Food Byproducts as Sustainable Ingredients for Innovative and Healthy Dairy Foods
Abstract
:1. Introduction
2. Materials and Methods
3. Byproducts Used as Novel Ingredients in Dairy Foods
4. Approaches in the Application of Food Byproducts in the Dairy Industry
4.1. Technological Applications of Food Byproducts in Dairy Formulations
4.1.1. Use of Byproducts as Antioxidants
4.1.2. Use of Byproducts as Antimicrobials
4.1.3. Action Against Dairy Food Spoilage Microorganisms
4.1.4. Action Against Foodborne Pathogens in Dairy Foods
4.1.5. Use of Byproducts as Colorants
4.1.6. Use of Byproducts as Texturizing Agents
4.2. Health-Promoting Applications of Food Byproducts in Dairy Formulations
4.2.1. Use of Byproducts in the Development of Functional Dairy Foods Containing Polyphenols
4.2.2. Use of Byproducts in the Development of Functional Dairy Foods Containing Dietary Fiber
4.2.3. Use of Animal Origin Byproducts in the Development of Functional Dairy Foods
5. Sensory Challenges and Consumer Perspective of Using Byproducts in Dairy Foods
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Sharma, S.K.; Bansal, S.; Mangal, M.; Dixit, A.K.; Gupta, R.K.; Mangal, A.K. Utilization of food processing by-products as dietary, functional, and novel fiber: A review. Crit. Rev. Food Sci. Nutr. 2015, 56, 1647–1661. [Google Scholar] [CrossRef] [PubMed]
- Godfray, H.C.J.; Beddington, J.R.; Crute, I.R.; Haddad, L.; Lawrence, D.; Muir, J.F.; Pretty, J.; Robinson, S.; Thomas, S.M.; Toulmin, C. Food security: The challenge of feeding 9 billion people. Science 2010, 327, 812–818. [Google Scholar] [CrossRef] [PubMed]
- Fan, S.; Brzeska, J. Sustainable food security and nutrition: Demystifying conventional beliefs. Glob. Food Sec. 2016, 11, 11–16. [Google Scholar] [CrossRef]
- Granato, D.; Branco, G.F.; Cruz, A.G.; de Faria, J.A.F.; Shah, N.P. Probiotic dairy products as functional foods. Compr. Rev. Food Sci. Food Saf. 2010, 9, 455–470. [Google Scholar] [CrossRef]
- Menrad, K. Market and marketing of functional food in Europe. J. Food Eng. 2003, 56, 181–188. [Google Scholar] [CrossRef]
- Urala, N.; Lähteenmäki, L. Consumers’ changing attitudes towards functional foods. Food Qual. Prefer. 2007, 18, 1–12. [Google Scholar] [CrossRef]
- FAO. Definitional Framework of Food Loss; Working Paper; Food and Agriculture Organization of the United Nations: Rome, Italy, 2014; pp. 1–18. [Google Scholar]
- A Report by the High Level Panel of Experts on Food Security and Nutrition of the Committee on World Food Security: HLPE Food Losses and Waste in the Context of Sustainable Food Systems; Hlpe Report: Rome, Italy, 2014.
- FAO FAOSTAT. Available online: http://www.fao.org/faostat (accessed on 18 December 2017).
- Pasotti, L.; Zucca, S.; Casanova, M.; Micoli, G.; Cusella De Angelis, M.G.; Magni, P. Fermentation of lactose to ethanol in cheese whey permeate and concentrated permeate by engineered Escherichia coli. BMC Biotechnol. 2017, 17, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Kaur, D.; Wani, A.A.; Singh, D.P.; Sogi, D.S. Shelf Life Enhancement of Butter, Ice-Cream, and Mayonnaise by Addition of Lycopene. Int. J. Food Prop. 2011, 14, 1217–1231. [Google Scholar] [CrossRef]
- Vital, A.C.P.; Croge, C.; Gomes-da-Costa, S.M.; Matumoto-Pintro, P.T. Effect of addition of Agaricus blazei mushroom residue to milk enriched with Omega-3 on the prevention of lipid oxidation and bioavailability of bioactive compounds after in vitro gastrointestinal digestion. Int. J. Food Sci. Technol. 2017, 52, 1483–1490. [Google Scholar] [CrossRef]
- Servili, M.; Rizzello, C.G.; Taticchi, A.; Esposto, S.; Urbani, S.; Mazzacane, F.; Di Maio, I.; Selvaggini, R.; Gobbetti, M.; Di Cagno, R. Functional milk beverage fortified with phenolic compounds extracted from olive vegetation water, and fermented with functional lactic acid bacteria. Int. J. Food Microbiol. 2011, 147, 45–52. [Google Scholar] [CrossRef] [PubMed]
- Demirci, T.; Aktaş, K.; Sözeri, D.; Öztürk, H.İ.; Akın, N. Rice bran improve probiotic viability in yoghurt and provide added antioxidative benefits. J. Funct. Foods 2017, 36, 396–403. [Google Scholar] [CrossRef]
- Akalın, A.S.; Unal, G.; Dinkci, N.; Hayaloglu, A.A. Microstructural, textural, and sensory characteristics of probiotic yogurts fortified with sodium calcium caseinate or whey protein concentrate. J. Dairy Sci. 2012, 95, 3617–3628. [Google Scholar] [CrossRef] [PubMed]
- Saffon, M.; Richard, V.; Jiménez-Flores, R.; Gauthier, S.; Britten, M.; Pouliot, Y. Behavior of heat-denatured whey:buttermilk protein aggregates during the yogurt-making process and their influence on set-type yogurt properties. Foods 2013, 2, 444–459. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.; Mccarthy, J.; Wang, G.; Liu, Y.; Guo, M. Physiochemical properties, microstructure, and probiotic survivability of nonfat goats’ milk yogurt using heat-treated whey protein concentrate as fat replacer. J. Food Sci. 2015, 80, M788–M794. [Google Scholar] [CrossRef] [PubMed]
- Ozcan, T.; Yilmaz-Ersan, L.; Akpinar-Bayizit, A.; Delikanli, B. Antioxidant properties of probiotic fermented milk supplemented with chestnut flour (C astanea sativa Mill). J. Food Process. Preserv. 2016. [Google Scholar] [CrossRef]
- Do Espírito Santo, A.P.; Cartolano, N.S.; Silva, T.F.; Soares, F.A.; Gioielli, L.A.; Perego, P.; Converti, A.; Oliveira, M.N. Fibers from fruit by-products enhance probiotic viability and fatty acid profile and increase CLA content in yoghurts. Int. J. Food Microbiol. 2012, 154, 135–144. [Google Scholar] [CrossRef] [PubMed]
- Issar, K.; Sharma, P.C.; Gupta, A. Utilization of apple pomace in the preparation of fiber-enriched acidophilus yoghurt. J. Food Process. Preserv. 2016. [Google Scholar] [CrossRef]
- Sah, B.N.; Vasiljevic, T.; McKechnie, S.; Donkor, O.N. Physicochemical, textural and rheological properties of probiotic yogurt fortified with fibre-rich pineapple peel powder during refrigerated storage. LWT-Food Sci. Technol. 2016, 65, 978–986. [Google Scholar] [CrossRef]
- Sah, B.N.; Vasiljevic, T.; McKechnie, S.; Donkor, O.N. Effect of refrigerated storage on probiotic viability and the production and stability of antimutagenic and antioxidant peptides in yogurt supplemented with pineapple peel. J. Dairy Sci. 2015, 98, 5905–5916. [Google Scholar] [CrossRef] [PubMed]
- Espírito-Santo, A.P.; Lagazzo, A.; Sousa, A.L.O.P.; Perego, P.; Converti, A.; Oliveira, M.N. Rheology, spontaneous whey separation, microstructure and sensorial characteristics of probiotic yoghurts enriched with passion fruit fiber. Food Res. Int. 2013, 50, 224–231. [Google Scholar] [CrossRef]
- Do Espírito Santo, A.P.; Perego, P.; Converti, A.; Oliveira, M.N. Influence of milk type and addition of passion fruit peel powder on fermentation kinetics, texture profile and bacterial viability in probiotic yoghurts. LWT-Food Sci. Technol. 2012, 47, 393–399. [Google Scholar] [CrossRef] [Green Version]
- Perina, N.P.; Granato, D.; Hirota, C.; Cruz, A.G.; Bogsan, C.S.B.; Oliveira, M.N. Effect of vegetal-oil emulsion and passion fruit peel-powder on sensory acceptance of functional yogurt. Food Res. Int. 2015, 70, 134–141. [Google Scholar] [CrossRef]
- Chen, W.; Duizer, L.; Corredig, M.; Goff, H.D. Addition of soluble soybean polysaccharides to dairy products as a source of dietary fiber. J. Food Sci. 2010, 75, 478–484. [Google Scholar] [CrossRef] [PubMed]
- Aliakbarian, B.; Casale, M.; Paini, M.; Casazza, A.; Lanteri, S.; Perego, P. Production of a novel fermented milk fortified with natural antioxidants and its analysis by NIR spectroscopy. Food Sci. Technol. Int. 2015, 62, 376–383. [Google Scholar] [CrossRef]
- Frumento, D.; do Espirito Santo, A.P.; Aliakbarian, B.; Casazza, A.A.; Gallo, M.; Converti, A.; Perego, P. Development of milk fermented with Lactobacillus acidophilus fortified with Vitis vinifera marc flour. Food Technol. Biotechnol. 2013, 51, 370–375. [Google Scholar]
- Tseng, A.; Zhao, Y. Wine grape pomace as antioxidant dietary fibre for enhancing nutritional value and improving storability of yogurt and salad dressing. Food Chem. 2013, 138, 356–365. [Google Scholar] [CrossRef] [PubMed]
- Dos Santos, K.M.O.; de Oliveira, I.C.; Lopes, M.A.C.; Cruz, A.P.G.; Buriti, F.C.A.; Cabral, L.M. Addition of grape pomace extract to probiotic fermented goat milk: The effect on phenolic content, probiotic viability and sensory acceptability. J. Sci. Food Agric. 2017, 97, 1108–1115. [Google Scholar] [CrossRef] [PubMed]
- Hashim, I.B.; Khalil, A.H.; Afifi, H.S. Quality characteristics and consumer acceptance of yogurt fortified with date fiber. J. Dairy Sci. 2009, 92, 5403–5407. [Google Scholar] [CrossRef] [PubMed]
- Nontasan, S.; Moongngarm, A.; Deeseenthum, S. Application of Functional Colorant Prepared from Black Rice Bran in Yogurt. APCBEE Procedia 2012, 2, 62–67. [Google Scholar] [CrossRef]
- Sandoval-Castilla, O.; Lobato-Calleros, C.; Aguirre-Mandujano, E.; Vernon-Carter, E.J. Microstructure and texture of yogurt as influenced by fat replacers. Int. Dairy J. 2004, 14, 151–159. [Google Scholar] [CrossRef]
- Yi, T.; Huang, X.; Pan, S.; Wang, L. Physicochemical and functional properties of micronized jincheng orange by-products (Citrus sinensis Osbeck) dietary fiber and its application as a fat replacer in yogurt. Int. J. Food 2014, 86, 565–572. [Google Scholar] [CrossRef] [PubMed]
- Sendra, E.; Kuri, V.; Fernández-López, J.; Sayas-Barberá, E.; Navarro, C.; Pérez-Alvarez, J.A. Viscoelastic properties of orange fiber enriched yogurt as a function of fiber dose, size and thermal treatment. LWT-Food Sci. Technol. 2010, 43, 708–714. [Google Scholar] [CrossRef]
- García-Pérez, F.J.; Lario, Y.; Fernández-López, J.; Sayas, E.; Pérez-Alvarez, J.A.; Sendra, E. Effect of orange fiber addition on yogurt color during fermentation and cold storage. Color. Res. Appl. 2005, 30, 457–463. [Google Scholar] [CrossRef]
- El-Said, M.M.; Haggag, H.F.; Fakhr El-Din, H.M.; Gad, A.S.; Farahat, A.M. Antioxidant activities and physical properties of stirred yoghurt fortified with pomegranate peel extracts. Ann. Agric. Sci. 2014, 59, 207–212. [Google Scholar] [CrossRef]
- Bertolino, M.; Belviso, S.; Dal Bello, B.; Ghirardello, D.; Giordano, M.; Rolle, L.; Gerbi, V.; Zeppa, G. Influence of the addition of different hazelnut skins on the physicochemical, antioxidant, polyphenol and sensory properties of yogurt. LWT-Food Sci. Technol. 2015, 63, 1145–1154. [Google Scholar] [CrossRef] [Green Version]
- Ersöz, E.; Kınık, Ö.; Yerlikaya, O.; Açu, M. Effect of phenolic compounds on characteristics of strained yoghurts produced from sheep milk. Afr. J. Agric. Res. 2011, 6, 5351–5359. [Google Scholar] [CrossRef]
- Ghorbanzade, T.; Jafari, S.M.; Akhavan, S.; Hadavi, R. Nano-encapsulation of fish oil in nano-liposomes and its application in fortification of yogurt. Food Chem. 2017, 216, 146–152. [Google Scholar] [CrossRef] [PubMed]
- Zhong, J.; Yang, R.; Cao, X.; Liu, X.; Qin, X. Improved physicochemical properties of yogurt fortified with fish oil/γ-oryzanol by nanoemulsion technology. Molecules 2018, 23, 56. [Google Scholar] [CrossRef] [PubMed]
- Sanz, T.; Salvador, A.; Jiménez, A.; Fiszman, S.M. Yogurt enrichment with functional asparagus fibre. Effect of fibre extraction method on rheological properties, colour, and sensory acceptance. Eur. Food Res. Technol. 2008, 227, 1515–1521. [Google Scholar] [CrossRef]
- Chouchouli, V.; Kalogeropoulos, N.; Konteles, S.J.; Karvela, E.; Makris, D.P.; Karathanos, V.T. Fortification of yoghurts with grape (Vitis vinifera) seed extracts. LWT-Food Sci. Technol. 2013, 53, 522–529. [Google Scholar] [CrossRef]
- Karnopp, A.R.; Oliveira, K.G.; de Andrade, E.F.; Postingher, B.M.; Granato, D. Optimization of an organic yogurt based on sensorial, nutritional, and functional perspectives. Food Chem. 2017, 233, 401–411. [Google Scholar] [CrossRef] [PubMed]
- Marchiani, R.; Bertolino, M.; Belviso, S.; Giordano, M.; Ghirardello, D.; Torri, L.; Piochi, M.; Zeppa, G. Yogurt enrichment with grape pomace: Effect of grape cultivar on physicochemical, microbiological and sensory properties. J. Food Qual. 2016, 39, 77–89. [Google Scholar] [CrossRef]
- Jridi, M.; Souissi, N.; Salem, M.B.; Ayadi, M.A.; Nasri, M.; Azabou, S. Tunisian date (Phoenix dactylifera L.) by-products: Characterization and potential effects on sensory, textural and antioxidant properties of dairy desserts. Food Chem. 2015, 188, 8–15. [Google Scholar] [CrossRef] [PubMed]
- Crizel, T. de M.; Araujo, R.R. de; Rios, A. de O.; Rech, R.; Flôres, S.H. Orange fiber as a novel fat replacer in lemon ice cream. Food Sci. Technol. 2014, 34, 332–340. [Google Scholar] [CrossRef]
- Çam, M.; Erdoǧan, F.; Aslan, D.; Dinç, M. Enrichment of functional properties of ice cream with pomegranate by-products. J. Food Sci. 2013, 78, 1543–1550. [Google Scholar] [CrossRef]
- Rizk, E.M.; El-Kady, A.T.; El-Bialy, A.R. Charactrization of carotenoids (lyco-red) extracted from tomato peels and its uses as natural colorants and antioxidants of ice cream. Ann. Agric. Sci. 2014, 59, 53–61. [Google Scholar] [CrossRef]
- Hwang, J.Y.; Shyu, Y.S.; Hsu, C.K. Grape wine lees improves the rheological and adds antioxidant properties to ice cream. LWT-Food Sci. Technol. 2009, 42, 312–318. [Google Scholar] [CrossRef]
- Nadeem, M.; Mahud, A.; Imran, M.; Khalique, A. Enhancement of the oxidative stability of whey butter through almond (Prunus dulcis) peel extract. J. Food Process. Preserv. 2014, 39, 591–598. [Google Scholar] [CrossRef]
- Abid, Y.; Azabou, S.; Jridi, M.; Khemakhem, I.; Bouaziz, M.; Attia, H. Storage stability of traditional Tunisian butter enriched with antioxidant extract from tomato processing by-products. Food Chem. 2017, 233, 476–482. [Google Scholar] [CrossRef] [PubMed]
- Lucera, A.; Costa, C.; Marinelli, V.; Saccotelli, M.A.; Alessandro, M.; Nobile, D.; Conte, A. Fruit and vegetable by-products to fortify spreadable cheese. Antioxidants 2018. [Google Scholar] [CrossRef] [PubMed]
- Terpou, A.; Bekatorou, A.; Bosnea, L.; Kanellaki, M.; Ganatsios, V.; Koutinas, A.A. Wheat bran as prebiotic cell immobilisation carrier for industrial functional Feta-type cheese making: Chemical, microbial and sensory evaluation. Biocatal. Agric. Biotechnol. 2018, 13, 75–83. [Google Scholar] [CrossRef]
- Erbay, Z.; Koca, N. Effects of whey or maltodextrin addition during production on physical quality of white cheese powder during storage. J. Dairy Sci. 2015, 98, 8391–8404. [Google Scholar] [CrossRef] [PubMed]
- Shan, B.; Cai, Y.-Z.; Brooks, J.D.; Corke, H. Potential application of spice and herb extracts as natural preservatives in cheese. J. Med. Food 2011, 14, 284–290. [Google Scholar] [CrossRef] [PubMed]
- Göksel Saraç, M.; Dogan, M. Incorporation of dietary fiber concentrates from fruit and vegetable wastes in butter: Effects on physicochemical, textural, and sensory properties. Eur. Food Res. Technol. 2016, 242, 1331–1342. [Google Scholar] [CrossRef]
- Felix da Silva, D.; Matumoto-Pintro, P.T.; Bazinet, L.; Couillard, C.; Britten, M. Effect of commercial grape extracts on the cheese-making properties of milk. J. Dairy Sci. 2015, 98, 1552–1562. [Google Scholar] [CrossRef] [PubMed]
- Marchiani, R.; Bertolino, M.; Ghirardello, D.; McSweeney, P.L.H.; Zeppa, G. Physicochemical and nutritional qualities of grape pomace powder-fortified semi-hard cheeses. J. Food Sci. Technol. 2015, 53, 1585–1596. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ayala-Zavala, J.F.; Vega-Vega, V.; Rosas-Domínguez, C.; Palafox-Carlos, H.; Villa-Rodriguez, J.A.; Siddiqui, M.W.; Dávila-Aviña, J.E.; González-Aguilar, G.A. Agro-industrial potential of exotic fruit byproducts as a source of food additives. Food Res. Int. 2011, 44, 1866–1874. [Google Scholar] [CrossRef]
- Codex Alimentarius General Standard for Food Additives Codex Stan 192–1995; Food and Agriculture Organization of the United Nations: Rome, Italy, 2017.
- Serra, M.; Trujillo, A.J.; Pereda, J.; Guamis, B.; Ferragut, V. Quantification of lipolysis and lipid oxidation during cold storage of yogurts produced from milk treated by ultra-high pressure homogenization. J. Food Eng. 2008, 89, 99–104. [Google Scholar] [CrossRef]
- Marsili, R. Flavors and off-flavors in dairy foods. In Encyclopedia of Dairy Sciences, 2nd ed.; Fuquay, J.W., Fox, P.F., McSweeney, P.L.H., Eds.; Elsevier Ltd.: Atlanta, GA, USA, 2011; pp. 533–551. [Google Scholar]
- Grażyna, C.; Hanna, C.; Adam, A.; Magdalena, B.M. Natural antioxidants in milk and dairy products. Int. J. Dairy Technol. 2017, 70, 165–178. [Google Scholar] [CrossRef]
- Hedegaard, R.V.; Kristensen, D.; Nielsen, J.H.; Frøst, M.B.; Østdal, H.; Hermansen, J.E.; Kröger-Ohlsen, M.; Skibsted, L.H. Comparison of descriptive sensory analysis and chemical analysis for oxidative changes in milk. J. Dairy Sci. 2006, 89, 495–504. [Google Scholar] [CrossRef]
- Herr, B. Types and functions of additives in dairy products. In Encyclopedia of Dairy Sciences; Fuquay, J.W., Fox, P.F., McSweeney, P.L.H., Eds.; Elsevier Ltd.: Atlanta, GA, USA, 2011; pp. 34–40. ISBN 978-0-12-374407-4. [Google Scholar]
- Frede, E. Properties and analysis. In Encyclopedia of Dairy Sciences; Fuquay, J.W., Fox, P.F., McSweeney, P.L.H., Eds.; Elsevier Ltd.: Rockford, IL, USA, 2011; pp. 506–514. [Google Scholar]
- Phan-Thi, H.; Durand, P.; Prost, M.; Prost, E.; Wach??, Y. Effect of heat-processing on the antioxidant and prooxidant activities of b-carotene from natural and synthetic origins on red blood cells. Food Chem. 2016, 190, 1137–1144. [Google Scholar] [CrossRef] [PubMed]
- McSweeney, P.L.H.; Sousa, M.J. Biochemical pathways for the production of flavour compounds in cheeses during ripening: A review. Lait 2000, 80, 293–324. [Google Scholar] [CrossRef] [Green Version]
- Fox, P.F. Proteolysis During Cheese Manufacture and Ripening. J. Dairy Sci. 1989, 72, 1379–1400. [Google Scholar] [CrossRef]
- Ribeiro Júnior, J.C.; de Oliveira, A.M.; Silva, F.D.G.; Tamanini, R.; de Oliveira, A.L.M.; Beloti, V. The main spoilage-related psychrotrophic bacteria in refrigerated raw milk. J. Dairy Sci. 2018, 101, 75–83. [Google Scholar] [CrossRef] [PubMed]
- Deeth, H.; Fitz-Gerald, C.H. Lipolytic enzymes and hydrolitic rancidity in milk and milk products. In Advanced Dairy Chemistry Vol 2: Lipids; Fox, P.F., McSweeney, P.L.H., Eds.; Springer: New York, NY, USA, 2006; pp. 481–556. [Google Scholar]
- Deeth, H.C. Lipolysis and Hydrolitic Rancidity. In Encyclopedia of Dairy Sciences; Fuquay, J.W., Fox, P.F., McSweeney, P.L.H., Eds.; Elsevier Ltd.: Atlanta, GA, USA, 2011; pp. 721–726. [Google Scholar]
- Forsythe, S.J. The Microbiology of Safe Food, 2nd ed.; Wiley Blackwell: Hoboken, NJ, USA, 2010. [Google Scholar]
- Baglinière, F.; Jardin, J.; Gaucheron, F.; de Carvalho, A.F.; Vanetti, M.C.D. Proteolysis of casein micelles by heat-stable protease secreted by Serratia liquefaciens leads to the destabilisation of UHT milk during its storage. Int. Dairy J. 2017, 68, 38–45. [Google Scholar] [CrossRef]
- Lu, M.; Wang, N.S. Spoilage of milk and dairy products. In The Microbiological Quality of Food-Foodborne Spoilers; Bevilacqua, A., Corbo, M.R., Milena, S., Eds.; Woodhead Publishing Limited: Cambridge, UK, 2017; pp. 151–178. [Google Scholar]
- Baglinière, F.; Tanguy, G.; Salgado, R.L.; Jardin, J.; Rousseau, F.; Robert, B.; Harel-Oger, M.; Vanetti, M.C.D.; Gaucheron, F. Ser2 from Serratia liquefaciens L53: A new heat stable protease able to destabilize UHT milk during its storage. Food Chem. 2017, 229, 104–110. [Google Scholar] [CrossRef] [PubMed]
- Rehman, S.U.; Farkye, N.Y. Phosphatases. In Encyclopedia of Dairy Sciences; Fuquay, J.W., Fox, P.F., McSweeney, P.L.H., Eds.; Elsevier Ltd.: Atlanta, GA, USA, 2011; pp. 314–318. [Google Scholar]
- Brisabois, A.; Lafarge, V.; Brouillaud, A.; de Buyser, M.L.; Collette, C.; Garin-Bastuji, B.; Thorel, M.F. Pathogenic organisms in milk and milk products: The situation in France and in Europe. Rev. Sci. Tech. 1997, 16, 467–471. [Google Scholar]
- De Buyser, M.L.; Dufour, B.; Maire, M.; Lafarge, V. Implication of milk and milk products in food-borne diseases in France and in different industrialised countries. Int. J. Food Microbiol. 2001, 67, 1–17. [Google Scholar] [CrossRef]
- Yoon, Y.; Lee, S.; Choi, K.H. Microbial benefits and risks of raw milk cheese. Food Control. 2016, 63, 201–215. [Google Scholar] [CrossRef]
- Donnelly, C.W. Growth and survival or microbial pathogens in cheese. In Cheese: Chemistry, Physics and Microbiology; Fox, P.F., McSweeney, P.L.H., Cogan, M.T., Guinee, T.P., Eds.; Elsevier: Atlanta, GA, USA, 2004; pp. 541–559. [Google Scholar]
- Fuselli, F.; Guarino, C. Preservatives in cheeses. In Handbook of Cheese in Health: Production, Nutrition and Medical Sciences; Human Health Handbooks no. 6; Preedy, V.R., Watson, R.R., Patel, V.B., Eds.; Wageningen Academic Publishers: Wageningen, The Netherlands, 2013. [Google Scholar]
- Morgan, D.; Newman, C.P.; Hutchinson, D.N.; Walker, A.M.; Rowe, B.; Majid, F. Verotoxin producing Escherichia coli 0 157 infections associated with the consumption of yoghurt. Epidemiol. Infect. 1993, 111, 181–187. [Google Scholar] [CrossRef] [PubMed]
- Cheng, H.Y.; Yu, R.C.; Chou, C.C. Increased acid tolerance of Escherichia coli O157:H7 as affected by acid adaptation time and conditions of acid challenge. Food Res. Int. 2003, 36, 49–56. [Google Scholar] [CrossRef]
- Ultee, A.; Bennik, M.H.J.; Moezelaar, R. The phenolic hydroxyl group of carvacrol is essential for action against the food-borne pathogen Bacillus cereus. Appl. Environ. Microbiol. 2002, 68, 1561–1568. [Google Scholar] [CrossRef] [PubMed]
- Daglia, M. Polyphenols as antimicrobial agents. Curr. Opin. Biotechnol. 2012, 23, 174–181. [Google Scholar] [CrossRef] [PubMed]
- Cowan, M.M. Plant products as antimicrobial agents. Clin. Microbiol. Rev. 1999, 12, 564–582. [Google Scholar] [CrossRef] [PubMed]
- Wilson, P.D.; Brocklehurst, T.F.; Arino, S.; Thuault, D.; Jakobsen, M.; Lange, M.; Farkas, J.; Wimpenny, J.W.T.; Van Impe, J.F. Modelling microbial growth in structured foods: Towards a unified approach. Int. J. Food Microbiol. 2002, 73, 275–289. [Google Scholar] [CrossRef]
- Tayel, A.A.; Hussein, H.; Sorour, N.M.; El-Tras, W.F. Foodborne pathogens prevention and sensory attributes enhancement in processed cheese via flavoring with plant extracts. J. Food Sci. 2015, 80, M2886–M2891. [Google Scholar] [CrossRef] [PubMed]
- Marinho, M.T.; Zielinski, A.A.; Demiate, I.M.; dos Bersot, L.S.; Granato, D.; Nogueira, A. Ripened semihard cheese covered with lard and dehydrated rosemary (Rosmarinus officinalis L.) leaves: Processing, characterization, and quality traits. J. Food Sci. 2015, 80, 2045–2054. [Google Scholar] [CrossRef] [PubMed]
- Llamas, N.E.; Garrido, M.; Di Nezio, M.S.; Band, B.S.F. Second order advantage in the determination of amaranth, sunset yellow FCF and tartrazine by UV-vis and multivariate curve resolution-alternating least squares. Anal. Chim. Acta 2009, 655, 38–42. [Google Scholar] [CrossRef] [PubMed]
- Martins, N.; Roriz, C.L.; Morales, P.; Barros, L.; Ferreira, I.C.F.R. Food colorants: Challenges, opportunities and current desires of agro-industries to ensure consumer expectations and regulatory practices. Trends Food Sci. Technol. 2016, 52, 1–15. [Google Scholar] [CrossRef] [Green Version]
- Santos-Buelga, C.; Mateus, N.; De Freitas, V. Anthocyanins. Plant pigments and beyond. J. Agric. Food Chem. 2014, 62, 6879–6884. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Amaya, D.B. Natural food pigments and colorants. Curr. Opin. Food Sci. 2016, 7, 20–26. [Google Scholar] [CrossRef]
- Teixeira, A.; Baenas, N.; Dominguez-Perles, R.; Barros, A.; Rosa, E.; Moreno, D.A.; Garcia-Viguera, C. Natural bioactive compounds from winery by-products as health promoters: A review. Int. J. Mol. Sci. 2014, 15, 15638–15678. [Google Scholar] [CrossRef] [PubMed]
- Giusti, M.M.; Wrolstad, R.E. Acylated anthocyanins from edible sources and their applications in food systems. Biochem. Eng. J. 2003, 14, 217–225. [Google Scholar] [CrossRef]
- Prata, E.R.; Oliveira, L.S. Fresh coffee husks as potential sources of anthocyanins. LWT-Food Sci. Technol. 2007, 40, 1555–1560. [Google Scholar] [CrossRef]
- FDA Color Additives Listed for Use in Food. Available online: https://www.fda.gov/ForIndustry/ColorAdditives/ColorAdditivesinSpecificProducts/InFood/ucm130054.htm (accessed on 26 June 2018).
- Prudencio, I.D.; Prudêncio, E.S.; Gris, E.F.; Tomazi, T.; Bordignon-Luiz, M.T. Petit suisse manufactured with cheese whey retentate and application of betalains and anthocyanins. LWT-Food Sci. Technol. 2008, 41, 905–910. [Google Scholar] [CrossRef]
- Saunders, A.B. Dairy desserts. In Encyclopedia of Dairy Sciences, 2nd ed.; Fuquay, J., Fox, P.F., McSweeney, P.L., Eds.; Elsevier Ltd.: Atlanta, GA, USA, 2011; pp. 905–912. [Google Scholar]
- Krog, N. Emulsifiers. In Encyclopedia of Dairy Sciences, 2nd ed.; Fuquay, J., Fox, P.F., McSweeney, P.L.H., Eds.; Elsevier Ltd.: Atlanta, GA, USA, 2011; pp. 61–71. [Google Scholar]
- Müller-Maatsch, J.; Bencivenni, M.; Caligiani, A.; Tedeschi, T.; Bruggeman, G.; Bosch, M.; Petrusan, J.; Van Droogenbroeck, B.; Elst, K.; Sforza, S. Pectin content and composition from different food waste streams in memory. Food Chem. 2016, 201, 37–45. [Google Scholar] [CrossRef] [PubMed]
- De Oliveira, C.F.; Giordani, D.; Gurak, P.D.; Cladera-Olivera, F.; Marczak, L.D.F. Extraction of pectin from passion fruit peel using moderate electric field and conventional heating extraction methods. Innov. Food Sci. Emerg. Technol. 2015, 29, 201–208. [Google Scholar] [CrossRef]
- Min, B.; Lim, J.; Ko, S.; Lee, K.G.; Lee, S.H.; Lee, S. Environmentally friendly preparation of pectins from agricultural byproducts and their structural/rheological characterization. Bioresour. Technol. 2011, 102, 3855–3860. [Google Scholar] [CrossRef] [PubMed]
- Jeong, H.S.; Kim, H.Y.; Ahn, S.H.; Oh, S.C.; Yang, I.; Choi, I.G. Optimization of enzymatic hydrolysis conditions for extraction of pectin from rapeseed cake (Brassica napus L.) using commercial enzymes. Food Chem. 2014, 157, 332–338. [Google Scholar] [CrossRef] [PubMed]
- US General Accounting Office Food Safety. Improvements needed in overseing the safety of dietary supplements and “functional foods”. Available online: https://www.gao.gov/products/GAO/RCED-00-156 (accessed on 31 July 2018).
- Hasler, C.M. Functional foods: Benefits, concerns and challenges—A position paper from the American Council on Science and Health. J. Nutr. 2002, 132, 3772–3781. [Google Scholar] [CrossRef] [PubMed]
- Mozaffarian, D. Dietary and policy priorities for cardiovascular disease, diabetes, and obesity—A comprehensive review. Circulation 2016, 133, 187–225. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization Cardiovascular Disease. Available online: http://www.who.int/cardiovascular_diseases/en/ (accessed on 31 July 2018).
- Naczk, M.; Shahidi, F. Extraction and analysis of phenolics in food. J. Chromatogr. A 2004, 1054, 95–111. [Google Scholar] [CrossRef]
- Costa, C.; Tsatsakis, A.; Mamoulakis, C.; Teodoro, M.; Briguglio, G.; Caruso, E.; Tsoukalas, D.; Margina, D.; Dardiotis, E.; Kouretas, D.; et al. Current evidence on the effect of dietary polyphenols intake on chronic diseases. Food Chem. Toxicol. 2017, 110, 286–299. [Google Scholar] [CrossRef] [PubMed]
- Bahadoran, Z.; Mirmiran, P.; Azizi, F. Dietary polyphenols as potential neutraceuticals in management of diabetes: A review. J. Diabetes Metab. Disord. 2013, 12, 43. [Google Scholar] [CrossRef] [PubMed]
- Scalbert, A.; Manach, C.; Morand, C.; Rémésy, C.; Jiménez, L. Dietary polyphenols and the prevention of diseases. Crit. Rev. Food Sci. Nutr. 2005, 45, 287–306. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Jiménez, J.; Neveu, V.; Vos, F.; Scalbert, A. Identification of the 100 richest dietary sources of polyphenols: An application of the Phenol-Explorer database. Eur. J. Clin. Nutr. 2010, 64, S112–S120. [Google Scholar] [CrossRef] [PubMed]
- Boskou, D. Sources of natural phenolic antioxidants. Trends Food Sci. Technol. 2006, 17, 505–512. [Google Scholar] [CrossRef]
- Tomas-Barberan, F.; Gil-Izquierdo, A.; Moreno, D. Bioavailability and metabolism of phenolic compounds and glucosinolates. In Designing Functional Foods; Elsevier: Atlanta, GA, USA, 2009; pp. 194–229. ISBN 978-1-84569-432-6. [Google Scholar]
- Shahidi, F.; Naczk, M. Phenolics in Food and Nutraceuticals; CRC Press: Boca Raton, FL, USA, 2006; ISBN 0203508734. [Google Scholar]
- Haslam, E.; Lilley, T.H. Natural astringency in foodstuffs—A molecular interpretation. Crit. Rev. Food Sci. Nutr. Nat. 1988, 37–41. [Google Scholar] [CrossRef]
- Tseng, A.; Zhao, Y. Effect of different drying methods and storage time on the retention of bioactive compounds and antibacterial activity of wine grape pomace (Pinot Noir and Merlot). J. Food Sci. 2012, 77, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Del Rio, D.; Costa, L.G.; Lean, M.E.J.; Crozier, A. Polyphenols and health: What compounds are involved? Nutr. Metab. Cardiovasc. Dis. 2010, 20, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Jakobek, L. Interactions of polyphenols with carbohydrates, lipids and proteins. Food Chem. 2015, 175, 556–567. [Google Scholar] [CrossRef] [PubMed]
- McKee, L.H.; Latner, T.A. Underutilized sources of dietary fiber: A review. Plant. Foods Hum. Nutr. 2000, 55, 285–304. [Google Scholar] [CrossRef] [PubMed]
- European Food Safety Authority Scientific opinion on dietary reference values for carbohydrates and dietary fibre. EFSA J. 2010, 8, 1–77. [CrossRef]
- Álvarez, E.E.; González, P. La fibra dietética. Nutr. Hosp. 2006, 21, 61–72. [Google Scholar]
- AECOSAN Report of the Scientific Committee of the Spanish Agency for Consumer Affairs, Food Safety and Nutrition (AECOSAN) about objectives as well as nutritional and physical activity recommendations to tackle obesity in the framework of the NAOS Strategy. Rev. Com. Cient. 2014, 19, 95–209.
- Pereira, M.; O’Reilly, E.; Augustsson, K.; Fraser, G.; Goldbourt, U.; Heitmann, B.; Hallmans, G.; Knekt, P.; Liu, S.; Pietinen, P.; et al. Dietary Fiber and Risk of Coronary Heart Disease. Arch. Intern. Med. 2004, 164, 370–376. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lindström, J.; Peltonen, M.; Eriksson, J.G.; Louheranta, A.; Fogelholm, M.; Uusitupa, M.; Tuomilehto, J. High-fibre, low-fat diet predicts long-term weight loss and decreased type 2 diabetes risk: The Finnish Diabetes Prevention Study. Diabetologia 2006, 49, 912–920. [Google Scholar] [CrossRef] [PubMed]
- Peters, U.; Sinha, R.; Chatterjee, N.; Subar, A.F.; Ziegler, R.G.; Kulldorff, M.; Bresalier, R.; Weissfeld, J.L.; Flood, A.; Schatzkin, A.; et al. Dietary fibre and colorectal adenoma in a colorectal cancer early detection programme. Lancet 2003, 361, 1491–1495. [Google Scholar] [CrossRef]
- European Parliament and Council Regulation (EC) No 1924/2006 on Nutrition and Health Claims Made on Foods. 2006. Available online: https://eur-lex.europa.eu/LexUriServ/LexUriServ.do?uri=OJ:L:2007:012:0003:0018:EN:PDF (accessed on 31 July 2018).
- Guillon, F.; Champ, M. Structural and physical properties of dietary fibres, and consequences of processing on human physiology. Food Res. Int. 2000, 33, 233–245. [Google Scholar] [CrossRef]
- Grabitske, H.A.; Slavin, J.L. Gastrointestinal effects of low-digestible carbohydrates. Crit. Rev. Food Sci. Nutr. 2009, 49, 327–360. [Google Scholar] [CrossRef] [PubMed]
- Marafon, A.P.; Sumi, A.; Alcântara, M.R.; Tamime, A.Y.; Nogueira de Oliveira, M. Optimization of the rheological properties of probiotic yoghurts supplemented with milk proteins. LWT-Food Sci. Technol. 2011, 44, 511–519. [Google Scholar] [CrossRef]
- Sodini, I.; Lucas, A.; Tissier, J.P.; Corrieu, G. Physical properties and microstructure of yoghurts supplemented with milk protein hydrolysates. Int. Dairy J. 2005, 15, 29–35. [Google Scholar] [CrossRef]
- Yadav, H.; Jain, S.; Sinha, P.R. Production of free fatty acids and conjugated linoleic acid in probiotic dahi containing Lactobacillus acidophilus and Lactobacillus casei during fermentation and storage. Int. Dairy J. 2007, 17, 1006–1010. [Google Scholar] [CrossRef]
- Do Espírito Santo, A.P.; Silva, R.C.; Soares, F.A.S.M.; Anjos, D.; Gioielli, L.A.; Oliveira, M.N. Açai pulp addition improves fatty acid profile and probiotic viability in yoghurt. Int. Dairy J. 2010, 20, 415–422. [Google Scholar] [CrossRef]
- Jayathilakan, K.; Sultana, K.; Radhakrishna, K.; Bawa, A.S. Utilization of byproducts and waste materials from meat, poultry and fish processing industries: A review. J. Food Sci. Technol. 2012, 49, 278–293. [Google Scholar] [CrossRef] [PubMed]
- Childs, N.M.; Childs, N.M.; Poryzees, G.H. Foods that help prevent disease: Consumer attitudes and public policy implications. J. Consum. Mark. 1997, 14, 433–447. [Google Scholar] [CrossRef]
- Verbeke, W. Functional foods: Consumer willingness to compromise on taste for health? Food Qual. Prefer. 2006, 17, 126–131. [Google Scholar] [CrossRef]
- Urala, N.; Liisa, L. Attitudes behind consumers’ willingness to use functional foods. Food Qual. Prefer. 2004, 15, 793–803. [Google Scholar] [CrossRef]
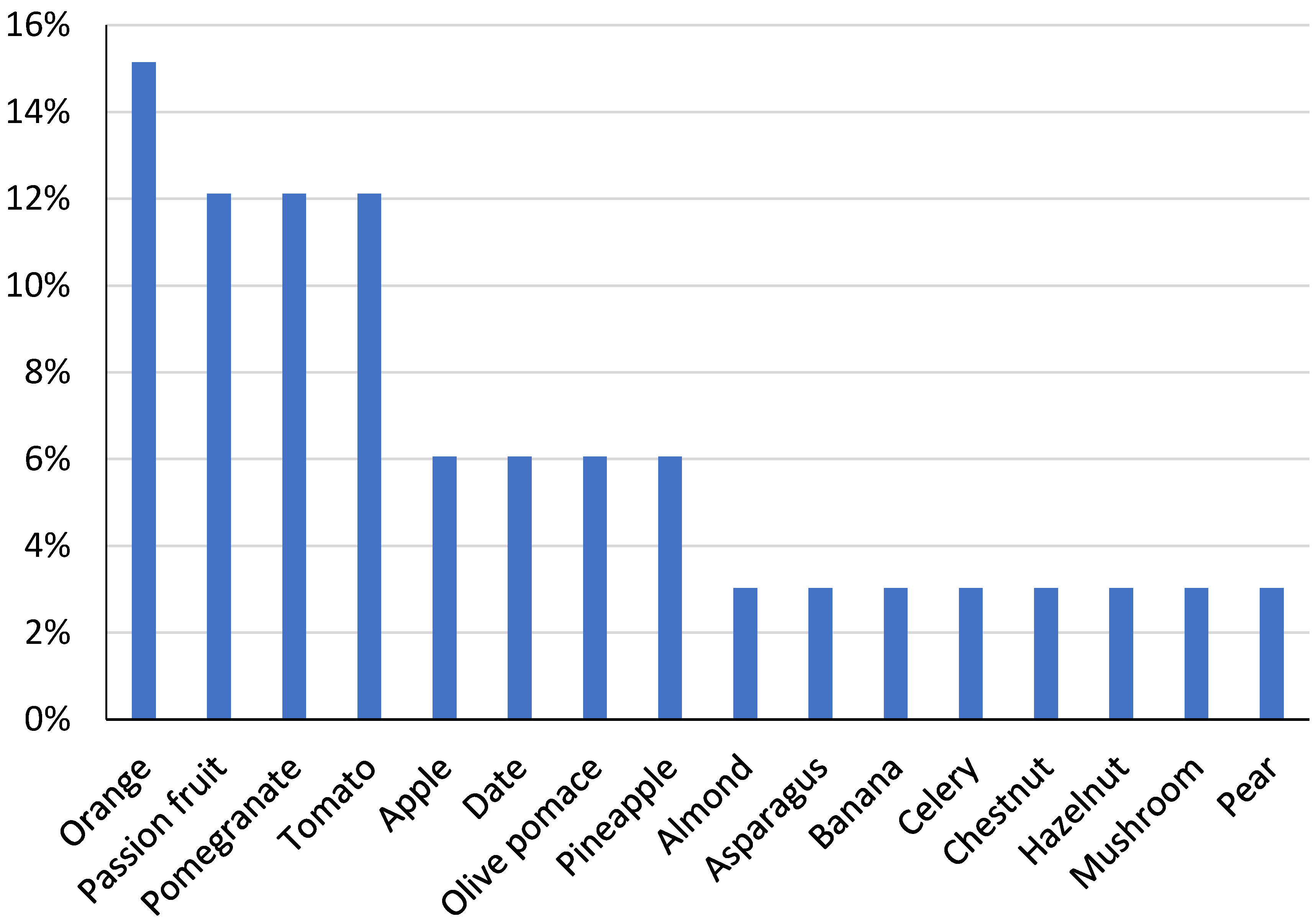
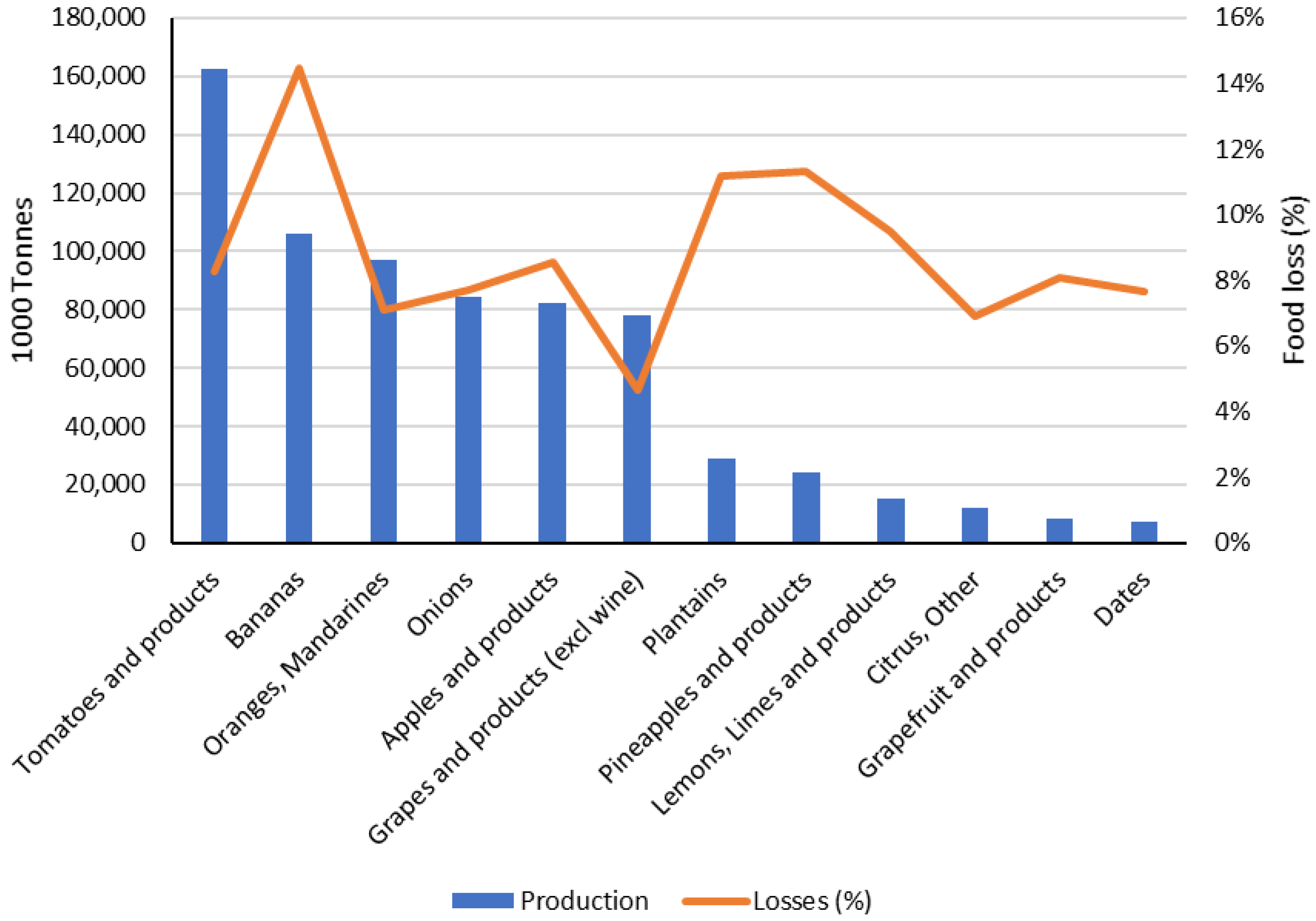
| Dairy Product | Food Industry | Byproduct | Doses | Function | Reference |
|---|---|---|---|---|---|
| Dairy beverage | Vegetable | Mushroom residue | 1, 2 and 3 g/kg | Technological (antioxidant) Health-promoting (source of phenols) | Vital et al., 2017 [12] |
| Olive vegetable water | 100 mg/L to 200 mg/L | Health-promoting (source of phenols, probiotic protection) | Servili et al., 2011 [13] | ||
| Fermented milk | Cereal | Rice bran | 1% to 3% | Health-promoting (source of fiber and phenols, probiotic protection) | Demirci et al., 2017 [14] |
| Dairy | Whey protein | 2% | Technological (texturizing agent) Health-promoting (source of protein) | Akalin et al., 2012 [15] | |
| Whey protein and buttermilk | 0% to 100% replacement of skim milk powder | Technological (texturizing agent) Health-promoting (source of protein) | Saffon et al., 2013 [16] | ||
| Whey protein | 8% to 14% | Technological (texturizing agent) Health-promoting (source of protein, probiotic protection) | Zhang et al., 2015 [17] | ||
| Fruit | Chestnut flour | 2% | Health-promoting (source of phenols, probiotic protection) | Ozcan et al., 2016 [18] | |
| Apple | 1% | Health-promoting (source of fiber, probiotic protection) | Do Espírito Santo et al., 2012 [19] | ||
| Apple pomace | 2.5% to 10% | Health-promoting (source of fiber) | Issar et al., 2016 [20] | ||
| Banana | 1% | Health-promoting (source of fiber, probiotic protection) | Do Espírito Santo et al., 2012 [19] | ||
| Pineapple peel powder | 1% | Technological (texturizing agent) | Sah et al., 2016 [21] | ||
| Pineapple peel powder | 1% | Health-promoting (probiotic protection) | Sah et al., 2015 [22] | ||
| Passion fruit peels | 1% | Technological (texturizing agent) | Espírito-Santo et al., 2012 [23] | ||
| Passion fruit peels | 0.7% | Health-promoting (source of fiber) | Do Espírito Santo et al., 2012 [24] | ||
| Vegetable | Passion fruit | 1% | Health-promoting (source of fiber, probiotic protection) | Do Espírito Santo et al., 2012 [19] | |
| Passion fruit peels | 1% | Health-promoting (source of fiber) | Perina et al., 2015 [25] | ||
| Okara | 3% to 10% | Health-promoting (source of fiber) | Chen et al., 2010 [26] | ||
| Olive pomace | 100 mg/L TPC | Health-promoting (source of phenols, probiotic protection) | Aliakbarian et al., 2015 [27] | ||
| Winery | Wine pomace extract | 100 mg/L TPC | Health-promoting (source of phenols, probiotic protection) | Aliakbarian et al., 2015 [27] | |
| Grape marc flour | 10, 20 and 50 g/L | Health-promoting (source of phenols, probiotic protection) | Aliakbarian et al., 2013 [28] | ||
| Wine pomace extract and flour | 1% to 3% 1% to 2% | Technological (antioxidant, colorant) Health-promoting (source of fiber and phenols) | Tseng and Zhao 2013 [29] | ||
| Wine pomace extract | 788 mg GAE/100 g | Health-promoting (source of phenols, probiotic protection) | Dos Santos et al., 2017 [30] | ||
| Wine pomace flour | 10, 20 and 50 g/L | Health-promoting (source of phenols, probiotic protection) | Frumento et al., 2013 [28] | ||
| Yogurt | Cereal | Wheat bran | 1.5% | Health-promoting (source of fiber) | Hashim et al., 2009 [31] |
| Rice bran | 0.2% to 0.6% | Technological (colorant) | Nontasan et al. 2012 [32] | ||
| Dairy | Whey protein | 3.3, 5 and 10 g/L | Technological (texturizing agent) Health-promoting (source of protein) | Sandoval-Castilla et al., 2004 [33] | |
| Fruit | Date byproducts | 1.5% to 4.5% | Health-promoting (source of fiber) | Hashim et al., 2009 [31] | |
| Orange peels, pulp, seed powders | 1% to 3% | Technological (texturizing agent) | Yi et al., 2014 [34] | ||
| Orange byproducts | 0.2 to 1 g/mL | Technological (texturizing agent) | Sendra et al., 2010 [35] | ||
| Orange albedo, flavedo and pulp powders | 0.6% to 1% | Health-promoting (source of fiber) | García-Pérez et al., 2005 [36] | ||
| Pomegranate peel extract | 5% to 35% | Health-promoting (source of phenols) | El Said et al., 2014 [37] | ||
| Hazelnut skin powder | 3% to 6% | Health-promoting (source of fiber) | Bertolino et al., 2015 [38] | ||
| Pomegranate seed | 25 mg/L | Technological (antioxidant) | Ersöz et al., 2011 [39] | ||
| Marine | Fish oil | 15 mL/100 g | Health-promoting (source of omega-3) | Ghorbanzade et al., 2017 [40] | |
| Fish oil | 13 g/100 g | Health-promoting (source of omega-3) | Zhong et al., 2018 [41] | ||
| Vegetable | Asparagus byproducts | 1% | Health-promoting (source of fiber) | Sanz et al., 2008 [42] | |
| Winery | Grape seed extract | 100 mg/150 g | Health-promoting (source of phenols) | Chouchouli et al., 2013 [43] | |
| Grape skin flour | 0.167 to 1 g/100 g | Health-promoting (source of phenols) | Karnopp et al., 2017 [44] | ||
| Grape skin flour | 60 g/kg | Health-promoting (source of phenols) | Marchiani et al., 2016 [45] | ||
| Grape seed | 25 mg/L | Technological (antioxidant) | Ersöz et al., 2011 [39] | ||
| Dairy dessert | Fruit | Date byproduct | 0.5, 1 and 2 ratio dried date powder/date syrup | Technological (texturizing agent)Health-promoting (source of phenols) | Jridi et al., 2015 [46] |
| Vegetable | Okara | 3% to 10% | Health-promoting (source of fiber) | Chen et al., 2010 [26] | |
| Ice-cream | Fruit | Orange peels, pulp, seed powders | 1% to 1.5% | Technological (texturizing agent) | Crizel et al., 2014 [47] |
| Pomegranate peels | 0.1% and 0.4% | Health-promoting (source of phenols) | Çam et al., 2013 [48] | ||
| Vegetable | Lycopene from tomato byproducts | 70 mg/kg | Technological (antioxidant, colorant, antimicrobial) | Kaur et al., 2011 [11] | |
| Carotenoids from tomato peels | 1% to 5% | Technological (antioxidant, colorant) | Rizk et al., 2014 [49] | ||
| Winery | Grape wine lees | 50, 100 and 150 g/kg | Health-promoting (source of phenols) | Hwang et al., 2009 [50] | |
| Butter | Fruit | Almond peel extract | 100 ppm to 400 ppm | Technological (antioxidant) | Nadeem et al., 2014 [51] |
| Vegetable | Lycopene from tomato byproducts | 20 mg/kg | Technological (antioxidant, colorant, antimicrobial) | Kaur et al., 2011 [11] | |
| Tomato processing byproduct | 400 and 800 mg/kg | Technological (antioxidant) | Abid et al., 2017 [52] | ||
| Cheese | Cereal | Corn bran | 5% | Health-promoting (source of phenols) | Lucera et al., 2018 [53] |
| Wheat bran | 10 g/500g | Health-promoting (probiotic protection) | Terpou et al., 2018 [54] | ||
| Dairy | Fluid whey | Water substitution | Technological (texturizing agent) | Erbay et al., 2015 [55] | |
| Fruit | Pomegranate peel | 100 mL/25 g | Technological (antioxidant, antimicrobial) | Shan et al., 2011 [56] | |
| Orange byproduct fibers | 3% to 5% | Technological (texturizing agent) | Saraç and Dogan 2016 [57] | ||
| Pear stones | 3% to 5% | Technological (texturizing agent) | Saraç and Dogan 2016 [57] | ||
| Vegetable | Spinach | 3% to 5% | Technological (texturizing agent) | Saraç and Dogan 2016 [57] | |
| Celery byproduct fibers | 3% to 5% | Technological (texturizing agent) | Saraç and Dogan 2016 [57] | ||
| Okara | 1% to 4% | Health-promoting (source of fiber) | Chen et al., 2010 [26] | ||
| Tomato peels | 5% | Health-promoting (source of phenols) | Lucera et al., 2018 [53] | ||
| Broccoli stems and leaves | 5% | Health-promoting (source of phenols) | Lucera et al., 2018 [53] | ||
| Artichoke external leaves | 5% | Health-promoting (source of phenols) | Lucera et al., 2018 [53] | ||
| Winery | Grape seed | 100 mL/25 g | Technological (antioxidant, antimicrobial) | Shan et al., 2011 [56] | |
| Wine pomace, skin and seed extracts | 0.1, 0.2 and 0.3 wt/vol | Health-promoting (source of phenols) | Da Silva et al., 2015 [58] | ||
| Wine pomace flour | 0.8 and 1.6 w/w | Health-promoting (source of phenols) | Marchiani et al., 2015 [59] | ||
| Grape pomace | 5% | Health-promoting (source of phenols) | Lucera et al., 2018 [53] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Iriondo-DeHond, M.; Miguel, E.; Del Castillo, M.D. Food Byproducts as Sustainable Ingredients for Innovative and Healthy Dairy Foods. Nutrients 2018, 10, 1358. https://doi.org/10.3390/nu10101358
Iriondo-DeHond M, Miguel E, Del Castillo MD. Food Byproducts as Sustainable Ingredients for Innovative and Healthy Dairy Foods. Nutrients. 2018; 10(10):1358. https://doi.org/10.3390/nu10101358
Chicago/Turabian StyleIriondo-DeHond, Maite, Eugenio Miguel, and María Dolores Del Castillo. 2018. "Food Byproducts as Sustainable Ingredients for Innovative and Healthy Dairy Foods" Nutrients 10, no. 10: 1358. https://doi.org/10.3390/nu10101358
APA StyleIriondo-DeHond, M., Miguel, E., & Del Castillo, M. D. (2018). Food Byproducts as Sustainable Ingredients for Innovative and Healthy Dairy Foods. Nutrients, 10(10), 1358. https://doi.org/10.3390/nu10101358







