4-ol, in rats: Role of the autonomic nervous system. Planta Med. 2002, 68, 1097–1102. [Google Scholar] [CrossRef] [PubMed]
- Leal-Cardoso, J.H.; Moreira, M.R.; Cruz, G.P.; Morais, S.M.; Lahlou, M.S.; Coelho-de-Souza, A.N. Effects of essential oil of Alpinia zerumbet on the compound action potential of the rat sciatic nerve. Phytomedicine 2004, 11, 549–553. [Google Scholar] [CrossRef] [PubMed]
- Pinto, N.V.; Assreuy, A.M.; Coelho-de-Souza, A.N.; Ceccatto, V.M.; Magalhães, P.J.; Lahlou, S.; Leal-Cardoso, J.H. Endothelium-dependent vasorelaxant effects of the essential oil from aerial parts of Alpinia zerumbet and its main constituent 1,8-cineole in rats. Phytomedicine 2009, 16, 1151–1155. [Google Scholar] [CrossRef] [PubMed]
- Satou, T.; Kasuya, H.; Takahashi, M.; Murakami, S.; Hayashi, S.; Sadamoto, K. Relationship between duration of exposure and anxiolytic-like effects of essential oil from Alpinia zerumbet. Flavour Fragr. J. 2011, 26, 180–185. [Google Scholar] [CrossRef]
- Jezler, C.N.; Batista, R.S.; Alves, P.B.; Silva, D.C.; Costa, L.C.B. Histochemistry, content and chemical composition of essential oil in different organs of Alpinia zerumbet. Cienc. Rural 2013, 43, 1811–1816. [Google Scholar] [CrossRef]
- The US National Center for Health Statistics. Available online: https://www.cdc.gov/nchs/index.htm (accessed on 19 November 2017).
- Cause of Death Statistics—People over 65. Available online: http://ec.europa.eu/eurostat/statisticsexplained/index.php/Causes_of_death_statistics_-_people_over_65 (accessed on 19 November 2017).
- The Japan Institute of Health. Available online: http://www.healthdata.org/japan (accessed on 19 November 2017).
- Hadjzadeh, M.A.R.; Ghanbari, H.; Keshavarzi, Z.; Tavakol-Afshari, J. The effects of aqueous extract of Alpinia galanga on gastric cancer cells (AGS) and L929 cells in vitro. Iran. J. Cancer Prev. 2014, 7, 142–146. [Google Scholar] [PubMed]
- Xuan, T.D.; Fukuta, M.; Ao, C.W.; Elzaawely, A.A.; Khanh, T.D.; Tawata, S. Efficacy of extracting solvents to chemical components of kava (Piper methysticum) roots. J. Nat. Med. 2008, 62, 188–194. [Google Scholar] [CrossRef] [PubMed]
- Mokdad, A.H.; Ford, E.S.; Bowman, B.A.; Dietz, W.H.; Vinicor, F.; Bales, V.S.; Marks, J.S. Prevalence of obesity, diabetes, and obesity-related health risk factors, 2001. J. Am. Med. Assoc. 2003, 289, 76–79. [Google Scholar] [CrossRef]
- Klop, B.; Elte, J.W.F.; Cabezas, M.C. Dyslipidemia in obesity: Mechanisms and potential targets. Nutrients 2013, 5, 1218–1240. [Google Scholar] [CrossRef] [PubMed]
- Corey, K.E.; Kaplan, L.M. Obesity and liver disease: The epidemic of the twenty-first century. Clin. Liver Dis. 2014, 18, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Marseglia, L.; Manti, S.; D’Angelo, G.; Nicotera, A.; Parisi, E.; Di Rosa, G.; Gitto, E.; Arrigo, T. Oxidative stress in obesity: A critical component in human diseases. Int. J. Mol. Sci. 2015, 16, 378–400. [Google Scholar] [CrossRef] [PubMed]
- Chan, Y.C.; Suzuki, M.; Yamamoto, S. Dietary, anthropometric, hematological and biochemical assessment of the nutritional status of centenarians and elderly people in Okinawa, Japan. J. Am. Coll. Nutr. 1997, 16, 229–235. [Google Scholar] [CrossRef] [PubMed]
- Subedi, B.H.; Joshi, P.H.; Jones, S.R.; Martin, S.S.; Blaha, M.J.; Michos, E.D. Current guidelines for high-density lipoprotein cholesterol in therapy and future directions. Vasc. Health Risk Manag. 2014, 10, 205–216. [Google Scholar] [CrossRef] [PubMed]
- Nowak, W.N.; Deng, J.; Ruan, X.Z.; Xu, Q. Reactive oxygen species generation and atherosclerosis. Arterioscler. Thromb. Vasc. Biol. 2017, 37, e41–e52. [Google Scholar] [CrossRef] [PubMed]
- Nomura, S.; Sakamoto, H.; Glenn, S.; Tsugawa, Y.; Abe, S.K.; Rahman, M.M.; Brown, J.C.; Ezoe, S.; Fitzmaurice, C.; Inokuchi, T.; et al. Population health and regional variations of disease burden in Japan, 1990–2015: A systematic subnational analysis for the Global Burden of Disease Study 2015. Lancet 2017, 390, 1521–1538. [Google Scholar] [CrossRef]
- Hadenablog. Annual Change of the Main Causes of Death in Okinawa. Available online: http://nbakki.hatenablog.com/entry/Causes_of_Death_in_Okinawa (accessed on 19 November 2017).
- Okinawa Centenarian Study. Available online: http://www.okicent.org/team.html (accessed on 19 November 2017).
- Akisaka, M.; Suzuki, M.; Inoko, H. Molecular genetic studies on DNA polymorphism of the HLA class II genes associated with human longevity. Tissue Antigens 1997, 50, 489–493. [Google Scholar] [CrossRef] [PubMed]
- Inoue, M.; Iso, H.; Yamamoto, S.; Kurahashi, N.; Iwasaki, M.; Sasazuki, S.; Tsugane, S.; Japan Public Health Center-Based Prospective Study Group. Daily total physical activity level and premature death in men and women: Results from a large-scale population-based cohort study in Japan (JPHC Study). Ann. Epidemiol. 2008, 18, 522–530. [Google Scholar] [CrossRef] [PubMed]
- Willcox, D.C.; Willcox, B.J.; Todoriki, H.; Curb, J.D.; Suzuki, M. Caloric restriction and human longevity: What can we learn from the Okinawans? Biogerontology 2006, 7, 173–177. [Google Scholar] [CrossRef] [PubMed]
- Le Bourg, E. Dietary restriction studies in humans: Focusing on obesity, forgetting longevity. Gerontology 2012, 58, 126–128. [Google Scholar] [CrossRef] [PubMed]
- Willcox, B.J.; Yano, K.; Chen, R.; Willcox, D.C.; Rodriguez, B.L.; Masaki, K.H.; Donion, T.; Tanaka, B.; Curb, J.D. How much should we eat? The association between energy intake and mortality in a 36-year follow-up study of Japanese-American men. J. Gerontol. A Biol. Sci. Med. Sci. 2004, 59, 789–795. [Google Scholar] [CrossRef] [PubMed]
- Madeo, F.; Pietrocola, F.; Eisenberg, T.; Kroemer, G. Caloric restriction mimetics: Towards a molecular definition. Nat. Rev. Drug Discov. 2014, 13, 727–740. [Google Scholar] [CrossRef] [PubMed]
- Willcox, B.J.; Willcox, D.C.; Todoriki, H.; Fujiyoshi, A.; Yano, K.; He, Q.; Curb, J.D.; Suzuki, M. Caloric restriction, the traditional Okinawan diet, and healthy aging. The diet of the world’s longest-lived people and its potential impact on morbidity and life span. Ann. N. Y. Acad. Sci. 2007, 1114, 434–455. [Google Scholar] [CrossRef] [PubMed]
- Okinawa: Japan’s exotic southern islands. Available online: https://www3.nhk.or.jp/nhkworld/en/food/articles/38.html (accessed on 19 November 2017).
- Barnes, J.; Anderson, L.A.; Phillipson, J.D. Herbal Medicines, 3rd ed.; Pharmaceutical Press: London, UK, 2007; p. 690. ISBN 9780-8536-96230. [Google Scholar]
- Willcox, D.C.; Willcox, B.J.; Wang, N.C.; He, Q.; Rosenbaum, M.; Suzuki, M. Genetic and environmental determinants of healthy aging. Life at the extreme limit: Phenotypic characteristics of supercentenarians in Okinawa. J. Gerontol. Med. Sci. 2008, 63A, 1201–1208. [Google Scholar] [CrossRef]
- Forrest, H.; Nielsen, F.H.; Johnson, L.A.K. Data from controlled metabolic ward studies provide guidance for the determination of status indicators and dietary requirements for magnesium. Biol. Trace Elem. Res. 2017, 177, 43–52. [Google Scholar]
- Boberg, J.; Gustafsson, I.B.; Karlström, B.; Lithell, H.; Vessby, B.; Werner, I. Treatment of hyperlipoproteinemia with “type-specific” diets under metabolic ward conditions. Effects on serum lipoproteins and serum lipid esters. Ann. Nutr. Metab. 1981, 25, 320–333. [Google Scholar] [CrossRef] [PubMed]
- Schmitt, J.; Ferro, A. Nutraceuticals: Is there good science behind the hype? Br. J. Clin. Pharmacol. 2013, 75, 585–587. [Google Scholar] [CrossRef] [PubMed]
- Teschke, R.; Wolff, A.; Frenzel, C.; Eickhoff, A.; Schulze, J. Herbal traditional Chinese medicine and its evidence base in gastrointestinal disorders. World J. Gastroenterol. 2015, 21, 4466–4490. [Google Scholar] [CrossRef] [PubMed]
- Teschke, R.; Zhang, L.; Long, H.; Schwarzenboeck, A.; Schmidt-Taenzer, W.; Genthner, A.; Wolff, A.; Frenzel, C.; Schulze, J.; Eickhoff, A. Traditional Chinese Medicine and herbal hepatotoxicity: A tabular compilation of reported cases. Ann. Hepatol. 2015, 14, 7–19. [Google Scholar] [CrossRef]
- Zambrone, F.A.D.; Corrêa, C.L.; Amaral, L.M.S.D. A critical analysis of the hepatotoxicity cases described in the literature related to Herbalife (r) products. Braz. J. Pharm. Sci. 2015, 51, 785–796. [Google Scholar] [CrossRef]
- Teschke, R.; Schulze, J.; Eickhoff, A.; Wolff, A.; Frenzel, C. Mysterious Hawaii liver disease case—Naproxen overdose as cause rather than Oxy ELITE Pro? J. Liver Clin. Res. 2015, 2, 1013. Available online: http://www.jscimedcentral.com/Liver/liver-2-1013.pdf (accessed on 19 November 2017).
- Teschke, R.; Andrade, R.J. Drug, herb, and dietary supplement hepatotoxicity. Int. J. Mol. Sci. 2016, 17, 1488. [Google Scholar] [CrossRef] [PubMed]
- Teschke, R.; Eickhoff, A. The Honolulu Liver disease cluster at the Medical Center: Its mysteries and challenges. Int. J. Mol. Sci. 2016, 17, 476. [Google Scholar] [CrossRef] [PubMed]
- Teschke, R.; Schwarzenboeck, A.; Frenzel, C.; Schulze, J.; Eickhoff, A.; Wolff, A. The mystery of the Hawaii liver disease cluster in summer 2013: A pragmatic and clinical approach to solve the problem. Ann. Hepatol. 2016, 15, 91–119. Available online: http://www.annalsofhepatology.com.mx/revista/numeros/2016/HP161-12-Mystery%20(web)%20(FF_041215V)_PROTEGIDO%20(1).pdf (accessed on 19 November 2017). [CrossRef] [PubMed]
- Brown, A. Liver toxicity related to herbs and dietary supplements: Online table of case reports. Part 2 of 5 series. Food Chem. Toxicol. 2017, 107, 472–501. [Google Scholar] [CrossRef] [PubMed]
- Lane, M.A.; Ingram, D.K.; Roth, G.S. 2-Deoxy-d-glucose feeding in rats mimics physiologic effects of calorie restriction. J. Anti-Aging Med. 1998, 1, 327–337. [Google Scholar] [CrossRef]
- Cantó, C.; Auwerkx, J. Caloric restriction: Is AMPK a key sensor and effector? Physiology 2011, 26, 214–224. [Google Scholar] [CrossRef] [PubMed]
- Chen, D.; Guarente, L. SIR2: A potential target for caloric restriction mimetics. Trends Mol. Med. 2007, 13, 64–71. [Google Scholar] [CrossRef] [PubMed]
- Test, G.; Biasi, F.; Poli, G.; Chiarpotto, E. Caloric restriction mimetics: A strategy for improving healthy aging and longevity. Curr. Pharm. Des. 2014, 20, 2950–2977. [Google Scholar] [CrossRef]
- Guarente, L. Sir2 links chromatin silencing, metabolism, and aging. Genes Dev. 2000, 214, 1021–1026. [Google Scholar]
- Joshi, P.K.; Pirastu, N.; Kentistou, K.A.; Fischer, K.; Hofer, E.; Schraut, K.E.; Clark, D.W.; Nutile, T.; Barnes, C.L.; Timmers, P.R.; et al. Genome-wide meta-analysis associates HLA-DQA1/DRB1 and LPA and lifestyle factors with human longevity. Nat. Commun. 2017, 8, 910. [Google Scholar] [CrossRef] [PubMed]
- Yang, F.; Sun, L.; Zhu, X.; Han, J.; Zeng, Y.; Nie, C.; Yuan, H.; Li, X.; Shi, X.; Yang, Y.; et al. Identification of new genetic variants of HLA-DQB1 associated with human longevity and lipid homeostasis—A cross-sectional study in a Chinese population. Aging 2017, 9, 2311–2333. [Google Scholar] [CrossRef] [PubMed]
- Pilling, L.C.; Kuo, C.L.; Sicinski, K.; Tamosauskaite, J.; Kuchel, G.A.; Harries, L.W.; Herd, P.; Wallace, R.; Ferrucci, L.; Melzer, D. Human longevity: 25 genetic loci associated in 389,166 UK biobank participants. Aging 2017, 9, 2504–2520. [Google Scholar] [CrossRef] [PubMed]

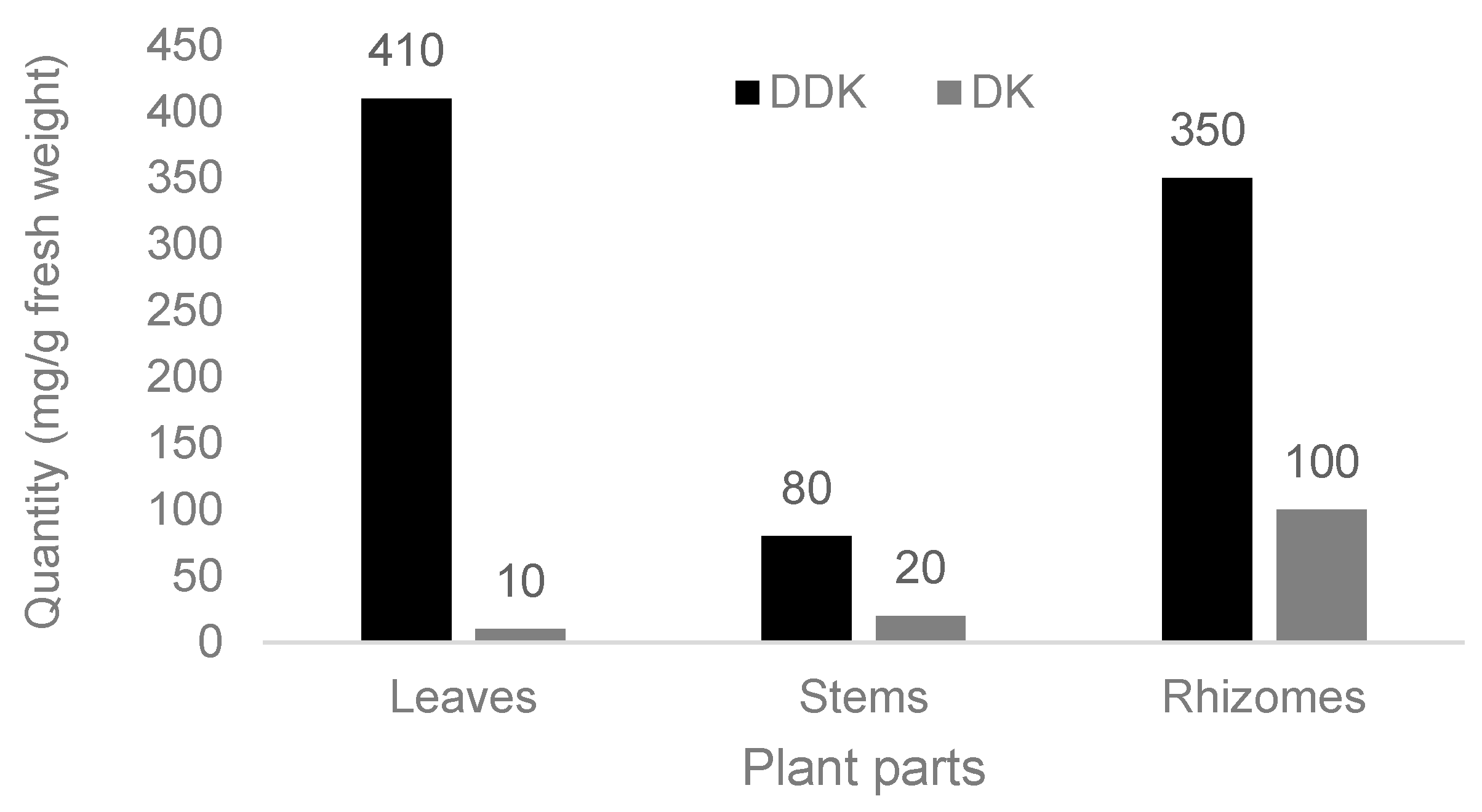
| Category | Description |
|---|---|
| Preferred scientific name | Alpinia zerumbet (Pers.) B.L. Burtt & R.M. Sm. |
| Preferred common name | Shell ginger |
| Taxonomic tree | Domain: Eukaryota; Kingdom: Plantae; Phylum: Spermatophyta; Subphylum: Angiospermae; Class: Monocotyledonae; Order: Zingiberaceae; Genus: Alpinia; Species: Alpinia zerumbet |
| Other scientific names | Alpinia speciosa (J.C.Wendl.) K. Schum.; Amomum nutans (Andrews) Schult.; Catimbium speciosum (J.C.Wendl.) Holttum; Costus zerumbet Pers.; Languas speciosa (J.C.Wendl.) Small; Renealmia nutans Andrews; Zerumbet speciosum J.C.Wendl. |
| International common names | English: light galangal; pink porcelain lily; shell flower; French: atoumau; Chinese: yan shan jiang |
| Other names in distributed countries | Japan: getto; Brazil: colonia; Cook Islands: kaopui; Cuba: boca de lobo; Dominican Republic: burriquito; Haiti: de tui maux; tous maux; Indonesia: galoba merah; Myanmar: padegaw-gyi; the Philippines: langkuas na pula; Puerto Rico: boca de dragon; Thailand: khaa khom; Tonga: teuila; Vietnam: Riềng ấm |
| Distribution | South, Southeast, and East Asia (from India to Japan), and southward to Oceania and Australia. Cultivated as an ornamental plant in tropics and subtropics. |
| Uses | A spice, food, medicine, beverage, pharmacy, cosmetics, paper, and ornament |
| References | Information in this histogram is from literature and references [10,11,12,13,14,15,16,17,18,19,20,21,22]. |
| Chemical Name | Plant Parts | Quantity | References |
|---|---|---|---|
| Kavalactones Dihydro-5,6-dehydrokawain (DDK) | Leaves, stems, rhizomes, fruits | 80–410 mg g−1 | [18,27,28,29] |
| 5,6-Dehydrokawain (DK) | Leaves, stems, rhizomes, fruits | 10–100 mg g−1 | [18,19,27,28] |
| Methyltriacetolactone | Leaves | 0.44% ** | [26] |
| Non-kavalactones Essential oils | Leaves, petal, roots | <150 mg g−1 | [18,19,20,24,27,30,31,32,33,34,35,36,37,38,39,40,41,42,43,44,45,46] |
| (E)-labda-8(17)-12-diene-15-ol-16-al | Rhizomes | [47] | |
| (E)-15,16-bisnorlabda-8(17)-11-diene-13-one | Rhizomes | [47] | |
| Phenolic acids | Leaves, stems, rhizomes | <1.0 mg g−1 | [15,18,27,29] |
| Phenols | Leaves | 108.27 mg g−1 | [27] |
| 12-Labdaiene-15,16-dial (labdadiene) | Rhizomes, stems, leaves, flowers, pericarps, seeds | 0.75–1.0 mg g−1 | [12,48] |
| Rutin | Leaves, seeds | [23,49] | |
| Kaempferol-3-o-glucuronide | Leaves | [28,49] | |
| Quercetin | Seeds | [23] | |
| (+) Catechin | Leaves | [49] | |
| (−) Epicatechin | Leaves | [49] | |
| p-hydroxycinnamaldehyde | Rhizomes | [21] | |
| (i-(p-hydroxy-cis-styryl)) methane | Rhizomes | [21] | |
| Kaempferol-3-o-rutinoside | Leaves | [28] | |
| Dihydroflavokavain B | Rhizomes | [15] | |
| Chalcones (2′,4′-Dihydroxy-6′-methoxy chalcone; 2',4'-dihydroxy-5'-methoxy chalcone) | Seeds, leaves | 0.25% * | [27] |
| trans-1-(4′-Hydroxy-3′-methoxyphenyl-7-phenylhept-1-en-3-one (yakuchinone-B) | Pericarps | [22] | |
| Labda-8(17),12-diene-15,16-dial | Rhizomes | [28] | |
| Bisabolanes | Leaves | [28] | |
| Steroids | Leaves, seeds | <1% * | [27] |
| Fatty acids | Leaves | 62 µg g−1 | [27] |
| Phenanthrin | Leaves | 4.65% * | [27] |
| Activity | Extracts and Doses | References |
|---|---|---|
| Antioxidant DPPH | Aqueous (IC50): 10.3–165.6 µg mL−1 Ethanol (IC50): 136.6–599.0 µg mL−1 | [13] |
| Antioxidant ABTS | Aqueous 73.9–163.4 µg mL−1 Ethanol: 137.9–293.9 µg mL−1 | [13] |
| Antioxidant PMS-NADH | Aqueous: 58.6–215.1 µg mL−1 Ethanol: 56.1–210.5 µg mL−1 | [13] |
| Total phenols | Aqueous: 47.8–187.8 mg g−1 GAE Ethanol: 9.6–56.3 mg g−1 GAE | [13] |
| Tyrosinase | 2.30–312.5 µg L−1 | [12] |
| Pancreatic lipase | 5.0–27.8 µg L−1 | [12] |
| 15-Lipoxygenase | 1.26–1866.8 µg L−1 | [12] |
| LDL oxidation | 15.4–515.5 µg L−1 | [12] |
| Longevity | Leaf extracts: 50–100 µg mL−1 increased 22.6% life span of Caenorhabditis elegans | [20] |
| Anti-cancer on A549 cell line | Compounds isolated from A. zerumbet: IC 50 values: labdadiene: 67.1 µM; MTD: 98.9 µM; TMOQ 90.8 µM; KOG 81.4 µM | [26] |
| PAK1 inhibitory | Compounds isolated from A. zerumbet: IC50 value: labdadiene IC50: 52.1 µM; MTD: 58.6 µM; TMOQ 49.3 µM; KOG 12.9 µM | [26] |
| Anti-obesity | Compounds including hispidin, DK, and DDK isolated from A. zerumbet: Dose of 250 µg mL−1. Glycerol release increased 276.4%, 225.1%, and 137.1%; cAMP promoted 81.2%, 67.0%, and 56.9%; Inhibition on lipid accumulation: 47.8%, 48.0%, and 36.8%, respectively. | [10] |
| a | b | c | d | e | f | g | h | |
|---|---|---|---|---|---|---|---|---|
| Japan | 44.7 * | 31.7 * | 25.1 † | 18.2 † | 10.3 † | 8.3 † | 5.7 * | 4.6 * |
| Okinawa | 46.6 * | 29.9 * | 19.7 † | 12.1 † | 7.8 † | 5.9 † | 5.4 | 4.8 * |
| a | b | c | d | e | f | g | h | i | j | k | l | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Japan | 813.2 * | 466.8 * | 438.5 * | 437.9 † | 373.5 † | 341.5 † | 298.4 † | 290.0 * | 280.0 * | 275.0 † | 264.3 * | 246.8 * |
| Okinawa | 813.1 * | 472.1 * | 443.8 * | 400.6 † | 353.4 † | 234.0 † | 213.5 † | 286.1 * | 279.9 * | 236.6 † | 264.4 * | 251.2 * |
| Alpinia-derived products in Okinawa | Details of Alpinia preparation and use in the traditional Okinawa cuisine |
| Tea | One pack includes dried Alpinia powder from leaves, flowers, and rhizome. Few teas use only the leaves, flowers, or rhizome individually, others use mixtures. The amount of one pack is on average 2.5 g, dipped in 200–300 mL of hot water. Daily or intermittent consumption. |
| Mochi | Traditional sticky rice cake that is sold commonly in Okinawan markets. The rice cake is prepared with brown sugar or local purple sweet potato, covered with several fresh leaves of Alpinia, and steamed. When consumed, the rice cake has a strong characteristic flavour of Alpinia due to its essential oils, similar to the common ginger vegetable. Most Okinawans eat this cake regularly, some intermittently. |
| Steamed bun | Common rice powder prepared with meat or other ingredients, covered with fresh Alpinia leaves and steamed: not used daily but restricted to special occasions. |
| Fried meat or fish | The meat or fish is covered with fresh Alpinia leaves, steamed, and then fried before consumption on a non-regular basis. |
| Buckwheat | The dried powder of Alpinia leaves is mixed with buckwheat powder, boiled, and consumed. |
| Ice cream | Uses extract of Alpinia leaves to provide a fragrance to ice cream: it is consumed on special occasions. |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Teschke, R.; Xuan, T.D. Viewpoint: A Contributory Role of Shell Ginger (Alpinia zerumbet) for Human Longevity in Okinawa, Japan? Nutrients 2018, 10, 166. https://doi.org/10.3390/nu10020166
Teschke R, Xuan TD. Viewpoint: A Contributory Role of Shell Ginger (Alpinia zerumbet) for Human Longevity in Okinawa, Japan? Nutrients. 2018; 10(2):166. https://doi.org/10.3390/nu10020166
Chicago/Turabian StyleTeschke, Rolf, and Tran Dang Xuan. 2018. "Viewpoint: A Contributory Role of Shell Ginger (Alpinia zerumbet) for Human Longevity in Okinawa, Japan?" Nutrients 10, no. 2: 166. https://doi.org/10.3390/nu10020166
APA StyleTeschke, R., & Xuan, T. D. (2018). Viewpoint: A Contributory Role of Shell Ginger (Alpinia zerumbet) for Human Longevity in Okinawa, Japan? Nutrients, 10(2), 166. https://doi.org/10.3390/nu10020166






