Acute Effect of Resistant Starch on Food Intake, Appetite and Satiety in Overweight/Obese Males
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Test Products and Supplements
2.3. Study Protocol
2.4. Ad Libitum Pasta Meal
2.5. Biochemistry
2.6. Insulin Sensitivity
2.7. Calculations and Statistical Analysis
3. Results
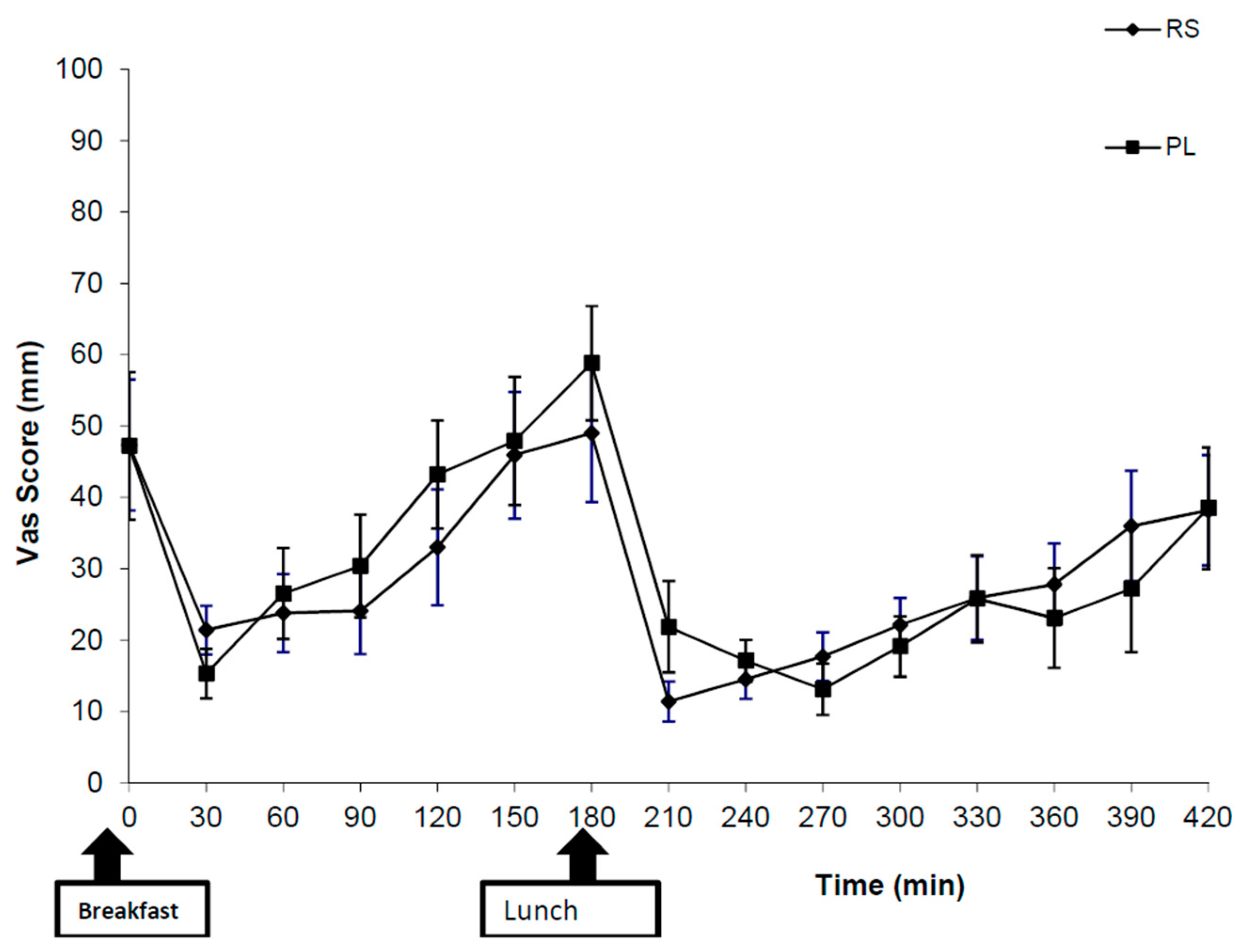
3.1. Quantitative Appetite Assessment
3.2. Qualitative Appetite Assessment
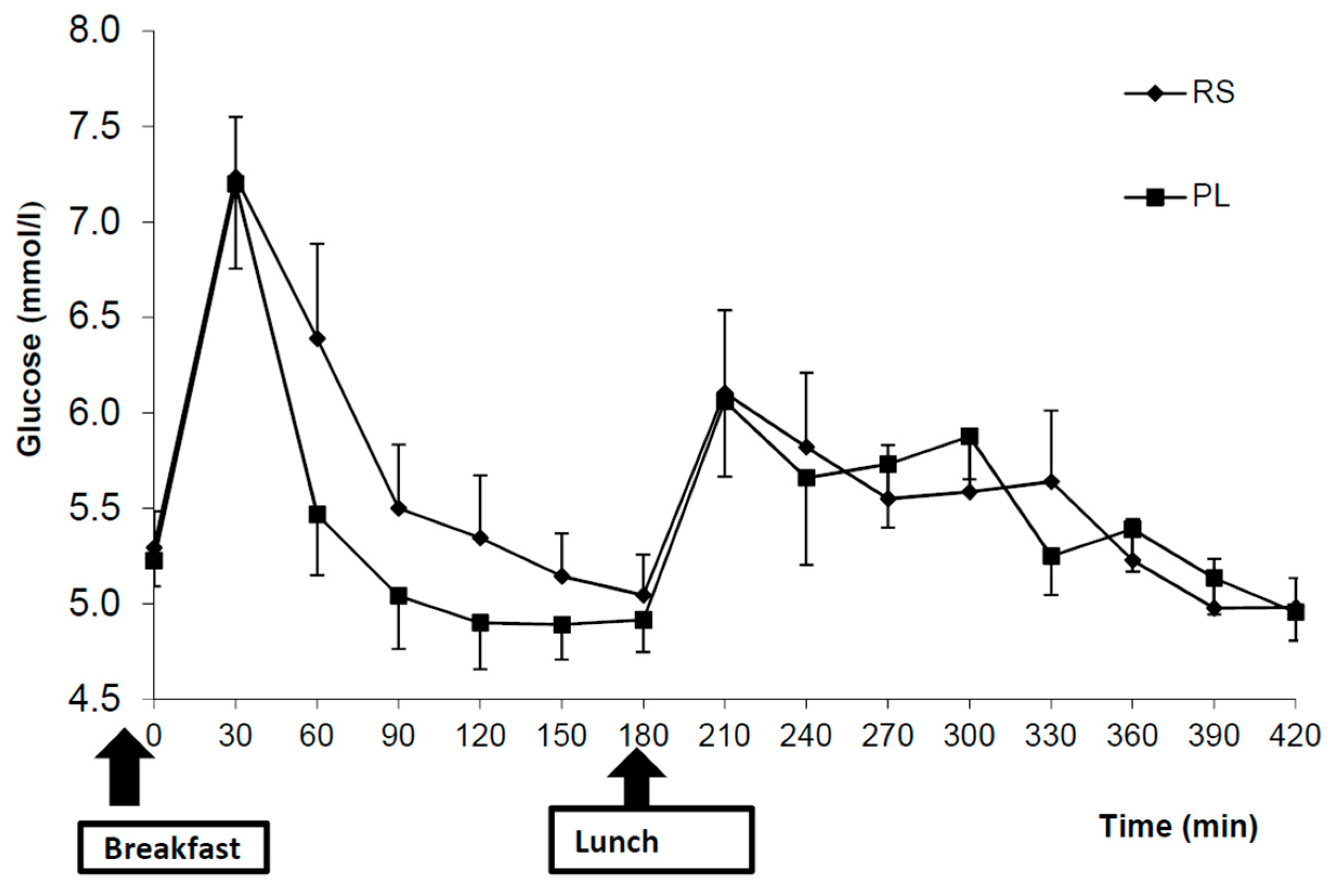
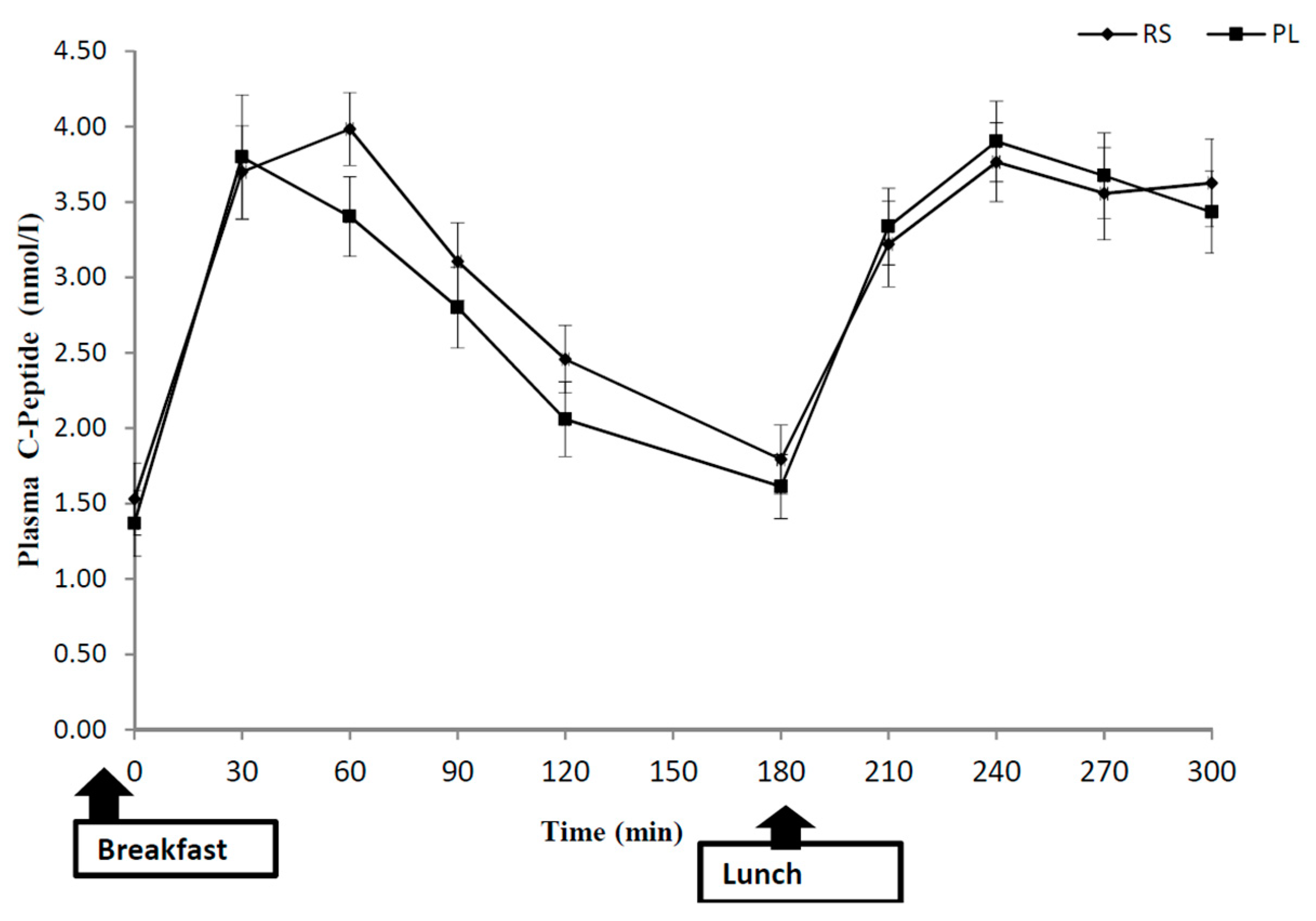
3.3. Postprandial Metabolites
3.4. Insulin Sensitivity
3.5. Palatability and Gastrointestinal Symptoms
4. Discussion
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Al-Nuaim, A.R.; Al-Rubeaan, K.; Al-Mazrou, Y.; Al-Attas, O.; Al-Daghari, N.; Khoja, T. High prevalence of overweight and obesity in Saudi Arabia. Int. J. Obes. Relat. Metab. Disord. 1996, 20, 547–552. [Google Scholar] [PubMed]
- Al-Sendi, A.M.; Shetty, P.; Musaiger, A.O. Prevalence of overweight and obesity among Bahraini adolescents: A comparison between three different sets of criteria. Eur. J. Clin. Nutr. 2003, 57, 471–474. [Google Scholar] [CrossRef] [PubMed]
- Flegal, K.M.; Carroll, M.D.; Ogden, C.L.; Curtin, L.R. Prevalence and trends in obesity among US adults, 1999–2008. JAMA 2010, 303, 235–241. [Google Scholar] [CrossRef] [PubMed]
- Ng, S.W.; Zaghloul, S.; Ali, H.I.; Harrison, G.; Popkin, B.M. The prevalence and trends of overweight, obesity and nutrition-related non-communicable diseases in the Arabian Gulf States. Obes. Rev. 2011, 12, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Seidell, J.C.; Flegal, K.M. Assessing obesity: Classification and epidemiology. Br. Med. Bull. 1997, 53, 238–252. [Google Scholar] [CrossRef]
- WHO. Obesity and Overweight. Available online: http://www.who.int/mediacentre/factsheets/fs311/en/#.WO9bKDNFkY8.mendeley&title=Obesityandoverweight&description=Overweightandobesityaredefinedasabnormalorexcessivefataccumulationthatmayimpairhealth (accessed on 16 February 2018).
- Jokela, M.; Kivimaki, M.; Elovainio, M.; Viikari, J.; Raitakari, O.T.; Keltikangas-Jarvinen, L. Urban/rural differences in body weight: Evidence for social selection and causation hypotheses in Finland. Soc. Sci. Med. 2009, 68, 867–875. [Google Scholar] [CrossRef]
- Mozaffarian, D.; Hao, T.; Rimm, E.B.; Willett, W.C.; Hu, F.B. Changes in diet and lifestyle and long-term weight gain in women and men. N. Engl. J. Med. 2011, 364, 2392–2404. [Google Scholar] [CrossRef]
- Du, H.; van Der, A.D.; Boshuizen, H.C.; Forouhi, N.G.; Wareham, N.J.; Halkjaer, J.; Tjonneland, A.; Overvad, K.; Jakobsen, M.U.; Boeing, H.; et al. Dietary fiber and subsequent changes in body weight and waist circumference in European men and women. Am. J. Clin. Nutr. 2010, 91, 329–336. [Google Scholar] [CrossRef]
- Howarth, N.C.; Murphy, S.P.; Wilkens, L.R.; Hankin, J.H.; Kolonel, L.N. Dietary energy density is associated with overweight status among 5 ethnic groups in the multiethnic cohort study. J. Nutr. 2006, 136, 2243–2248. [Google Scholar] [CrossRef]
- Kendall, C.W.; Esfahani, A.; Sanders, L.M.; Potter, S.M.; Vidgen, E. The effect of a pre-load meal containing resistant starch on spontaneousfood intake and glucose and insulin responses. J. Food Technol. 2010, 8, 67–73. [Google Scholar] [CrossRef]
- Lattimer, J.M.; Haub, M.D. Effects of dietary fiber and its components on metabolic health. Nutrients 2010, 2, 1266–1289. [Google Scholar] [CrossRef] [PubMed]
- Slavin, J.L. Dietary fiber and body weight. Nutrition 2005, 21, 411–418. [Google Scholar] [CrossRef] [PubMed]
- Papathanasopoulos, A.; Camilleri, M. Dietary fiber supplements: Effects in obesity and metabolic syndrome and relationship to gastrointestinal functions. Gastroenterology 2010, 138, 65–72.e61. [Google Scholar] [CrossRef] [PubMed]
- Slavin, J. Fiber and prebiotics: Mechanisms and health benefits. Nutrients 2013, 5, 1417–1435. [Google Scholar] [CrossRef]
- Brown, I.L. Applications and uses of resistant starch. J. AOAC Int. 2004, 87, 727–732. [Google Scholar] [PubMed]
- Nugent, A.P. Health properties of resistant starch. Nutr. Bull. 2005, 30, 27–54. [Google Scholar] [CrossRef]
- Phillips, G.O. Dietary fibre: A chemical category or a health ingredient? Bioact. Carbohydr. Diet. Fibre 2013, 1, 3–9. [Google Scholar] [CrossRef]
- Bodinham, C.L.; Al-Mana, N.M.; Smith, L.; Robertson, M.D. Endogenous plasma glucagon-like peptide-1 following acute dietary fibre consumption. Br. J. Nutr. 2013, 110, 1429–1433. [Google Scholar] [CrossRef]
- Keenan, M.J.; Zhou, J.; McCutcheon, K.L.; Raggio, A.M.; Bateman, H.G.; Todd, E.; Jones, C.K.; Tulley, R.T.; Melton, S.; Martin, R.J.; et al. Effects of resistant starch, a non-digestible fermentable fiber, on reducing body fat. Obesity 2006, 14, 1523–1534. [Google Scholar] [CrossRef]
- Willis, H.J.; Eldridge, A.L.; Beiseigel, J.; Thomas, W.; Slavin, J.L. Greater satiety response with resistant starch and corn bran in human subjects. Nutr. Res. 2009, 29, 100–105. [Google Scholar] [CrossRef]
- Ble-Castillo, J.L.; Juárez-Rojop, I.E.; Tovilla-Zárate, C.A.; García-Vázquez, C.; Servin-Cruz, M.Z.; Rodríguez-Hernández, A.; Araiza-Saldaña, C.I.; Nolasco-Coleman, A.M.; Díaz-Zagoya, J.C. Acute consumption of resistant starch reduces food intake but has no effect on appetite ratings in healthy subjects. Nutrients 2017, 9, 696. [Google Scholar] [CrossRef] [PubMed]
- Bodinham, C.L.; Frost, G.S.; Robertson, M.D. Acute ingestion of resistant starch reduces food intake in healthy adults. Br. J. Nutr. 2010, 103, 917–922. [Google Scholar] [CrossRef] [PubMed]
- Van Strien, T.; Frijters, J.E.R.; Bergers, G.P.A.; Defares, P.B. The Dutch eating behavior questionnaire (DEBQ) for assessment of restrained, emotional, and external eating behavior. Int. J. Eat. Disord. 1986, 5, 295–315. [Google Scholar] [CrossRef]
- AOAC. AOAC. AOAC Method 991.43. Total, insoluble and soluble dietary fiber in food—Enzymatic-gravimetric method, MES-TRIS buffer. In Official Methods of Analysis; AOAC International: Gaithersburg, MD, USA, 1995. [Google Scholar]
- Flint, A.; Raben, A.; Blundell, J.E.; Astrup, A. Reproducibility, power and validity of visual analogue scales in assessment of appetite sensations in single test meal studies. Int. J. Obes. Relat. Metab. Disord. 2000, 24, 38–48. [Google Scholar] [CrossRef] [PubMed]
- Weststrate, J.A.; van Amelsvoort, J.M. Effects of the amylose content of breakfast and lunch on postprandial variables in male volunteers. Am. J. Clin. Nutr. 1993, 58, 180–186. [Google Scholar] [CrossRef] [PubMed]
- Darzi, J.; Frost, G.S.; Robertson, M.D. Effects of a novel propionate-rich sourdough bread on appetite and food intake. Eur. J. Clin. Nutr. 2012, 66, 789–794. [Google Scholar] [CrossRef] [PubMed]
- Levy, J.C.; Matthews, D.R.; Hermans, M.P. Correct homeostasis model assessment (HOMA) evaluation uses the computer program. Diabetes Care 1998, 21, 2191–2192. [Google Scholar] [CrossRef] [PubMed]
- Matsuda, M.; DeFronzo, R.A. Insulin sensitivity indices obtained from oral glucose tolerance testing: Comparison with the euglycemic insulin clamp. Diabetes Care 1999, 22, 1462–1470. [Google Scholar] [CrossRef]
- Matthews, D.R.; Hosker, J.P.; Rudenski, A.S.; Naylor, B.A.; Treacher, D.F.; Turner, R.C. Homeostasis model assessment: Insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia 1985, 28, 412–419. [Google Scholar] [CrossRef]
- Wallace, T.M.; Levy, J.C.; Matthews, D.R. Use and abuse of HOMA modeling. Diabetes Care 2004, 27, 1487–1495. [Google Scholar] [CrossRef]
- Bergman, R.N.; Ader, M.; Huecking, K.; Van Citters, G. Accurate assessment of beta-cell function: The hyperbolic correction. Diabetes 2002, 51 (Suppl. 1), S212–S220. [Google Scholar] [CrossRef]
- Lorenzo, C.; Wagenknecht, L.E.; Rewers, M.J.; Karter, A.J.; Bergman, R.N.; Hanley, A.J.G.; Haffner, S.M. Disposition index, glucose effectiveness, and conversion to type 2 diabetes: The insulin resistance atherosclerosis study (IRAS). Diabetes Care 2010, 33, 2098–2103. [Google Scholar] [CrossRef] [PubMed]
- Goedecke, J.H.; Dave, J.A.; Faulenbach, M.V.; Utzschneider, K.M.; Lambert, E.V.; West, S.; Collins, M.; Olsson, T.; Walker, B.R.; Seckl, J.R.; et al. Insulin response in relation to insulin sensitivity. Diabetes Care 2009, 32, 860–865. [Google Scholar] [CrossRef] [PubMed]
- Singh, B.; Saxena, A. Surrogate markers of insulin resistance: A review. World J. Diabetes 2010, 1, 36–47. [Google Scholar] [CrossRef] [PubMed]
- Meier, J.J.; Holst, J.J.; Schmidt, W.E.; Nauck, M.A. Reduction of hepatic insulin clearance after oral glucose ingestion is not mediated by glucagon-like peptide 1 or gastric inhibitory polypeptide in humans. Am. J. Physiol. Endocrinol. Metab. 2007, 293, E849–856. [Google Scholar] [CrossRef] [PubMed]
- De Natale, C.; Annuzzi, G.; Bozzetto, L.; Mazzarella, R.; Costabile, G.; Ciano, O.; Riccardi, G.; Rivellese, A.A. Effects of a plant-based high-carbohydrate/high-fiber diet versus high-monounsaturated fat/low-carbohydrate diet on postprandial lipids in type 2 diabetic patients. Diabetes Care 2009, 32, 2168–2173. [Google Scholar] [CrossRef] [PubMed]
- Anderson, G.H.; Cho, C.E.; Akhavan, T.; Mollard, R.C.; Luhovyy, B.L.; Finocchiaro, E.T. Relation between estimates of cornstarch digestibility by the Englyst in vitro method and glycemic response, subjective appetite, and short-term food intake in young men. Am. J. Clin. Nutr. 2010, 91, 932–939. [Google Scholar] [CrossRef]
- Flint, A.; Raben, A.; Astrup, A.; Holst, J.J. Glucagon-like peptide 1 promotes satiety and suppresses energy intake in humans. J. Clin. Investig. 1998, 101, 515–520. [Google Scholar] [CrossRef]
- Pereira, M.A.; Ludwig, D.S. Dietary fiber and body-weight regulation. Observations and mechanisms. Pediatr. Clin. North Am. 2001, 48, 969–980. [Google Scholar] [CrossRef]
- Zhou, J.; Hegsted, M.; McCutcheon, K.L.; Keenan, M.J.; Xi, X.; Raggio, A.M.; Martin, R.J. Peptide YY and proglucagon mRNA expression patterns and regulation in the gut. Obesity 2006, 14, 683–689. [Google Scholar] [CrossRef]
- Zhou, J.; Martin, R.J.; Tulley, R.T.; Raggio, A.M.; McCutcheon, K.L.; Shen, L.; Danna, S.C.; Tripathy, S.; Hegsted, M.; Keenan, M.J. Dietary resistant starch upregulates total GLP-1 and PYY in a sustained day-long manner through fermentation in rodents. Am. J. Physiol. Endocrinol. Metab. 2008, 295, E1160–E1166. [Google Scholar] [CrossRef] [PubMed]
- Brites, C.M.; Trigo, M.J.; Carrapico, B.; Alvina, M.; Bessa, R.J. Maize and resistant starch enriched breads reduce postprandial glycemic responses in rats. Nutr. Res. 2011, 31, 302–308. [Google Scholar] [CrossRef] [PubMed]
- Hallstrom, E.; Sestili, F.; Lafiandra, D.; Bjorck, I.; Ostman, E. A novel wheat variety with elevated content of amylose increases resistant starch formation and may beneficially influence glycaemia in healthy subjects. Food Nutr. Res. 2011, 55, 7074. [Google Scholar] [CrossRef] [PubMed]
- Verdich, C.; Toubro, S.; Buemann, B.; Lysgard Madsen, J.; Juul Holst, J.; Astrup, A. The role of postprandial releases of insulin and incretin hormones in meal-induced satiety--effect of obesity and weight reduction. Int. J. Obes. Relat. Metab. Disord. 2001, 25, 1206–1214. [Google Scholar] [CrossRef]
- Chaput, J.P.; Tremblay, A. The glucostatic theory of appetite control and the risk of obesity and diabetes. Int. J. Obes. 2009, 33, 46–53. [Google Scholar] [CrossRef] [PubMed]
- Levin, B.E. Role of neuronal glucosensing in the regulation of energy homeostasis. Diabetes Care 2006, 55, S122–S130. [Google Scholar] [CrossRef]
- Mayer, J. Glucostatic mechanism of regulation of food intake. N. Engl. J. Med. 1953, 249, 13–16. [Google Scholar] [CrossRef] [PubMed]
- Klosterbuer, A.S.; Greaves, K.A.; Slavin, J. Fiber intake inconsistently alters gut hormone levels in humans following acute or chronic intake. J. Food Res. 2012, 1, 255–273. [Google Scholar] [CrossRef]
| Overall (n = 10) | |||
|---|---|---|---|
| Mean | SD | Range | |
| Age (years) | 22 | 3.7 | 18–31 |
| Height (cm) | 176.4 | 6.5 | 167–188 |
| Weight (kg) | 99.7 | 12.4 | 82–119.5 |
| BMI (kgm2) | 32 | 2.7 | 28–37 |
| DEBQ Restrained | 2.5 | 0.70 | 1.7–3.8 |
| DEBQ Emotional | 2.2 | 0.77 | 1–4 |
| DEBQ External | 3.2 | 0.71 | 1.7–4 |
| Fasting Glucose (mmol/L) | 4.9 | 0.65 | 3.7–5.9 |
| Haemoglobin (g/dL) | 16.6 | 1.52 | 14.1–19.4 |
| Breakfast | Lunch | |||
|---|---|---|---|---|
| Mean | SEM | Mean | SEM | |
| Energy (kcal) | 442 | 1.18 | 1161 | 59.1 |
| Protein (g) | 9.6 | 0.05 | 42.6 | 0.09 |
| Carbohydrate (g) | 83.0 | 0.33 | 138 | 0.34 |
| Fat (g) | 8.0 | 0.05 | 45.1 | 4.80 |
| Fibre (g) | 0.5 (24.7 *) | 0.08 (0.15) * | 5.9 (30.1 *) | 0.08 (0.15) * |
| PL | RS | p-Value | |||
|---|---|---|---|---|---|
| Mean | SEM | Mean | SEM | ||
| (A) Intake of ad libitum Test Dinner | |||||
| Energy (kJ) | 5197 | 561 | 4551 | 617 | 0.017 |
| (B) 24 h Intake | |||||
| Energy (kJ) | 12,553 | 722.5 | 12,955 | 1198.98 | 0.77 |
| Protein (g) | 105.4 | 8.1 | 111.7 | 11.7 | 0.66 |
| Carbohydrate (g) | 419.00 | 24.0 | 431.9 | 36.1 | 0.75 |
| Sugar (g) | 102.3 | 11.6 | 97.6 | 12.3 | 0.81 |
| Fat (g) | 97.61 | 6.7 | 99.7 | 11.89 | 0.88 |
| SFA (g) | 35.32 | 2.7 | 41.1 | 6.1 | 0.40 |
| Fibre (g) | 63.42 | 1.0 | 17.2 | 1.57 | 0.005 |
| RS | PL | p | |||
|---|---|---|---|---|---|
| Mean | SEM | Mean | SEM | ||
| * AUC C-peptide | |||||
| AUC (nmol·L−1 120 min) | 383,377 | 21,302 | 351,348 | 0.065 | 0.065 |
| AUC (nmol·L−1 300 min) | 397,467 | 27,410 | 3039 | 22,770 | 0.58 |
| AUC (nmol·L−1 300 min) | 780,844 | 45,898 | 754,387 | 44,383 | 0.19 |
| * AUC Insulin | |||||
| AUC 0–120 min (pmol/L) | 88,606 | 10,205 | 84,766 | 10,493 | 0.60 |
| AUC 180–300 min (pmol/L) | 96,047 | 14,930 | 97,754 | 15,964 | 0.86 |
| AUC 0–300 min (pmol/L) | 269,460 | 36,671 | 228,746 | 33,646 | 0.39 |
| * C-peptide to insulin ratio | |||||
| B/fast (0–120) | 5.02 | 0.81 | 4.68 | 0.57 | 0.54 |
| Lunch (180–300) | 5.13 | 0.75 | 5.08 | 0.72 | 0.90 |
| Total (0–300) | 4.93 | 0.71 | 4.82 | 0.63 | 0.74 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Al-Mana, N.M.; Robertson, M.D. Acute Effect of Resistant Starch on Food Intake, Appetite and Satiety in Overweight/Obese Males. Nutrients 2018, 10, 1993. https://doi.org/10.3390/nu10121993
Al-Mana NM, Robertson MD. Acute Effect of Resistant Starch on Food Intake, Appetite and Satiety in Overweight/Obese Males. Nutrients. 2018; 10(12):1993. https://doi.org/10.3390/nu10121993
Chicago/Turabian StyleAl-Mana, Najlaa M., and M. Denise Robertson. 2018. "Acute Effect of Resistant Starch on Food Intake, Appetite and Satiety in Overweight/Obese Males" Nutrients 10, no. 12: 1993. https://doi.org/10.3390/nu10121993
APA StyleAl-Mana, N. M., & Robertson, M. D. (2018). Acute Effect of Resistant Starch on Food Intake, Appetite and Satiety in Overweight/Obese Males. Nutrients, 10(12), 1993. https://doi.org/10.3390/nu10121993





