Annurca Apple Polyphenols Protect Murine Hair Follicles from Taxane Induced Dystrophy and Hijacks Polyunsaturated Fatty Acid Metabolism toward β-Oxidation
Abstract
1. Introduction
2. Materials and Methods
2.1. Reagents and Nutraceuticals
2.2. Animals
2.2.1. Animals for Ex-Vivo Culturing of Murine HFs
2.2.2. Animals for In-Vivo Experiments
2.3. Metabolite Extraction from Murine Tissues
2.4. Mass Spectrometry-Based Metabolomic, Statistics and Analysis
3. Results
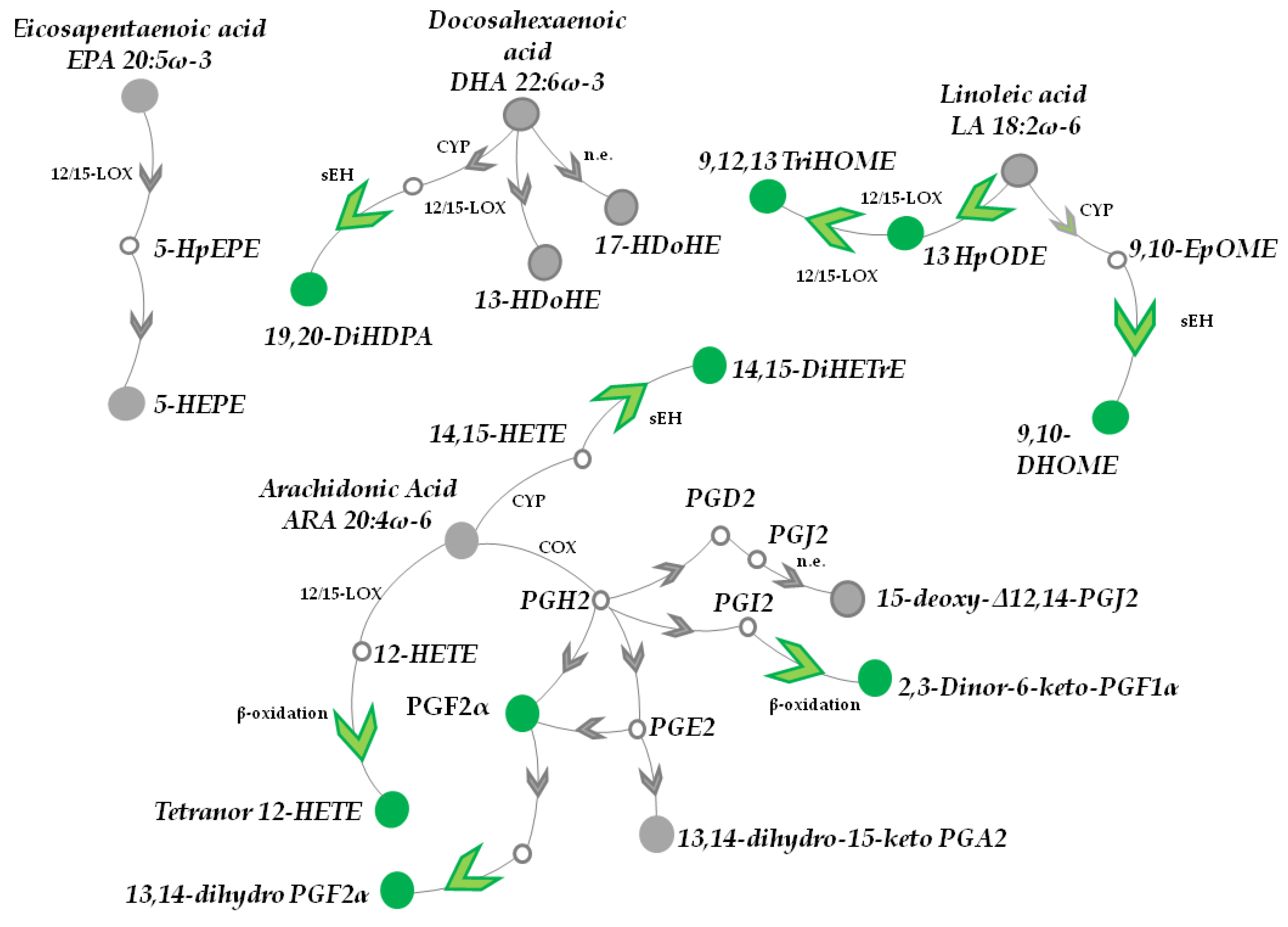
3.1. Topical Treatment with AAE Alters PUFAs Metabolism in Murine HFs
3.1.1. DHA, EPA, LA and α-LA Metabolites
3.1.2. ARA Catabolites (CYP and LOX Metabolites)
3.1.3. AAE Accelerates Epoxides Conversion into Inactive Diols and β-Oxidation of PUFA Metabolites
3.1.4. Prostaglandins
3.1.5. AAE Selectively Activates Prostanoid Metabolism in HF Cells
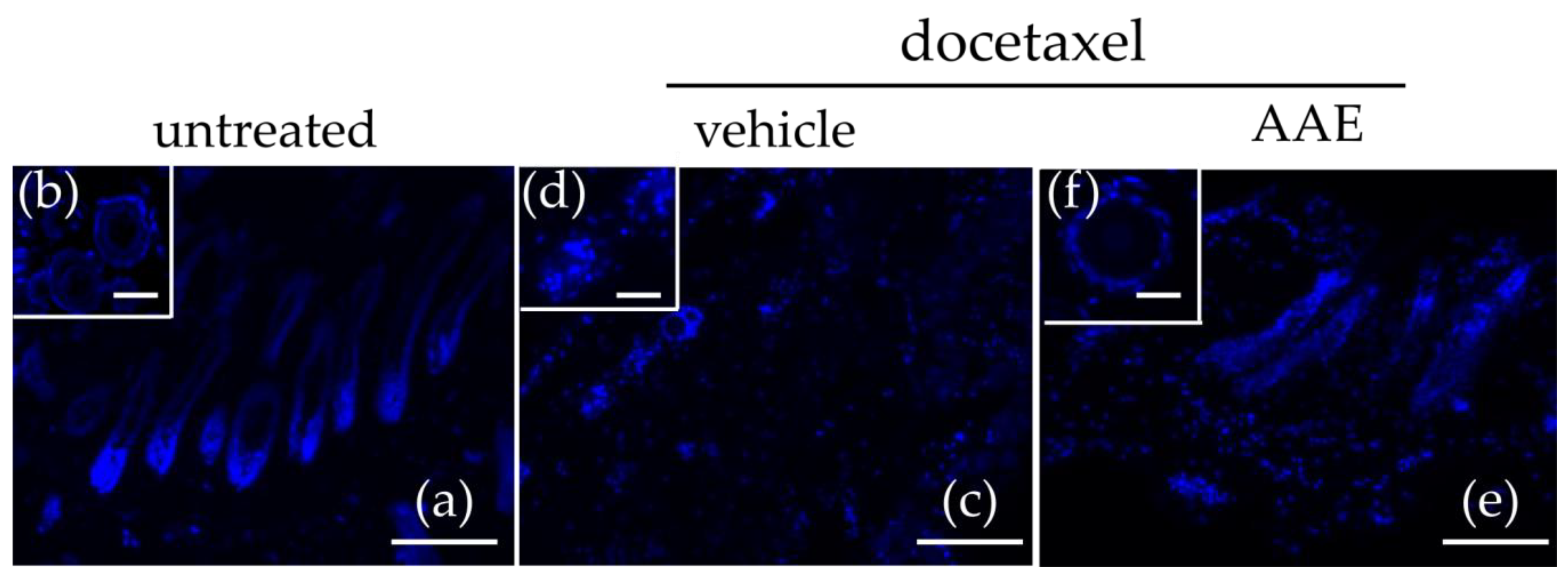
3.2. AAE Protects Murine HFs from Taxane Induced Follicular Dystrophy
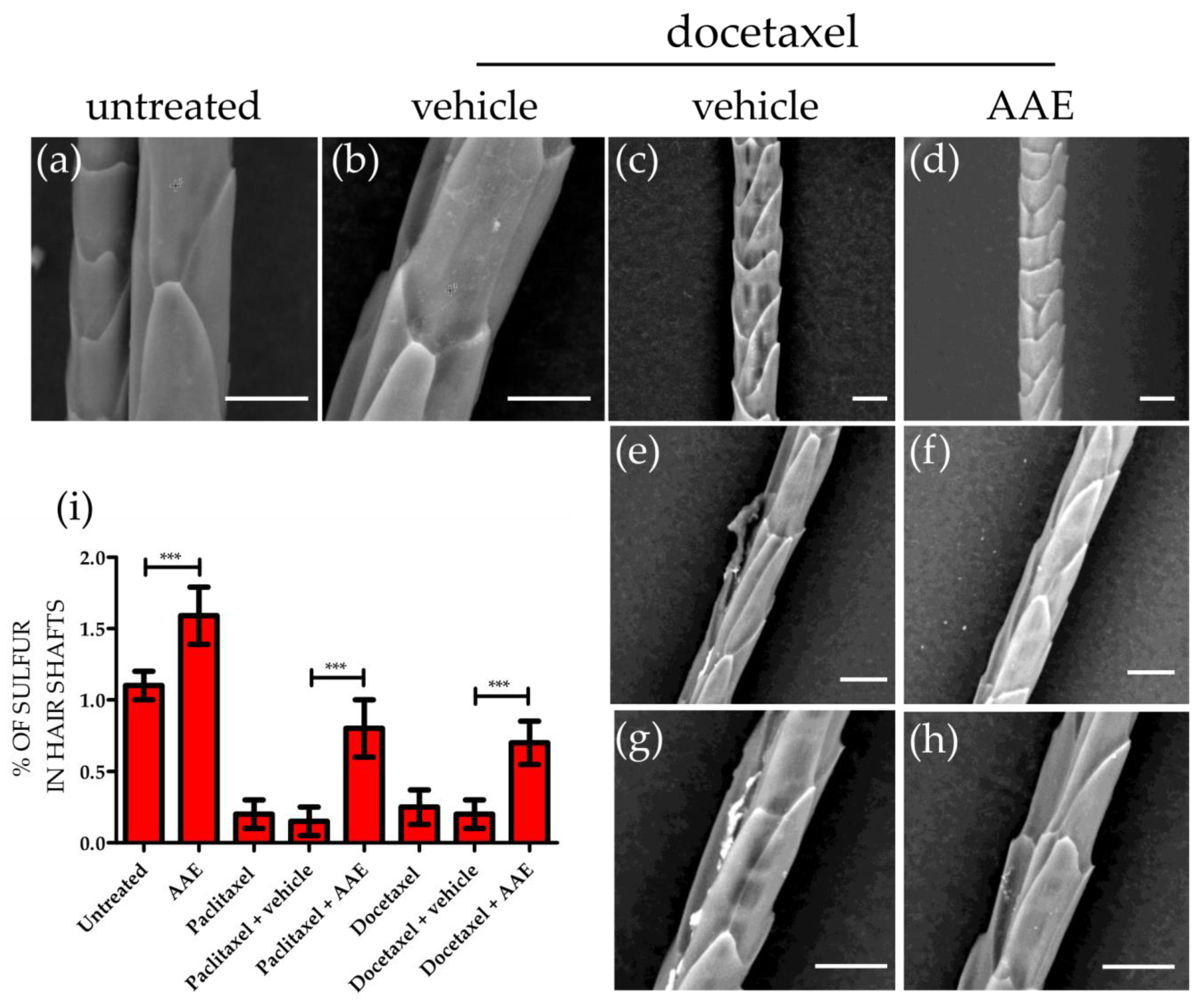
3.3. AAE Preserves Keratin Production in Murine HFs Treated with Taxanes
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Fuchs, E. The Tortoise and the Hair-Slow-Cycling Cells in the Stem Cell Race.pdf. Cell 2009, 137, 811–819. [Google Scholar] [CrossRef] [PubMed]
- Fuchs, E.; Merrill, B.J.; Jamora, C.; DasGupta, R. At the roots of a never-ending Cycle. Dev. Cell 2001, 1, 13–25. [Google Scholar] [CrossRef]
- Bernard, B. Advances in Understanding Hair Growth. F1000Research 2016, 5, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Sonthalia, S. Hair Restoration in Androgenetic Alopecia: Looking Beyond Minoxidil, Finasteride and Hair Transplantation. J. Cosmetol. Trichol. 2016, 2, 1–13. [Google Scholar] [CrossRef]
- Epstein, F.H.; Paus, R.; Cotsarelis, G. The Biology of Hair Follicles. N. Engl. J. Med. 1999, 341, 491–497. [Google Scholar] [CrossRef]
- Botchkarev, V.A. Molecular mechanisms of chemotherapy-induced hair loss. J. Investig. Dermatol. Symp. Proc. 2003, 8, 72–75. [Google Scholar] [CrossRef] [PubMed]
- Trueb, R. Oxidative stress in ageing of hair. Int. J. Trichol. 2009, 1, 6. [Google Scholar] [CrossRef] [PubMed]
- Paus, R. Migrating melanocyte stem cells: Masters of disaster? Nat. Med. 2013, 19, 818–819. [Google Scholar] [CrossRef] [PubMed]
- Hendrix, S.; Handjiski, B.; Peters, E.M.J.; Paus, R. A guide to assessing damage response pathways of the hair follicle: Lessons from cyclophosphamide-induced alopecia in mice. J. Investig. Dermatol. 2005, 125, 42–51. [Google Scholar] [CrossRef] [PubMed]
- Cece, R.; Cazzaniga, S.; Morelli, D.; Sfondrini, L.; Bignotto, M.; Menard, S.; Colnaghi, M.I.; Balsari, A. Apoptosis of hair follicle cells during doxorubicin-induced alopecia in rats. Lab. Investig. 1996, 75, 601–609. [Google Scholar] [PubMed]
- Lindner, G.; Botchkarev, V.A.; Botchkareva, N.V.; Ling, G.; van der Veen, C.; Paus, R. Analysis of apoptosis during hair follicle regression (catagen). Am. J. Pathol. 1997, 151, 1601–1617. [Google Scholar] [PubMed]
- Bodó, E.; Tobin, D.J.; Kamenisch, Y.; Bíró, T.; Berneburg, M.; Funk, W.; Paus, R. Dissecting the impact of chemotherapy on the human hair follicle: A pragmatic in vitro assay for studying the pathogenesis and potential management of hair follicle dystrophy. Am. J. Pathol. 2007, 171, 1153–1167. [Google Scholar] [CrossRef] [PubMed]
- Markman, M. Management of toxicities associated with the administration of taxanes. Expert Opin. Drug Saf. 2003, 2, 141–146. [Google Scholar] [CrossRef] [PubMed]
- Baati, I.; Mnif, L.; Masmoudi, J.; Allouche, C.; Damak, R.; Halwani, N.; Feki, A.; Ayadi, N.; Jaoua, A. P03-10-Psychological impact of chemotherapy induced alopecia. Eur. Psychiatry 2010, 25, 869. [Google Scholar] [CrossRef]
- Leirõs, G.J.; Attorresi, A.I.; Balañá, M.E. Hair follicle stem cell differentiation is inhibited through cross-talk between Wnt/β-catenin and androgen signalling in dermal papilla cells from patients with androgenetic alopecia. Br. J. Dermatol. 2012, 166, 1035–1042. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.-H.; Yoon, J.; Shin, S.H.; Zahoor, M.; Kim, H.J.; Park, P.J.; Park, W.-S.; Min, D.S.; Kim, H.-Y.; Choi, K.-Y. Valproic Acid Induces Hair Regeneration in Murine Model and Activates Alkaline Phosphatase Activity in Human Dermal Papilla Cells. PLoS ONE 2012, 7, e34152. [Google Scholar] [CrossRef] [PubMed]
- Zimber, M.P.; Ziering, C.; Zeigler, F.; Hubka, M.; Mansbridge, J.N.; Baumgartner, M.; Hubka, K.; Kellar, R.; Perez-Meza, D.; Sadick, N.; et al. Hair regrowth following a Wnt- and follistatin containing treatment: Safety and efficacy in a first-in-man phase 1 clinical trial. J. Drugs Dermatol. 2011, 10, 1308–1312. [Google Scholar] [PubMed]
- Takahashi, T.; Kamimura, A.; Yokoo, Y.; Honda, S.; Watanabe, Y. The first clinical trial of topical application of procyanidin B-2 to investigate its potential as a hair growing agent. Phyther. Res. 2001, 15, 331–336. [Google Scholar] [CrossRef] [PubMed]
- Tenore, G.C.; Campiglia, P.; Ritieni, A.; Novellino, E. In vitro bioaccessibility, bioavailability and plasma protein interaction of polyphenols from Annurca apple (M. pumila Miller cv Annurca). Food Chem. 2013, 141, 3519–3524. [Google Scholar] [CrossRef] [PubMed]
- Tenore, G.C.; Caruso, D.; Buonomo, G.; D’Avino, M.; Santamaria, R.; Irace, C.; Piccolo, M.; Maisto, M.; Novellino, E. Annurca Apple Nutraceutical Formulation Enhances Keratin Expression in a Human Model of Skin and Promotes Hair Growth and Tropism in a Randomized Clinical Trial. J. Med. Food 2018, 21, 90–103. [Google Scholar] [CrossRef] [PubMed]
- Badolati, N.; Sommella, E.; Riccio, G.; Salviati, E.; Heintz, D.; Bottone, S.; Di Cicco, E.; Dentice, M.; Tenore, G.; Campiglia, P.; et al. Annurca Apple Polyphenols Ignite Keratin Production in Hair Follicles by Inhibiting the Pentose Phosphate Pathway and Amino Acid Oxidation. Nutrients 2018, 10, 1406. [Google Scholar] [CrossRef] [PubMed]
- Williams, R.; Philpott, M.P.; Kealey, T. Metabolism of freshly isolated human hair follicles capable of hair elongation: A glutaminolytic, aerobic glycolytic tissue. J. Investig. Dermatol. 1993, 100, 834–840. [Google Scholar] [CrossRef] [PubMed]
- Colombe, L.; Vindrios, A.; Michelet, J.-F.; Bernard, B.A. Prostaglandin metabolism in human hair follicle. Exp. Dermatol. 2007, 16, 762–769. [Google Scholar] [CrossRef] [PubMed]
- Choi, Y.M.; Diehl, J.; Levins, P.C. Promising alternative clinical uses of prostaglandin F2α analogs: Beyond the eyelashes. J. Am. Acad. Dermatol. 2015, 72, 712–716. [Google Scholar] [CrossRef] [PubMed]
- Garza, L.A.; Liu, Y.; Yang, Z.; Alagesan, B.; Lawson, J.A.; Norberg, S.M.; Loy, D.E.; Zhao, T.; Blatt, H.B.; Stanton, D.C.; et al. Prostaglandin D2inhibits hair growth and is elevated in bald scalp of men with androgenetic alopecia. Sci. Transl. Med. 2012, 4, 126ra34. [Google Scholar] [CrossRef] [PubMed]
- Bernard, B.A.; Michelet, J.F.; Colombe, L. Prostanoid receptors in anagen human hair follicles. Exp. Dermatol. 2008, 17, 63–72. [Google Scholar] [CrossRef]
- Abel, S.; Riedel, S.; Gelderblom, W.C.A. Dietary PUFA and cancer. Proc. Nutr. Soc. 2014, 73, 361–367. [Google Scholar] [CrossRef] [PubMed]
- Bartsch, H.; Nair, J.; Owen, R.W. Dietary polyunsaturated fatty acids and cancers of the breast and colorectum: Emerging evidence for their role as risk modifiers. Carcinogenesis 1999, 20, 2209–2218. [Google Scholar] [CrossRef] [PubMed]
- Munkhbayar, S.; Jang, S.; Cho, A.R.; Choi, S.J.; Shin, C.Y.; Eun, H.C.; Kim, K.H.; Kwon, O. Role of arachidonic acid in promoting hair growth. Ann. Dermatol. 2016, 28, 55–64. [Google Scholar] [CrossRef] [PubMed]
- Le Floc’h, C.; Cheniti, A.; Connétable, S.; Piccardi, N.; Vincenzi, C.; Tosti, A. Effect of a nutritional supplement on hair loss in women. J. Cosmet. Dermatol. 2015, 14, 76–82. [Google Scholar] [CrossRef] [PubMed]
- Blume-Peytavi, U.; Lönnfors, S.; Hillmann, K.; Garcia Bartels, N. A randomized double-blind placebo-controlled pilot study to assess the efficacy of a 24-week topical treatment by latanoprost 0.1% on hair growth and pigmentation in healthy volunteers with androgenetic alopecia. J. Am. Acad. Dermatol. 2012, 66, 794–800. [Google Scholar] [CrossRef] [PubMed]
- Sasaki, S.; Hozumi, Y.; Kondo, S. Influence of prostaglandin F2alpha and its analogues on hair regrowth and follicular melanogenesis in a murine model. Exp. Dermatol. 2005, 14, 323–328. [Google Scholar] [CrossRef] [PubMed]
- Sommella, E.; Conte, G.M.; Salviati, E.; Pepe, G.; Bertamino, A.; Ostacolo, C.; Sansone, F.; Prete, F.D.; Aquino, R.P.; Campiglia, P. Fast profiling of natural pigments in different spirulina (arthrospira platensis) dietary supplements by DI-FT-ICR and evaluation of their antioxidant potential by pre-column DPPH-UHPLC assay. Molecules 2018, 23, 1132. [Google Scholar] [CrossRef] [PubMed]
- Riccio, G.; Maisto, M.; Bottone, S.; Badolati, N.; Rossi, G.B.; Tenore, G.C.; Stornaiuolo, M.; Novellino, E. WNT inhibitory activity of malus pumila miller cv annurca and malus domestica cv limoncella apple extracts on human colon-rectal cells carrying familial adenomatous polyposis mutations. Nutrients 2017, 9, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Riccio, G.; Bottone, S.; La Regina, G.; Badolati, N.; Passacantilli, S.; Rossi, G.B.; Accardo, A.; Dentice, M.; Silvestri, R.; Novellino, E.; et al. A Negative Allosteric Modulator of WNT Receptor Frizzled 4 Switches into an Allosteric Agonist. Biochemistry 2018, 57, 839–851. [Google Scholar] [CrossRef] [PubMed]
- Forslind, B. Clinical applications of scanning electron microscopy and energy dispersive X-ray analysis in dermatology—An up-date. Scanning Microsc. 1988, 2, 959–976. [Google Scholar] [PubMed]
- Ser, Z.; Liu, X.; Tang, N.N.; Locasale, J.W. Extraction parameters for metabolomics from cell extracts Zheng. Anal. Biochem. 2015, 475, 87–92. [Google Scholar] [CrossRef] [PubMed]
- Pepe, G.; Pagano, F.; Adesso, S.; Sommella, E.; Ostacolo, C.; Manfra, M.; Chieppa, M.; Sala, M.; Russo, M.; Marzocco, S.; et al. Bioavailable Citrus sinensis extract: Polyphenolic composition and biological activity. Molecules 2017, 22. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.H.; Cho, S.; Lee, S.; Kim, K.H.; Cho, K.H.; Eun, H.C.; Chung, J.H. Photoprotective and anti-skin-aging effects of eicosapentaenoic acid in human skin in vivo. J. Lipid Res. 2006, 47, 921–930. [Google Scholar] [CrossRef] [PubMed]
- Müller-Röver, S.; Handjiski, B.; Van Der Veen, C.; Eichmüller, S.; Foitzik, K.; McKay, I.A.; Stenn, K.S.; Paus, R. A comprehensive guide for the accurate classification of murine hair follicles in distinct hair cycle stages. J. Investig. Dermatol. 2001, 117, 3–15. [Google Scholar] [CrossRef] [PubMed]
- Askari, A.A.; Thomson, S.; Edin, M.L.; Lih, F.B.; Zeldin, D.C.; Bishop-Bailey, D. Basal and inducible anti-inflammatory epoxygenase activity in endothelial cells. Biochem. Biophys. Res. Commun. 2014, 446, 633–637. [Google Scholar] [CrossRef] [PubMed]
- Roda-Navarro, P.; Reyburn, H.T. The Traffic of the NKG2D/Dap10 Receptor Complex during Natural Killer (NK) Cell Activation. J. Biol. Chem. 2009, 284, 16463–16472. [Google Scholar] [CrossRef] [PubMed]
- Nieves, A.; Garza, L.A. Does prostaglandin D2hold the cure to male pattern baldness? Exp. Dermatol. 2014, 23, 224–227. [Google Scholar] [CrossRef] [PubMed]
- Hanson, W.R.; Pelka, A.E.; Nelson, A.K.; Malkinson, F.D. Subcutaneous or topical administration of 16,16 dimethyl prostaglandin E2protects from radiation-induced alopecia in mice. Int. J. Radiat. Oncol. Biol. Phys. 1992, 23, 333–337. [Google Scholar] [CrossRef]
- Malkinson, D.; Hanson, R. Prostaglandins Protect Against Murine Hair Injury Produced by Ionizing Radiation or Doxorubicin. J. Investig. Dermatol. 1993, 101, 135–137. [Google Scholar] [CrossRef]
- De Souza, F.H.M.; Fernando, L.; Torres, B. Quantitative evaluation of transverse scalp sections * Avaliação quantitativa em cortes histológicos transversais. Anais Brasileiros de Dermatologia 2006, 81, 227–232. [Google Scholar] [CrossRef]
- Sommella, E.; Pepe, G.; Pagano, F.; Ostacolo, C.; Tenore, G.C.; Russo, M.T.; Novellino, E.; Manfra, M.; Campiglia, P. Detailed polyphenolic profiling of Annurca apple (M. pumila Miller cv Annurca) by a combination of RP-UHPLC and HILIC, both hyphenated to IT-TOF mass spectrometry. Food Res. Int. 2015, 76, 466–477. [Google Scholar] [CrossRef] [PubMed]
- Stirpe, M.; Palermo, V.; Bianchi, M.M.; Silvestri, R.; Falcone, C.; Tenore, G.; Novellino, E.; Mazzoni, C. Annurca apple (M. pumila Miller cv Annurca) extracts act against stress and ageing in S. cerevisiae yeast cells. BMC Complement. Altern. Med. 2017, 17, 200. [Google Scholar] [CrossRef] [PubMed]
- Tenore, G.C.; Caruso, D.; Buonomo, G.; D’Urso, E.; D’Avino, M.; Campigli, P.; Marinelli, L.; Novellino, E. Annurca (Malus pumilamiller cv. Annurca) apple as a functional food for the contribution to a healthy balance of plasma cholesterol levels: Results of a randomized clinical trial. J. Sci. Food Agric. 2016, 97, 2107–2115. [Google Scholar] [CrossRef] [PubMed]
- Tenore, G.C.; Calabrese, G.; Stiuso, P.; Ritieni, A.; Giannetti, D.; Novellino, E. Effects of Annurca apple polyphenols on lipid metabolism in HepG2 cell lines: A source of nutraceuticals potentially indicated for the metabolic syndrome. Food Res. Int. 2014, 63, 252–257. [Google Scholar] [CrossRef]
| PUFA | Metabolite | Fold Change 1 | PUFA | Metabolite | Fold Change 1 |
|---|---|---|---|---|---|
| ARA | DHA | ||||
| ARA 20:4ω-6 * | 1.0 ± 0.1 | DHA 22:6ω-3 * | 0.9 ± 0.2 | ||
| Tetranor 12-HETE | 3.4 ± 0.2 | 17-HDoHE | 1.0 ± 0.1 | ||
| 14,15-DiHETrE | 3.5 ± 0.1 | 13-HDoHE | 1.1 ± 0.1 | ||
| 2,3-Dinor-6-keto-PGF1α | 1.6 ± 0.2 | 19,20-DiHDPA | 1.3 ± 0.1 | ||
| 15-Keto-13,14-dihydroPGA2 | 1.0 ± 0.1 | ||||
| PGF2α | 1.6 ± 0.1 | α-LA | |||
| 13,14-dihydro-PGF2α | 2.3 ± 0.2 | α-LA 18:3ω-3 * | 1.1 ± 0.1 | ||
| 15-deoxy-Δ12,14-PGJ2 | 1.1 ± 0.1 | LA | |||
| LA 18:2ω-6 * | 0.9 ± 0.1 | ||||
| EPA | 9,10-DHOME | specific for AAE | |||
| EPA 20:5ω-3 * | 0.9 ± 0.1 | 13-HpODE | 2.3 ± 0.1 | ||
| 5-HEPE | 1.0 ± 0.1 | 9,12,13-TriHOME | 1.3 ± 0.1 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Riccio, G.; Sommella, E.; Badolati, N.; Salviati, E.; Bottone, S.; Campiglia, P.; Dentice, M.; Tenore, G.C.; Stornaiuolo, M.; Novellino, E. Annurca Apple Polyphenols Protect Murine Hair Follicles from Taxane Induced Dystrophy and Hijacks Polyunsaturated Fatty Acid Metabolism toward β-Oxidation. Nutrients 2018, 10, 1808. https://doi.org/10.3390/nu10111808
Riccio G, Sommella E, Badolati N, Salviati E, Bottone S, Campiglia P, Dentice M, Tenore GC, Stornaiuolo M, Novellino E. Annurca Apple Polyphenols Protect Murine Hair Follicles from Taxane Induced Dystrophy and Hijacks Polyunsaturated Fatty Acid Metabolism toward β-Oxidation. Nutrients. 2018; 10(11):1808. https://doi.org/10.3390/nu10111808
Chicago/Turabian StyleRiccio, Gennaro, Eduardo Sommella, Nadia Badolati, Emanuela Salviati, Sara Bottone, Pietro Campiglia, Monica Dentice, Gian Carlo Tenore, Mariano Stornaiuolo, and Ettore Novellino. 2018. "Annurca Apple Polyphenols Protect Murine Hair Follicles from Taxane Induced Dystrophy and Hijacks Polyunsaturated Fatty Acid Metabolism toward β-Oxidation" Nutrients 10, no. 11: 1808. https://doi.org/10.3390/nu10111808
APA StyleRiccio, G., Sommella, E., Badolati, N., Salviati, E., Bottone, S., Campiglia, P., Dentice, M., Tenore, G. C., Stornaiuolo, M., & Novellino, E. (2018). Annurca Apple Polyphenols Protect Murine Hair Follicles from Taxane Induced Dystrophy and Hijacks Polyunsaturated Fatty Acid Metabolism toward β-Oxidation. Nutrients, 10(11), 1808. https://doi.org/10.3390/nu10111808











