Whole Fruits and Fruit Fiber Emerging Health Effects
Abstract
1. Introduction
2. Fruit as a Prebiotic Source
2.1. Fruit Fiber Components and Fermentability
2.2. “In Vitro” Human Colonic Microbiota Model System Studies
2.2.1. Pectin
2.2.2. Fruits
2.3. Human Trials
3. Emerging Health Benefits Associated with Whole Fruits and Fruit Fiber
3.1. Gastrointestinal Tract
3.1.1. Constipation
3.1.2. Irritable Bowel Syndrome
3.1.3. Inflammatory Bowel Diseases
Crohn’s Disease (CD)
Ulcerative Colitis (UC)
3.1.4. Diverticular Disease
3.2. Weight Control
3.2.1. Observational Studies
Fruit
Fiber-Rich Diets
3.2.2. Randomized Controlled Trials (RCTs)
Fruits and Vegetables
Fiber-Rich Diets
3.2.3. Mechanisms
Colonic Microbiota
Energy Density (ED)
Satiety and Energy Intake
Metabolizable Energy
3.3. Cardiovascular Disease (CVD)
3.3.1. Fruit and Fruit Fiber
Vascular Aging (Atherosclerosis)
Blood Lipids
Hypertension
3.4. Type 2 Diabetes (Diabetes)
3.4.1. Fruit and Fruit Fiber
Observational Studies
RCTs
3.5. Metabolic Syndrome
3.5.1. Fruit
3.5.2. Fruit Fiber
3.6. Cancer
3.6.1. Colorectal Cancer
3.6.2. Lung Cancer
3.7. Successful Aging
3.7.1. Fruit and Fiber
3.7.2. Mechanisms
3.8. Lung Function
3.8.1. Asthma Severity and Wheezing
3.8.2. Chronic Obstructive Pulmonary Disease (COPD)
3.9. Psychological Well-Being
3.9.1. Children
3.9.2. Adults
3.10. Depression
3.11. Bone Mineral Density
3.11.1. Observational Studies
3.11.2. RCTs
3.12. Seborrheic Dermatitis
3.13. Autism Spectrum Disorder (ASD)
Author Contributions
Funding
Conflicts of Interest
References
- U.S. Department of Health and Human Services and U.S. Department of Agriculture. Dietary Guidelines for Americans 2015–2020. Available online: http://health.gov/dietaryguidelines/ 2015/guidelines/ (accessed on 4 August 2018).
- Slavin, J.C.; Lloyd, B. Health benefits of fruits and vegetables. Adv. Nutr. 2012, 3, 506–512. [Google Scholar] [CrossRef] [PubMed]
- Veronese, N.; Solmi, M.; Caruso, M.G.; Giannelli, M.G.; Osella, A.R.; Evangelou, E.; Maggi, S.; Fontana, L.; Stubbs, B.; Tzoulaki, I. Dietary fiber and health outcomes: An umbrella review of systematic reviews and meta-analyses. Am. J. Clin. Nutr. 2018, 107, 436–444. [Google Scholar] [CrossRef] [PubMed]
- Miller, V.; Mente, A.; Dehghan, S.; Rangarajan, S.; Zhang, X.; Swaminathan, S.; Dagenais, G.; Gupta, R.; Mohan, Y.; Lear, S.; et al. Fruit, vegetable, and legume intake, and cardiovascular disease and deaths in 18 countries (PURE): A prospective cohort study. Lancet 2017, 390, 2037–2049. [Google Scholar] [CrossRef]
- Dreher, M.L. Connection between fiber, colonic microbiota, and health across the human life cycle. In Dietary Fiber in Health and Disease; Springer International Publishing AG: Basel, The Switzerland, 2018; pp. 67–93. [Google Scholar]
- Marlett, J.A.; Cheung, T.-F. Database and quick methods of assessing typical dietary fiber intakes for 228 commonly consumed foods. J. Am. Diet. Assoc. 1997, 97, 1139–1148. [Google Scholar] [CrossRef]
- USDA. National Nutrient Database for Standard Reference; Release Legacy; USDA: Washington, DC, USA, 2018.
- Baker, B.A. Reassessment of some fruit and vegetable pectin levels. J. Food. Sci. 1997, 62, 225–229. [Google Scholar] [CrossRef]
- Padayachee, A.; Day, L.; Howell, K.; Gidley, M.J. Complexity and health functionality of plant cell wall fibers from fruits and vegetables. Crit. Rev. Food Sci. Nutr. 2017, 57, 59–81. [Google Scholar] [CrossRef] [PubMed]
- Byrd-Bredbenner, C.; Ferruzz, M.G.; Fulgoni, V.L.; Murray, R.; Pivonka, E.; Wallace, T.C. Satisfying America’s fruit gap: Summary of an expert roundtable on the role of 100% fruit juice. J. Food Sci. 2017, 82, 1523–1534. [Google Scholar] [CrossRef] [PubMed]
- Burton-Freeman, B.M.; Guenther, P.M.; Oh, M.; Stuart, D.; Jensen, H.H. Assessing the consumption of berries and associated factors in the United States using the National Health and Nutrition Examination Survey (NHANES), 2007–2012. Food Funct. 2018, 9, 1009–1016. [Google Scholar] [CrossRef] [PubMed]
- U.S. HHS; USDA. Part D, Chapter 2: Dietary patterns, foods and nutrients and health outcomes. In 2015–2020 Dietary Guidelines for Americans; Scientific Report; Dietary Guidelines Advisory Committee (DGAC): Washington, DC, USA, 2015; pp. 1–35. [Google Scholar]
- Jew, S.; Abumweis, S.S.; Jones, P.J.H. Evolution of the human diet: Linking our ancestral diet to modern functional foods as a means of disease prevention. J. Med. Foods 2009, 12, 925–934. [Google Scholar] [CrossRef] [PubMed]
- Deehan, E.C.; Walter, J. The fiber gap and disappearing gut microbiome: Implications for human health. Trends Endocrinol. Metab. 2016, 27, 239–241. [Google Scholar] [CrossRef] [PubMed]
- Lie, L.; Brown, L.; Forrester, T.E.; Plange-Rhule, J.; Bovet, P.; Lambert, E.V.; Layden, B.T.; Luke, A.; Dugas, A. The association of dietary fiber intake with cardiometabolic risk in four countries across the epidemiologic transition. Nutrients 2018, 10, 628. [Google Scholar] [CrossRef] [PubMed]
- Rivière, A.; Selak, M.; Lantin, D.; Leroy, F.; De Vuyst, L. Bifidobacteria and butyrate-producing colon bacteria: Importance and strategies for their stimulation in the human gut. Front. Microbiol. 2016, 7, 979. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.; Yu, T.; Huang, G.; Cai, D.; Liang, X.; Su, H.; Zhu, Z.; Li, D.; Yang, Y.; Shen, P.; et al. Gut microbiota community and its assembly associated with age and diet in Chinese centenarians. J. Microbiol. Biotechnol. 2015, 25, 1195–1204. [Google Scholar] [CrossRef] [PubMed]
- Institute of Medicine. Dietary Reference Intakes for Energy, Carbohydrate, Fiber, Fat, Fatty Acids, Cholesterol, Protein, and Amino Acids; National Academies Press: Washington, DC, USA, 2002/2005. [Google Scholar]
- European Food Safety Authority. Scientific opinion on dietary reference values for carbohydrates and dietary fibre. EFSA Panel on Dietetic Products, Nutrition, and Allergies. EFSA J. 2010, 8, 1462. [Google Scholar]
- Dahl, W.J.; Stewart, M.L. Position of the Academy of Nutrition and Dietetics: Health implications of dietary fiber. J. Acad. Nutr. Diet. 2015, 115, 1861–1870. [Google Scholar] [CrossRef] [PubMed]
- Stephen, A.M.; Champ, M.M.-J.; Cloran, S.J. Dietary fibre in Europe: Current state of knowledge on definitions, sources, recommendations, intakes and relationships to health. Nutr. Res. Rev. 2017, 30, 149–190. [Google Scholar] [CrossRef] [PubMed]
- Klurfeld, D.M.; Davis, C.D.; Karp, R.W.; Allen-Vercoe, E.; Chang, E.B.; Chassaing, B.; Fahey, G.V.; Hamaker, B.R.; Holscher, H.D.; Lampe, J.W.; et al. Considerations for best practices in studies of fiber or other dietary components and the intestinal microbiome. Am. J. Physiol. Endocrinol. Metab. 2018. [Google Scholar] [CrossRef] [PubMed]
- Schmier, J.K.; Miller, P.E.; Levine, J.A.; Perez, V.; Maki, K.C.; Rains, T.M.; Devareddy, L.; Sanders, L.M.; Alexander, D.D. Cost savings of reduced constipation rates attributed to increased dietary fiber intakes: A decision-analytic model. BMC Public Health 2014, 14, 374. [Google Scholar] [CrossRef] [PubMed]
- Slavin, J. Fiber and prebiotics: Mechanisms and health benefits. Nutrients 2013, 5, 1417–1435. [Google Scholar] [CrossRef] [PubMed]
- Kashtanova, D.A.; Popenko, A.S.; Tkacheva, O.N.; Tyakht, A.B.; Alexeev, D.G.; Boytsov, S.A. Association between the gut microbiota and diet: Fetal life, early childhood, and further life. Nutrition 2016, 32, 620–627. [Google Scholar] [CrossRef] [PubMed]
- Williams, B.A.; Grant, L.J.; Gidley, M.J.; Mikkelsen, D. Gut fermentation of dietary fibres: Physico-chemistry of plant cell walls and implications for health. Int. J. Mol. Sci. 2017, 18, 2203. [Google Scholar] [CrossRef] [PubMed]
- Low, D.Y.; Williams, B.A.; D’Arcy, B.R.; Flanagan, B.M.; Gidley, M.J. In vitro fermentation of chewed mango and banana: Particle size, starch and vascular fibre effects. Food Funct. 2015, 6, 2464–2474. [Google Scholar] [CrossRef] [PubMed]
- Tuohy, K.M.; Conterno, L.; Gasperotti, M.; Viola, R. Up-regulating the human intestinal microbiome using whole plant foods, polyphenols, and/or fiber. J. Agric. Food Chem. 2012, 60, 8776–8782. [Google Scholar] [CrossRef] [PubMed]
- Tap, J.; Furet, J.P.; Bensaada, M.; Philippe, M.; Roth, H.; Rabot, S.; Lakhdari, O.; Lombard, V.; Henrissat, B.; Corthier, G.; et al. Gut microbiota richness promotes its stability upon increased dietary fibre intake in healthy adults. Environ. Microbiol. 2015, 17, 4954–4964. [Google Scholar] [CrossRef] [PubMed]
- O’Keefe, J.D.; Li, J.V.; Lahti, L.; Ou, J.; Carbonero, F.; Mohammed, K.; Posma, J.M.; Kinross, J.; Wahl, E.; Ruder, E.; et al. Fat, fiber and cancer risk in African Americans and rural Africans. Nat. Commun. 2015, 6, 6342. [Google Scholar] [CrossRef]
- De Filippis, F.; Pellegrini, N.; Vannini, L.; Jeffery, I.B.; La Storia, A.; Laghi, L.; Serrazanetti, D.I.; Di Cagno, R.; Ferrocino, I.; Lazzi, C.; et al. High-level adherence to a Mediterranean diet beneficially impacts the gut microbiota and associated metabolome. Gut 2016, 65, 1812–1821. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Mantrana, I.; Selma-Royo, M.; Alcantara, C.; Collado, M.C. Shifts on gut microbiota associated to Mediterranean Diet adherence and specific dietary intakes on general adult population. Front. Microbiol. 2018, 9, 890. [Google Scholar] [CrossRef] [PubMed]
- Hjorth, M.F.; Beaedel, T.; Bendtsen, L.Q.; Lorenzen, J.K.; Holm, J.B.; Killerich, P.; Roager, H.M.; Kristiansen, K.; Larsen, L.H.; Astrup, A. Prevotella-to-Bacteroides ratio predicts body weight and fat loss success on 24-week diets varying in macronutrient composition and dietary fiber: Results from a post-hoc analysis. Int. J. Obes. (Lond.) 2018. [Google Scholar] [CrossRef] [PubMed]
- Tabernero, M.; Venema, K.; Maathuis, A.J.H.; Saura-Calixto, F.D. Metabolite production during in vitro colonic fermentation of dietary fiber: Analysis and comparison of two European diets. J. Agric. Food Chem. 2011, 59, 8968–8975. [Google Scholar] [CrossRef] [PubMed]
- Klinder, A.; Shen, Q.; Heppel, S.; Lovegrove, J.A.; Rowland, I.; Tuohy, K.M. Impact of increasing fruit and vegetables and flavonoid intake on the human gut microbiota. Food Funct. 2016, 7, 1788–1796. [Google Scholar] [CrossRef] [PubMed]
- Chung, W.S.F.; Meijerink, M.; Zeuner, B.; Holck, J.; Louis, P.; Meyer, A.S.; Wells, J.M.; Flint, H.J.; Duncan, S.H. Prebiotic potential of pectin and pectic oligosaccharides to promote anti-inflammatory commensal bacteria in the human colon. FEMS Microbiol. Ecol. 2017, 93. [Google Scholar] [CrossRef] [PubMed]
- Paniagua, C.; Pose, S.; Morris, V.J.; Kirby, A.R.; Quesada, M.A.; Mercado, J.A. Fruit softening and pectin disassembly: An overview of nanostructural pectin modifications assessed by atomic force microscopy. Ann. Bot. 2014, 114, 1375–1383. [Google Scholar] [CrossRef] [PubMed]
- Langkilde, A.M.; Champ, M.; Andersson, H. Effects of high-resistant-starch banana flour (RS2) on in vitro fermentation and the small-bowel excretion of energy, nutrients, and sterols: An ileostomy study. Am. J. Clin. Nutr. 2002, 75, 104–111. [Google Scholar] [CrossRef] [PubMed]
- Bang, S.-J.; Kim, G.; Young Lim, M.; Song, E.J.; Jung, D.H.; Kum, J.S.; Nam, Y.D.; Park, C.S.; Seo, D.H. The influence of in vitro pectin fermentation on the human fecal microbiome. AMB Expr. 2018, 8, 98. [Google Scholar] [CrossRef] [PubMed]
- Chung, W.S.F.; Walker, A.W.; Louis, P.; Parkhill, J.; Vermeiren, J.; Bosscher, D.; Duncan, S.H.; Flint, H.J. Modulation of the human gut microbiota by dietary fibres occurs at the species level. BMC Boil. 2016, 14, 3. [Google Scholar] [CrossRef] [PubMed]
- Bianchi, F.; Larsen, N.; de Mello Tieghi, T.; Adorno, M.A.T.; Kot, W.; Saad, S.M.I.; Jespersen, L.; Sivieri, K. Modulation of gut microbiota from obese individuals by in vitro fermentation of citrus pectin in combination with Bifidobacterium longum BB-46. Appl. Microbiol. Biotechnol. 2018, 102, 8827–8840. [Google Scholar] [CrossRef] [PubMed]
- Larsen, N.; Cahu, T.B.; Isay Saad, S.M.; Blennow, A.; Jespersen, L. The effects of pectin on survival of probiotic Lactobacillus spp. in gastrointestinal juices is related to their structure and physical properties. Food Microbiol. 2018, 74, 11–20. [Google Scholar] [CrossRef] [PubMed]
- Luis, A.S.; Briggs, J.; Zhang, X.; Farnell, B.; Ndeh, D.; Labourwl, A.; Basle, A.; Cartmell, A.; Terrapon, N.; Stott, K.; et al. Dietary pectic glycans are degraded by coordinated enzyme pathways in human colonic Bacteroides. Nat. Microbiol. 2018, 3, 210–219. [Google Scholar] [CrossRef] [PubMed]
- Barry, J.-L.; Hoebler, C.; MacFarlane, G.T.; MacFarlane, S.; Mathers, J.C.; Reed, K.A.; Mortensen, P.B.; Nordgaard, I.; Rowland, I.R.; Rumney, C.J. Estimation of the fermentability of dietary fibre in vitro: A European interlaboratory study. Br. J. Nutr. 1995, 74, 303–322. [Google Scholar] [CrossRef] [PubMed]
- Condezo-Hoyos, L.; Mohanty, I.P.; Noratto, G.D. Assessing non-digestible compounds in apple cultivars and their potential as modulators of obese faecal microbiota in vitro. Food Chem. 2014, 161, 208–215. [Google Scholar] [CrossRef] [PubMed]
- Simpson, H.L.; Campbell, B.J. Review article: Dietary fibre–microbiota interactions. Aliment. Pharmacol. Ther. 2015, 42, 158–179. [Google Scholar] [CrossRef] [PubMed]
- Vong, M.H.; Stewart, M.L. In vitro bacterial fermentation of tropical fruit fibres. Benef. Microbes 2013, 4, 291–295. [Google Scholar] [CrossRef] [PubMed]
- de Vries, J.; Birkett, A.; Hulshof, T.; Verbeke, K.; Gibes, K. Effects of cereal, fruit and vegetable fibers on human fecal weight and transit time: A comprehensive review of intervention trials. Nutrients 2016, 8, 130. [Google Scholar] [CrossRef] [PubMed]
- Koutsos, A.; Lima, M.; Conterno, L.; Gasperotti, M.; Bianchi, M.; Fava, F.; Vrhovsek, U.; Lovegrove, J.A.; Tuohy, K.M. Effects of commercial apple varieties on human gut microbiota composition and metabolic output using an in vitro colonic model. Nutrients 2017, 9, 533. [Google Scholar] [CrossRef] [PubMed]
- Mandalari, G.; Chessa, S.; Bisignano, C.; Chan, L.; Carughi, A. The effect of sun-dried raisins (Vitis vinifera L.) on the in vitro composition of the gut microbiota. Food Funct 2016, 7, 4048–4060. [Google Scholar] [CrossRef] [PubMed]
- Berding, K.; Holscher, H.D.; Arthur, A.E.; Donovan, S.M. Fecal microbiome composition and stability in 4- to 8-year old children is associated with dietary patterns and nutrient intake. J. Nutr. Biochem. 2018, 56, 165–174. [Google Scholar] [CrossRef] [PubMed]
- Shinohara, K.; Ohashi, Y.; Kawasumi, K.; Terada, A.; Fujisawa, T. Effect of apple intake on fecal microbiota and metabolites in humans. Anaerobe 2010, 16, 510–515. [Google Scholar] [CrossRef] [PubMed]
- Lever, E.; Scott, M.S.; Louis, P.; Emery, P.W.; Whelan, K. The effect of prunes on stool output, gut transit time and gastrointestinal microbiota: A randomised controlled trial. Clin. Nutr. 2018. [Google Scholar] [CrossRef] [PubMed]
- Mitsou, E.K.; Kougia, E.; Normikos, T.; Yannakoulia, M.; Mountzouris, K.C.; Kyriacou, A. Effect of banana consumption on faecal microbiota: A randomized, controlled trial. Anaerobe 2011, 17, 384–387. [Google Scholar] [CrossRef] [PubMed]
- Rabbani, G.H.; Ahmed, S.; Hossain, I.; Islam, R.; Marni, F.; Akhtar, M.; Majid, N. Green banana reduces clinical severity of childhood shigellosis: A double-blind, randomized, controlled clinical trial. Pediatr. Infect. Dis. J. 2009, 28, 420–425. [Google Scholar] [CrossRef] [PubMed]
- Rabbani, G.H.; Teka, T.; Zaman, B.; Majid, N.; Khatun, M.; Fuchs, G.J. Clinical studies in persistent diarrhea: Dietary management with green bananas or pectin in Bangladeshi children. Gastroenterology 2001, 121, 554–560. [Google Scholar] [CrossRef] [PubMed]
- Emery, E.A.; Ahmad, S.; Koethe, J.D.; Skipper, A.; Perlmutter, S.; Paskin, D.L. Banana flakes control diarrhea in enterally fed patients. Nutr. Clin. Pract. 1997, 12, 72–75. [Google Scholar] [CrossRef] [PubMed]
- Alvarez-Acosta, T.; Leon, C.; Acosta-Gonzalez, S.; Parra-Soto, H.; Cluet-Rodriguez, I.; Rossell, M.R.; Colina-Chourio, J.A. Beneficial role of green plantain [Musa paradisiaca] in the management of persistent diarrhea: A prospective randomized trial. J. Am. Coll. Nutr. 2009, 28, 169–176. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.K.; Low, K.Y.; Siah, K.; Drummond, L.M.; Gwee, K.A. Kiwifruit (Actinidia deliciosa) changes intestinal microbial profile. Microb. Ecol. Health Dis. 2012, 23, 18572. [Google Scholar] [CrossRef] [PubMed]
- Eid, N.; Osmanova, H.; Natchez, C.; Walton, G.; Costabile, A.; Gibson, G.; Rowland, I.; Spencer, J.P. Impact of palm date consumption on microbiota growth and large intestinal health: A randomised, controlled, cross-over, human intervention study. Br. J. Nutr. 2015, 114, 1226–1236. [Google Scholar] [CrossRef] [PubMed]
- Forootan, M.; Bagheri, N.; Darvishi, M. Chronic constipation a review of literature. Medicine 2018, 97, 20. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.; Yu, W.; Jiang, J.; Li, N. Clinical benefits after soluble dietary fiber supplementation: A randomized clinical trial in adults with slow-transit constipation. Zhonghua Yi Xue Za Zhi 2014, 94, 3813–3816. [Google Scholar] [PubMed]
- Kelsay, J.L.; Behall, K.M.; Prather, E.S. Effect of fiber from fruits and vegetables on metabolic responses of human subjects. Bowel transit time, number of defecations, fecal weight, urinary excretions of energy and nitrogen and apparent digestibilities of energy, nitrogen, and fat. Am. J. Clin. Nutr. 1978, 31, 1149–1153. [Google Scholar] [CrossRef] [PubMed]
- Venancio, V.P.; Kim, H.; Sirven, M.A.; Ekwe, C.D.; Honvoh, G.; Talcott, S.T.; Mertens-Talcott, S.U. Polyphenol-rich mango (Mangifera indica L.) ameliorate functional constipation symptoms in humans beyond equivalent amount of fiber. Mol. Nutr. Food Res. 2018, 62, 1701034. [Google Scholar] [CrossRef] [PubMed]
- Lever, E.; Cole, J.; Scott, S.M.; Emery, P.W.; Whelan, K. Systematic review: The effect of prunes on gastrointestinal function. Aliment. Pharmacol. Ther. 2014, 40, 750–758. [Google Scholar] [CrossRef] [PubMed]
- Attaluri, A.; Donahoe, R.; Valestin, J.; Brown, K.; Rao, S.S. Randomised clinical trial: Dried plums (prunes) vs. psyllium for constipation. Aliment. Pharmacol. Ther. 2011, 33, 822–828. [Google Scholar] [CrossRef] [PubMed]
- Rush, E.C.; Patel, M.; Plank, L.D.; Ferguson, L.R. Kiwifruit promotes laxation in the elderly. Asia Pac. J. Clin. Nutr. 2002, 11, 164–168. [Google Scholar] [CrossRef] [PubMed]
- Chan, A.O.; Leung, G.; Tong, T.; Wong, N.Y.H. Increasing dietary fiber intake in terms of kiwifruit improves constipation in Chinese patients. World J. Gastroenterol. 2007, 13, 4771–4775. [Google Scholar] [CrossRef] [PubMed]
- Salem, A.E.; Singh, R.; Ayoub, Y.K.; Khairy, A.M.; Mullin, G.E. The gut microbiome and irritable bowel syndrome: State of art review. Arab. J. Gastroenterol. 2018. [Google Scholar] [CrossRef] [PubMed]
- Pozuelo, M.; Panda, S.; Santiago, A.; Mendez, S.; Accarino, A.; Santos, J.; Guarner, F.; Azpiroz, F.; Manichanh, C. Reduction of butyrate- and methane-producing microorganisms in patient with irritable bowel syndrome. Sci. Rep. 2015, 5, 12693. [Google Scholar] [CrossRef] [PubMed]
- El-Salhy, M.; Ystad, S.O.; Mazzawi, T.; Gundersen, D. Dietary fiber in irritable bowel syndrome (review). Int. J. Mol. Med. 2017, 40, 607–613. [Google Scholar] [CrossRef] [PubMed]
- Nagarajana, N.; Mordena, A.; Bischof, D.; King, E.A.; Kosztowski, M.; Wick, E.C.; Stein, E.M. The role of fiber supplementation in the treatment of irritable bowel syndrome: A systematic review and meta-analysis. Eur. J. Gastroenterol. Hepatol. 2015, 27, 1002–1010. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.; Yu, W.; Jiang, J.; Feng, X.; Li, N. Efficacy of pectin in the treatment of diarrhea predominate irritable bowel syndrome. Zhonghua Wei Chang Wai Ke Za Zhi 2015, 18, 267–271. [Google Scholar] [PubMed]
- Chang, C.-C.; Lin, Y.-T.; Lu, Y.-T.; Liu, Y.S.; Liu, J.F. Kiwifruit improves bowel function in patients with irritable bowel syndrome with constipation. Asia Pac. J. Clin. Nutr. 2010, 19, 451–457. [Google Scholar] [PubMed]
- Cozma-Petruţ, A.; Loghin, F.; Miere, D.; Dumitraşcu, D.L. Diet in irritable bowel syndrome: What to recommend, not what to forbid to patients! World J. Gastroenterol. 2017, 23, 3771–3783. [Google Scholar] [CrossRef] [PubMed]
- Haskey, N.; Gibson, D.L. An examination of diet for the maintenance of remission in inflammatory bowel disease. Nutrients 2017, 9, 259. [Google Scholar] [CrossRef] [PubMed]
- Laserna-Mendieta, E.J.; Clooney, A.G.; Carretero-Gomez, J.F.; Moran, C.; Sheehan, D.; Nolan, J.A.; Hill, C.; Gahan, C.G.M.; Joyce, S.A.; Shanahan, F.; et al. Determinants of reduced genetic capacity for butyrate synthesis by the gut microbiome in Crohn’s disease and ulcerative colitis. J. Crohn’s Colitis 2018, 12, 204–216. [Google Scholar] [CrossRef] [PubMed]
- Behzadi, P.; Behzadi, E.; Ranjbar, R. The incidence and prevalence of Crohn’s disease in global scale. SOJ Immunol. 2015, 3, 1–6. [Google Scholar]
- Li, F.; Liu, X.; Wang, W.; Zhang, D. Consumption of vegetables and fruit and the risk of inflammatory bowel disease: A meta-analysis. Eur. J. Gastroenterol. Hepatol. 2015, 27, 623–630. [Google Scholar] [CrossRef] [PubMed]
- Hou, J.K.; Abraham, B.; El-Serag, H. Dietary intake and risk of developing inflammatory bowel disease: A systematic review of the literature. Am. J. Gastroenterol. 2011, 563–573. [Google Scholar] [CrossRef] [PubMed]
- Ananthakrishnan, A.N.; Khalili, H.; Konijeti, G.G.; Higuchi, L.M.; de Silva, P.; Korzenik, J.R.; Fuchs, C.S.; Willett, W.C.; Richter, J.M.; Chan, A.T. A prospective study of long-term intake of dietary fiber and risk of Crohn’s Disease and ulcerative colitis. Gastroenterology 2013, 145, 970–977. [Google Scholar] [CrossRef] [PubMed]
- Amre, D.K.; D’Souza, S.; Morgan, K.; Seidman, G.; Lambrette, P.; Grimard, G.; Israel, D.; Mack, D.; Ghadirian, P.; Deslandres, C.; et al. Imbalances in dietary consumption of fatty acids, vegetables, and fruits are associated with risk for Crohn’s disease in children. Am. J. Gastroenterol. 2007, 102, 2016–2025. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Wu, Y.; Li, F.; Zhang, D. Dietary fiber intake reduces risk of inflammatory bowel disease: Results from a meta-analysis. Nutr. Res. 2015. [Google Scholar] [CrossRef] [PubMed]
- Brotherton, C.S.; Martin, C.A.; Long, M.D.; et al. Avoidance of fiber is associated with greater risk of Crohn’s Disease flare in a 6-month period. Clin. Gastroenterol. Hepatol. 2016, 14, 1130–1136. [Google Scholar] [CrossRef] [PubMed]
- Andersen, V.; Chan, S.; Luben, R.; Khaw, K.T.; Olsen, A.; Tjonneland, A.; Kaaks, R.; Grio, O.; Bergmann, M.M.; Boeing, H.; et al. Fibre intake and the development of inflammatory bowel disease: A European prospective multi-centre cohort study (EPIC-IBD). J. Crohn’s Colitis 2018, 129–136. [Google Scholar] [CrossRef] [PubMed]
- Carabotti, M.; Annibale, B. Treatment of diverticular disease: An update on latest evidence and clinical implications. Drugs Context 2018, 7, 212526. [Google Scholar] [CrossRef] [PubMed]
- Spiller, R.C. Changing views on diverticular disease: Impact of aging, obesity, diet and microbiota. Neurogastroenterol. Motil. 2015, 27, 305–312. [Google Scholar] [CrossRef] [PubMed]
- Barbara, G.; Scaioli, E.; Barbaro, M.R.; Biagi, E.; Laghi, L.; Cremon, C.; Marasco, G.; Colecchia, A.; Picone, G.; Salfi, N.; et al. Gut microbiota, metabolome and immune signatures in patients with uncomplicated diverticular disease. Gut 2017, 66, 1252–1261. [Google Scholar] [CrossRef] [PubMed]
- Mahmood, M.W.; Abraham-Nordling, M.; Hakansson, N.; Wolk, A.; Hjern, F. High intake of dietary fibre from fruits and vegetables reduces the risk of hospitalization for diverticular disease. Eur. J. Nutr. 2018. [Google Scholar] [CrossRef] [PubMed]
- Crowe, F.L.; Balkwill, A.; Cairns, B.J.; Appleby, P.N.; Green, J.; Key, T.J.; Beral, V. Source of dietary fibre and diverticular disease incidence: A prospective study of UK women. Gut 2014, 63, 1450–1456. [Google Scholar] [CrossRef] [PubMed]
- Aldoori, W.; Ryan-Harshman, M. Preventing diverticular disease. Review of recent evidence on high-fibre diets. Can. Fam. Physician 2002, 48, 1632–1637. [Google Scholar] [PubMed]
- Crowe, F.L.; Appleby, P.N.; Allen, N.E.; Key, T.J. Diet and risk of diverticular disease in Oxford cohort of European Prospective Investigation into Cancer and Nutrition (EPIC): Prospective study of British vegetarians and non-vegetarians. BMJ 2011, 343. [Google Scholar] [CrossRef] [PubMed]
- Dahl, C.; Crichton, M.; Jenkins, J.; Nucera, R.; Mahoney, S.; Marx, W.; Marshall, S. Evidence for dietary fibre modification in the recovery and prevention of reoccurrence of acute, uncomplicated diverticulitis: A systematic literature review. Nutrients 2018, 10, 137. [Google Scholar] [CrossRef] [PubMed]
- Schwingshackl, L.; Hoffmann, G.; Kalle-Uhlmann, T.; Arregui, M.; Buijsse, B.; Boeing, H. Fruit and vegetable consumption and changes in anthropometric variables in adult populations: A systematic review and meta-analysis of prospective cohort studies. PLoS ONE 2015, 10, e0140846. [Google Scholar] [CrossRef] [PubMed]
- Sharma, S.P.; Chung, H.J.; Kim, H.J.; Hong, S.T. Paradoxical effects of fruit on obesity. Nutrients 2016, 8, 633. [Google Scholar] [CrossRef] [PubMed]
- Bertoia, M.L.; Mukamal, K.J.; Cahill, L.E.; Hou, T.; Ludwig, D.S.; Mozaffarian, D.; Willett, W.C.; Hu, F.B.; Rimm, E.B. Changes in intake of fruits and vegetables and weight change in United States men and women followed for up to 24 years: Analysis from three prospective cohort studies. PLoS Med. 2015, 12. [Google Scholar] [CrossRef] [PubMed]
- Aljadani, H.M.; Patterson, A.; Sibbritt, D.; Hutchesson, M.J.; Jensen, M.E.; Collins, C.E. Diet quality, measured by fruit and vegetable intake, predicts weight change in young women. J. Obesity 2013, 2013, 525161. [Google Scholar] [CrossRef] [PubMed]
- Rautiainen, S.; Wang, L.; Lee, I.M.; Manson, J.E.; Buring, J.E.; Sesso, H.D. Higher intake of fruit, but not vegetables or fiber, at baseline is associated with lower risk of becoming overweight or obese in middle-aged and older women of normal BMI at baseline. J. Nutr. 2015, 145, 960–968. [Google Scholar] [CrossRef] [PubMed]
- Yuan, S.; Yu, H.J.; Liu, M.W.; Huang, Y.; Yang, X.H.; Tang, B.W.; Song, Y.; Cao, Z.K.; Wu, H.J.; He, Q.O.; et al. The association of fruit and vegetable consumption with changes in weight and body mass index in Chinese adults: A cohort study. Public Heal 2018, 157, 121–126. [Google Scholar] [CrossRef] [PubMed]
- Yu, Z.M.; DeClercq, V.; Cui, Y.; Forbes, C.; Grandy, S.; Keats, M.; Parker, L.; Sweeney, E.; Dummer, T.J.B. Fruit and vegetable intake and body adiposity among populations in Eastern Canada: The Atlantic Partnership for Tomorrow’s Health Study. BMJ Open 2018, 8, e018060. [Google Scholar] [CrossRef] [PubMed]
- Cavallo, D.N.; Horino, M.; McCarthy, W.J. Adult intake of minimally processed fruits and vegetables: Associated with cardiometabolic disease factors. J. Acad. Nutr. Diet. 2016, 116, 1387–1394. [Google Scholar] [CrossRef] [PubMed]
- Auerbach, B.J.; Littman, A.J.; Krieger, J.; Young, B.A.; Larson, J.; Tinker, L.; Neuhouser, M.L. Association of 100% fruit juice consumption and 3-year weight change among postmenopausal women in the Women’s Health Initiative. Prev. Med. 2018, 109, 8–10. [Google Scholar] [CrossRef] [PubMed]
- Fogelholm, M.; Anderssen, S.; Gunnarsdottir, I.; Lahti-Koski, M. Dietary macronutrients, and food consumption as determinants of long-term weight change in adult populations: A systematic literature review. Food Nutr. Res. 2012, 56. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Willett, W.C.; Manson, J.E.; Hu, F.B.; Rosner, B.; Colditz, G. Relation between changes in intakes of dietary fiber nd grain products and changes in weight and development of obesity among middle-aged women. Am. J. Clin. Nutr. 2003, 78, 920–927. [Google Scholar] [CrossRef] [PubMed]
- Tucker, L.A.; Thomas, K.S. Increasing total fiber intake reduces risk of weight and fat gains in women. J. Nutr. 2009, 139, 576–581. [Google Scholar] [CrossRef] [PubMed]
- Grooms, K.N.; Ommerborn, M.J.; Pham, D.Q.; Djousse, L.; Clark, C.R. Dietary fiber intake and cardiometabolic risk among US adults, NHANES 1999–2010. Am. J. Med. 2013, 126, 1059–1067. [Google Scholar] [CrossRef] [PubMed]
- Romaguera, D.; Angquist, L.; Du, H.; Jakobsen, M.U.; Forouhi, N.G.; Halkjaer, J.; Feskens, E.J.; van der A, D.L.; Masala, G.; Steffen, A.; et al. Dietary determinants of changes in waist circumference adjusted for body mass index—A proxy measure of visceral adiposity. PLoS ONE 2010, 5, e11588. [Google Scholar] [CrossRef] [PubMed]
- Hairston, K.G.; Vitolins, M.Z.; Norris, J.M.; Anderson, A.M.; Hanley, A.J.; Wagenknecht, L.E. Lifestyle factors and 5-year abdominal fat accumulation in a minority cohort: The IRAS Family Study. Obesity (Silver Spring) 2012, 20. [Google Scholar] [CrossRef] [PubMed]
- Othman, F.; Nor, N.S.M.; Appannah, G.; Zaki, N.A.M.; Ambak, R.; Omar, A.; Fazliana, M.; Salleh, R.; Yusof, B.N.M.; Muksan, N.; et al. Prediction of body fat loss in relation to change in nutrient intake among housewives participating in the MyBFF@home study. BMC Women’s Heal 2018, 18 (Suppl. 1), 102. [Google Scholar] [CrossRef] [PubMed]
- Kaiser, K.A.; Brown, A.W.; Shikany, J.M.; Shikany, J.M.; Mattes, R.D.; Allison, D.B. Increased fruit and vegetable intake has no discernable effect on weight loss: A systematic review and meta-analysis. Am. J. Clin. Nutr. 2014, 100, 567–576. [Google Scholar] [CrossRef] [PubMed]
- Mytton, O.T.; Nnoaham, K.; Eyles, H.; Scarborough, P.; Ni Mhurchu, C. Systematic review and meta-analysis of the effect of increased vegetable and fruit consumption on body weight and energy intake. BMC Public 2014, 14, 886. [Google Scholar] [CrossRef] [PubMed]
- Sylvetsky, A.C.; Edelstein, S.L.; Walford, G.; Boyko, E.J.; Horton, E.S.; Ibebuogu, U.N.; Knowler, W.C.; Montez, M.G.; Temprosa, M.; Hoskin, M.; et al. A high-carbohydrate, high-fiber, low-fat diet results in weight loss among adults at high risk of type 2 diabetes. J. Nutr. 2017, 147, 2060–2066. [Google Scholar] [CrossRef] [PubMed]
- Champagne, C.M.; Broyles, S.T.; Moran, L.D.; Cash, K.C.; Levy, E.J.; Lin, P.H.; Batch, B.C.; Lien, L.F.; Funk, K.L.; Dalcin, A.; et al. Dietary intakes associated with successful weight loss and maintenance during the Weight Loss Maintenance Trial. J. Am. Diet. Assoc. 2011, 111, 1826–1835. [Google Scholar] [CrossRef] [PubMed]
- De Oliveira, M.C.; Sichieri, R.; Venturim Mozzer, R.A. low-energy-dense diet adding fruit reduces weight and energy intake in women. Appetite 2008, 51, 291–295. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Rasmussen, A.; Woo, S.L.; Yaceczko, S.; Gilbuena, L.V.N.; Thames, G.; Shao, P.; Yang, A.; Henning, S.; Yang, J.; et al. Daily avocado supplementation on weight loss via hypocaloric diet: A prospective randomized trial. Nutrition 2018. American Society for Nutrition Poster 23-062, #689, Boston, MA, USA, presentation June 10, 2018. [Google Scholar]
- Ma, Y.; Olendzki, B.C.; Wang, J.; Persuitte, G.M.; Li, W.; Fang, H.; Merriam, P.A.; Wedick, N.M.; Ockene, I.S.; Culver, A.L.; et al. Single-component versus multi-component dietary goals for the metabolic syndrome: A randomized trial. Ann. Intern. Med. 2015, 162, 248–257. [Google Scholar] [CrossRef] [PubMed]
- Pal, S.; Khossousi, A.; Binns, C.; Dhaliwal, S.; Ellis, V. The effect of a fibre supplement compared to a healthy diet on body composition, lipids, glucose, insulin and other metabolic syndrome risk factors in overweight and obese individuals. Br. J. Nutr. 2011, 105, 90–100. [Google Scholar] [CrossRef] [PubMed]
- Ferdowsian, H.R.; Barnard, N.D.; Hoover, V.I.; Katcher, H.I.; Levin, S.M.; Green, A.A.; Cohen, J.L. A multi-component intervention reduced body weight and cardiovascular risk at a GEICO corporate site. Am. J. Health Promt. 2010, 24, 384–387. [Google Scholar] [CrossRef] [PubMed]
- Lindstrom, J.; Peltonen, M.; Eriksson, J.G.; Louheranta, A.; Fogelholm, M.; Uusitupa, M.; Tuomilehto, J. High-fibre, low-fat diet predicts long-term weight loss and decreased type 2 diabetes risk: The Finnish Diabetes Prevention Study. Diabetologia 2006, 49, 912–920. [Google Scholar] [CrossRef] [PubMed]
- Bajerska, J.; Chmurzynska, A.; Muzsik, A.; Krzyzanowska, P.; Madry, E.; Malinowska, A.M.; Walkowiak, J. Weight loss and metabolic health effects from energy-restricted Mediterranean and Central-European diets in postmenopausal women: A randomized controlled trial. Sci. Reps. 2018, 8, 11170. [Google Scholar] [CrossRef] [PubMed]
- Davis, H.C. Can the gastrointestinal microbiota be modulated by dietary fibre to treat obesity? Ir. J. Med. Sci. 2018, 187, 393–402. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Navarro, T.; Salazar, N.; Gutiérrez-Díaz, I.; de Los Reyes-Gavilan, C.G.; Gueimonde, M.; Gonzalez, S. Different intestinal microbial profile in over-weight and obese subjects consuming a diet with low content of fiber and antioxidants. Nutrients 2017, 9, 551. [Google Scholar] [CrossRef]
- Aguirre, M.; Jonkers, D.M.A.E.; Troost, F.J.; Roeselers, G.; Venema, K. In vitro characterization of the impact of different substrates on metabolite production, energy extraction and composition of gut microbiota from lean and obese subjects. PLoS ONE 2014, 9, e113864. [Google Scholar] [CrossRef] [PubMed]
- Hjorth, M.F.; Roager, H.M.; Larsen, T.M.; Poulsen, S.K.; Licht, T.R.; Bahl, M.I.; Zohar, Y.; Astrup, A. Pre-treatment microbial Prevotella-to-Bacteroides ratio, determines body fat loss success during a 6-month randomized controlled diet intervention. Int. J. Obes. (Lond.) 2018, 42, 580–583. [Google Scholar] [CrossRef] [PubMed]
- Ivan, J.; Major, E.; Sipos, A.; Kovacs, K.; Hovath, D.; Tamas, I.; Bay, P.; Dombradi, V.; Lontay, B. The short-chain fatty acid propionate inhibits adipogenic differentiation of human chorion-derived mesenchymal stem cells through the free fatty acid receptor 2. Stem Cells Dev. 2017, 26, 1724–1733. [Google Scholar] [CrossRef] [PubMed]
- Byrne, C.S.; Chambers, E.S.; Alhabeeb, H.; Chhina, N.; Morrison, D.J.; Preston, T.; Tedford, T.; Fitzpatrick, J.; Irani, C.; Busza, A. Increased colonic propionate reduces anticipatory reward responses in the human striatum to high-energy foods. Am. J. Clin. Nutr. 2016, 104, 5–14. [Google Scholar] [CrossRef] [PubMed]
- Samuel, B.S.; Shaito, A.; Motoike, T.; Rey, F.E.; Backhed, F.; Manchester, J.K.; Hammer, R.E.; Williams, S.C.; Crowley, J.; Yanagisawa, M.; et al. Effects of the gut microbiota on host adiposity are modulated by the short-chain fatty-acid binding G protein-coupled receptor, Gpr41. Proc. Natl. Acad. Sci. USA 2008, 105, 16767–16772. [Google Scholar] [CrossRef] [PubMed]
- Rolls, B.J. Dietary energy density: Applying behavioural science to weight management. Nutr. Bull. 2017, 42, 246–253. [Google Scholar] [CrossRef] [PubMed]
- CDC National Center for Chronic Disease Prevention and Health Promotion. Can eating fruits and vegetables help people to manage their weight? Research to Practice Series; No. 1. Available online: https://www.cdc.gov/nccdphp/dnpa/nutrition/pdf/rtp_practitioner_10_07.pdf (accessed on 6 August 2018).
- Brunstrom, J.M.; Drake, A.C.L.; Forde, C.G.; Rogers, P.J. Undervalued and ignored: Are humans poorly adapted to energy dense foods? Appetite 2018, 120, 589–595. [Google Scholar] [CrossRef] [PubMed]
- Kant, A.K.; Graubard, B.I. Energy density of diets reported by American adults: Association with food group intake, nutrient intake and body weight. Int. J. Obes. (Lond.) 2005, 29, 950–956. [Google Scholar] [CrossRef] [PubMed]
- Vernarelli, J.A.; Mitchell, D.C.; Rolls, B.J.; Hartman, T.J. Dietary energy density and obesity: How consumption patterns differ by body weight status. Eur. J. Nutr. 2018, 57, 351–361. [Google Scholar] [CrossRef] [PubMed]
- Fernando, N.N.T.; Campbell, K.J.; McNaughton, S.A.; Zheng, M.; Lacy, K.E. Predictors of dietary energy density among preschool aged children. Nutrients 2018, 10, 178. [Google Scholar] [CrossRef] [PubMed]
- Raynor, H.A.; Van Walleghen, E.L.; Bachman, J.L. Dietary energy density and successful weight loss maintenance. Eat Behav. 2011, 12, 119–125. [Google Scholar] [CrossRef] [PubMed]
- Wanders, A.J.; van den Borne, J.J.; de Graaf, C.; Hulshof, T.; Jonathan, M.C.; Kristensen, M.; Mars, M.; Schols, H.A.; Feskens, E.J. Effects of dietary fibre on subjective appetite, energy intake and body weight: A systematic review of randomized controlled trials. Obes. Rev. 2011, 12, 724–739. [Google Scholar] [CrossRef] [PubMed]
- Tiwary, C.M.; Ward, J.A.; Jackson, B.A. Effect of pectin on satiety in healthy US Army adults. J. Am. Coll. Nutr. 1997, 16, 423–428. [Google Scholar] [CrossRef] [PubMed]
- Wanders, A.J.; Mars, M.; Borgonjen-van den Berg, K.J.; de Graaf, C.; Feskens, E.J. Satiety and energy intake after single and repeated exposure to gel-forming dietary fiber: Post-ingestive effects. Int. J. Obes. (Lond.) 2014, 38, 794–800. [Google Scholar] [CrossRef] [PubMed]
- Dong, H.; Sargent, L.J.; Chatzidiakou, Y.; Saunders, C.; Harkness, L.; Bordenave, N.; Rowland, I.; Spencer, J.P.E.; Lovegrove, J.A. Orange pomace fibre increases a composite scoring of subjective ratings of hunger and fullness in healthy adults. Appetite 2016, 107, 478–485. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann Sardá, F.A.; Giuntini, E.B.; Gomez, M.L.P.A.; Lui, M.G.Y.; Negrini, J.A.E.; Tadini, C.C.; Lajolo, F.M.; Menezes, E.W. Impact of resistant starch from unripe banana flour on hunger, satiety, and glucose homeostasis in healthy volunteers. J. Funct. Foods 2016, 24, 63–74. [Google Scholar] [CrossRef]
- Rogers, P.J.; Shahrokni, R. A comparison of the satiety effects of a smoothie, its fresh fruit equivalent and other drinks. Nutrients 2018, 10, 431. [Google Scholar] [CrossRef] [PubMed]
- Maljaars, P.W.; Peters, H.P.; Mela, D.J.; Masclee, A.A. Ileal brake: A sensible food target for appetite control. A review. Physiol. Behav. 2008, 95, 271–281. [Google Scholar] [CrossRef] [PubMed]
- Flood-Obbagy, J.E.; Rolls, B.J. The effect of fruit in different forms on energy intake and satiety at a meal. Appetite 2009, 52, 416–422. [Google Scholar] [CrossRef] [PubMed]
- James, L.J.; Funnell, M.P.; Milner, S. An afternoon snack of berries reduces subsequent energy intake compared to an isoenergetic confectionary snack. Appetite 2015, 95, 132–137. [Google Scholar] [CrossRef] [PubMed]
- Patel, B.P.; Bellissimo, N.; Luhovyy, B.; Bennett, L.J.; Hurton, E.; Painter, J.E.; Anderson, G.H. An after-school snack of raisins lowers cumulative food intake in young children. J. Food Sci. 2013, 78, A5–A10. [Google Scholar] [CrossRef] [PubMed]
- Wien, M.; Haddad, E.; Oda, K.; Sabaté, J. A randomized 3×3 crossover study to evaluate the effect of Hass avocado intake on post-ingestive satiety, glucose and insulin levels, and subsequent energy intake in overweight adults. Nutr. J. 2013, 12, 155. [Google Scholar] [CrossRef] [PubMed]
- Haddad, E.; Wien, M.; Oda, K.; Sabate, J. Postprandial gut hormone responses to Hass avocado meals and their association with visual analog scores in overweight adults; a randomized 3x3 crossover trial. Eat Behav. 2018, 31, 35–40. [Google Scholar] [CrossRef] [PubMed]
- Farajian, P.; Katsagani, M.; Zampelas, A. Short-term effects of a snack including dried prunes on energy intake and satiety in normal-weight individuals. Eat Behav. 2010, 11, 201–203. [Google Scholar] [CrossRef] [PubMed]
- Miles, C.W. The metabolizable energy of diets differing in dietary fat and fiber measured in humans. J. Nutr. 1992, 122, 306–311. [Google Scholar] [CrossRef] [PubMed]
- Miles, C.W.; Kelsay, J.L.; Wong, N.P. Effect of dietary fiber on the metabolizable energy of human diets. J. Nutr. 1988, 118, 1075–1081. [Google Scholar] [CrossRef] [PubMed]
- Baer, D.J.; Rumpler, W.V.; Miles, C.W.; Fahey, G.C. Dietary fiber decreases the metabolizable energy content and nutrient digestibility of mixed diets fed to humans. J. Nutr. 1997, 127, 579–586. [Google Scholar] [CrossRef] [PubMed]
- Beyer, P.L.; Flynn, M.A. Effects of high- and low-fiber diets on human feces. J. Am. Diet. Assoc. 1978, 72, 271–277. [Google Scholar] [PubMed]
- Wisker, E.; Feldheim, W. Metabolizable energy of diets low in dietary fiber from fruits and vegetables when consumed by humans. J. Nutr. 1990, 120, 1331–1337. [Google Scholar] [CrossRef] [PubMed]
- Webb, P.; Annis, J.F. Adaptation to overeating in lean and overweight men and women. Hum. Nutr. Clin. Nutr. 1983, 37, 117–131. [Google Scholar] [PubMed]
- Mendis, S.; Puska, P.; Norrving, B. Global Atlas on Cardiovascular Disease Prevention and Control; World Health Organization: Geneva, Switzerland, 2011; Available online: www.who.int/cardiovasculardiseases/publications/atlascvd/en/ (accessed on 6 August 2018).
- Pearson-Stuttard, J.; Bandosz, P.; Rehm, C.D.; Penalvo, J.; Whitsel, L.; Gaziano, T.; Conrad, Z.; Wilde, P.; Micha, R.; Lloyd-Williams, F.; et al. Reducing US cardiovascular disease burden and disparities through national and targeted dietary policies: A modelling study. PLoS Med. 2017, 14. [Google Scholar] [CrossRef] [PubMed]
- Appiah, D.; Capistrant, B.D. Cardiovascular disease risk assessment in the United States and low- and middle-income countries using predicted heart/vascular age. Sci. Rep. 2017, 7, 16673. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.; Dwyer, K.M.; Fan, Z.; Shircore, A.; Fan, J.; Dwyer, J.H. Dietary fiber and progression of atherosclerosis: The Los Angeles Atherosclerosis Study. Am. J. Clin. Nutr. 2003, 78, 1085–1091. [Google Scholar] [CrossRef] [PubMed]
- Juonala, M.; Viikari, J.S.A.; Kahonen, M.; Taittonen, L.; Laitinen, T.; Hutri-Kahonen, N.; Lehtimaki, T.; Jula, A.; Pietikainen, M.; Jokinen, E. Life-time risk factors and progression of carotid atherosclerosis in young adults: The Cardiovascular Risk in Young Finns Study. Eur. Hear. J. 2010, 31, 1745–1751. [Google Scholar] [CrossRef] [PubMed]
- Buil-Cosiales, P.; Martinez-Gonzalez, M.A.; Ruiz-Canela, M.; Diez-Espino, J.; Garcia-Prellano, A.; Toledo, E. Consumption of fruit and fruit fiber decrease the risk of cardiovascular disease in a Mediterranean young cohort. Nutrients 2017, 9, 295. [Google Scholar] [CrossRef] [PubMed]
- Bondonno, N.P.; Lewis, J.R.; Prince, R.L.; Lim, W.H.; Wong, G.; Schousboe, J.T.; Woodman, R.J.; Kiel, D.P.; Bondonno, C.P.; Wars, N.C.; et al. Fruit intake and abdominal aortic calcification in elderly women: A prospective cohort study. Nutrients 2016, 8, 159. [Google Scholar] [CrossRef] [PubMed]
- Tian, X.; Du, H.; Li, L.; Bennett, D.; Gao, R.; Li, S.; Wang, S.; Guo, Y.; Bian, Z.; Yang, L.; et al. Fruit consumption and physical activity in relation to all-cause and cardiovascular mortality among 70,000 Chinese adults with pre-existing vascular disease. PLoS ONE 2017, 12. [Google Scholar] [CrossRef] [PubMed]
- Lai, H.T.M.; Threapleton, D.E.; Day, A.J.; Williamson, G.; Cade, J.E.; Burley, V.J. Fruit intake and cardiovascular disease mortality in the UK Women’s Cohort Study. Eur. J. Epidemiol. 2015, 30, 1035–1048. [Google Scholar] [CrossRef] [PubMed]
- Threapleton, D.E.; Greenwood, D.C.; Evans, C.E.L.; Cleghorn, C.L.; Nykjaer, C.; Woodhead, C.; Cade, J.E.; Gale, C.P.; Burley, V.J. Dietary fibre intake and risk of cardiovascular disease: Systematic review and meta-analysis. BMJ 2013, 347. [Google Scholar] [CrossRef] [PubMed]
- Najjar, R.S.; Moore, C.E.; Montgomery, B.D. A defined, plant-based diet utilized in an outpatient cardiovascular clinic effectively treats hypercholesterolemia and hypertension and reduces medications. Clin. Cardiol. 2018, 41, 307–313. [Google Scholar] [CrossRef] [PubMed]
- Theuwissen, E.; Mensink, R.P. Water-soluble dietary fibers and cardiovascular disease. Physiol. Behav. 2008, 94, 285–292. [Google Scholar] [CrossRef] [PubMed]
- Brown, L.; Rosner, B.; Willett, W.W.; Sacks, F.M. Cholesterol lowering effects of dietary fiber. A meta-analysis. Am. J. Clin. Nutr. 1999, 69, 30–42. [Google Scholar] [CrossRef] [PubMed]
- Brouns, F.; Theuwissen, E.; Adam, A.; Bell, M.; Berger, A.; Mensink, R.P. Cholesterol-lowering properties of different pectin types in mildly hyper-cholesterolemic men and women. Eur. J. Clin. Nutr. 2012, 66, 591–599. [Google Scholar] [CrossRef] [PubMed]
- Ravn-Haren, G.; Dragsted, L.O.; Buch-Andersen, T.; Jensen, E.N.; Jensen, R.I.; Nemeth-Balogh, M.; Paulovicsova, B.; Bergstrom, A.; Wilcks, A.; Licht, T.R.; et al. Intake of whole apples or clear apple juice has contrasting effects on plasma lipids in healthy volunteers. Eur. J. Nutr. 2013, 52, 1875–1889. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Bordi, P.L.; Fleming, J.A.; Hill, A.M.; Kris-Etherton, P.M. Effects of a moderate fat diet with and without avocados on lipoprotein particle number, size and subclasses in overweight and obese adults: A randomized, controlled trial. J. Am. Heart Assoc. 2015, 4, e001355. [Google Scholar] [CrossRef] [PubMed]
- Park, E.; Edirisinghe, I.; Burton-Freeman, B. Avocado fruit on postprandial markers of cardio-metabolic risk: A randomized controlled dose response trial in overweight and obese men and women. Nutrients 2018, 10, 1287. [Google Scholar] [CrossRef] [PubMed]
- Peou, S.; Milliard-Hasting, B.; Shah, S. Impact of avocado-enriched diets on plasma lipoproteins: A meta-analysis. J. Clin. Lipidol. 2016, 10, 161–171. [Google Scholar] [CrossRef] [PubMed]
- Mahmassani, H.A.; Avendano, E.E.; Raman, G.; Johnson, E.J. Avocado consumption and risk factors for heart disease: A systematic review and meta-analysis. Am. J. Clin. Nutr. 2018, 107, 523–536. [Google Scholar] [CrossRef] [PubMed]
- Tinker, L.F.; Schneeman, B.O.; Davis, P.A.; Gallaher, D.D.; Waggoner, C.R. Consumption of prunes as a source of dietary fiber in men with mild hypercholesterolemia. Am. J. Clin. Nutr. 1991, 53, 1259–1265. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.; Chen, G.; Liao, D.; Zhu, Y.; Xue, X. Effects of berries consumption on cardiovascular risk factors: A meta-analysis with trial sequential analysis of randomized controlled trials. Sci. Rep. 2016, 6, 233625. [Google Scholar] [CrossRef] [PubMed]
- Borgi, L.; Muraki, I.; Satija, A.; Willett, W.C.; Rimm, E.B.; Forman, J.P. Fruit and vegetable consumption and the incidence of hypertension in three prospective cohort studies. Hypertension 2016, 67, 288–893. [Google Scholar] [CrossRef] [PubMed]
- Schwingshackl, L.; Schwedhelm, C.; Hoffmann, G.; Knuppel, S.; Iqbal, K.; Andriolo, V.; Bechthold, A.; Schlesinger, S.; Boeing, H. Food groups and risk of hypertension: A systematic review and dose-response meta-analysis of prospective studies. Adv. Nutr. 2017, 8, 93–803. [Google Scholar] [CrossRef] [PubMed]
- Li, B.; Li, F.; Wang, L.; Zhang, D. Fruit and vegetables consumption and risk of hypertension: A meta-analysis. J. Clin. Hypertens. (Greenwich.) 2016, 18, 468–476. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.; Sun, D.; He, Y. Fruit and vegetable consumption and incident hypertension: Dose response meta-analysis of prospective cohort studies. J. Hum. Hypertens. 2016, 30, 573–580. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Manson, J.E.; Gaziano, J.M.; Buring, J.E.; Sesso, H.D. Fruit and vegetable intake and the risk of hypertension in middle-aged and older women. Am. J. Hypertens. 2012, 25, 180–189. [Google Scholar] [CrossRef] [PubMed]
- Ascherio, A.; Stampfer, M.J.; Colditz, G.A.; Willet, W.C.; McKinlay, J. Nutrient intake and blood pressure in normotensive males. Int. J. Epidem. 1991, 20, 886–891. [Google Scholar] [CrossRef]
- Nunez-Cordoba, J.M.; Alonso, A.; Beunza, J.J.; Palma, S.; Gomez-Gracia, E.; Martinez-Gonzalez, M.A. Role of vegetables and fruits in Mediterranean diets to prevent hypertension. Eur. J. Clin. Nutr. 2009, 63, 605–612. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Kim, J. Association between fruit and vegetable consumption and risk of hypertension in middle-aged and older Korean adults. J. Acad. Nutr. Diet. 2018, 118, 1438–1449. [Google Scholar] [CrossRef] [PubMed]
- Mellendick, K.; Shanahan, L.; Wideman, L.; Calkins, S.; Keane, S.; Lovelady, C. Diets rich in fruits and vegetables are associated with lower cardiovascular disease risk in adolescents. Nutrients 2018, 10, 136. [Google Scholar] [CrossRef] [PubMed]
- Aljuraiban, G.S.; Griep, L.M.O.; Chan, Q.; Daviglus, M.L.; Stamler, J.; Van Horn, L.; Elliott, P.; Frost, G.S. Total, insoluble and soluble dietary fibre intake in relation to blood pressure: The INTERMAP Study. Br. J. Nutr. 2015, 114, 1480–1486. [Google Scholar] [CrossRef] [PubMed]
- Ascherio, A. A prospective study of nutritional factors and hypertension among US men. Circulation 1992, 86, 1475–1484. [Google Scholar] [CrossRef] [PubMed]
- Ascherio, A.; Hennekens, C.; Willett, W.; Sacks, F.; Rosner, B.; Manson, J.; Witteman, J.; Stampfer, M.J. Prospective study of nutritional factors, blood pressure, and hypertension among US women. Hypertension 1996, 27, 1065–1072. [Google Scholar] [CrossRef] [PubMed]
- Sun, L.J.; Shi, X.; Wang, T.; Zhang, D. Exploration of the association between dietary fiber intake and hypertension among U.S. adults using 2017 American College of Cardiology/American Heart Association Blood Pressure Guidelines: NHANES 2007–2014. Nutrients 2018, 10, 1091. [Google Scholar] [CrossRef] [PubMed]
- Hartley, L.; Igbinedion, E.; Holmes, J.; Flowers, N.; Thorogood, M.; Clarke, A.; Stranges, S.; Hooper, L.; Rees, K. Increased consumption of fruit and vegetables for the primary prevention of cardiovascular diseases. Cochrane Database Syst. Rev. 2013, 6. [Google Scholar] [CrossRef] [PubMed]
- Appel, L.J.; Moore, T.J.; Obarzanek, E.; Vollmer, W.M.; Svetkey, L.P; Bray, G.A.; Vogt, T.M.; Culter, J.A.; Windhauser, M.M.; et al. A clinical trial of the effects of dietary patterns on blood pressure. N. Engl. J. Med. 1997, 336, 1117–1124. [Google Scholar] [CrossRef] [PubMed]
- McCall, D.O.; McGartland, C.P.; McKinley, M.C.; Patterson, C.C.; Sharpe, P.; McCance, D.R.; Young, I.S.; Woodside, J.V. Dietary intake of fruits and vegetables improves microvascular function in hypertensive subjects in a dose-dependent manner. Circulation 2009, 119, 2153–2160. [Google Scholar] [CrossRef] [PubMed]
- John, J.H.; Ziebland, S.; Yudkin, P.; Roe, L.S.; Neil, H.A. Effects of fruit and vegetable consumption on plasma antioxidant concentrations and blood pressure: A randomized controlled trial. Lancet 2002, 359, 1969–1974. [Google Scholar] [CrossRef]
- Krishnan, S.; Adams, S.H.; Allen, L.H.; Laugero, K.D.; Newman, J.W.; Stephensen, C.B.; Burnett, D.J.; Witbracht, M.; Welch, L.C.; Que, E.S.; et al. A randomized controlled-feeding trial based on the Dietary Guidelines for Americans on cardiometabolic health indexes. Am. J. Clin. Nutr. 2018, 108, 266–278. [Google Scholar] [CrossRef] [PubMed]
- Siervo, M.; Lara, J.; Chowdhury, S.; Ashor, A.; Oggoni, C.; Mathers, J.C. Effects of the Dietary Approach to Stop Hypertension (DASH) diet on cardiovascular risk factors: A systematic review and meta-analysis. Br. J. Nutr. 2015, 113, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Khan, K.; Jovanovski, E.; Ho, H.V.T.; Marques, A.C.R.; Zurbau, A.; Mejia, S.B.; Sievenpiper, J.C.; Vuksan, V. The effect of viscous soluble fiber on blood pressure: A systematic review and meta-analysis of randomized controlled trials. Nutr. Metab. Cardiov. Dis. 2018, 28, 3–13. [Google Scholar] [CrossRef] [PubMed]
- Hartley, L.; May, M.D.; Loveman, E.; Colquitt, J.L; Rees, K. Dietary fibre for the primary prevention of cardiovascular disease. Cochrane Database Syst. Rev. 2016, 1, CD011472. [Google Scholar] [CrossRef] [PubMed]
- Streppel, M.T.; Arends, L.R.; van’t Veer, P.; Grobbee, D.E.; Gelenijnse, J.M. Dietary fiber and blood pressure: A meta-analysis of randomized placebo-controlled trials. Arch. Intern. Med. 2005, 165, 150–156. [Google Scholar] [CrossRef] [PubMed]
- Whelton, S.P.; Hyre, A.D.; Pedersen, B.; Yi, Y.; Whelton, P.K.; He, J. Effect of dietary fiber intake on blood pressure: A meta-analysis of randomized, controlled clinical trials. J. Hypertens. 2005, 23, 475–481. [Google Scholar] [CrossRef] [PubMed]
- Zhou, M.S.; Schulman, I.H.; Raij, L. Vascular inflammation, insulin resistance, and endothelial dysfunction in salt-sensitive hypertension: Role of nuclear factor kappa B activation. J. Hypertens. 2010, 28, 527–535. [Google Scholar] [CrossRef] [PubMed]
- Yang, T.; Santisteban, M.M.; Rodriguez, V.; Li, E.; Ahmari, N.; Carvajal, J.M.; Zadeh, M.; Gong, M.; Qi, Y.; Zubcevic, J.; et al. Gut dysbiosis is linked to hypertension. Hypertension 2015, 65, 1331–1340. [Google Scholar] [CrossRef] [PubMed]
- Pluznick, J.L. A novel SCFA receptor, the microbiota, and blood pressure regulation. Gut Microbes 2014, 5, 202–207. [Google Scholar] [CrossRef] [PubMed]
- Miyamoto, J.; Kascubuchi, M.; Nakajima, A.; Irie, J.; Itoh, H.; Kimura, I. The role of short-chain fatty acid on blood pressure regulation. Curr. Opin. Nephrol. Hypertens. 2016, 25, 379–383. [Google Scholar] [CrossRef] [PubMed]
- Weisman, A.; Fazli, G.S.; Johns, A.; Booth, G.L. Evolving trends in the epidemiology, risk factors, and prevention of type 2 diabetes: A review. Can. J. Cardiol. 2018, 34, 552–564. [Google Scholar] [CrossRef] [PubMed]
- Williamson, D.A. Prevention of type 2 diabetes: What is the right target population? Expert Rev. Endocrinol. Metab. 2018, 13. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, M.; Tuomilehto, J.; Aschner, P.; Beseler, L.; Cahn, A.; Eckel, R.H.; Fischl, A.H.; Guthrie, G.; Hill, J.O.; Kumwenda, M.; et al. Global status of diabetes prevention and prospects for action: A consensus statement. Diabetes Metab. Res. Rev. 2018, 34, e3021. [Google Scholar] [CrossRef] [PubMed]
- Wang, P.-Y.; Fang, J.-C.; Gao, Z.-H.; Zhang, C.; Xie, S.Y. Higher intake of fruits, vegetables or their fiber reduces the risk of type 2 diabetes: A meta-analysis. J. Diabetes Investig. 2016, 7, 56–69. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Miao, S.; Huang, Y.; Liu, Z.; Tian, H.; Yin, X.; Tang, W.; Steffen, L.; Xi, B. Fruit intake decreases risk of incident type 2 diabetes: An updated meta-analysis. Endocrine 2015, 48, 454–460. [Google Scholar] [CrossRef] [PubMed]
- Mursu, J.; Virtanen, J.K.; Tuomainen, T.-P.; Nurmi, T.; Voulainen, S. Intake of fruit, berries, and vegetables and risk of type 2 diabetes in Finnish men: The Kuopio Ischaemic Heart Disease Risk Factor Study. Am. J. Clin. Nutr. 2014, 99, 328–333. [Google Scholar] [CrossRef] [PubMed]
- Muraki, I.; Imamura, F.; Manson, J.E.; Hu, F.B.; Willett, W.C.; van Dam, R.M.; Sun, Q. Fruit consumption and risk of type 2 diabetes: Results from three prospective longitudinal cohort studies. BMJ 2013, 347, 5001. [Google Scholar] [CrossRef] [PubMed]
- Du, H.; Li, L.; Bennett, D.; Guo, Y.; Turnbull, I.; Yang, L.; Bragg, F.; Bian, Z.; Chen, Y.; Chen, J.; et al. Fresh fruit consumption in relation to incident diabetes and diabetic vascular complications: A 7-y prospective study of 0.5 million Chinese adults. PLoS Med. 2017, 14, e1002279. [Google Scholar] [CrossRef] [PubMed]
- Yao, B.; Fang, H.; Xu, W.; Yan, Y.; Xu, H.; Liu, Y.; Mo, M.; Zhang, H.; Zhao, Y. Dietary fiber intake and risk of type 2 diabetes: A dose-response analysis of prospective studies. Eur. J. Epidemiol. 2014, 29, 79–88. [Google Scholar] [CrossRef] [PubMed]
- Schulze, M.B.; Schulz, M.; Heidemann, C.; et al. Fiber and magnesium intake and incidence of type 2 diabetes. Arch. Intern. Med. 2007, 167, 956–965. [Google Scholar] [CrossRef] [PubMed]
- Looman, M.; Schoenaker, D.A.J.M.; Soedaman-Muthu, S.S.; Geelen, A.; Feskens, E.J.M.; Mishra, G.D. Pre-pregnancy dietary carbohydrate quantity and quality, and risk of developing gestational diabetes: The Australian Longitudinal Study in Women’s Health. Br. J. Nutr. 2018, 120, 435–444. [Google Scholar] [CrossRef] [PubMed]
- Sargeant, L.A.; Khaw, K.T.; Bingham, S.; Day, N.E.; Luben, R.N.; Oakes, S.; Welch, A.; Wareham, N.J. Fruit and vegetable intake and population glycosylated haemoglobin levels: The EPIC-Norfolk Study. Eur. J. Clin. Nutr. 2001, 55, 342–348. [Google Scholar] [CrossRef] [PubMed]
- Carraro, J.C.; Hermsdorff, H.H.; Mansego, M.L.; Zulet, M.A.; Milagro, F.I.; Bressan, J.; Martinez, J.A. Higher fruit intake is related to TNF-α hypomethylation and better glucose tolerance in healthy subjects. J. Nutr. Nutr. 2016, 9, 95–105. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, S.; Yoshimura, Y.; Kawasaki, R.; Kamada, C.; Tanaka, S.; Horikawa, C.; Ohashi, Y.; Araki, A.; Ito, H.; Akanuma, Y.; et al. Fruit intake and incident diabetic retinopathy with type 2 diabetes. Epidemiology 2013, 24, 204–211. [Google Scholar] [CrossRef] [PubMed]
- Breneman, C.B.; Tucker, L. Dietary fibre consumption and insulin resistance—The role of body fat and physical activity. Br. J. Nutr. 2013, 110, 375–383. [Google Scholar] [CrossRef] [PubMed]
- Morimoto, N.; Kasuga, C.; Tanaka, A.; Kamachi, K.; Ai, M.; Urayama, K.Y.; Tanaka, A. Association between dietary fibre: Carbohydrate intake ratio and insulin resistance in Japanese adults without type 2 diabetes. Br. J. Nutr. 2018, 119, 620–628. [Google Scholar] [CrossRef] [PubMed]
- AlEssa, H.B.; Ley, S.H.; Rosner, B.; Malik, U.S.; Willett, W.C.; Campos, H.; Hu, F.B. High fiber and low starch intakes are associated with circulating intermediate biomarkers of type 2 diabetes among women. J. Nutr. 2016, 146, 306–317. [Google Scholar] [CrossRef] [PubMed]
- Den Biggelaar, L.J.C.J.; Eussen, S.J.P.M.; Sep, S.J.S.; Mari, A.; Ferranini, E.; van Greevenbroek, M.M.; van der Kallen, C.J.; Schalkwijk, C.G.; Arts, I.C.W.; Stehouwer, C.D.A.; et al. Prospective associations of dietary carbohydrate, fat, and protein intake with β-cell function in the CODAM study. Eur. J. Nutr. 2018. [Google Scholar] [CrossRef] [PubMed]
- AlEssa, H.B.; Bhupathiraju, S.N.; Malik, V.S.; Wedick, N.M.; Campos, H.; Rosen, B.; Willett, W.C.; Hu, F.B. Carbohydrate quality and quantity and risk of type 2 diabetes in US women. Am. J. Clin. Nutr. 2015, 102, 1543–1553. [Google Scholar] [CrossRef] [PubMed]
- Haber, G.B.; Heaton, K.W.; Murphy, D. Depletion and disruption of dietary fibre effects on satiety, plasma-glucose, and serum insulin. Lancet 1977, 2, 679–682. [Google Scholar] [CrossRef]
- Bolton, R.P.; Heaton, K.W.; Burroughs, L.F. The role of dietary fiber in satiety, glucose and insulin: Studies with fruit and fruit juice. Am. J. Clin. Nutr. 1981, 34, 211–217. [Google Scholar] [CrossRef] [PubMed]
- Oliver, C.-Y.; Rasmussen, H.; Kamil, A.; Du, P.; Blumberg, J.B. Orange pomace improves postprandial glycemic responses: An acute, randomized, placebo-controlled, double-blind, crossover trial in overweight men. Nutrients 2017, 9, 130. [Google Scholar] [CrossRef]
- Torronen, R.; Kolehmainen, M.; Sarkkinen, E.; Poutanen, K.; Mykkanen, H.; Niskanaen, L. Berries reduce postprandial insulin responses to wheat and rye breads in healthy women. J. Nutr. 2013, 143, 430–436. [Google Scholar] [CrossRef] [PubMed]
- De Carvalho, C.; de Paula, T.P.; Viana, L.V.; Machado, V.M.; de Almeida, J.C.; Azevedo, M.J. Plasma glucose and insulin responses after consumption of breakfasts with different sources of soluble fiber in type 2 diabetes patients: A randomized crossover clinical trial. Am. J. Clin. Nutr. 2017, 106, 1238–1245. [Google Scholar] [CrossRef] [PubMed]
- Galvao Candido, F.; Silva Ton, W.T.; de Cassia Goncalves Alfenas, R. Addition of dietary fiber sources to shakes reduces postprandial glycemia and alters food intake. Nutr. Hosp. 2015, 31, 299–306. [Google Scholar] [CrossRef]
- Esfahani, A.; Lam, J.; Kendall, C.W.C. Acute effects of raisin consumption on glucose and insulin responses in healthy individuals. J. Nutr. Sci. 2014, 3, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Jenkins, D.J.; Jenkins, A.L. Dietary fiber and the glycemic response. Proc. Soc. Exp. Biol. Med. 1985, 180, 422–431. [Google Scholar] [CrossRef] [PubMed]
- McRorie, J.W. Evidence-based approach to fiber supplement and clinical meaningful health benefits, part 1. Nutr. Today 2015, 50, 82–89. [Google Scholar] [CrossRef] [PubMed]
- Boland, B.B.; Rhodes, C.J.; Grimsby, J.S. The dynamic plasticity of insulin production in β-cells. Mol. Metab. 2017, 6, 958–973. [Google Scholar] [CrossRef] [PubMed]
- Stull, A.J.; Cash, K.C.; Johnson, W.D.; Champagne, C.M.; Cefalu, W.T. Bioactives in blueberries improve insulin sensitivity in obese, insulin-resistant men and women. J. Nutr. 2010, 140, 1764–1768. [Google Scholar] [CrossRef] [PubMed]
- Anderson, J.W.; Weiter, K.M.; Christian, A.L.; Ritchey, M.B.; Bays, H.E. Raisins compared with other snack effects on glycemia and blood pressure: A randomized, controlled trial. Post-Grad. Med. 2014, 126, 37–43. [Google Scholar] [CrossRef] [PubMed]
- Christensen, A.S.; Viggers, L.; Hasselström, K.; Gregersen, S. Effect of fruit restriction on glycemic control in patients with type 2 diabetes--a randomized trial. Nutr. J. 2013, 12, 29. [Google Scholar] [CrossRef] [PubMed]
- Silva, F.M.; Kramer, C.K.; de Almeida, J.C.; Steemburgo, T.; Gross, J.L.; Azevedo, M.J. Fiber intake and glycemic control in patients with type 2 diabetes mellitus: A systematic review with meta-analysis of randomized controlled trials. Nutr. Rev. 2013, 71, 790–801. [Google Scholar] [CrossRef] [PubMed]
- Post, R.E.; Mainous, A.G.; King, D.E.; Simpson, K.N. Dietary fiber for the treatment of type 2 diabetes mellitus: A meta-Analysis. J. Am. Board Fam. Med. 2012, 25, 16–23. [Google Scholar] [CrossRef] [PubMed]
- Xi, P.; Liu, R.H. Whole food approach for type 2 diabetes prevention. Mol. Nutr. Food Res. 2016, 60, 1819–1836. [Google Scholar] [CrossRef] [PubMed]
- Weickert, M.O.; Pfeiffer, A.F.H. Impact of dietary fiber consumption on insulin resistance and the prevention of type 2 diabetes. J. Nutr. 2018, 148, 7–12. [Google Scholar] [CrossRef] [PubMed]
- McRorie, J.W. Evidence-based approach to fiber supplements and clinically meaningful health benefits, part 2. Nutr. Today 2015, 50, 90–97. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Zhang, F.; Ding, X.; Wu, G.; Lam, Y.Y.; Wang, X.; Fu, H.; Lu, C.; Ma, J.; Yu, L.; et al. Gut bacteria selectively promoted by dietary fiber alleviate type 2 diabetes. Science 2018, 359, 1151–1156. [Google Scholar] [CrossRef] [PubMed]
- Weitkunat, K.; Schumann, S.; Nickel, D.; Hornemann, S.; Petzke, K.J.; Schulze, M.B.; Pfeiffer, A.F.; Klaus, S. Odd-chain acids as a biomarker for dietary fiber intake: A novel pathway for endogenous production from propionate. Am. J. Clin. Nutr. 2017, 105, 1544–1551. [Google Scholar] [CrossRef] [PubMed]
- Saklayen, M.G. The global epidemic of the metabolic syndrome. Curr. Hypertens. Rep. 2018, 20, 12. [Google Scholar] [CrossRef] [PubMed]
- Tian, Y.; Su, L.; Wang, J.; Duan, X.; Jiang, X. Fruit and vegetable consumption and risk of the metabolic syndrome: A meta-analysis. Public Health Nutr. 2018, 21, 756–765. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Zhang, D.-Z. Associations of vegetable and fruit consumption with metabolic syndrome. A meta-analysis of observational studies. Public Health Nutr. 2018, 21, 1693–1703. [Google Scholar] [CrossRef] [PubMed]
- Yeo, R.; Yoon, S.R.; Kim, O.Y. The association between food group consumption patterns and early metabolic syndrome risk in non-diabetic healthy people. Clin. Nutr. Res. 2017, 6, 172–182. [Google Scholar] [CrossRef] [PubMed]
- Basu, A.; Lyons, T.J. Strawberries, Blueberries, and cranberries in the metabolic syndrome: Clinical perspectives. J. Agric. Food Chem. 2011, 60, 5687–5692. [Google Scholar] [CrossRef] [PubMed]
- Vendrame, S.; Bo, D.; Ciappellano, S.; Riso, P.; Zacas, D. Berry fruit consumption and metabolic syndrome. Antioxidants (Basel) 2016, 5, 34. [Google Scholar] [CrossRef] [PubMed]
- Fulgoni, V.L., III; Dreher, M.; Davenport, A.J. Avocado consumption is associated with better diet quality and nutrient intake, and lower metabolic syndrome risk in US adults: Results from the National Health and Nutrition Examination Survey (NHANES) 2001–2008. Nutr. J. 2013, 12, 1. [Google Scholar] [CrossRef] [PubMed]
- Tabeshpour, J.; Razavi, B.M.; Hosseinzadeh, H. Effects of avocado (Persea americana) on metabolic syndrome; a comprehensive systematic review. Phytother. Res. 2017, 31, 819–837. [Google Scholar] [CrossRef] [PubMed]
- Fulgoni, V.L., III; Painter, J.; Carughi, A. Association of raisin consumption with nutrient intake, diet quality, and health risk factors in US adults: National Health and Nutrition Examination Survey 2001–2012. Food Nutr. Res. 2017, 61, 1378567. [Google Scholar] [CrossRef] [PubMed]
- Esmaillzadeh, A.; Kimiagar, M.; Mehrabi, Y.; Azadbakht, L.; Hu, F.B.; Willett, W.C. Fruit and vegetable intakes, C-reactive protein, and the metabolic syndrome. Am. J. Clin. Nutr. 2006, 84, 1489–1497. [Google Scholar] [CrossRef] [PubMed]
- Wei, B.; Liu, Y.; Lin, X.; Fabg, Y.; Cui, J.; Wan, J. Dietary fiber intake and risk of metabolic syndrome: A meta-analysis of observational studies. Clin. Nutr. 2017. [Google Scholar] [CrossRef] [PubMed]
- Hosseinpour-Niazi, S.; Mirmiran, P.; Mirzaei, S.; Azizi, F. Cereal, fruit, and vegetable fibre intake and the risk of the metabolic syndrome: A prospective study in the Tehran Lipid and Glucose Study. J. Hum. Nutr. Diet. 2015, 28, 236–245. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Montalvo, I.A.; Mendez Diaz, S.Y.; Aguirre Guzman, M.A.; Sanchez Medina, M.A.; Matias Perez, D.; Perez Campos, E. Increasing consumption of dietary fiber complementary to the treatment of metabolic syndrome. Nutr. Hosp. 2018, 35, 582–587. [Google Scholar] [CrossRef] [PubMed]
- Sekgala, M.D.; Mchiza, Z.J.; Parker, W.; Monyeki, K.D. Dietary fiber intake and metabolic syndrome risk factors among young South African adults. Nutrients 2018, 10, 504. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.-P.; Chen, G.-C.; Wang, X.-P.; Qin, L.; Bai, Y. Dietary fiber and metabolic syndrome: A meta-analysis and review of related mechanisms. Nutrients 2018, 10, 24. [Google Scholar] [CrossRef] [PubMed]
- Behrens, G.; Gredner, T.; Stock, C.; Leitzmann, M.F.; Brenner, H.; Mons, U. Cancers due to excess weight, low physical activity, and unhealthy diet. Dtsch. Arztebl. Int. 2018, 115, 578–585. [Google Scholar] [CrossRef] [PubMed]
- Kushi, L.H.; Doyle, C.; McCullough, M.; Rock, C.L.; Demark-Wahnefried, W.; Bandera, E.V.; Gapstur, S.; Patel, A.V.; Andrews, K.; Gansler, T. American Cancer Society Guidelines on nutrition and physical activity for cancer prevention: Reducing the risk of cancer with healthy food choices and physical activity. CA Cancer J. Clin. 2012, 62, 30–67. [Google Scholar] [CrossRef] [PubMed]
- World Cancer Research Fund/American Institute for Cancer Research. Food, Nutrition, Physical Activity, and the Prevention of Colorectal Cancer. Continuous Update Project Report. 2011. Available online: http://www.aicr.org/continuousupdate-project/reports/Colorectal-Cancer-2011-Report.pdf (accessed on 27 July 2018).
- World Cancer Research Fund/American Institute for Cancer Research expert Report. Food, Nutrition, Physical Activity, and the Prevention of Cancer: A Global Perspective. 2007, pp. 280–288. Available online: www.aicr.org/assets/docs/pdf/reports/Second_Expert_Report.pdf (accessed on 5 August 2018).
- Aune, D.; Lau, R.; Chan, D.S.M.; Vieira, R.; Greenwood, D.C.; Kampman, E.; Norat, T. Nonlinear reduction in risk for colorectal cancer by fruit and vegetable intake based on meta-analysis of prospective studies. Gastroenterology 2011, 141, 106–118. [Google Scholar] [CrossRef] [PubMed]
- Bradbury, K.E.; Appleby, P.N.; Key, T.J. Fruit, vegetable, and fiber intake in relation to cancer risk: Findings from the European Prospective Investigation into Cancer and Nutrition (EPIC). Am. J. Clin. Nutr. 2014, 100 (Suppl. 1), 394S–398S. [Google Scholar] [CrossRef] [PubMed]
- Vogtmann, E.; Xiang, Y.-B.; Li, H.-L.; Levitan, E.B.; Yang, G.; Waterbor, J.W.; Gao, J.; Cai, H.; Wu, Q.J.; et al. Fruit and vegetable intake and the risk of colorectal cancer: Results from the Shanghai Men’s Health Study. Cancer Causes Control 2013, 24, 1935–1945. [Google Scholar] [CrossRef] [PubMed]
- Schwingshackl, L.; Schwedhelm, C.; Hoffmann, G.; Knuppel, S.; Laure Preterre, A.; Iqbal, K.; Bechthold, A.; De Henauw, S.; Michels, N.; Devleeschauwer, B.; et al. Food groups and risk of colorectal cancer. Int. J. Cancer 2018, 142, 1748–1758. [Google Scholar] [CrossRef] [PubMed]
- Fabiani, R.; Minelli, L.; Rosignoli, P. Apple intake and cancer risk: A systematic review and meta-analysis of observational studies. Public Health Nutr. 2016, 19, 2603–2617. [Google Scholar] [CrossRef] [PubMed]
- Vulcan, A.; Brandstedt, J.; Manjer, J.; Jirdtrom, K.; Ohlsson, B.; Ericson, U. Fibre intake and incident colorectal cancer depending on fibre source, sex, tumour location and tumour, node, metastasis stage. Br. J. Nutr. 2015, 114, 959–969. [Google Scholar] [CrossRef] [PubMed]
- Gianfredi, V.; Salvatori, T.; Villarini, M.; Moretti, M.; Nucci, D.; Realdon, S. dietary fibre truly protective against colon cancer? A systematic review and meta-analysis. Int. J. Food Sci. Nutr. 2018,, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Ben, Q.; Sun, Y.; Chai, R.; Qian, A.; Xu, B.; Yuan, Y. Dietary fiber intake reduces risk for colorectal adenoma: Meta-analysis. Gastroenterology 2014, 146, 689–699. [Google Scholar] [CrossRef] [PubMed]
- Ben, Q.; Zhong, J.; Liu, J.; Wang, L.; Sun, Y.L.; Yuan, Y. Association between consumption of fruits and vegetables and risk of colorectal adenoma A PRISMA-Compliant meta-analysis of observational studies. Medicine 2015, 94, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Sansbury, L.B.; Wanke, K.; Albert, P.S.; Kahle, L.; Schatzkin, A.; Lanza, E. The effect of strict adherence to a high-fiber, high-fruit and -vegetable, and low-fat eating pattern on adenoma recurrence. Am. J. Epidem. 2009, 170, 576–584. [Google Scholar] [CrossRef] [PubMed]
- Kunzmann, A.T.; Coleman, H.G.; Huang, W.-Y.; Cantwell, M.M.; Kitharn, C.M.; Berndt, S.I. Fruit and vegetable intakes and risk of colorectal cancer and incident and recurrent adenomas in the PLCO cancer screening trial. Int. J. Cancer 2016, 138, 1851–1861. [Google Scholar] [CrossRef] [PubMed]
- Song, M.; Wu, K.; Meyerhardt, J.A.; Ogino, S.; Wang, M.; Fuchs, C.S.; Giovannucci, E.L.; Chan, A.T. Fiber intake and survival after colorectal cancer diagnosis. JAMA Oncol. 2018, 4, 71–79. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Vitetta, L. Inflammatory-modulating effect of butyrate in the prevention of colon cancer by dietary fiber. Clin. Color. Cancer 2018. [Google Scholar] [CrossRef] [PubMed]
- O’Keeffe, L.M.; Taylor, G.; Huxley, R.R.; Mitchell, P.; Woodward, M.; Petters, S.A.E. Smoking as a risk factor for lung cancer in women and men: A systematic review and meta-analysis. BMJ Open 2018, 8, e021611. [Google Scholar] [CrossRef]
- Wang, Y.; Li, F.; Wang, Z.; Qiu, T.; Shen, Y.; Wang, M. Fruit and vegetable consumption and risk of lung cancer: Dose response meta-analysis of prospective cohort studies. Lung Cancer 2015, 88, 124–130. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Qin, S.; Zhang, T.; Song, X.; Zhang, S. The effect of fruit and vegetable intake on the development of lung cancer: A meta-analysis of 32 publications and 20,414 cases. Eur. J. Clin. Nutr. 2015, 69, 1184–1192. [Google Scholar] [CrossRef] [PubMed]
- Vieira, A.R.; Abar, L.; Vingeliene, S.; Chan, D.S.; Aune, D.; Navarro-Rosenblatt, D.; Steven, C.; Greenwood, D.; Norat, T. Fruits, vegetables and lung cancer risk: A systematic review and meta-analysis. Ann. Oncol. 2016, 27, 81–96. [Google Scholar] [CrossRef] [PubMed]
- Nomura, S.J.O.; Dash, C.; Rosenberg, L.; Palmer, J.; Adams-Campbell, L.L. Fruit and vegetable intake and lung cancer incidence among black women according to cigarette smoking status. Nutr. Cancer 2018. [Google Scholar] [CrossRef] [PubMed]
- Milte, C.M.; McNaughton, S.A. Dietary patterns and successful ageing; a systematic review. Eur. J. Nutr. 2016, 55, 423–450. [Google Scholar] [CrossRef] [PubMed]
- Assmann, K.E.; Lassale, C.; Andreeva, V.A.; Jeandel, C.; Hercberg, S.; Galan, P.; Kesse-Guyot, E. A healthy dietary pattern at midlife, combined with a regulated energy intake, is related to increased odds for healthy aging. J. Nutr. 2015, 145, 2139–2145. [Google Scholar] [CrossRef] [PubMed]
- Myint, P.K.; Welch, A.A.; Bingham, S.A.; Surtees, P.G.; Wainwright, N.W.; Luben, R.N.; Wareham, N.J.; Smith, R.D.; Harvey, I.M.; Day, N.E.; Khaw, K.T. Fruit and vegetable consumption and self-reported functional health in men and women in the European Prospective Investigation into Cancer-Norfolk (EPIC-Norfolk): A population based cross-sectional study. Public Health Nutr. 2007, 10, 34–41. [Google Scholar] [CrossRef] [PubMed]
- Hodge, A.M.; O’Dea, K.; English, D.R.; Giles, G.G.; Flicker, L. Dietary patterns as predictors of successful ageing. J. Nutr. Health Aging 2014, 18, 221–227. [Google Scholar] [CrossRef] [PubMed]
- Gopinath, B.; Russell, J.; Kifley, A.; Flood, V.M.; Mitchell, P. Adherence to Dietary Guidelines and Successful Aging over 10 Years. J. Gerontol. A Biol. Sci. Med. Sci. 2016, 71, 349–355. [Google Scholar] [CrossRef] [PubMed]
- Gopinath, B.; Flood, V.M.; Kifley, A.; Louie, J.C.; Mitchell, P. Association between carbohydrate nutrition and successful aging over 10 years. J. Gerontol. A Biol. Sci. Med. Sci. 2016, 71, 1335–1340. [Google Scholar] [CrossRef] [PubMed]
- Hu, D.; Huang, J.; Wang, Y.; Zhang, D.; Qu, Y. Fruits and vegetables consumption and risk of stroke. A meta-analysis of prospective cohort studies. Stroke 2014, 45, 1613–1619. [Google Scholar] [CrossRef] [PubMed]
- Aune, D.; Giovannucci, E.; Boffetta, P.; Fadnes, L.T.; Keum, N.; Norat, T.; Greenwood, D.C.; Riboli, E.; Vatten, L.J.; Tonstad, S. Fruit and vegetable intake and the risk of cardiovascular disease, total cancer and all-cause mortality-a systematic review and dose response meta-analysis of prospective studies. Int. J. Epidemiol. 2017, 1–28. [Google Scholar] [CrossRef] [PubMed]
- Buil-Cosiales, P.; Zazpe, I.; Toledo, E.; Corella, D.; Salas-Salvado, J.; Diez-Espino, J.; Ros, E.; Fernandez-Creuet Navajas, J.; Santos-Lozano, J.M.; Aros, F. Fiber intake and all-cause mortality in the Prevencion con Dieta Mediterranea (PREDIMED) study. Am. J. Clin. Nutr. 2014, 100, 1498–1507. [Google Scholar] [CrossRef] [PubMed]
- Root, M.M.; McGinn, M.C.; Nieman, D.C.; Henson, D.A.; Heinz, S.A.; Shanely, R.A.; Knab, A.M.; Jin, F. Combined fruit and vegetable intake is correlated with improved inflammatory and oxidant status from a cross-sectional study in a community setting. Nutrients 2012, 4, 29–41. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Rimando, J.; Sandler, D.P. Fruit and vegetable intake and urinary levels of prostaglandin E2 metabolite in postmenopausal women. Nutr. Cancer 2015, 67, 580–586. [Google Scholar] [CrossRef] [PubMed]
- Abdullah, M.M.; Gyles, C.L.; Marinangeli, C.P.; Carlberg, J.G.; Jones, P.J. Cost-of-illness analysis reveals potential healthcare savings with reductions in type 2 diabetes and cardiovascular disease following recommended intakes of dietary fiber in Canada. Front. Pharmacol. 2015, 6, 167. [Google Scholar] [CrossRef] [PubMed]
- Fellet, M. Increasing Healthspan: Tissue engineering and medicinal chemistry for aging bodies and brains. ACS Chem. Life 2017, 1, 1–33. [Google Scholar]
- Tucker, L.A. Dietary fiber and telomere length in 5674 U.S. adults: An NHANES study of biological aging. Nutrients 2018, 10, 400. [Google Scholar] [CrossRef] [PubMed]
- Garcıa-Calzon, S.; Gea, A.; Razquin, C.; Corella, D.; Lamuela-Raventos, R.M.; Martinez, J.A.; Martinez-Gonzalez, M.A.; Zalba, G.; Marti, A. Longitudinal association of telomere length and obesity indices in an intervention study with a Mediterranean diet: The PREDIMED-NAVARRA trial. Int. J. Obesity 2014, 38, 177–182. [Google Scholar] [CrossRef] [PubMed]
- García-Calzón, S.; Zalba, G.; Ruiz-Canela, M.; Shivappa, N.; Hebert, J.R.; Martinez, J.A.; Fito, M.; Gomez-Gracia, E.; Martinez-Gonzalez, M.A.; Marti, A. Dietary inflammatory index and telomere length in subjects with a high cardiovascular disease risk from the PREDIMED-NAVARRA study: Cross-sectional and longitudinal analyses over 5 y. Am. J. Clin. Nutr. 2015, 102, 897–904. [Google Scholar] [CrossRef] [PubMed]
- Nicodemus-Johnson, J.; Sinnott, R.A. Fruit and juice epigenetic signatures are associated with independent immunoregulatory pathways. Nutrients 2017, 9, 752. [Google Scholar] [CrossRef] [PubMed]
- Keenan, M.J.; Marco, M.L.; Ingram, D.K.; Martin, R.J. Improving healthspan via changes in gut microbiota and fermentation. Age 2015, 37, 98. [Google Scholar] [CrossRef] [PubMed]
- Hosseini, B.; Berthon, B.S.; Wark, P.; Wood, LG. Effects of fruit and vegetable consumption on risk of asthma, wheezing and immune responses: A systematic review and meta-analysis. Nutrients 2017, 9, 41. [Google Scholar] [CrossRef] [PubMed]
- Bakolis, I.; Hooper, R.; Bachert, C.; Lange, B.; Haahtela, T.; Keil, T.; Hofmaier, S.; Fokkens, S.; Rymarczyk, B.; Janson, C. Dietary patterns and respiratory health in adults from nine European countries—Evidence from the GA2 LEN study. Clin. Exp. Allergy 2018. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Larsen, V.; Del Giacco, S.R.; Moreira, A.; Bonini, M.; Charles, D.; Reeves, T.; Carlsen, K.H.; Haahtela, T.; Bonini, S.; Fonseca, J.; et al. Asthma and dietary intake: An overview of systematic reviews. Allergy 2016, 71, 433–442. [Google Scholar] [CrossRef] [PubMed]
- Willers, S.M.; Wijga, A.H.; Brunekreef, B.; Scholtens, S.; Postma, D.S.; Kerkhof, M.; de Jongste, J.C.; Smit, H.A. Childhood diet and asthma and atopy at 8 years of age: The PIAMA birth cohort study. Eur. Respir. J. 2011, 37, 1060–1067. [Google Scholar] [CrossRef] [PubMed]
- Mendes, A.P.; Zhang, L.; Prietsch, S.O.; Franco, O.S.; Gonzales, K.P.; Fabris, A.G.; Catharino, A. Factors associated with asthma severity in children: A case control study. J. Asthma 2011, 48, 235–240. [Google Scholar] [CrossRef] [PubMed]
- Barros, R.; Moreira, A.; Fonseca, J.; de Oliveira, J.F.; Delgado, L.; Castel-Branco, M.G.; Haahtela, T.; Lopes, C.; Moreira, P. Adherence to the Mediterranean diet and fresh fruit intake is associated with improved asthma control. Allergy 2008, 63, 917–923. [Google Scholar] [CrossRef] [PubMed]
- Seyedrezazadeh, E.; Moghaddam, M.P.; Ansarin, K.; Vafa, M.R.; Sharma, S.; Kolahdooz, F. Fruit and vegetable intake and risk of wheezing and asthma: A systematic review and meta-analysis. Nutr. Rev. 2014, 72, 411–428. [Google Scholar] [CrossRef] [PubMed]
- Ma, J.; Strub, P.; Lv, N.; Xiao, L.; Camargo, C.A; Buist, A.S.; Lavori, P.W.; Wilson, S.R.; Nadeau, K.C.; Rosas, L.G. Pilot randomised trial of a healthy eating behavioural intervention in uncontrolled asthma. Eur. Respir. J. 2016, 47, 122–132. [Google Scholar] [CrossRef] [PubMed]
- Cepeda, A.M.; Thawer, S.; Boyle, R.J.; Villalba, S.; Jaller, R.; Tapias, E.; Segura, A.M.; Villegas, R.; Garcia-Larsen, V. Diet and respiratory health in children from 11 Latin American countries: Evidence from ISAAC Phase III. Lung 2017, 195, 683–692. [Google Scholar] [CrossRef] [PubMed]
- Hosseini, B.; Berthon, B.S.; Saedisomeolia, A.; Starkey, M.R.; Collison, A.; Wark, P.A.B.; Wood, L.G. Effects of fruit and vegetable consumption on inflammatory biomarkers and immune cell populations: A systematic literature review and meta-analysis. Am. J. Clin. Nutr. 2018, 108, 136–155. [Google Scholar] [CrossRef] [PubMed]
- Statovci, D.; Aguilera, M.; MacSharry, J.; Melgar, S. The impact of Western diet and nutrients on the microbiota and immune response at mucosal interfaces. Front. Immunol. 2017, 8, 838. [Google Scholar] [CrossRef] [PubMed]
- Berthon, B.S.; Macdonald-Wicks, L.K.; Gibson, P.G.; Wood, L.G. Investigation of the association between dietary intake, disease severity and airway inflammation in asthma. Respirology 2013, 18, 447–454. [Google Scholar] [CrossRef] [PubMed]
- Folkerts, J.; Stadhouders, R.; Redegeld, F.A.; Tam, S.Y.; Hendriks, R.W.; Galli, S.J.; Maurer, M. Effect of dietary fiber and metabolites on mast cell activation and mast cell-associated diseases. Front. Immunol. 2018, 9, 1067. [Google Scholar] [CrossRef] [PubMed]
- Cuello-Garcia, C.; Fiocchi, A.; Pawankar, R.; Yepes-Nuez, J.J.; Morgano, G.P.; Zhang, Y.; Agarwal, A.; Gandhi, S.; Terracciano, L.; Schunemann, H.J.; et al. Prebiotics for the prevention of allergies: A systematic review and meta-analysis of randomized controlled trials. Clin. Exp. Allergy 2017, 47, 1468–1477. [Google Scholar] [CrossRef] [PubMed]
- Adeloye, D.; Chua, S.; Lee, C.; Basquill, C.; Papana, A.; Theodoratou, E.; Nair, H.; Gasevic, D.; Sridhar, D.; Campbell, H.; et al. Global and reginal estimates of COPD prevalence: Systematic review and meta-analysis. J. Glob. Health 2015, 5, 020415. [Google Scholar] [CrossRef] [PubMed]
- Salvi, S. Tobacco smoking and environmental risk factors for chronic obstructive pulmonary disease. Clin. Chest Med. 2014, 35, 17–27. [Google Scholar] [CrossRef] [PubMed]
- Steinemann, N.; Grize, L.; Pons, M.; Rothe, T.; Stolz, D.; Turk, A.; Schindler, C.; Brombach, C.; Probst-Hensch, N. Associations between dietary patterns and post-bronchodilation lung function in the SAPALDIA Cohort. Respiration 2018. [Google Scholar] [CrossRef] [PubMed]
- Brigham, E.P.; Steffen, L.M.; London, S.J.; Boyce, D.; Diette, G.B.; Hansel, N.N.; Rice, J.; McCormack, M.C. Diet pattern and respiratory morbidity in the Atherosclerosis Risk in Communities Study. Ann. Am. Thorac. Soc. 2018, 15, 675–682. [Google Scholar] [CrossRef] [PubMed]
- Kaluza, J.; Larsson, S.C.; Orsini, N.; Linden, A.; Wolk, A. Fruit and vegetable consumption and risk of COPD: A prospective cohort study of men. Thorax 2017, 72, 500–509. [Google Scholar] [CrossRef] [PubMed]
- Kaluza, J.; Harris, H.R.; Linden, A.; Wolk, A. Long-term consumption of fruits and vegetables and risk of chronic obstructive pulmonary disease: A prospective cohort study of women. Int. J. Epidemiol. 2018. [Google Scholar] [CrossRef] [PubMed]
- Hanson, C.; Lyden, E.; Rennard, S.; Mannino, D.M.; Rutten, E.P.; Hopkins, R.; Young, R. The relationship between dietary fiber intake and lung function in the National Health and Nutrition Examination Surveys. Ann. Am. Thorac. Soc. 2016, 13, 643–650. [Google Scholar] [CrossRef] [PubMed]
- Kaluza, J.; Harris, H.; Wallin, A.; Linden, A.; Wolk, A. Dietary fiber intake and risk of chronic obstructive pulmonary disease: A prospective cohort study of men. Epidemiology 2018, 29, 254–260. [Google Scholar] [CrossRef] [PubMed]
- Young, R.P.; Hopkins, R.J.; Marsland, B. The gut-liver-lung axis. Modulation of the innate immune response and its possible role in chronic obstruction pulmonary disease. Am. J. Respir. Cell Mol. Biol. 2016, 54, 161–169. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.K.; Shin, C. The microbiota-gut brain axis in neuropsychiatric disorders: Pathophysiological mechanisms and novel treatments. Curr. Neuropharmacol. 2018, 16, 559–573. [Google Scholar] [CrossRef] [PubMed]
- Taylor, A.M.; Holscher, H.D. A review of dietary and microbial connections to depression, anxiety, and stress. Nutr. Neurosci. 2018, 9, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, B.; Ding, D.; Mihrshahi, S. Fruit and vegetable consumption and psychological distress: Cross sectional and longitudinal analyses based on a large Australian sample. BMJ Open 2017, 7, e014201. [Google Scholar] [CrossRef] [PubMed]
- Lai, J.S.; Hiles, S.; Bisquera, A.; Hure, A.J.; McEvoy, M.; Attia, J. A systematic review and meta-analysis of dietary patterns and depression in community-dwelling adults. Am. J. Clin. Nutr. 2014, 99, 181–197. [Google Scholar] [CrossRef] [PubMed]
- Goyal, M.S.; Iannotti, L.; Raichle, M.E. Brain nutrition: A life span approach. Annu. Rev. Nutr. 2018, 38, 17–19. [Google Scholar] [CrossRef] [PubMed]
- Bolduc, F.V.; Lau, A.; Rosenfelt, C.S.; Langer, S.; Wang, N.; Smithson, L.; Letebvre, D.; Alexander, T.; Dickson, C.T.; Li, L.; et al. Cognitive enhancement in infants associated with increased maternal fruit intake during pregnancy: Result from a birth cohort study with validation in an animal model. EBioMedicine 2016, 8, 331–340. [Google Scholar] [CrossRef] [PubMed]
- Renzaho, A.M.N.; Kumanyika, S.; Tucker, K.L. Family functioning, parental psychological distress, child behavioural problems, socio-economic disadvantage and fruit and vegetable consumption among 4-12-year-old Victorians, Australia. Health Promot. Int. 2010, 26, 263–275. [Google Scholar] [CrossRef] [PubMed]
- Oddy, W.H.; Robinson, M.; Ambrosini, G.L.; O’Sullivan, T.A.; de Klerk, N.H.; Beilin, L.J.; Silburn, S.R.; Zubrick, S.R.; Stanley, F.J. The association between dietary patterns and mental health in early adolescence. Prev. Med. 2009, 49, 39–44. [Google Scholar] [CrossRef] [PubMed]
- Meegan, A.P.; Perry, I.J.; Phillips, C.M. The association between dietary quality and dietary guideline adherence with mental health outcomes in adults: A cross-sectional analysis. Nutrients 2017, 9, 238. [Google Scholar] [CrossRef] [PubMed]
- Brookie, K.L.; Best, G.I.; Conner, T.S. Intake of raw fruits and vegetables is associated with better mental health than intake of processed fruits and vegetables. Front. Psychol. 2018, 9, 487. [Google Scholar] [CrossRef] [PubMed]
- Mujcic, R.; Oswald, A.J. Evolution of well-being and happiness after increases in consumption of fruit and vegetables. Am. J. Public Heal. 2016, 106, 1504–1510. [Google Scholar] [CrossRef] [PubMed]
- Conner, T.S.; Brookie, K.L.; Richardson, A.C.; Polak, M.A. On carrots and curiosity: Eating fruit and vegetables is associated with greater flourishing in daily life. Br. J. Health Psychol. 2015, 20, 413–427. [Google Scholar] [CrossRef] [PubMed]
- Conner, T.S.; Thompson, L.M.; Knight, R.L.; Flett, J.A.; Richardson, A.C.; Brookie, K.L. The role of personality traits in young adult fruit and vegetable consumption. Front. Psychol. 2017, 8, 119. [Google Scholar] [CrossRef] [PubMed]
- Conner, T.S.; Brookie, K.L.; Carr, A.C.; Mainvil, L.A.; Vissers, M.C. Let them eat fruit! The effect of fruit and vegetable consumption on psychological well-being in young adults: A randomized controlled trial. PLoS ONE 2017, 12, e0171206. [Google Scholar] [CrossRef] [PubMed]
- Saghafian, F.; Malmir, H.; Saneei, P.; Keshteli, A.H.; Hosseinzadeh-Attar, M.J.; Afshar, H.; Siassi, F.; Esmaillzadeh, A.; Adubi, P. Consumption of fruit and vegetables in relation with psychological disorders in Iranian adults. Eur. J. Nutr. 2018. [Google Scholar] [CrossRef] [PubMed]
- Saghafian, F.; Malmir, H.; Saneei, P.; Milajerdi, A.; Larijani, B. Esmaillzadeh, A. Fruit and vegetable consumption and risk of depression: Accumulative evidence from an updated systematic review and meta-analysis of epidemiological studies. Br. J. Nutr. 2018, 119, 1087–1101. [Google Scholar] [CrossRef] [PubMed]
- Kingsbury, M.; Dupuis, G.; Jacka, F.; Roy-Gagnon, M.H.; McMartin, S.E.; Colman, I. Associations between fruit and vegetable consumption and depressive symptoms: Evidence from a national Canadian longitudinal survey. J. Epidemiol. Community Heal. 2016, 70, 155–161. [Google Scholar] [CrossRef] [PubMed]
- Miki, T.; Eguchi, M.; Kurotani, K.; Kochi, T.; Kuwahara, K.; Ito, R.; Kimura, Y.; Tsuruoka, H.; Akter, S.; Kashino, I. et al.; et al. Dietary fiber intake and depressive symptoms in Japanese employees: The Furukawa Nutrition and Health Study. Nutrition 2016, 32, 584–589. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.; Li, S.; Song, X.; Li, Z.; Zhang, D. Exploration of the association between dietary fiber intake and depressive symptoms in adults. Nutrition 2018, 54, 48–53. [Google Scholar] [CrossRef] [PubMed]
- Steptoe, A.; Easterlin, E.; Kirschbaum, C. Conscientious, hair cortisol concentration, and health behaviour in older men and women. Psychoneuroendocrinology 2017, 86, 122–127. [Google Scholar] [CrossRef] [PubMed]
- Whisner, C.M.; Castillo, L.F. Prebiotic, bone and mineral metabolism. Calcif. Tissue Int. 2018, 102, 443–479. [Google Scholar] [CrossRef] [PubMed]
- Berger, C.; Langsetmo, L.; Joseph, L.; Hanley, D.A.; Davison, K.S.; Josse, R.; Kreiger, N.; Tenenhouse, A.; Goltzman, D. Change in bone mineral density as a function of age in women and men and association with use of antiresorptive agents. CMAJ 2008, 178, 1660–1668. [Google Scholar] [CrossRef] [PubMed]
- Lucas, S.; Omata, Y.; Hofmann, J.; Bottcher, M.; IIjazovic, A.; Starter, K.; Albrecht, O.; Schulz, O.; Krishnacoumar, B.; Kronke, G.; et al. Short-chain fatty acids regulate systemic bone mass and protect from pathological bone loss. Nat. Commun. 2018, 9, 55. [Google Scholar] [CrossRef] [PubMed]
- McGartland, C.P.; Robson, P.J.; Murray, L.J.; Cran, G.W.; Savage, M.J.; Watkins, D.C.; Rooney, M.M.; Boreham, C.A. Fruit and vegetable consumption and bone mineral density: The Northern Ireland Young Hearts Project. Am. J. Clin. Nutr. 2004, 80, 1019–1023. [Google Scholar] [CrossRef] [PubMed]
- Prynne, C.J.; Mishra, G.D.; O’Connell, M.A.; Muniz, G.; Laskey, M.A.; Yan, L.; Prentice, A.; Ginty, F. Fruit and vegetable intakes and bone mineral status: A cross-sectional study in 5 age and sex cohorts. Am. J. Clin. Nutr. 2006, 83, 1420–1428. [Google Scholar] [CrossRef] [PubMed]
- Dai, Z.; Zhang, Y.; Lu, N.; Felson, D.T.; Kiel, D.P.; Sahni, S. J. Association between dietary fiber intake and bone loss in the Framingham Offspring Study. J. Bone Min. Res. 2018, 33, 241–249. [Google Scholar] [CrossRef] [PubMed]
- Qiu, R.; Cao, W.-T.; Tian, H.-Y.; He, J.; Chen, G.D.; Ghen, Y.M. Greater intake of fruit and vegetables is associated with greater bone mineral density and lower osteoporosis risk in middle-aged and elderly adults. PLoS ONE 2017, 12, e0168906. [Google Scholar] [CrossRef] [PubMed]
- Li, J.-J.; Huang, Z.-W.; Wang, R.-Q.; ma, X.M.; Zhang, Z.Q.; Liu, Z.; Chen, Y.M.; Su, Y.X. Fruit and vegetable intake and bone mass in Chinese adolescents, young and postmenopausal women. Public Health Nutr. 2012, 16, 78–86. [Google Scholar] [CrossRef] [PubMed]
- Hooshmand, S.; Chai, S.C.; Saadat, R.L.; Payton, M.E.; Brummel-Smith, K.; Arjmandi, B.H. Comparative effects of dried plum and dried apple on bone in postmenopausal women. Br. J. Nutr. 2011, 106, 923–930. [Google Scholar] [CrossRef] [PubMed]
- Hooshmand, S.; Kern, M.; Metti, D.; Shamloufard, P.; Chai, S.C.; Johnson, S.A.; Payton, M.E.; Arjmandi, B.H. The effect of two doses of dried plum on bone density and bone biomarkers in osteopenic postmenopausal women: A randomized, controlled trial. Osteoporos Int. 2016, 27, 2271–2279. [Google Scholar] [CrossRef] [PubMed]
- Sanders, M.G.H.; Pardo, L.M.; Ginger, R.S.; Kiefte-de Jong, K.C; Nijsten, T. Association between diet and seborrheic dermatitis: A cross-sectional study. J. Investig. Dermatol. 2018. [Google Scholar] [CrossRef] [PubMed]
- Pascal, M.; Perez-Gordo, M.; Caballero, T.; Escribese, M.M.; Lopez Longo, M.N; Luengo, O.; Manso, L.; Mateu, V.; Seoane, E.; Zamorano, M.; et al. Microbiome and allergic disease. Front. Immunol. 2018, 9, 1584. [Google Scholar] [CrossRef] [PubMed]
- Grimaldi, R.; Cela, D.; Swann, J.R.; Vulevic, J.; Gibson, G.R.; Tzortzis, G.; Costabile, A. In vitro fermentation of B-GOS: Impact of faecal bacterial populations and metabolic activity in autistic and non-autistic children. FEMS Microbiol. Ecol. 2017, 93, fiw233. [Google Scholar] [CrossRef] [PubMed]
- Sanctuary, M.R.; Kain, J.N.; Angkustsiri, K.; German, J.B. Dietary considerations in autism spectrum disorders: The potential role of protein digestion and microbial putrefaction in the gut-brain axis. Front. Nutr. 2018, 5, 40. [Google Scholar] [CrossRef] [PubMed]
- Berding, K.; Donovan, S.M. Diet can impact microbiota composition in children with autism spectrum disorder. Front. Neurosci. 2018. [Google Scholar] [CrossRef]
- Sharp, W.G.; Postorino, V.; McCracken, C.E.; Berry, R.C.; Criado, K.K.; Burrell, T.L; Scahill, L. Dietary intake, nutrient status, and growth parameters in children with autism spectrum disorder and severe food sensitivity: An electronic medical record review. J. Acad. Nutr. Diet. 2018, 118, 1943–1950. [Google Scholar] [CrossRef] [PubMed]
- Grimaldi, R.; Gibson, G.R.; Vulevic, J.; Giallourou, N.; Castro-Mejia, J.L.; Hansen, L.H.; Leigh Gibson, E.; Nielsen, D.S.; Costabile, A. A prebiotic intervention study in children with autism spectrum disorders (ASDs). Microbiome 2018, 6, 133. [Google Scholar] [CrossRef] [PubMed]
- Saavedra, M.J.; Aziz, J.; Cacchiarelli San Roman, N. Scurvy due to restrictive diet in a child with autism spectrum disorder: Case report. Arch. Argent. Pediatr. 2018, 116, e684–e687. [Google Scholar] [CrossRef] [PubMed]
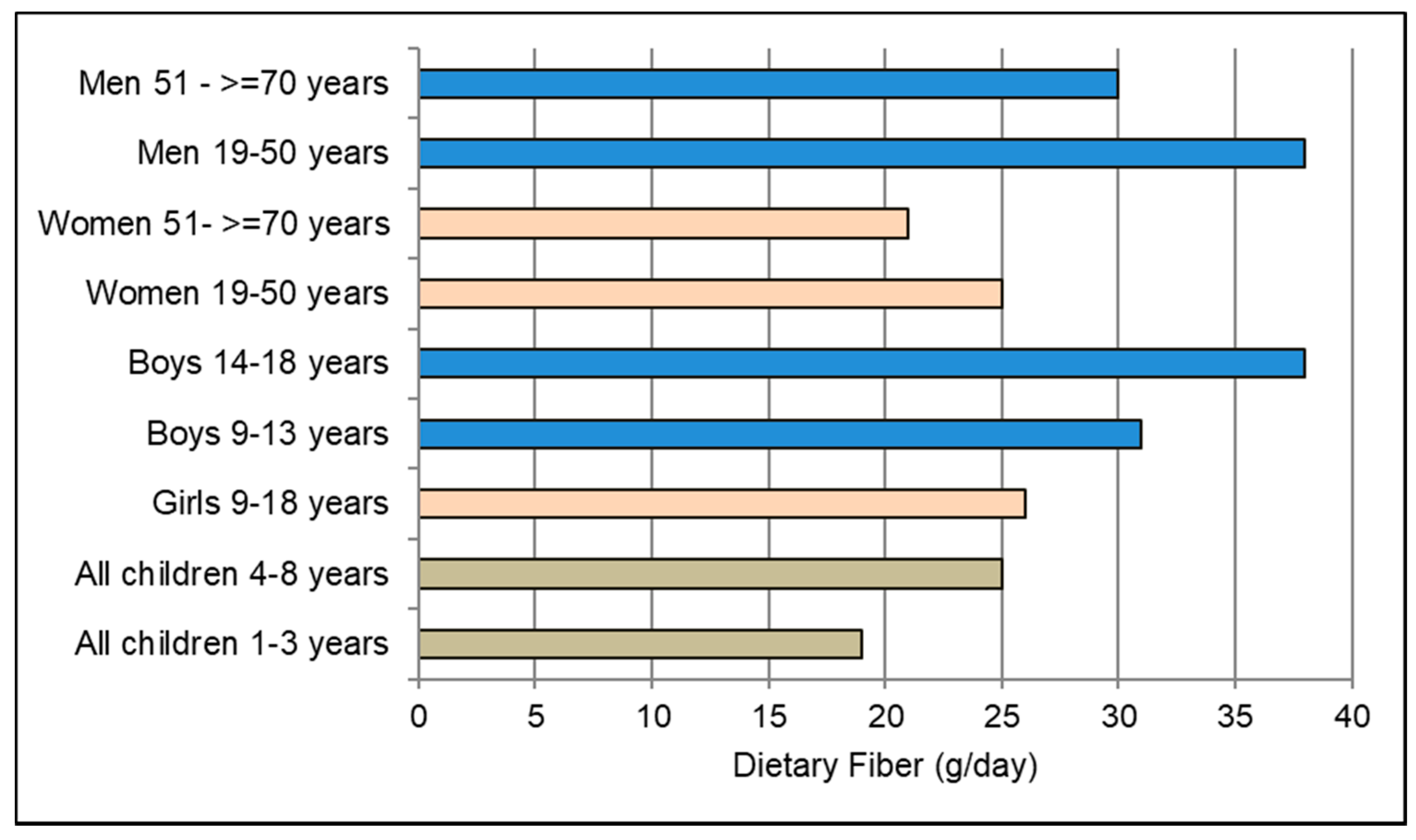
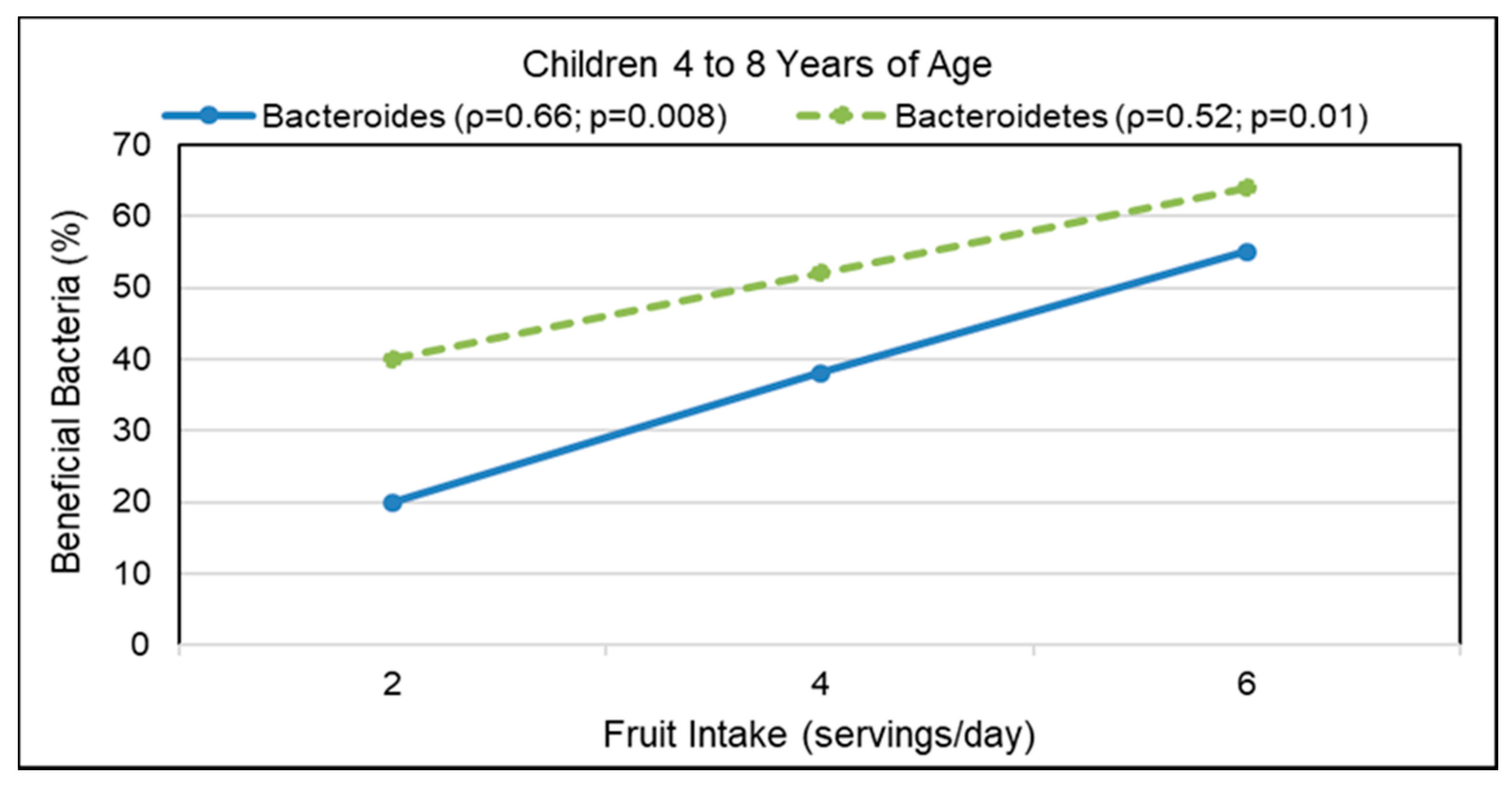
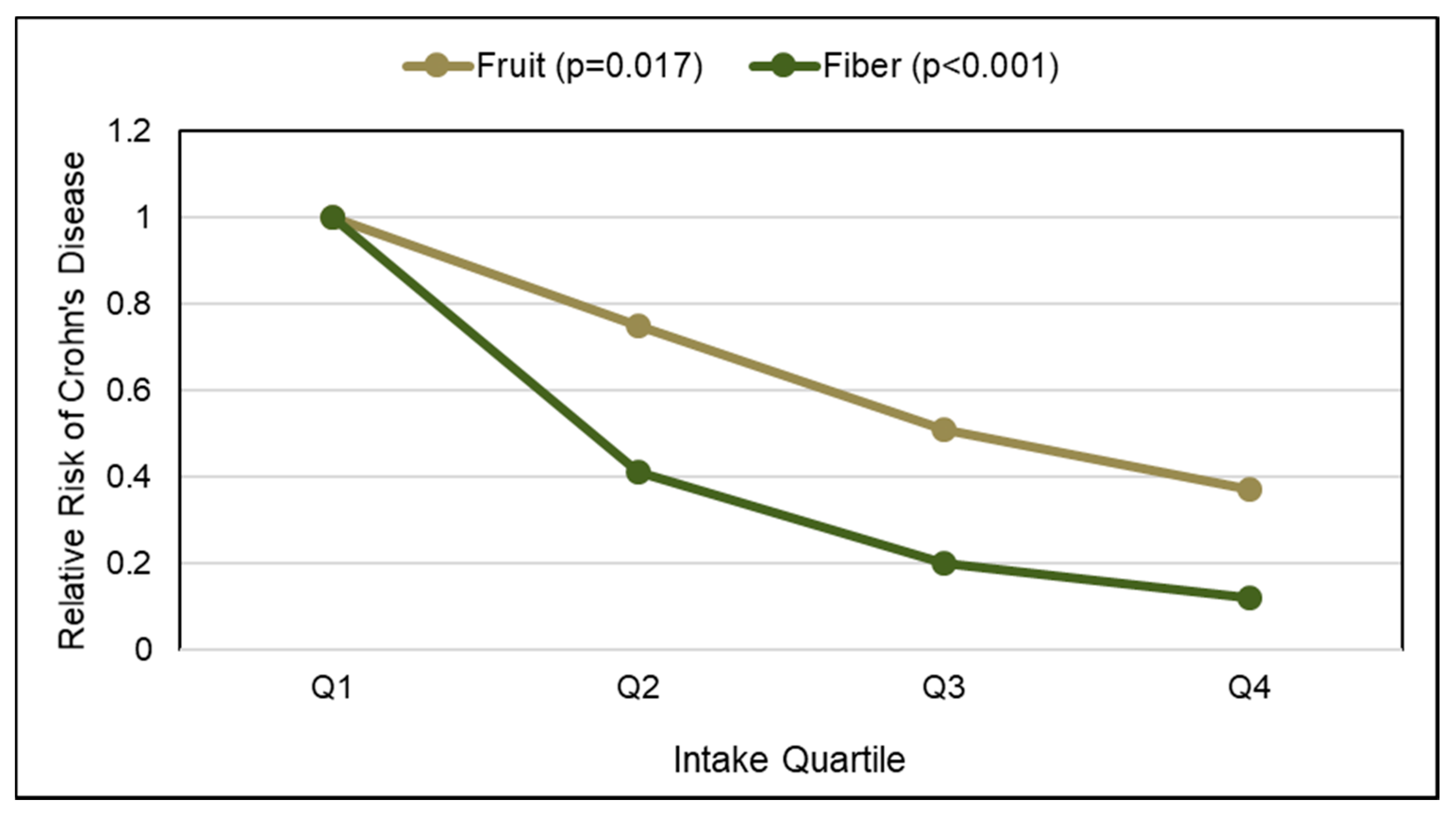
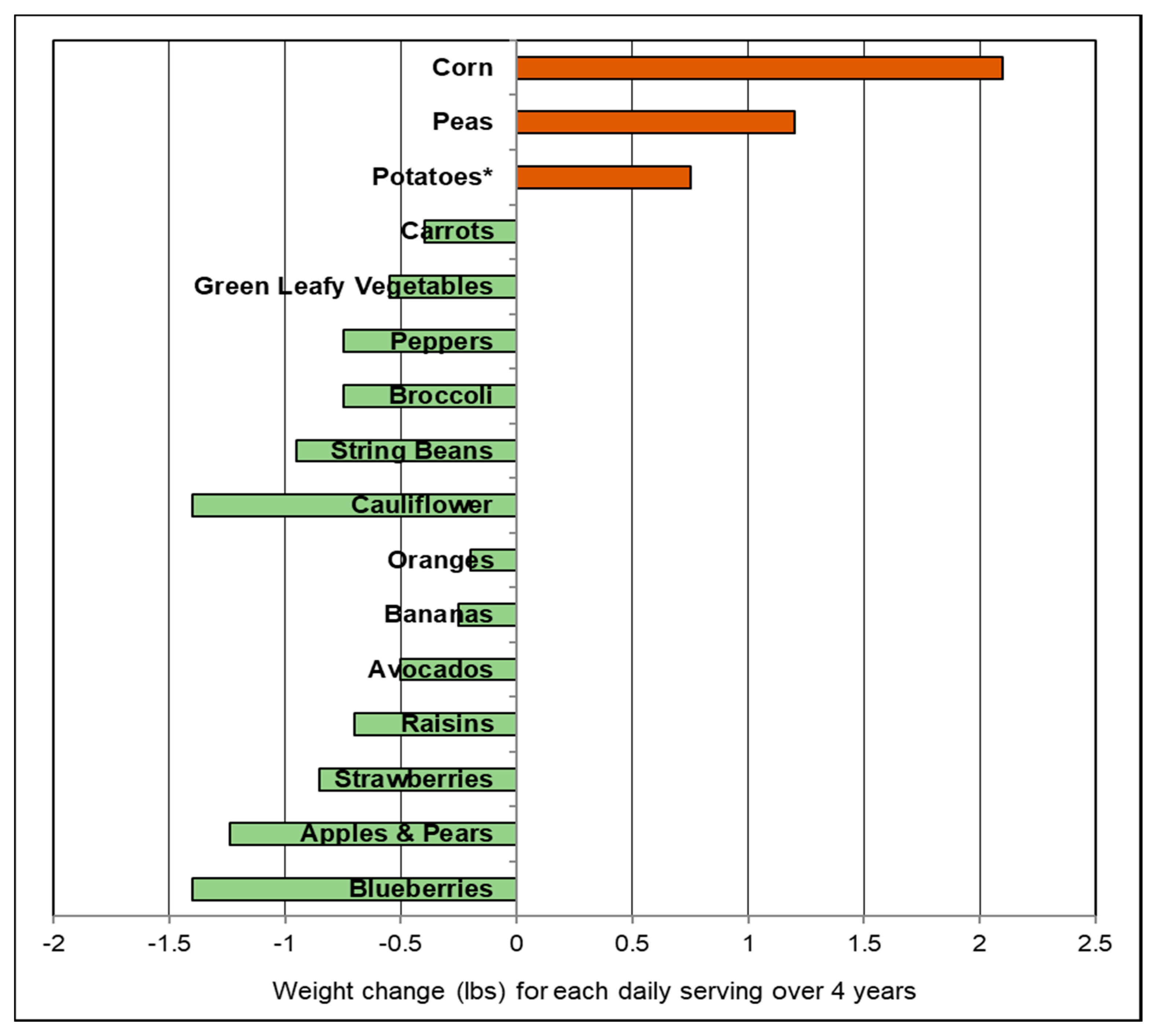
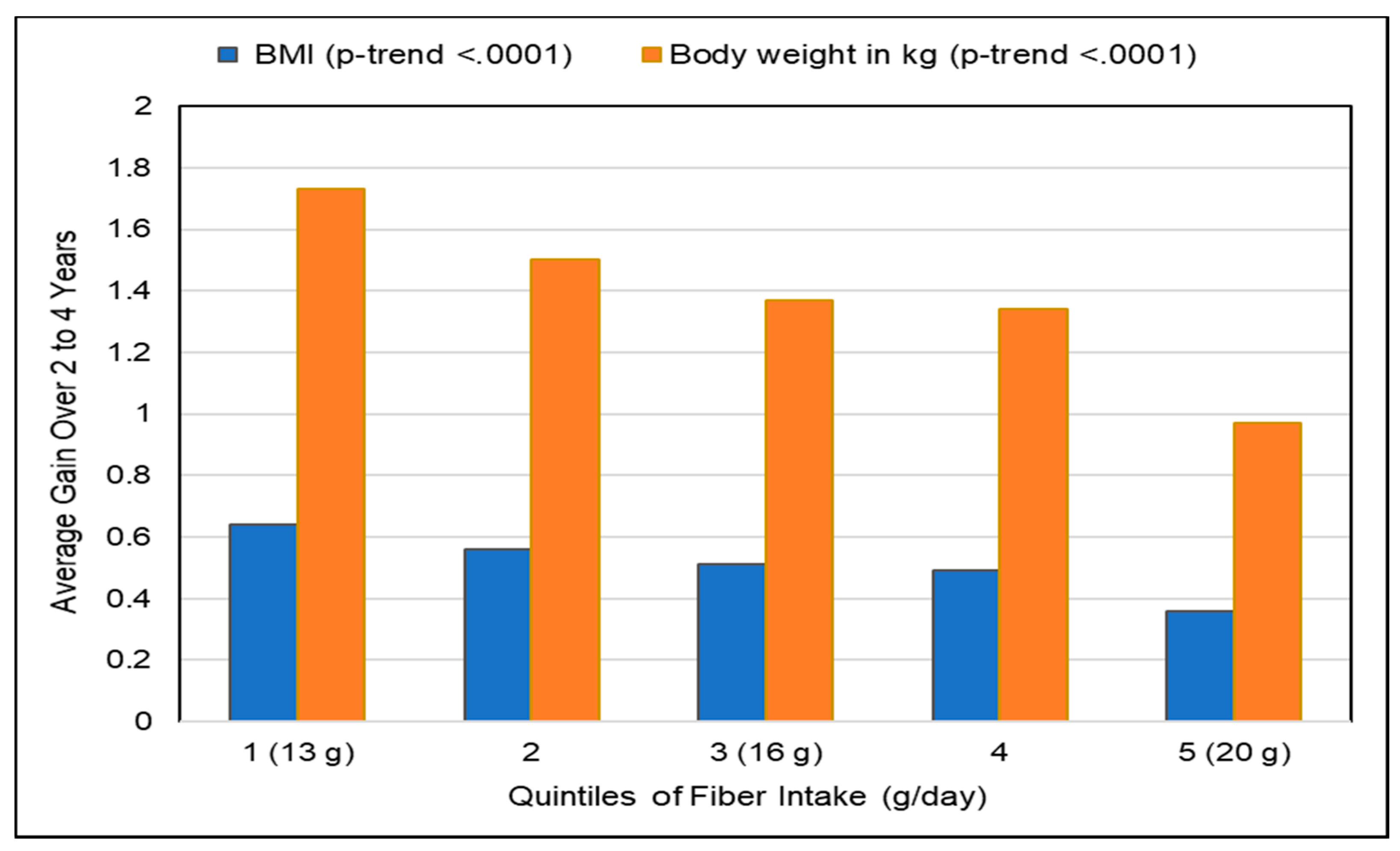
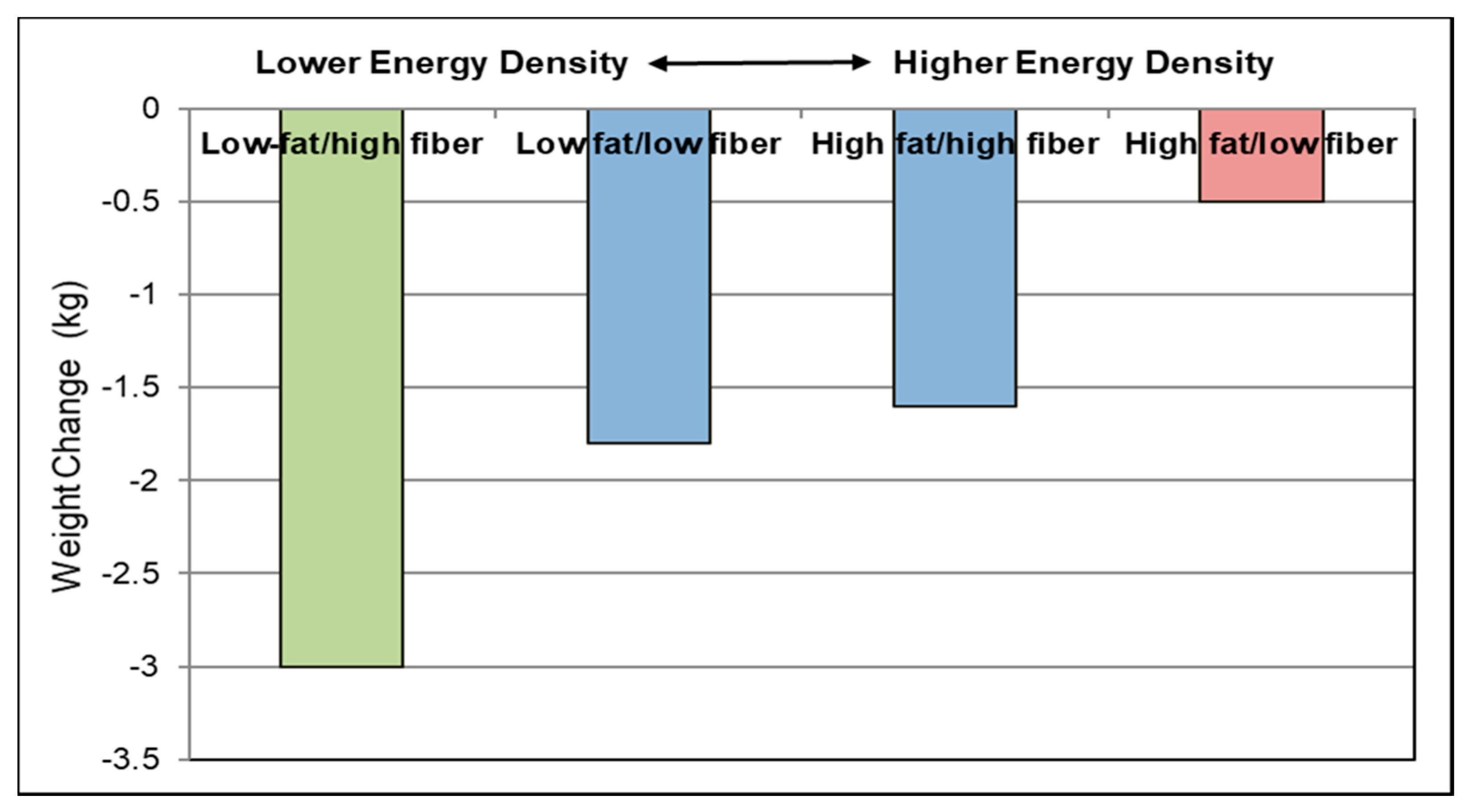
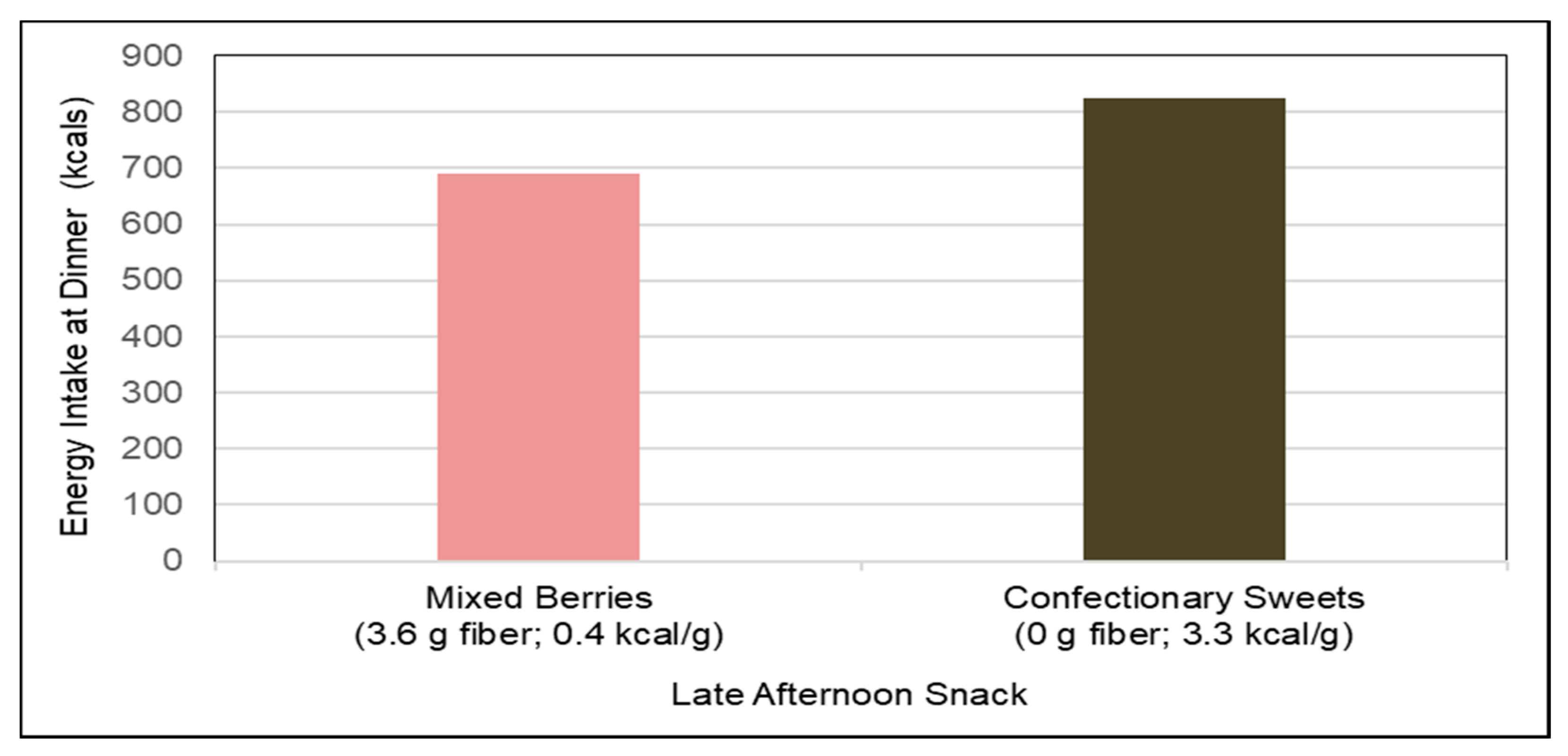
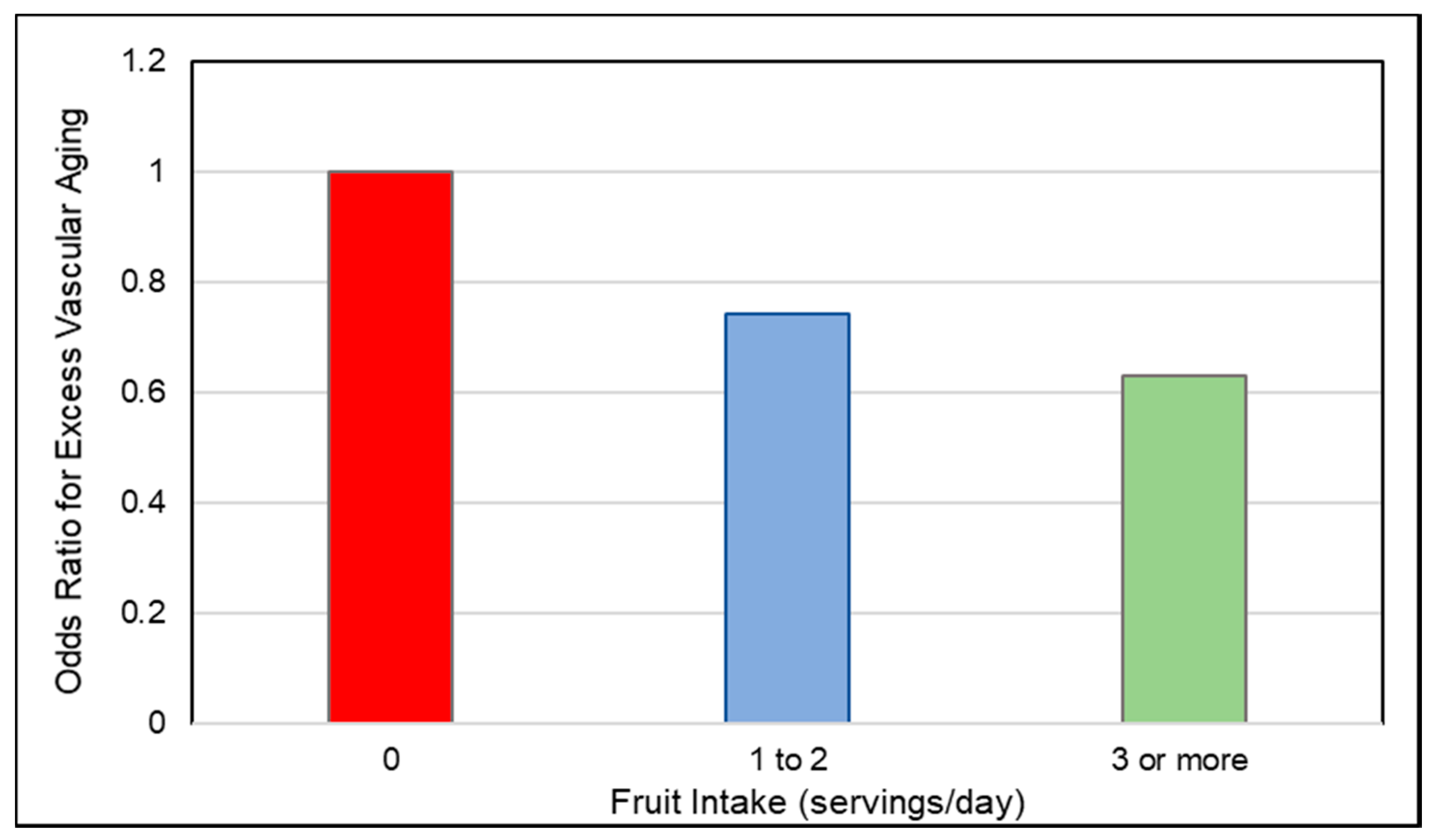
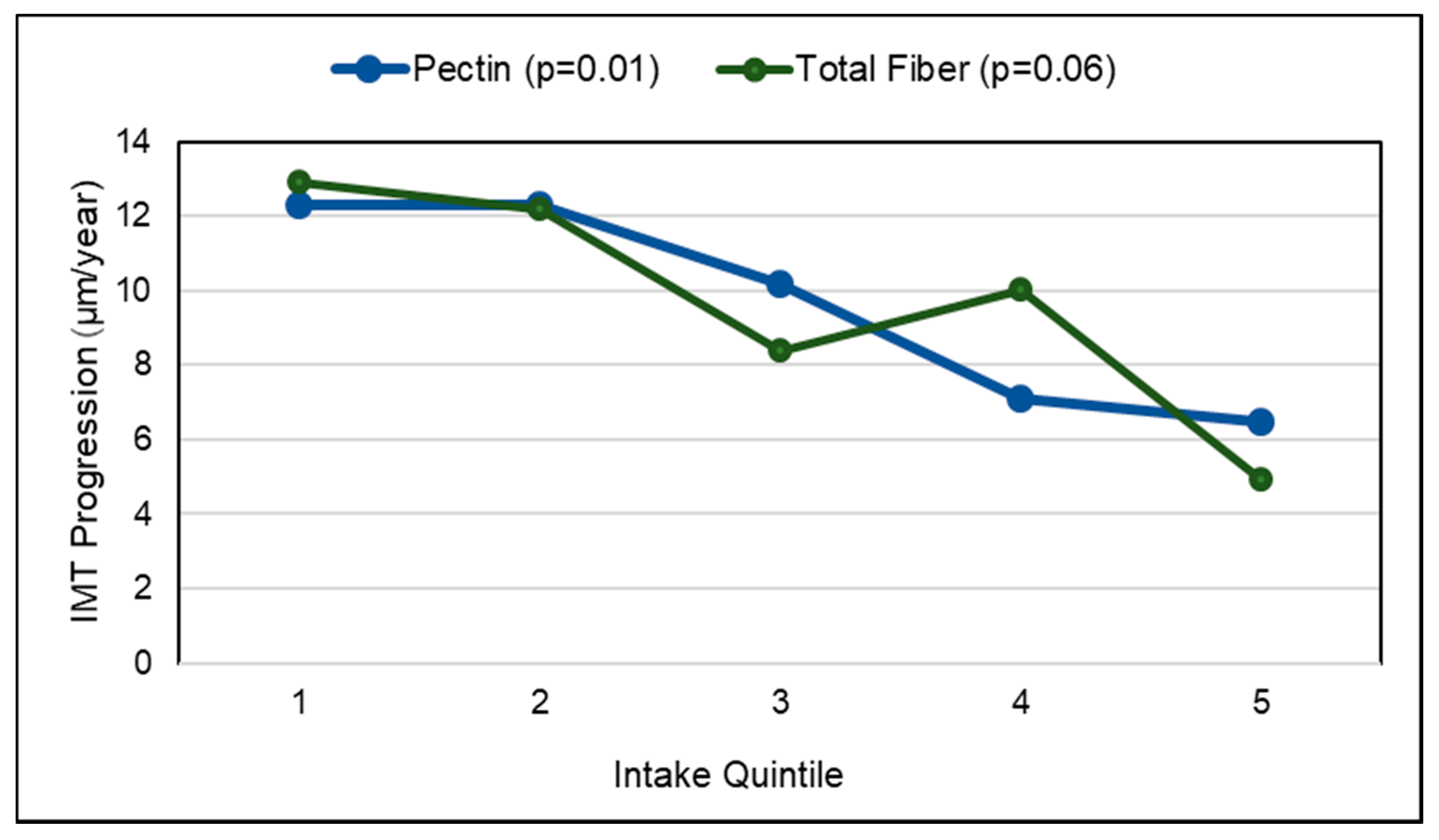
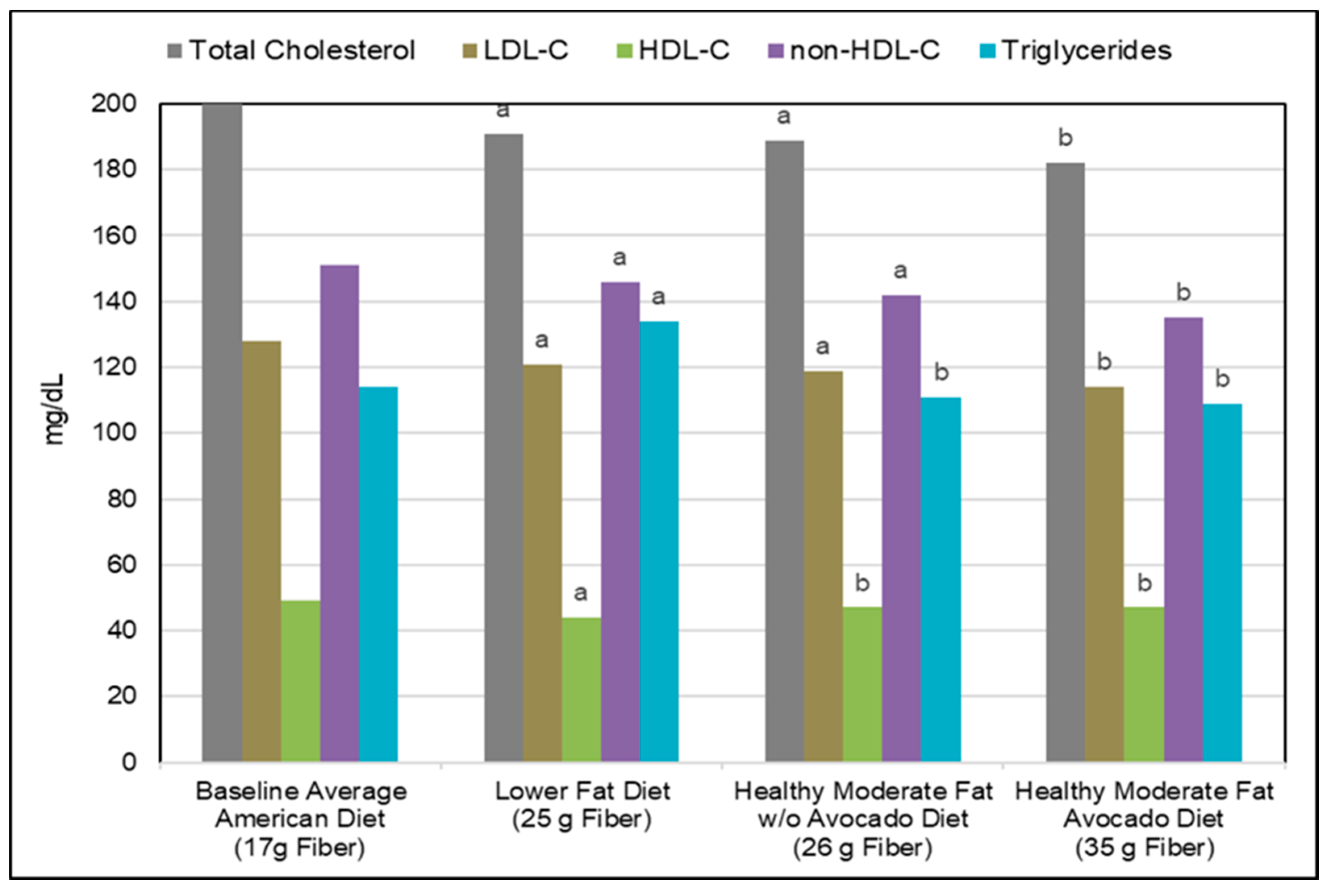
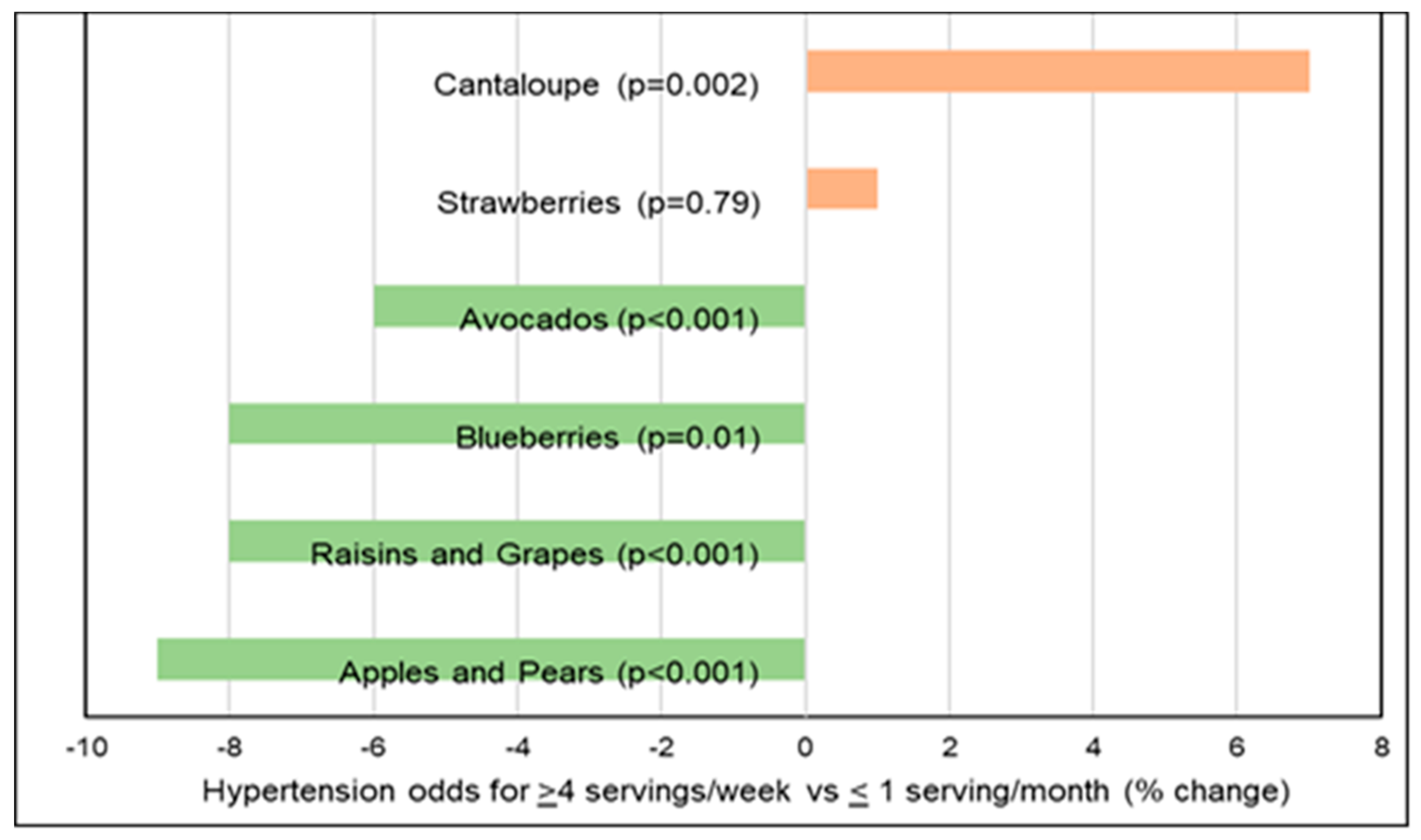
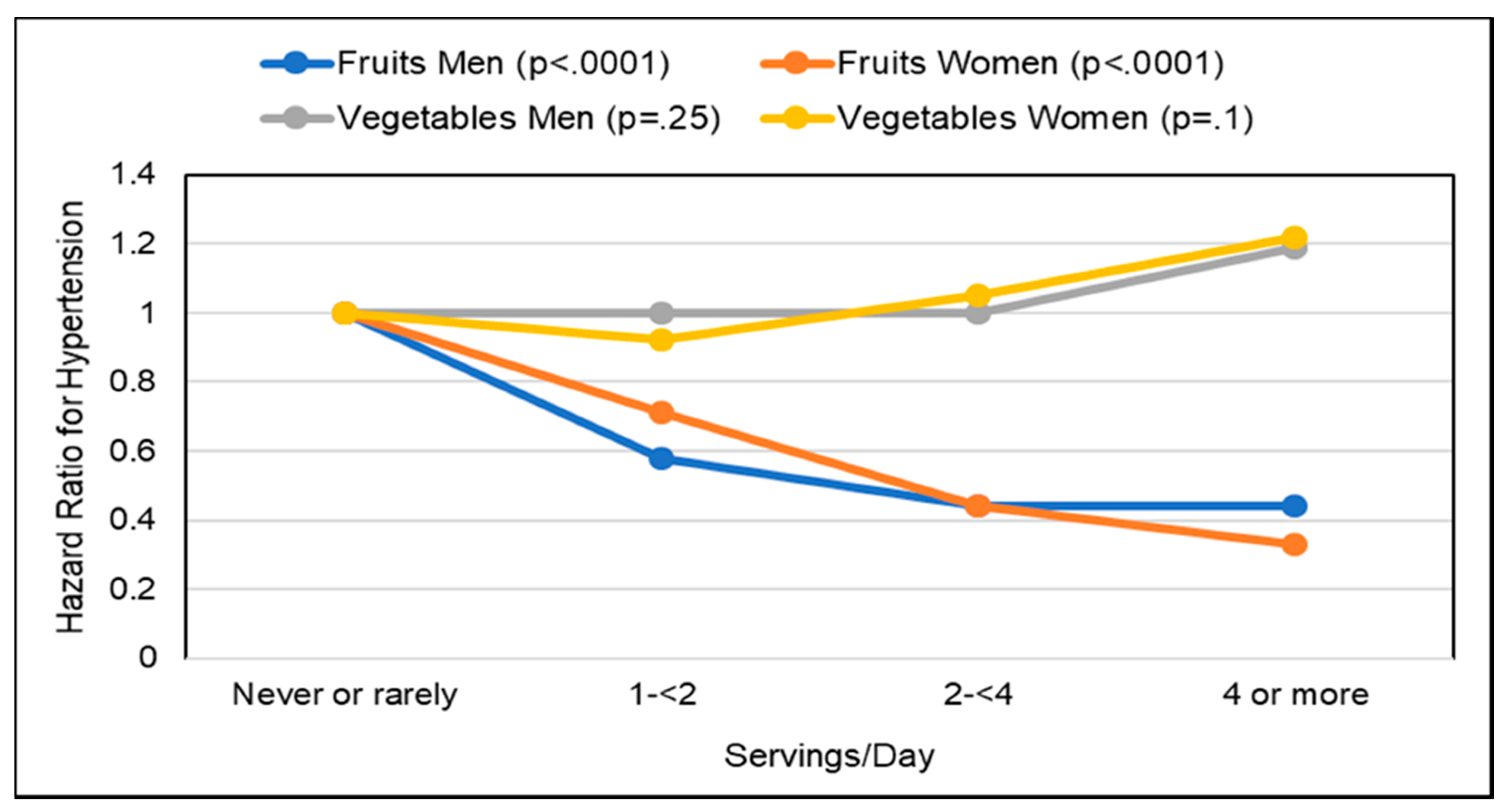

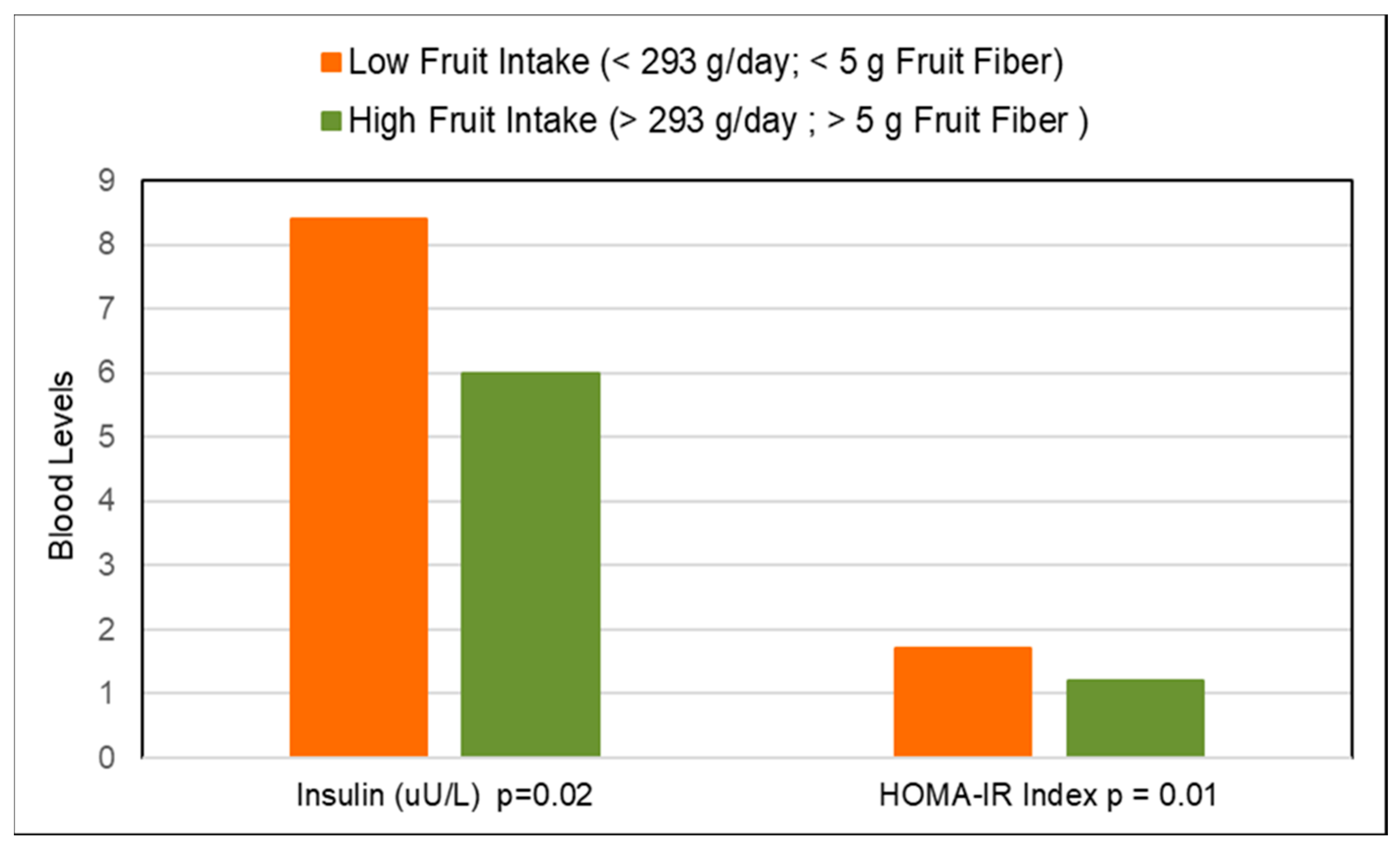
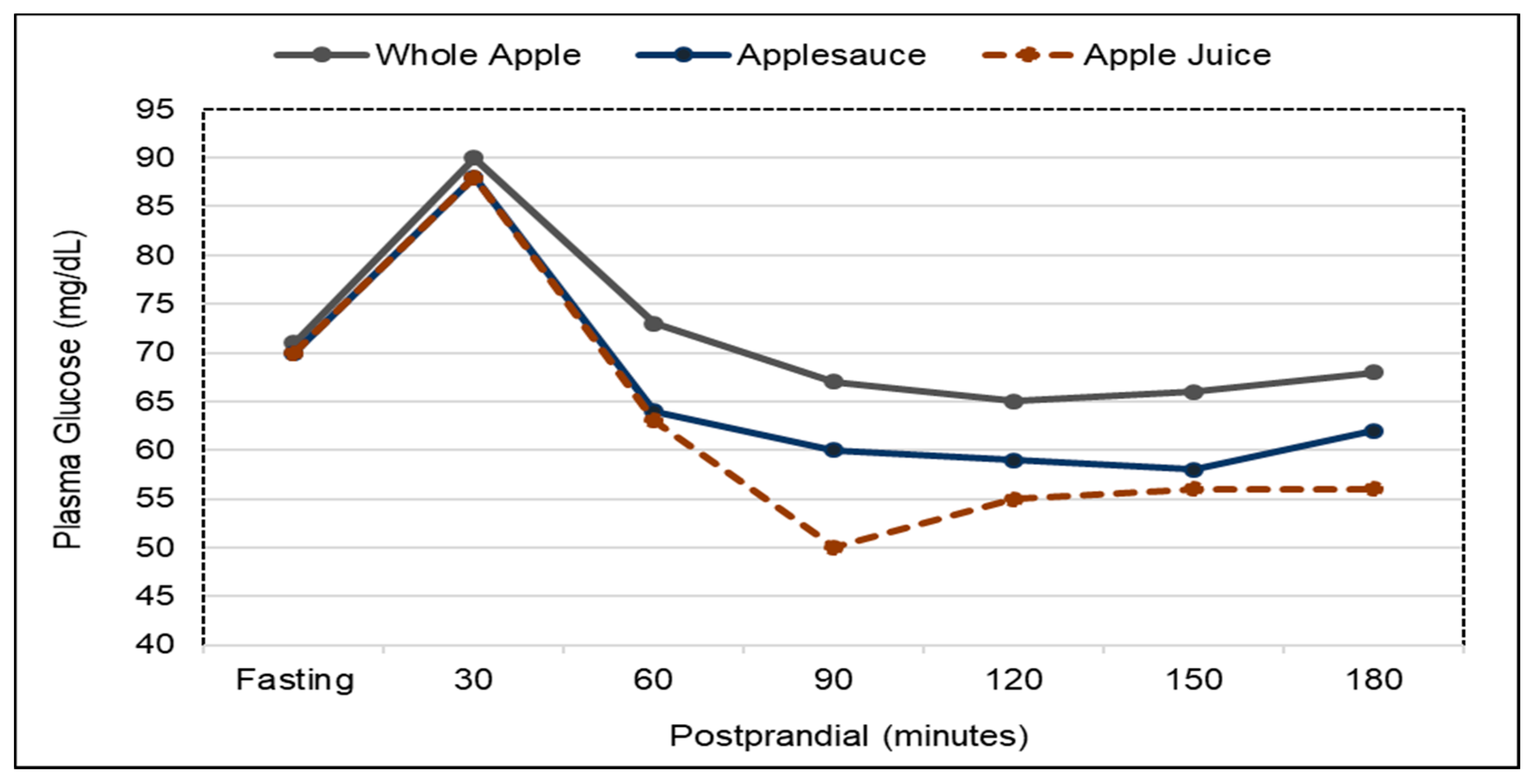
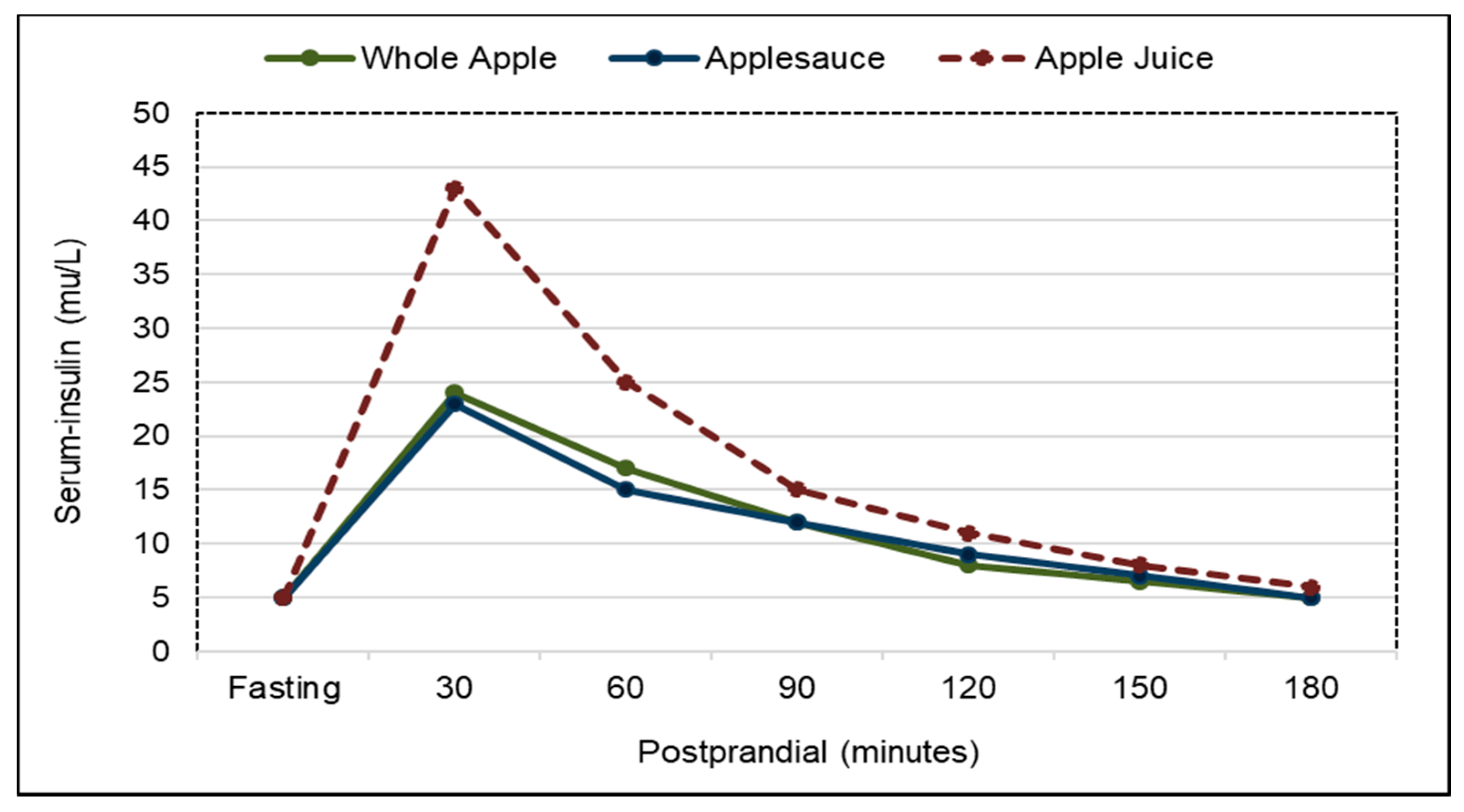
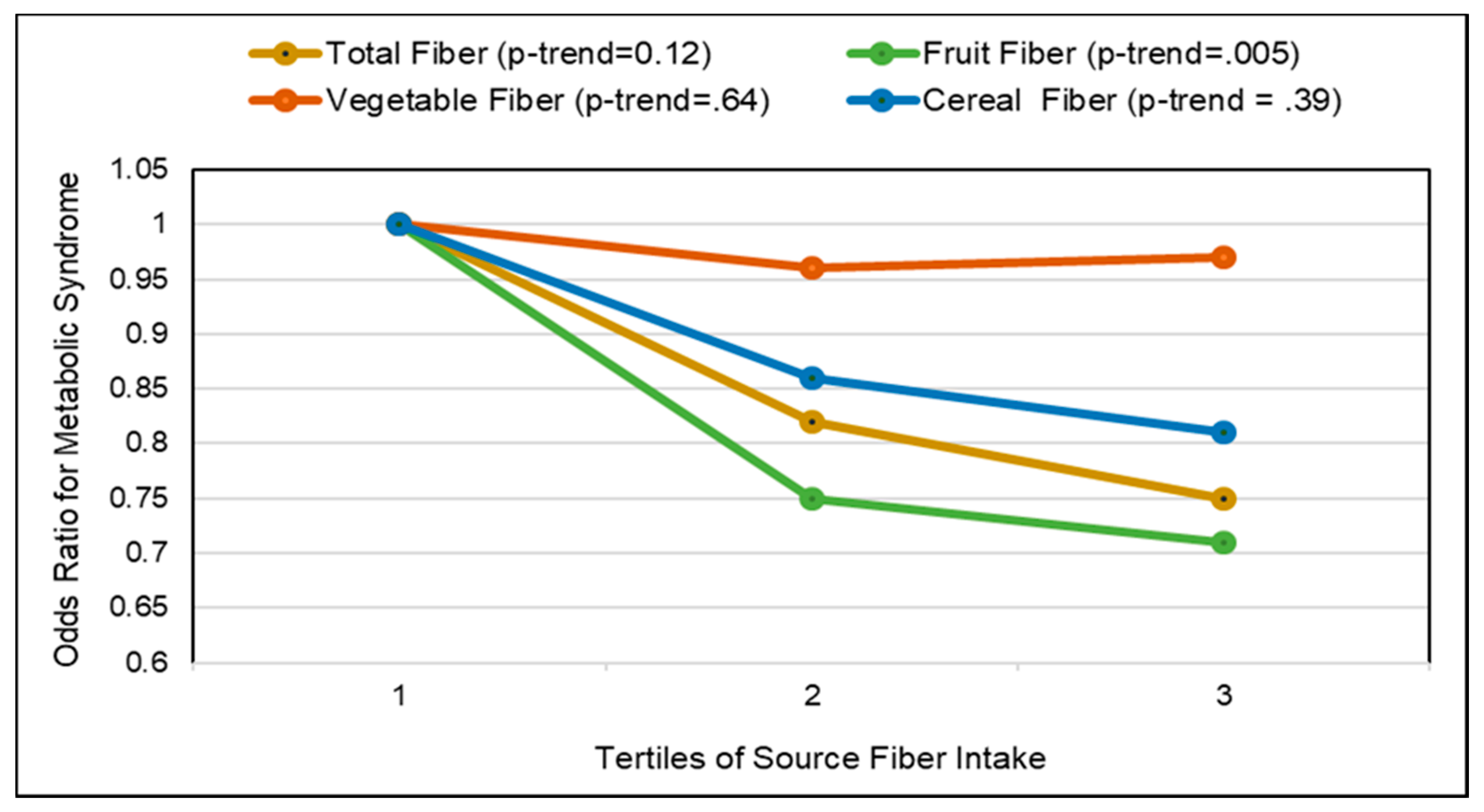
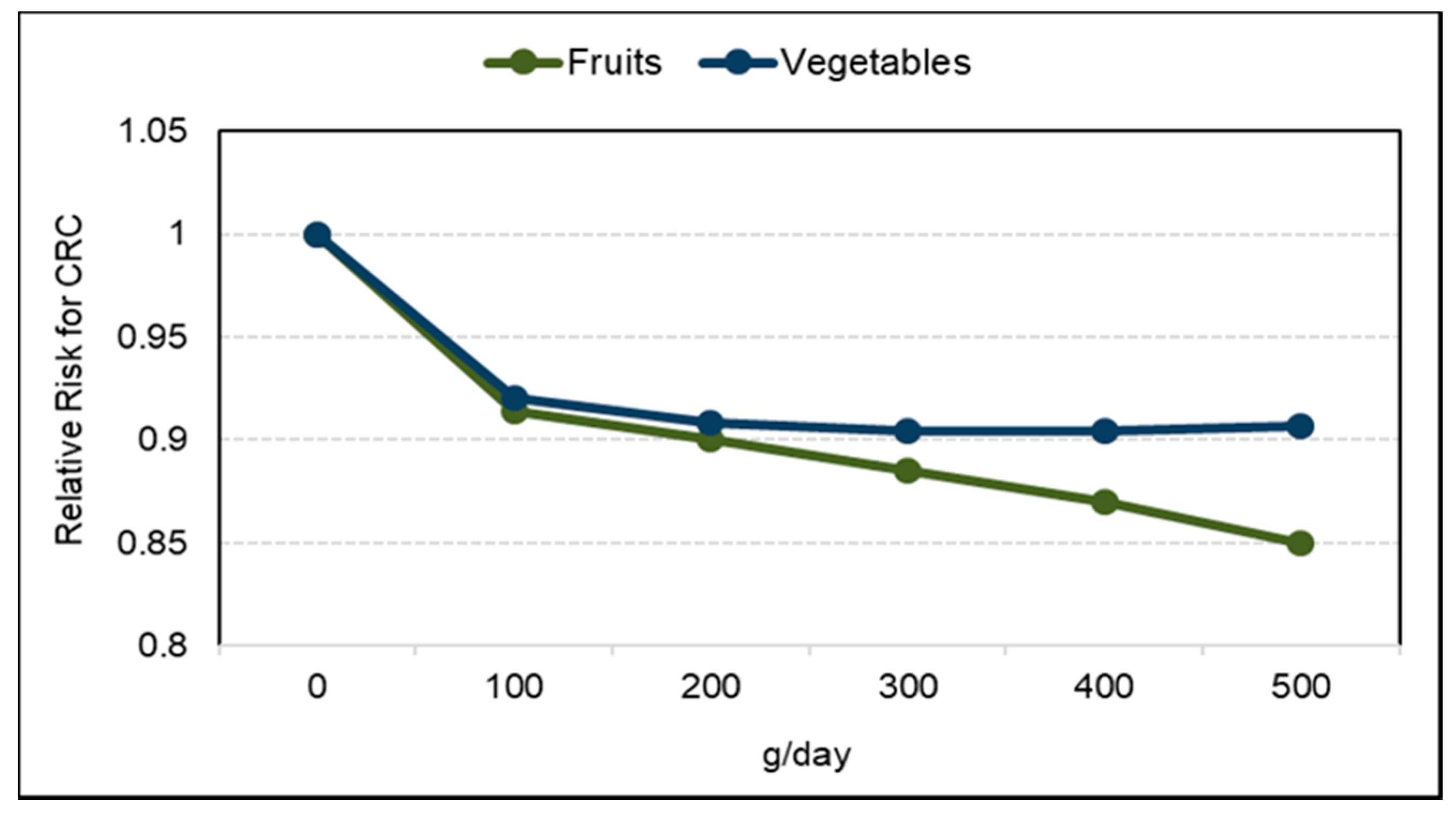
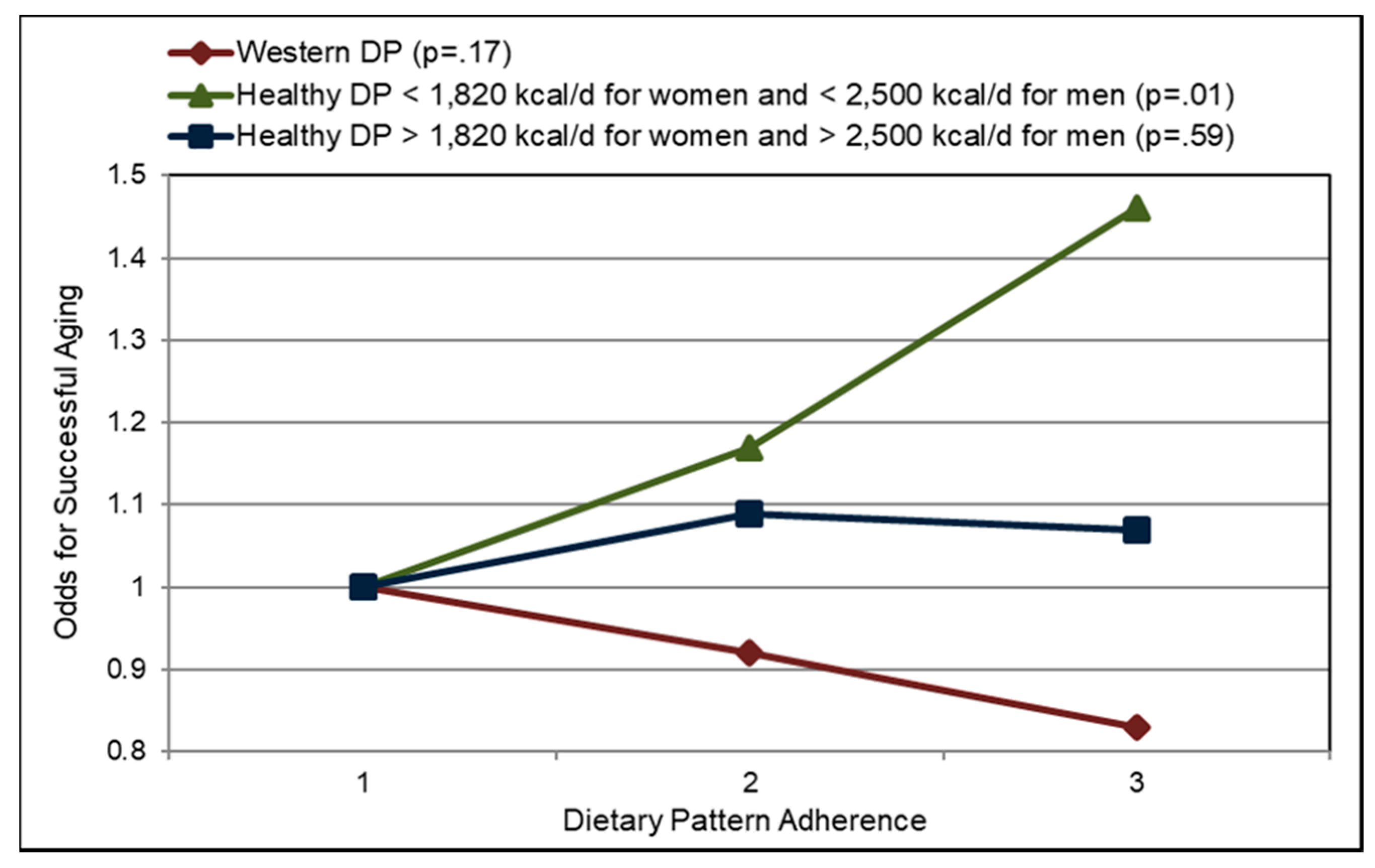
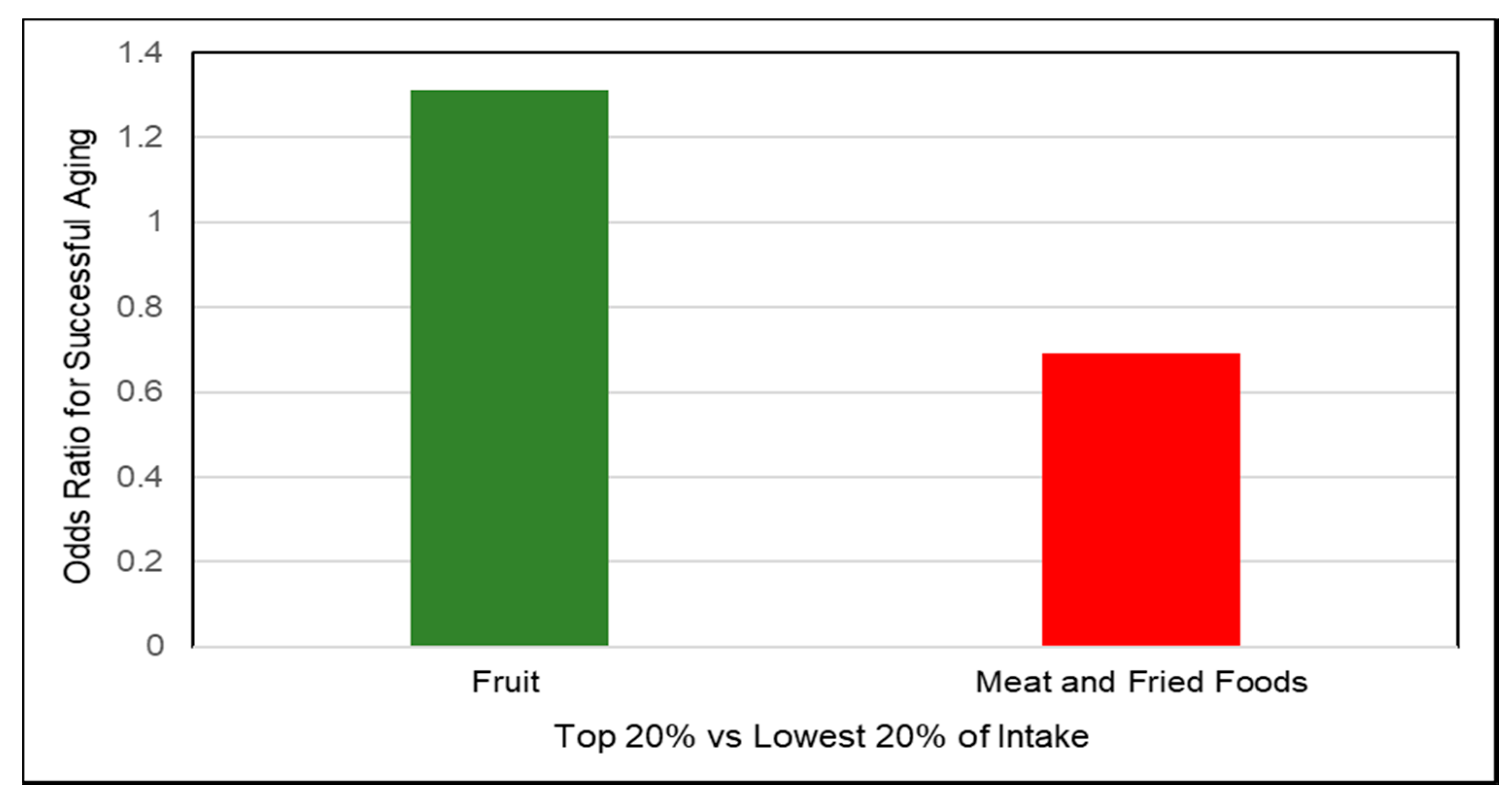
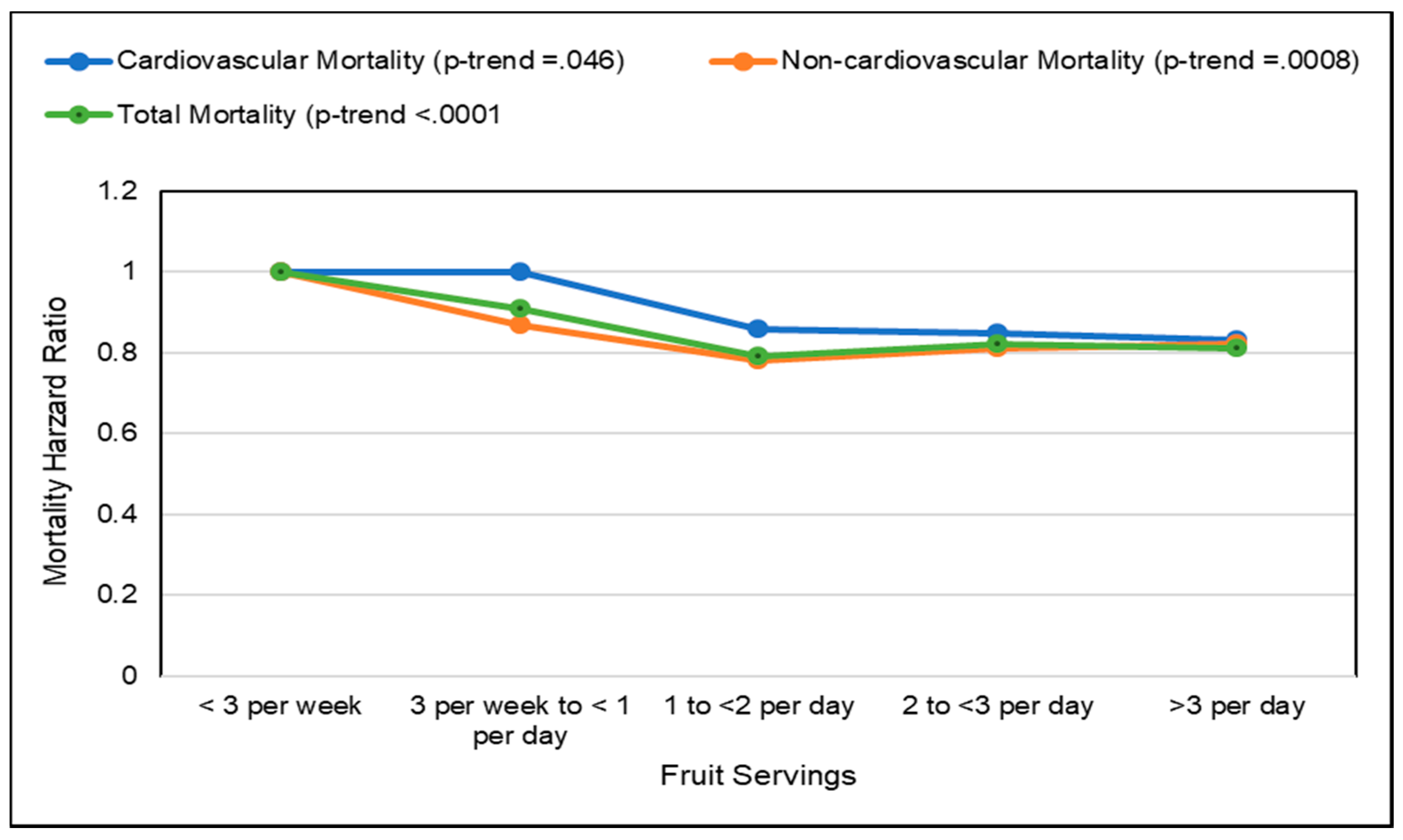
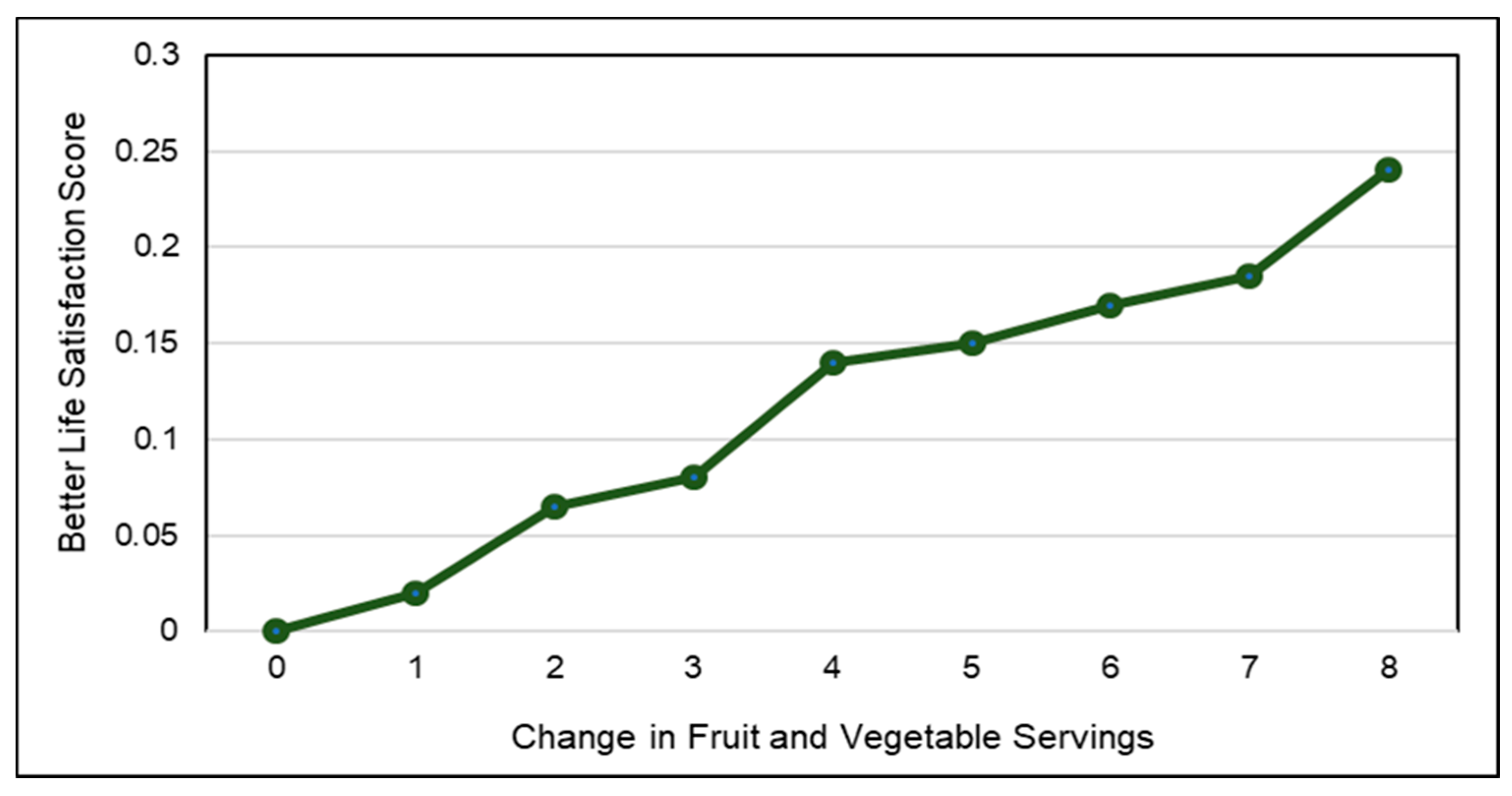
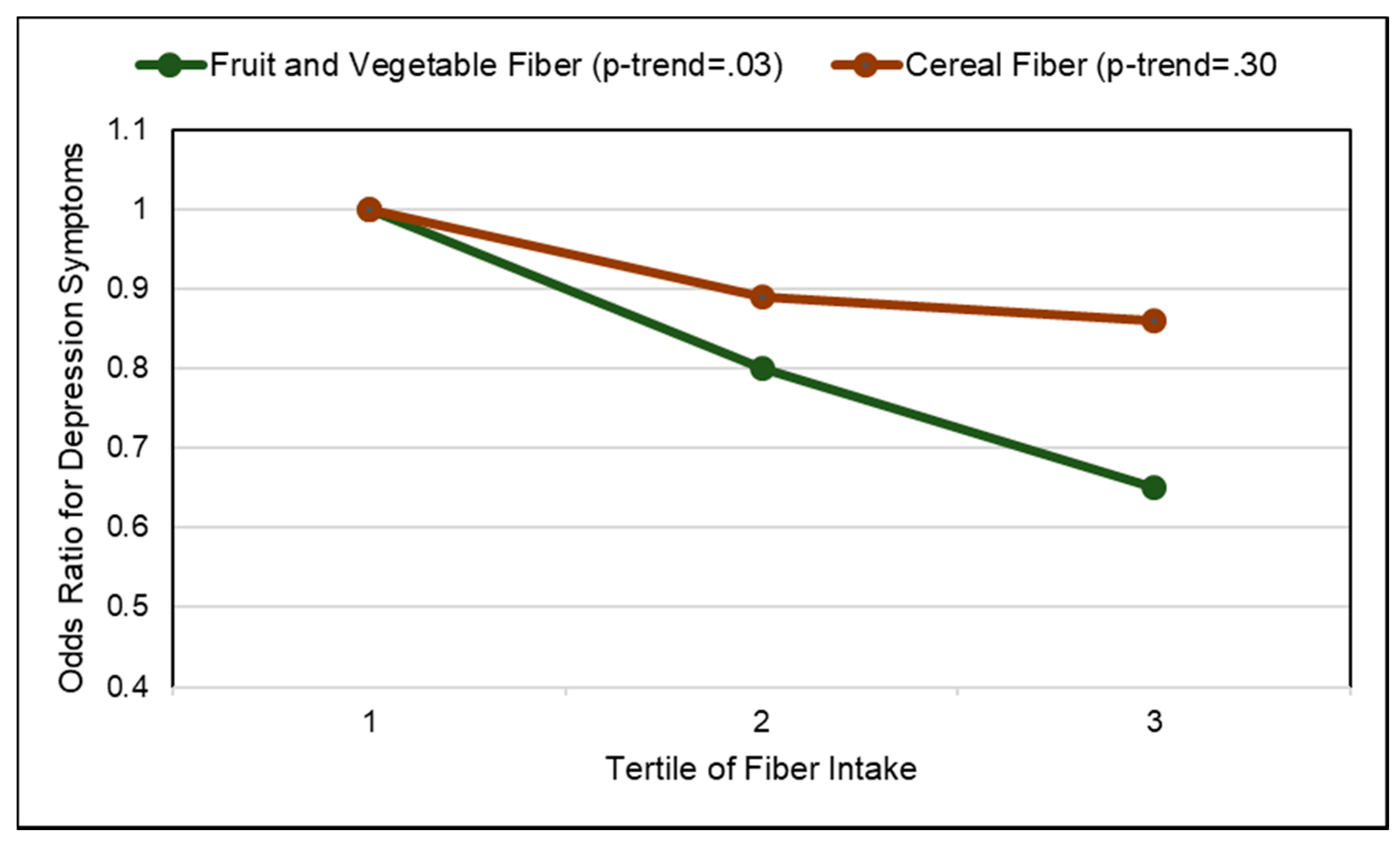
| Fruit Source | Serving Size * | Estimated Fiber Components | Sugar (Starch) (100-g) | Energy Density (kcal/g) | Energy (kcal) 100-g | |||
|---|---|---|---|---|---|---|---|---|
| Total (100-g) | Insoluble (100-g) | Soluble (100-g) | Pectin ** (100-g) | |||||
| Fresh Fruit | ||||||||
| Apples | 1 (182 g) | 2.4 | 1.7 | 0.5 | 0.8 | 10 | 0.5 | 52 |
| Avocados | 1/3 (50 g) | 6.8 | 4.4 | 2.4 | 2.4 | < 1.0 | 1.6 | 160 |
| Bananas | 1 (118 g) | 2.6 | 1.8 | 0.8 | 0.6 | 12 (5) | 0.9 | 89 |
| Blackberries | 1 cup(144 g) | 5.3 | 4.7 | 0.6 | 1.4 | 4.9 | 0.4 | 43 |
| Blueberries | 1 cup(148 g) | 2.8 | 2.4 | 0.3 | 0.8 | 10 | 0.6 | 57 |
| Cherries | 1 cup(138 g) | 2.2 | 1.6 | 0.6 | 0.7 | 13 | 0.6 | 63 |
| Figs | 1 (100 g) | 3.0 | 2.4 | 0.6 | 1.0 | 16 | 0.7 | 74 |
| Grapefruits | 1 cup(154 g) | 1.6 | 1.1 | 0.5 | 0.6 | 7.0 | 0.4 | 42 |
| Guavas | 1 cup(165 g) | 5.4 | 4.2 | 1.8 | 1.5 | 9.0 | 0.7 | 68 |
| Kiwis | 2 (138 g) | 3.0 | 2.2 | 0.9 | 0.7 | 9.0 | 0.6 | 61 |
| Mangoes | 1 cup(165 g) | 1.6 | 1.0 | 0.6 | 0.5 | 14 | 0.6 | 60 |
| Oranges | 1 (131 g) | 2.4 | 1.4 | 1.0 | 0.8 | 9.1 | 0.5 | 47 |
| Papayas | 1 cup(145 g) | 1.7 | 1.4 | 0.3 | 0.5 | 7.6 | 0.4 | 43 |
| Pears | 1 (166 g) | 3.1 | 2.2 | 0.9 | 1.0 | 10 | 0.6 | 60 |
| Plantains | ½ (134 g) | 2.2 | 1.5 | 0.7 | 0.4 | 2.2 (30) | 1.5 | 149 |
| Pomegranate arils | 1 cup(122 g) | 5.7 | 4.1 | 1.6 | 2.0 | 12 | 0.8 | 81 |
| Raspberries | 1 cup(123 g) | 6.5 | 5.3 | 1.2 | 1.6 | 4.4 | 0.5 | 52 |
| Strawberries | 1 cup(152 g) | 2.0 | 1.5 | 0.5 | 0.7 | 4.7 | 0.3 | 33 |
| Apricots | 2 (70 g) | 2.0 | 1.0 | 1.0 | 0.7 | 9.1 | 0.5 | 49 |
| Cantaloupes | 1 cup(177 g) | 0.9 | 0.6 | 0.3 | 0.3 | 7.9 | 0.3 | 34 |
| Green grapes | 1 cup (92 g) | 0.9 | 0.6 | 0.3 | 0.2 | 17 | 0.7 | 69 |
| Peaches | 1 (150 g) | 1.5 | 0.9 | 0.6 | 0.5 | 9.0 | 0.4 | 39 |
| Pineapples | 1 cup(165 g) | 1.4 | 1.1 | 0.3 | 0.5 | 10 | 0.5 | 50 |
| Plums | 2 (132 g) | 1.4 | 0.9 | 0.5 | 0.4 | 11 | 0.5 | 45 |
| Watermelons | 1 wedge (286 g) | 0.4 | 0.3 | 0.1 | 0.1 | 6.3 | 0.3 | 29 |
| Dried Fruit | ||||||||
| Apricots | 6 (40 g) | 10 | 7.5 | 2.5 | 3.0 | 37 | 2.6 | 258 |
| Cranberries | ¼ cup(40 g) | 7.5 | 5.0 | 2.5 | 2.5 | 72 | 3.3 | 325 |
| Dates, pitted | 5–6 (40 g) | 7.5 | 6.0 | 1.5 | 2.5 | 72 | 3.0 | 300 |
| Dried Figs | 1/3 cup (40 g) | 12 | 8.8 | 37 | 4.3 | 50 | 2.8 | 275 |
| Prunes | 7 (40 g) | 7.5 | 5.0 | 2.5 | 2.5 | 37 | 2.5 | 250 |
| Raisins | ¼ cu(40 g) | 5.0 | 3.7 | 1.2 | 1.7 | 73 | 3.0 | 300 |
| Variable | Baseline Value | 4-Week Value | % Change | p-Value |
|---|---|---|---|---|
| Total cholesterol (mg/dL) | 217 | 183 | −34 | <0.0005 |
| LDL-C (mg/dL) | 143 | 118 | −25 | <0.0005 |
| HDL-C (mg/dL) | 55 | 105 | −5 | <0.0005 |
| Triglycerides (mg/dL) | 124 | 105 | −20 | =0.008 |
| Insulin (µIU/mL) | 14.6 | 10.3 | −4.3 | <0.0005 |
| Glucose (mg/dL) | 90 | 87 | −2.9 | =0.25 |
| HbA1c (%) | 5.9 | 5.7 | −0.2 | =0.002 |
| hs-CRP (mg/L) | 7.8 | 5.3 | −2.5 | =0.001 |
| Weight (kg) | 108 | 101 | −7.0 | <0.0005 |
| BMI (kg/m2) | 37.5 | 35.2 | −2.3 | <0.0005 |
| Diastolic BP (mm Hg) | 91 | 82 | −9.0 | <0.0005 |
| Systolic BP (mm Hg) | 147 | 130 | −17 | <0.0005 |
| Energy (kcal/day) | 2053 | 1369 | −684 | <0.0005 |
| Dietary Fiber (g/day) | 20 | 51 | +31 | <0.0005 |
| Variables | <2 Fruit Servings/Day | 2 Fruit Servings/Day | >2 Fruit Servings/Day | p-Trend |
|---|---|---|---|---|
| BMI (kg/m2) | 27.9 ± 5.8 | 26.9 ± 5.8 | 25.9 ± 5.0 | <0.0001 |
| CRP (mg/L) | 1.80 (1.55–2.08) | 1.42 (1.26–1.62) | 1.16 (1.02–1.33) | <0.0032 |
| IL-6 (pg/mL) | 1.81 (1.69–1.94) | 1.59 (1.50–1.69) | 1.34 (1.26–1.42) | <0.0001 |
| TNF-α (pg/mL) | 1.95 (1.79–2.13) | 1.67 (1.54–1.80) | 1.46 (1.35–1.58) | <0.0001 |
| F2-isoprostanes (pg/mL) | 43.9 (42.3–45.4) | 40.9 (39.6–42.1) | 36.8 (35.6–37.9) | <0.0001 |
© 2018 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dreher, M.L. Whole Fruits and Fruit Fiber Emerging Health Effects. Nutrients 2018, 10, 1833. https://doi.org/10.3390/nu10121833
Dreher ML. Whole Fruits and Fruit Fiber Emerging Health Effects. Nutrients. 2018; 10(12):1833. https://doi.org/10.3390/nu10121833
Chicago/Turabian StyleDreher, Mark L. 2018. "Whole Fruits and Fruit Fiber Emerging Health Effects" Nutrients 10, no. 12: 1833. https://doi.org/10.3390/nu10121833
APA StyleDreher, M. L. (2018). Whole Fruits and Fruit Fiber Emerging Health Effects. Nutrients, 10(12), 1833. https://doi.org/10.3390/nu10121833




