Exercise and the Timing of Snack Choice: Healthy Snack Choice is Reduced in the Post-Exercise State
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Procedures
2.3. Food Choice Paradigm
2.4. Data Analyses
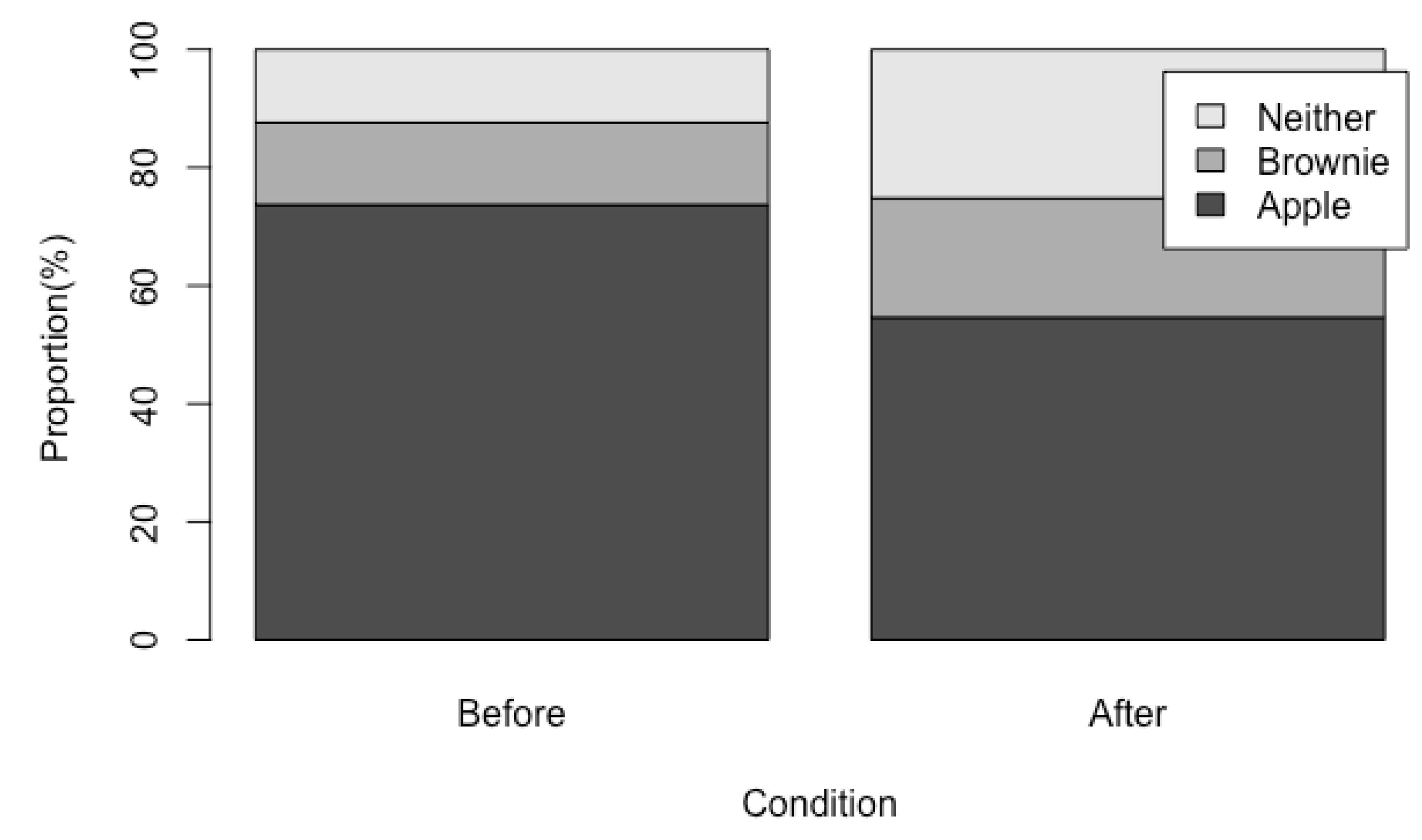
3. Results
4. Discussion
4.1. Interindividual Variation in Post-Exercise Food Choices: Compensatory Eating and Exercise-Induced Anorexia
4.2. Impact of Gender and Body Mass Index
4.3. Limitations
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Cornelissen, V.A.; Smart, N.A. Exercise training for blood pressure: A systematic review and meta-analysis. J. Am. Heart Assoc. 2013, 2, e004473. [Google Scholar] [CrossRef] [PubMed]
- Goodyear, L.J.; Kahn, B.B. Exercise, glucose transport, and insulin sensitivity. Annu. Rev. Med. 1998, 49, 235–261. [Google Scholar] [CrossRef] [PubMed]
- Moore, S.C.; Lee, I.-M.; Weiderpass, E.; Campbell, P.T.; Sampson, J.N.; Kitahara, C.M.; Keadle, S.K.; Arem, H.; Berrington de Gonzalez, A.; Hartge, P.; et al. Association of leisure-time physical activity with risk of 26 types of cancer in 1.44 million adults. JAMA Internal Medicine 2016, 176, 816–825. [Google Scholar] [CrossRef] [PubMed]
- Cox, E.P.; O’Dwyer, N.; Cook, R.; Vetter, M.; Cheng, H.L.; Rooney, K.; O’Connor, H. Relationship between physical activity and cognitive function in apparently healthy young to middle-aged adults: A systematic review. J. Sci. Med. Sport. 2016, 19, 616–628. [Google Scholar] [CrossRef] [PubMed]
- Strohle, A. Physical activity, exercise, depression and anxiety disorders. J. Neural. Transm. (Vienna) 2009, 116, 777–784. [Google Scholar] [CrossRef] [PubMed]
- Simpson, D.; Callisaya, M.L.; English, C.; Thrift, A.G.; Gall, S.L. Self-Reported Exercise Prevalence and Determinants in the Long Term After Stroke: The North East Melbourne Stroke Incidence Study. J. Stroke Cerebrovasc. Dis. 2017, 26, 2855–2863. [Google Scholar] [CrossRef]
- Gutierrez, P.M.; Davidson, C.L.; Friese, A.H.; Forster, J.E. Physical Activity, Suicide Risk Factors, and Suicidal Ideation in a Veteran Sample. Suicide Life Threat Behav. 2016, 46, 284–292. [Google Scholar] [CrossRef] [PubMed]
- Pompili, M.; Venturini, P.; Campi, S.; Seretti, M.E.; Montebovi, F.; Lamis, D.A.; Serafini, G.; Amore, M.; Girardi, P. Do Stroke Patients have an Increased Risk of Developing Suicidal Ideation or Dying by Suicide? An Overview of the Current Literature. CNS Neurosci. Ther. 2012, 18, 711–721. [Google Scholar] [CrossRef] [PubMed]
- Dombrowski, S.U.; Knittle, K.; Avenell, A.; Araujo-Soares, V.; Sniehotta, F.F. Long term maintenance of weight loss with non-surgical interventions in obese adults: Systematic review and meta-analyses of randomised controlled trials. BMJ 2014, 348, g2646. [Google Scholar] [CrossRef]
- Schubert, M.M.; Desbrow, B.; Sabapathy, S.; Leveritt, M. Acute exercise and subsequent energy intake. A meta-analysis. Appetite 2013, 63, 92–104. [Google Scholar] [CrossRef] [PubMed]
- Stein, A.T.; Greathouse, L.J.; Otto, M.W. Eating in response to exercise cues: Role of self-control fatigue, exercise habits, and eating restraint. Appetite 2016, 96, 56–61. [Google Scholar] [CrossRef] [PubMed]
- Bellisle, F. Food choice, appetite and physical activity. Public Health Nutr. 1999, 2, 357–361. [Google Scholar] [CrossRef] [PubMed]
- Moshier, S.J.; Landau, A.J.; Hearon, B.A.; Stein, A.T.; Greathouse, L.; Smits, J.A.J.; Otto, M.W. The Development of a Novel Measure to Assess Motives for Compensatory Eating in Response to Exercise: The CEMQ. Behav. Med. 2016, 42, 93–104. [Google Scholar] [CrossRef] [PubMed]
- Blundell, J.E.; Gibbons, C.; Caudwell, P.; Finlayson, G.; Hopkins, M. Appetite control and energy balance: Impact of exercise. Obes. Rev. 2015, 16, 67–76. [Google Scholar] [CrossRef] [PubMed]
- Thomas, D.M.; Bouchard, C.; Church, T.; Slentz, C.; Kraus, W.E.; Redman, L.M.; Martin, C.K.; Silva, A.M.; Vossen, M.; Westerterp, K. Why do individuals not lose more weight from an exercise intervention at a defined dose? An. energy balance analysis. Obes. Rev. 2012, 13, 835–847. [Google Scholar] [PubMed]
- Blundell, J.E.; King, N.A. Physical activity and regulation of food intake: Current evidence. Med. Sci. Sports Exerc. 1999, 31, S573–S583. [Google Scholar] [CrossRef] [PubMed]
- Vatansever-Ozen, S.; Tiryaki-Sonmez, G.; Bugdayci, G.; Ozen, G. The effects of exercise on food intake and hunger: Relationship with acylated ghrelin and leptin. J. Sports Sci. Med. 2011, 10, 283–291. [Google Scholar] [PubMed]
- Ueda, S.Y.; Yoshikawa, T.; Katsura, Y.; Usui, T.; Nakao, H.; Fujimoto, S. Changes in gut hormone levels and negative energy balance during aerobic exercise in obese young males. The Journal of Endocrinology 2009, 201, 151–159. [Google Scholar] [CrossRef]
- Ueda, S.Y.; Yoshikawa, T.; Katsura, Y.; Usui, T.; Fujimoto, S. Comparable effects of moderate intensity exercise on changes in anorectic gut hormone levels and energy intake to high intensity exercise. J. Endocrinol. 2009, 203, 357–364. [Google Scholar] [CrossRef]
- Blundell, J.E.; Stubbs, R.J.; Hughes, D.A.; Whybrow, S.; King, N.A. Cross talk between physical activity and appetite control: Does physical activity stimulate appetite? Proceedings Nutr. Soc. 2003, 62, 651–661. [Google Scholar] [CrossRef]
- Finlayson, G.; Arlotti, A.; Dalton, M.; King, N.; Blundell, J.E. Implicit wanting and explicit liking are markers for trait binge eating. A susceptible phenotype for overeating. Appetite 2011, 57, 722–728. [Google Scholar] [CrossRef] [PubMed]
- Finlayson, G.; Bryant, E.; Blundell, J.E.; King, N.A. Acute compensatory eating following exercise is associated with implicit hedonic wanting for food. Physiol. Behav. 2009, 97, 62–67. [Google Scholar] [CrossRef] [PubMed]
- Alonso-Alonso, M.; Woods, S.C.; Pelchat, M.; Grigson, P.S.; Stice, E.; Farooqi, S. Food reward system: Current perspectives and future research needs. Nutr. Rev. 2015, 73, 296–307. [Google Scholar] [CrossRef] [PubMed]
- Singh, M. Mood, food, and obesity. Front Psychol. 2014, 5, 925. [Google Scholar] [CrossRef] [PubMed]
- Evero, N.; Hackett, L.C.; Clark, R.D.; Phelan, S.; Hagobian, T.A. Aerobic exercise reduces neuronal responses in food reward brain regions. J. Appl. Physiol. (1985) 2012, 112, 1612–1619. [Google Scholar] [CrossRef] [PubMed]
- Crabtree, D.R.; Chambers, E.S.; Hardwick, R.M.; Blannin, A.K. The effects of high-intensity exercise on neural responses to images of food. Am. J. Clin. Nutr. 2014, 99, 258–267. [Google Scholar] [CrossRef] [PubMed]
- McNeil, J.; Cadieux, S.; Finlayson, G.; Blundell, J.E.; Doucet, É. The effects of a single bout of aerobic or resistance exercise on food reward. Appetite 2015, 84, 264–270. [Google Scholar] [CrossRef]
- Panek, L.M.; Jones, K.R.; Temple, J.L. Short term aerobic exercise alters the reinforcing value of food in inactive adults. Appetite 2014, 81, 320–329. [Google Scholar] [CrossRef]
- King, J.A.; Wasse, L.K.; Ewens, J.; Crystallis, K.; Emmanuel, J.; Batterham, R.L.; Stensel, D.J. Differential acylated ghrelin, peptide yy3–36, appetite, and food intake responses to equivalent energy deficits created by exercise and food restriction. J. Clin. Endocrinol. Metab. 2011, 96, 1114–1121. [Google Scholar] [CrossRef]
- Read, D.; van Leeuwen, B. Predicting Hunger: The Effects of Appetite and Delay on Choice. Organ Behav. Hum. Decis. Process. 1998, 76, 189–205. [Google Scholar] [CrossRef]
- Ikeda, S.; Kang, M.I.; Ohtake, F. Hyperbolic discounting, the sign effect, and the body mass index. J. Health Econ. 2010, 29, 268–284. [Google Scholar] [CrossRef] [PubMed]
- Laibson, D. Golden Eggs and Hyperbolic Discounting*. Q. J. Econ. 1997, 112, 443–478. [Google Scholar] [CrossRef]
- Loewenstein, G.; Brennan, T.; Volpp, K.G. Asymmetric paternalism to improve health behaviors. JAMA 2007, 298, 2415–2417. [Google Scholar] [CrossRef] [PubMed]
- Schwartz, J.; Mochon, D.; Wyper, L.; Maroba, J.; Patel, D.; Ariely, D. Healthier by Precommitment. Psychol. Sci. 2014, 25, 538–546. [Google Scholar] [CrossRef] [PubMed]
- Barlow, P.; Reeves, A.; McKee, M.; Galea, G.; Stuckler, D. Unhealthy diets, obesity and time discounting: A systematic literature review and network analysis. Obes. Rev. 2016, 17, 810–819. [Google Scholar] [CrossRef] [PubMed]
- Nichols, A.L.; Maner, J.K. The good-subject effect: Investigating participant demand characteristics. J. Gen Psychol. 2008, 135, 151–165. [Google Scholar] [CrossRef] [PubMed]
- Adams, S.A.; Matthews, C.E.; Ebbeling, C.B.; Moore, C.G.; Cunningham, J.E.; Fulton, J.; Hebert, J.R. The effect of social desirability and social approval on self-reports of physical activity. Am. J. Epidemiol. 2005, 161, 389–398. [Google Scholar] [CrossRef]
- Hebert, J.R.; Clemow, L.; Pbert, L.; Ockene, I.S.; Ockene, J.K. Social desirability bias in dietary self-report may compromise the validity of dietary intake measures. Int. J. Epidemiol. 1995, 24, 389–398. [Google Scholar] [CrossRef]
- United States Department of Agriculture Agricultural Research Service. USDA Food Composition Databases 2018. Available online: https://ndb.nal.usda.gov/ndb/ (accessed on 29 October 2018).
- King, N.A.; Burley, V.J.; Blundell, J.E. Exercise-induced suppression of appetite: Effects on food intake and implications for energy balance. Eur. J. Clin. Nutr. 1994, 48, 715–724. [Google Scholar]
- Broom, D.R.; Batterham, R.L.; King, J.A.; Stensel, D.J. Influence of resistance and aerobic exercise on hunger, circulating levels of acylated ghrelin, and peptide yy in healthy males. AJP: Regul. Integr. Comp. Physiol. 2009, 296, R29–R35. [Google Scholar] [CrossRef]
- Van Kleef, E.; Shimizu, M.; Wansink, B. Food compensation: Do exercise ads change food intake? Int. J. Behav. Nutr. Phys. Act. 2011, 8, 6. [Google Scholar] [CrossRef] [PubMed]
- Cobb-Clark, D.A.; Kassenboehmer, S.C.; Schurer, S. Healthy habits: The connection between diet, exercise, and locus of control. J. Econ. Behav. Organ. 2014, 98, 1–28. [Google Scholar] [CrossRef]
- Middleman, A.B.; Vazquez, I.; Durant, R.H. Eating patterns, physical activity, and attempts to change weight among adolescents. J. Adolesc. Health 1998, 22, 37–42. [Google Scholar] [CrossRef]
- Privitera, G.J.; Dickinson, E.K. Control your cravings: Self-controlled food choice varies by eating attitudes, sex, and food type among Division I collegiate athletes. Psychol. Sport Exerc. 2015, 19, 18–22. [Google Scholar] [CrossRef]
- Bryan, A.D.; Rocheleau, C.A. Predicting aerobic versus resistance exercise using the theory of planned behavior. Am. J. Health Behav. 2002, 26, 83–94. [Google Scholar] [CrossRef] [PubMed]
- Ainsworth, B.E.; Haskell, W.L.; Herrmann, S.D.; Meckes, N.; Bassett, D.R., Jr.; Tudor-Locke, C. 2011 Compendium of Physical Activities: A second update of codes and MET values. Med. Sci. Sports Exerc. 2011, 43, 1575–1581. [Google Scholar] [CrossRef] [PubMed]
- Schultes, B.; Ernst, B.; Wilms, B.; Thurnheer, M.; Hallschmid, M. Hedonic hunger is increased in severely obese patients and is reduced after gastric bypass surgery. Am. J. Clin. Nutr. 2010, 92, 277–283. [Google Scholar] [CrossRef]
- Barkeling, B.; King, N.A.; Näslund, E.; Blundell, J.E. Characterization of obese individuals who claim to detect no relationship between their eating pattern and sensations of hunger or fullness. Int. J. Obes. (Lond.) 2007, 31, 435–439. [Google Scholar] [CrossRef] [PubMed]
- Schiff, S.; Amodio, P.; Testa, G.; Nardi, M.; Montagnese, S.; Caregaro, L.; di Pellegrino, G.; Sellitto, M. Impulsivity toward food reward is related to BMI: Evidence from intertemporal choice in obese and normal-weight individuals. Brain Cogn. 2016, 110, 112–119. [Google Scholar] [CrossRef] [PubMed]
- Garland, T.; Schutz, H.; Chappell, M.A.; Keeney, B.K.; Meek, T.H.; Copes, L.E.; Acosta, W.; Drenowatz, C.; Maciel, R.C.; van Dijk, G.; et al. The biological control of voluntary exercise, spontaneous physical activity and daily energy expenditure in relation to obesity: Human and rodent perspectives. J. Exp. Biol. 2011, 214, 206–229. [Google Scholar] [CrossRef] [PubMed]
- Sharifi, N.; Mahdavi, R.; Ebrahimi-Mameghani, M. Perceived Barriers to Weight loss Programs for Overweight or Obese Women. Health Promot. Perspect. 2013, 3, 11–22. [Google Scholar] [PubMed]
- Goonasegaran, A.R.; Nabila, F.N.; Shuhada, N.S. Comparison of the effectiveness of body mass index and body fat percentage in defining body composition. Singapore Med. J. 2012, 53, 403–408. [Google Scholar] [PubMed]
- Pomerleau, M.; Imbeault, P.; Parker, T.; Doucet, E. Effects of exercise intensity on food intake and appetite in women. Am. J. Clin. Nutr. 2004, 80, 1230–1236. [Google Scholar] [CrossRef] [PubMed]
- Gastin, P.B.; Cayzer, C.; Robertson, S.; Dwyer, D. Validity of the ActiGraph GT3X+ and BodyMedia SenseWear Armband to estimate energy expenditure during physical activity and sport. J. Sci. Med. Sport 2018, 21, 291–295. [Google Scholar] [CrossRef] [PubMed]
- Scharff, R.L. Obesity and Hyperbolic Discounting: Evidence and Implications. J. Consum. Policy 2009, 32, 3–21. [Google Scholar] [CrossRef]
- Loewenstein, G.; O’Donoghue, T.; Rabin, M. Projection Bias in Predicting Future Utility*. Q. J. Econ. 2003, 118, 1209–1248. [Google Scholar] [CrossRef]
- De-Magistris, T.; Gracia, A. Assessing Projection Bias in Consumers’ Food Preferences. PLoS ONE 2016, 11, e0146308. [Google Scholar] [CrossRef] [PubMed]
| All Participants (n = 256) | “Before” Condition (n = 137) | “After” Condition (n = 119) | p-Value * | |
|---|---|---|---|---|
| Age | 22.1 ± 3.1 | 22.0 ± 2.9 | 22.1 ± 3.4 | 0.72 |
| Gender (Male) | 135 (52.7%) | 75 (54.7%) | 60 (50.4%) | 0.49 |
| BMI (kg/m2) Underweight/Normal weight Overweight/Obese | 24.7 ± 3.7 148 (57.8%) 108 (42.2%) | 24.8 ± 3.6 75 (54.8%) 62 (45.2%) | 24.6 ± 3.8 73 (62.3%) 46 (38.7%) | 0.55 |
| 0.29 | ||||
| 0.29 | ||||
| Workout Duration (min) | 65.3 ± 22.5 | 67.3 ± 25.5 | 63.0 ± 18.3 | 0.12 |
| Food Consumption (Yes) | 8 (3.1%) | 5 (3.6%) | 3 (2.5%) | 0.60 |
| Aerobic Exercise (Yes) | 172 (67.2%) | 87 (63.5%) | 85 (73.4%) | 0.18 |
| Resistance Exercise (Yes) | 192 (75%) | 102 (74.5%) | 90 (75.6%) | 0.83 |
| Other Exercise (Yes) | 9 (3.5%) | 7 (5.1%) | 2 (1.7%) | 0.13 |
| Apple | Brownie | |||
|---|---|---|---|---|
| OR | 95% CI | OR | 95% CI | |
| Condition: “After” | 0.33 | (0.16–0.66) | 0.64 | (0.26–1.55) |
| BMI Status (>25 kg/m2) | 0.61 | (0.31–1.23) | 0.37 | (0.15–0.92) |
| Age | 0.98 | (0.87–1.10) | 1.09 | (0.96–1.24) |
| Gender (male) | 1.17 | (0.56–2.43) | 1.81 | (0.71–4.64) |
| Workout Duration (min) | 1.00 | (0.98–1.02) | 1.00 | (0.98–1.02) |
| Food Consumption (Yes) | 1.51 | (0.17–13.62) | 1.16 | (0.07–20.00) |
| Aerobic Exercise (Yes) | 1.11 | (0.48–2.59) | 0.85 | (0.30–2.40) |
| Resistance Exercise (Yes) | 1.03 | (0.43–2.43) | 0.91 | (0.30–2.77) |
| Other Exercise (Yes) | 0.47 | (0.04–1.56) | 0.47 | (0.05–4.45) |
| Intercept | 10.65 | (0.53–214.60) | 0.31 | (0.01–9.75) |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gustafson, C.R.; Rakhmatullaeva, N.; Beckford, S.E.; Ammachathram, A.; Cristobal, A.; Koehler, K. Exercise and the Timing of Snack Choice: Healthy Snack Choice is Reduced in the Post-Exercise State. Nutrients 2018, 10, 1941. https://doi.org/10.3390/nu10121941
Gustafson CR, Rakhmatullaeva N, Beckford SE, Ammachathram A, Cristobal A, Koehler K. Exercise and the Timing of Snack Choice: Healthy Snack Choice is Reduced in the Post-Exercise State. Nutrients. 2018; 10(12):1941. https://doi.org/10.3390/nu10121941
Chicago/Turabian StyleGustafson, Christopher R., Nigina Rakhmatullaeva, Safiya E. Beckford, Ajai Ammachathram, Alexander Cristobal, and Karsten Koehler. 2018. "Exercise and the Timing of Snack Choice: Healthy Snack Choice is Reduced in the Post-Exercise State" Nutrients 10, no. 12: 1941. https://doi.org/10.3390/nu10121941
APA StyleGustafson, C. R., Rakhmatullaeva, N., Beckford, S. E., Ammachathram, A., Cristobal, A., & Koehler, K. (2018). Exercise and the Timing of Snack Choice: Healthy Snack Choice is Reduced in the Post-Exercise State. Nutrients, 10(12), 1941. https://doi.org/10.3390/nu10121941






