Blackcurrant Supplementation Improves Trabecular Bone Mass in Young but Not Aged Mice
Abstract
1. Introduction
2. Methods
2.1. Animals and Diets
2.2. Bone Microcomputed Tomography (µCT) Analysis
2.3. Bone Histomorphometric Analysis
2.4. Dual-Energy X-Ray Absorptiometry (DXA) Analysis
2.5. Bone Homogenate Biomarkers
2.6. Statistical Analysis
3. Results
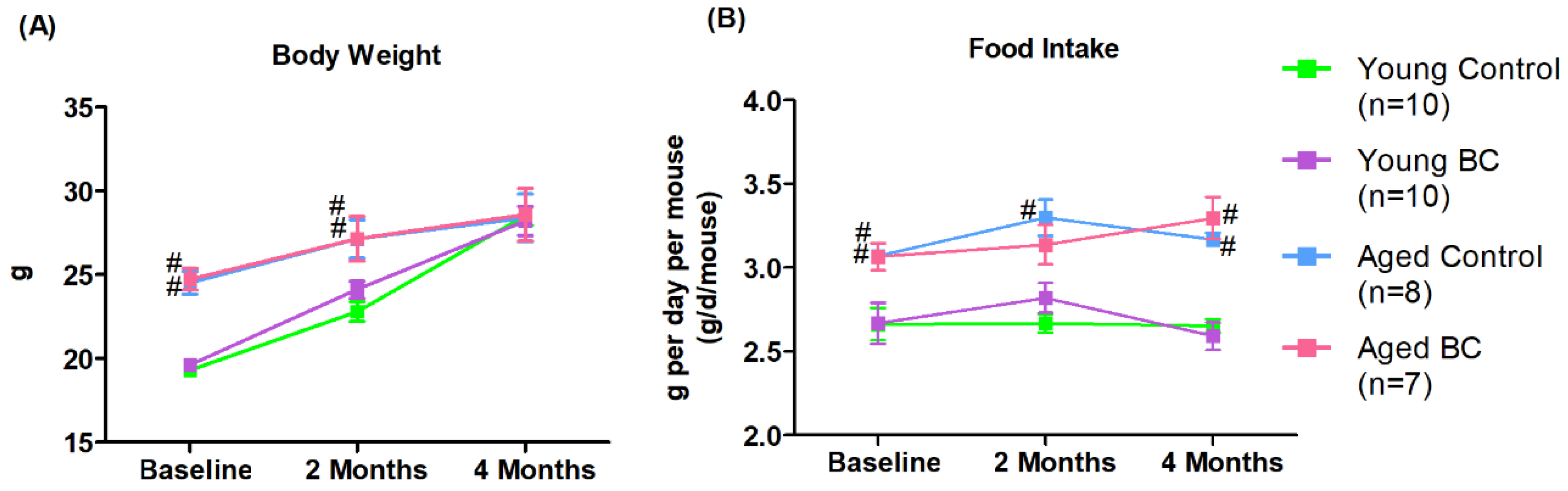
3.1. Body Weight and Food Intake
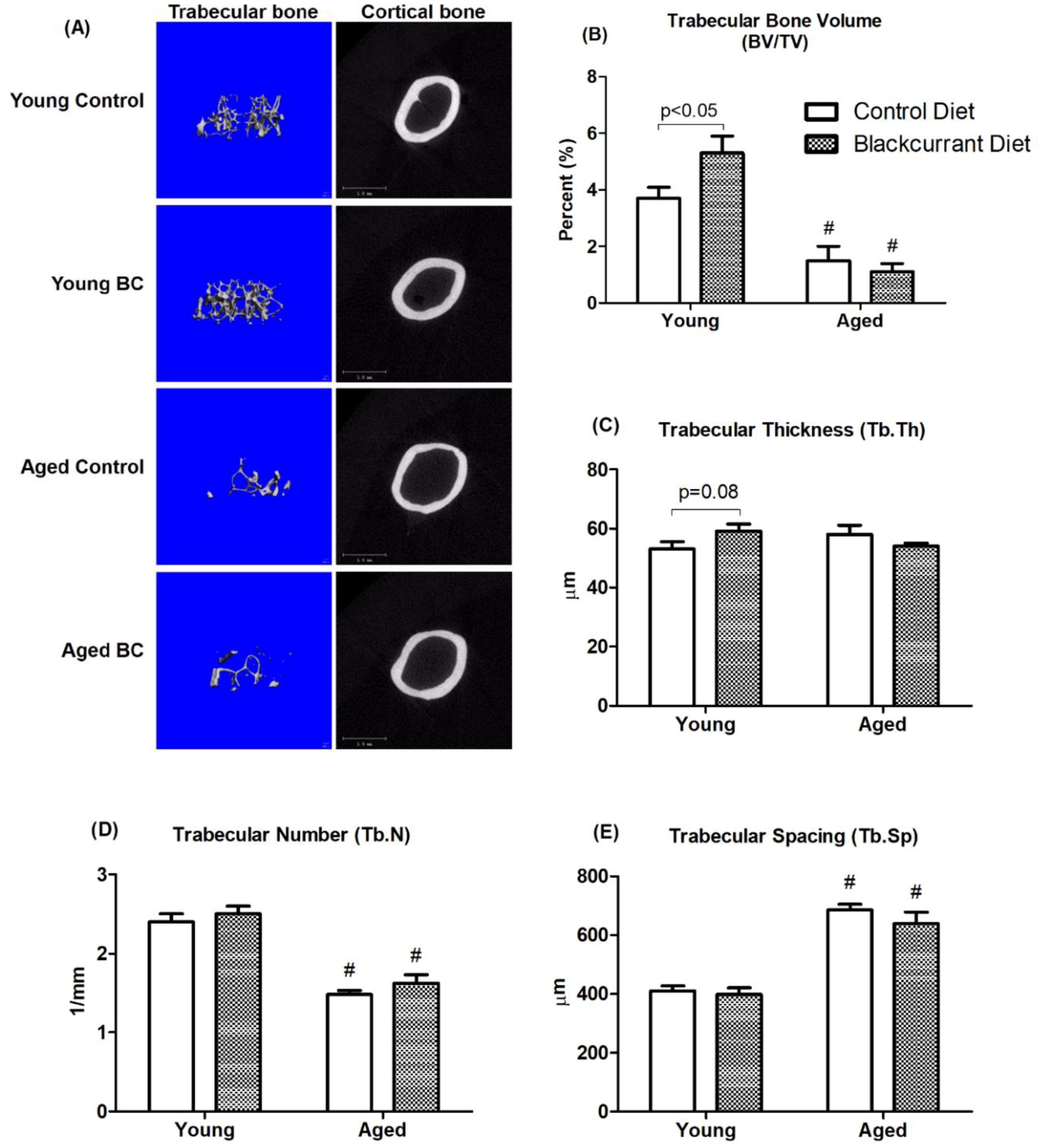
3.2. Bone µCT Analysis
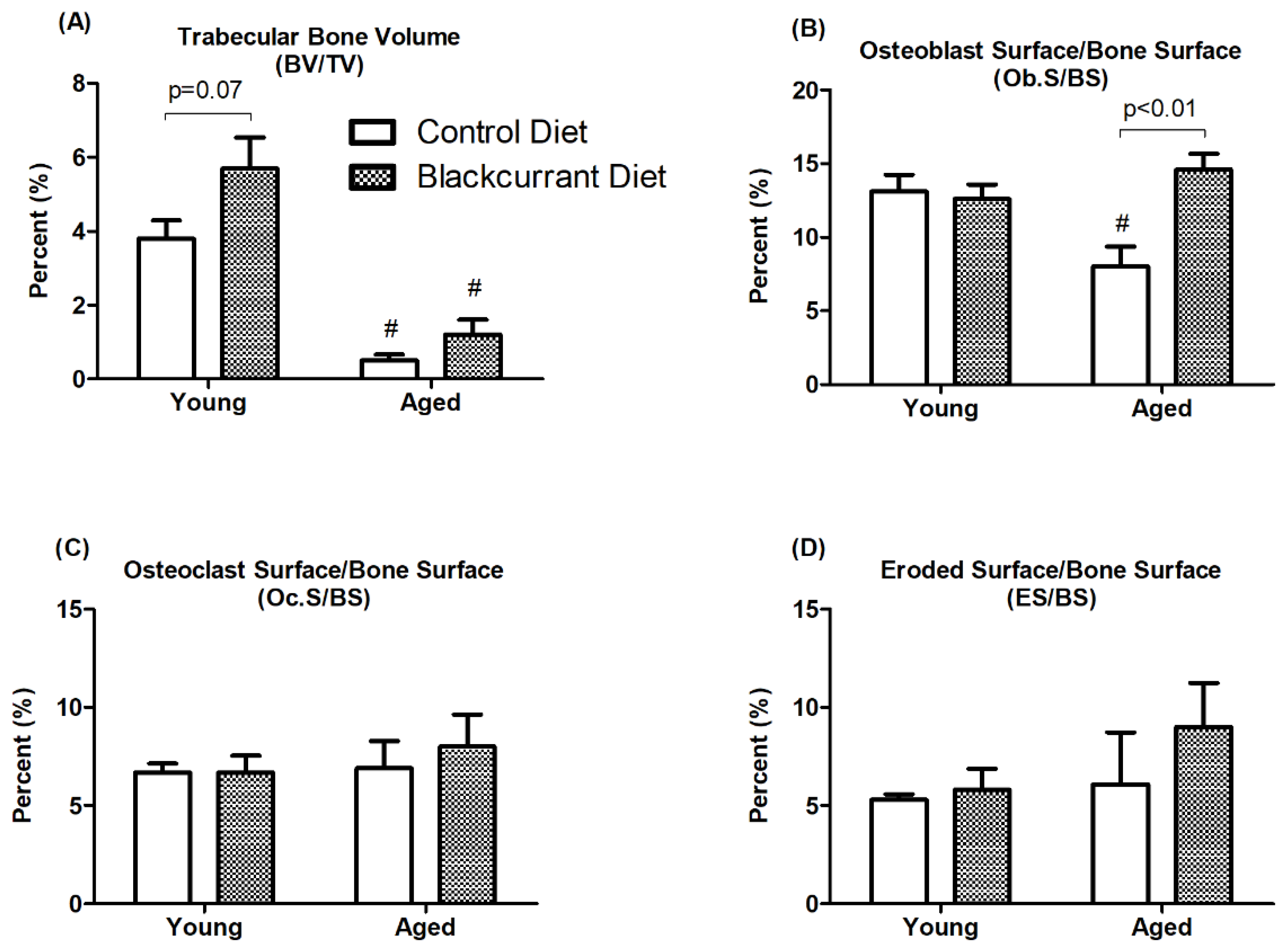
3.3. Bone Histomorphometric Analysis
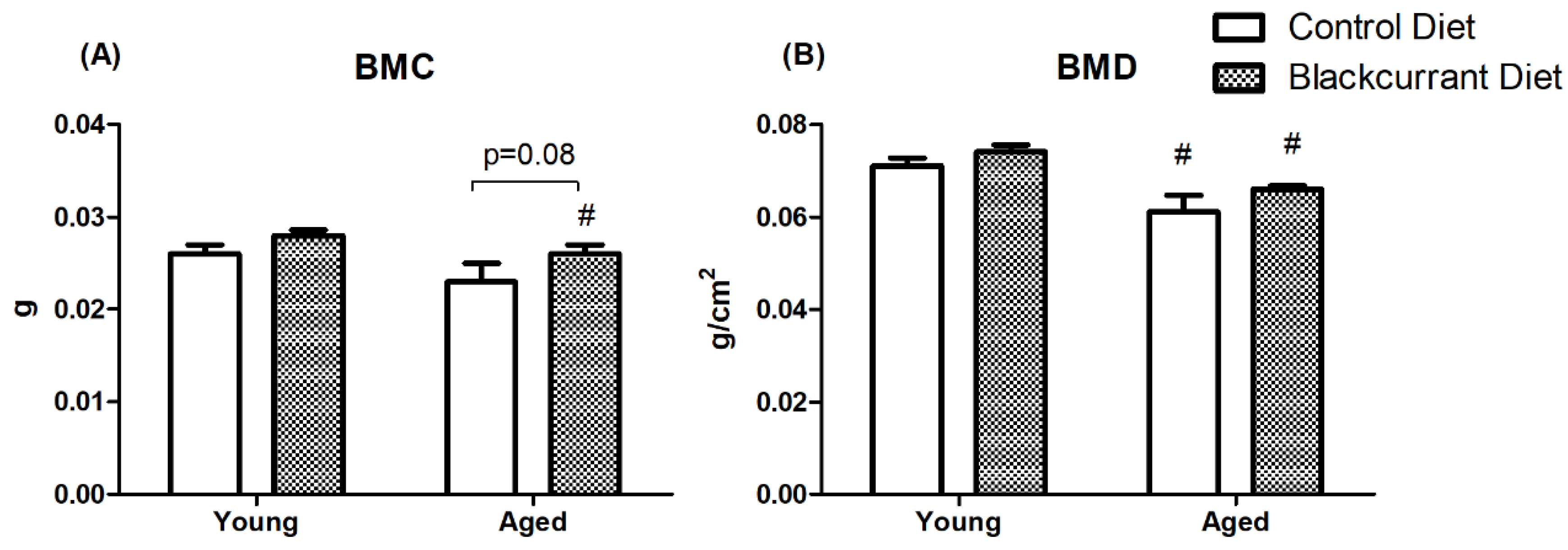
3.4. DXA Analysis
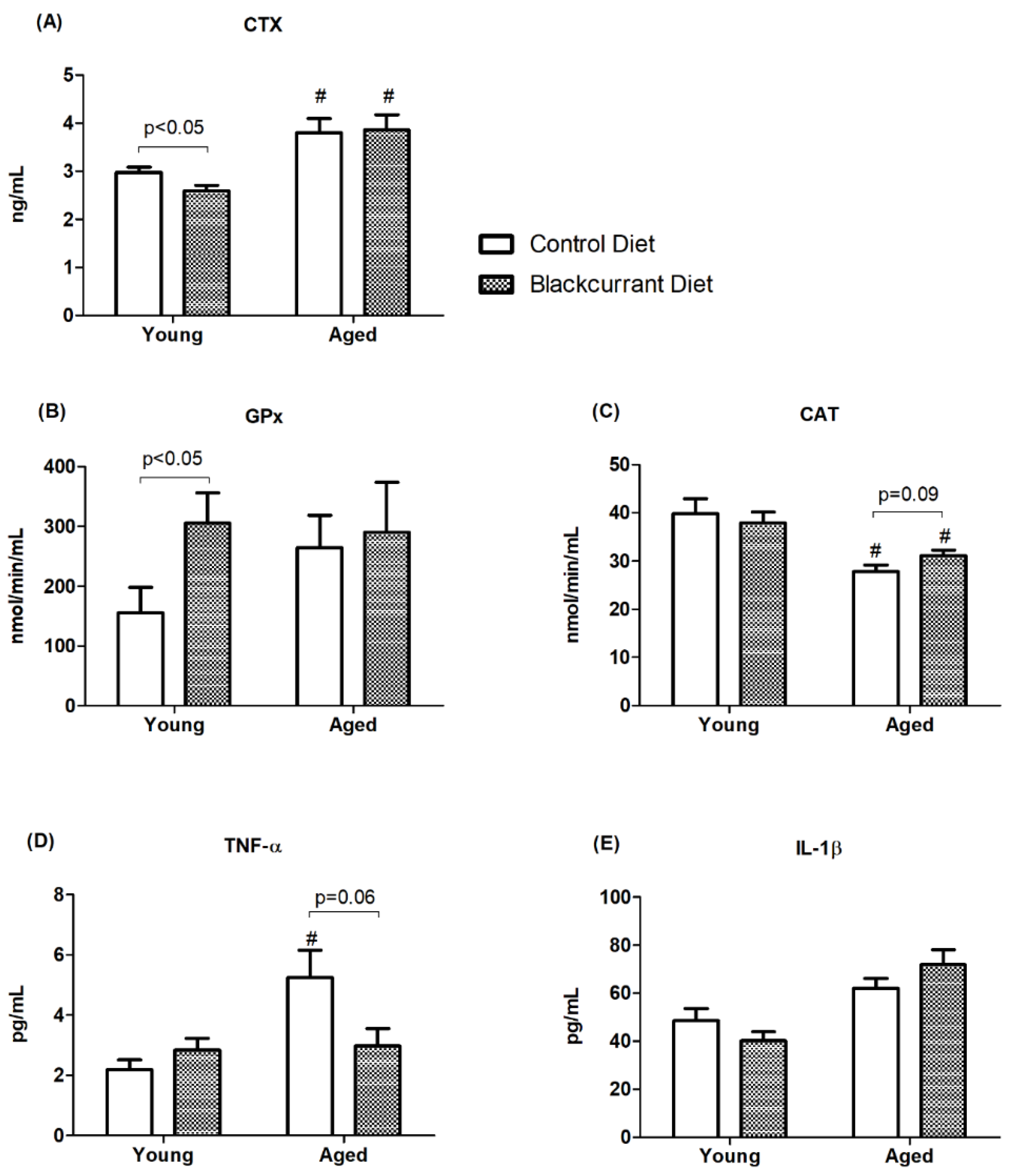
3.5. Humerus Homogenate Biomarkers
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Demontiero, O.; Vidal, C.; Duque, G. Aging and bone loss: New insights for the clinician. Ther. Adv. Musculoskelet. Dis. 2012, 4, 61–76. [Google Scholar] [CrossRef] [PubMed]
- Vedi, S.; Compston, J.; Webb, A.; Tighe, J. Histomorphometric analysis of dynamic parameters of trabecular bone formation in the iliac crest of normal British subjects. Metab. Bone Dis. Relat. Res. 1984, 5, 69–74. [Google Scholar] [CrossRef]
- Ericksen, E. Normal and pathological remodeling of human trabecular bone: Three dimensional reconstruction of the remodelling sequence in normals and in metabolic disease. Endoc. Rev. 1986, 4, 379–408. [Google Scholar] [CrossRef] [PubMed]
- Lips, P.; Courpron, P.; Meunier, P. Mean wall thickness of trabecular bone packets in the human iliac crest: Changes with age. Calcif. Tissue Res. 1978, 26, 13–17. [Google Scholar] [CrossRef] [PubMed]
- Bjørnerem, Å.; Ghasem-Zadeh, A.; Bui, M.; Wang, X.; Rantzau, C.; Nguyen, T.; Hopper, J.; Zebaze, R.; Seeman, E. Remodeling markers are associated with larger intracortical surface area but smaller trabecular surface area: A twin study. Bone 2011, 49, 1125–1130. [Google Scholar] [CrossRef] [PubMed]
- Mosekilde, L.; Mosekilde, L. Sex differences in age-related changes in vertebral body size, density and biomechanical competence in normal individuals. Bone 1990, 11, 67–73. [Google Scholar] [CrossRef]
- Zebaze, R.; Ghasem-Zadeh, A.; Bohte, A.; Iuliano-Burns, S.; Mirams, M.; Price, R.; Mackie, E.; Seeman, E. Intracortical remodelling and porosity in the distal radius and post-mortem femurs of women: A cross-sectional study. Lancet 2010, 375, 1729–1736. [Google Scholar] [CrossRef]
- Lacativa, P.G.S.; De Farias, M.L.F. Osteoporosis and inflammation. Arq. Bras. Endocrinol. Metab. 2010, 54, 123–132. [Google Scholar] [CrossRef]
- Shen, C.; Chyu, M.; Wang, J. Tea and bone health: Steps forward in translational nutrition. Am. J. Clin. Nutr. 2013, 98, 1694–1699. [Google Scholar] [CrossRef] [PubMed]
- Manolagas, S.C. From estrogen-centric to aging and oxidative stress: A revised perspective of the pathogenesis of osteoporosis. Endocr. Rev. 2010, 31, 266–300. [Google Scholar] [CrossRef] [PubMed]
- Qaseem, A.; Forciea, M.A.; McLean, R.M.; Denberg, T.D. Treatment of low bone density or osteoporosis to prevent fractures in men and women: A clinical practice guideline update from the American college of physicians. Ann. Intern. Med. 2017, 166, 818–839. [Google Scholar] [CrossRef] [PubMed]
- Rossini, M.; Adami, S.; Bertoldo, F.; Diacinti, D.; Gatti, D.; Giannini, S.; Giusti, A.; Malavolta, N.; Minisola, S.; Osella, G.; et al. Guidelines for the diagnosis, prevention and management of osteoporosis. Reumatismo 2016, 68, 1–39. [Google Scholar] [CrossRef] [PubMed]
- Kanis, J.A.; McCloskey, E.V.; Johansson, H.; Cooper, C.; Rizzoli, R.; Reginster, J.Y. European guidance for the diagnosis and management of osteoporosis in postmenopausal women. Osteoporos Int. 2013, 24, 23–57. [Google Scholar] [CrossRef] [PubMed]
- Akkawi, I.; Zmerly, H. Osteoporosis: Current Concepts. Joints 2018, 6, 122–127. [Google Scholar] [PubMed]
- Lee, S.G.; Vance, T.M.; Nam, T.G.; Kim, D.O.; Koo, S.I.; Chun, O.K. Contribution of Anthocyanin Composition to Total Antioxidant Capacity of Berries. Plant Foods Hum. Nutr. 2015, 70, 427–432. [Google Scholar] [CrossRef] [PubMed]
- Moriwaki, S.; Suzuki, K.; Muramatsu, M.; Nomura, A.; Inoue, F.; Into, T.; Yoshiko, Y.; Niida, S. Delphinidin, one of the major anthocyanidins, prevents bone loss through the inhibition of excessive osteoclastogenesis in osteoporosis model mice. PLoS ONE 2014, 9, e97177. [Google Scholar] [CrossRef] [PubMed]
- Shahnazari, M.; Turner, R.T.; Iwaniec, U.T.; Wronski, T.J.; Li, M.; Ferruzzi, M.G.; Nissenson, R.A.; Halloran, B.P. Dietary dried plum increases bone mass, suppresses proinflammatory cytokines and promotes attainment of peak bone mass in male mice. J. Nutr. Biochem. Elsevier Inc. 2016, 34, 73–82. [Google Scholar] [CrossRef] [PubMed]
- Zheng, X.; Mun, S.; Lee, S.G.; Vance, T.M.; Hubert, P.; Koo, S.I.; Lee, S.-K.; Chun, O.K. Anthocyanin-Rich Blackcurrant Extract Attenuates Ovariectomy-Induced Bone Loss in Mice. J. Med. Food 2016, 19, 390–397. [Google Scholar] [CrossRef] [PubMed]
- Kaume, L.; Gilbert, W.; Smith, B.J.; Devareddy, L. Cyanidin 3-O-β-d-Glucoside Improves Bone Indices. J. Med. Food 2015, 18, 690–697. [Google Scholar] [CrossRef] [PubMed]
- Lean, J.M.; Jagger, C.J.; Kirstein, B.; Fuller, K.; Chambers, T.J. Hydrogen peroxide is essential for estrogen-deficiency bone loss and osteoclast formation. Endocrinology 2005, 146, 728–735. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.G.; Kim, B.; Soung, D.Y.; Vance, T.; Lee, J.S.; Lee, J.; Koo, S.I.; Kim, D.-O.; Drissi, H.; Chun, O.K. Relationship Between Oxidative Stress and Bone Mass in Obesity and Effects of Berry Supplementation on Bone Remodeling in Obese Male Mice: An Exploratory Study. J. Med. Food 2015, 18, 476–482. [Google Scholar] [CrossRef] [PubMed]
- Benn, T.; Kim, B.; Park, Y.K.; Wegner, C.J.; Harness, E.; Nam, T.G.; Kim, D.O.; Lee, J.S.; Lee, J.Y. Polyphenol-rich blackcurrant extract prevents inflammation in diet-induced obese mice. J. Nutr. Biochem. 2014, 25, 1019–1025. [Google Scholar] [CrossRef] [PubMed]
- Halloran, B.P.; Wronski, T.J.; VonHerzen, D.C.; Chu, V.; Xia, X.; Pingel, J.E.; Williams, A.A.; Smith, B.J. Dietary Dried Plum Increases Bone Mass in Adult and Aged Male Mice. J. Nutr. 2010, 140, 1781–1787. [Google Scholar] [CrossRef] [PubMed]
- Smith, B.J.; Graef, J.L.; Wronski, T.J.; Rendina, E.; Williams, A.A.; Clark, K.A.; Clarke, S.L.; Lucas, E.A.; Halloran, B.P. Effects of dried plum supplementation on bone metabolism in adult C57BL/6 male mice. Calcif. Tissue Int. 2014, 94, 442–453. [Google Scholar] [CrossRef] [PubMed]
- Bouxsein, M.L.; Boyd, S.K.; Christiansen, B.A.; Guldberg, R.E.; Jepsen, K.J.; Müller, R. Guidelines for assessment of bone microstructure in rodents using micro-computed tomography. J. Bone Miner Res. 2010, 25, 1468–1486. [Google Scholar] [CrossRef] [PubMed]
- Parfitt, A.; Drezner, M.; Glorieux, F.; Kanis, J.; Malluche, H.; Meunier, P.; Ott, S.; Recker, R. Bone histomorphometry: Standardization of nomenclature, symbols, and units. Report of the ASBMR Histomorphometry Nomenclature Committee. J. Bone Miner Res. 1987, 2, 595–610. [Google Scholar] [CrossRef] [PubMed]
- Plotkin, L.I.; Laird, D.W.; Amedee, J. Role of connexins and pannexins during ontogeny, regeneration, and pathologies of bone. BMC Cell Biol. 2016, 17, 19. [Google Scholar] [CrossRef] [PubMed]
- Looker, A.C.; Wahner, H.W.; Dunn, W.L.; Calvo, M.S.; Harris, T.B.; Heyse, S.P.; Johnston, C.C.; Lindsay, R. Updated data on proximal femur bone mineral levels of US adults. Osteoporos Int. 1998, 8, 468–489. [Google Scholar] [CrossRef] [PubMed]
- Khosla, S.; Riggs, B.L. Pathophysiology of age-related bone loss and osteoporosis. Endocrinol. Metab. Clin. N. Am. 2005, 34, 1015–1030. [Google Scholar] [CrossRef] [PubMed]
- Syed, F.A.; Ng, A.C. The pathophysiology of the aging skeleton. Curr. Osteoporos Rep. 2010, 8, 235–240. [Google Scholar] [CrossRef] [PubMed]
- Baek, K.H.; Oh, K.W.; Lee, W.Y.; Lee, S.S.; Kim, M.K.; Kwon, H.S.; Rhee, E.J.; Han, J.H.; Song, K.H.; Cha, B.Y.; et al. Association of oxidative stress with postmenopausal osteoporosis and the effects of hydrogen peroxide on osteoclast formation in human bone marrow cell cultures. Calcif.Tissue Int. 2010, 87, 226–235. [Google Scholar] [CrossRef] [PubMed]
- Glatt, V.; Canalis, E.; Stadmeyer, L.; Bouxsein, M.L. Age-related changes in trabecular architecture differ in female and male C57BL/6J mice. J. Bone Miner Res. 2007, 22, 1197–1207. [Google Scholar] [CrossRef] [PubMed]
- Byers, R.; Denton, J.; Hoyland, J.; Freemont, A. Differential patterns of osteoblast dysfunction in trabecular bone in patients with established osteoporosis. J. Clin. Pathol. 1997, 50, 760–764. [Google Scholar] [CrossRef] [PubMed]
- Russell, S.J.; Kahn, C.R. Endocrine regulation of ageing. Nat. Rev. Mol. Cell Biol. 2007, 8, 681–691. [Google Scholar] [CrossRef] [PubMed]
- Shimizu, S.; Matsushita, H.; Morii, Y.; Ohyama, Y.; Morita, N.; Tachibana, R.; Watanabe, K.; Wakatsuki, A. Effect of anthocyanin-rich bilberry extract on bone metabolism in ovariectomized rats. Biomed. Rep. 2018, 8, 198–204. [Google Scholar] [CrossRef] [PubMed]
- Bai, X.C.; Lu, D.; Bai, J.; Zheng, H.; Ke, Z.Y.; Li, X.M.; Luo, S.Q. Oxidative stress inhibits osteoblastic differentiation of bone cells by ERK and NF-κB. Biochem. Biophys. Res. Commun. 2004, 314, 197–207. [Google Scholar] [CrossRef] [PubMed]
- Bax, B.E.; Alam, A.S.M.T.; Banerji, B.; Bax, C.M.R.; Bevis, P.J.R.; Stevens, C.R.; Moonga, B.S.; Blake, D.R.; Zaidi, M. Stimulation of osteoclastic bone resorption by hydrogen peroxide. Biochem. Biophys. Res. Commun. 1992, 183, 1153–1158. [Google Scholar] [CrossRef]
- Sanada, F.; Taniyama, Y.; Muratsu, J.; Otsu, R.; Shimizu, H.; Rakugi, H.; Morishita, R. Source of Chronic Inflammation in Aging. Front. Cardiovasc. Med. 2018, 5, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Mundy, G. Osteoporosis and inflammation. Nutr. Rev. 2007, 65, 147–151. [Google Scholar] [CrossRef]
- Weitzmann, M.N.; Pacifici, R. The role of T lymphocytes in bone metabolism. Immunol. Rev. 2005, 208, 154–168. [Google Scholar] [CrossRef] [PubMed]
- Teitelbaum, S.L. Osteoclasts: What do they do and how do they do it? Am. J. Pathol. 2007, 170, 427–435. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.G.; Kim, B.; Yang, Y.; Pham, T.X.; Park, Y.K.; Manatou, J.; Koo, S.I.; Chun, O.K.; Lee, J.Y. Berry anthocyanins suppress the expression and secretion of proinflammatory mediators in macrophages by inhibiting nuclear translocation of NF-κB independent of NRF2-mediated mechanism. J. Nutr. Biochem. 2014, 25, 404–411. [Google Scholar] [CrossRef] [PubMed]

© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sakaki, J.; Melough, M.; Lee, S.G.; Kalinowski, J.; Koo, S.I.; Lee, S.-K.; Chun, O.K. Blackcurrant Supplementation Improves Trabecular Bone Mass in Young but Not Aged Mice. Nutrients 2018, 10, 1671. https://doi.org/10.3390/nu10111671
Sakaki J, Melough M, Lee SG, Kalinowski J, Koo SI, Lee S-K, Chun OK. Blackcurrant Supplementation Improves Trabecular Bone Mass in Young but Not Aged Mice. Nutrients. 2018; 10(11):1671. https://doi.org/10.3390/nu10111671
Chicago/Turabian StyleSakaki, Junichi, Melissa Melough, Sang Gil Lee, Judy Kalinowski, Sung I. Koo, Sun-Kyeong Lee, and Ock K. Chun. 2018. "Blackcurrant Supplementation Improves Trabecular Bone Mass in Young but Not Aged Mice" Nutrients 10, no. 11: 1671. https://doi.org/10.3390/nu10111671
APA StyleSakaki, J., Melough, M., Lee, S. G., Kalinowski, J., Koo, S. I., Lee, S.-K., & Chun, O. K. (2018). Blackcurrant Supplementation Improves Trabecular Bone Mass in Young but Not Aged Mice. Nutrients, 10(11), 1671. https://doi.org/10.3390/nu10111671






