Impact of a Healthy Dietary Pattern on Gut Microbiota and Systemic Inflammation in Humans
Abstract
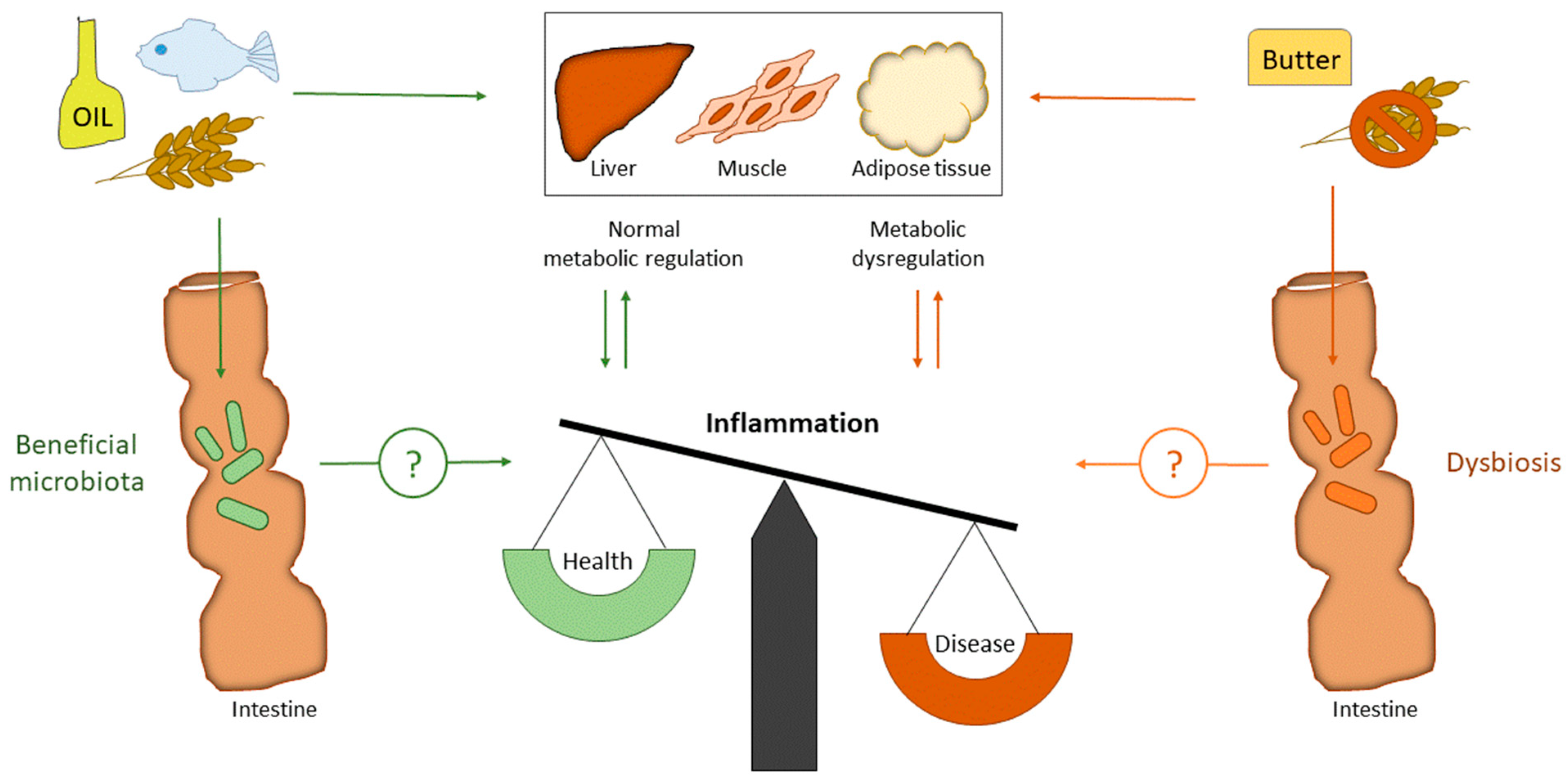
1. Introduction
2. Materials and Methods
3. Results and Discussion
3.1. Dietary Fiber and Whole Grains
3.2. Dietary Fat
3.3. Other Dietary Factors
3.4. Diets
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Tabas, I.; Glass, C.K. Anti-inflammatory therapy in chronic disease: Challenges and opportunities. Science 2013, 339, 166–172. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, C.M.; Vieira, A.T.; Vinolo, M.A.; Oliveira, F.A.; Curi, R.; Martins, F.S. The central role of the gut microbiota in chronic inflammatory diseases. J. Immunol. Res. 2014, 2014, 689492. [Google Scholar] [CrossRef] [PubMed]
- Hansen, T.H.; Gobel, R.J.; Hansen, T.; Pedersen, O. The gut microbiome in cardio-metabolic health. Genome. Med. 2015, 7, 33. [Google Scholar] [CrossRef] [PubMed]
- Barko, P.C.; McMichael, M.A.; Swanson, K.S.; Williams, D.A. The Gastrointestinal Microbiome: A Review. J. Vet. Intern. Med. 2018, 32, 9–25. [Google Scholar] [CrossRef] [PubMed]
- Sekirov, I.; Russell, S.L.; Antunes, L.C.; Finlay, B.B. Gut microbiota in health and disease. Physiol. Rev. 2010, 90, 859–904. [Google Scholar] [CrossRef] [PubMed]
- Guarner, F.; Malagelada, J.R. Gut flora in health and disease. Lancet 2003, 361, 512–519. [Google Scholar] [CrossRef]
- Qin, J.; Li, R.; Raes, J.; Arumugam, M.; Burgdorf, K.S.; Manichanh, C.; Nielsen, T.; Pons, N.; Levenez, F.; Yamada, T.; et al. A human gut microbial gene catalogue established by metagenomic sequencing. Nature 2010, 464, 59–65. [Google Scholar] [CrossRef] [PubMed]
- Tremaroli, V.; Backhed, F. Functional interactions between the gut microbiota and host metabolism. Nature 2012, 489, 242–249. [Google Scholar] [CrossRef] [PubMed]
- Ottman, N.; Smidt, H.; de Vos, W.M.; Belzer, C. The function of our microbiota: Who is out there and what do they do? Front. Cell. Infect. Microbiol. 2012, 2, 104. [Google Scholar] [CrossRef] [PubMed]
- Hakansson, A.; Molin, G. Gut microbiota and inflammation. Nutrients 2011, 3, 637–682. [Google Scholar] [CrossRef] [PubMed]
- Candido, F.G.; Valente, F.X.; Grzeskowiak, L.M.; Moreira, A.P.B.; Rocha, D.; Alfenas, R.C.G. Impact of dietary fat on gut microbiota and low-grade systemic inflammation: Mechanisms and clinical implications on obesity. Int. J. Food Sci. Nutr. 2018, 69, 125–143. [Google Scholar] [CrossRef] [PubMed]
- Salonen, A.; de Vos, W.M. Impact of diet on human intestinal microbiota and health. Annu. Rev. Food Sci. Technol. 2014, 5, 239–262. [Google Scholar] [CrossRef] [PubMed]
- Underwood, M.A. Intestinal dysbiosis: Novel mechanisms by which gut microbes trigger and prevent disease. Prev. Med. 2014, 65, 133–137. [Google Scholar] [CrossRef] [PubMed]
- Walker, A.W.; Lawley, T.D. Therapeutic modulation of intestinal dysbiosis. Pharmacol. Res. 2013, 69, 75–86. [Google Scholar] [CrossRef] [PubMed]
- Brun, P.; Castagliuolo, I.; Di Leo, V.; Buda, A.; Pinzani, M.; Palu, G.; Martines, D. Increased intestinal permeability in obese mice: New evidence in the pathogenesis of nonalcoholic steatohepatitis. Am. J. Physiol. Gastrointest. Liver Physiol. 2007, 292, 518–525. [Google Scholar] [CrossRef] [PubMed]
- Le Chatelier, E.; Nielsen, T.; Qin, J.; Prifti, E.; Hildebrand, F.; Falony, G.; Almeida, M.; Arumugam, M.; Batto, J.M.; Kennedy, S.; et al. Richness of human gut microbiome correlates with metabolic markers. Nature 2013, 500, 541–546. [Google Scholar] [CrossRef] [PubMed]
- Peng, L.; Li, Z.R.; Green, R.S.; Holzman, I.R.; Lin, J. Butyrate enhances the intestinal barrier by facilitating tight junction assembly via activation of AMP-activated protein kinase in Caco-2 cell monolayers. J. Nutr. 2009, 139, 1619–1625. [Google Scholar] [CrossRef] [PubMed]
- Ohata, A.; Usami, M.; Miyoshi, M. Short-chain fatty acids alter tight junction permeability in intestinal monolayer cells via lipoxygenase activation. Nutrition 2005, 21, 838–847. [Google Scholar] [CrossRef] [PubMed]
- Correa-Oliveira, R.; Fachi, J.L.; Vieira, A.; Sato, F.T.; Vinolo, M.A. Regulation of immune cell function by short-chain fatty acids. Clin. Transl. Immunol. 2016, 5, e73. [Google Scholar] [CrossRef] [PubMed]
- Kristensen, N.B.; Bryrup, T.; Allin, K.H.; Nielsen, T.; Hansen, T.H.; Pedersen, O. Alterations in fecal microbiota composition by probiotic supplementation in healthy adults: A systematic review of randomized controlled trials. Genome. Med. 2016, 8, 52. [Google Scholar] [CrossRef] [PubMed]
- Hartley, L.; May, M.D.; Loveman, E.; Colquitt, J.L.; Rees, K. Dietary fibre for the primary prevention of cardiovascular disease. Cochrane Database Syst. Rev. 2016, 1, CD011472. [Google Scholar] [CrossRef] [PubMed]
- Tosh, S.M. Review of human studies investigating the post-prandial blood-glucose lowering ability of oat and barley food products. Eur. J. Clin. Nutr. 2013, 67, 310–317. [Google Scholar] [CrossRef] [PubMed]
- Gunness, P.; Gidley, M.J. Mechanisms underlying the cholesterol-lowering properties of soluble dietary fibre polysaccharides. Food Funct. 2010, 1, 149–155. [Google Scholar] [CrossRef] [PubMed]
- Rideout, T.C.; Harding, S.V.; Jones, P.J.; Fan, M.Z. Guar gum and similar soluble fibers in the regulation of cholesterol metabolism: Current understandings and future research priorities. Vasc. Health Risk Manag. 2008, 4, 1023–1033. [Google Scholar] [CrossRef] [PubMed]
- Othman, R.A.; Moghadasian, M.H.; Jones, P.J. Cholesterol-lowering effects of oat beta-glucan. Nutr. Rev. 2011, 69, 299–309. [Google Scholar] [CrossRef] [PubMed]
- De Filippo, C.; Cavalieri, D.; Di Paola, M.; Ramazzotti, M.; Poullet, J.B.; Massart, S.; Collini, S.; Pieraccini, G.; Lionetti, P. Impact of diet in shaping gut microbiota revealed by a comparative study in children from Europe and rural Africa. Proc. Natl. Acad. Sci. USA 2010, 107, 14691–14696. [Google Scholar] [CrossRef] [PubMed]
- Halmos, E.P.; Christophersen, C.T.; Bird, A.R.; Shepherd, S.J.; Gibson, P.R.; Muir, J.G. Diets that differ in their FODMAP content alter the colonic luminal microenvironment. Gut 2015, 64, 93–100. [Google Scholar] [CrossRef] [PubMed]
- Schwab, U.; Lauritzen, L.; Tholstrup, T.; Haldorssoni, T.; Riserus, U.; Uusitupa, M.; Becker, W. Effect of the amount and type of dietary fat on cardiometabolic risk factors and risk of developing type 2 diabetes, cardiovascular diseases, and cancer: A systematic review. Food Nutr. Res. 2014, 58, 25145. [Google Scholar] [CrossRef] [PubMed]
- Taylor, F.; Huffman, M.D.; Macedo, A.F.; Moore, T.H.; Burke, M.; Davey, S.G.; Ward, K.; Ebrahim, S. Statins for the primary prevention of cardiovascular disease. Cochrane Database Syst. Rev. 2011, 1, CD004816. [Google Scholar]
- Gabert, L.; Vors, C.; Louche-Pelissier, C.; Sauvinet, V.; Lambert-Porcheron, S.; Drai, J.; Laville, M.; Desage, M.; Michalski, M.C. 13C tracer recovery in human stools after digestion of a fat-rich meal labelled with [1,1,1-13C3]tripalmitin and [1,1,1-13C3]triolein. Rapid Commun. Mass Spectrom. 2011, 25, 2697–2703. [Google Scholar] [CrossRef] [PubMed]
- Caesar, R.; Tremaroli, V.; Kovatcheva-Datchary, P.; Cani, P.D.; Backhed, F. Crosstalk between Gut Microbiota and Dietary Lipids Aggravates WAT Inflammation through TLR Signaling. Cell Metab. 2015, 22, 658–668. [Google Scholar] [CrossRef] [PubMed]
- Allin, K.H.; Nielsen, T.; Pedersen, O. Mechanisms in endocrinology: Gut microbiota in patients with type 2 diabetes mellitus. Eur. J. Endocrinol. 2015, 172, 167–177. [Google Scholar] [CrossRef] [PubMed]
- Vanegas, S.M.; Meydani, M.; Barnett, J.B.; Goldin, B.; Kane, A.; Rasmussen, H.; Brown, C.; Vangay, P.; Knights, D.; Jonnalagadda, S.; et al. Substituting whole grains for refined grains in a 6-wk randomized trial has a modest effect on gut microbiota and immune and inflammatory markers of healthy adults. Am. J. Clin. Nutr. 2017, 105, 635–650. [Google Scholar] [CrossRef] [PubMed]
- Morales, P.; Fujio, S.; Navarrete, P.; Ugalde, J.A.; Magne, F.; Carrasco-Pozo, C.; Tralma, K.; Quezada, M.; Hurtado, C.; Covarrubias, N.; et al. Impact of Dietary Lipids on Colonic Function and Microbiota: An Experimental Approach Involving Orlistat-Induced Fat Malabsorption in Human Volunteers. Clin. Transl. Gastroenterol. 2016, 7, e161. [Google Scholar] [CrossRef] [PubMed]
- Nicolucci, A.C.; Hume, M.P.; Martinez, I.; Mayengbam, S.; Walter, J.; Reimer, R.A. Prebiotics Reduce Body Fat and Alter Intestinal Microbiota in Children Who Are Overweight or With Obesity. Gastroenterology 2017, 153, 711–722. [Google Scholar] [CrossRef] [PubMed]
- Nilsson, A.; Johansson-Boll, E.; Sandberg, J.; Bjorck, I. Gut microbiota mediated benefits of barley kernel products on metabolism, gut hormones, and inflammatory markers as affected by co-ingestion of commercially available probiotics: A randomized controlled study in healthy subjects. Clin. Nutr. ESPEN 2016, 15, 49–56. [Google Scholar] [CrossRef] [PubMed]
- Benjamin, J.L.; Hedin, C.R.; Koutsoumpas, A.; Ng, S.C.; McCarthy, N.E.; Hart, A.L.; Kamm, M.A.; Sanderson, J.D.; Knight, S.C.; Forbes, A.; et al. Randomised, double-blind, placebo-controlled trial of fructo-oligosaccharides in active Crohn’s disease. Gut 2011, 60, 923–929. [Google Scholar] [CrossRef] [PubMed]
- Vulevic, J.; Juric, A.; Tzortzis, G.; Gibson, G.R. A mixture of trans-galactooligosaccharides reduces markers of metabolic syndrome and modulates the fecal microbiota and immune function of overweight adults. J. Nutr. 2013, 143, 324–331. [Google Scholar] [CrossRef] [PubMed]
- Vetrani, C.; Costabile, G.; Luongo, D.; Naviglio, D.; Rivellese, A.A.; Riccardi, G.; Giacco, R. Effects of whole-grain cereal foods on plasma short chain fatty acid concentrations in individuals with the metabolic syndrome. Nutrition 2016, 32, 217–221. [Google Scholar] [CrossRef] [PubMed]
- Pedersen, C.; Gallagher, E.; Horton, F.; Ellis, R.J.; Ijaz, U.Z.; Wu, H.; Jaiyeola, E.; Diribe, O.; Duparc, T.; Cani, P.D.; et al. Host-microbiome interactions in human type 2 diabetes following prebiotic fibre (galacto-oligosaccharide) intake. Br. J. Nutr. 2016, 116, 1869–1877. [Google Scholar] [CrossRef] [PubMed]
- Canfora, E.E.; van der Beek, C.M.; Hermes, G.D.A.; Goossens, G.H.; Jocken, J.W.E.; Holst, J.J.; van Eijk, H.M.; Venema, K.; Smidt, H.; Zoetendal, E.G.; et al. Supplementation of Diet with Galacto-oligosaccharides Increases Bifidobacteria, but Not Insulin Sensitivity, in Obese Prediabetic Individuals. Gastroenterology 2017, 153, 87–97. [Google Scholar] [CrossRef] [PubMed]
- Martin-Pelaez, S.; Castaner, O.; Sola, R.; Motilva, M.J.; Castell, M.; Perez-Cano, F.J.; Fito, M. Influence of Phenol-Enriched Olive Oils on Human Intestinal Immune Function. Nutrients 2016, 8, 213. [Google Scholar] [CrossRef] [PubMed]
- Balfego, M.; Canivell, S.; Hanzu, F.A.; Sala-Vila, A.; Martinez-Medina, M.; Murillo, S.; Mur, T.; Ruano, E.G.; Linares, F.; Porras, N.; et al. Effects of sardine-enriched diet on metabolic control, inflammation and gut microbiota in drug-naive patients with type 2 diabetes: A pilot randomized trial. Lipids Health Dis. 2016, 15, 78. [Google Scholar] [CrossRef] [PubMed]
- Ravn-Haren, G.; Dragsted, L.O.; Buch-Andersen, T.; Jensen, E.N.; Jensen, R.; Nemeth-Balogh, M.; Paulovicsova, B.; Bergstrom, A.; Wilcks, A.; Licht, T.R.; et al. Intake of whole apples or clear apple juice has contrasting effects on plasma lipids in healthy volunteers. Eur. J. Nutr. 2013, 52, 1875–1889. [Google Scholar] [CrossRef] [PubMed]
- Clemente-Postigo, M.; Queipo-Ortuno, M.I.; Boto-Ordonez, M.; Coin-Araguez, L.; Roca-Rodriguez, M.M.; Delgado-Lista, J.; Cardona, F.; Andres-Lacueva, C.; Tinahones, F.J. Effect of acute and chronic red wine consumption on lipopolysaccharide concentrations. Am. J. Clin. Nutr. 2013, 97, 1053–1061. [Google Scholar] [CrossRef] [PubMed]
- Accardi, G.; Aiello, A.; Gargano, V.; Gambino, C.M.; Caracappa, S.; Marineo, S.; Vesco, G.; Carru, C.; Zinellu, A.; Zarcone, M.; et al. Nutraceutical effects of table green olives: A pilot study with Nocellara del Belice olives. Immun. Ageing 2016, 13, 11. [Google Scholar] [CrossRef] [PubMed]
- Cotillard, A.; Kennedy, S.P.; Kong, L.C.; Prifti, E.; Pons, N.; Le Chatelier, E.; Almeida, M.; Quinquis, B.; Levenez, F.; Galleron, N.; et al. Dietary intervention impact on gut microbial gene richness. Nature 2013, 500, 585–588. [Google Scholar] [CrossRef] [PubMed]
- Umoh, F.I.; Kato, I.; Ren, J.; Wachowiak, P.L.; Ruffin, M.T.; Turgeon, D.K.; Sen, A.; Brenner, D.E.; Djuric, Z. Markers of systemic exposures to products of intestinal bacteria in a dietary intervention study. Eur. J. Nutr. 2016, 55, 793–798. [Google Scholar] [CrossRef] [PubMed]
- Pataky, Z.; Genton, L.; Spahr, L.; Lazarevic, V.; Terraz, S.; Gaia, N.; Rubbia-Brandt, L.; Golay, A.; Schrenzel, J.; Pichard, C. Impact of Hypocaloric Hyperproteic Diet on Gut Microbiota in Overweight or Obese Patients with Nonalcoholic Fatty Liver Disease: A Pilot Study. Dig. Dis. Sci. 2016, 61, 2721–2731. [Google Scholar] [CrossRef] [PubMed]
- Marlow, G.; Ellett, S.; Ferguson, I.R.; Zhu, S.; Karunasinghe, N.; Jesuthasan, A.C.; Han, D.Y.; Fraser, A.G.; Ferguson, L.R. Transcriptomics to study the effect of a Mediterranean-inspired diet on inflammation in Crohn’s disease patients. Hum. Genom. 2013, 7, 24. [Google Scholar] [CrossRef] [PubMed]
- Cani, P.D.; Amar, J.; Iglesias, M.A.; Poggi, M.; Knauf, C.; Bastelica, D.; Neyrinck, A.M.; Fava, F.; Tuohy, K.M.; Chabo, C.; et al. Metabolic endotoxemia initiates obesity and insulin resistance. Diabetes 2007, 56, 1761–1772. [Google Scholar] [CrossRef] [PubMed]
- Gibson, G.R.; Beatty, E.R.; Wang, X. Cummings JH: Selective stimulation of bifidobacteria in the human colon by oligofructose and inulin. Gastroenterology 1995, 108, 975–982. [Google Scholar] [CrossRef]
- Ramirez-Farias, C.; Slezak, K.; Fuller, Z.; Duncan, A.; Holtrop, G.; Louis, P. Effect of inulin on the human gut microbiota: Stimulation of Bifidobacterium adolescentis and Faecalibacterium prausnitzii. Br. J. Nutr. 2009, 101, 541–550. [Google Scholar] [CrossRef] [PubMed]
- David, L.A.; Maurice, C.F.; Carmody, R.N.; Gootenberg, D.B.; Button, J.E.; Wolfe, B.E.; Ling, A.V.; Devlin, A.S.; Varma, Y.; Fischbach, M.A.; et al. Diet rapidly and reproducibly alters the human gut microbiome. Nature 2014, 505, 559–563. [Google Scholar] [CrossRef] [PubMed]
- Johnson, E.L.; Heaver, S.L.; Walters, W.A.; Ley, R.E. Microbiome and metabolic disease: Revisiting the bacterial phylum Bacteroidetes. J. Mol. Med. 2017, 95, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Hiippala, K.; Jouhten, H.; Ronkainen, A.; Hartikainen, A.; Kainulainen, V.; Jalanka, J.; Satokari, R. The Potential of Gut Commensals in Reinforcing Intestinal Barrier Function and Alleviating Inflammation. Nutrients 2018, 10, 988. [Google Scholar] [CrossRef] [PubMed]
- Wexler, H.M. Bacteroides: The good, the bad, and the nitty-gritty. Clin. Microbiol. Rev. 2007, 20, 593–621. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Zhi, F. Lower Level of Bacteroides in the Gut Microbiota Is Associated with Inflammatory Bowel Disease: A Meta-Analysis. Biomed. Res. Int. 2016, 2016, 5828959. [Google Scholar] [CrossRef]
- Gibiino, G.; Lopetuso, L.R.; Scaldaferri, F.; Rizzatti, G.; Binda, C.; Gasbarrini, A. Exploring Bacteroidetes: Metabolic key points and immunological tricks of our gut commensals. Dig. Liver. Dis. 2018, 50, 635–639. [Google Scholar] [CrossRef] [PubMed]
- Komaroff, A.L. The Microbiome and Risk for Obesity and Diabetes. JAMA 2017, 317, 355–356. [Google Scholar] [CrossRef] [PubMed]
- Ley, R.E.; Turnbaugh, P.J.; Klein, S.; Gordon, J.I. Microbial ecology: Human gut microbes associated with obesity. Nature 2006, 444, 1022–1023. [Google Scholar] [CrossRef] [PubMed]
- Turnbaugh, P.J.; Ley, R.E.; Mahowald, M.A.; Magrini, V.; Mardis, E.R.; Gordon, J.I. An obesity-associated gut microbiome with increased capacity for energy harvest. Nature 2006, 444, 1027–1031. [Google Scholar] [CrossRef] [PubMed]
- Queipo-Ortuno, M.I.; Boto-Ordonez, M.; Murri, M.; Gomez-Zumaquero, J.M.; Clemente-Postigo, M.; Estruch, R.; Cardona, D.F.; Andres-Lacueva, C.; Tinahones, F.J. Influence of red wine polyphenols and ethanol on the gut microbiota ecology and biochemical biomarkers. Am. J. Clin. Nutr. 2012, 95, 1323–1334. [Google Scholar] [CrossRef] [PubMed]
- Koutsos, A.; Tuohy, K.M.; Lovegrove, J.A. Apples and cardiovascular health--is the gut microbiota a core consideration? Nutrients 2015, 7, 3959–3998. [Google Scholar] [CrossRef] [PubMed]
- Estruch, R.; Ros, E.; Salas-Salvado, J.; Covas, M.I.; Corella, D.; Aros, F.; Gomez-Gracia, E.; Ruiz-Gutierrez, V.; Fiol, M.; Lapetra, J.; et al. Primary Prevention of Cardiovascular Disease with a Mediterranean Diet Supplemented with Extra-Virgin Olive Oil or Nuts. N. Engl. J. Med. 2018, 378, e34. [Google Scholar] [CrossRef] [PubMed]
- Del Chierico, F.; Vernocchi, P.; Dallapiccola, B.; Putignani, L. Mediterranean diet and health: Food effects on gut microbiota and disease control. Int. J. Mol. Sci. 2014, 15, 11678–11699. [Google Scholar] [CrossRef] [PubMed]
- Kim, C.S.; Park, H.S.; Kawada, T.; Kim, J.H.; Lim, D.; Hubbard, N.E.; Kwon, B.S.; Erickson, K.L.; Yu, R. Circulating levels of MCP-1 and IL-8 are elevated in human obese subjects and associated with obesity-related parameters. Int. J. Obes. 2006, 30, 1347–1355. [Google Scholar] [CrossRef] [PubMed]
- Gangarapu, V.; Yildiz, K.; Ince, A.T.; Baysal, B. Role of gut microbiota: Obesity and NAFLD. Turk. J. Gastroenterol. 2014, 25, 133–140. [Google Scholar] [CrossRef] [PubMed]
| Taxonomic Levels of Bacteria | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Domain/Kingdom | Bacteria | ||||||||
| Phylum | Firmicutes | Bacteroidetes | |||||||
| Class | Clostridia | Bacilli | Bacteroidetes | ||||||
| order | Clostridiales | Lactobacilliales | Bacteroidales | ||||||
| family | Clostridiaceae | Lachnospiraceae | Lactobacillacea | Streptococcaceae | Prevotellaceae | Bacteroidaceae | |||
| genus | Clostridium | Dorea | Lachnospira | Roseburia | Lactobacillus | Streptococcus | Enterococcus | Prevotella | Bacteroides |
| Study | Population | Time of Intervention | Treatment | Changes in Gut Microbiota * | Changes in Inflammatory Markers * |
|---|---|---|---|---|---|
| Dietary fiber and wholegrain | |||||
| Vanegas SM et al.; AJCN, 2017, USA | n = 81, M/F, RG gr: 54 year, BMI 26 WG gr: 55 year, BMI 26 | 6 weeks | (1) Recommendations + Refined grain diet (8 g/1000 kcal) (C) (2) Recommendations + Wholegrain diet (16 g/1000 kcal) | ↑ Lachnospira ↓ Enterobacteriaceae ↔ Firmicutes, Bacteroidetes, Proteobacteria, Actinobacteria, Tenericutes, Verrucomicrobia, Cyanobacteria, Fusobacteria, Lentisphaerae, Roseburia Within groups: RG: ↓ Total SCFA, acetate, propionate WG: ↓ Total SCFA, propionate | ↔ TNFα, IL-8, IL-6, IL-1β, LBP ↑ TNFα (LPS stimulated blood) ↔ IFNγ, IL-8, IL-10, IL-6, IL-1β (LPS stimulated blood) ↔ IFNγ, IL-17, TNFα, IL-6, TGF-β (feces) |
| Morales P et al.; Clin Transl Gastroenterol, 2016, Chile | n = 41, M/F, C: 24 year, BMI 22.2 P: 24 year, BMI 23.1 O: 26.5 year, BMI 23.0 OP: 25.5 year, BMI 22.2 | 1 week | A fat-standardized diet (60 g/day) plus one of the following: (1) Maltodextrin (2 × 120 mg maltodextrin+ 16 g maltodextrin) (C) (2) Prebiotic (2 × 120 mg maltodextrin + 16 g oligofructose) (P) (3) Orlistat (2 × 120 mg Orlistat + 16 g maltodextrin) (O) (4) Orlistat/Prebiotic (2 × 120 mg Orlistat + 16 g oligofructose) (OP) | ↔ Gut barrier function ↔ Cyanobacteria, Actinobacteria, Erysipelotrichaceae, Bifidobacteroaceae, Tissierellaceae, Enterobacteriaceae, Barnesiellaceae, Verrucomicrobiaceae ↔ SCFA (feces) Within groups: OP: ↑ Bifidobacterium, OP: ↓ Isobutyrate, isovalerate (feces) | ↔ hsCRP, IL-6 ↔ Calprotectin Within groups: OP: ↓ hsCRP C, P, and O: ↓ IL-6 O: ↑ Calprotectin P and OP: ↓ Calprotectin |
| Nicolucci A et al. Gastroenterology, 2017, Canada | n = 42, M/F, C: 10.2 year, BMI 26.9 OI: 10.4 y, BMI 26.3 | 16 weeks | (1) Maltodextrin (3.3 g/day) (isocaloric) (C) (2) Oligofructose-enriched inulin (OI) (8 g/day) | ↑ Bifidobacterium spp. Within groups: OI: ↑ Actinobacteria, ↑ Bifidobacterium, Collinsella, ↓ Ruminococcus C: ↓ Clostridium XVIII, Actinomyces, Dorea, Eggerthella | ↔ CRP, IFNγ, IL-10, IL-1b, TNFα, IL-4, IL-33, MCP-1, LPS ↓ IL-6 |
| Nilsson A, et al.; Clin Nutr ESPEN, 2016, Sweden | n = 21, M/F, 23.9 year, BMI 22.6 | 3 × 14 days (crossover) | (1) White wheat flour bread (WWB-ref) (148 g/day) (C) (2) Barley kernel bread without probiotics (BB) (256 g/day) (3) Barley kernel bread with probiotics (BB-pro) (256 g/day) Post-study: breakfast with WWB | ↑ Breath H2 after BB and BB-pro groups | ↔ CRP, IL-6, IL-18 ↑ s-PAI-1 after BB-pro |
| Other dietary components | |||||
| Ravn-Haren G et al.; EJN, 2013, Germany | n = 23, M/F, 36.2 year, BMI 22.3 | 5 × 4 weeks (crossover) | (1) No supplement (C) (2) Whole apples (550 g/day) (3) Apple pomace (22 g/day) (4) Clear apple juice (500 mL/day) (5) Cloudy apple juice (500 mL/day) | ↔ Bifidobacterium adolescentis, Bifidobacterium pseudocatenulatum, Bifidobacterium bifidum, Clostridium clusters XI and XVIa, Bacteroides | ↔ hsCRP |
| Clemente-Postigo M et al.; AJCN, 2013, Spain | n = 10, M, 48 year, BMI 27.6 Post-study fat overload: n = 5, M, 41.8 year, BMI 28.4 | 3 × 20 days (crossover) | (1) Red wine (RW) (272 mL/day) (2) Dealcoholized red wine (DRW) (272 mL/day) (3) Gin (100 mL/day) Post-study fat overload challenge: (1) 50 g FO (2) 50 g FO +RW (272 mL) (3) 50 g FO + DRW (272 mL) (4) 50 g FO + gin (100 mL) | ↑ Firmicutes, Bacteoidetes after RW compared to DRW and gin ↓ Firmicutes, Bacteoidetes after gin compared to RW and DRW ↑ Prevotella after RW compared to DRW and gin ↑ Bifidobacterium after RW and DRW compared to gin | ↔ LPS and LBP ↔ LPS and LBP post-study fat overload |
| Accardi G et al.; Immun Ageing, 2016, Italy | n = 25, M/F, 38,3 year, BMI 24.4 | 30 days | (1) No control group (2) Table green olives (12 olives/d) | ↔ Lactobacilli | ↓ IL-6 |
| Diets | |||||
| Cotillard A et al. Nature, 2013, France | n = 49, M/F, - year, BMI 33.2 | 12 weeks | No control group Period 1: Energy-restricted, high-protein diet (6 weeks) Period 2: Weight maintenance diet (6 weeks) | LGC: ↑ gene richness at 6 weeks, and remained higher than baseline at week 12 HGC: ↔ gene richness at 6 week | ↓ CRP 12 weeks ↓ IL-6 12 weeks Split the group to LGC (40%) and HGC (60%): LGC: ↑ CRP at 6 weeks compared to HGC |
| Umoh FI et al.; EJN, 2016, US | n = 120, M/F, healthy, 52 year, BMI 27.4 | 6 months | (1) Healthy eating diet (C) (2) Mediterranean diet | Within groups: ↓ Branched-chain bacterial fatty acids within both groups | ↔ LBP, IL-1β, IL-6, IL-8, IFNγ, TNFα, IL-4, IL-10, IL-13 |
| Study | Population | Duration | Treatment | Changes in Gut Microbiota * | Changes in Inflammatory Markers * |
|---|---|---|---|---|---|
| Dietary fiber and wholegrain | |||||
| Benjamin JL et al.; Gut, 2011, UK | n = 103, M/F, Crohn’s disease, C: 39 year FOS: 40 year BMI - | 4 weeks | (1) Maltodextrin (15g/day) (C) (2) FOS (15g/day) | ↔ Bifidobacteria, F. prausnitzii | ↓ IL-6 (dendritic cells) ↔ IL-12p40 (dendritic cells) Within groups: FOS: ↑ IL-10 (dendritic cells) |
| Vulevic J et al.; JN, 2013, UK | n = 45, M/F, ≥3 risk factors of MetS M: 42.8 year, BMI 30.7 F: 46.4 year, BMI 32.1 | 12 weeks | (1) Maltodextrin (5.5g/day) (C) (2) B-GOS (5.5g/day) | ↑ Bifidobacteria ↓ Bacteroides spp.; C. histolyticum group, Desulfovibrio spp. ↔ Total bacteria, Lactobacillus/Enterococcus spp.; Clostridium coccoides/Eubacterium rectale group, Atopobium cluster, E. cylindroides, E. hallii, β-Proteobacteria, Clostridium cluster IX, F. prausnitzii cluster | ↓ CRP, calprotectin ↔ G-CSF, IL-6, IL-10, IL-8, TNFα |
| Vetrani C et al.; Nutrition. 2016, Italy | n = 54, M/F, MetS C: 58.4 year, BMI 31.5 Wholegrain: 57.2 year, BMI 32.1 | 12 weeks | (1) Refined cereal goods (C) (2) Whole-grain cereal products | ↑ Propionate | ↔ hsCRP, TNFα, IL-1RA, IL-6 |
| Pedersen C, et al.; Br J Nutr.; 2016, UK | n = 29, M, T2D, C: 58.1 year, BMI 28.4, GOS: 56.7, BMI 28.0 | 12 weeks | (1) Maltodextrin (5.5 g/day) (C) (2) GOS mixture (5.5 g/day) | ↔ Bacterial abundance, diversity and richness Within groups: ↔ Total bacteria, Lactobacillus Roseburia, Enteroacteriaceae, Clostridium leptum or Clostridium coccoides groups. | ↔ LPS, LBP, sCD14, hsCRP, IL-6, TNFα |
| Canfora EE et al. Gastroenterology, 2017, Nederlands | n = 44, M/F Prediabetic, C: 58.4 year, BMI 32.3 GOS: 59.2, BMI 33.3 | 12 weeks | (1) Maltodextrin (15g/day) (C) (2) GOS (15 g/day) | ↔ Microbial richness and diversity ↑ Bifidobacterium species, Prevotella oralis et rel.; Prevotella melaninogenica et rel, Bacteroides stercoris et rel and Sutterella wadsworthia et rel ↔ Acetate, propionate, butyrate ↔ Acetate, propionate, butyrate (feces) | ↔ IL-6, IL-8, TNF-α, LBP |
| Dietary fat | |||||
| Martin-Pelaez S et al.; Nutrients, 2016, Spain | n = 10, M/F, hypercholesterolemia, 35–80 year, VOO BMI 28.6 FVOO BMI 28.3, FVOOT BMI 28.4 | 3 weeks (crossover) | (1) OO with 80 mg PC/kg (VOO) (25 mL/day) (2) OO with 500 mg PC/kg from OO (FVOO) (25 mL/day) (3) OO with a mixture of 500 mg PC/kg from OO and thyme (1:1, FVOOT) (25 mL/day) | (1) compared to (2): ↓ Total bacteria (1) compared to (3), and (2) to (3): ↔ ↔ Firmicutes/Bacteroidetes ratios ↔ IgA-coated bacteria or IgA (feces) Within groups: (1) ↓ Total bacteria (2) ↑ Ig-A coated bacteria | ↔ TNFα (feces), IL-6 (feces) ↔ calprotectin (feces) ↑ CRP in (2) compared to (1) and (3) |
| Balfego M et al.; Lipids Health Dis, 2016, Spain | n = 35, M/F, T2D, C: 61.2 year, BMI 28.8, Sardine: 60.0 yrear, BMI 30.5 | 6 months | (1) Standard diet (C) (2) Standard diet with 100 g sardines 5 days a week | ↔ Firmicutes, Bacteroidetes, Firmicutes/Bacteroidetes ratio, Bacteroides‒Prevotella group, E. rectale‒C. coccoides group, F. prausnitzii, E.coli Within groups: ↓ Firmicutes, ↑ E.coli in (1) and (2) ↓ Firmicutes/bacteroidetes ratio, ↑ Bacteroides‒Prevotella group in (2) | ↔ CRP, TNFα, IL-6, IL-10 Within groups: ↑ TNFα in group (1) |
| Diets | |||||
| Pataky Z et al.; Dig Dis Sci, 2016, Switzerland | n = 15, M/F, NAFLD, 50 year, BMI 34.6 | 3 weeks | (1) No control group (2) Eurodiet (4 products/d: breakfast, one meal, two snacks) (Hypocaloric hyperproteic diet) | ↔ Firmicutes, Bacteroidetes ↓ Lachnospira ↑ Blautia, Butyricicoccus | ↓ CRP ↔ IL-6, IL-10, TNFα |
| Marlow G et al.; Human Genomics, 2013, New Zealand | n = 8, M/F, Crohn’s disease, 45.4 year, BMI nd | 6 weeks | (1) No control group (2) Mediterranean-inspired anti-inflammatory diet | ↔ Enterobacteriaceae, Enterococcales, Streptococcaceae, Fusobacterium, Bacillaceae, Akkermansia, Actinobacteria, Lactobacillaceae, Clostridium clusters (XI, III, IX, IV, XIVa), Bacteroides/Prevotella | ↔ CRP |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Telle-Hansen, V.H.; Holven, K.B.; Ulven, S.M. Impact of a Healthy Dietary Pattern on Gut Microbiota and Systemic Inflammation in Humans. Nutrients 2018, 10, 1783. https://doi.org/10.3390/nu10111783
Telle-Hansen VH, Holven KB, Ulven SM. Impact of a Healthy Dietary Pattern on Gut Microbiota and Systemic Inflammation in Humans. Nutrients. 2018; 10(11):1783. https://doi.org/10.3390/nu10111783
Chicago/Turabian StyleTelle-Hansen, Vibeke H., Kirsten B. Holven, and Stine M. Ulven. 2018. "Impact of a Healthy Dietary Pattern on Gut Microbiota and Systemic Inflammation in Humans" Nutrients 10, no. 11: 1783. https://doi.org/10.3390/nu10111783
APA StyleTelle-Hansen, V. H., Holven, K. B., & Ulven, S. M. (2018). Impact of a Healthy Dietary Pattern on Gut Microbiota and Systemic Inflammation in Humans. Nutrients, 10(11), 1783. https://doi.org/10.3390/nu10111783






