Calorie Restriction Mimetics: Upstream-Type Compounds for Modulating Glucose Metabolism
Abstract
1. Introduction
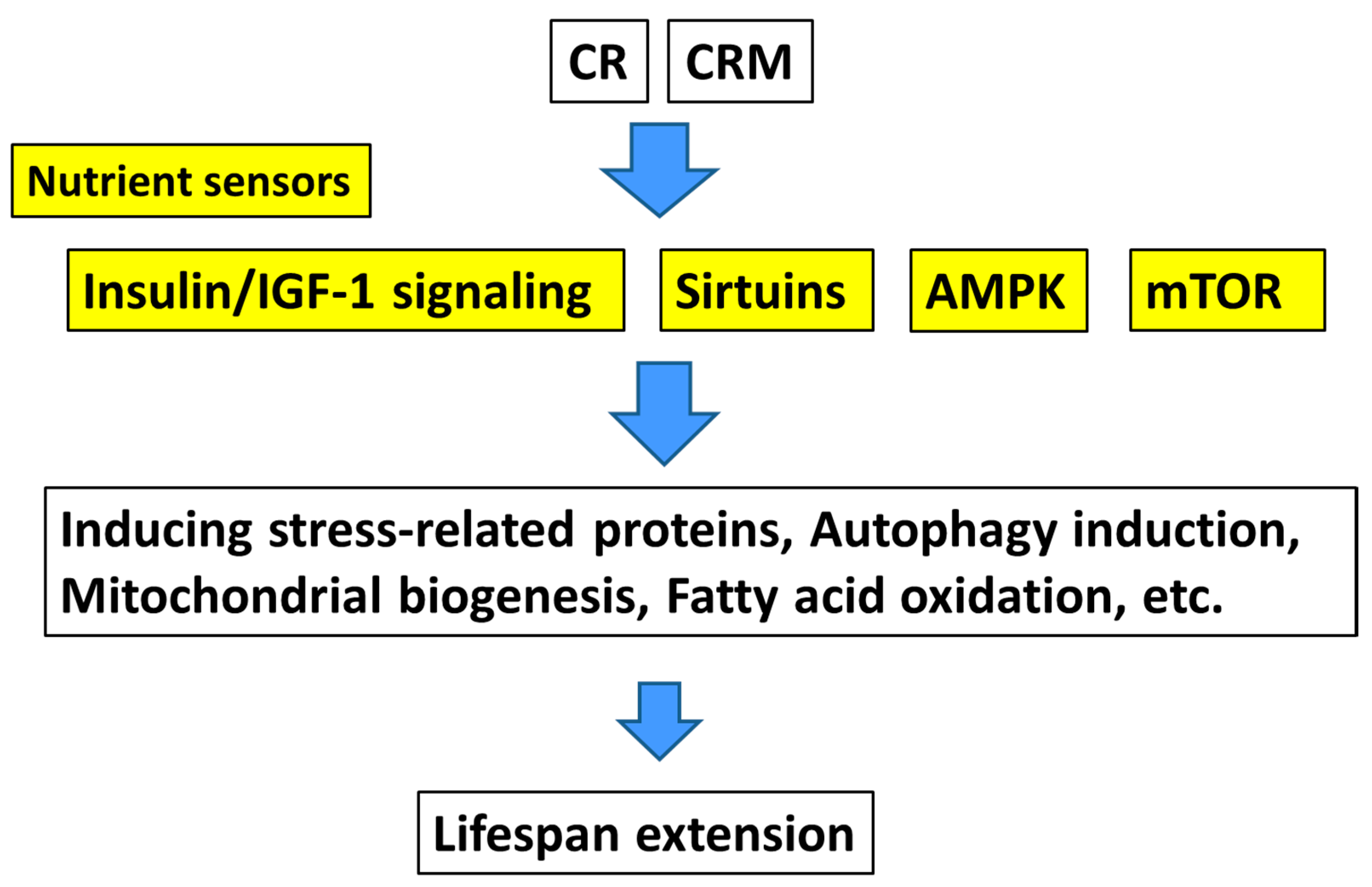
2. Calorie Restriction
3. Calorie Restriction Mimetics
3.1. Downstream-Type Calorie Restriction Mimetics
3.1.1. Metformin
3.1.2. Rapamycin
3.1.3. Resveratrol
3.1.4. Polyamines
3.1.5. Other Downstream-Type Calorie Restriction Mimetics
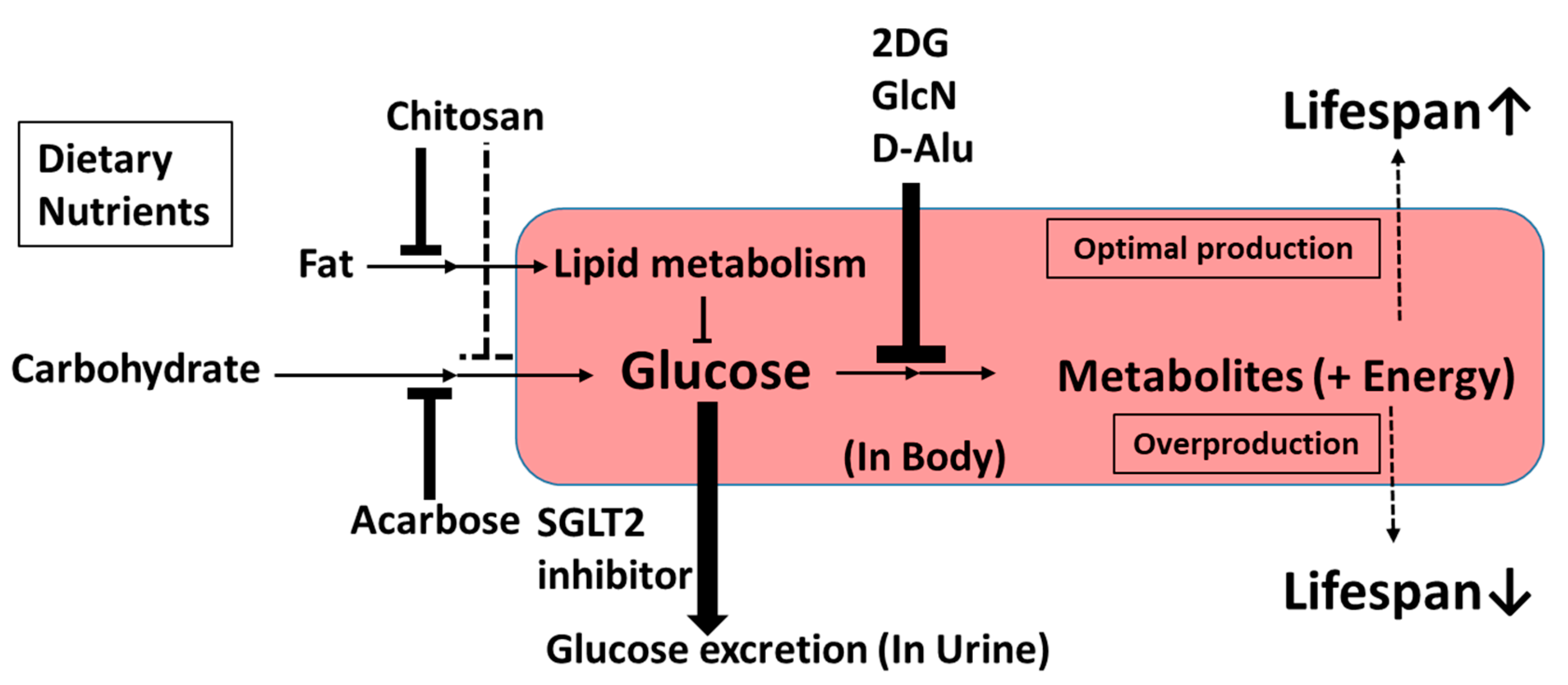
3.2. Upstream-Type Calorie Restriction Mimetics
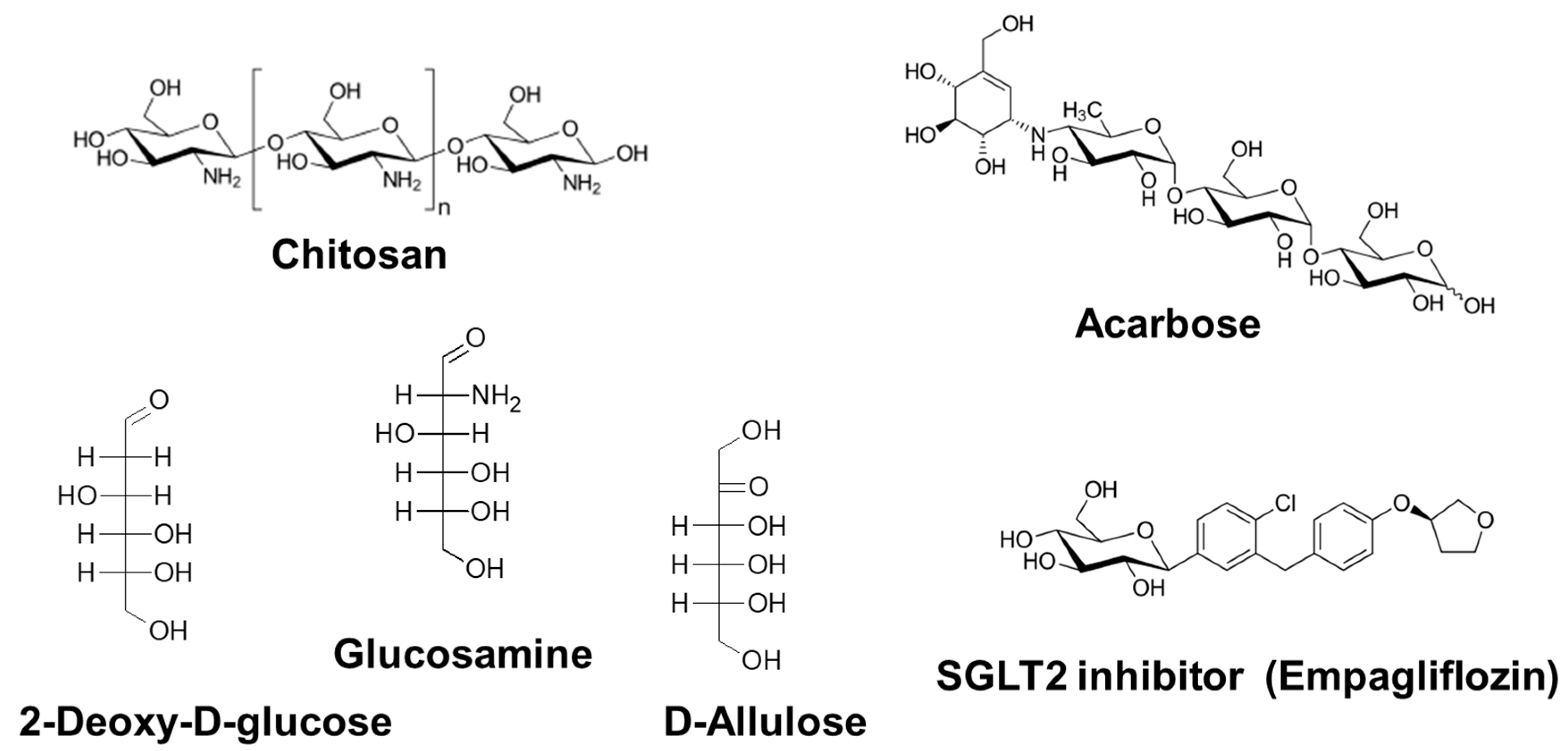
3.2.1. Chitosan
3.2.2. Acarbose
3.2.3. 2-Deoxy-d-Glucose
3.2.4. d-Glucosamine
3.2.5. d-Allulose
3.2.6. Sodium-Glucose Cotransporter 2 Inhibitors
4. Discussion and Summary
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- The World Health Organization. Life Expectancy Increased by 5 Years Since 2000, But Health Inequalities Persist. Available online: http://www.who.int/en/news-room/detail/19-05-2016-life-expectancy-increased-by-5-years-since-2000-but-health-inequalities-persist (accessed on 11 October 2018).
- Colman, R.J.; Anderson, R.M.; Johnson, S.C.; Kastman, E.K.; Kosmatka, K.J.; Beasley, T.M.; Allison, D.B.; Cruzen, C.; Simmons, H.A.; Kemnitz, J.W.; et al. Caloric restriction delays disease onset and mortality in rhesus monkeys. Science 2009, 325, 201–204. [Google Scholar] [CrossRef] [PubMed]
- Mattison, J.A.; Roth, G.S.; Mark Beasley, T.; Tilmont, E.M.; Handy, A.M.; Herbert, R.L.; Longo, D.L.; Allison, D.B.; Young, J.E.; Bryant, M.; et al. Impact of caloric restriction on health and survival in rhesus monkeys from the NIA study. Nature 2012, 489, 318–321. [Google Scholar] [CrossRef] [PubMed]
- Mattison, J.A.; Colman, R.J.; Beasley, T.M.; Allison, D.B.; Kemnitz, J.W.; Roth, G.S.; Ingram, D.K.; Weindruch, R.; De Cabo, R.; Anderson, R.M. Caloric restriction improves health and survival of rhesus monkeys. Nat. Commun. 2017, 8, 14063. [Google Scholar] [CrossRef] [PubMed]
- CALERIE Research Network CALERIE (Comprehensive Assessment of Long Term Effects of Reducing Intake of Energy). Available online: https://calerie.duke.edu/ (accessed on 11 October 2018).
- Martin, C.K.; Bhapkar, M.; Pittas, A.G.; Pieper, C.F.; Das, S.K.; Williamson, D.A.; Scott, T.; Redman, L.M.; Stein, R.; Gilhooly, C.H.; et al. Effect of calorie restriction on mood, quality of life, sleep, and sexual function in healthy nonobese adults: The CALERIE 2 Randomized Clinical Trial for the Comprehensive Assessment of Long-term Effects of Reducing Intake of Energy (CALERIE) Phase 2 study group. JAMA Int. Med. 2016, 176, 743–752. [Google Scholar] [CrossRef]
- Redman, L.M.; Smith, S.R.; Burton, J.H.; Martin, C.K.; Il’yasova, D.; Ravussin, E. Metabolic Slowing and reduced oxidative damage with sustained caloric restriction support the rate of living and oxidative damage theories of aging. Cell Metab. 2018, 27, 805–815. [Google Scholar] [CrossRef] [PubMed]
- Lane, M.A.; Ingram, D.K.; Roth, G.S. 2-Deoxy-d-glucose feeding in rats mimics physiologic effects of calorie restriction. J. Anti Aging Med. 1998, 1, 327–337. [Google Scholar] [CrossRef]
- Ingram, D.K.; Roth, G.S. Glycolytic inhibition as a strategy for developing calorie restriction mimetics. Exp. Gerontol. 2011, 46, 148–154. [Google Scholar] [CrossRef] [PubMed]
- Roth, G.S.; Lane, M.A.; Ingram, D.K. Caloric restriction mimetics: The next phase. Ann. N. Y. Acad. Sci. 2005, 1057, 365–371. [Google Scholar] [CrossRef] [PubMed]
- Ingram, D.K.; Anson, R.M.; De Cabo, R.; Mamczarz, J.; Zhu, M.; Mattison, J.; Lane, M.A.; Roth, G.S. Development of calorie restriction mimetics as a prolongevity strategy. Ann. N. Y. Acad. Sci. 2004, 1019, 412–423. [Google Scholar] [CrossRef] [PubMed]
- Shintani, T.; Okuma, K.; Sakoguchi, H.; Sato, M. Rare Sugars d-psicose and d-allose as calorie restriction mimetic-anti-metabolic syndrome effects and anti-aging effects. J. Brew. Soc. Jpn. 2013, 108, 565–574. [Google Scholar] [CrossRef]
- Ingram, D.K.; Roth, G.S. Calorie restriction mimetics: Can you have your cake and eat it, too? Ageing Res. Rev. 2015, 20, 46–62. [Google Scholar] [CrossRef] [PubMed]
- Madeo, F.; Pietrocola, F.; Eisenberg, T.; Kroemer, G. Caloric restriction mimetics: Towards a molecular definition. Nat. Rev. Drug Discov. 2014, 13, 727–740. [Google Scholar] [CrossRef] [PubMed]
- American Diabetes Association. Pharmacologic approaches to glycemic treatment: Standards of medical care in Diabetes-2018. Diabetes Care 2018, 41, S73–S85. [Google Scholar] [CrossRef] [PubMed]
- Inzucchi, S.E.; Bergenstal, R.M.; Buse, J.B.; Diamant, M.; Ferrannini, E.; Nauck, M.; Peters, A.L.; Tsapas, A.; Wender, R.; Matthews, D.R. Management of hyperglycemia in type 2 diabetes, 2015: A patient-centered approach: Update to a position statement of the American Diabetes Association and the European Association for the Study of Diabetes. Diabetes Care 2015, 38, 140–149. [Google Scholar] [CrossRef] [PubMed]
- Zhou, G.; Goodyear, L.J.; Moller, D.E. Role of AMP-activated protein kinase in mechanism of metformin action. J. Clin. Investig. 2001, 108, 1167–1174. [Google Scholar] [CrossRef] [PubMed]
- Foretz, M.; Guigas, B.; Bertrand, L.; Pollak, M.; Viollet, B. Metformin: From mechanisms of action to therapies. Cell Metab. 2014, 20, 953–966. [Google Scholar] [CrossRef] [PubMed]
- Buse, J.B.; DeFronzo, R.A.; Rosenstock, J.; Kim, T.; Burns, C.; Skare, S.; Baron, A.; Fineman, M. The primary glucose-lowering effect of metformin resides in the gut, not the circulation: Results from short-term pharmacokinetic and 12-week dose-ranging studies. Diabetes Care 2016, 39, 198–205. [Google Scholar] [CrossRef] [PubMed]
- Onken, B.; Driscoll, M. Metformin induces a dietary restriction-like state and the oxidative stress response to extend C. elegans healthspan via AMPK, LKB1, and SKN-1. PLoS ONE 2010, 5, e8758. [Google Scholar] [CrossRef] [PubMed]
- Cabreiro, F.; Au, C.; Leung, K.Y.; Vergara-Irigaray, N.; Cochemé, H.M.; Noori, T.; Weinkove, D.; Schuster, E.; Greene, N.D.E.; Gems, D. Metformin retards aging in C. elegans by altering microbial folate and methionine metabolism. Cell 2013, 153, 228–239. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.; Zhou, B.; Oshiro-Rapley, N.; Li, M.; Paulo, J.A.; Webster, C.M.; Mou, F.; Kacergis, M.C.; Talkowski, M.E.; Carr, C.E.; et al. An ancient, unified mechanism for metformin growth inhibition in C. elegans and cancer. Cell 2016, 167, 1705–1718.e13. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Ou, Y.; Li, Y.; Hu, S.; Shao, L.W.; Liu, Y. Metformin extends C. elegans lifespan through lysosomal pathway. Elife 2017, 6. [Google Scholar] [CrossRef] [PubMed]
- Slack, C.; Foley, A.; Partridge, L. Activation of AMPK by the putative dietary restriction mimetic metformin is insufficient to extend lifespan in Drosophila. PLoS ONE 2012, 7, e47699. [Google Scholar] [CrossRef] [PubMed]
- Abrat, O.B.; Storey, J.M.; Storey, K.B.; Lushchak, V.I. High amylose starch consumption induces obesity in Drosophila melanogaster and metformin partially prevents accumulation of storage lipids and shortens lifespan of the insects. Comput. Biochem. Physiol. Part A Mol. Integr. Physiol. 2018, 215, 55–62. [Google Scholar] [CrossRef] [PubMed]
- Martin-Montalvo, A.; Mercken, E.M.; Mitchell, S.J.; Palacios, H.H.; Mote, P.L.; Scheibye-Knudsen, M.; Gomes, A.P.; Ward, T.M.; Minor, R.K.; Blouin, M.J.; et al. Metformin improves healthspan and lifespan in mice. Nat. Commun. 2013, 4, 2192. [Google Scholar] [CrossRef] [PubMed]
- Turner, R. Effect of intensive blood-glucose control with metformin on complications in overweight patients with type 2 diabetes (UKPDS 34). Lancet 1998, 352, 854–865. [Google Scholar] [CrossRef]
- Campbell, J.M.; Bellman, S.M.; Stephenson, M.D.; Lisy, K. Metformin reduces all-cause mortality and diseases of ageing independent of its effect on diabetes control: A systematic review and meta-analysis. Ageing Res. Rev. 2017, 40, 31–44. [Google Scholar] [CrossRef] [PubMed]
- Sehgal, S.N.; Baker, H.; Vezina, C. Rapamycin (AY-22,989), a new antifungal antibiotic. II. Fermentation, isolation and characterization. J. Antibiot. 1975, 28, 727–732. [Google Scholar] [CrossRef] [PubMed]
- Lamming, D.W.; Ye, L.; Sabatini, D.M.; Baur, J.A. Rapalogs and mTOR inhibitors as anti-aging therapeutics. J. Clin. Investig. 2013, 123, 980–989. [Google Scholar] [CrossRef] [PubMed]
- Ingle, G.R.; Sievers, T.M.; Holt, C.D. Transplant Immunosuppression. Ann. Pharmacother. 2000, 34, 1044–1055. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, S.; Mukherjee, U. A Comprehensive review of immunosuppression used for liver transplantation. J. Transplant. 2009, 2009, 701464. [Google Scholar] [CrossRef] [PubMed]
- Robida-Stubbs, S.; Glover-Cutter, K.; Lamming, D.W.; Mizunuma, M.; Narasimhan, S.D.; Neumann-Haefelin, E.; Sabatini, D.M.; Blackwell, T.K. TOR signaling and rapamycin influence longevity by regulating SKN-1/Nrf and DAF-16/FoxO. Cell Metab. 2012, 15, 713–724. [Google Scholar] [CrossRef] [PubMed]
- Mü ller, C.; Zidek, L.M.; Ackermann, T.; de Jong, T.; Liu, P.; Kliche, V.; Amr Zaini, M.; Kortman, G.; Harkema, L.; Verbeek, D.S.; et al. Reduced expression of C/EBPb-LIP extends health and lifespan in mice. Elife 2018, 7, e34985. [Google Scholar] [CrossRef] [PubMed]
- Pan, H.; Finkel, T. Key proteins and pathways that regulate lifespan. J. Biol. Chem. 2017, 292, 6452–6460. [Google Scholar] [CrossRef] [PubMed]
- Sun, X.; Komatsu, T.; Lim, J.; Laslo, M.; Yolitz, J.; Wang, C.; Poirier, L.; Alberico, T.; Zou, S. Nutrient-dependent requirement for SOD1 in lifespan extension by protein restriction in Drosophila melanogaster. Aging Cell 2012, 11, 783–793. [Google Scholar] [CrossRef] [PubMed]
- Wang, A.; Mouser, J.; Pitt, J.; Promislow, D.; Kaeberlein, M. Rapamycin enhances survival in a Drosophila model of mitochondrial disease. Oncotarget 2016, 7, 80131–80139. [Google Scholar] [CrossRef] [PubMed]
- Harrison, D.E.; Strong, R.; Sharp, Z.D.; Nelson, J.F.; Astle, C.M.; Flurkey, K.; Nadon, N.L.; Wilkinson, J.E.; Frenkel, K.; Carter, C.S.; et al. Rapamycin fed late in life extends lifespan in genetically heterogeneous mice. Nature 2009, 460, 392–395. [Google Scholar] [CrossRef] [PubMed]
- Bitto, A.; Ito, T.K.; Pineda, V.V.; Letexier, N.J.; Huang, H.Z.; Sutlief, E.; Tung, H.; Vizzini, N.; Chen, B.; Smith, K.; et al. Transient rapamycin treatment can increase lifespan and healthspan in middle-aged mice. Elife 2016, 5, e16351. [Google Scholar] [CrossRef] [PubMed]
- Siegmund, S.E.; Yang, H.; Sharma, R.; Javors, M.; Skinner, O.; Mootha, V.; Hirano, M.; Schon, E.A. Low-dose rapamycin extends lifespan in a mouse model of mtDNA depletion syndrome. Hum. Mol. Genet. 2017, 26, 4588–4605. [Google Scholar] [CrossRef] [PubMed]
- Lesniewski, L.A.; Seals, D.R.; Walker, A.E.; Henson, G.D.; Blimline, M.W.; Trott, D.W.; Bosshardt, G.C.; LaRocca, T.J.; Lawson, B.R.; Zigler, M.C.; et al. Dietary rapamycin supplementation reverses age-related vascular dysfunction and oxidative stress, while modulating nutrient-sensing, cell cycle, and senescence pathways. Aging Cell 2017, 16, 17–26. [Google Scholar] [CrossRef] [PubMed]
- Wilkinson, J.E.; Burmeister, L.; Brooks, S.V.; Chan, C.C.; Friedline, S.; Harrison, D.E.; Hejtmancik, J.F.; Nadon, N.; Strong, R.; Wood, L.K.; et al. Rapamycin slows aging in mice. Aging Cell 2012. [Google Scholar] [CrossRef] [PubMed]
- Deblon, N.; Bourgoin, L.; Veyrat-Durebex, C.; Peyrou, M.; Vinciguerra, M.; Caillon, A.; Maeder, C.; Fournier, M.; Montet, X.; Rohner-Jeanrenaud, F.; et al. Chronic mTOR inhibition by rapamycin induces muscle insulin resistance despite weight loss in rats. Br. J. Pharmacol. 2012, 165, 2325–2340. [Google Scholar] [CrossRef] [PubMed]
- Siemann, E.H.; Creasy, L.L. Concentration of the phytoalexin resveratrol in wine. Am. J. Enol. Vitic. 1992, 43, 49. [Google Scholar]
- De Ligt, M.; Timmers, S.; Schrauwen, P. Resveratrol and obesity: Can resveratrol relieve metabolic disturbances? Biochim. Biophys. Acta Mol. Basis Dis. 2015, 1852, 1137–1144. [Google Scholar] [CrossRef] [PubMed]
- Pan, Y.; Zhang, H.; Zheng, Y.; Zhou, J.; Yuan, J.; Yu, Y.; Wang, J. Resveratrol exerts antioxidant effects by activating SIRT2 to deacetylate Prx1. Biochemistry 2017, 56, 6325–6328. [Google Scholar] [CrossRef] [PubMed]
- Viswanathan, M.; Kim, S.K.; Berdichevsky, A.; Guarente, L. A role for SIR-2.1 regulation of ER stress response genes in determining C. elegans life span. Dev. Cell 2005, 9, 605–615. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Rezaizadehnajafi, L.; Wink, M. Influence of resveratrol on oxidative stress resistance and life span in Caenorhabditis elegans. J. Pharm. Pharmacol. 2013, 65, 682–688. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Wheeler, C.T.; Alberico, T.; Sun, X.; Seeberger, J.; Laslo, M.; Spangler, E.; Kern, B.; De Cabo, R.; Zou, S. The effect of resveratrol on lifespan depends on both gender and dietary nutrient composition in Drosophila melanogaster. Age 2013, 35, 69–81. [Google Scholar] [CrossRef] [PubMed]
- Staats, S.; Wagner, A.E.; Kowalewski, B.; Rieck, F.T.; Soukup, S.T.; Kulling, S.E.; Rimbach, G. Dietary resveratrol does not affect life span, body composition, stress response, and longevity-related gene expression in Drosophila melanogaster. Int. J. Mol. Sci. 2018, 19. [Google Scholar] [CrossRef] [PubMed]
- Johnson, A.A.; Riehle, M.A. Resveratrol fails to extend lifespan in the mosquito Anopheles stephensi. Rejuv. Res. 2015, 18, 473–478. [Google Scholar] [CrossRef] [PubMed]
- Baur, J.A.; Pearson, K.J.; Price, N.L.; Jamieson, H.A.; Lerin, C.; Kalra, A.; Prabhu, V.V.; Allard, J.S.; Lopez-Lluch, G.; Lewis, K.; et al. Resveratrol improves health and survival of mice on a high-calorie diet. Nature 2006, 444, 337–342. [Google Scholar] [CrossRef] [PubMed]
- Da Luz, P.L.; Tanaka, L.; Brum, P.C.; Dourado, P.M.M.; Favarato, D.; Krieger, J.E.; Laurindo, F.R.M. Red wine and equivalent oral pharmacological doses of resveratrol delay vascular aging but do not extend life span in rats. Atherosclerosis 2012, 224, 136–142. [Google Scholar] [CrossRef] [PubMed]
- Witte, A.V.; Kerti, L.; Margulies, D.S.; Flöel, A. Effects of resveratrol on memory performance, hippocampal functional connectivity, and glucose metabolism in healthy older adults. J. Neurosci. 2014, 34, 7862–7870. [Google Scholar] [CrossRef] [PubMed]
- Timmers, S.; Konings, E.; Bilet, L.; Houtkooper, R.H.; Van De Weijer, T.; Goossens, G.H.; Hoeks, J.; Van Der Krieken, S.; Ryu, D.; Kersten, S.; et al. Calorie restriction-like effects of 30 days of resveratrol supplementation on energy metabolism and metabolic profile in obese humans. Cell Metab. 2011, 14, 612–622. [Google Scholar] [CrossRef] [PubMed]
- Kumar Bhatt, J.; Thomas, S.; Nanjan, M.J. Resveratrol supplementation improves glycemic control in type 2 diabetes mellitus. Nutr. Res. 2012, 32, 537–541. [Google Scholar] [CrossRef] [PubMed]
- Pollack, R.M.; Barzilai, N.; Anghel, V.; Kulkarni, A.S.; Golden, A.; O’Broin, P.; Sinclair, D.A.; Bonkowski, M.S.; Coleville, A.J.; Powell, D.; et al. Resveratrol improves vascular function and mitochondrial number but not glucose metabolism in older adults. J. Gerontol. Ser. A 2017, 72, 1703–1709. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, M.; Kurihara, S.; Kibe, R.; Ashida, H.; Benno, Y. Longevity in mice is promoted by probiotic-induced suppression of colonic senescence dependent on upregulation of gut bacterial polyamine production. PLoS ONE 2011, 6, e23652. [Google Scholar] [CrossRef] [PubMed]
- Eisenberg, T.; Knauer, H.; Schauer, A.; Büttner, S.; Ruckenstuhl, C.; Carmona-Gutierrez, D.; Ring, J.; Schroeder, S.; Magnes, C.; Antonacci, L.; et al. Induction of autophagy by spermidine promotes longevity. Nat. Cell Biol. 2009, 11, 1305–1314. [Google Scholar] [CrossRef] [PubMed]
- Eisenberg, T.; Abdellatif, M.; Schroeder, S.; Primessnig, U.; Stekovic, S.; Pendl, T.; Harger, A.; Schipke, J.; Zimmermann, A.; Schmidt, A.; et al. Cardioprotection and lifespan extension by the natural polyamine spermidine. Nat. Med. 2016, 22, 1428–1438. [Google Scholar] [CrossRef] [PubMed]
- Madeo, F.; Eisenberg, T.; Pietrocola, F.; Kroemer, G. Spermidine in health and disease. Science 2018, 359. [Google Scholar] [CrossRef] [PubMed]
- Williams, D.S.; Cash, A.; Hamadani, L.; Diemer, T. Oxaloacetate supplementation increases lifespan in Caenorhabditis elegans through an AMPK/FOXO-dependent pathway. Aging Cell 2009, 8, 765–768. [Google Scholar] [CrossRef] [PubMed]
- Yoshikawa, K. Studies on anti-diabetic effect of sodium oxaleacetate. Tohoku J. Exp. Med. 1968, 96, 127–141. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.H.; Cai, F.Y.; Chiang, M.T. Long-term feeding of chitosan ameliorates glucose and lipid metabolism in a high-fructose-diet-impaired rat model of glucose tolerance. Mar. Drugs 2015, 13, 7302–7313. [Google Scholar] [CrossRef] [PubMed]
- Bahijri, S.M.; Alsheikh, L.; Ajabnoor, G.; Borai, A. Effect of supplementation with chitosan on weight, cardiometabolic, and other risk indices in Wistar rats fed normal and high-fat/high-cholesterol diets ad libitum. Nutr. Metab. Insights 2017, 10. [Google Scholar] [CrossRef] [PubMed]
- Yao, H.T.; Huang, S.Y.; Chiang, M.T. A comparative study on hypoglycemic and hypocholesterolemic effects of high and low molecular weight chitosan in streptozotocin-induced diabetic rats. Food Chem. Toxicol. 2008, 46, 1525–1534. [Google Scholar] [CrossRef] [PubMed]
- Saltiel, R.A.; Kahn, C.R. Insulin signaling and the regulation of glucose and lipid metabolism. Nature 2001, 414, 799–806. [Google Scholar] [CrossRef] [PubMed]
- Baker, W.L.; Tercius, A.; Anglade, M.; White, C.M.; Coleman, C.I. A meta-analysis evaluating the impact of chitosan on serum lipids in hypercholesterolemic patients. Ann. Nutr. Metab. 2009, 55, 368–374. [Google Scholar] [CrossRef] [PubMed]
- Rizzo, M.; Giglio, R.V.; Nikolic, D.; Patti, A.M.; Campanella, C.; Cocchi, M.; Katsiki, N.; Montalto, G. Effects of chitosan on plasma lipids and lipoproteins: A 4-month prospective pilot study. Angiology 2014, 65, 538–542. [Google Scholar] [CrossRef] [PubMed]
- Lütjohann, D.; Marinova, M.; Wolter, K.; Willinek, W.; Bitterlich, N.; Coenen, M.; Coch, C.; Stellaard, F. Influence of chitosan treatment on surrogate serum markers of cholesterol metabolism in obese subjects. Nutrients 2018, 10. [Google Scholar] [CrossRef] [PubMed]
- Van Der Gronde, T.; Hartog, A.; Van Hees, C.; Pellikaan, H.; Pieters, T. Systematic review of the mechanisms and evidence behind the hypocholesterolaemic effects of HPMC, pectin and chitosan in animal trials. Food Chem. 2016, 199, 746–759. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.; Zou, Y.; Chi, H. Quantitative assessment of the effects of chitosan intervention on blood pressure control. Drug Des. Dev. Ther. 2017, 12, 67–75. [Google Scholar] [CrossRef] [PubMed]
- Harrison, D.E.; Strong, R.; Allison, D.B.; Ames, B.N.; Astle, C.M.; Atamna, H.; Fernandez, E.; Flurkey, K.; Javors, M.A.; Nadon, N.L.; et al. Acarbose, 17-α-estradiol, and nordihydroguaiaretic acid extend mouse lifespan preferentially in males. Aging Cell 2014, 13, 273–282. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, M.; Otsuki, M. Effect of inhibition of α-glucosidase on age-related glucose intolerance and pancreatic atrophy in rats. Metabolism 2006, 55, 533–540. [Google Scholar] [CrossRef] [PubMed]
- Chiasson, J.-L.; Josse, R.G.; Gomis, R.; Hanefeld, M.; Karasik, A.; Laakso, M. Acarbose for prevention of type 2 diabetes mellitus: The STOP-NIDDM randomised trial. Lancet 2002, 359, 2072–2077. [Google Scholar] [CrossRef]
- Chiasson, J.; Posse, R.G.; Gomis, R. Acarbose treatment and the risk of cardiovascular disease and hypertension in patients with impaired glucose tolerance: The stop-niddm trial. JAMA 2003, 290, 486–494. [Google Scholar] [CrossRef] [PubMed]
- Schnell, O.; Weng, J.; Sheu, W.H.-H.; Watada, H.; Kalra, S.; Soegondo, S.; Yamamoto, N.; Rathod, R.; Zhang, C.; Grzeszczak, W. Acarbose reduces body weight irrespective of glycemic control in patients with diabetes: Results of a worldwide, non-interventional, observational study data pool. J. Diabetes Complicat. 2016, 30, 628–637. [Google Scholar] [CrossRef] [PubMed]
- Lane, M.; Ingram, D. Roth George 2-Deoxy-d-Glucose Feeding in Rats Mimics Physiologic Effects of Calorie Restriction. J. Anti Aging Med. 2009, 1, 327–337. [Google Scholar] [CrossRef]
- Schulz, T.J.; Zarse, K.; Voigt, A.; Urban, N.; Birringer, M.; Ristow, M. Glucose restriction extends Caenorhabditis elegans life span by inducing mitochondrial respiration and increasing oxidative stress. Cell Metab. 2007, 6, 280–293. [Google Scholar] [CrossRef] [PubMed]
- Wan, R.; Camandola, S.; Mattson, M.P. Intermittent fasting and dietary supplementation with 2-deoxy-d-glucose improve functional and metabolic cardiovascular risk factors in rats. FASEB J. 2003, 17, 1133–1134. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Bruce-Keller, A.J.; Kruman, Y.; Chan, S.L.; Mattson, M.P. 2-Deoxy-d-glucose protects hippocampal neurons against excitotoxic and oxidative injury: Evidence for the involvement of stress proteins. J. Neurosci. Res. 1999, 57, 48–61. [Google Scholar] [CrossRef]
- Duan, W.; Mattson, M.P. Dietary restriction and 2-deoxyglucose administration improve behavioral outcome and reduce degeneration of dopaminergic neurons in models of Parkinson’s disease. J. Neurosci. Res. 1999, 57, 195–206. [Google Scholar] [CrossRef]
- Singh, S.; Pandey, S.; Bhatt, A.N.; Chaudhary, R.; Bhuria, V.; Kalra, N.; Soni, R.; Roy, B.G.; Saluja, D.; Dwarakanath, B.S. Chronic dietary administration of the glycolytic inhibitor 2-deoxy-d-glucose (2-DG) inhibits the growth of implanted Ehrlich’s ascites tumor in mice. PLoS ONE 2015, 10, e0132089. [Google Scholar] [CrossRef] [PubMed]
- Minor, R.K.; Smith, D.L.; Sossong, A.M.; Kaushik, S.; Poosala, S.; Spangler, E.L.; Roth, G.S.; Lane, M.; Allison, D.B.; de Cabo, R.; et al. Chronic ingestion of 2-deoxy-d-glucose induces cardiac vacuolization and increases mortality in rats. Toxicol. Appl. Pharmacol. 2010, 243, 332–339. [Google Scholar] [CrossRef] [PubMed]
- Reginster, J.Y.; Deroisy, R.; Rovati, L.C.; Lee, R.L.; Lejeune, E.; Bruyere, O.; Giacovelli, G.; Henrotin, Y.; Dacre, J.E.; Gossett, C. Long-term effects of glucosamine sulphate on osteoarthritis progression: A randomised, placebo-controlled clinical trial. Lancet 2001, 357, 251–256. [Google Scholar] [CrossRef]
- Weimer, S.; Priebs, J.; Kuhlow, D.; Groth, M.; Priebe, S.; Mansfeld, J.; Merry, T.L.; Dubuis, S.; Laube, B.; Pfeiffer, A.F.; et al. d-Glucosamine supplementation extends life span of nematodes and of ageing mice. Nat. Commun. 2014, 5, 3563. [Google Scholar] [CrossRef] [PubMed]
- Shintani, T.; Sakoguchi, H.; Yoshihara, A.; Izumori, K.; Sato, M. d-Allulose, a stereoisomer of d-fructose, extends Caenorhabditis elegans lifespan through a dietary restriction mechanism: A new candidate dietary restriction mimetic. Biochem. Biophys. Res. Commun. 2017, 493, 1528–1533. [Google Scholar] [CrossRef] [PubMed]
- Barrientos, C.; Racotta, R.; Quevedo, L. Glucosamine attenuates increases of intraabdominal fat, serum leptin levels, and insulin resistance induced by a high-fat diet in rats. Nutr. Res. 2010, 30, 791–800. [Google Scholar] [CrossRef] [PubMed]
- Shintani, T.; Yamazaki, F.; Katoh, T.; Umekawa, M.; Matahira, Y.; Hori, S.; Kakizuka, A.; Totani, K.; Yamamoto, K.; Ashida, H. Glucosamine induces autophagy via an mTOR-independent pathway. Biochem. Biophys. Res. Commun. 2010, 391, 1775–1779. [Google Scholar] [CrossRef] [PubMed]
- Katoh, A.; Kai, H.; Harada, H.; Niiyama, H.; Ikeda, H. Oral administration of glucosamine improves vascular endothelial function by modulating intracellular redox state. Int. Heart J. 2017, 58, 926–932. [Google Scholar] [CrossRef] [PubMed]
- Pocobelli, G.; Kristal, A.R.; Patterson, R.E.; Potter, J.D.; Lampe, J.W.; Kolar, A.; Evans, I.; White, E. Total mortality risk in relation to use of less-common dietary supplements. Am. J. Clin. Nutr. 2010, 91, 1791–1800. [Google Scholar] [CrossRef] [PubMed]
- Iida, T.; Hayashi, N.; Yamada, T.; Yoshikawa, Y.; Miyazato, S.; Kishimoto, Y.; Okuma, K.; Tokuda, M.; Izumori, K. Failure of d-psicose absorbed in the small intestine to metabolize into energy and its low large intestinal fermentability in humans. Metabolism. 2010, 59, 206–214. [Google Scholar] [CrossRef] [PubMed]
- Yoshihara, A.; Kozakai, T.; Shintani, T.; Matsutani, R.; Ohtani, K.; Iida, T.; Akimitsu, K.; Izumori, K.; Gullapalli, P.K. Purification and characterization of d-allulose 3-epimerase derived from Arthrobacter globiformis M30, a GRAS microorganism. J. Biosci. Bioeng. 2017, 123, 170–176. [Google Scholar] [CrossRef] [PubMed]
- Mooradian, A.D.; Smith, M.; Tokuda, M. The role of artificial and natural sweeteners in reducing the consumption of table sugar: A narrative review. Clin. Nutr. ESPEN 2017, 18, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Hossain, A.; Yamaguchi, F.; Matsuo, T.; Tsukamoto, I.; Toyoda, Y.; Ogawa, M.; Nagata, Y.; Tokuda, M. Rare sugar d-allulose: Potential role and therapeutic monitoring in maintaining obesity and type 2 diabetes mellitus. Pharmacol. Ther. 2015, 155, 49–59. [Google Scholar] [CrossRef] [PubMed]
- Matsuo, T.K.; Baba, Y.; Hashiguchi, M.; Takeshita, K.; Izumori, K.; Suzuki, H. Less Body Fat Accumulation with d-Psicose Diet versus d-Fructose Diet. J. Clin. Biochem. Nutr. 2001, 30, 55–65. [Google Scholar] [CrossRef]
- Ochiai, M.; Onishi, K.; Yamada, T.; Iida, T.; Matsuo, T. d-psicose increases energy expenditure and decreases body fat accumulation in rats fed a high-sucrose diet. Int. J. Food Sci. Nutr. 2014, 65, 245–250. [Google Scholar] [CrossRef] [PubMed]
- Ochiai, M.; Nakanishi, Y.; Yamada, T.; Iida, T.; Matsuo, T. Inhibition by dietary d-psicose of body fat accumulation in adult rats fed a high-sucrose diet. Biosci. Biotechnol. Biochem. 2013, 77, 1123–1126. [Google Scholar] [CrossRef] [PubMed]
- Shintani, T.; Yamada, T.; Hayashi, N.; Iida, T.; Nagata, Y.; Ozaki, N.; Toyoda, Y. Rare sugar syrup containing d-allulose but not high-fructose corn syrup maintains glucose tolerance and insulin sensitivity partly via hepatic glucokinase translocation in Wistar rats. J. Agric. Food Chem. 2017, 65, 2888–2894. [Google Scholar] [CrossRef] [PubMed]
- Sato, M.; Kurose, H.; Yamasaki, T.; Izumori, K. Potential anthelmintic: d-psicose inhibits motility, growth and reproductive maturity of L1 larvae of Caenorhabditis elegans. J. Nat. Med. 2008, 62, 244–246. [Google Scholar] [CrossRef] [PubMed]
- Shintani, T.; Kosuge, Y.; Ashida, H. Glucosamine extends the lifespan of Caenorhabditis elegans via autophagy induction glucosamine extends nematode lifespan via autophagy induction. J. Appl. Glycosci. 2018, 65, 37–43. [Google Scholar] [CrossRef]
- Nagata, Y.; Kanasaki, A.; Tamaru, S.; Tanaka, K. d-psicose, an epimer of d-fructose, favorably alters lipid metabolism in Sprague-Dawley rats. J. Agric. Food Chem. 2015, 63, 3168–3176. [Google Scholar] [CrossRef] [PubMed]
- Iida, T.; Kishimoto, Y.; Yoshikawa, Y.; Hayashi, N.; Okuma, K.; Toshi, M.; Yagi, K.; Matsuo, T.; Izumori, K. Acute d-psicose administration decreases the glycemic responses to an oral maltodextrin tolerance test in normal adults. J. Nutr. Sci. Vitaminol. 2008, 54, 511–514. [Google Scholar] [CrossRef] [PubMed]
- Hayashi, N.; Iida, T.; Yamada, T.; Okura, K.; Takehara, I.; Yamamoto, T.; Yamada, K.; Tokuda, M. Study on the postprandial blood glucose suppression effect of d-psicose in borderline diabetes and the safety of long-term ingestion by normal human subjects. Biosci. Biotechnol. Biochem. 2010, 74, 510–519. [Google Scholar] [CrossRef] [PubMed]
- Yamada, T.; Shintani, S.; Iida, T.; Kishimoto, Y.; Okuma, K. Effect of ingestion of rare sugar syrup on the blood glucose response in humans. J. Jpn. Soc. Nutr. Food Sci. 2017, 70, 271–278. [Google Scholar] [CrossRef]
- Kimura, T.; Kanasaki, A.; Hayashi, N.; Yamada, T.; Iida, T.; Nagata, Y.; Okuma, K. d-Allulose enhances postprandial fat oxidation in healthy humans. Nutrition 2017, 43–44, 16–20. [Google Scholar] [CrossRef] [PubMed]
- Han, Y.; Kwon, E.-Y.; Yu, M.; Lee, S.; Kim, H.-J.; Kim, S.-B.; Kim, Y.; Choi, M.-S. A preliminary study for evaluating the dose-dependent effect of d-allulose for fat Mass reduction in adult humans: A randomized, double-blind, placebo-controlled trial. Nutrients 2018, 10. [Google Scholar] [CrossRef] [PubMed]
- Kalra, S. Sodium glucose co-transporter-2 (SGLT2) inhibitors: A review of their basic and clinical pharmacology. Diabetes Ther. 2014, 5, 355–366. [Google Scholar] [CrossRef] [PubMed]
- Lee, P.C.; Ganguly, S.; Goh, S.-Y. Obesity treatment/etiology and pathophysiology weight loss associated with sodium-glucose cotransporter-2 inhibition: A review of evidence and underlying mechanisms. Obes. Rev. 2018. [Google Scholar] [CrossRef] [PubMed]
- Thomas, M.C.; Cherney, D.Z.I. The actions of SGLT2 inhibitors on metabolism, renal function and blood pressure. Diabetologia 2018, 61, 2098–2107. [Google Scholar] [CrossRef] [PubMed]
- Nair, S.; Wilding, J.P.H. Sodium glucose cotransporter 2 inhibitors as a new treatment for diabetes mellitus. J. Clin. Endocrinol. Metab. 2010, 95, 34–42. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Luo, Y.; Wang, X.; Orlicky, D.J.; Myakala, K.; Yang, P.; Levi, M. The sodium-glucose cotransporter 2 inhibitor dapagliflozin prevents renal and liver disease in western diet induced obesity mice. Int. J. Mol. Sci. 2018, 19. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Welihinda, A.; Mechanic, J.; Ding, H.; Zhu, L.; Lu, Y.; Deng, Z.; Sheng, Z.; Lv, B.; Chen, Y.; et al. EGT1442, a potent and selective SGLT2 inhibitor, attenuates blood glucose and HbA 1c levels in db/db mice and prolongs the survival of stroke-prone rats. Pharmacol. Res. 2011, 63, 284–293. [Google Scholar] [CrossRef] [PubMed]
- Zinman, B.; Wanner, C.; Lachin, J.M.; Fitchett, D.; Bluhmki, E.; Hantel, S.; Mattheus, M.; Devins, T.; Johansen, O.E.; Woerle, H.J.; et al. Empagliflozin, cardiovascular outcomes, and mortality in type 2 diabetes. N. Engl. J. Med. 2015, 373, 2117–2128. [Google Scholar] [CrossRef] [PubMed]
- Bruce, N.; Vlado, P.; Kenneth, M.; Dick, Z.; Greg, F.; Ngoz, E.; Wayne, S.; Gordon, L.; Mehul, D.; David, M.; CANVAS Program Collaborative Group. Canagliflozin and Cardiovascular and Renal Events in Type 2 Diabetes. N. Engl. J. Med. 2017, 377, 644–657. [Google Scholar]
- Kalra, S.; Jacob, J.; Gupta, Y. Newer antidiabetic drugs and calorie restriction mimicry. Indian J. Endocrinol. Metab. 2016, 20, 142–146. [Google Scholar] [CrossRef] [PubMed]

| Compound | Mode of Action |
|---|---|
| Metformin (antidiabetic drug) | AMPK activation |
| Rapamycin (immunosuppressant drug) | mTOR inhibition |
| Resveratrol (food component) | Sirtuin activation |
| Polyamines (food component) | Epigenetic control |
| Oxaloacetic acid (dietary supplement) | Redox balance |
| Compound | Mode of Action |
|---|---|
| Chitosan (dietary supplement) | Glucose diminution |
| Acarbose (antidiabetic drug) | Glycosidase inhibition |
| 2-Deoxy-d-glucose (anticancer drug) | Glycolysis inhibition |
| d-Glucosamine (dietary supplement) | Glycolysis adjustment |
| d-Allulose (food component) | Glycolysis improvement |
| SGLT2 inhibitor (antidiabetic drug) | Glucose excretion |
| Main Experimental Animal Model | Lifespan | Glucose Level | Body Weight | Body Fat | Autophagy | Energy Consumption | Stress Tolerance | |
|---|---|---|---|---|---|---|---|---|
| CR | Human | Extend? | Lower | Lose | Decrease | Enhance in excessive CR | Change | Decrease stress markers |
| Nematode/Mouse | Extend | Lower | Lose | Decrease | Enhance in excessive CR | Change | Decrease stress markers | |
| Chitosan | Mouse | Extend? | Lower | Lose | Decrease | NR | NR | NR |
| Acarbose | Human/Mouse | Extend? | Lower | Lose | Decrease | NR | NR | NR |
| 2DG | Nematode/Mouse | Extend | Lower | Lose | Decrease | Enhance | NR | Increase antioxidant enzyme |
| GlcN | Nematode/Mouse | Extend | Lower | Lose | Decrease | Enhance | Change | Increase antioxidant enzyme |
| d-Alu | Nematode/Mouse | Extend | Lower | Lose | Decrease | NR | Change | Increase antioxidant enzyme |
| SGLT2 inhibitors | Human/Mouse | Extend? | Lower | Lose | Decrease | NR | Change | Decrease stress markers |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shintani, H.; Shintani, T.; Ashida, H.; Sato, M. Calorie Restriction Mimetics: Upstream-Type Compounds for Modulating Glucose Metabolism. Nutrients 2018, 10, 1821. https://doi.org/10.3390/nu10121821
Shintani H, Shintani T, Ashida H, Sato M. Calorie Restriction Mimetics: Upstream-Type Compounds for Modulating Glucose Metabolism. Nutrients. 2018; 10(12):1821. https://doi.org/10.3390/nu10121821
Chicago/Turabian StyleShintani, Hideya, Tomoya Shintani, Hisashi Ashida, and Masashi Sato. 2018. "Calorie Restriction Mimetics: Upstream-Type Compounds for Modulating Glucose Metabolism" Nutrients 10, no. 12: 1821. https://doi.org/10.3390/nu10121821
APA StyleShintani, H., Shintani, T., Ashida, H., & Sato, M. (2018). Calorie Restriction Mimetics: Upstream-Type Compounds for Modulating Glucose Metabolism. Nutrients, 10(12), 1821. https://doi.org/10.3390/nu10121821





