1. Introduction
Salt marshes are among the most productive natural ecosystems in the world [
1,
2] and provide numerous ecological services and economic benefits [
3]. These important ecosystems are at risk from current and anticipated stressors, including sea level rise, higher average and peak temperatures, altered freshwater discharges and precipitation patterns, and more frequent droughts [
4,
5,
6,
7,
8]. To date, climate induced changes have been associated with shifting vegetation phenologies, variations in biomass and/or net annual primary production (NAPP), and shifting species composition within marsh ecosystems [
9,
10,
11,
12,
13]. Understanding how salt marsh ecosystems will continue to respond to a changing climate and additional anthropogenic disturbances are key questions for scientists, coastal reserve managers, conservationists, and coastal residents.
Vegetation indicators such as aboveground biomass and community composition are commonly used to evaluate overall health of coastal wetlands [
14,
15]. To understand the effects of climate-induced changes on ecosystem health and resilience, analysis of the influences abiotic drivers have on marsh productivity is required. However, traditional methods for monitoring plant biomass are field and lab intensive, expensive, and normally constrained to one or several accessible local sites. Remote sensing with aerial and satellite sensor platforms provides an attractive alternative to estimate biomass and monitor ecosystem health at greater spatial and temporal scales [
15,
16]. Synoptic remote sensing methods can estimate canopy chlorophyll, green leaf area, aboveground biomass, and vegetation fraction at regional levels [
17,
18]. Remote estimations of these ecological indicators are possible using spectral reflectance transformations, referred to as vegetation indices (VIs) [
14,
19].
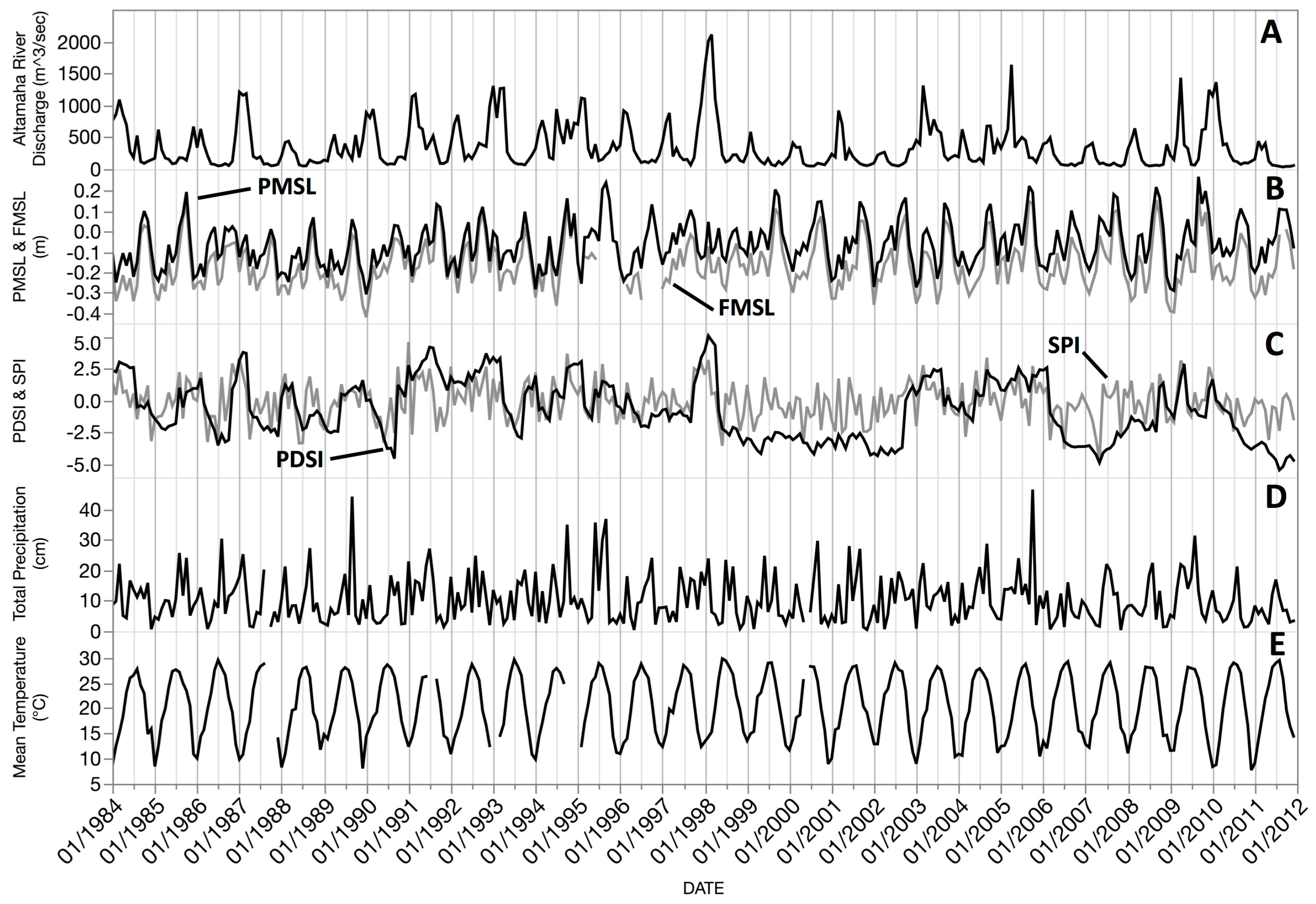
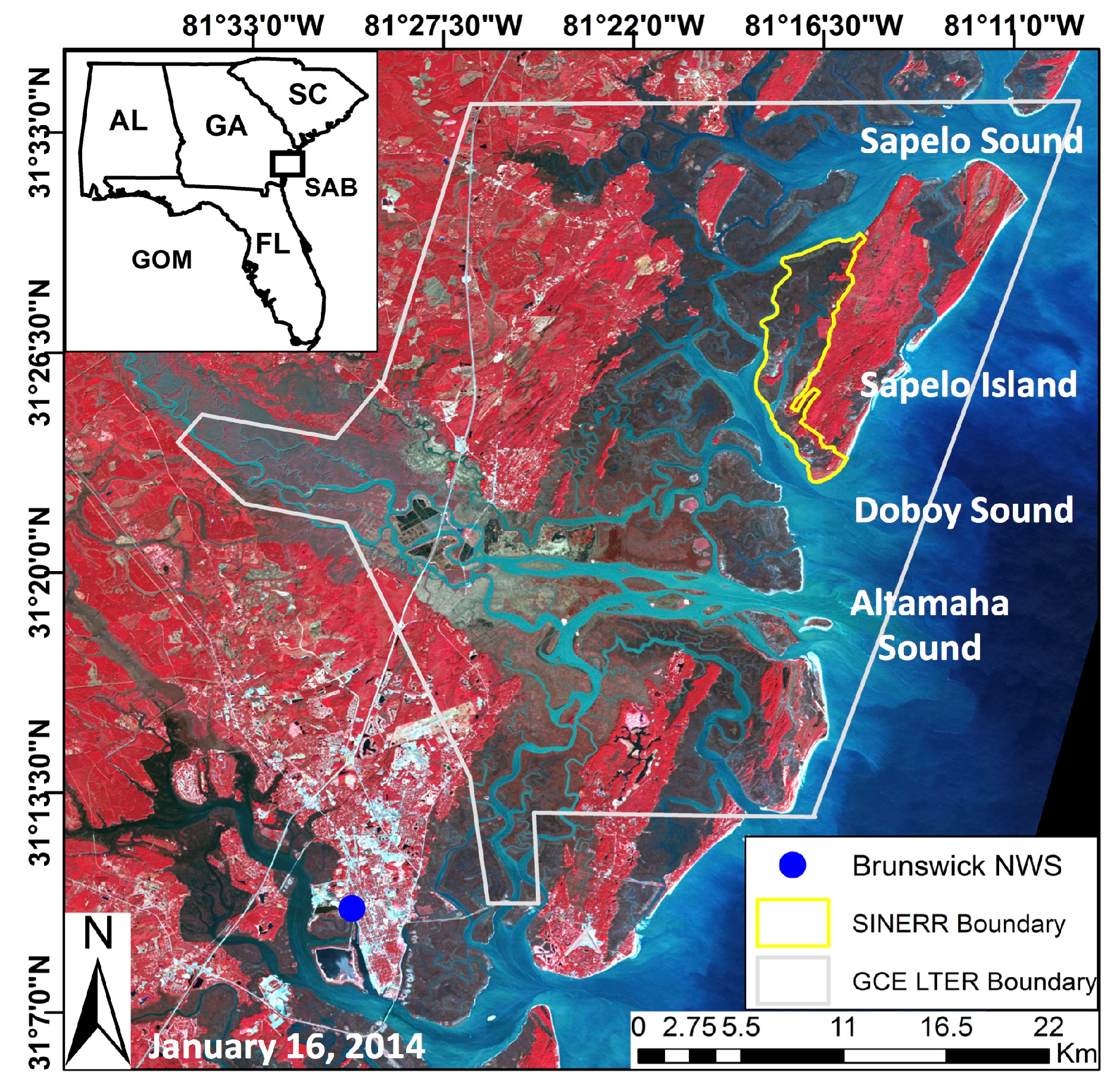
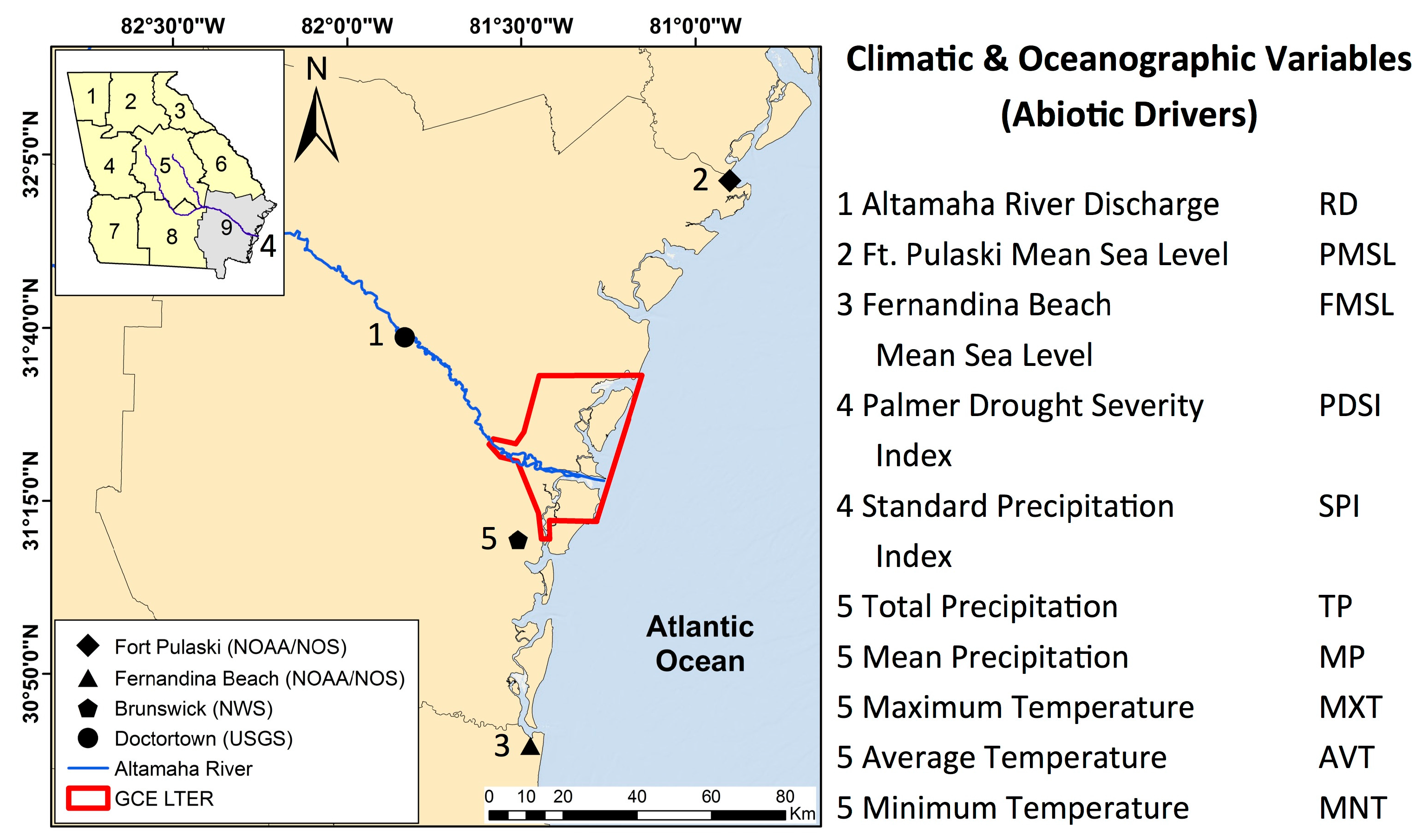
This study investigated the inter- and intra-annual dynamics of
Spartina alterniflora biomass on the Central Georgia Coast (USA) and the influence of abiotic drivers on spatial and temporal biomass patterns during the operational period of Landsat 5 TM (1984–2011). In a recent study, Więski and Pennings [
20] found that inter-annual differences in fall season
S. alterniflora biomass on the Central Georgia Coast were best explained by Altamaha River discharge, sea level, and maximum summer temperature. Our primary objectives were to: (1) establish a data extraction and analysis scheme for identification of statistically significant relationships between biomass and abiotic drivers using Landsat data; (2) identify trends of
S. alterniflora biomass throughout the 28-year study period and across each of three size classes; and (3) identify which climatic and other external variables best explained variations in
S. alterniflora biomass across all seasons and with data binned by seasons. In this paper, we present an effective approach using Earth Observation Satellite (EOS) sensors for long-term observations of salt marsh response to changing climate patterns and efficient monitoring of salt marsh health.
4. Discussion
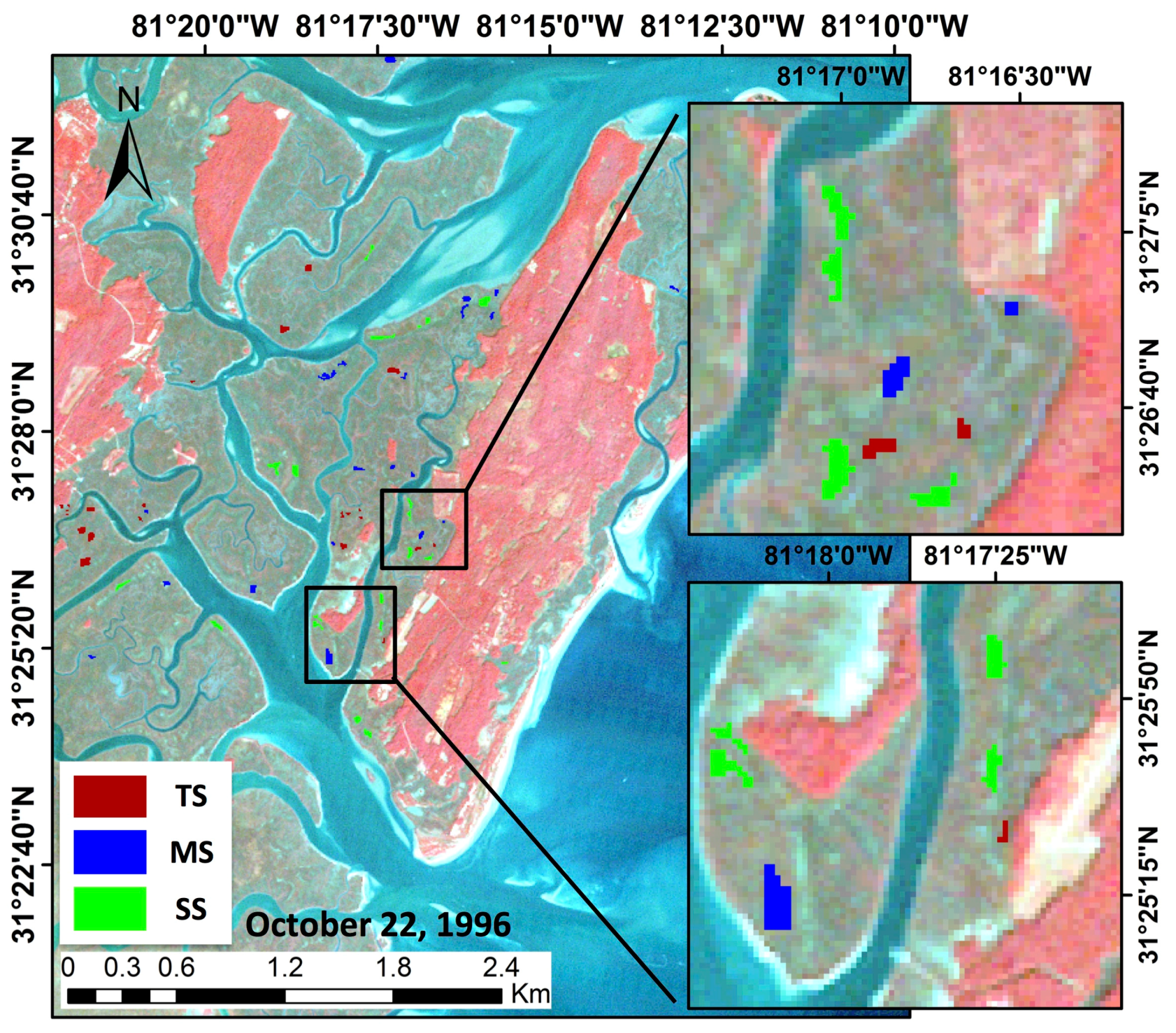
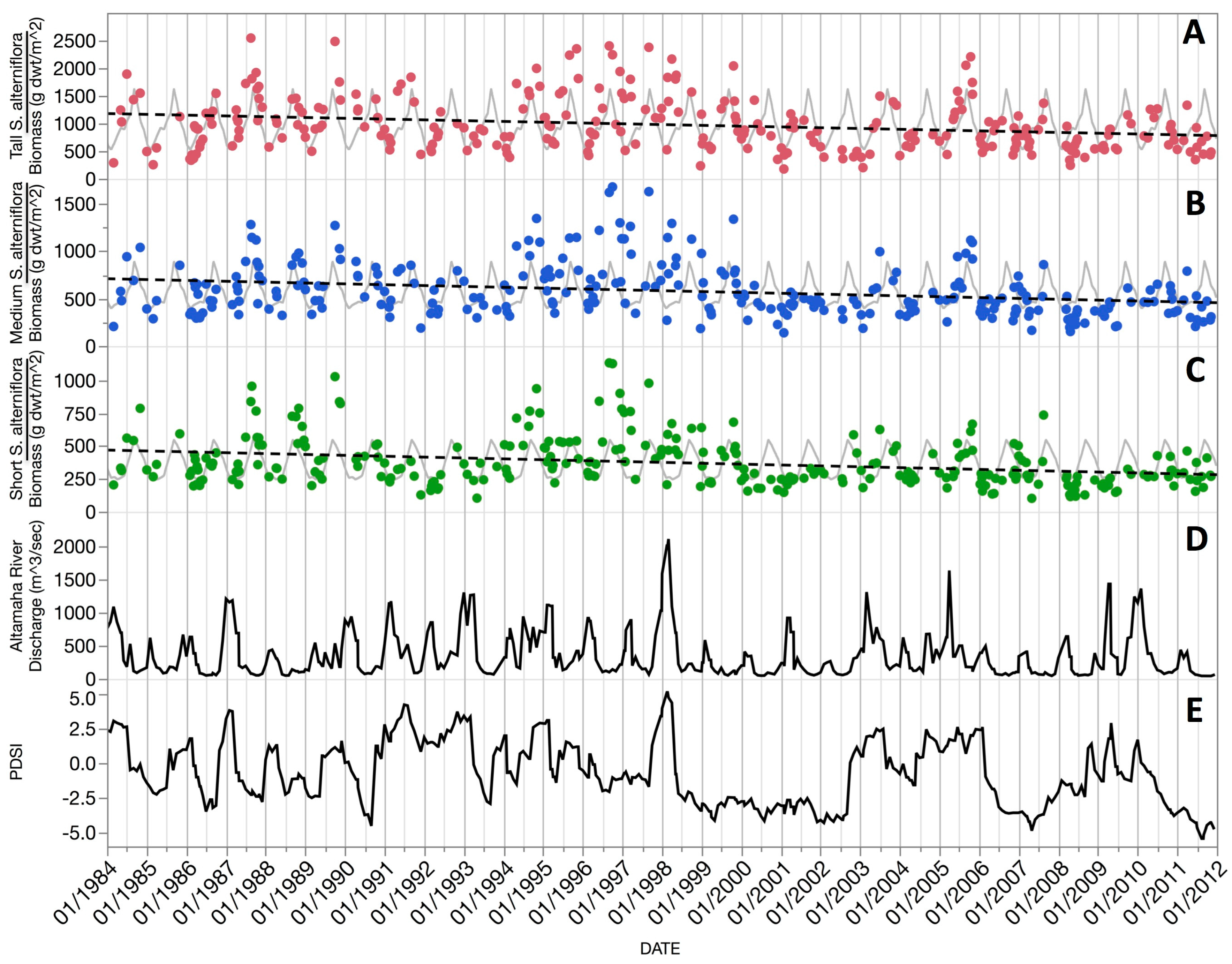
This study demonstrated the utility of using long-term Thematic Mapper optical data from a single NASA Earth Observation Satellite (EOS) mission, Landsat 5, that provided the longest continuous record of any EOS. We developed a scheme to: (1) scale-up ground truth clip-plot S. alterniflora aboveground biomass from our study area to match the 30 m resolution of Landsat 5; (2) create and extract polygons from 28 years of imagery that represented clusters of three operational size classes occurring across the marsh elevation gradient and covered ~1.1 million m2 of Central Georgia salt marsh; and (3) combine long-term biomass observations with climate, hydrology, and meteorological data to account for short to longer duration vegetation behavior.
We established that
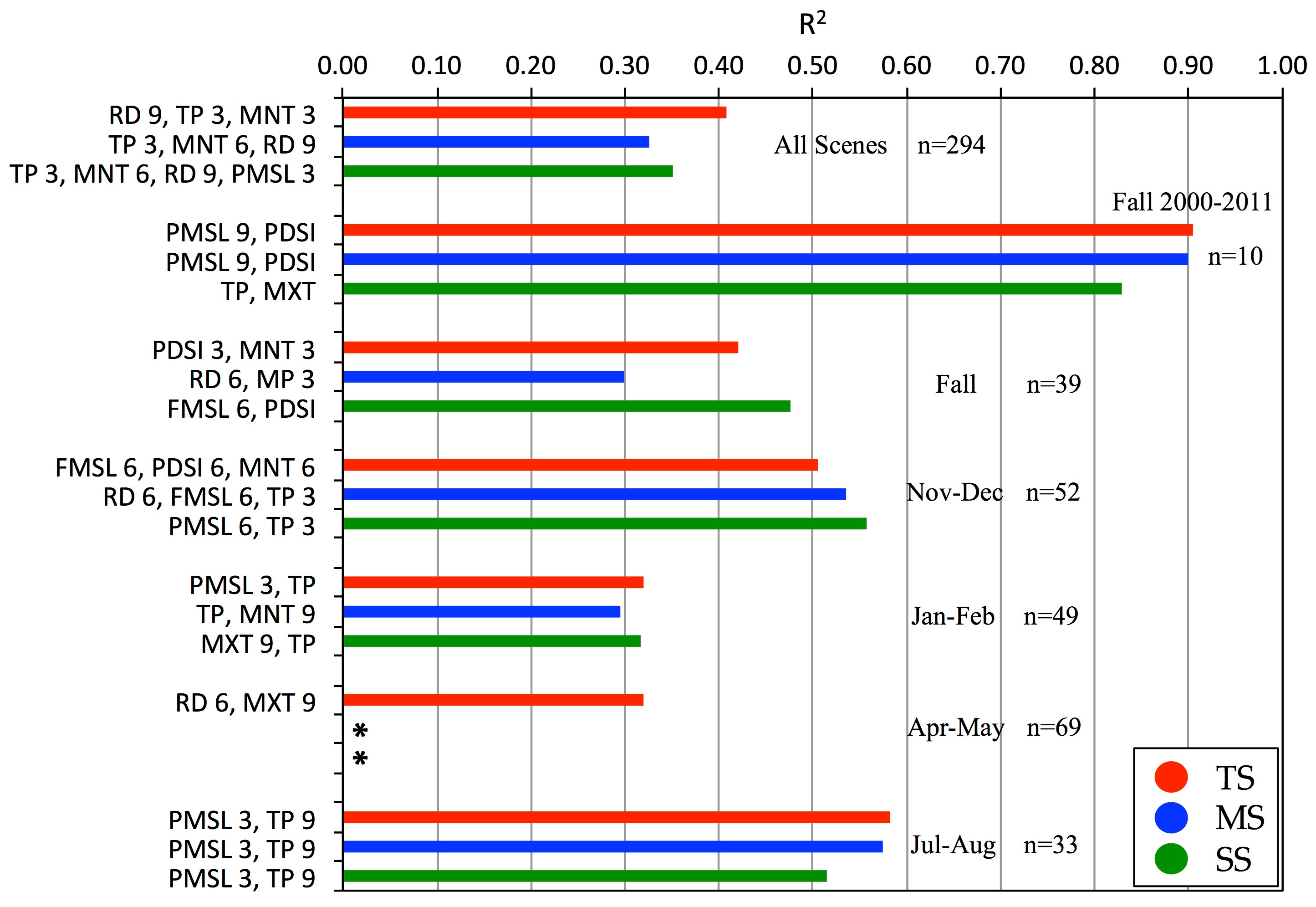
S. alterniflora production in Southeastern U.S. subtropical marshes is responsive at any time of year to abiotic drivers. For example, overwintering aboveground biomass is variable and appears most vulnerable to moisture stress from reduced precipitation and lower sea level. The duration of stress is important. Three, six, and nine month periods prior to image acquisitions all emerged as significant at certain times of year and with one or more size classes (
Figure 7). Not surprisingly, our best regression results (
Figure 7) matched the periods of greatest biomass change (notably mid-summer and mid-fall, see
Figure 5) or the longer durations and greater deviations from average phenology biomass (day-of-year gray line in
Figure 6) that occurred in the second half of our study (1998–2011).
Overall, temperature accounted for the greatest amount of temporal variability, primarily as a positive influence on the phenology of net primary production. However, the entire suite of variables determine the degree to which biomass deviates according to stress-subsidy responses [
50] from the average phenology (
Figure 6). In the winter, higher sea level, precipitation, and temperature promote higher biomass (
Figure 7). Increased river discharges and precipitation in winter lead to higher spring biomass. By mid-summer, however, higher sea level and/or total precipitation account for higher biomass whereas drought and high temperature stress result in lower than average biomass. By mid-fall to early winter, vegetation biomass responds most strongly to higher sea level, precipitation, and temperatures.
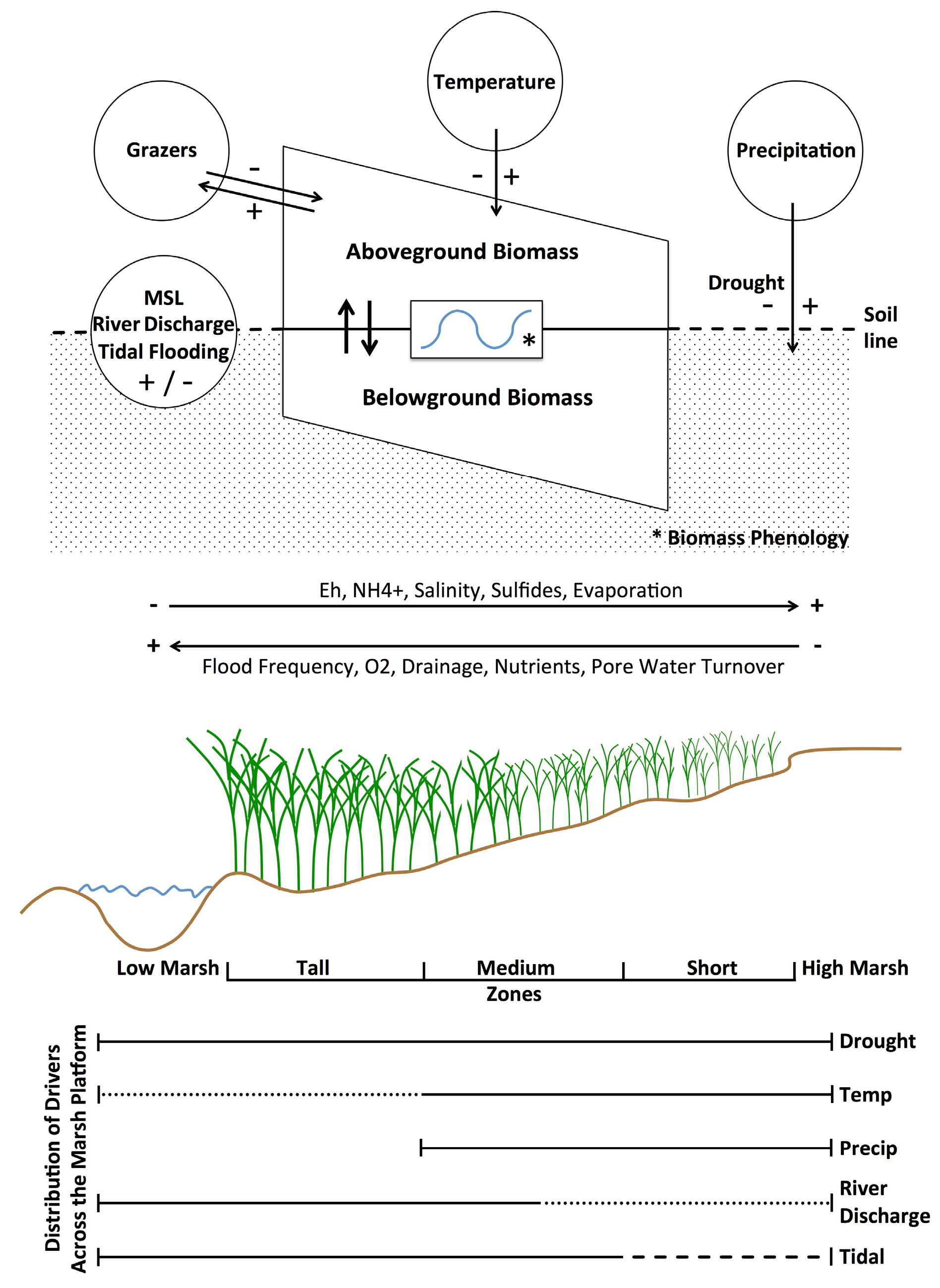
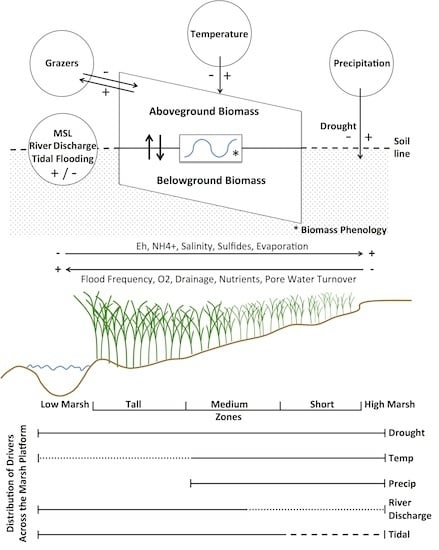
S. alterniflora physiology and productivity are directly driven by the physical and chemical properties within salt marsh sediments. Overall, soil salinities and other stressors in marshes (
Figure 8) are determined by the salinity of ambient tidal water, the frequency of tidal flooding along marsh elevation gradients, recent precipitation on the marsh, and solar radiation as a driver of evapotranspiration and soil hypersalinization [
51,
52]. These effects vary across the
S. alterniflora elevation stress gradient. Plant stress can also lead to stronger herbivory and top-down controls on marsh plants [
53].
Precipitation in any season directly dilutes salt that accumulates in soils and provides moisture (
Figure 8). For example, increased precipitation was associated with higher biomass in July and August, but also in our coldest two periods (November-December and January-February,
Figure 7). These results are consistent with previous findings by Dame and Kenny [
54], Gross
et al. [
55], and Więski and Pennings [
20].
High Altamaha River discharges decrease the salinities of interconnected nearshore channels and the inner shelf waters [
56] and increase nitrogen and phosphorous supplies [
57]. When coupled with higher sea levels, tidal flushing lowers porewater salinities, increases dissolved oxygen and limiting nutrients, and removes excess wastes (metabolites) and detritus (
Figure 8) [
52].
Both our study and Więski and Pennings [
20] found strong intra-annual differences in drivers and biomass responses for early fall during a common period of study (2000–2011). Biomass ranges were comparable, even though Więski and Pennings used non-destructive, site-specific allometric equations from fewer and smaller sampling areas
versus our more synoptic remote sensing procedure. Both studies established that Altamaha River discharge, sea level, temperature, and precipitation had positive correlations with and were generally the best predictors of mid-fall biomass and that maximum temperature had a negative correlation with late growing season biomass. Also, in both studies, higher sea levels (Fort Pulaski and, or Fernandina gauges) were associated with increased aboveground biomass for all three size classes along the marsh elevation gradient. Several studies have shown an increase in
S. alterniflora biomass productivity with increasing water levels at mid-marsh elevations [
58]. In our period of our study, sea level variations were more affected by meteorology (changes in the direction and force of winds) [
56] than the slow, additive rates of sea level rise.
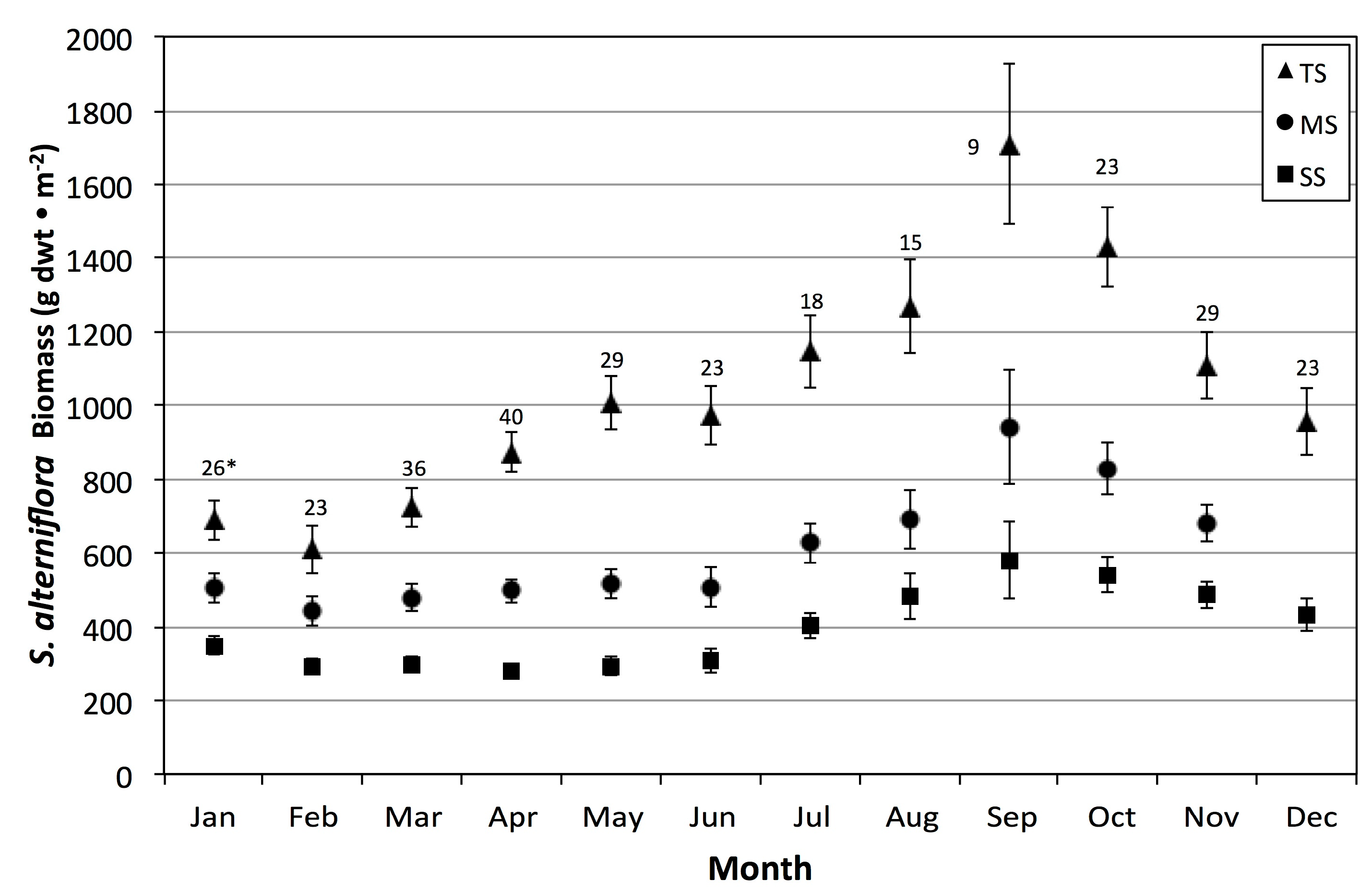
Unlike Więski and Penning’s study, we were also able to examine the influence of abiotic drivers on other periods of the year. In the subtropical climate of the Central Georgia Coast, primary production and net growth are possible at any time of the year, although the period between October and February had overall declines in average monthly aboveground biomass (
Figure 5). To increase sample sizes but avoid strong inter-monthly variation, we selected four bimonthly binned periods to examine the effects of abiotic variables at different phenology stages across our 28-year study period.
Consistent with our temperature response data, carbon fixation and net photosynthesis increased in all seasons in
S. alterniflora with modest temperature increases [
59]. Conversely, temperature stress was linked to decreased biomass during periods of drought and above average summer temperatures. During summer chamber experiments in Sapelo Island marshes, Giurgevich and Dunn [
59] found that net photosynthesis rates peaked at about 30–32 °C for both short and tall form
S. alterniflora. They measured significant reductions in net photosynthesis, leaf diffusive conductance to water vapor, and water use efficiencies between 35 and 45 °C. With anticipated increases in summer maximum temperatures [
60],
S. alterniflora may become more constrained by reduced periods of optimal temperatures and decreased NPP and through stress induced by a combination of higher temperatures and soil salinities and by lower soil redox potential (Eh).
Recent, prolonged droughts were associated with small to large-scale die-back events of
S. alterniflora [
61]. The direct causes of dieback events are complex, but both abiotic and biotic stressors have been identified as contributing factors [
62]. Droughts have profound impacts on the interactions between marsh plants, grazers, and fungi. For example, during extended periods of drought
Littoraria irrorata snails exert strong negative effects on short form
S. alterniflora biomass [
63]; however, these effects can extend into healthier stands at lower elevations [
53]. Fungi in the genus
Fusarium directly damaged
S. alterniflora under high salt stress and made plants more susceptible to purple marsh crab (
Sesarma reticulatum) and root nematode herbivory [
61]. The recurrent, highly reduced biomass between 2000 and 2011 (
Figure 6) may thus be explained by a combination of drought induced plant stress combined with snail, crab, and
Fusarium damage. Salt marsh biomass declines and diebacks could be similar to recent, widespread forest declines [
64], where drought intensification leads to negative plant carbon balance, increased vulnerability to herbivores (bark beetles), and, when prolonged, tree death spirals.
The extended periods of drought and related maximum summer temperature regimes since 2000 help to explain the net
S. alterniflora biomass decline over our 28-year study (
Table 4). Whether this decline is a portent of longer-term declines is unknown. Challenges to this keystone species and marsh ecosystem health are being addressed by the Georgia Coastal Ecosystems LTER long-term research program and the development of a comprehensive, linked set of models to portray plant ecophysiology and gross and primary production in a spatially explicit manner. Our remote sensing approach can play an important role in addressing this question not only on the Central Georgia Coast, but in many other coastal locations.
5. Conclusions
The ability to remotely differentiate size classes and thus analyze biomass responses along the marsh platform’s elevation gradient affords insights of
S. alterniflora’s adaptations to resources and stressors (
Figure 8). Our study identified river discharge, mean sea level, temperature, precipitation, drought, and previous year’s biomass as important predictors of
S. alterniflora production. Although most evident in seasonal or inter-annual periods of accelerated biomass change, the drivers we studied exert affects throughout the year and their magnitudes largely determine deviations from average biomass phenology.
Our 28-year record of aboveground biomass revealed a serious and recent decline in the emergent macrophyte,
S. alterniflora, in Coastal Georgia. It seems likely that this aggregate response is largely drought driven and the result of reduced flushing of marsh soils and warmer temperatures, leading to reduced nutrient loading, increased salinities, more extensive anaerobic conditions with elevated sulfide and ammonia levels, and greater herbivore damage (
Figure 8). Declines at lower elevations may also be related to variations in sea levels and accelerated physical and biological erosion along river and creekbanks and the margins of more isolated intertidal mudflats [
65].
This study has demonstrated the value of using remote sensing techniques and Earth Observing Satellites to monitor seasonal and long-term patterns of primary production as opposed to studies primarily based on end-of-season biomass (NAPP). The ability to accurately monitor and capture phenological trends can assist modeling efforts to better understand the interactions of environmental controls on vegetation across all seasons. However, in coastal habitats, selection of imagery requires careful assessment of tidal inundation as well as proper atmospheric corrections and georectification. This sampling method can accurately reveal large-scale patterns, making it easier to extrapolate hypotheses and conclusions to the ecosystem and landscape levels, as opposed to reliance on plot-based measurements. Finally, this study established a methodological approach that can be continued at our site and carried to other sites or regions, allowing scientists and reserve managers to better examine the influence of climatic and other external drivers via bottom-up and top-down controls on primary production within salt marsh ecosystems.