Mediterranean Lifestyle in Relation to Cognitive Health: Results from the HELIAD Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Setting and Study Population
2.2. Neuropshychological Evaluation and Clinical Diagnoses
2.3. Dietary Intake and Adherence to the Meditteranean Diet
2.4. Physical Activity
2.5. Sleep Quality
2.6. Instrumental Activities of Daily Living (IADL)
2.7. Total Lifestyle Index (TLI)
2.8. Statistical Analyses
3. Results
3.1. Total Sample Charachteristics
3.2. Excluding Participants with Dementia
3.3. Excluding Both Participants with Dementia and MCI
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- World Population Prospects: The 2017 Revision, Key Findings and Advance Tables. Available online: https://esa.un.org/unpd/wpp/publications/files/wpp2017_keyfindings.pdf (accessed on 11 June 2018).
- Prince, M.; Bryce, R.; Albanese, E.; Wimo, A.; Ribeiro, W.; Ferri, C.P. The global prevalence of dementia: A systematic review and metaanalysis. Alzheimers Dement. 2013, 9, 63–75. [Google Scholar] [CrossRef] [PubMed]
- Prince, M.; Ali, G.-C.; Guerchet, M.; Prina, A.M.; Albanese, E.; Wu, Y.-T. Recent global trends in the prevalence and incidence of dementia, and survival with dementia. Alzheimers Res. Ther. 2016, 8, 23. [Google Scholar] [CrossRef] [PubMed]
- Muir, S.W.; Gopaul, K.; Montero Odasso, M.M. The role of cognitive impairment in fall risk among older adults: A systematic review and meta-analysis. Age Ageing 2012, 41, 299–308. [Google Scholar] [CrossRef] [PubMed]
- Pusswald, G.; Tropper, E.; Kryspin-Exner, I.; Moser, D.; Klug, S.; Auff, E.; Dal-Bianco, P.; Lehrner, J. Health-related quality of life in patients with subjective cognitive decline and mild cognitive impairment and its relation to activities of daily living. J. Alzheimers Dis. 2015, 47, 479–486. [Google Scholar] [CrossRef] [PubMed]
- Loy, C.T.; Schofield, P.R.; Turner, A.M.; Kwok, J.B. Genetics of dementia. Lancet 2014, 383, 828–840. [Google Scholar] [CrossRef]
- Norton, S.; Matthews, F.E.; Barnes, D.E.; Yaffe, K.; Brayne, C. Potential for primary prevention of alzheimer’s disease: An analysis of population-based data. Lancet Neurol. 2014, 13, 788–794. [Google Scholar] [CrossRef]
- Singh, B.; Parsaik, A.K.; Mielke, M.M.; Erwin, P.J.; Knopman, D.S.; Petersen, R.C.; Roberts, R.O. Association of mediterranean diet with mild cognitive impairment and alzheimer’s disease: A systematic review and meta-analysis. J. Alzheimers Dis. 2014, 39, 271–282. [Google Scholar] [CrossRef] [PubMed]
- Psaltopoulou, T.; Sergentanis, T.N.; Panagiotakos, D.B.; Sergentanis, I.N.; Kosti, R.; Scarmeas, N. Mediterranean diet, stroke, cognitive impairment, and depression: A meta-analysis. Ann. Neurol. 2013, 74, 580–591. [Google Scholar] [CrossRef] [PubMed]
- Sofi, F.; Abbate, R.; Gensini, G.F.; Casini, A. Accruing evidence on benefits of adherence to the mediterranean diet on health: An updated systematic review and meta-analysis. Am. J. Clin. Nutr. 2010, 92, 1189–1196. [Google Scholar] [CrossRef] [PubMed]
- Yannakoulia, M.; Kontogianni, M.; Scarmeas, N. Cognitive health and mediterranean diet: Just diet or lifestyle pattern? Ageing Res. Rev. 2015, 20, 74–78. [Google Scholar] [CrossRef] [PubMed]
- Bach-Faig, A.; Berry, E.M.; Lairon, D.; Reguant, J.; Trichopoulou, A.; Dernini, S.; Medina, F.X.; Battino, M.; Belahsen, R.; Miranda, G.; et al. Mediterranean diet pyramid today. Science and cultural updates. Public Health Nutr. 2011, 14, 2274–2284. [Google Scholar] [CrossRef] [PubMed]
- Sofi, F.; Valecchi, D.; Bacci, D.; Abbate, R.; Gensini, G.F.; Casini, A.; Macchi, C. Physical activity and risk of cognitive decline: A meta-analysis of prospective studies. J. Intern. Med. 2011, 269, 107–117. [Google Scholar] [CrossRef] [PubMed]
- Laurin, D.; Verreault, R.; Lindsay, J.; MacPherson, K.; Rockwood, K. Physical activity and risk of cognitive impairment and dementia in elderly persons. Arch. Neurol. 2001, 58, 498–504. [Google Scholar] [CrossRef] [PubMed]
- Di Marco, L.Y.; Marzo, A.; Munoz-Ruiz, M.; Ikram, M.A.; Kivipelto, M.; Ruefenacht, D.; Venneri, A.; Soininen, H.; Wanke, I.; Ventikos, Y.A.; et al. Modifiable lifestyle factors in dementia: A systematic review of longitudinal observational cohort studies. J. Alzheimers Dis. 2014, 42, 119–135. [Google Scholar] [CrossRef] [PubMed]
- Fieo, R.; Zahodne, L.; Tang, M.X.; Manly, J.J.; Cohen, R.; Stern, Y. The historical progression from adl scrutiny to iadl to advanced adl: Assessing functional status in the earliest stages of dementia. J. Gerontol. A Biol. Sci. Med. Sci. 2017. [Google Scholar] [CrossRef] [PubMed]
- Fieo, R.; Manly, J.J.; Schupf, N.; Stern, Y. Functional status in the young-old: Establishing a working prototype of an extended-instrumental activities of daily living scale. J. Gerontol A Biol. Sci. Med. Sci. 2014, 69, 766–772. [Google Scholar] [CrossRef] [PubMed]
- Potvin, O.; Lorrain, D.; Forget, H.; Dubé, M.; Grenier, S.; Préville, M.; Hudon, C. Sleep quality and 1-year incident cognitive impairment in community-dwelling older adults. Sleep 2012, 35, 491–499. [Google Scholar] [CrossRef] [PubMed]
- Tsapanou, A.; Gu, Y.; Manly, J.; Schupf, N.; Tang, M.X.; Zimmerman, M.; Scarmeas, N.; Stern, Y. Daytime sleepiness and sleep inadequacy as risk factors for dementia. Dement. Geriatr. Cogn. Dis. Extra 2015, 5, 286–295. [Google Scholar] [CrossRef] [PubMed]
- Scarmeas, N.; Luchsinger, J.A.; Schupf, N.; Brickman, A.M.; Cosentino, S.; Tang, M.X.; Stern, Y. Physical activity, diet, and risk of alzheimer disease. Am. J. Geriatr. Psychiatry 2009, 302, 627–637. [Google Scholar] [CrossRef] [PubMed]
- Norton, M.C.; Dew, J.; Smith, H.; Fauth, E.; Piercy, K.W.; Breitner, J.C.; Tschanz, J.; Wengreen, H.; Welsh-Bohmer, K. Lifestyle behavior pattern is associated with different levels of risk for incident dementia and alzheimer’s disease: The cache county study. J. Am. Geriatr. Soc. 2012, 60, 405–412. [Google Scholar] [CrossRef] [PubMed]
- Weng, P.H.; Chen, J.H.; Chiou, J.M.; Tu, Y.K.; Chen, T.F.; Chiu, M.J.; Tang, S.C.; Yeh, S.J.; Chen, Y.C. The effect of lifestyle on late-life cognitive change under different socioeconomic status. PLoS ONE 2018, 13, e0197676. [Google Scholar] [CrossRef] [PubMed]
- Dardiotis, E.; Kosmidis, M.H.; Yannakoulia, M.; Hadjigeorgiou, G.M.; Scarmeas, N. The hellenic longitudinal investigation of aging and diet (heliad): Rationale, study design, and cohort description. Neuroepidemiology 2014, 43, 9–14. [Google Scholar] [CrossRef] [PubMed]
- Anastasiou, C.A.; Yannakoulia, M.; Kosmidis, M.H.; Dardiotis, E.; Hadjigeorgiou, G.M.; Sakka, P.; Arampatzi, X.; Bougea, A.; Labropoulos, I.; Scarmeas, N. Mediterranean diet and cognitive health: Initial results from the hellenic longitudinal investigation of ageing and diet. PLoS ONE 2017, 12, e0182048. [Google Scholar] [CrossRef] [PubMed]
- Kosmidis, M.H.; Vlachos, G.S.; Anastasiou, C.A.; Yannakoulia, M.; Dardiotis, E.; Hadjigeorgiou, G.; Sakka, P.; Ntanasi, E.; Scarmeas, N. Dementia prevalence in greece: The hellenic longitudinal investigation of aging and diet (heliad). Alzheimer Dis. Assoc. Disord. 2018, 32, 232–239. [Google Scholar] [CrossRef] [PubMed]
- Folstein, M.F.; Folstein, S.E.; McHugh, P.R. “Mini-mental state”. A practical method for grading the cognitive state of patients for the clinician. J. Psychiatr. Res. 1975, 12, 189–198. [Google Scholar] [CrossRef]
- Vlahou, C.H.; Kosmidis, M.H.; Dardagani, A.; Tsotsi, S.; Giannakou, M.; Giazkoulidou, A.; Zervoudakis, E.; Pontikakis, N. Development of the greek verbal learning test: Reliability, construct validity, and normative standards. Arch. Clin. Neuropsychol. 2013, 28, 52–64. [Google Scholar] [CrossRef] [PubMed]
- Lezak, M.D.; Howieson, D.B.; Loring, D.W. Neuropsychological Assessment; Oxford University Press: New York, NY, USA, 2004. [Google Scholar]
- Kosmidis, M.H.; Vlahou, C.H.; Panagiotaki, P.; Kiosseoglou, G. The verbal fluency task in the greek population: Normative data, and clustering and switching strategies. J. Int. Neuropsychol. Soc. 2004, 10, 164–172. [Google Scholar] [CrossRef] [PubMed]
- Tsapkini, K.; Vlahou, C.H.; Potagas, C. Adaptation and validation of standardized aphasia tests in different languages: Lessons from the boston diagnostic aphasia examination—Short form in Greek. Behav. Neurol. 2010, 22, 111–119. [Google Scholar] [CrossRef] [PubMed]
- Benton, A.L.; Sivan, A.B.; Hamsher, K.D.; Varney, N.R.; Spreen, O. Contributions to Neuropsychological Assessment: A Clinical Manual, 2nd ed.; Oxford University Press: New York, NY, USA, 1994. [Google Scholar]
- Kosmidis, M.H.; Tsotsi, S.; Karambela, O.; Takou, E.; Vlahou, C.H. Cultural factors influencing performance on visuoperceptual neuropsychological tasks. Behav. Neurol. 2010, 23, 245–247. [Google Scholar] [CrossRef] [PubMed]
- Bozikas, V.P.; Giazkoulidou, A.; Hatzigeorgiadou, M.; Karavatos, A.; Kosmidis, M.H. Do age and education contribute to performance on the clock drawing test? Normative data for the greek population. J. Clin. Exp. Neuropsychol. 2008, 30, 199–203. [Google Scholar] [CrossRef] [PubMed]
- Vlahou, C.H.; Kosmidis, M.H. The greek trail making test: Preliminary norms for clinical and research use. Psychology 2002, 9, 336–352. [Google Scholar]
- Giaglis, G.; Kyriazidou, S.; Paraskevopoulou, E.; Tascos, N.; Kosmidis, M.H. Evaluating premorbid level: Preliminary findings regarding the vulnerability of scores on cognitive measures in patients with MS (abstract). J. Int. Neuropsychol. Soc. 2010, 165 (Suppl. 1), 159. [Google Scholar]
- McKhann, G.; Drachman, D.; Folstein, M.; Katzman, R.; Price, D.; Stadlan, E.M. Clinical diagnosis of alzheimer’s disease: Report of the nincds-adrda work group under the auspices of department of health and human services task force on alzheimer’s disease. Neurology 1984, 34, 939–944. [Google Scholar] [CrossRef] [PubMed]
- Bountziouka, V.; Bathrellou, E.; Giotopoulou, A.; Katsagoni, C.; Bonou, M.; Vallianou, N.; Barbetseas, J.; Avgerinos, P.C.; Panagiotakos, D.B. Development, repeatability and validity regarding energy and macronutrient intake of a semi-quantitative food frequency questionnaire: Methodological considerations. Nutr. Metab. Cardiovasc. Dis. 2012, 22, 659–667. [Google Scholar] [CrossRef] [PubMed]
- Ministry of Health and Welfare. Dietary guidelines for adults in Greece. Arch. Hellenic Med. 1999, 16, 516–524. [Google Scholar]
- Panagiotakos, D.B.; Pitsavos, C.; Arvaniti, F.; Stefanadis, C. Adherence to the mediterranean food pattern predicts the prevalence of hypertension, hypercholesterolemia, diabetes and obesity, among healthy adults; the accuracy of the meddietscore. Prev. Med. 2007, 44, 335–340. [Google Scholar] [CrossRef] [PubMed]
- Kavouras, S.A.; Maraki, M.I.; Kollia, M.; Gioxari, A.; Jansen, L.T.; Sidossis, L.S. Development, reliability and validity of a physical activity questionnaire for estimating energy expenditure in greek adults. Sci. Sports 2016, 31, e47–e53. [Google Scholar] [CrossRef]
- Spritzer, K.H.; Hays, R. Mos Sleep Scale: A Manual for Use And Scoring, Version 1.0; RAND: Los Angeles, CA, USA, 2003. [Google Scholar]
- Hays, R.D.; Sherbourne, C.D.; Mazel, R.M. User’s Manual for The Medical Outcomed Study (Mos) Core Measures of Health-Related Quality of Life. 1995. Available online: https://www.rand.org/pubs/monograph_reports/MR162.html (accessed on 3 October 2017).
- Spencer, C.A.; Jamrozik, K.; Norman, P.E.; Lawrence-Brown, M. A simple lifestyle score predicts survival in healthy elderly men. Prev. Med. 2005, 40, 712–717. [Google Scholar] [CrossRef] [PubMed]
- Mawditt, C.; Sacker, A.; Britton, A.; Kelly, Y.; Cable, N. The clustering of health-related behaviours in a british population sample: Testing for cohort differences. Prev. Med. 2016, 88, 95–107. [Google Scholar] [CrossRef] [PubMed]
- Campanini, M.Z.; Guallar-Castillon, P.; Rodriguez-Artalejo, F.; Lopez-Garcia, E. Mediterranean diet and changes in sleep duration and indicators of sleep quality in older adults. Sleep 2017. [Google Scholar] [CrossRef] [PubMed]
- Mamalaki, E.; Anastasiou, C.A.; Ntanasi, E.; Tsapanou, A.; Kosmidis, M.H.; Dardiotis, E.; Hadjigeorgiou, G.M.; Sakka, P.; Scarmeas, N.; Yannakoulia, M. Associations between the mediterranean diet and sleep in older adults: Results from the hellenic longitudinal investigation of aging and diet study. Geriatr. Gerontol. Int. 2018. [Google Scholar] [CrossRef] [PubMed]
- Gaziano, T.A. Lifestyle and cardiovascular disease: More work to do. J. Am. Coll. Cardiol. 2017, 69, 1126–1128. [Google Scholar] [CrossRef] [PubMed]
- Andrieu, S.; Guyonnet, S.; Coley, N.; Cantet, C.; Bonnefoy, M.; Bordes, S.; Bories, L.; Cufi, M.N.; Dantoine, T.; Dartigues, J.F.; et al. Effect of long-term omega 3 polyunsaturated fatty acid supplementation with or without multidomain intervention on cognitive function in elderly adults with memory complaints (mapt): A randomised, placebo-controlled trial. Lancet Neurol. 2017, 16, 377–389. [Google Scholar] [CrossRef]
- Van Charante, E.P.M.; Eurelings, L.S.; van Dalen, J.W.; Ligthart, S.A.; van Bussel, E.F.; Hoevenaar-Blom, M.P.; Vermeulen, M.; van Gool, W.A. Effectiveness of a 6-year multidomain vascular care intervention to prevent dementia (prediva): A cluster-randomised controlled trial. Lancet 2016, 388, 797–805. [Google Scholar] [CrossRef]
- Ngandu, T.; Lehtisalo, J.; Solomon, A.; Levalahti, E.; Ahtiluoto, S.; Antikainen, R.; Backman, L.; Hanninen, T.; Jula, A.; Laatikainen, T.; et al. A 2 year multidomain intervention of diet, exercise, cognitive training, and vascular risk monitoring versus control to prevent cognitive decline in at-risk elderly people (finger): A randomised controlled trial. Lancet 2015, 385, 2255–2263. [Google Scholar] [CrossRef]
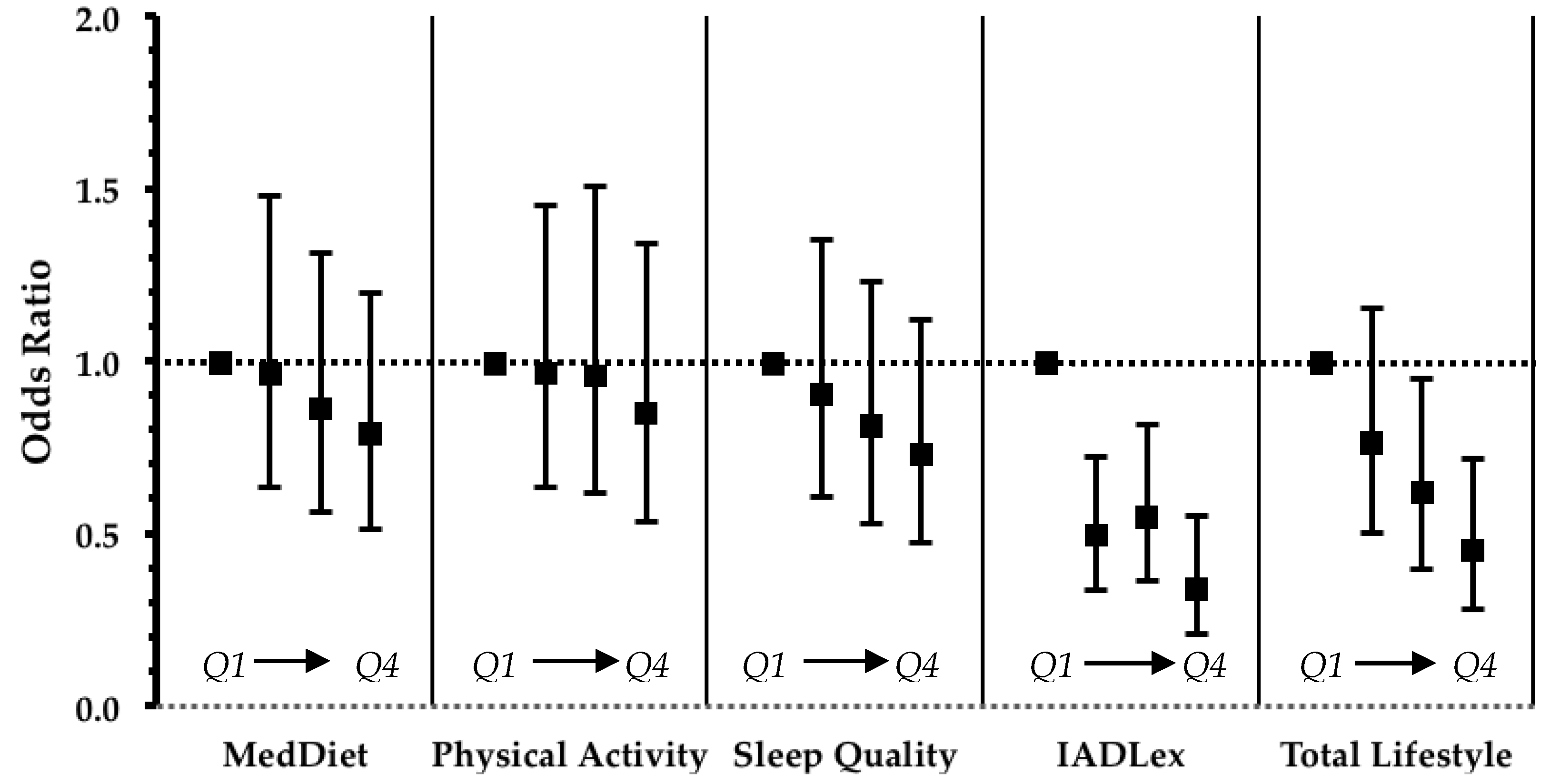

| All | Normal Cognition | MCI | Dementia | p1 | |
|---|---|---|---|---|---|
| n = 1716 | n = 1450 | n = 206 | n = 60 | ||
| Age, years | 72.9 ± 6.1 | 72.4 ± 6.0 | 74.8 ± 5.5 * | 77.8 ± 5.4 *, # | <0.001 |
| Sex, % male | 40.4% | 39.9% | 42.2% | 48.3% | 0.363 |
| Education, years | 7.7 ± 4.8 | 7.9 ± 4.7 | 6.9 ± 5.2 * | 5.8 ± 5.0 * | <0.001 |
| Illiteracy, % yes | 5.1% | 3.9% | 10.2% | 15.2% | <0.001 |
| MMSE score Literate, range 0–30 Illiterate, range 0–23 | 26.8 ± 3.1 18.9 ± 3.7 | 27.4 ± 2.3 20.4 ± 2.0 | 24. 5± 3.3 * 18.6 ± 2.3 * | 17.9 ± 6.2 *, # 12.0 ± 4.2 *, # | <0.001 <0.001 |
| MedDiet Score, range 0–55 | 33.0 ± 4.5 | 33.2 ± 4.5 | 32.6 ± 4.4 | 31.4 ± 4.8 * | 0.003 |
| Physical Activity, per 200 MET·min/day | 7.4 ± 1.4 | 7.5 ± 1.4 | 7.3 ± 1.4 | 6.8 ± 1.3 * | <0.001 |
| Sleep Quality, range 1–54 | 37.3 ± 7.8 | 37.4 ± 7.7 | 36.4.6 ± 7.6 | 36.1 ± 9.0 | 0.117 |
| IADLex, range 0–9 | 4.5 ± 1.3 | 4.6 ± 1.2 | 4.1 ± 1.2 * | 2.6 ± 1.6 *, # | <0.001 |
| Total Lifestyle Index, range 0–12 | 6.1 ± 2.4 | 6.3 ± 2.3 | 5.4 ± 2.4 * | 4.2 ± 2.3 *, # | <0.001 |
| Cognitive Domains | Beta | p | R2 |
|---|---|---|---|
| Memory | 0.098 | <0.001 | 0.301 |
| Executive | 0.071 | <0.001 | 0.388 |
| Visual-Spatial | 0.068 | 0.003 | 0.255 |
| Language | 0.072 | <0.001 | 0.478 |
| Attention-Speed | 0.039 | 0.084 | 0.287 |
| Composite | 0.085 | <0.001 | 0.477 |
| Cognitive Domains | MedDiet Score | Physical Activity | Sleep Quality | IADLex | R2 | ||||
|---|---|---|---|---|---|---|---|---|---|
| Beta | p | Beta | p | Beta | p | Beta | p | ||
| Memory | 0.059 | 0.007 | 0.057 | 0.012 | 0.037 | 0.091 | 0.037 | 0.090 | 0.304 |
| Executive | 0.016 | 0.437 | 0.044 | 0.037 | −0.002 | 0.931 | 0.085 | <0.001 | 0.391 |
| Visual-Spatial | 0.056 | 0.013 | 0.037 | 0.121 | 0.054 | 0.016 | −0.021 | 0.350 | 0.257 |
| Language | 0.044 | 0.019 | 0.016 | 0.416 | 0.016 | 0.405 | 0.052 | 0.006 | 0.478 |
| Attention- Speed | −0.021 | 0.354 | 0.066 | 0.007 | 0.001 | 0.951 | 0.041 | 0.074 | 0.290 |
| Composite | 0.039 | 0.039 | 0.056 | 0.004 | 0.023 | 0.210 | 0.051 | 0.007 | 0.478 |
| Total Lifestyle Index (Continuous) | Total Lifestyle Index (in Quartiles) | ||||
|---|---|---|---|---|---|
| OR [95% CI] | p | OR [95% CI] | p | p-Trend | |
| Unadjusted | 0.883 [0.818–0.952] | 0.001 | Q1: 1 (reference) | <0.001 | |
| Q2: 0.695 [0.460–1.052] | 0.085 | ||||
| Q3: 0.519 [0.339–0.794] | 0.003 | ||||
| Q4: 0.342 [0.216–0.540] | <0.001 | ||||
| Adjusted 1 | 0.925 [0.855–1.001] | 0.054 | Q1: 1 (reference) | 0.001 | |
| Q2: 0.757 [0.497–1.152] | 0.193 | ||||
| Q3: 0.614 [0.397–0.950] | 0.028 | ||||
| Q4: 0.446 [0.277–0.717] | 0.001 | ||||
| Q1 | Q2 | Q3 | Q4 | p1 | |
|---|---|---|---|---|---|
| Age, years | 76.4 ± 5.3 | 73.1 ± 5.1 | 71.4 ± 5.4 | 69.7 ± 6.2 | <0.001 |
| Sex, % male | 40.4% | 39.9% | 42.2% | 48.3% | 0.363 |
| Education, years | 4.3 ± 2.9 | 6.0 ± 2.9 | 8.8 ± 4.0 | 12.4± 4.2 | <0.001 |
| MedDiet Score, range 0–55 | 32.2 ± 4.4 | 33.1 ± 4.4 | 33.3 ± 4.5 | 34.1 ± 4.5 | <0.001 |
| Physical Activity, per 200 MET·min/day | 7.3 ± 1.4 | 7.5 ± 1.5 | 7.5 ± 1.4 | 7.6 ± 1.3 | 0.033 |
| Sleep Quality, range 1–54 | 36.2 ± 8.0 | 36.7 ± 7.9 | 38.8 ± 6.9 | 38.0 ± 7.8 | <0.001 |
| IADLex, range 0–9 | 4.4 ± 1.2 | 4.5 ± 1.1 | 4.7 ± 1.2 | 4.9 ± 1.4 | <0.001 |
| Total Lifestyle index, range 0–12 | 6.2 ± 2.1 | 6.7 ± 2.0 | 7.0 ± 2.2 | 7.5 ± 2.1 | <0.001 |
| Total Lifestyle Index (Continuous) | Total Lifestyle Index (in Quartiles) | ||||
|---|---|---|---|---|---|
| OR [95% CI] | p | OR [95% CI] | p | p-Trend | |
| Unadjusted | 0.824 [0.768–0.883] | <0.001 | Q1: 1 (reference) | <0.001 | |
| Q2: 0.545 [0.358–0.829] | 0.005 | ||||
| Q3: 0.567 [0.354–0.910] | 0.019 | ||||
| Q4: 0.324 [0.214–0.492] | <0.001 | ||||
| Adjusted 1 | 0.920 [0.845–1.003] | 0.057 | Q1: 1 (reference) | 0.076 | |
| Q2: 0.653 [0.389–1.095] | 0.106 | ||||
| Q3: 0.801 [0.450–1.424] | 0.450 | ||||
| Q4: 0.567 [0.339–0.947] | 0.030 | ||||
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Anastasiou, C.A.; Yannakoulia, M.; Kontogianni, M.D.; Kosmidis, M.H.; Mamalaki, E.; Dardiotis, E.; Hadjigeorgiou, G.; Sakka, P.; Tsapanou, A.; Lykou, A.; et al. Mediterranean Lifestyle in Relation to Cognitive Health: Results from the HELIAD Study. Nutrients 2018, 10, 1557. https://doi.org/10.3390/nu10101557
Anastasiou CA, Yannakoulia M, Kontogianni MD, Kosmidis MH, Mamalaki E, Dardiotis E, Hadjigeorgiou G, Sakka P, Tsapanou A, Lykou A, et al. Mediterranean Lifestyle in Relation to Cognitive Health: Results from the HELIAD Study. Nutrients. 2018; 10(10):1557. https://doi.org/10.3390/nu10101557
Chicago/Turabian StyleAnastasiou, Costas A., Mary Yannakoulia, Meropi D. Kontogianni, Mary H. Kosmidis, Eirini Mamalaki, Efthimios Dardiotis, Giorgos Hadjigeorgiou, Paraskevi Sakka, Angeliki Tsapanou, Anastasia Lykou, and et al. 2018. "Mediterranean Lifestyle in Relation to Cognitive Health: Results from the HELIAD Study" Nutrients 10, no. 10: 1557. https://doi.org/10.3390/nu10101557
APA StyleAnastasiou, C. A., Yannakoulia, M., Kontogianni, M. D., Kosmidis, M. H., Mamalaki, E., Dardiotis, E., Hadjigeorgiou, G., Sakka, P., Tsapanou, A., Lykou, A., & Scarmeas, N. (2018). Mediterranean Lifestyle in Relation to Cognitive Health: Results from the HELIAD Study. Nutrients, 10(10), 1557. https://doi.org/10.3390/nu10101557






