Non-Pharmacological Interventions to Reduce Unhealthy Eating and Risky Drinking in Young Adults Aged 18–25 Years: A Systematic Review and Meta-Analysis
Abstract
:1. Introduction
2. Materials and Methods
2.1. Eligibility Criteria
2.2. Search Strategy
2.3. Study Selection and Data Extraction
2.4. Data Synthesis
2.5. Risk of Bias and Appraisal of Methodological Quality
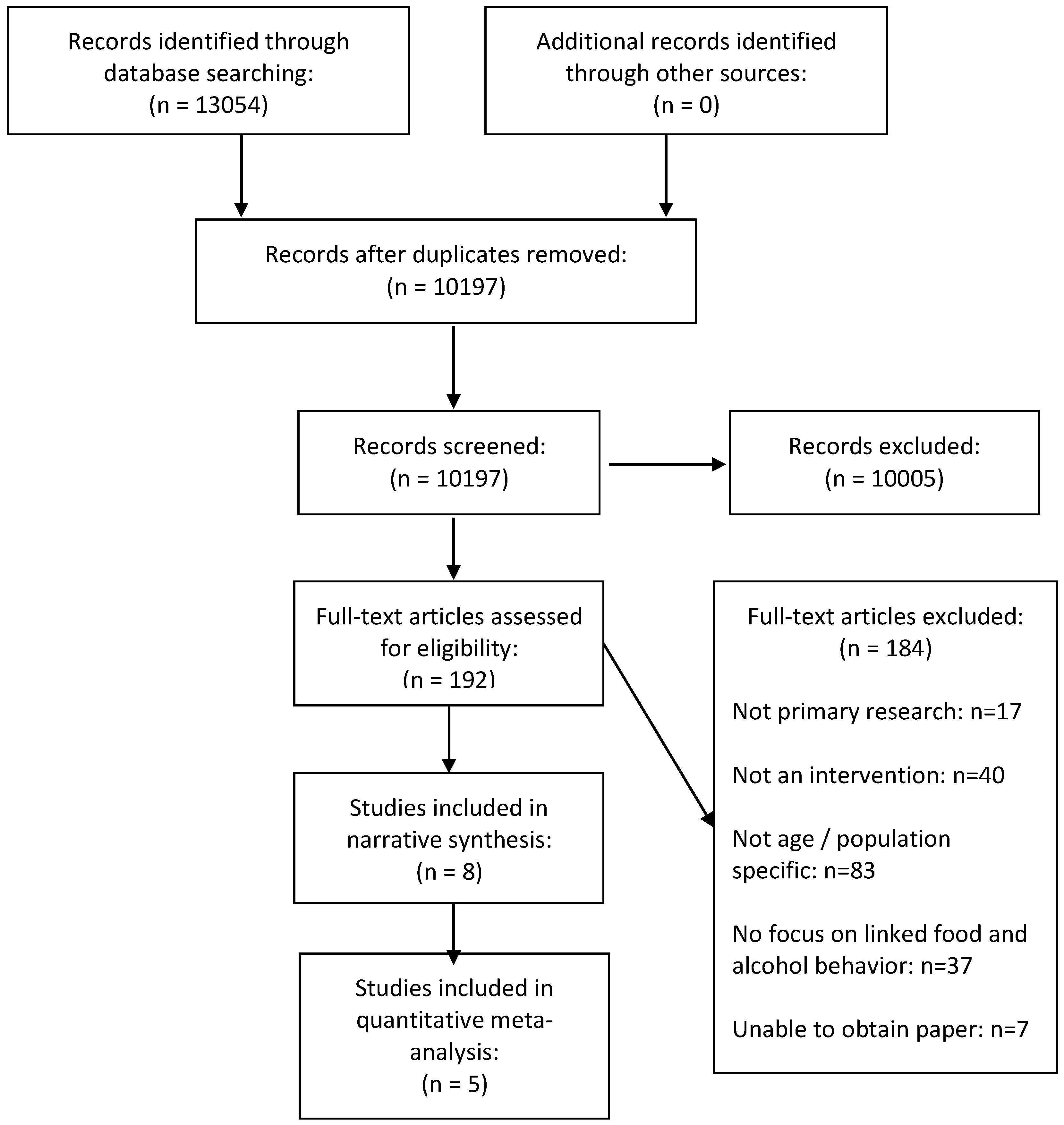
3. Results
3.1. Description of Included Studies
3.2. Critical Appraisal
3.3. Effects of Interventions
3.3.1. Interventions Providing Linked Feedback
3.3.2. Interventions Targeting Alcohol and Diet Separately
3.4. Physiological Outcomes
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- World Health Organisation. Global Status Report on Alcohol and Health. 2014. Available online: http://www.who.int/substance_abuse/activities/gsrah/en/ (accessed on 1 September 2018).
- World Health Organisation. Global Health Risks: Mortality and Burden of Disease Attributable to Selected Major Risks. 2009. Available online: http://Apps.who.int/iris/handle/10665/44203 (accessed on 1 September 2018).
- Hart, C.L.; Morrison, D.S.; Batty, G.D.; Mitchell, R.J.; Davey Smith, G. Effect of body mass index and alcohol consumption on liver disease: Analysis of data from two prospective cohort studies. Br. Med. J. 2010, 340. [Google Scholar] [CrossRef] [PubMed]
- Pinto Pereira, S.M.; van Veldhoven, K.; Li, L.; Power, C. Combined early and adult life risk factor associations for mid-life obesity in a prospective birth cohort: Assessing potential public health impact. BMJ Open 2016, 6, e011044. [Google Scholar] [CrossRef] [PubMed]
- Mokdad, A.H.; Forouzanfar, M.H.; Daoud, F.; Mokdad, A.A.; El Bcheraoui, C.; Moradi-Lakeh, M.; Kyu, H.H.; Barber, R.M.; Wagner, J.; Cercy, K.; et al. Global burden of diseases, injuries, and risk factors for young people’s health during 1990–2013: A systematic analysis for the global burden of disease study 2013. Lancet 2016, 387, 2383–2401. [Google Scholar] [CrossRef]
- Michels Blanck, H.; Gillespie, C.; Kimmons, J.E.; Seymour, J.D.; Serdula, M.K. Trends in fruit and vegetable consumption among U.S. men and women, 1994–2005. Prev. Chronic Dis. 2008, 5, A35. [Google Scholar]
- Hebden, L.; Chan, H.N.; Louie, J.C.; Rangan, A.; Allman-Farinelli, M. You are what you choose to eat: Factors influencing young adults’ food selection behaviour. J. Hum. Nutr. Diet. 2015, 28, 401–408. [Google Scholar] [CrossRef] [PubMed]
- Wymond, B.; Dickinson, K.; Riley, M. Alcoholic beverage intake throughout the week and contribution to dietary energy intake in Australian adults. Public Health Nutr. 2016, 19, 2592–2602. [Google Scholar] [CrossRef] [PubMed]
- Fazzino, T.L.; Fleming, K.; Sher, K.J.; Sullivan, D.K.; Befort, C. Heavy drinking in young adulthood increases risk of transitioning to obesity. Am. J. Prev. Med. 2017, 53, 169–175. [Google Scholar] [CrossRef] [PubMed]
- Eisenberg, M.H.; Fitz, C.C. “Drunkorexia”: Exploring the who and why of a disturbing trend in college students’ eating and drinking behaviors. J. Am. Coll. Health 2014, 62, 570–577. [Google Scholar] [CrossRef] [PubMed]
- Knight, A.; Castelnuovo, G.; Pietrabissa, G.; Manzoni, G.M.; Simpson, S. Drunkorexia: An empirical investigation among Australian female university students. Aust. Psychol. 2017, 52, 414–423. [Google Scholar] [CrossRef]
- Wilkerson, A.H.; Hackman, C.L.; Rush, S.E.; Usdan, S.L.; Smith, C.S. “Drunkorexia”: Understanding eating and physical activity behaviors of weight conscious drinkers in a sample of college students. J. Am. Coll. Health 2017, 65, 492–501. [Google Scholar] [CrossRef] [PubMed]
- Lupi, M.; Martinotti, G.; Di Giannantonio, M. Drunkorexia: An emerging trend in young adults. Eat. Weight Disord. 2017, 22, 619–622. [Google Scholar] [CrossRef] [PubMed]
- Patte, K.A.; Laxer, R.E.; Qian, W.; Leatherdale, S.T. An analysis of weight perception and physical activity and dietary behaviours among youth in the compass study. SSM Popul. Health 2016, 2, 841–849. [Google Scholar] [CrossRef] [PubMed]
- Barry, A.E.; Piazza-Gardner, A.K.; Holton, M.K. Assessing the alcohol-bmi relationship in a us national sample of college students. Health Educ. J. 2015, 74, 496–504. [Google Scholar] [CrossRef]
- Schlissel, A.C.; Skeer, M.R. Trying to lose weight and alcohol misuse among high school girls: Findings from the U.S. National 2011 youth risk behavior survey. Subst. Use Misuse 2015, 50, 1599–1605. [Google Scholar] [CrossRef] [PubMed]
- Yeomans, M.R. Short term effects of alcohol on appetite in humans. Effects of context and restrained eating. Appetite 2010, 55, 565–573. [Google Scholar] [PubMed]
- Sayon-Orea, C.; Martinez-Gonzalez, M.A.; Bes-Rastrollo, M. Alcohol consumption and body weight: A systematic review. Nutr. Rev. 2011, 69, 419–431. [Google Scholar] [CrossRef] [PubMed]
- Gray, C.M.; Wyke, S.; Hunt, K.; Mutrie, N.; Anderson, A.S.; Treweek, S. Can a gender-sensitised weight management programme delivered by Scottish premier league football clubs help men lose weight? A feasibility study for a randomised controlled trial. J. Epidemiol. Community Health 2011, 65, A37–A38. [Google Scholar] [CrossRef]
- Crombie, I.K.; Irvine, L.; Falconer, D.W.; Williams, B.; Ricketts, I.W.; Jones, C.; Humphris, G.; Norrie, J.; Slane, P.; Rice, P. Alcohol and disadvantaged men: A feasibility trial of an intervention delivered by mobile phone. Drug Alcohol Rev. 2017, 36, 468–476. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Anderson, A.S.; Craigie, A.M.; Caswell, S.; Treweek, S.; Stead, M.; Macleod, M.; Daly, F.; Belch, J.; Rodger, J.; Kirk, A.; et al. The impact of a bodyweight and physical activity intervention (bewel) initiated through a national colorectal cancer screening programme: Randomised controlled trial. Br. Med. J. 2014, 348. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- MacArthur, G.; Kipping, R.; White, J.; Chittleborough, C.; Lingam, R.; Pasch, K.; Gunnell, D.; Hickman, M.; Campbell, R. Individual-, family-, and school-led interventions for preventing multiple risk behaviours in individuals aged 8 to 25 years. Cochrane Database Syst. Rev. 2012. [Google Scholar] [CrossRef]
- Scott, S.; Parkinson, K.; Kaner, E.; Robalino, S.; Stead, M.; Power, C.; Fitzgerald, N.; Wrieden, W.; Adamson, A. Non-pharmacological interventions designed to reduce health risks due to unhealthy eating behaviour and linked risky or excessive drinking in adults aged 18–25 years: A systematic review protocol. Syst. Rev. 2017, 6, 42. [Google Scholar] [CrossRef] [PubMed]
- Moher, D.; Shamseer, L.; Clarke, M.; Ghersi, D.; Liberati, A.; Petticrew, M.; Shekelle, P.; Stewart, L.A.; PRISMA-P Group. Preferred reporting items for systematic review and meta-analysis protocols (prisma-p) 2015 statement. Syst. Rev. 2015, 4. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G. Preferred reporting items for systematic reviews and meta-analyses: The prisma statement. PLoS Med. 2009, 6. [Google Scholar] [CrossRef] [PubMed]
- Stockings, E.; Hall, W.D.; Lynskey, M.; Morley, K.I.; Reavley, N.; Strang, J.; Patton, G.; Degenhardt, L. Prevention, early intervention, harm reduction, and treatment of substance use in young people. Lancet Psychiatry 2016, 3, 280–296. [Google Scholar] [CrossRef]
- Winett, R.A. A framework for health promotion and disease prevention programs. Am. Psychol. 1995, 50, 341–350. [Google Scholar] [CrossRef] [PubMed]
- Scott, S.; Kaner, E. Brief alcohol interventions. In Alcohol Abuse and Liver Disease; Neuberger, J., DiMartini, A., Eds.; John Wiley & Sons: Oxford, UK, 2015. [Google Scholar]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.; PRISMA Group. Preferred reporting items for systematic reviews and meta-analyses: The prisma statement. Ann. Intern. Med. 2009, 151, 264–269. [Google Scholar] [CrossRef] [PubMed]
- Higgins, J.P.T.; Altman, D.G.; Sterne, J.A.C. Chapter 8: Assessing Risk of Bias in Included Studies. In Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 (Updated March 2011); Higgins, J.P.T., Green, S., Eds.; The Cochrane Collaboration: London, UK, 2011; Available online: http://handbook.cochrane.org (accessed on 1 September 2018).
- Thomas, H. Quality Assessment Tool for Quantitative Studies; McMaster University: Hamilton, ON, Canada, 2003. [Google Scholar]
- Epton, T.; Norman, P.; Dadzie, A.-S.; Harris, P.R.; Webb, T.L.; Sheeran, P.; Julious, S.A.; Ciravegna, F.; Brennan, A.; Meier, P.S.; et al. A theory-based online health behaviour intervention for new university students (u@uni): Results from a randomised controlled trial. BMC Public Health 2014, 14, 563. [Google Scholar] [CrossRef] [PubMed]
- Cameron, D.; Epton, T.; Norman, P.; Sheeran, P.; Harris, P.R.; Webb, T.L.; Julious, S.A.; Brennan, A.; Thomas, C.; Petroczi, A.; et al. A theory-based online health behaviour intervention for new university students (u@uni:Lifeguide): Results from a repeat randomized controlled trial. Trials 2015, 16, 555. [Google Scholar] [CrossRef] [PubMed]
- Kypri, K.; McAnally, H.M. Randomized controlled trial of a web-based primary care intervention for multiple health risk behaviors. Prev. Med. 2005, 41, 761–766. [Google Scholar] [CrossRef] [PubMed]
- Quartiroli, A.; Zizzi, S. A tailored wellness intervention for college students using internet-based technology: A pilot study. Int. Electron. J. Health Educ. 2012, 15, 37–50. [Google Scholar]
- Werch, C.E.; Bian, H.; Moore, M.J.; Ames, S.; DiClemente, C.C.; Weiler, R.M. Brief multiple behavior interventions in a college student health care clinic. J. Adolescent Health 2007, 41, 577–585. [Google Scholar] [CrossRef] [PubMed]
- Werch, C.E.; Moore, M.J.; Bian, H.; DiClemente, C.C.; Ames, S.C.; Weiler, R.M.; Thombs, D.; Pokorny, S.B.; Huang, I.C. Efficacy of a brief image-based multiple-behavior intervention for college students. Ann. Behav. Med. 2008, 36, 149–157. [Google Scholar] [CrossRef] [PubMed]
- Ashton, L.M.; Morgan, P.J.; Hutchesson, M.J.; Rollo, M.E.; Collins, C.E. Feasibility and preliminary efficacy of the ‘HEYMAN’ healthy lifestyle program for young men: A pilot randomised controlled trial. Nutr. J. 2017, 16, 2. [Google Scholar] [CrossRef] [PubMed]
- Leiva, O.A.M.; Martínez, S.M.A.; Celis-Morales, C. Efecto de una intervención centrada en la reducción de factores de riesgo cardiovascular en estudiantes universitarios. Rev. Med. Chil. 2015, 143, 971–978. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rehm, J.; Anderson, P.; Prieto, J.A.A.; Armstrong, I.; Aubin, H.-J.; Bachmann, M.; Bastus, N.B.; Brotons, C.; Burton, R.; Cardoso, M.; et al. Towards new recommendations to reduce the burden of alcohol-induced hypertension in the european union. BMC Med. 2017, 15, 173. [Google Scholar] [CrossRef] [PubMed]
- Kraus, L.; Schulte, B.; Manthey, J.; Rehm, J. Alcohol screening and alcohol interventions among patients with hypertension in primary health care: An empirical survey of german general practitioners. Addict. Res. Theor. 2017, 25, 285–292. [Google Scholar] [CrossRef]
- McCambridge, J.; Saitz, R. Rethinking brief interventions for alcohol in general practice. Br. Med. J. 2017, 356. [Google Scholar] [CrossRef] [PubMed]
- Davis, C.G.; Thake, J.; Vilhena, N. Social desirability biases in self-reported alcohol consumption and harms. Add. Behav. 2010, 35, 302–311. [Google Scholar] [CrossRef] [PubMed]
- Gemming, L.; Jiang, Y.; Swinburn, B.; Utter, J.; Mhurchu, C.N. Under-reporting remains a key limitation of self-reported dietary intake: An analysis of the 2008/09 new zealand adult nutrition survey. Eur. J. Clin. Nutr. 2013, 68, 259–264. [Google Scholar] [CrossRef] [PubMed]
- Subar, A.F.; Freedman, L.S.; Tooze, J.A.; Kirkpatrick, S.I.; Boushey, C.; Neuhouser, M.L.; Thompson, F.E.; Potischman, N.; Guenther, P.M.; Tarasuk, V.; et al. Addressing current criticism regarding the value of self-report dietary data. J. Nutr. 2015, 145, 2639–2645. [Google Scholar] [CrossRef] [PubMed]
- Ashton, L.M.; Morgan, P.J.; Hutchesson, M.J.; Rollo, M.E.; Young, M.D.; Collins, C.E. A systematic review of snapo (smoking, nutrition, alcohol, physical activity and obesity) randomized controlled trials in young adult men. Prev. Med. 2015, 81, 221–231. [Google Scholar] [CrossRef] [PubMed]
- Fang, M.L.; Gerbrandt, J.; Liwander, A.; Pederson, A. Exploring promising gender-sensitive tobacco and alcohol use interventions: Results of a scoping review. Subst. Use Misuse 2014, 49, 1400–1416. [Google Scholar] [CrossRef] [PubMed]
- Fitzgerald, N.; Angus, K.; Emslie, C.; Shipton, D.; Bauld, L. Gender differences in the impact of population-level alcohol policy interventions: Evidence synthesis of systematic reviews. Addiction 2016, 111, 1735–1747. [Google Scholar] [CrossRef] [PubMed]
- Dennison, L.; Morrison, L.; Conway, G.; Yardley, L. Opportunities and challenges for smartphone applications in supporting health behavior change: Qualitative study. J. Med. Internet Res. 2013, 15, e86. [Google Scholar] [CrossRef] [PubMed]
- Lally, P.; Gardner, B. Promoting habit formation. Health Psychol. Rev. 2013, 7, S137–S158. [Google Scholar] [CrossRef]
- Lindsay, J. Young Australians and the staging of intoxication and self-control. J. Youth Stud. 2009, 12, 371–384. [Google Scholar] [CrossRef]
- Harrison, L.; Kelly, P.; Lindsay, J.; Advocat, J.; Hickey, C. ‘I don’t know anyone that has two drinks a day’: Young people, alcohol and the government of pleasure. Health Risk Soc. 2011, 13, 469–486. [Google Scholar] [CrossRef]
- Advocat, J.; Lindsay, J. To drink or not to drink? Young Australians negotiating the social imperative to drink to intoxication. J. Sociol. 2013, 51, 139–153. [Google Scholar] [CrossRef]
- Dempster, S. I drink, therefore i’m man: Gender discourses, alcohol and the construction of British undergraduate masculinities. Gender Educ. 2011, 23, 635–653. [Google Scholar] [CrossRef]
- Mullen, K.; Watson, J.; Swift, J.; Black, D. Young men, masculinity and alcohol. Drugs Educ. Prev. Policy 2007, 14, 151–165. [Google Scholar] [CrossRef]
- Robertson, A.; Mullan, B.; Todd, J. A qualitative exploration of experiences of overweight young and older adults. An application of the integrated behaviour model. Appetite 2014, 75, 157–164. [Google Scholar] [CrossRef] [PubMed]
- Ashton, L.M.; Hutchesson, M.J.; Rollo, M.E.; Morgan, P.J.; Thompson, D.I.; Collins, C.E. Young adult males’ motivators and perceived barriers towards eating healthily and being active: A qualitative study. Int. J. Behav. Nutr. Phys. Act. 2015, 12, 93. [Google Scholar] [CrossRef] [PubMed]
- National Institute for Clinical and Health Excellence. Alcohol Use Disorders: Preventing the Development of Hazardous and Harmful Drinking; Nice Public Health Guidance 24; National Institute for Health and Clinical Excellence: London, UK, 2010. [Google Scholar]
- Medical Research Council. Developing and Evaluating Complex Interventions; Medical Research Council: London, UK, 2008. [Google Scholar]
| Reference. Country | Setting | Study Design | RoB/Quality Assessment | Follow Up | Study Aim | Sample Characteristics | Inclusion/Exclusion Criteria | Outcome Measures: | Statistical Methods Used |
|---|---|---|---|---|---|---|---|---|---|
| Ashton et al. (2017) Australia | Region/community wide | Two-arm pilot RCT with waitlist control | Low Risk | 3-months post-intervention | To evaluate the feasibility of a targeted healthy lifestyle program for young adult men aged 18–25 years; to estimate the treatment effect of HEYMAN on improving objective physical activity levels (steps/day), diet quality and subjective well-being and other lifestyle, psychological, anthropometric and physiological measures. |
| Inclusion: male; aged 18–25; available for assessment sessions; access to a computer, tablet or smartphone with e-mail and Internet facilities. Exclusion: self-reported meeting national recommendations for F&V intakes and/or physical activity; currently participating in an alternative healthy lifestyle program; history of major medical problems (such as heart disease or diabetes that requires insulin injections) and not granted GP approval to participate; reported psychological distress and no GP approval (or associated expert) to participate; diagnosed with an eating disorder; non-English speaking; disability that precluded participation. | Primary: physical activity (pedometer steps/day); diet quality; subjective wellbeing and mental health. Secondary: AUDIT-C; BMI; waist circumference; energy intake (KJ/day); daily servings of fruit and vegetables; proportion of energy from alcohol; proportion of ED-NP foods; MVPA minutes/week. Biomarkers: Fasting Total cholesterol, HDL-Cholesterol, LDL-Cholesterol and Triglycerides (composite measures); Systolic and diastolic blood pressure (composite measures), resting heart rate and augmentation index; salivary cortisol. | Independent t-tests and chi-squared (χ2) tests; generalized linear mixed models for intention-to-treat (ITT) populations. Differences of means and 95% confidence intervals (CIs) determined using the mixed models. All health outcomes were included in the model, the predictors included time (treated as categorical with levels baseline and 3 months), treatment group (intervention and control), and an interaction term for time by treatment group. Models adjusted for baseline values of BMI, pedometer steps and proportion of energy from energy-dense, nutrient-poor foods. |
| Epton et al. (2014) Cameron et al. (2015) UK | University | Two-arm RCT followed by a two-arm Repeat RCT | Low risk | 6-months post-intervention | To assess the efficacy and cost-effectiveness of a theory-based online health behaviour intervention targeting health behaviours in new university students (fruit and vegetable intake, physical activity, alcohol consumption and smoking status), in comparison to a measurement-only control. |
| Inclusion: Incoming first year undergraduates Exclusion: NR | Primary: Portions of fruit and vegetables per day; physical activity in the last week; alcohol consumption in the last week (units/week; binge/week); AUDIT; smoking status at 6-month follow up. Secondary: health status; recreational drug use; BMI; health service usage; academic performance; social cognitive variables. Biomarkers: hair sample (3 cm long) liquefied and analysed for biochemical markers of various health behaviours related to alcohol consumption, cigarette smoking, and recreational drug use. | A series of ANCOVAs and logistic regression analyses were used to assess the impact of the intervention on performance of the targeted behaviours at 6-month follow-up, controlling for corresponding baseline scores, sex, age and nationality (i.e., UK or non-UK). For primary outcomes, the Bonferroni correction was used to account for multiple tests. Statistical significance was declared if any of the primary endpoints were significant at 0.0127. |
| Kypri and McAnally (2005) New Zealand | Student Health Service | Three-arm parallel group RCT | Unclear risk | 6-weeks post-intervention | To examine the efficacy of a brief web-based intervention for multiple risk behaviors in a primary care setting for young people. |
| Inclusion: NR Exclusion: NR | Daily fruit and vegetable consumption; alcohol consumption (age at first drink, alcohol consumption in the past year, largest amount consumed in the last 4 weeks, AUDIT); smoking, physical activity, mental health. * Primary outcome(s) not defined. | Dichotomous variables analysed using Pearson’s Chi-squared test with one degree of freedom for the following pairwise comparisons: A vs. C, A vs. B, and B vs. C (see Supplementary Table S2). Mean peak EBACs and 95% confidence intervals computed for each experimental group; mean differences analysed using analysis of variance. |
| Leiva et al. (2015) Chile | University | Pre/post | Weak | Immediately post-intervention | To evaluate the effect of a lifestyle-based intervention on reducing cardiovascular risk factors in university students. |
| Inclusion: Third year students. Exclusion: NR | BMI; physical activity; fruit and vegetable consumption; tobacco use; alcohol consumption. Biomarkers: glucose; total cholesterol (TC); triglyceride (TG); LDL cholesterol; HDL cholesterol; blood pressure. * Primary outcome(s) not defined. | Results presented as mean values with their respective standard deviation (continuous variables). To determine significance between pre and post intervention, t-test was applied for paired samples. For categorical variables, results were presented as prevalence. To determine significant changes in prevalence pre and post-intervention, X2 test was applied. |
| Quartiroli and Zizzi (2012) USA | University | Pseudo experimental (two-arm) | Weak | 8 weeks post-intervention | To pilot test a theory-based, computer-tailored feedback system for improvement of lifestyles among college students at a large, public university. |
| Inclusion: NR Exclusion: NR | Physical activity (days with moderate physical activity, days with stretching, days with strength activity); daily fruit and vegetable servings; alcohol use (days with at least one drink, number of drinks per day, days with 5+ drinks in a week, number of episodes with 5+ drinks in a month). * Primary outcome(s) not defined. | The impact of intervention was analysed by running a series of 2 (feedback type) × 3 (time) repeated measure ANOVAs, run for each of the dependent variables. In these analyses the independent variables were the assigned group (Normative vs. Personalized) and the time points during the intervention (T1, T2, T3). |
| Werch et al. (2008) USA | University | Two-arm RCT | High risk | 12 weeks post-intervention | To examine the efficacy of a brief, image-based Multiple Behaviour Intervention (MBI) compared against a standard care control for influencing risk behaviors (i.e., alcohol, cigarette, and marijuana consumption and problems) and health-promoting behaviors (i.e., exercise, nutrition, sleep, stress management) as well as health quality of life, among a sample of college students 3 months post-intervention. |
| Inclusion: Students aged 18–21 years currently enrolled at the target university and who visited the campus medical services center. Exclusion: NR | Alcohol, cigarette and marijuana consumption (initiation of use, 30-day frequency, 30-day quantity, 30-day heavy use); 18-item measure of alcohol and drug problems; physical activity (initiation of exercise, 30-day vigorous physical activity, 30-day moderate physical activity, 7-day strenuous exercise, 7-day moderate exercise); nutrition habits (past 30-day servings of fruit and vegetables, number of times eating healthy carbohydrates and fats); sleep habits; self-reported health status. * Primary outcome(s) not defined. | Baseline measures were compared across treatment group using chi-square tests for categorical variables and independent sample t-tests for continuous variables. Repeated measures MANOVAs and ANOVAs were used to test intervention effects over time. Repeated-measures MANOVAs were performed to more efficiently address the multiple health behaviours targeted by the intervention. Effect sizes were calculated based on mean pre–post change in the treatment group minus the mean pre–post change in the control group, divided by the pooled pre-test standard deviation. |
| Werch et al. (2007) USA | University | Three-arm RCT | High risk | 1-month post-intervention | To examine the effects of brief image-based interventions, including a multiple behavior health contract, a one-on-one tailored consultation, and a combined consultation plus contract intervention, for impacting multiple health behaviors of students in a university health clinic. |
| Inclusion: Students currently enrolled at the target university. Exclusion: NR | Alcohol, cigarette and marijuana consumption (length of use, 30-day frequency, 30-day quantity); physical activity (30-day vigorous physical activity, 30-day moderate physical activity, 7-day strenuous exercise, 7-day moderate exercise); nutrition habits (past 7-day servings of fruit and vegetables, number of times eating good carbohydrates and fats); sleep habits; self-reported health status. * Primary outcome(s) not defined. | Baseline measures were compared across treatment group using chi-square tests for categorical data and ANOVA tests for continuous scores. Both ANOVAs and repeated-measures MANOVAs were used to test intervention effects over time, first, on behaviour measures and, second, on image and belief measures. Repeated-measures MANOVAs were performed to more efficiently address the multiple health behaviours targeted by the intervention, and because the dependent variables were not perfectly correlated. |
| Reference: | Results: |
|---|---|
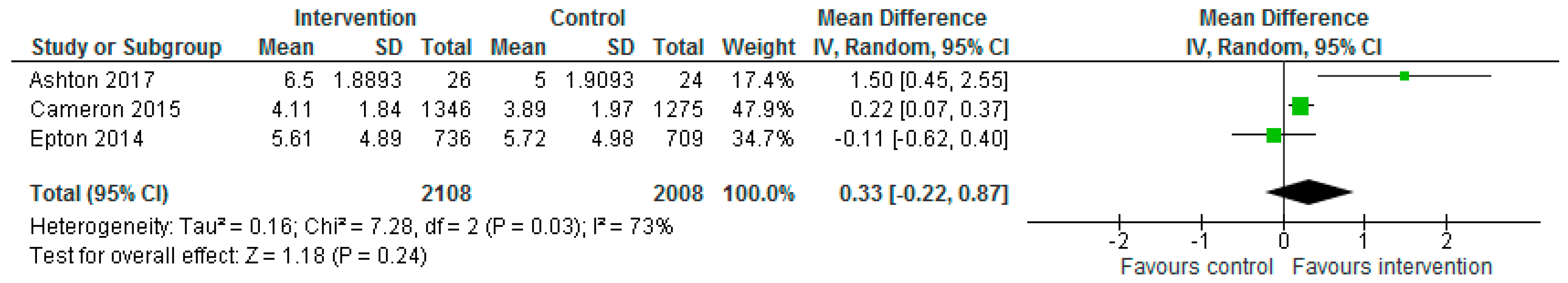
| Ashton et al. (2017) | No significant differences between groups observed for alcohol consumption (0.7, 95% CI = −0.3, 1.8, p = 0.181, d = 0.36) or diet quality score (3.6, 95% CI = −0.4, 7.6, p = 0.081, d = 0.48). Significant within-group differences evident in the intervention group for diet quality score (5.9 95% CI = 3.1, 8.7). Significant differences favouring the intervention group at 3-months were observed for daily vegetable servings (p < 0.05, d = 0.62), percentage energy from EDNP foods (p < 0.01, d = 0.73), weight (p < 0.05, d = 0.63), percentage weight loss (p < 0.05, d = 0.67), waist circumference (p < 0.001, d = 0.89), BMI (p < 0.01, d = 0.81), body fat mass (p < 0.05, d = 0.67), plasma total cholesterol (p < 0.05, d = 0.60), LDL cholesterol (p < 0.01, d = 0.83) and ratio of total cholesterol-to-HDL cholesterol (p < 0.05, d = 0.60). |
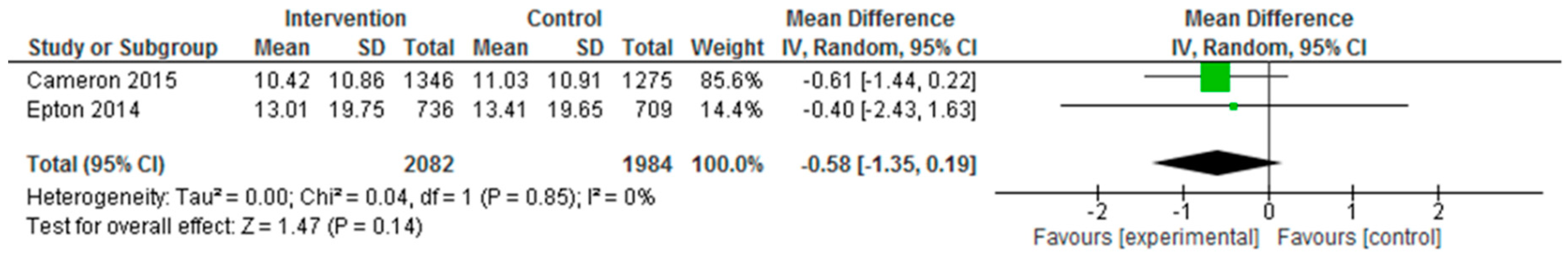
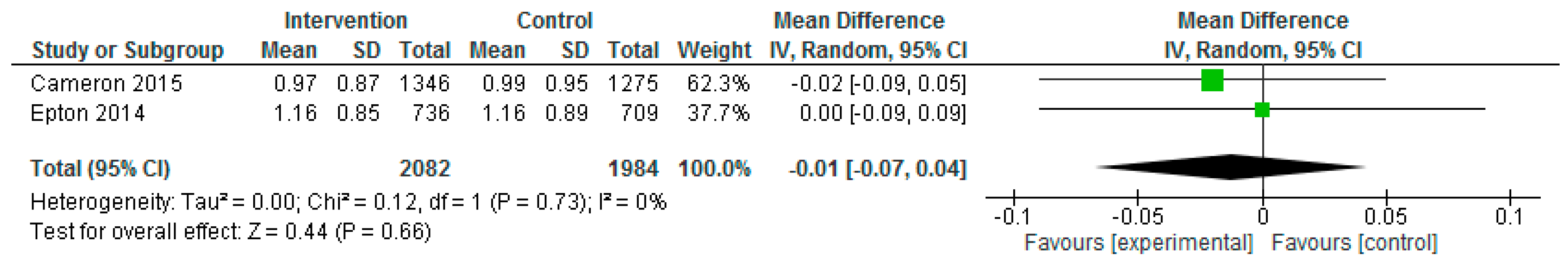
| Epton et al. (2014) Cameron et al. (2015) | Epton et al.: At 6-month follow-up, fruit and vegetable intake and alcohol consumption did not differ significantly between the two arms (F&V portions: Control: mean: 5.72, SD: 4.98; Intervention: mean: 5.61, SD: 4.89, p = 0.708, d = −0.02; mean alcohol units in past 7 days: control: 13.41, SD: 19.65, intervention: 13.01, SD: 19.75, p = 0.737, d = 0.02; mean no. binge drinking days in past 7 days: control: 1.16, SD: 0.89, intervention: 1.16, SD: 0.85, p = 0.973, d = 0.00). Cameron et al.: No significant differences between the intervention and control conditions on the primary outcomes at 6-month follow-up, although the effect of the intervention on fruit and vegetable intake approached significance (fruit and vegetable mean portions per day (control: 3.89, SD: 1.97; intervention: 4.11, SD: 1.84; p = 0.024); mean alcohol units in the past 7 days (control: 11.03, SD: 10.91; intervention: 10.42, SD: 10.86, p = 0.277). Repeating the primary analyses without data imputation produced consistent results. No significant differences between the intervention and control conditions at 6-month follow up in relation to no. days binge drinking in past 7 days (control: 0.99, SD: 0.95; intervention: 0.97, SD: 0.87, p = 0.674). Effect sizes found in the repeat trial were comparable to those found in the original trial for fruit and vegetable intake, Q(1) = 2.93, p = 0.087, and mean alcohol units in past 7 days, Q(1) = 0.25, p = 0.619. A marginally larger effect size was found for fruit and vegetable intake in the repeat trial (d = 0.12) than in the original trial (d = −0.02). Per-protocol analyses demonstrated intervention participants reported consuming significantly more portions of fruit and vegetables, F(1, 1068) = 7.19, p = 0.007, than those in the control condition (mean = 4.23, 3.89; SD = 0.11 and 0.07, respectively). Like the primary analyses, there was no significant effect of the intervention on alcohol units consumed, F(1, 1030) = 1.30, p = 0.254. The intervention had a significant effect on the biochemical marker of alcohol use (fatty acid ethyl esters) at 6-month follow-up, with lower levels of alcohol use observed among participants in the intervention versus control condition (control: 7.29, SD: 7.85; intervention: 5.00, SD: 4.33, p = 0.038). |
| Kypri and McAnally (2005) | Fruit and vegetable consumption: Group A had significantly greater compliance with recommendations than group C. Differences between A and B, and B vs. C were non-significant (p value: A vs. C: 0.02; A vs. B: 0.44; B vs. C:0.08). Alcohol consumption: None of the groups differed significantly in their compliance with recommended limits for episodic alcohol consumption (based on binge criteria) (p value: A vs. C: 0.84; A vs. B: 0.44; B vs. C: 0.70). The mean (95% confidence interval) peak EBACs in groups A, B, and C were 0.11 (0.08, 0.14), 0.12 (0.09, 0.15), and 0.13 (0.10, 0.15), F = 0.208, p = 0.813. |
| Leiva et al. (2015) | Significant reductions in the prevalence of hyperglycaemia (−10.0%; p =0.048) and high blood pressure (−16.7%; p = 0.0008) observed post-intervention. Reduction in prevalence of high blood pressure was particularly significant for women (−15.9%; p = 0.005). Significant reductions in body weight (pre: 67.7 ± 10.8; post: 65.6 ± 9.3; p ≤ 0.0001), body mass index (pre: 24.8 ± 3.9; post: 23.9 ± 3.3; p ≤ 0.0001), blood pressure (pre: 103.2 ± 11.2; post: 98.0 ± 8.3; p ≤ 0.0001), and an increase in HDL cholesterol (pre: 48.7 ± 11.5; post: 53.5 ± 10.4; p ≤ 0.0001) observed post-intervention. Waist circumference (pre: 78.2 ± 10.1; post: 79.2 ± 12.0; p = 0.0005), total cholesterol (pre: 206.1 ± 43.3; post: 185.8 ± 29.9; p ≤ 0.0001) and triglycerides (pre: 134.0 ± 69.9; post: 117.0 ± 41.2; p = 0.003) only decreased significantly in women. Other health markers such as baseline glycaemia, PAD and cholesterol LDL, were unchanged in the study population. Post-intervention, there was no change in the percentage of students who drank alcohol, but the amount of drinks they declared decreased to 38.4% drinkers at risk (from 50.0% pre-intervention; p = 0.201). Post-intervention, the daily consumption of fruits and vegetables increased to 55%, with 20% of students declaring that they consume 5+ servings daily, thus complying with the recommendations. However, these changes were not significant. |
| Quartiroli and Zizzi (2012) | Fruit and vegetable consumption: No significant main effects or interactions were found over time for fruit and vegetable intake (Time 1: mean servings: 7.40, SD: 3.396; Time 2: mean servings: 7.39, SD: 3.423; Time 3: mean servings: 6.98, SD: 3.257). Alcohol consumption: Days w/1 drink: Time 1: mean: 0.93, SD: 1.260; Time 2: mean: 1.08, SD: 1.297; Time 3: mean: 1.15, SD: 1.365. Drinks per day: Time 1: mean: 0.70, SD: 1.094; Time 2: mean: 0.93, SD: 1.436; Time 3: mean: 1.00, SD: 1.0547. Binge/Week: Time 1: mean: 0.60, SD: 1.045; Time 2: mean: 0.61, SD: 1.061; Time 3: mean: 0.69, SD: 1.288 Binge/Month: Time 1: mean: 0.83, SD: 1.044; Time 2: mean: 0.92, SD: 1.076; Time 3: mean: 0.76, SD: 0.862 Alcohol use over time: The interaction of the two variables (days w/1 drink and drinks per day) suggested a small, significant increase in drinks per day [F(2,120)=3.53, p = 0.03, ES = 0.058, Obs.Pow = 0.647]. Individuals in the normative feedback group showed a slightly larger change in their average of drinks per day than the personalized feedback group (Personalized −0.14; Normative 0.50). Change in drinking habits was also in the opposite direction for the individuals in the personalized feedback group. Overall, there was not a significant main effect for time (p = 0.238) or for group (p = 0.527). |
| Werch et al. (2008) | Post intervention, univariate tests for alcohol behaviours found that students exposed to the brief intervention drank alcohol less frequently (intervention: M=2.41, SE=0.12; control: M = 2.77, SE = 0.12; D = 0.27, p = 0.00), drank heavily less frequently (intervention: M = 1.74, SE = 0.10; control: M = 2.03, SE = 0.10; D = 0.29, p = 0.00) and drove after drinking less frequently (intervention: M = 0.50, SE = 0.11; control: M = 0.71, SE = 0.10; D = 0.23, p = 0.02). No omnibus treatment by time interactions were found for nutrition behaviours (fruits/vegetables: intervention: M = 4.31, SE = 0.17; control: M = 3.73, SE = 0.16, D = 0.20; good carbohydrate: intervention: M = 5.46, SE = 0.23; control: M = 4.76, SD = 0.23, D= 0.23; good fats: intervention: M = 4.34, SD = 0.21; control: M = 3.59, SE = 0.21, D = 0.22). Omnibus treatment by time multivariate analysis of variance interactions were significant for alcohol consumption behaviours groupings (F(6, 261) = 2.73, p = 0.01). Small positive effects were found for increases on all three nutrition behaviours (F = 1.33, df = 3, 279, p = 0.27), even though overall MANOVA tests were not significant. |
| Werch et al. (2007) | Omnibus repeated-measures MANOVAs were significant for drinking driving behaviours (F(2,136), 4.43, p = 0.01) and nutrition habits, inclusive of fruit and vegetable consumption (F(3,143), 5.37, p = 0.00), with improvements on each of these behaviours across time. No differences were seen over time on alcohol consumption measures (F(3,142), 0.48, p = 0.69); univariate analyses showed decreases in the frequency of riding with a drunk driver (F(1,145), 9.63, p = 0.01), and a near significant decrease in driving while drunk (F(1,137), 3.64, p = 0.06). Univariate tests also showed increases in the consumption of foods containing healthy fats in the past 7 days (e.g., vegetable oil, seeds, nuts, olive oil, or fish) over time (F(1,145), 4.67, p = 0.03). |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Scott, S.; Beyer, F.; Parkinson, K.; Muir, C.; Graye, A.; Kaner, E.; Stead, M.; Power, C.; Fitzgerald, N.; Bradley, J.; et al. Non-Pharmacological Interventions to Reduce Unhealthy Eating and Risky Drinking in Young Adults Aged 18–25 Years: A Systematic Review and Meta-Analysis. Nutrients 2018, 10, 1538. https://doi.org/10.3390/nu10101538
Scott S, Beyer F, Parkinson K, Muir C, Graye A, Kaner E, Stead M, Power C, Fitzgerald N, Bradley J, et al. Non-Pharmacological Interventions to Reduce Unhealthy Eating and Risky Drinking in Young Adults Aged 18–25 Years: A Systematic Review and Meta-Analysis. Nutrients. 2018; 10(10):1538. https://doi.org/10.3390/nu10101538
Chicago/Turabian StyleScott, Stephanie, Fiona Beyer, Kathryn Parkinson, Cassey Muir, Alice Graye, Eileen Kaner, Martine Stead, Christine Power, Niamh Fitzgerald, Jen Bradley, and et al. 2018. "Non-Pharmacological Interventions to Reduce Unhealthy Eating and Risky Drinking in Young Adults Aged 18–25 Years: A Systematic Review and Meta-Analysis" Nutrients 10, no. 10: 1538. https://doi.org/10.3390/nu10101538





