Daily Yogurt Consumption Improves Glucose Metabolism and Insulin Sensitivity in Young Nondiabetic Japanese Subjects with Type-2 Diabetes Risk Alleles
Abstract
1. Introduction
2. Materials and Methods
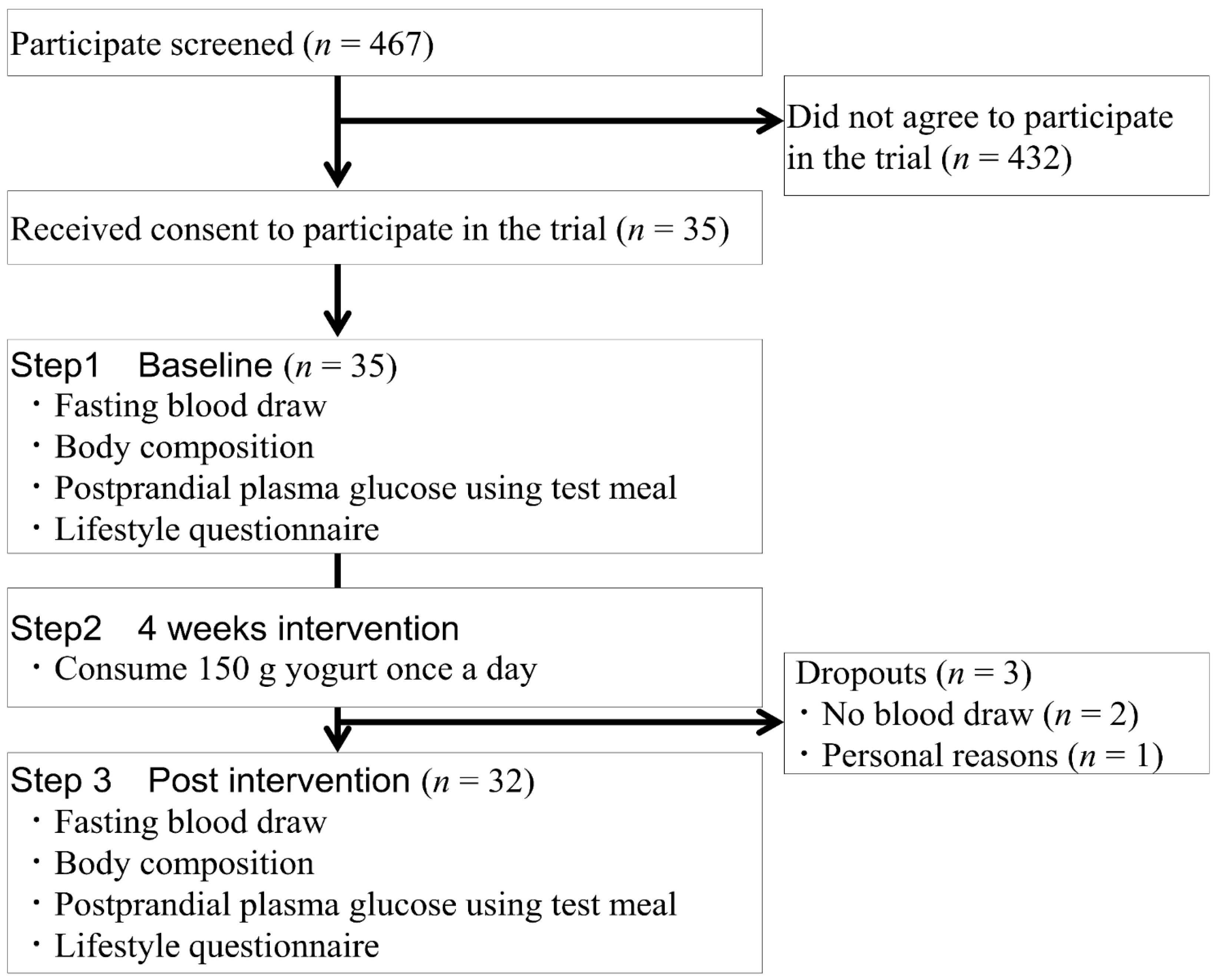
2.1. Study Participants
2.2. Study Design
2.3. Study Procedure
2.4. DNA Extraction and Genotyping
2.5. Study Outcomes
2.6. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Shi, Z.; Zhen, S.; Qi, L.; Zhou, Y.; Taylor, A.W. Association between rice intake and all-cause mortality among Chinese adults: Findings from the Jiangsu Nutrition Study. Asia Pac. J. Clin. Nutr. 2017, 26, 1152–1157. [Google Scholar] [CrossRef] [PubMed]
- Kodama, K.; Tojjar, D.; Yamada, S.; Toda, K.; Patel, C.J.; Butte, A.J. Ethnic differences in the relationship between insulin sensitivity and insulin response: A systematic review and meta-analysis. Diabetes Care 2013, 36, 1789–1796. [Google Scholar] [CrossRef] [PubMed]
- Aune, D.; Norat, T.; Romundstad, P.; Vatten, L.J. Dairy products and the risk of type 2 diabetes: A systematic review and dose-response meta-analysis of cohort studies. Am. J. Clin. Nutr. 2013, 98, 1066–1083. [Google Scholar] [CrossRef] [PubMed]
- Pereira, M.A.; Jacobs, D.R., Jr.; Van Horn, L.; Slattery, M.L.; Kartashov, A.I.; Ludwig, D.S. Dairy consumption, obesity, and the insulin resistance syndrome in young adults: The CARDIA Study. JAMA 2002, 287, 2081–2089. [Google Scholar] [CrossRef] [PubMed]
- Kimura, I.; Ozawa, K.; Inoue, D.; Imamura, T.; Kimura, K.; Maeda, T.; Terasawa, K.; Kashihara, D.; Hirano, K.; Tani, T.; et al. The gut microbiota suppresses insulin-mediated fat accumulation via the short-chain fatty acid receptor GPR43. Nat. Commun. 2013, 4, 1829. [Google Scholar] [CrossRef] [PubMed]
- Bergholdt, H.K.; Nordestgaard, B.G.; Ellervik, C. Milk intake is not associated with low risk of diabetes or overweight-obesity: A Mendelian randomization study in 97,811 Danish individuals. Am. J. Clin. Nutr. 2015, 102, 487–496. [Google Scholar] [CrossRef] [PubMed]
- Grarup, N.; Andersen, G. Gene-environment interactions in the pathogenesis of type 2 diabetes and metabolism. Curr. Opin. Clin. Nutr. Metab. Care 2007, 10, 420–426. [Google Scholar] [CrossRef] [PubMed]
- Wareham, N.J.; Franks, P.W.; Harding, A.H. Establishing the role of gene-environment interactions in the etiology of type 2 diabetes. Endocrinol. Metab. Clin. North. Am. 2002, 31, 553–566. [Google Scholar] [CrossRef]
- Matsuba, R.; Sakai, K.; Imamura, M.; Tanaka, Y.; Iwata, M.; Hirose, H.; Kaku, K.; Maegawa, H.; Watada, H.; Tobe, K.; et al. Replication Study in a Japanese Population to Evaluate the Association between 10 SNP Loci, Identified in European Genome-Wide Association Studies, and Type 2 Diabetes. PLoS ONE 2015, 10, e0126363. [Google Scholar] [CrossRef] [PubMed]
- Yamauchi, T.; Hara, K.; Maeda, S.; Yasuda, K.; Takahashi, A.; Horikoshi, M.; Nakamura, M.; Fujita, H.; Grarup, N.; Cauchi, S.; et al. A genome-wide association study in the Japanese population identifies susceptibility loci for type 2 diabetes at UBE2E2 and C2CD4A-C2CD4B. Nat. Genet. 2010, 42, 864–868. [Google Scholar] [CrossRef] [PubMed]
- Staiger, H.; Machicao, F.; Fritsche, A.; Haring, H.U. Pathomechanisms of type 2 diabetes genes. Endocr. Rev. 2009, 30, 557–585. [Google Scholar] [CrossRef] [PubMed]
- McCarthy, M.I. Genomics, type 2 diabetes, and obesity. N. Engl. J. Med. 2010, 363, 2339–2350. [Google Scholar] [CrossRef] [PubMed]
- Hart, L.M.; Simonis-Bik, A.M.; Nijpels, G.; van Haeften, T.W.; Schafer, S.A.; Houwing-Duistermaat, J.J.; Boomsma, D.I.; Groenewoud, M.J.; Reiling, E.; van Hove, E.C.; et al. Combined risk allele score of eight type 2 diabetes genes is associated with reduced first-phase glucose-stimulated insulin secretion during hyperglycemic clamps. Diabetes 2010, 59, 287–292. [Google Scholar] [CrossRef] [PubMed]
- Imamura, M.; Shigemizu, D.; Tsunoda, T.; Iwata, M.; Maegawa, H.; Watada, H.; Hirose, H.; Tanaka, Y.; Tobe, K.; Kaku, K.; et al. Assessing the clinical utility of a genetic risk score constructed using 49 susceptibility alleles for type 2 diabetes in a Japanese population. J. Clin. Endocrinol. Metab. 2013, 98, E1667–E1673. [Google Scholar] [CrossRef] [PubMed]
- Qi, L.; Cornelis, M.C.; Zhang, C.; van Dam, R.M.; Hu, F.B. Genetic predisposition, Western dietary pattern, and the risk of type 2 diabetes in men. Am. J. Clin. Nutr. 2009, 89, 1453–1458. [Google Scholar] [CrossRef] [PubMed]
- Mattei, J.; Qi, Q.; Hu, F.B.; Sacks, F.M.; Qi, L. TCF7L2 genetic variants modulate the effect of dietary fat intake on changes in body composition during a weight-loss intervention. Am. J. Clin. Nutr. 2012, 96, 1129–1136. [Google Scholar] [CrossRef] [PubMed]
- Gjesing, A.P.; Kjems, L.L.; Vestmar, M.A.; Grarup, N.; Linneberg, A.; Deacon, C.F.; Holst, J.J.; Pedersen, O.; Hansen, T. Carriers of the TCF7L2 rs7903146 TT genotype have elevated levels of plasma glucose, serum proinsulin and plasma gastric inhibitory polypeptide (GIP) during a meal test. Diabetologia 2011, 54, 103–110. [Google Scholar] [CrossRef] [PubMed]
- Villareal, D.T.; Robertson, H.; Bell, G.I.; Patterson, B.W.; Tran, H.; Wice, B.; Polonsky, K.S. TCF7L2 variant rs7903146 affects the risk of type 2 diabetes by modulating incretin action. Diabetes 2010, 59, 479–485. [Google Scholar] [CrossRef] [PubMed]
- Schafer, S.A.; Tschritter, O.; Machicao, F.; Thamer, C.; Stefan, N.; Gallwitz, B.; Holst, J.J.; Dekker, J.M.; Hart, L.M.; Nijpels, G.; et al. Impaired glucagon-like peptide-1-induced insulin secretion in carriers of transcription factor 7-like 2 (TCF7L2) gene polymorphisms. Diabetologia 2007, 50, 2443–2450. [Google Scholar] [CrossRef] [PubMed]
- Villegas, R.; Goodloe, R.J.; McClellan, B.E., Jr.; Boston, J.; Crawford, D.C. Gene-carbohydrate and gene-fiber interactions and type 2 diabetes in diverse populations from the National Health and Nutrition Examination Surveys (NHANES) as part of the Epidemiologic Architecture for Genes Linked to Environment (EAGLE) study. BMC Genet. 2014, 15, 69. [Google Scholar] [CrossRef] [PubMed]
- Danquah, I.; Othmer, T.; Frank, L.K.; Bedu-Addo, G.; Schulze, M.B.; Mockenhaupt, F.P. The TCF7L2 rs7903146 (T) allele is associated with type 2 diabetes in urban Ghana: A hospital-based case-control study. BMC Med. Genet. 2013, 14, 96. [Google Scholar] [CrossRef] [PubMed]
- Yin, X.; Cheng, H.; Lin, Y.; Wineinger, N.E.; Zhou, F.; Sheng, Y.; Yang, C.; Li, P.; Li, F.; Shen, C.; et al. A weighted polygenic risk score using 14 known susceptibility variants to estimate risk and age onset of psoriasis in Han Chinese. PLoS ONE 2015, 10, e0125369. [Google Scholar] [CrossRef] [PubMed]
- Matthews, D.R.; Hosker, J.P.; Rudenski, A.S.; Naylor, B.A.; Treacher, D.F.; Turner, R.C. Homeostasis model assessment: Insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia 1985, 28, 412–419. [Google Scholar] [CrossRef] [PubMed]
- Utzschneider, K.M.; Prigeon, R.L.; Faulenbach, M.V.; Tong, J.; Carr, D.B.; Boyko, E.J.; Leonetti, D.L.; McNeely, M.J.; Fujimoto, W.Y.; Kahn, S.E. Oral disposition index predicts the development of future diabetes above and beyond fasting and 2-h glucose levels. Diabetes Care 2009, 32, 335–341. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, S.; Honda, S.; Murakami, K.; Sasaki, S.; Okubo, H.; Hirota, N.; Notsu, A.; Fukui, M.; Date, C. Both comprehensive and brief self-administered diet history questionnaires satisfactorily rank nutrient intakes in Japanese adults. J. Epidemiol. 2012, 22, 151–159. [Google Scholar] [CrossRef] [PubMed]
- Craig, C.L.; Marshall, A.L.; Sjostrom, M.; Bauman, A.E.; Booth, M.L.; Ainsworth, B.E.; Pratt, M.; Ekelund, U.; Yngve, A.; Sallis, J.F.; et al. International physical activity questionnaire: 12-country reliability and validity. Med. Sci. Sports Exerc. 2003, 35, 1381–1395. [Google Scholar] [CrossRef] [PubMed]
- Mahabir, S.; Baer, D.J.; Giffen, C.; Clevidence, B.A.; Campbell, W.S.; Taylor, P.R.; Hartman, T.J. Comparison of energy expenditure estimates from 4 physical activity questionnaires with doubly labeled water estimates in postmenopausal women. Am. J. Clin. Nutr. 2006, 84, 230–236. [Google Scholar] [CrossRef] [PubMed]
- Kommoju, U.J.; Maruda, J.; Kadarkarai, S.; Irgam, K.; Kotla, J.P.; Velaga, L.; Mohan Reddy, B. No detectable association of IGF2BP2 and SLC30A8 genes with type 2 diabetes in the population of Hyderabad, India. Meta. Gene 2013, 1, 15–23. [Google Scholar] [CrossRef] [PubMed]
- Lyssenko, V.; Almgren, P.; Anevski, D.; Perfekt, R.; Lahti, K.; Nissen, M.; Isomaa, B.; Forsen, B.; Homstrom, N.; Saloranta, C.; et al. Predictors of and longitudinal changes in insulin sensitivity and secretion preceding onset of type 2 diabetes. Diabetes 2005, 54, 166–174. [Google Scholar] [CrossRef] [PubMed]
- Groenewoud, M.J.; Dekker, J.M.; Fritsche, A.; Reiling, E.; Nijpels, G.; Heine, R.J.; Maassen, J.A.; Machicao, F.; Schafer, S.A.; Haring, H.U.; et al. Variants of CDKAL1 and IGF2BP2 affect first-phase insulin secretion during hyperglycaemic clamps. Diabetologia 2008, 51, 1659–1663. [Google Scholar] [CrossRef] [PubMed]
- Rideout, T.C.; Marinangeli, C.P.; Martin, H.; Browne, R.W.; Rempel, C.B. Consumption of low-fat dairy foods for 6 months improves insulin resistance without adversely affecting lipids or bodyweight in healthy adults: A randomized free-living cross-over study. Nutr. J. 2013, 12, 56. [Google Scholar] [CrossRef] [PubMed]
- Akter, S.; Kurotani, K.; Nanri, A.; Pham, N.M.; Sato, M.; Hayabuchi, H.; Mizoue, T. Dairy consumption is associated with decreased insulin resistance among the Japanese. Nutr. Res. 2013, 33, 286–292. [Google Scholar] [CrossRef] [PubMed]
- Stancliffe, R.A.; Thorpe, T.; Zemel, M.B. Dairy attentuates oxidative and inflammatory stress in metabolic syndrome. Am. J. Clin. Nutr. 2011, 94, 422–430. [Google Scholar] [CrossRef] [PubMed]
- Miele, L.; Giorgio, V.; Alberelli, M.A.; De Candia, E.; Gasbarrini, A.; Grieco, A. Impact of Gut Microbiota on Obesity, Diabetes, and Cardiovascular Disease Risk. Curr. Cardiol. Rep. 2015, 17, 120. [Google Scholar] [CrossRef] [PubMed]
- Tilg, H.; Moschen, A.R. Microbiota and diabetes: An evolving relationship. Gut 2014, 63, 1513–1521. [Google Scholar] [CrossRef] [PubMed]
- Klein, A.; Friedrich, U.; Vogelsang, H.; Jahreis, G. Lactobacillus acidophilus 74-2 and Bifidobacterium animalis subsp lactis DGCC 420 modulate unspecific cellular immune response in healthy adults. Eur. J. Clin. Nutr. 2008, 62, 584–593. [Google Scholar] [CrossRef] [PubMed]
- Wolever, T.M.; Brighenti, F.; Royall, D.; Jenkins, A.L.; Jenkins, D.J. Effect of rectal infusion of short chain fatty acids in human subjects. Am. J. Gastroenterol. 1989, 84, 1027–1033. [Google Scholar] [PubMed]
- Brown, A.J.; Goldsworthy, S.M.; Barnes, A.A.; Eilert, M.M.; Tcheang, L.; Daniels, D.; Muir, A.I.; Wigglesworth, M.J.; Kinghorn, I.; Fraser, N.J.; et al. The Orphan G protein-coupled receptors GPR41 and GPR43 are activated by propionate and other short chain carboxylic acids. J. Biol. Chem. 2003, 278, 11312–11319. [Google Scholar] [CrossRef] [PubMed]
| L-GRS (n = 17) | H-GRS (n = 15) | All (n = 32) | p Value | |
|---|---|---|---|---|
| Age (years) b | 20.5 ± 0.8 | 20.7 ± 1.0 | 20.3 ± 0.6 | 0.665 |
| % men (%) c | 11.8 | 6.7 | 9.4 | 0.618 |
| Current smoker (%) c | 0 | 0 | 0 | 1.000 |
| Height (cm) b | 161.8 ± 5.6 | 159.9 ± 6.5 | 160.9 ± 6.0 | 0.382 |
| Weight (kg) b | 55.2 ± 6.1 | 52.5 ± 12.7 | 54.0 ± 9.8 | 0.447 |
| BMI (kg/m2) b,d | 21.1 ± 2.0 | 20.4 ± 3.9 | 20.8 ± 3.0 | 0.534 |
| Skeletal muscle mass (kg) b | 40.1 ± 5.5 | 37.2 ± 7.2 | 38.8 ± 6.3 | 0.196 |
| Body fat mass (kg) b | 14.3 ± 3.3 | 13.0 ± 7.0 | 13.7 ± 5.4 | 0.507 |
| Waist circumference (cm) b | 74.2 ± 4.3 | 72.6 ± 10.9 | 73.4 ± 8.1 | 0.562 |
| Family history of diabetes (%) c | 41.2 | 26.7 | 34.4 | 0.386 |
| Weighted genetic risk score b | 0.85 ± 0.25 | 1.31 ± 0.13 | 1.07 ± 0.20 | <0.001 |
| Genetic risk score b | 4.6 ± 1.4 | 7.1 ± 0.8 | 5.8 ± 1.2 | <0.001 |
| KCNQ1 (rs2237892) c,e | ||||
| C/C | 2 (11.8) | 9 (60.0) | 11 (34.4) | 0.012 |
| C/T | 11 (64.7) | 5 (33.3) | 16 (50.0) | |
| T/T | 4 (23.5) | 1 (6.7) | 5 (15.6) | |
| CDKAL1 (rs2206734) c,e | ||||
| T/T | 1 (5.9) | 5 (33.3) | 6 (18.8) | 0.096 |
| T/C | 9 (52.9) | 7 (46.7) | 16 (50.0) | |
| C/C | 7 (41.2) | 3 (20.0) | 10 (31.2) | |
| CDKN2B (rs2383208) c,e | ||||
| A/A | 1 (5.9) | 9 (60.0) | 10 (31.2) | <0.001 |
| A/G | 12 (70.6) | 6 (40.0) | 18 (56.3) | |
| G/G | 4 (23.5) | 0 (0.0) | 4 (12.5) | |
| UBE2E2 (rs6780569) c,e | ||||
| G/G | 11 (64.7) | 12 (80.0) | 23 (71.9) | 0.405 |
| G/A | 5 (29.4) | 3 (20.0) | 8 (25.0) | |
| A/A | 1 (5.9) | 0 (0.0) | 1 (3.1) | |
| IGF2BP2 (rs1470579) c,e | ||||
| C/C | 2 (11.7) | 4 (26.7) | 6 (18.7) | 0.384 |
| C/A | 7 (41.2) | 7 (46.7) | 14 (43.8) | |
| A/A | 8 (47.1) | 4 (26.6) | 12 (37.5) | |
| Baseline a | Intervention a | Mean Difference b | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| L-GRS | H-GRS | p Value | L-GRS | H-GRS | p Value | L-GRS | p Value | H-GRS | p Value | |
| FPG (mg/dL) | 80.6 ± 1.8 | 81.7 ± 2.2 | 0.565 | 82.1 ± 2.6 | 80.0 ± 3.1 | 0.950 | 1.5 (−1.0 to 4.0) | 0.228 | −1.7 (−5.7 to 2.3) | 0.388 |
| FSI (μg/mL) | 5.43 ± 0.94 | 5.51 ± 1.17 | 0.401 | 4.82 ± 1.03 | 3.95 ± 1.23 | 0.484 | −0.61 (−2.26 to 1.05) | 0.448 | −1.55 (−0.40 to −2.71) | 0.012 |
| Glu-AUC0–30 min (mg/dL.min) | 944 ± 141 | 1338 ± 176 | 0.003 | 1062 ± 187 | 1116 ± 223 | 0.731 | 118 (−88 to 324) | 0.243 | −222 (−506 to 63) | 0.117 |
| Glu-AUC30–120 min (mg/dL.min) | .404 ± 94 | 837 ± 117 | <0.001 | 452 ± 156 | 600 ± 186 | 0.269 | 48 (−91 to 187) | 0.473 | −236 (−455 to −18) | 0.036 |
| Glu-AUC0–120 min (mg/dL.min) | 1348 ± 199 | 2175 ± 248 | <0.001 | 1514 ± 313 | 1717 ± 374 | 0.448 | −166 (−152 to 484) | 0.284 | −458 (−902 to −15) | 0.044 |
| Ins-AUC0–30 min (μg/mL.min) | 358 ± 123 | 76 ± 153 | 0.003 | 228 ± 103 | 68 ± 123 | 0.072 | −130 (−306 to 45) | 0.135 | −8 (−76 to 60) | 0.802 |
| Ins-AUC30–120 min (μg/mL.min) | 213 ± 138 | 69 ± 172 | 0.127 | 222 ± 144 | 26 ± 172 | 0.189 | 9 (−339 to 356) | 0.959 | −43 (−83 to −3) | 0.039 |
| Ins-AUC0–120 min (μg/mL.min) | 571 ± 253 | 145 ± 316 | 0.020 | 450 ± 183 | 94 ± 218 | 0.044 | −122 (−564 to 320) | 0.568 | −51 (−148 to 46) | 0.278 |
| HbA1c (%) | 5.1 ± 0.1 | 5.1 ± 0.1 | 0.481 | 5.1 ± 0.1 | 5.2 ± 0.1 | 0.192 | −0.02 (−0.08 to 0.04) | 0.548 | 0.03 (−0.03 to 0.08) | 0.334 |
| TC (mg/dL) | 173 ± 13 | 196 ± 14 | 0.005 | 165 ± 10 | 185 ± 10 | 0.010 | −8 (−15 to −1) | 0.032 | −11 (−21 to −1) | 0.028 |
| HDL-C (mg/dL) | 65 ± 5 | 72 ± 5 | 0.109 | 64 ± 4 | 69 ± 4 | 0.229 | −1 (−6 to 3) | 0.499 | −4 (−8 to 1) | 0.086 |
| LDL-C (mg/dL) | 95 ± 10 | 113 ± 11 | 0.006 | 90 ± 8 | 105 ± 8 | 0.019 | −5 (−10 to 0) | 0.055 | −8 (−15 to −1) | 0.029 |
| TG (mg/dL) | 70 ± 11 | 64 ± 11 | 0.536 | 56 ± 12 | 63 ± 11 | 0.295 | −14 (−21 to −7) | <0.001 | −1 (−17 to 15) | 0.903 |
| SBP (mmHg) | 98 ± 5 | 97 ± 6 | 0.693 | 97 ± 5 | 95 ± 5 | 0.715 | −0.4 (−3.6 to 2.9) | 0.822 | −1.1 (−6.3 to 4.0) | 0.644 |
| DBP (mmHg) | 54 ± 5 | 53 ± 5 | 0.812 | 55 ± 4 | 52 ± 4 | 0.309 | 1.7 (−1.5 to 4.9) | 0.281 | −0.4 (−7.5 to 6.7) | 0.905 |
| HOMA-IR c | 1.09 ± 0.20 | 1.14 ± 0.25 | 0.490 | 0.99 ± 0.22 | 0.80 ± 0.27 | 0.495 | −0.10 (−0.44 to 0.25) | 0.568 | −0.34 (−0.61 to −0.07) | 0.016 |
| HOMA-β d | 115 ± 18 | 104 ± 22 | 0.162 | 89 ± 29 | 63 ± 34 | 0.962 | −27 (−60 to 7) | 0.110 | −42 (−88 to 5) | 0.076 |
| 1/FSI | 0.23 ± 0.04 | 0.24 ± 0.06 | 0.117 | 0.27 ± 0.09 | 0.33 ± 0.10 | 0.471 | 0.04 (−0.05 to 0.12) | 0.368 | 0.09 (0.15 to 0.03) | 0.012 |
| Insulinogenic Index e | 0.30 ± 0.09 | 0.05 ± 0.11 | <0.001 | 0.22 ± 0.12 | 0.06 ± 0.14 | 0.068 | −0.07 (−0.22 to 0.07) | 0.308 | 0.01 (−0.04 to 0.06) | 0.665 |
| IncAUCins/glu (μg/mg) | 0.21 ± 0.07 | 0.04 ± 0.09 | 0.002 | 0.44 ± 0.28 | 0.05 ± 0.33 | 0.126 | 0.24 (−0.29 to 0.76) | 0.354 | 0.02 (−0.02 to 0.06) | 0.357 |
| Oral disposition index f | 0.46 ± 0.21 | 0.06 ± 0.27 | 0.006 | 0.30 ± 0.15 | 0.06 ± 0.18 | 0.096 | −0.16 (−0.41 to 0.09) | 0.190 | 0.00 (−0.05 to 0.04) | 0.866 |
| Baseline a | Intervention a | Mean Difference b | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| L-GRS | H-GRS | p Value | L-GRS | H-GRS | p Value | L-GRS | p Value | H-GRS | p Value | |
| Energy (kcal/day) | 1458 ± 178 | 1632 ± 203 | 0.175 | 1766 ± 193 | 1607 ± 220 | 0.717 | 308 (89 to 497) | 0.003 | −25 (−240 to 191) | 0.810 |
| Protein (g/day) | 50.5 ± 6.8 | 58.3 ± 7.7 | 0.114 | 68.7 ± 7.0 | 63.1 ± 8.0 | 0.811 | 18.2 (12.2 to 24.1) | <0.001 | 4.8 (−3.0 to 12.6) | 0.208 |
| Fat (g/day) | 46.0 ± 5.7 | 49.7 ± 6.4 | 0.309 | 53.6 ± 6.1 | 49.7 ± 6.9 | 0.788 | 7.6 (1.9 to 13.3) | 0.012 | 0.0 (−7.9 to 8.0) | 0.998 |
| Carbohydrate (g/day) | 194 ± 26 | 221 ± 30 | 0.176 | 231 ± 30 | 214 ± 34 | 0.833 | 37 (10 to 65) | 0.011 | −6 (−37 to 24) | 0.658 |
| Calcium (mg/day) | 302 ± 48 | 384 ± 55 | 0.074 | 782 ± 61 | 735 ± 69 | 0.784 | 480 (377 to 583) | <0.001 | 352 (289 to 415) | <0.001 |
| Weight (kg) | 55.2 ± 2.4 | 52.5 ± 2.8 | 0.524 | 54.8 ± 2.3 | 51.7 ± 2.6 | 0.405 | −0.5 (−0.9 to −0.1) | 0.041 | −0.8 (−1.5 to −0.1) | 0.038 |
| Skeletal muscle mass (kg) | 40.1 ± 1.6 | 37.2 ± 1.8 | 0.141 | 39.5 ± 1.4 | 37.2 ± 1.5 | 0.273 | −0.6 (−2.7 to 1.5) | 0.550 | 0.0 (−0.4 to 0.5) | 0.839 |
| Body fat mass (kg) | 14.3 ± 1.5 | 13.0 ± 1.7 | 0.658 | 12.7 ± 1.5 | 12.2 ± 1.7 | 0.858 | −1.6 (−2.9 to −0.3) | 0.022 | −0.9 (−1.6 to −0.1) | 0.022 |
| Waist circumference (cm) | 74.2 ± 2.1 | 72.6 ± 2.4 | 0.715 | 73.7 ± 2.1 | 71.7 ± 2.4 | 0.612 | −0.6 (−1.4 to 0.2) | 0.151 | −0.8 (−1.8 to 0.1) | 0.079 |
| PAEE (kcal/day) c | 421 ± 108 | 328 ± 123 | 0.789 | 438 ± 165 | 298 ± 188 | 0.546 | .17 (−237 to 271) | 0.891 | −30 (−117 to 58) | 0.478 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Watanabe, D.; Kuranuki, S.; Sunto, A.; Matsumoto, N.; Nakamura, T. Daily Yogurt Consumption Improves Glucose Metabolism and Insulin Sensitivity in Young Nondiabetic Japanese Subjects with Type-2 Diabetes Risk Alleles. Nutrients 2018, 10, 1834. https://doi.org/10.3390/nu10121834
Watanabe D, Kuranuki S, Sunto A, Matsumoto N, Nakamura T. Daily Yogurt Consumption Improves Glucose Metabolism and Insulin Sensitivity in Young Nondiabetic Japanese Subjects with Type-2 Diabetes Risk Alleles. Nutrients. 2018; 10(12):1834. https://doi.org/10.3390/nu10121834
Chicago/Turabian StyleWatanabe, Daiki, Sachi Kuranuki, Akiko Sunto, Naoki Matsumoto, and Teiji Nakamura. 2018. "Daily Yogurt Consumption Improves Glucose Metabolism and Insulin Sensitivity in Young Nondiabetic Japanese Subjects with Type-2 Diabetes Risk Alleles" Nutrients 10, no. 12: 1834. https://doi.org/10.3390/nu10121834
APA StyleWatanabe, D., Kuranuki, S., Sunto, A., Matsumoto, N., & Nakamura, T. (2018). Daily Yogurt Consumption Improves Glucose Metabolism and Insulin Sensitivity in Young Nondiabetic Japanese Subjects with Type-2 Diabetes Risk Alleles. Nutrients, 10(12), 1834. https://doi.org/10.3390/nu10121834





