Immune Function and Micronutrient Requirements Change over the Life Course
Abstract
:1. Introduction
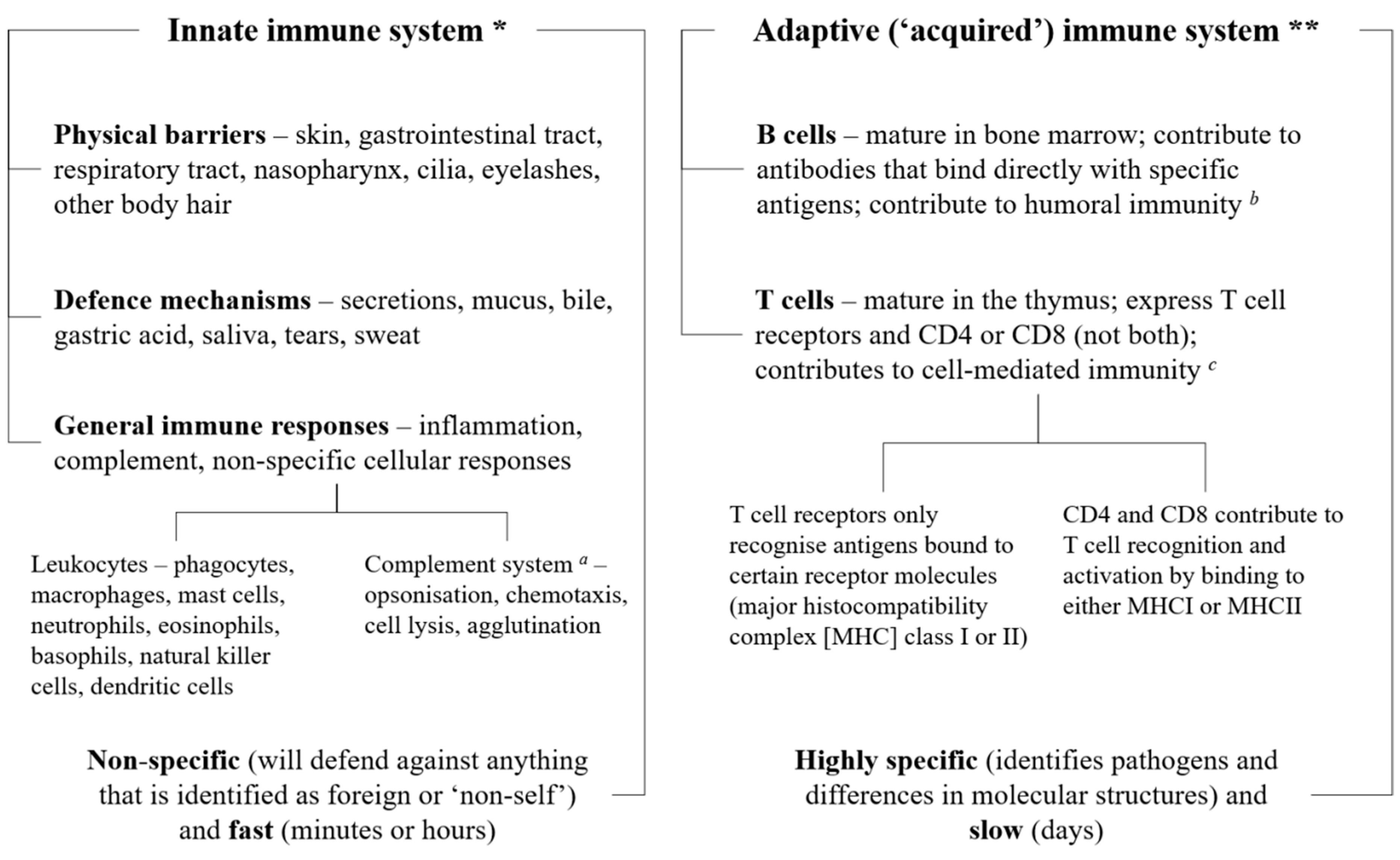
2. The Immune System
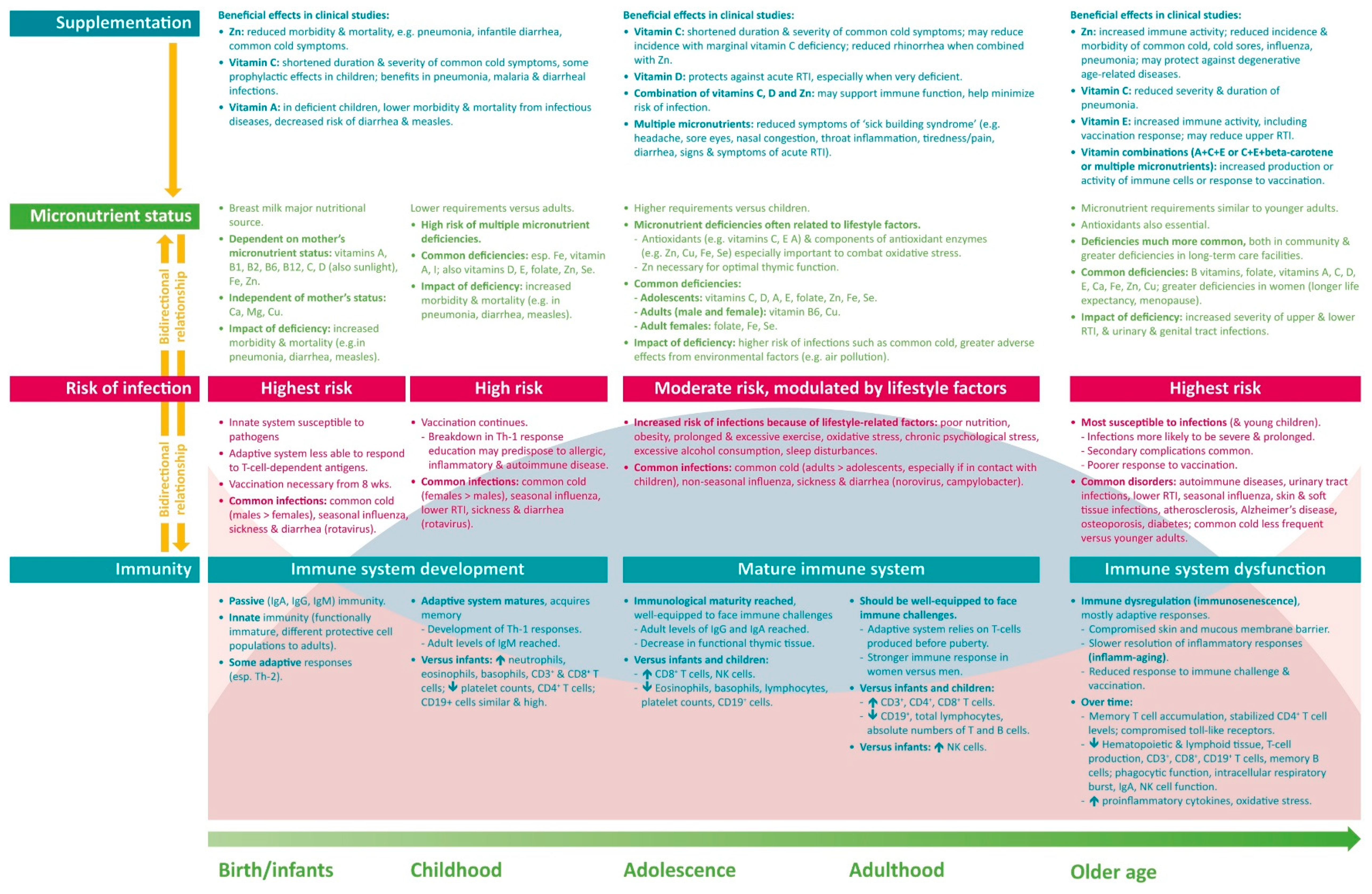
2.1. Infants and Children
2.2. Adolescents and Adults
2.3. Older People
3. Response to Infection
3.1. Infants and Children
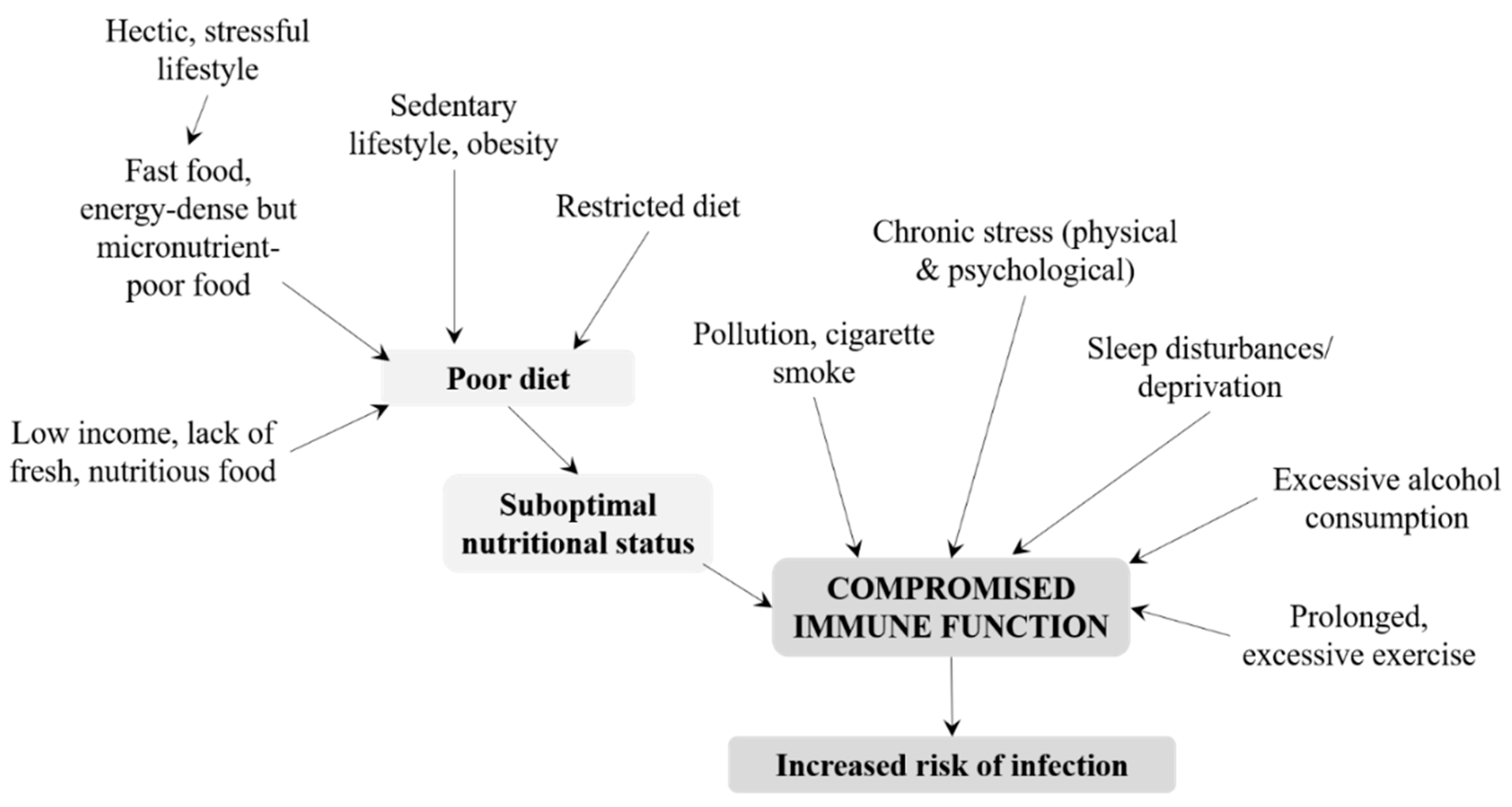
3.2. Adolescents and Adults
3.3. Older People
4. Micronutrient Requirements and Reported Deficiencies
4.1. Infants and Children
4.2. Adolescents and Adults
4.3. Older People
5. Clinical Impact of Micronutrient Deficiencies and Supplementation
5.1. Infants and Children
5.2. Adolescents and Adults
5.3. Older People
6. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Castelo-Branco, C.; Soveral, I. The immune system and aging: A review. Gynecol. Endocrinol. 2014, 30, 16–22. [Google Scholar] [CrossRef] [PubMed]
- Pandya, P.H.; Murray, M.E.; Pollok, K.E.; Renbarger, J.L. The immune system in cancer pathogenesis: Potential therapeutic approaches. J. Immunol. Res. 2016, 2016, 4273943. [Google Scholar] [CrossRef] [PubMed]
- Maggini, S.; Maldonado, P.; Cardim, P.; Fernandez Newball, C.; Sota Latino, E. Vitamins C., D and zinc: Synergistic roles in immune function and infections. Vitam. Miner. 2017, 6, 167. [Google Scholar] [CrossRef]
- Alpert, P. The role of vitamins and minerals on the immune system. Home Health Care Manag. Pract. 2017, 29, 199–202. [Google Scholar] [CrossRef]
- Calder, P. Conference on ‘Transforming the nutrition landscape in Africa’. Plenary Session 1: Feeding the immune system. Proc. Nutr. Soc. 2013, 72, 299–309. [Google Scholar] [CrossRef] [PubMed]
- Watson, R.R.; Zibadi, S.; Preedy, V.R. Dietary Components and Immune Function; Springer Science & Business Media: Berlin, Germany, 2010. [Google Scholar]
- Biebinger, R.; Hurrell, R.F. 3—Vitamin and mineral fortification of foods. In Food Fortification and Supplementation; Ottaway, P.B., Ed.; Woodhead Publishing: Cambridge, UK, 2008; pp. 27–40. [Google Scholar]
- Schaefer, E. Micronutrient deficiency in women living in industrialized countries during the reproductive years: Is there a basis for supplementation with multiple micronutrients? J. Nutr. Disord. Ther. 2016, 6, 199. [Google Scholar] [CrossRef]
- Wintergerst, E.; Maggini, S.; Hornig, D. Immune-enhancing role of vitamin C and zinc and effect on clinical conditions. Ann. Nutr. Metab. 2006, 50, 85–94. [Google Scholar] [CrossRef] [PubMed]
- Haryanto, B.; Suksmasari, T.; Wintergerst, E.; Maggini, S. Multivitamin supplementation supports immune function and ameliorates conditions triggered by reduced air quality. Vitam. Miner. 2015, 4, 1–15. [Google Scholar]
- Maggini, S.; Beveridge, S.; Sorbara, J.; Senatore, G. Feeding the immune system: The role of micronutrients in restoring resistance to infections. CAB Rev. 2008, 3, 1–21. [Google Scholar] [CrossRef]
- Carr, A.; Maggini, S. Vitamin C and immune function. Nutrients 2017, 9, 1211. [Google Scholar] [CrossRef] [PubMed]
- Saeed, F.; Nadeem, M.; Ahmed, R.; Nadeem, M.; Arshad, M.; Ullah, A. Studying the impact of nutritional immunology underlying the modulation of immune responses by nutritional compounds—A review. Food Agric. Immunol. 2016, 27, 205–229. [Google Scholar] [CrossRef]
- Micronutrient Information Center. Immunity in Depth. Available online: http://lpi.oregonstate.edu/mic/health-disease/immunity (accessed on 17 April 2018).
- Meydani, S.; Ribaya-Mercado, J.; Russell, R.; Sahyoun, N.; Morrow, F.; Gershoff, S. Vitamin B-6 deficiency impairs interleukin 2 production and lymphocyte proliferation in elderly adults. Am. J. Clin. Nutr. 1991, 53, 1275–1280. [Google Scholar] [CrossRef] [PubMed]
- Wintergerst, E.; Maggini, S.; Hornig, D. Contribution of selected vitamins and trace elements to immune function. Nutr. Metab. 2007, 51, 301–323. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization; Food and Agricultural Organization of the United Nations. Part 2. Evaluating the public health significance of micronutrient malnutrition. In Guidelines on Food Fortification with Micronutrients; World Health Organization: Geneva, Switzerland, 2006. [Google Scholar]
- Maggini, S.; Wintergerst, E.S.; Beveridge, S.; Hornig, D.H. Selected vitamins and trace elements support immune function by strengthening epithelial barriers and cellular and humoral immune responses. Br. J. Nutr. 2007, 98, S29–S35. [Google Scholar] [CrossRef] [PubMed]
- Simon, A.; Hollander, G.; McMichael, A. Evolution of the immune system in humans from infancy to old age. Proc. R. Soc. B 2015, 282, 20143085. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Butler, E.; Kehrli, M. Immunoglobulins and immunocytes in the mammary gland and its secretions. In Mucosal Immunology; Mestecky, J., Lamm, M., Ogra, P., Strober, W., Bienenstock, J., McGhee, J., Mayer, L., Eds.; Elsevier: Amsterdam, The Netherland, 2005; pp. 1763–1793. [Google Scholar]
- Field, C.J. The immunological components of human milk and their effect on immune development in infants. J. Nutr. 2005, 135, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Witkowska-Zimny, M.; Kaminska-El-Hassan, E. Cells of human breast milk. Cell. Mol. Biol. Lett. 2017, 22, 11. [Google Scholar] [CrossRef] [PubMed]
- Brodin, P.; Davis, M. Human immune system variation. Nat. Rev. Immunol. 2017, 17, 21–29. [Google Scholar] [CrossRef] [PubMed]
- MacGillivray, D.; Kollmann, T. The role of environmental factors in modulating immune responses in early life. Front. Immunol. 2014, 5, 434. [Google Scholar] [CrossRef] [PubMed]
- Valiathan, R.; Ashman, M.; Asthana, D. Effects of ageing on the immune system: Infants to elderly. Scand. J. Immunol. 2016, 83, 255–266. [Google Scholar] [CrossRef] [PubMed]
- Hepworth, M.; Sonnenberg, G. Regulation of the adaptive immune system by innate lymphoid cells. Curr. Opin. Immunol. 2014, 27, 75–82. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tortura, G.J.; Derrickson, B. Metabolism and nutrition. In Principles of Anatomy and Physiology, 8th ed.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2014; pp. 940–978. [Google Scholar]
- Wang, K.; Wei, G.; Liu, D. CD19: A biomarker for B cell development, lymphoma diagnosis and therapy. Exp. Hematol. Oncol. 2012, 1, 36. [Google Scholar] [CrossRef] [PubMed]
- Berg, T.; Johansson, S. Immunoglobulin levels during childhood, with special regard to IgE. Acta Paediatr. 1969, 58, 513–524. [Google Scholar] [CrossRef]
- Pawelec, G. Hallmarks of human “immunosenescence”: Adaptation or dysregulation? Immun. Ageing 2012, 9, 15. [Google Scholar] [CrossRef] [PubMed]
- Klein, S.; Hodgson, A.; Robinson, D. Mechanisms of sex disparities in influenza pathogenesis. J. Leukoc. Biol. 2012, 92, 67–73. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fulop, T.; Witkowski, J.; Pawelec, G.; Alan, C.; Larbi, A. On the immunological theory of aging. In Aging: Facts and Theories; Robert, L., Fulop, T., Eds.; Karger: Basel, Switzerland, 2014; Volume 39, pp. 163–176. [Google Scholar]
- Chandra, R. Nutrition and the immune system from birth to old age. Eur. J. Clin. Nutr. 2002, 56, S73–S76. [Google Scholar] [CrossRef] [PubMed]
- Montecino-Rodriguez, E.; Berent-Maoz, B.; Dorshkind, K. Causes, consequences, and reversal of immune system aging. J. Clin. Investig. 2013, 123, 958–965. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ventura, M.; Casciaro, M.; Gangemi, S.; Buquicchio, R. Immunosenescence in aging: Between immune cells depletion and cytokines up-regulation. Clin. Mol. Allergy 2017, 15, 21. [Google Scholar] [CrossRef] [PubMed]
- Pawelec, G. Does the human immune system ever really become “senescent”? F1000Research 2017, 6, 1323. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Goronzy, J.; Weyand, C. Immune aging and autoimmunity. Cell. Mol. Life Sci. 2012, 69, 1615–1623. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jafarzadeh, A.; Sadeghi, M.; Karam, G.A.; Vazirinejad, R. Salivary IgA and IgE levels in healthy subjects: Relation to age and gender. Braz. Oral Res. 2010, 24, 21–27. [Google Scholar] [CrossRef] [PubMed]
- Grewe, M. Chronological ageing and photoageing of dendritic cells. Clin. Exp. Dermatol. 2001, 26, 608–612. [Google Scholar] [CrossRef] [PubMed]
- Pand, A.A.; Qian, F.; Mohanty, S.; van Duin, D.; Newman, F.K.; Zhang, L.; Chen, S.; Towle, V.; Belshe, R.B.; Fikrig, E.; et al. Age-associated decrease in TLR function in primary human dendritic cells predicts influenza vaccine response. J. Immunol. 2010, 184, 2518–2527. [Google Scholar] [CrossRef] [PubMed]
- Hemmi, H.; Akira, S. TLR signalling and the function of dendritic cells. Chem. Immunol. Allergy 2005, 86, 120–135. [Google Scholar] [PubMed]
- Zhang, Y.; Wallace, D.; de Lara, C.; Ghattas, H.; Asquith, B.; Worth, A.; Griffin, G.; Taylor, G.; Tough, D.; Beverley, P.; et al. In vivo kinetics of human natural killer cells: The effects of ageing and acute and chronic viral infection. Immunology 2007, 121, 258–265. [Google Scholar] [CrossRef] [PubMed]
- Hazeldine, J.; Lord, J. The impact of ageing on natural killer cell function and potential consequences for health in older adults. Ageing Res. Rev. 2013, 12, 1069–1078. [Google Scholar] [CrossRef] [PubMed]
- Fulop, T.; Larbi, A.; Dupuis, G.; Le Page, A.; Frost, E.; Cohen, A.; Witkowski, J.; Franceschi, C. Immunosenescence and inflamm-aging as two sides of the same coin: Friends or foes? Front. Immunol. 2017, 8, 1960. [Google Scholar] [CrossRef] [PubMed]
- Monto, A.; Ullman, B. Acute respiratory illness in an American community: The Tecumseh study. JAMA 1974, 227, 164–169. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Influenza (Seasonal). Fact Sheet. Available online: http://www.who.int/mediacentre/factsheets/fs211/en/ (accessed on 28 April 2018).
- National Institute for Health and Care Excellence Diarrhoea and Vomiting Caused by Gastroenteritis in under 5s: Diagnosis and Management. Clinical Guideline [CG84]. Available online: https://www.nice.org.uk/guidance/cg84 (accessed on 28 April 2018).
- GBD 2015 LRI Collaborators. Estimates of the global, regional, and national morbidity, mortality, and aetiologies of lower respiratory tract infections in 195 countries: A systematic analysis for the Global Burden of Disease Study 2015. Lancet Infect. Dis. 2017, 17, 1133–1161. [Google Scholar] [CrossRef]
- Calder, P.; Prescott, S.; Caplan, M. Scientific Review: The Role of Nutrients in Immune Function of Infants and Young Children; Emerging Evidence for Long-Chain Polyunsaturated Fatty Acids; Mead Johnson & Company: Glenview, IL, USA, 2007. [Google Scholar]
- Bresnahan, K.; Tanumihardjo, S. Undernutrition, the acute phase response to infection, and its effects on micronutrient status indicators. Adv. Nutr. 2014, 5, 702–711. [Google Scholar] [CrossRef] [PubMed]
- Milner, J.; Beck, M. Micronutrients, immunology and inflammation. The impact of obesity on the immune response to infection. Proc. Nutr. Soc. 2012, 71, 298–306. [Google Scholar] [CrossRef] [PubMed]
- Gleeson, M. Effects of exercise on immune function. Sports Sci. Exch. 2015, 28, 1–6. [Google Scholar]
- Gleeson, M. Immunological aspects of sports nutrition. Immunol. Cell Biol. 2016, 94, 117–123. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nieman, D. Immunonutrition support for athletes. Nutr. Rev. 2008, 66, 310–320. [Google Scholar] [CrossRef] [PubMed]
- Campbell, J.; Turner, J. Debunking the myth of exercise-induced immune suppression: Redefining the impact of exercise on immunological health across the lifespan. Front. Immunol. 2018, 9, 648. [Google Scholar] [CrossRef] [PubMed]
- Segerstrom, S.; Miller, G. Psychological stress and the human immune system: A meta-analytic study of 30 years of inquiry. Psychol. Bull. 2004, 130, 601–630. [Google Scholar] [CrossRef] [PubMed]
- Romeo, J.; Wärnberg, J.; Nova, E.; Díaz, L.E.; Gómez-Martinez, S.; Marcos, A. Moderate alcohol consumption and the immune system: A review. Br. J. Nutr. 2007, 98, S111–S115. [Google Scholar] [CrossRef] [PubMed]
- Besedovsky, L.; Lange, T.; Born, J. Sleep and immune function. Eur. J. Physiol. 2012, 163, 121–137. [Google Scholar] [CrossRef] [PubMed]
- Cohen, S.; Tyrrell, D.; Smith, A. Psychological stress and susceptibility to the common cold. N. Engl. J. Med. 1991, 325, 606–612. [Google Scholar] [CrossRef] [PubMed]
- Nieman, D. Exercise, upper respiratory tract infection, and the immune system. Med. Sci. Sports Exerc. 1994, 26, 128–139. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Risk Management Solutions. Learning from the 2009 H1N1 Influenza Pandemic. RMS Special Report. Available online: http://static.rms.com/email/documents/liferisks/reports/learning-from-the-2009-h1n1-influenza-pandemic.pdf (accessed on 28 April 2018).
- Marshall, J.A.; Bruggink, L.D. The dynamics of norovirus outbreak epidemics: Recent insights. Int. J. Environ. Res. Public Health 2011, 8, 1141–1149. [Google Scholar] [CrossRef] [PubMed]
- Man, S. The clinical importance of emerging Campylobacter species. Nat. Rev. Gastroenterol. Hepatol. 2011, 8, 669–685. [Google Scholar] [CrossRef] [PubMed]
- Centers for Disease Control and Prevention. Norovirus Worldwide. Available online: https://www.cdc.gov/norovirus/worldwide.html (accessed on 29 July 2018).
- Yoshikawa, T. Epidemiology and unique aspects of aging and infectious diseases. Clin. Infect. Dis. 2000, 30, 931–933. [Google Scholar] [CrossRef] [PubMed]
- Heikkinen, T.; Jarvinen, A. The common cold. Lancet 2003, 361, 51–59. [Google Scholar] [CrossRef]
- Eccles, R. Mechanisms of symptoms of common cold and flu. In Common Cold; Eccles, R., Weber, O., Eds.; Birkhauser Verlag: Basel, Switzerland, 2009; pp. 23–45. [Google Scholar]
- Ballinger, M.; Standiford, T. Postinfluenza bacterial pneumonia: Host defenses gone awry. J. Interferon Cytokine Res. 2010, 30, 643–652. [Google Scholar] [CrossRef] [PubMed]
- Albers, R.; Bourdet-Sicard, R.; Braun, D.; Calder, P.; Herz, U.; Lambert, C.; Lenoir-Wijnkoop, I.; Meheurst, A.; Ouwehand, A.; Phothirath, P.; et al. Monitoring immune modulation by nutrition in the general population: Identifying and substantiating effects on human health. Br. J. Nutr. 2013, S110–S130. [Google Scholar] [CrossRef] [PubMed]
- Bhaskaram, P. Micronutrient malnutrition, infection, and immunity: An overview. Nutr. Rev. 2002, 60, S40–S45. [Google Scholar] [CrossRef] [PubMed]
- Prentice, S. They are what you eat: Can nutritional factors during gestation and early infancy modulate the neonatal immune response? Front. Immunol. 2017, 8, 1641. [Google Scholar] [CrossRef] [PubMed]
- Butte, N.; Lopez-Alarcon, M.; Garza, C. Nutrient Adequacy of Exclusive Breastfeeding for the Term Infant during the First Six Months of Life. Available online: http://apps.who.int/iris/handle/10665/42519 (accessed on 12 December 2017).
- Björklund, K.; Vahter, M.; Palm, B.; Grandér, M.; Lignell, S.; Berglund, M. Metals and trace element concentrations in breast milk of first time healthy mothers: A biological monitoring study. Environ. Health 2012, 11, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Hall Moran, V.; Lowe, N.; Crossland, N.; Berti, C.; Cetin, I.; Hermoso, M.; Koletzko, B.; Dykes, F. Nutritional requirements during lactation. Towards European alignment of reference values: The EURRECA network. Matern. Child Health 2010, 6, 39–54. [Google Scholar] [CrossRef] [PubMed]
- Kominiarek, M.; Rajan, P. Nutrition recommendations in pregnancy and lactation. Med. Clin. N. Am. 2016, 100, 1199–1215. [Google Scholar] [CrossRef] [PubMed]
- Dawodu, A.; Tsang, R. Maternal vitamin D status: Effect on milk vitamin D content and vitamin D status of breastfeeding infants. Adv. Nutr. 2012, 3, 353–361. [Google Scholar] [CrossRef] [PubMed]
- Semba, R. Impact of micronutrient deficiencies on immune function. In Micronutrient Deficiencies during the Weaning Period and the First Years of Life, Proceedings of the 54th Nestlé Nutrition Workshop, Pediatric Program, São Paulo, Brazil, 26–30 October 2003; Pettifor, J., Zlotkin, S., Eds.; Nestlé Nutrition Institute Workshop Series: Lausanne, Switzerland, 2004. [Google Scholar]
- Institute of Medicine. Dietary Reference Intakes for Calcium and Vitamin D; The National Academies Press: Washington, DC, USA, 2011. [Google Scholar]
- Marasinghe, E.; Chackrewarthy, S.; Abeysena, C.; Rajindrajith, S. Micronutrient status and its relationship with nutritional status in preschool children in urban Sri Lanka. Asia Pac. J. Clin. Nutr. 2015, 24, 144–151. [Google Scholar] [PubMed]
- Luo, R.; Shi, Y.; Zhou, H.; Yue, A.; Zhang, L.; Sylvia, S.; Medina, A.; Rozelle, S. Micronutrient deficiencies and developmental delays among infants: Evidence from a cross-sectional survey in rural China. BMJ Open 2015, 5, e008400. [Google Scholar] [CrossRef] [PubMed]
- Özden, T.A.; Gökçay, G.; Cantez, M.S.; Durmaz, Ö.; İşsever, H.; Ömer, B.; Saner, G. Copper, zinc and iron levels in infants and their mothers during the first year of life: A prospective study. BMC Pediatr. 2015, 15, 157. [Google Scholar] [CrossRef] [PubMed]
- Jardim-Botelho, A.; Queiroz Gurgel, R.; Simeone Henriques, G.; Dos Santos, C.B.; Afonso Jordão, A.; Nascimento Faro, F.; Silveira Souto, F.M.; Rodrigues Santos, A.P.; Eduardo Cuevas, L. Micronutrient deficiencies in normal and overweight infants in a low socio-economic population in north-east Brazil. Paediatr. Int. Child Health 2016, 36, 198–202. [Google Scholar] [CrossRef] [PubMed]
- Bailey, R.; West, K.J.; Black, R. The epidemiology of global micronutrient deficiencies. Ann. Nutr. Metab. 2015, 66, 22–33. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Food and Agricultural Organization of the United Nations. Part I. The role of food fortification in the control of micronutrient malnutrition. In Guidelines on Food Fortification with Micronutrients; Allen, L., de Benoist, B., Dary, O., Hurrell, R., Eds.; World Health Organization: Geneva, Switzerland, 2006. [Google Scholar]
- Mackey, A.; Picciano, M. Maternal folate status during extended lactation and the effect of supplemental folic acid. Am. J. Clin. Nutr. 1999, 69, 285–292. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Houghton, L.; Sherwood, K.; O’Connor, D. How well do blood folate concentrations predict dietary folate intakes in a sample of Canadian lactating women exposed to high levels of folate? An observational study. BMC Pregnancy Childbirth 2007, 7, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Gellert, S.; Ströhle, A.; Hahn, A. Breastfeeding woman are at higher risk of vitamin D deficiency than nonbreastfeeding women - insights from the German VitaMinFemin study. Int. Breastfeed. J. 2017, 12, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Milman, N.; Hvas, A.-M.; Bergholt, T. Vitamin D status during normal pregnancy and postpartum. A longitudinal study in 141 Danish women. J. Perinat. Med. 2012, 40, 57–61. [Google Scholar] [CrossRef] [PubMed]
- Dawodu, A.; Zalla, L.; Woo, J.G.; Herbers, P.M.; Davidson, B.S.; Heubi, J.E.; Morrow, A.L. Heightened attention to supplementation is needed to improve the vitamin D status of breastfeeding mothers and infants when sunshine exposure is restricted. Matern. Child Health 2014, 10, 383–397. [Google Scholar] [CrossRef] [PubMed]
- Hannan, M.; Faraji, B.; Tanguma, J.; Longoria, N.; Rodriguez, R. Maternal milk concentration of zinc, iron, selenium, and iodine and its relationship to dietary intakes. Biol. Trace Elem. Res. 2009, 127, 6–15. [Google Scholar] [CrossRef] [PubMed]
- Charlton, K.; Yeatman, H.; Lucas, C.; Axford, S.; Gemming, L.; Houweling, F.; Goodfellow, A.; Ma, G. Poor knowledge and practices related to iodine nutrition during pregnancy and lactation in australian women: Pre- and post-iodine fortification. Nutrients 2012, 4, 1317–1327. [Google Scholar] [CrossRef] [PubMed]
- Mulrine, H.M.; Skeaff, S.A.; Ferguson, E.L.; Gray, A.R.; Valeix, P. Breast-milk iodine concentration declines over the first 6 mo postpartum in iodine-deficient women. Am. J. Clin. Nutr. 2010, 92, 849–856. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Valent, F.; Horvat, M.; Mazej, D.; Stibilj, V.; Barbone, F. Maternal diet and selenium concentration in human milk from an Italian population. J. Epidemiol. 2011, 21, 285–292. [Google Scholar] [CrossRef] [PubMed]
- Brenna, J.T.; Varamini, B.; Jensen, R.G.; Diersen-Schade, D.A.; Boettcher, J.A.; Arterburn, L.M. Docosahexaenoic and arachidonic acid concentrations in human breast milk worldwide. Am. J. Clin. Nutr. 2007, 85, 1457–1464. [Google Scholar] [CrossRef] [PubMed]
- Podzolkova, N.; Schaefer, E. Micronutrient intakes and status are frequently insufficient in breastfeeding women. BMC Pregnancy Childbirth 2018. submitted for publication. [Google Scholar]
- Elmadfa, I.; Meyer, A.; Nowak, V.; Hasenegger, V.; Putz, P.; Verstraeten, R.; Remaut-DeWinter, A.M.; Kolsteren, P.; Dostálová, J.; Dlouhý, P.; et al. European Nutrition and Health Report. Forum Nutr. 2009, 62, 1–405. [Google Scholar] [PubMed]
- Savino, W.; Dardenne, M. Nutritional imbalances and infections affect the thymus: Consequences on T-cell-mediated immune responses. Proc. Nutr. Soc. 2010, 69, 636–643. [Google Scholar] [CrossRef] [PubMed]
- Palacios, C.; Gonzalez, L. Is vitamin D deficiency a major global public health problem? J. Steroid Biochem. Mol. Biol. 2014, 144PA, 138–145. [Google Scholar] [CrossRef] [PubMed]
- Montgomery, S.; Streit, S.; Beebe, M.; Maxwell IV, P. Micronutrient needs of the elderly. Nutr. Clin. Pract. 2014, 29, 435–444. [Google Scholar] [CrossRef] [PubMed]
- Drenowski, A.; Shultz, J. Impact of aging on eating behaviors, food choices, nutrition, and health status. J. Nutr. Health Aging 2001, 5, 75–79. [Google Scholar]
- High, K. Nutritional strategies to boost immunity and prevent infection in elderly individuals. Clin. Infect. Dis. 2001, 33, 1892–1900. [Google Scholar] [CrossRef] [PubMed]
- Wiacek, M.; Zubrzycki, I.; Bojke, O.; Kim, H. Menopause and age-driven changes in blood level of fat- and water-soluble vitamins. Climacteric 2013, 16, 689–699. [Google Scholar] [CrossRef] [PubMed]
- Karaouzenea, N.; Merzouka, H.; Aribib, M.; Merzoukc, S.; Yahia Berrouiguet, A.; Tessiere, C.; Narce, M. Effects of the association of aging and obesity on lipids, lipoproteins and oxidative stress biomarkers: A comparison of older with young men. Nutr. Metab. Cardiovasc. Dis. 2011, 21, 792–799. [Google Scholar] [CrossRef] [PubMed]
- Hemilä, H. Vitamin C and infections. Nutrients 2017, 9, 339. [Google Scholar] [CrossRef] [PubMed]
- Hemilä, H.; Chalker, E. Vitamin C for preventing and treating the common cold. Cochrane Database Syst. Rev. 2013. Available online: https://www.cochranelibrary.com/cdsr/doi/10.1002/14651858.CD000980.pub4/full (accessed on 17 October 2018).[Green Version]
- Aranow, C. Vitamin D and the immune system. J. Investig. Med. 2011, 59, 881–886. [Google Scholar] [CrossRef] [PubMed]
- Mangin, M.; Sinha, R.; Fincher, K. Inflammation and vitamin D: The infection connection. Inflamm. Res. 2014, 63, 803–819. [Google Scholar] [CrossRef] [PubMed]
- Hemilä, H. Zinc lozenges may shorten the duration of colds: A systematic review. Open Respir. Med. J. 2011, 5, 51–58. [Google Scholar] [PubMed]
- Johnston, C.; Barkyoumb, G.M.; Schumacher, S.S. Vitamin C supplementation slightly improves physical activity levels and reduces cold incidence in men with marginal vitamin C status: A randomized controlled trial. Nutrients 2014, 6, 2572–2583. [Google Scholar] [CrossRef] [PubMed]
- Maggini, S.; Beveridge, S.; Suter, M. A combination of high-dose vitamin C plus zinc for the common cold. J. Int. Med. Res. 2012, 40, 28–42. [Google Scholar] [CrossRef] [PubMed]
- Graf, P.; Eccles, R.; Chen, S. Efficacy and safety of intranasal xylometazoline and ipratropium in patients with common cold. Expert Opin. Pharmacother. 2009, 10, 889–908. [Google Scholar] [CrossRef] [PubMed]
- Yamshchikov, A.; Desai, N.; Blumberg, H.; Ziegler, T.; Tangpricha, V. Vitamin D for treatment and prevention of infectious diseases: A systematic review of randomized controlled trials. Endocr. Pract. 2009, 15, 438–449. [Google Scholar] [CrossRef] [PubMed]
- Charan, J.; Goyal, J.; Saxena, D.; Yadav, P. Vitamin D for prevention of respiratory tract infections: A systematic review and meta-analysis. J. Pharmacol. Pharmacother. 2012, 3, 300–303. [Google Scholar] [CrossRef] [PubMed]
- Bergman, P.; Lindh, Å.; Björkhem-Bergman, L.; Lindh, J. Vitamin D and respiratory tract infections: A systematic review and meta-analysis of randomized controlled trials. PLoS ONE 2013, 8, e65835. [Google Scholar] [CrossRef] [PubMed]
- Martineau, A.; Jolliffe, D.; Hooper, R.; Greenberg, L.; Aloia, J.; Bergman, P.; Dubnov-Raz, G.; Esposito, S.; Ganmaa, D.; Ginde, A.A.; et al. Vitamin D supplementation to prevent acute respiratory infections: Systematic review and meta-analysis of individual participant data. BMJ 2017, 356, i6583. [Google Scholar] [CrossRef] [PubMed]
- Hamer, D.; Sempértegui, F.; Estrella, B.; Tucker, K.; Rodríguez, A.; Egas, J.; Dallal, G.; Selhub, J.; Griffiths, J.; Meydani, S. Micronutrient deficiencies are associated with impaired immune response and higher burden of respiratory infections in elderly Ecuadorians. J. Nutr. 2009, 139, 113–119. [Google Scholar] [CrossRef] [PubMed]
- Penn, N.D.; Purkins, L.; Kelleher, J.; Heatley, R.V.; Mascie-Taylor, B.H.; Belfield, P.W. The effect of dietary supplementation with vitamins A, C, and E on cell-mediated immune function in elderly log-stay patients: A randomized controlled trial. Age Ageing 1991, 20, 169–174. [Google Scholar] [CrossRef] [PubMed]
- Chandra, R. Effect of vitamin and trace-element supplementation on immune responses and infection in elderly subjects. Lancet 1992, 340, 1124–1127. [Google Scholar] [CrossRef]
- Schmoranzer, F.; Fuchs, N.; Markolin, G.; Carlin, E.; Sakr, L.; Sommeregger, U. Influence of a complex micronutrient supplement on the immune status of elderly individuals. Int. J. Vitam. Nutr. Res. 2009, 79, 308–318. [Google Scholar] [CrossRef] [PubMed]
- Prasad, A. Zinc: Mechanisms of host defense. J. Nutr. 2007, 137, 1345–1349. [Google Scholar] [CrossRef] [PubMed]
- Meydani, S.; Barnett, J.; Dallal, G.; Fine, B.; Jacques, P.; et al. Serum zinc and pneumonia in nursing home elderly. Am. J. Clin. Nutr. 2007, 86, 1167–1173. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mocchegiani, E.; Romeo, J.; Malavolta, M.; Costarelli, L.; Giacconi, R.; Diaz, L.; Marcos, A. Zinc: Dietary intake and impact of supplementation on immune function in elderly. Age (Dordr.) 2013, 35, 839–860. [Google Scholar] [CrossRef] [PubMed]
- De la Fuente, M.; Hernanz, A.; Guayerbas, N.; Victor, V.; Arnalich, F. Vitamin E ingestion improves several immune functions in elderly men and women. Free Radic. Res. 2008, 42, 272–280. [Google Scholar] [CrossRef] [PubMed]
- Meydani, S.; Meydani, M.; Blumberg, J.; Leka, L.; Siber, G.; Loszewski, R.; Thompson, C.; Pedrosa, M.; Diamond, R.; Stollar, B. Vitamin E supplementation and in vivo immune response in healthy elderly subjects. A randomized controlled trial. JAMA 1997, 277, 1380–1386. [Google Scholar] [CrossRef] [PubMed]
- Meydani, S.; Leka, L.; Fine, B.; Dallal, G.; Keusch, G.; Singh, M.; Hamer, D. Vitamin E and respiratory tract infections in elderly nursing home residents: A randomized controlled trial. JAMA 2004, 292, 828–836. [Google Scholar] [CrossRef] [PubMed]
| Micronutrient/Role | Innate Immunity | Adaptive Immunity |
|---|---|---|
| Vitamin C | Effective antioxidant that protects against ROS and RNS produced when pathogens are killed by immune cells [9,14] Regenerates other important antioxidants such as glutathione and vitamin E to their active state [9] Promotes collagen synthesis, thereby supporting the integrity of epithelial barriers [10] Stimulates production, function and movement of leukocytes (e.g., neutrophils, lymphocytes, phagocytes) [9,14] Increases serum levels of complement proteins [14] Has roles in antimicrobial and NK cell activities and chemotaxis [10] Involved in apoptosis and clearance of spent neutrophils from sites of infection by macrophages [12] | Can increase serum levels of antibodies [12,14] Has roles in lymphocyte differentiation and proliferation [10,12] |
| Vitamin D | Vitamin D receptor expressed in innate immune cells (e.g., monocytes, macrophages, dendritic cells) [14] Increases the differentiation of monocytes to macrophages [10] Stimulates immune cell proliferation and cytokine production and helps protect against infection caused by pathogens [14] 1,25-dihydroxyvitamin D3, the active form of vitamin D, regulates the antimicrobial proteins cathelicidin and defensin, which can directly kill pathogens, especially bacteria [14] | Mainly inhibitory effect in adaptive immunity [14]; for example, 1,25-dihydroxyvitamin D3 suppresses antibody production by B cells and inhibits T cell proliferation [14] |
| Vitamin A | Helps maintain structural and functional integrity of mucosal cells in innate barriers (e.g., skin, respiratory tract, etc.) [14] Important for normal functioning of innate immune cells (e.g., NK cells, macrophages, neutrophils) [14] | Necessary for proper functioning of T and B lymphocytes, and thus for generation of antibody responses to antigen [14] Involved in development and differentiation of Th1 and Th2 cells and supports Th2 anti-inflammatory response [10] |
| Vitamin E | An important fat-soluble antioxidant [10] Protects the integrity of cell membranes from damage caused by free radicals [14] Enhances IL-2 production and NK cell cytotoxic activity [10] | Enhances T cell-mediated functions and lymphocyte proliferation [10] Optimizes and enhances Th1 and suppresses Th2 response [10] |
| Vitamin B6 | Helps regulate inflammation [13] Has roles in cytokine production and NK cell activity [13,15] | Required in the endogenous synthesis and metabolism of amino acids, the building blocks of cytokines and antibodies [14] Has roles in lymphocyte proliferation, differentiation and maturation [14] Maintains Th1 immune response [10] Has roles in antibody production [13] |
| Vitamin B12 | Has roles in NK cell functions [13] | May act as an immunomodulator for cellular immunity, especially with effects on cytotoxic cells (NK cells, CD8+ T-cells) [10] Facilitates production of T lymphocytes [13] Involved in humoral and cellular immunity and one-carbon metabolism (interactions with folate) [13] |
| Folate | Maintains innate immunity (NK cells) [10] | Has roles in cell-mediated immunity [13] Important for sufficient antibody response to antigens [13] Supports Th 1-mediated immune response [13] |
| Zinc | Antioxidant effects protect against ROS and RNS [9] Helps modulate cytokine release and induces proliferation of CD8+ T cells [10,16] Helps maintain skin and mucosal membrane integrity [10] | Central role in cellular growth and differentiation of immune cells that have a rapid differentiation and turnover [17] Essential for intracellular binding of tyrosine kinase to T cell receptors, required for T lymphocyte development and activation [9] Supports Th1 response [10] |
| Iron | Involved in regulation of cytokine production and action [10] Forms highly-toxic hydroxyl radicals, thus involved in the process of killing bacteria by neutrophils [10] Important in the generation of ROS that kill pathogens [14] | Important in the differentiation and proliferation of T lymphocytes [14] Essential for cell differentiation and growth, component of enzymes critical for functioning of immune cells (e.g., ribonucleotide reductase involved in DNA synthesis) [10] |
| Copper | Free-radical scavenger [4] Antimicrobial properties [14] Accumulates at sites of inflammation, important for IL-2 production and response [13,14] May play a role in the innate immune response to bacterial infections [14] | Has roles in T cell proliferation [13] Has roles in antibody production and cellular immunity [18] |
| Selenium | Essential for the function of selenium-dependent enzymes (selenoproteins) that can act as redox regulators and cellular antioxidants, potentially counteracting ROS [10,14] Selenoproteins are important for the antioxidant host defense system affecting leukocyte and NK cell function [13] | Involved in T lymphocyte proliferation [4,13] Has roles in the humoral system (e.g., immunoglobulin production) [13] |
| Select Micronutrients | Recommended Dietary Allowance [78] | Tolerable Upper Intake Levels [78] | Reported Mean Micronutrient Intakes, Min–Max [96] | ||||||
| Children a 4–8 years 9–13 years 14–18 years: M/F | Adults 19–50 years: M/F b | Older age 51 to >70 years: M/F | Children a 4–8 years 9–13 years 14–18 years | Adults 19–50 years: b | Older age 51 to >70 years | Children 4–6 years: M/F 7–9 years: M/F 10–14 years: M/F 15–18 years: M/F | Adults 19–50 years: M/F | Older age 51 to >70 years: M/F | |
| Vitamin C, mg/day | 25 45 75/65 | 90/75 | 90/75 | 650 1200 1800/1800 | 2000 | 2000 | 60–157/61–157 63–172/57–172 73–197/77–222 71–201/67–205 | 64–153/62–153 | 59–142/60–160 |
| Vitamin D, μg/day | 15 | 15 | 15–20 | 75 100 100/100 | 100 | 100 | 1.8–5.8/1.5–6.5 1.5–6.4/1.5–5.1 1.5–4.8/1.2–4.5 1.8–7.5/1.5–7.1 | 1.6–10.9/1.2–10.1 | 0.7–15.0/0.7–12.9 |
| Vitamin A, μg/day | 400 600 900/700 | 900/700 | 900/700 | 900 1700 2800/2800 | 3000 | 3000 | 400–1100/400–1200 400–1300/400–1100 400–2400/300–2300 400–1800/300–1600 | 500–2200/500–2000 | 500–2500/400–2300 |
| Vitamin E, mg/day | 7 11 15 | 15 | 15 | 300 600 800 | 1000 | 1000 | 5.3–9.8/5.1–9.8 6.3–11.2/5.9–13.3 5.9–14.5/5.6–18.1 6.8–20.8/6.0–15.5 | 3.3–17.7/4.2–16.1 | 6.3–13.7/6.7–13.7 |
| Vitamin B6, mg/day | 0.6 1.0 1.3/1.2 | 1.3 | 1.7/1.5 | 40 60 80 | 100 | 100 | 1.3–1.8/1.0–1.9 1.2–2.5/1.1–1.9 1.2–2.8/1.1–2.7 1.5–3.1/1.2–2.5 | 1.6–3.5/1.3–2.1 | 1.2–3.0/1.2–2.9 |
| Vitamin B12, μg/day | 1.2 1.8 2.4 | 2.4 | 2.4 | ND | ND | ND | 2.7–5.3/2.6–5.0 3.6–5.5/2.2–5.3 3.2–11.8/2.2–11.1 4.9–7.5/3.5–5.2 | 1.9–9.3/1.0–8.8 | 3.1–8.2/2.5–7.5 |
| Folate, μg/day | 200 300 400 | 300-400 | 400 | 400 600 800 | 1000 | 1000 | 120–256/109–199 144–290/133–264 149–428/140–360 190–365/154–298 | 203–494/131–392 | 139–343/121–335 |
| Zinc, mg/day | 5 8 11/9 | 11/8 | 11/8 | 12 23 34 | 40 | 40 | 6.0–9.2/5.3–8.9 7.0–10.9/6.4–9.4 7.0–14.6/6.1–13.9 9.3–15.2/6.4–11.0 | 8.6–14.6/6.7–10.7 | 7.5–12.3/6.7–11.2 |
| Iron, mg/day | 10 8 11/15 | 8/18 | 8 | 40 40 45 | 45 | 45 | 7.3–10.6/6.8–10.6 8.4–11.8/7.7–11.8 9.2–19.4/7.7–14.8 10.2–19.0/7.8–14.0 | 10.6–26.9/8.2–22.2 | 10.2–25.2/8.5–20.9 |
| Copper, μg/day | 440 700 890 | 900 | 900 | 3000 5000 8000 | 10,000 | 10,000 | 700–2200/700–2000 900–2800/800–2600 800–2900/700-2800 1200–3400/800–2100 | 1100–2300/1000–2200 | 1100–1900/900–1900 |
| Selenium, μg/day | 30 40 55 | 55 | 55 | 150 280 400 | 400 | 400 | 23–61/24–61 27–41/26–58 29–110/28–104 39–59/30–38 | 36–73/31–54 | 39–62/34–55 |
| Micronutrient | Impact of Deficiency | Impact of Supplementation |
|---|---|---|
| Vitamin C | Increased oxidative damage [104] Increased incidence and severity of pneumonia and other infections [71,104] Decreased resistance to infection and cancer, decreased delayed-type hypersensitivity response, impaired wound healing [49] | Antioxidant properties protect leukocytes and lymphocytes from oxidative stress [14] Older people: possible reduction in incidence and duration of pneumonia [71] Children: reduced duration and severity of common cold symptoms [105]; improved outcomes in pneumonia, malaria and diarrheal symptoms [9] |
| Vitamin D | Increased susceptibility to infections, especially RTI [71] Increased morbidity and mortality, increased severity of infections, reduced number of lymphocytes, reduced lymphoid organ weight [49] Increased risk of autoimmune diseases (e.g., type 1 diabetes, multiple sclerosis, systemic lupus erythematosus, rheumatoid arthritis) [14] | Reduced acute respiratory tract infections if deficient [71] |
| Vitamin A | Affects many immune functions, including number and killing activity of NK cells, neutrophil function, macrophage ability to phagocytose pathogens, growth and differentiation of B cells, decreasing number and distribution of T cells, etc. [14] Increased susceptibility to infections (e.g., diarrhea, RTI, measles, malaria) [14,71] | Children: Reduces all-cause mortality, diarrhea incidence and mortality, and measles incidence and morbidity in deficient children (6 month to 5 years) [14,71]; decreased risk of morbidity and mortality from infectious diseases [77] Not beneficial in pneumonia [14] |
| Vitamin E | Deficiency rare in humans [49] Impairs both humoral and cell-mediated aspects of adaptive immunity, including B and T cell function [14] | Older people: reduced RTI [71] |
| Vitamin B6 | Lymphocytopenia, reduced lymphoid tissue weight, reduced responses to mitogens, general deficiencies in cell-mediated immunity, lowered antibody responses [49] | |
| Vitamin B12 | Depressed immune responses (e.g., delayed-type hypersensitivity response, T-cell proliferation) [49] * | |
| Folate | Depressed immune responses (e.g., delayed-type hypersensitivity response, T-cell proliferation) [49] * | |
| Zinc | Decreased lymphocyte number and function, particularly T cells, increased thymic atrophy, altered cytokine production that contributes to oxidative stress and inflammation [14] Increased bacterial, viral and fungal infections (particularly diarrhea and pneumonia) [71] and diarrheal and respiratory morbidity [49] Increased thymic atrophy and consequent risk of infection [97] | Restoration of thymulin activity, increased numbers of cytotoxic T cells, reduced numbers of activated T helper cells (which can contribute to autoimmunity), increased natural killer cell cytotoxicity, reduced incidence of infections [14] Children: reduction in duration of diarrhea and incidence of pneumonia in at-risk children >6 month, but not in children 2–6 month [71]; reduced duration and severity of common cold symptoms [108]; improved outcomes in pneumonia, malaria and diarrheal symptoms [9] |
| Iron | Reduced capacity for adequate immune response (decreased delayed-type hypersensitivity response, mitogen responsiveness, NK cell activity), decreased lymphocyte bactericidal activity, lower interleukin-6 levels [49] | May enhance or protect from infection with bacteria, viruses, fungi and protozoa depending on the level of iron [71] May theoretically enhance immunity to infectious diseases, but untargeted supplementation may increase availability of iron for pathogen growth and virulence and increase susceptibility to malaria and bacterial sepsis in particular [71] Children: potential detrimental effects in iron-replete children [14] |
| Copper | Abnormally low neutrophil levels [14] Potentially increased susceptibility to infection [14] | Children: increased ability of certain white blood cells to engulf pathogens if deficient [14] Reduced antibody production in response to influenza vaccine with chronic high doses in healthy young men [14] |
| Selenium | Impaired humoral and cell-mediated immunity [14] Increased viral virulence [14,71] Suppression of immune function, increased cancer incidence and cardiomyopathy with chronic deficiency [49] Children: increased risk of respiratory infections in the first 6 weeks of life [71] | Improves cell-mediated immunity and enhances immune response to viruses in deficient individuals, but may worsen allergic asthma and impair the immune response to parasites [14] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Maggini, S.; Pierre, A.; Calder, P.C. Immune Function and Micronutrient Requirements Change over the Life Course. Nutrients 2018, 10, 1531. https://doi.org/10.3390/nu10101531
Maggini S, Pierre A, Calder PC. Immune Function and Micronutrient Requirements Change over the Life Course. Nutrients. 2018; 10(10):1531. https://doi.org/10.3390/nu10101531
Chicago/Turabian StyleMaggini, Silvia, Adeline Pierre, and Philip C. Calder. 2018. "Immune Function and Micronutrient Requirements Change over the Life Course" Nutrients 10, no. 10: 1531. https://doi.org/10.3390/nu10101531
APA StyleMaggini, S., Pierre, A., & Calder, P. C. (2018). Immune Function and Micronutrient Requirements Change over the Life Course. Nutrients, 10(10), 1531. https://doi.org/10.3390/nu10101531





