Mediterranean Diet and Other Dietary Patterns in Primary Prevention of Heart Failure and Changes in Cardiac Function Markers: A Systematic Review
Abstract
1. Introduction
2. Materials and Methods
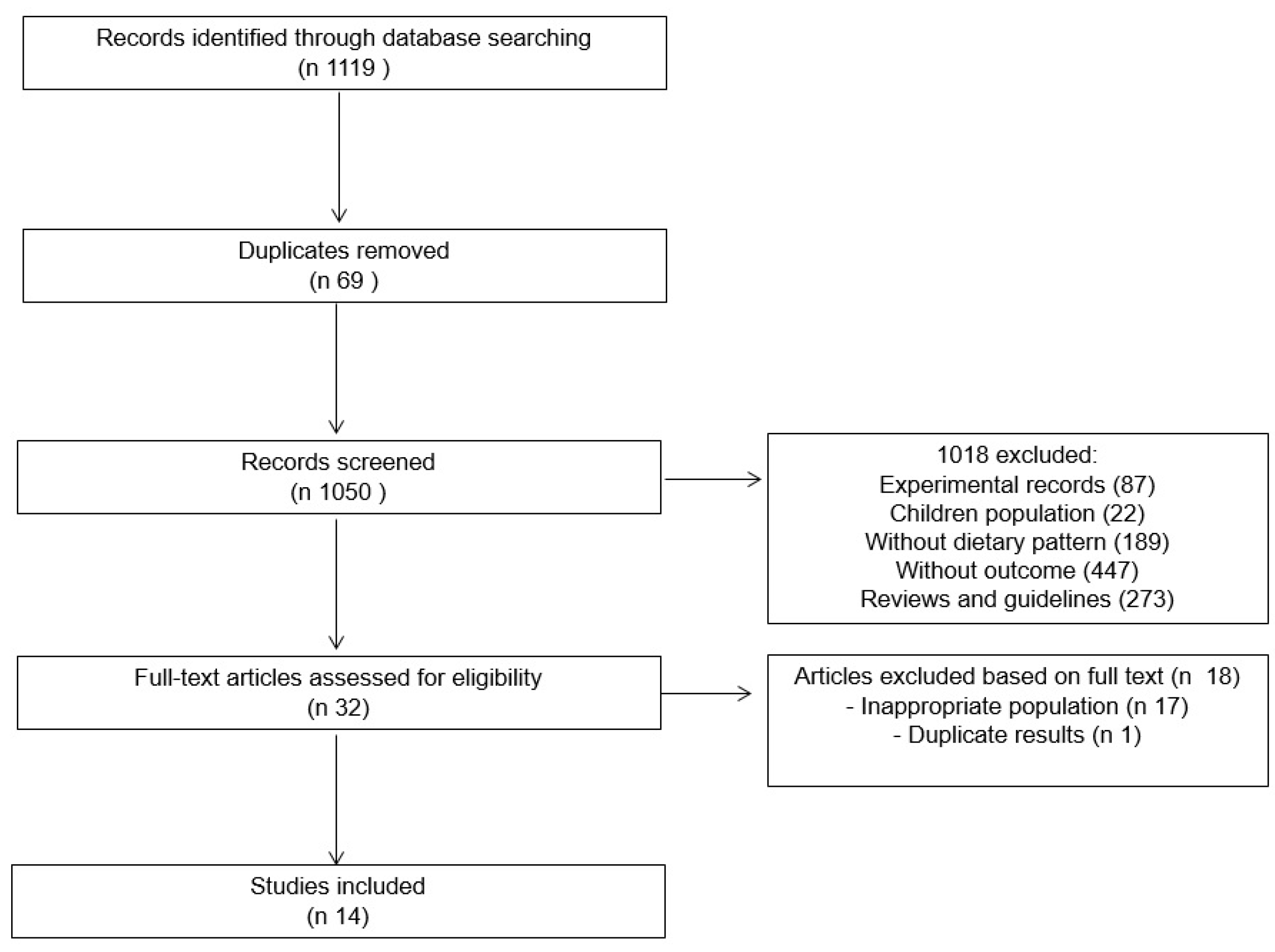
2.1. Search Strategy and Study Selection
2.2. Eligibility Criteria
2.3. Data Extraction
2.4. Assessment of Bias Risk and Study Quality
3. Results
3.1. Descriptions of Studies
3.2. Quality and Publication Bias Assessment
3.3. Mediterranean Diet
3.3.1. Definition in the Included Studies
3.3.2. Mediterranean Diet and HF
3.4. DASH Diet
3.4.1. Definition in the Included Studies
3.4.2. DASH Diet and HF
3.5. Other Diets and Markers of Cardiac Function
4. Discussion
5. Limitations
6. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- McMurray, J.J.; Adamopoulos, S.; Anker, S.D.; Auricchio, A.; Böhm, M.; Dickstein, K.; Falk, V.; Filippatos, G.; Fonseca, C.; Gomez-Sanchez, M.A.; et al. ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure 2012: The task force for the diagnosis and treatment of acute and chronic heart failure 2012 of the European society of cardiology. Developed in collaboration with the Heart Failure Association (HFA) of the ESC. Eur. Heart J. 2012, 33, 1787–1847. [Google Scholar] [CrossRef] [PubMed]
- Jhund, P.S.; MacIntyre, K.; Simpson, C.R.; Lewsey, J.D.; Stewart, S.; Redpath, A.; Chalmers, J.W.; Capewell, S.; McMurray, J.J. Long-term trends in first hospitalization for heart failure and subsequent survival between 1986 and 2003: A population study of 5.1 million people. Circulation 2009, 119, 515–523. [Google Scholar] [CrossRef] [PubMed]
- Lassus, J.; Gayat, E.; Mueller, C.; Peacock, W.F.; Spinar, J.; Harjola, V.P.; van Kimmenade, R.; Pathak, A.; Mueller, T.; Metra, M.; et al. Incremental value of biomarkers to clinical variables for mortality prediction in acutely decompensated heart failure: The Multinational Observational Cohort on Acute Heart Failure (MOCA) study. Int. J. Cardiol. 2013, 168, 2186–2194. [Google Scholar] [CrossRef] [PubMed]
- Jacob, J.; Miró, Ò.; Herrero, P.; Martín-Sánchez, F.J.; Gil, V.; Tost, J.; Aguirre, A.; Escoda, R.; Alquézar, A.; Andueza, J.A.; et al. Predicting short-term mortality in patients with acute exacerbation of chronic heart failure: The EAHFE-3D scale. Med. Intensiv. 2016, 40, 348–355. [Google Scholar] [CrossRef] [PubMed]
- Simão, A.F.; Précoma, D.B.; de Andrade, J.P.; Filho, H.C.; Saraiva, J.F.K.; Oliveira, G.M.M. I diretriz brasileira de prevenção cardiovascular. Arq. Bras. Cardiol. 2014, 102, 420–431. [Google Scholar] [CrossRef] [PubMed]
- Lim, S.S.; Vos, T.; Flaxman, A.D.; Danaei, G.; Shibuya, K.; Adair-Rohani, H.; AlMazroa, M.A.; Amann, M.; Anderson, H.R.; Andrews, K.G.; et al. A comparative risk assessment of burden of disease and injury attributable to 67 risk factors and risk factor clusters in 21 regions, 1990–2010: A systematic analysis for the Global Burden of Disease Study 2010. Lancet 2012, 380, 2224–2260. [Google Scholar] [CrossRef]
- Ravera, A.; Carubelli, V.; Sciatti, E.; Bonadei, I.; Gorga, E.; Cani, D.; Vizzardi, E.; Metra, M.; Lombardi, C. Nutrition and cardiovascular disease: Finding the perfect recipe for cardiovascular health. Nutrients 2016, 8, 363. [Google Scholar] [CrossRef] [PubMed]
- Anand, S.S.; Hawkes, C.; de Souza, R.J.; Mente, A.; Dehghan, M.; Nugent, R.; Zulyniak, M.A.; Weis, T.; Bernstein, A.M.; Krauss, R.M.; et al. Food consumption and its impact on cardiovascular disease: Importance of solutions focused on the globalized food system. J. Am. Coll. Cardiol. 2015, 66, 1590–1614. [Google Scholar] [CrossRef] [PubMed]
- Vogt, T.M.; Appel, L.J.; Obarzanek, E.V.A.; Moore, T.J.; Vollmer, W.M.; Svetkey, L.P.; Sacks, F.M.; Bray, G.A.; Cutler, J.A.; Windhauser, M.M.; et al. Dietary approaches to stop hypertension: Rationale, design, and methods. DASH Collaborative Research Group. J. Am. Diet. Assoc. 1999, 99, S12–S18. [Google Scholar] [CrossRef]
- Willett, W.C.; Sacks, F.; Trichopoulou, A.; Drescher, G.; Ferro-Luzzi, A.; Helsing, E.; Trichopoulos, D. Mediterranean diet pyramid: A cultural model for healthy eating. Am. J. Clin. Nutr. 1995, 61, 1402S–1406S. [Google Scholar] [CrossRef] [PubMed]
- Appel, L.J.; Moore, T.J.; Obarzanek, E.; Vollmer, W.M.; Svetkey, L.P.; Sacks, F.M.; Bray, G.A.; Vogt, T.M.; Cutler, J.A.; Windhauser, M.M.; et al. A clinical trial of the effects of dietary patterns on blood pressure. DASH Collaborative Research Group. N. Engl. J. Med. 1997, 336, 1117–1124. [Google Scholar] [CrossRef] [PubMed]
- Sacks, F.M.; Svetkey, L.P.; Vollmer, W.M.; Appel, L.J.; Bray, G.A.; Harsha, D.; Obarzanek, E.; Conlin, P.R.; Miller, E.R., III; Simons-Morton, D.G. Effects on blood pressure of reduced dietary sodium and the Dietary Approaches to Stop Hypertension (DASH) Diet. DASH-Sodium Collaborative Research Group. N. Engl. J. Med. 2001, 344, 3–10. [Google Scholar] [CrossRef] [PubMed]
- Appel, L.J.; Champagne, C.M.; Harsha, D.W.; Cooper, L.S.; Obarzanek, E.; Elmer, P.J.; Stevens, V.J.; Vollmer, W.M.; Lin, P.H.; Svetkey, L.P.; et al. Effects of comprehensive lifestyle modification on blood pressure control: Main results of the premier clinical trial. JAMA 2003, 289, 2083–2093. [Google Scholar] [PubMed]
- He, J.; Ogden, L.G.; Bazzano, L.A.; Vupputuri, S.; Loria, C.; Whelton, P.K. Risk factors for congestive heart failure in US men and women: NHANES I Epidemiologic Follow-Up Study. Arch. Intern. Med. 2001, 161, 996–1002. [Google Scholar] [CrossRef] [PubMed]
- Heckbert, S.R.; Post, W.; Pearson, G.D.; Arnett, D.K.; Gomes, A.S.; Jerosch-Herold, M.; Hundley, W.G.; Lima, J.A.; Bluemke, D.A. Traditional cardiovascular risk factors in relation to left ventricular mass, volume, and systolic function by cardiac magnetic resonance imaging: The Multi-Ethnic Study of Atherosclerosis. J. Am. Coll. Cardiol. 2006, 48, 2285–2292. [Google Scholar] [CrossRef] [PubMed]
- Levy, D.; Larson, M.G.; Vasan, R.S.; Kannel, W.B.; Ho, K.K. The progression from hypertension to congestive heart failure. JAMA 1996, 275, 1557–1562. [Google Scholar] [CrossRef] [PubMed]
- Panagiotakos, D.B.; Pitsavos, C.; Arvaniti, F.; Stefanadis, C. Adherence to the Mediterranean food pattern predicts the prevalence of hypertension, hypercholesterolemia, diabetes and obesity, among healthy adults; the accuracy of the MedDietScore. Prev. Med. 2007, 44, 335–340. [Google Scholar] [CrossRef] [PubMed]
- Panagiotakos, D.; Kalogeropoulos, N.; Pitsavos, C.; Roussinou, G.; Palliou, K.; Chrysohoou, C.; Stefanadis, C. Validation of the MedDietScore via the determination of fatty acids. Int. J. Food Sci. 2009, 60, 168–180. [Google Scholar] [CrossRef] [PubMed]
- Higgins, J.; Grren, S. Cochrane Handbook for Systematic Reviews of Interventions, Version 5.1.0 [Updated March 2011]; The Cochrane Collaboration, 2011. Available online: http://handbook.cochrane.org. (accessed on 5 October 2017).
- Moher, D.; Shamseer, L.; Clarke, M.; Ghersi, D.; Liberati, A.; Petticrew, M.; Shekelle, P.; Stewart, L.A. Preferred reporting items for systematic review and meta-analysis protocols (PRISMA-P) 2015 statement. Syst. Rev. 2015, 4, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Larsson, S.C.; Tektonidis, T.G.; Gigante, B.; Åkesson, A.; Wolk, A. Healthy lifestyle and risk of heart failure: Results from 2 prospective cohort studies. Circ. Heart Fail. 2016, 9, 2855. [Google Scholar] [CrossRef] [PubMed]
- Tuttle, K.R.; Shuler, L.A.; Packard, D.P.; Milton, J.E.; Daratha, K.B.; Bibus, D.M.; Short, R.A. Comparison of low-fat versus Mediterranean-style dietary intervention after first myocardial infarction (from The Heart Institute of Spokane Diet Intervention and Evaluation Trial). Am. J. Cardiol. 2008, 101, 1523–1530. [Google Scholar] [CrossRef] [PubMed]
- Papadaki, A.; Martínez-González, M.Á.; Alonso-Gómez, A.; Rekondo, J.; Salas-Salvadó, J.; Corella, D.; Ros, E.; Fitó, M.; Estruch, R.; Lapetra, J.; et al. Mediterranean diet and risk of heart failure: Results from the PREDIMED randomized controlled trial. Eur. J. Heart Fail. 2017, 30. [Google Scholar] [CrossRef] [PubMed]
- Fitó, M.; Estruch, R.; Salas-Salvadó, J.; Martínez-Gonzalez, M.A.; Arós, F.; Vila, J. Effect of the Mediterranean diet on heart failure biomarkers: A randomized sample from the PREDIMED trial. Eur. J. Heart Fail. 2014, 16, 543–550. [Google Scholar] [CrossRef] [PubMed]
- Lorgeril, M.; Salen, P.; Martin, J.L.; Monjaud, I.; Delaye, J.; Mamelle, N. Mediterranean diet, traditional risk factors and the rate of cardiovascular complications after myocardial infarction: Final report of the Lyon Diet Heart Study. Circulation 1999, 99, 779–785. [Google Scholar] [CrossRef] [PubMed]
- Wirth, J.; di Giuseppe, R.; Boeing, H.; Weikert, C. A Mediterranean-style diet, its components and the risk of heart failure: A prospective population-based study in a non-Mediterranean country. Eur. J. Clin. Nutr. 2016, 70, 1015–1021. [Google Scholar] [CrossRef] [PubMed]
- Chrysohoou, C.; Panagiotakos, D.B.; Aggelopoulos, P.; Kastorini, C.M.; Kehagia, I.; Pitsavos, C.; Stefanadis, C. The Mediterranean diet contributes to the preservation of left ventricular systolic function and to the long-term favorable prognosis of patients who have had an acute coronary event. Am. J. Clin. Nutr. 2010, 92, 47–54. [Google Scholar] [CrossRef] [PubMed]
- Tektonidis, T.G.; Åkesson, A.; Gigante, B.; Wolk, A.; Larsson, S.C.A. Mediterranean diet and risk of myocardial infarction, heart failure and stroke: A population-based cohort study. Atherosclerosis 2015, 243, 93–98. [Google Scholar] [CrossRef] [PubMed]
- Tektonidis, T.G.; Åkesson, A.; Gigante, B.; Wolk, A.; Larsson, S.C. Adherence to a Mediterranean diet is associated with reduced risk of heart failure in men. Eur. J. Heart Fail. 2016, 18, 253–259. [Google Scholar] [CrossRef] [PubMed]
- Del Gobbo, L.C.; Kalantarian, S.; Imamura, F.; Lemaitre, R.; Siscovick, D.S.; Psaty, B.M.; Mozaffarian, D. Contribution of major lifestyle risk factors for incident heart failure in older adults. The Cardiovascular Health Study. JACC Heart Fail. 2015, 3, 520–528. [Google Scholar] [CrossRef] [PubMed]
- Levitan, E.B.; Wolk, A.; Mittleman, M.A. Consistency with the DASH diet and incidence of heart failure. Arch. Intern. Med. 2009, 169, 851–857. [Google Scholar] [CrossRef] [PubMed]
- Levitan, E.B.; Wolk, A.; Mittleman, M.A. Relation of consistency with the dietary approaches to stop hypertension diet and incidence of heart failure in men aged 45 to 79 years. Am. J. Cardiol. 2009, 104, 1416–1420. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, H.T.; Bertoni, A.G.; Nettleton, J.A.; Bluemke, D.A.; Levitan, E.B.; Burke, G.L. DASH eating pattern is associated with favorable left ventricular function in the multi-ethnic study of atherosclerosis. J. Am. Coll. Nutr. 2012, 31, 401–407. [Google Scholar] [CrossRef] [PubMed]
- Andersson, J.; Mellberg, C.; Otten, J.; Ryberg, M.; Rinnström, D.; Larsson, C. Left ventricular remodelling changes without concomitant loss of myocardial fat after long-term dietary intervention. Int. J. Cardiol. 2016, 216, 92–96. [Google Scholar] [CrossRef] [PubMed]
- Pai, R.G.; Varadarajan, P.; Jabo, B.; Al-Mallah, M.H. Vegetarianism is independently associated with Lower risk of left ventricular diastolic dysfunction and stage B heart failure burden [abstract]. Circulation 2015, 132, A14601. [Google Scholar]
- Martinez-Gonzalez, M.A.; Fernandez-Jarne, E.; Serrano-Martinez, M.; Wright, M.; Gomez-Gracia, E. Development of a short dietary intake questionnaire for the quantitative estimation of adherence to a cardioprotective Mediterranean diet. Eur. J. Clin. Nutr. 2004, 58, 1550–1552. [Google Scholar] [CrossRef] [PubMed]
- Lorgeril, M.; Salen, P.; Caillat-Vallet, E.; Hanauer, M.T.; Barthelemy, J.C.; Mamelle, N. Control of bias in dietary trial to prevent coronary recurrences: The Lyon Diet Heart Study. Eur. J. Clin. Nutr. 1997, 51, 116–122. [Google Scholar] [CrossRef] [PubMed]
- Lorgeril, M.; Renaud, S.; Mamelle, N.; Salen, P.; Martin, J.L.; Monjaud, I. Mediterranean alpha-linolenic acid-rich 784 Mediterranean Diet and Coronary Heart Disease diet in secondary prevention of coronary heart disease. Lancet 1994, 343, 1454–1459. [Google Scholar] [CrossRef]
- Trichopoulou, A.; Kouris-Blazos, A.; Wahlqvist, M.L.; Gnardellis, C.; Lagiou, P.; Polychronopoulos, E.; Vassilakou, T.; Lipworth, L.; Trichopoulos, D. Diet and overall survival in elderly people. BMJ 1995, 311, 1457–1460. [Google Scholar] [CrossRef] [PubMed]
- Fung, T.T.; Chiuve, S.E.; McCullough, M.L.; Rexrode, K.M.; Logroscino, G.; Hu, F.B. Adherence to a DASH-style diet and risk of coronary heart disease and stroke in women. Arch. Intern. Med. 2008, 168, 713–720. [Google Scholar] [CrossRef] [PubMed]
- Gillingham, L.G.; Harris-Janz, S.; Jones, P.J. Dietary monounsaturated fatty acids are protective against metabolic syndrome and cardiovascular disease risk factors. Lipids 2011, 46, 209–228. [Google Scholar] [CrossRef] [PubMed]
- Cárdeno, A.; Sánchez-Hidalgo, M.; Alarcón-de-la-Lastra, C. An Up-date of Olive Oil Phenols in Inflammation and Cancer: Molecular Mechanisms and Clinical Implications. Curr. Med. Chem. 2013, 20, 4758–4776. [Google Scholar]
- Hu, T.; He, X.W.; Jiang, J.G.; Xu, X.L. Hydroxytyrosol and its potential therapeutic effects. J. Agric. Food Chem. 2014, 62, 1449–1455. [Google Scholar] [CrossRef] [PubMed]
- Moreno-Luna, R.; Muñoz-Hernandez, R.; Miranda, M.L.; Costa, A.F.; Jimenez-Jimenez, L.; Vallejo-Vaz, A.J.; Muriana, F.J.; Villar, J.; Stiefel, P. Olive oil polyphenols decrease blood pressure and improve endothelial function in young women with mild hypertension. Am. J. Hypertens. 2012, 25, 1299–1304. [Google Scholar] [CrossRef] [PubMed]
- Medina-Remón, A.; Estruch, R.; Tresserra-Rimbau, A.; Vallverdú-Queralt, A.; Lamuela-Raventos, R.M. The Effect of Polyphenol Consumption on Blood Pressure. Mini Rev. Med. Chem. 2012, 13, 1137–1149. [Google Scholar] [CrossRef]
- Santos, R.D.; Gagliardi, A.C.M.; Xavier, H.T.; Magnoni, C.D.; Cassani, R.; Lottenberg, A.M.P. Sociedade Brasileira de Cardiologia. I Diretriz sobre o consumo de Gorduras e Saúde Cardiovascular. Arq. Bras. Cardiol. 2013, 100, 1–40. [Google Scholar] [PubMed]
- He, J.; Ogden, L.G.; Bazzano, L.A.; Vupputuri, S.; Loria, C.; Whelton, P.K. Dietary sodium intake and incidence of congestive heart failure in overweight US men and women. Arch. Intern. Med. 2002, 162, 1619–1624. [Google Scholar] [CrossRef] [PubMed]
- Aurigemma, G.P.; Gottdiener, J.S.; Shemanski, L.; Gardin, J.; Kitzman, D. Predictive value of systolic and diastolic function for incident congestive heart failure in the elderly: The Cardiovascular Health Study. J. Am. Coll. Cardiol. 2001, 37, 1042–1048. [Google Scholar] [CrossRef]
- Bella, J.N.; Palmieri, V.; Roman, M.J.; Liu, J.E.; Welty, T.K.; Lee, E.T.; Fabsitz, R.R.; Howard, B.V.; Devereux, R.B. Mitral ratio of peak early to late diastolic filling velocity as a predictor of mortality in middleaged and elderly adults: The Strong Heart Study. Circulation 2002, 105, 1928–1933. [Google Scholar] [CrossRef] [PubMed]
- Gottdiener, J.S.; Arnold, A.M.; Aurigemma, G.P.; Polak, J.F.; Tracy, R.P.; Kitzman, D.W.; Gardin, J.M.; Rutledge, J.E.; Boineau, R.C. Predictors of congestive heart failure in the elderly: The Cardiovascular Health Study. J. Am. Coll. Cardiol. 2000, 35, 1628–1637. [Google Scholar] [CrossRef]
- Roman, M.J.; Pickering, T.G.; Schwartz, J.E.; Pini, R.; Devereux, R.B. Relation of arterial structure and function to left ventricular geometric patterns in hypertensive adults. J. Am. Coll. Cardiol. 1996, 28, 751–756. [Google Scholar] [CrossRef]
- Lim, P.H.; Allen, J.D.; Li, Y.J.; Yu, M.; Lien, L.F.; Svetkey, L.P. Blood pressure-lowering mechanisms of the DASH dietary pattern. J. Nutr. Metab. 2012, 2012. [Google Scholar] [CrossRef]
- Al-Solaiman, Y.; Jesri, A.; Zhao, Y.; Morrow, J.D.; Egan, B.M. Low-sodium DASH reduces oxidative stress and improves vascular function in salt-sensitive humans. J. Hum. Hypertens. 2009, 23, 826–835. [Google Scholar] [CrossRef] [PubMed]
- Azadbakht, L.; Surkan, P.J.; Esmaillzadeh, A.; Willett, W.C. The Dietary Approaches to Stop Hypertension eating plan affects C-reactive protein, coagulation abnormalities, and hepatic function tests among type 2 diabetic patients. J. Nutr. 2011, 141, 1083–1088. [Google Scholar] [CrossRef] [PubMed]
- Akita, S.; Sacks, F.M.; Svetkey, L.P.; Conlin, P.R.; Kimura, G. Effects of the Dietary Approaches to Stop Hypertension (DASH) diet on the pressure–natriuresis relationship. Hypertension 2003, 42, 8–13. [Google Scholar] [CrossRef] [PubMed]
- Edwards, K.M.; Wilson, K.L.; Sadja, J.; Ziegler, M.G.; Mills, P.J. Effects on blood pressure and autonomic nervous system function of a 12-week exercise or exercise plus DASH-diet intervention in individuals with elevated blood pressure. Acta Physiol. 2011, 203, 343–350. [Google Scholar] [CrossRef] [PubMed]
- Blumenthal, J.A.; Babyak, M.A.; Hinderliter, A.; Watkins, L.L.; Craighead, L.; Lin, P.H. Effects of the DASH diet al.one and in combination with exercise and weight loss on blood pressure and cardiovascular biomarkers in men and women with high blood pressure: The ENCORE study. Arch. Intern. Med. 2010, 170, 126–135. [Google Scholar] [CrossRef] [PubMed]
- Cheng, S.; Vasan, R.S. Advances in the epidemiology of heart failure and left ventricular remodeling. Circulation 2011, 124, e516–e519. [Google Scholar] [CrossRef] [PubMed]
- Brouwers, F.P.; de Boer, R.A.; van der Harst, P.; Voors, A.A.; Gansevoort, R.T.; Bakker, S.J.; Hillege, H.L.; van Veldhuisen, D.J.; van Gilst, W.H. Incidence and epidemiology of new onset heart failure with preserved vs. reduced ejection fraction in a community-based cohort: 11-year follow-up of PREVEND. Eur. Heart J. 2013, 34, 1424–1431. [Google Scholar] [CrossRef] [PubMed]
- Pilis, W.; Stec, K.; Zych, M.; Pilis, A. Health benefits and risk associated with adopting a vegetarian diet. Rocz. Panstw. Zakl. Hig. 2014, 65, 9–14. [Google Scholar] [PubMed]
- Jacobs, D.R., Jr.; Meyer, K.A.; Kushi, L.H.; Folsom, A.R. Is whole grain intake associated with reduced total and cause-specific death rates in older women? The Iowa Women’s Health Study. Am. J. Public Health 1999, 89, 322–329. [Google Scholar] [CrossRef] [PubMed]
- Jacobs, D.R., Jr.; Steffen, L.M. Nutrients, foods, and dietary patterns as exposures in research: A framework for food synergy. Am. J. Clin. Nutr. 2003, 78, 508–513. [Google Scholar]
| Study | SD | Condition or Cohort Name | N | Age | Intervention | Control | Duration of Intervention | Outcome |
|---|---|---|---|---|---|---|---|---|
| Mediterranean Diet | ||||||||
| Tuttle et al., 2008 [22] | RCT | Patients after a first myocardial infarction | 201 | Mediterranean diet: 58 ± 10 Low-fat diet: 58 ± 9 Usual diet: 57 ± 10 | Mediterranean diet or Low-fat diet | Usual diet | 2 years | HF development: Control: 3 patients Intervention: 0 patients (p < 0.25) Outcome-free survival: Control: 61 patients Intervention: 85 patients OR: 0.28 (95% 0.13 to 0.63, p < 0.002) |
| Papadaki et al., 2017 [23] | RCT | High risk of cardiovascular disease | 7403 | TMD + VOO: 67.0 ± 6.2 TMD + nuts: 66.7 ± 6.1 Low-fat: 67.3 ± 6.3 | TMD + VOO or TMD + nuts | Low-fat | 4.8 years | HF development: TMD + VOO: 29 patients TMD + nuts: 33 patients Low-fat: 32 patients TMD + VOO vs. control: HR 0.68 (CI 95% 0.41–1.13) TMD + nuts vs. control: HR 0.92 (CI 95% 0.56−1.49) |
| Fitó et al., 2014 [24] | RCT | High risk of cardiovascular disease | 930 | TMD + VOO: 66.4 ± 5.7 TMD + nuts: 66.2 ± 6.0 Low-fat: 67.6 ± 6.1 | TMD + VOO or TMD + nuts | Low-fat | 1 year | NT-proBNP (pg/mL) TMD + VOO vs. control: −70.3 (−133, −7.37) p = 0.029 TMD + nuts vs. control: −84.7 (−145, −24.5) p = 0.006 LDL (mg/dL) TMD + VOO vs. control: −8.27 (−13.9, −2.6) p = 0.004 TMD + nuts vs. control: −4.20 (−9.82, −1.42) p = 0.145 Lipoprotein (a) (mg/dL) TMD + VOO vs. control: −4.17 (−8.12, −0.23) p = 0.038 TMD + nuts vs. control: −2.62 (−6.36, −1.13) p = 0.170 |
| Lorgeril et al., 1999 [25] | RCT | Patients after a first myocardial infarction | 423 | NR | Mediterranean diet | Prudent Western-type diet | 4 years | HF development: Control: 11 patients Intervention: 6 patients Composite Outcomes(CO1:cardiac deaths and nonfatal AMI): Control: 44 patients Intervention: 14 patients (p = 0.0001) Composite Outcomes (CO2: CO1 + HF, Stroke, unstable angina, pulmonary embolism or peripheral embolism): Control: 90 patients Intervention: 27 patients (p = 0.0001) |
| Wirth et al., 2016 [26] | Cohort | European Prospective Investigation into Cancer and Nutrition (EPIC)-Potsdam | 24,008 | 35 to 65 | Mediterranean diet | NA | 8.2 years | HF development: 209 patients Q1 (0–2 points): 1 Q2 (3–4 points): 1.00 (0.70–1.43) Q3 (5–7 points): 0.66 (0.41–1.08) |
| Chrysohoou et al., 2010 [27] | Cohort | Patients after an acute coronary event | 1000 | EF < 40%: Male: 64 ± 14 Famale: 71 ± 12 EF ≥ 40%: Male: 62 ± 12 Famale: 67 ± 12 | Mediterranean diet | NA | 2 years | Left ventricular systolic dysfunction: OR 0.93 (0.88–0.99) Ventricular remodeling: OR 0.90 (0.78–1.03) Re-current events ACS: OR 0.88 (0.80–0.98) |
| Tektonidis et al., 2015 [28] | Cohort | Swedish mammography cohort | 32,921 | 48 to 83 | Mediterranean diet | NA | 10 years | HF development: Total: 1648 patients Q4: 244 patients RR 0.79 (0.68–0.93, p = 0.001) |
| Tektonidis et al., 2016 [29] | Cohort | Cohort of Swedish men | 37,308 | 45 to79 | Mediterranean diet | NA | 11 years | HF development: Total: 1269 patients Q4: 169 patients RR 0.69 (0.57–0.83, p < 0.001) HF mortality Total: 146 Q4: 16 patients RR 0.55 (0.31–0.98, p = 0.007) |
| DASH Diet | ||||||||
| Del Gobbo et al., 2015 [30] | Cohort | Cardiovascular health study of elderly patients with and without CVD | 4490 | Q1: 72.2 ± 5.3 Q5: 72.0 ± 5.1 | DASH | NA | 21 years | HF development: Total: 1380 patients Q5: 235 patients RR 1.05 (0.88–1.26, p = 0.36) |
| Levitan et al., 2009a [31] | Cohort | Swedish mammography cohort | 36,019 | 48–83 | DASH | NA | 7 years | HF development: Total: 443 patients Q4: 89 patients RR 0.63 (0.48–0.81, p < 0.001) |
| Levitan et al., 2009b [32] | Cohort | Cohort of Swedish men | 38,987 | 45 to 79 | DASH | NA | 9 years | HF development: Total: 807 patients Q4: 192 patients RR 0.78 (0.65–0.95, p = 0.006) |
| Nguyen et al., 2012 [33] | Cross-sectional | Multi-ethnic group free of clinical cardiovascular disease (CVD) | 4506 | 45 to 84 | DASH | NA | NA | A 1-unit increase in the DASH: Model 1 1: End-diastolic volume: 0.31 (0.08), p < 0.001 Stroke volume: 0.12 (0.03), p < 0.001 LVEF: 0.03 (0.02), p = 0.15 Model 2 2 End-diastolic volume: 0.31 (0.08), p < 0.001 Stroke volume: 0.12 (0.03), p < 0.001 LVEF: 0.03 (0.02), p = 0.15 Model 3 3 End-diastolic volume: 0.26 (0.08), p < 0.01 Stroke volume: 0.10 (0.03), p < 0.001 LVEF: 0.04 (0.02), p = 0.08 |
| Paleolithic Diet | ||||||||
| Andersson et al., 2016 [34] | RTC | Healthy postmenopausal women | 68 | NR | Paleolithic diet (30% PTN, 40% LIP, 30% CHO) | Nordic nutrition recommendation (15% PTN, 25–30% LIP, 55–60% CHO) | 2 years | NT-proBNP (pg/mL) Group Paleolithic diet: Baseline: 64.4 (6.1) After 24 months: 87.6 (18.6) Group Nordic Nutrition Recommendation: Baseline: 47.3 (4.8) After 24 months: 67.9 (9.6) (p = 0.764) Left ventricular mass (g) Group Paleolithic diet: Basline: 101 After 24 months: 90 Group Nordic Nutrition Recommendation: Baseline: 107 After 24 months: 99 (p < 0.05) |
| Vegetarian Diet | ||||||||
| Pai et al., 2015 [35] | Cross-sectional | Adventist health study-2 | 206 | 74.0 ± 10.0 | Vegetarian diet | non-vegetarian diet | NA | LV diastolic dysfunction: OR 0.42 (IC 95% 0.24–0.73) LV hypertrophy: OR 0.30 (IC 95% 0.08–0.86) |
| Study | SD | RSG | AC | Blinding of Research | Blinding of PP | Blinding of OA | LE | Do They Describe Confounders in an Adjusted Analysis? | Do You Assess the Balance Between the Groups at the Start of the Study? |
|---|---|---|---|---|---|---|---|---|---|
| Andersson et al. | RCT | High risk | Unclear risk | Unclear risk | Unclear risk | Low risk | Low risk | NA | NA |
| Fitó et al. | RCT | Low risk | Low risk | Unclear risk | Unclear risk | Low risk | Low risk | NA | NA |
| Lorgeril et al. | RCT | Unclear risk | Unclear risk | Unclear risk | Unclear risk | Low risk | Low risk | NA | NA |
| Papadaki et al. | RCT | Low risk | Low risk | Unclear risk | Unclear risk | Low risk | Low risk | NA | NA |
| Tuttle et al. | RCT | Low risk | Low risk | Unclear risk | Unclear risk | Low risk | Low risk | NA | NA |
| Chrysohoou et al. | RCT | Low risk | Low risk | Unclear risk | Unclear risk | Low risk | Low risk | NA | NA |
| Del Gobbo et al. | Cohort | NA | NA | NA | NA | NA | NA | Low risk | High risk |
| Levitan et al. | Cohort | NA | NA | NA | NA | NA | NA | Low risk | Low risk |
| Levitan et al. | Cohort | NA | NA | NA | NA | NA | NA | Low risk | Low risk |
| Tekitonidis et al. | Cohort | NA | NA | NA | NA | NA | NA | Low risk | Low risk |
| Tekitonidis et al. | Cohort | NA | NA | NA | NA | NA | NA | Low risk | Low risk |
| Wirth et al. | Cohort | NA | NA | NA | NA | NA | NA | Low risk | Low risk |
| Nguyen et al. | Cross-sectional study | NA | NA | NA | NA | NA | NA | Low risk | Low risk |
| Pai et al. * | Cross-sectional study | NA | NA | NA | NA | NA | NA | Low risk | High risk |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sanches Machado d’Almeida, K.; Ronchi Spillere, S.; Zuchinali, P.; Corrêa Souza, G. Mediterranean Diet and Other Dietary Patterns in Primary Prevention of Heart Failure and Changes in Cardiac Function Markers: A Systematic Review. Nutrients 2018, 10, 58. https://doi.org/10.3390/nu10010058
Sanches Machado d’Almeida K, Ronchi Spillere S, Zuchinali P, Corrêa Souza G. Mediterranean Diet and Other Dietary Patterns in Primary Prevention of Heart Failure and Changes in Cardiac Function Markers: A Systematic Review. Nutrients. 2018; 10(1):58. https://doi.org/10.3390/nu10010058
Chicago/Turabian StyleSanches Machado d’Almeida, Karina, Stefanny Ronchi Spillere, Priccila Zuchinali, and Gabriela Corrêa Souza. 2018. "Mediterranean Diet and Other Dietary Patterns in Primary Prevention of Heart Failure and Changes in Cardiac Function Markers: A Systematic Review" Nutrients 10, no. 1: 58. https://doi.org/10.3390/nu10010058
APA StyleSanches Machado d’Almeida, K., Ronchi Spillere, S., Zuchinali, P., & Corrêa Souza, G. (2018). Mediterranean Diet and Other Dietary Patterns in Primary Prevention of Heart Failure and Changes in Cardiac Function Markers: A Systematic Review. Nutrients, 10(1), 58. https://doi.org/10.3390/nu10010058




