Submerged Kelp Detection with Hyperspectral Data
Abstract
:1. Introduction
2. Materials and Methods
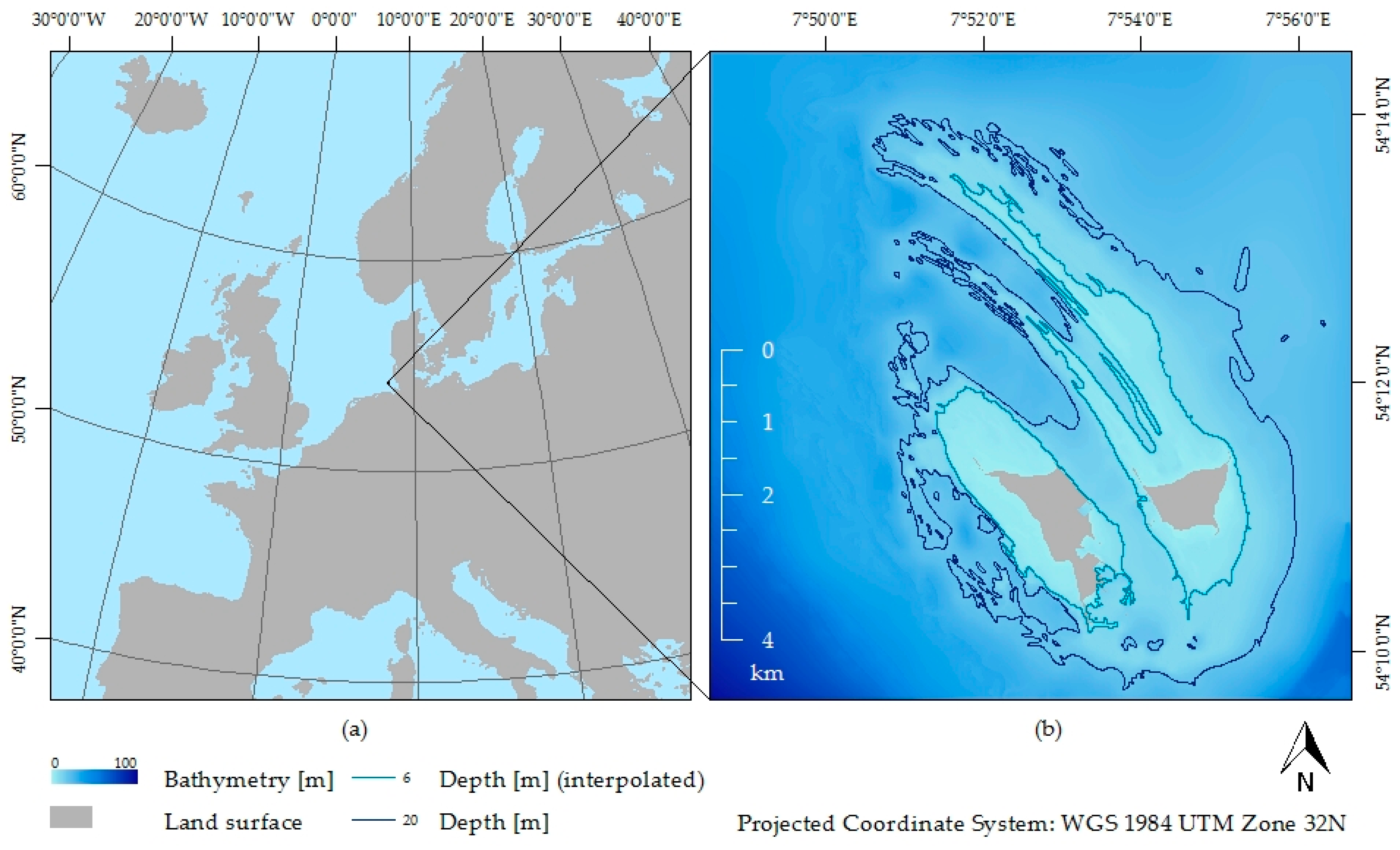
2.1. Study Site
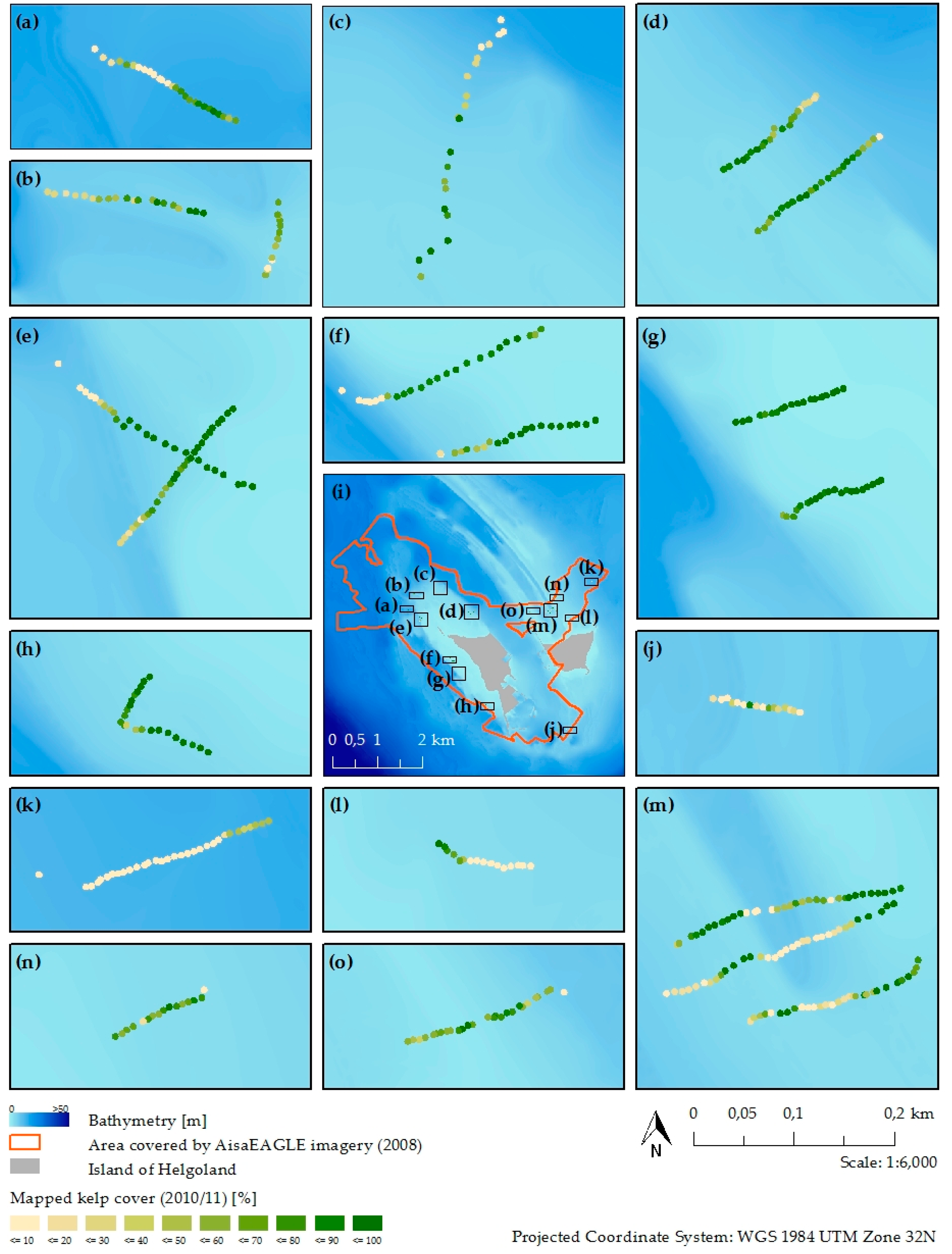
2.2. Field Survey
- Cover estimation of the four dominant brown macroalgae (Laminaria digitata, Laminaria hyperborea, Saccharina latissima, Desmarestia aculeata).
- Presence/absence of brown algae Water depth measurement using a digital depth gauge (Seemann Sub; precision: 40 cm) and transferred to sea chart level zero [56].
2.3. Hyperspectral Data
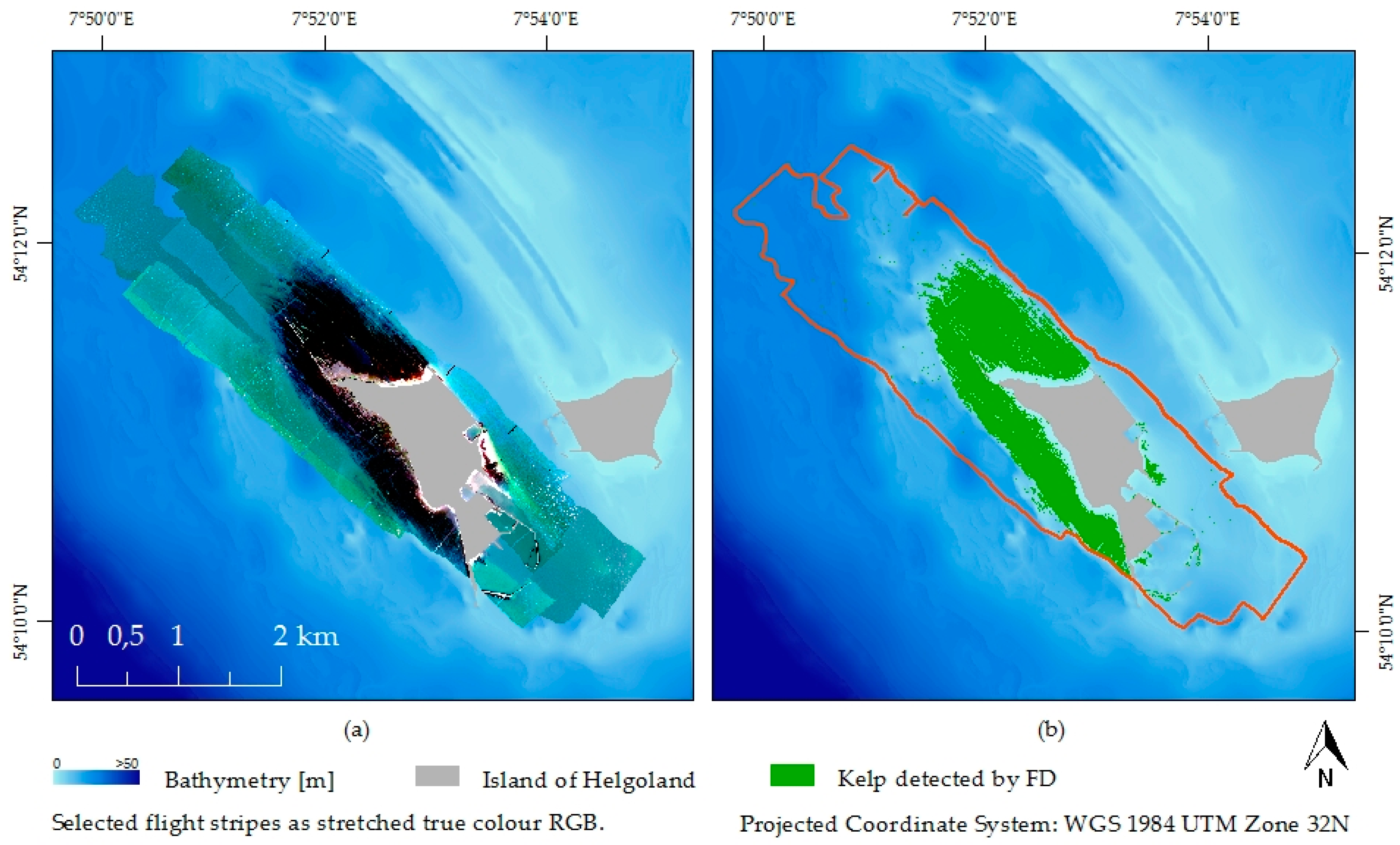
2.4. Kelp Detection
2.4.1. Water Anomaly Filter—WAF
2.4.2. Feature Detection—FD
2.5. Maximum Likelihood Classifier—MLC
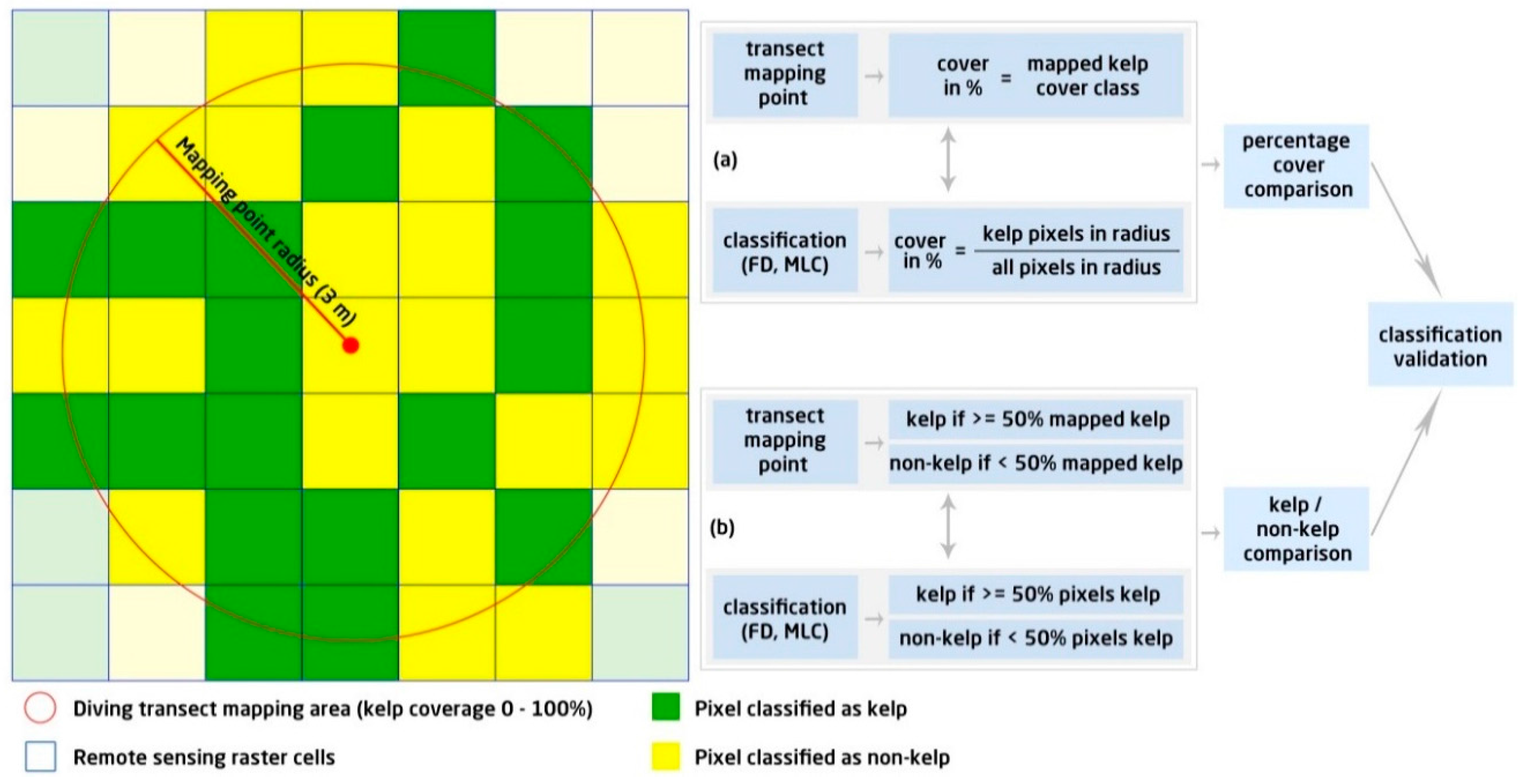
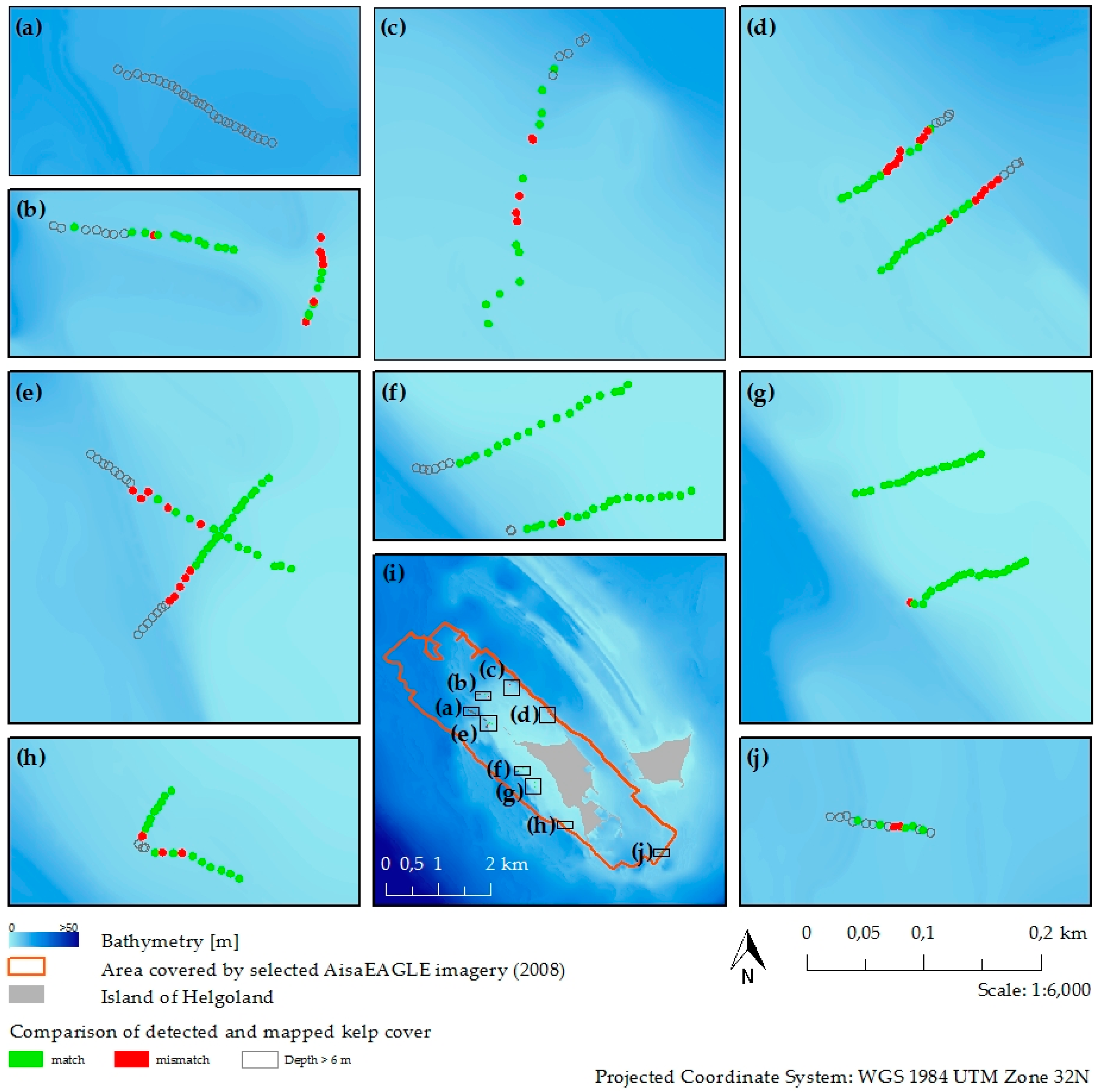
2.6. Validation of Classification Results
3. Results and Discussion
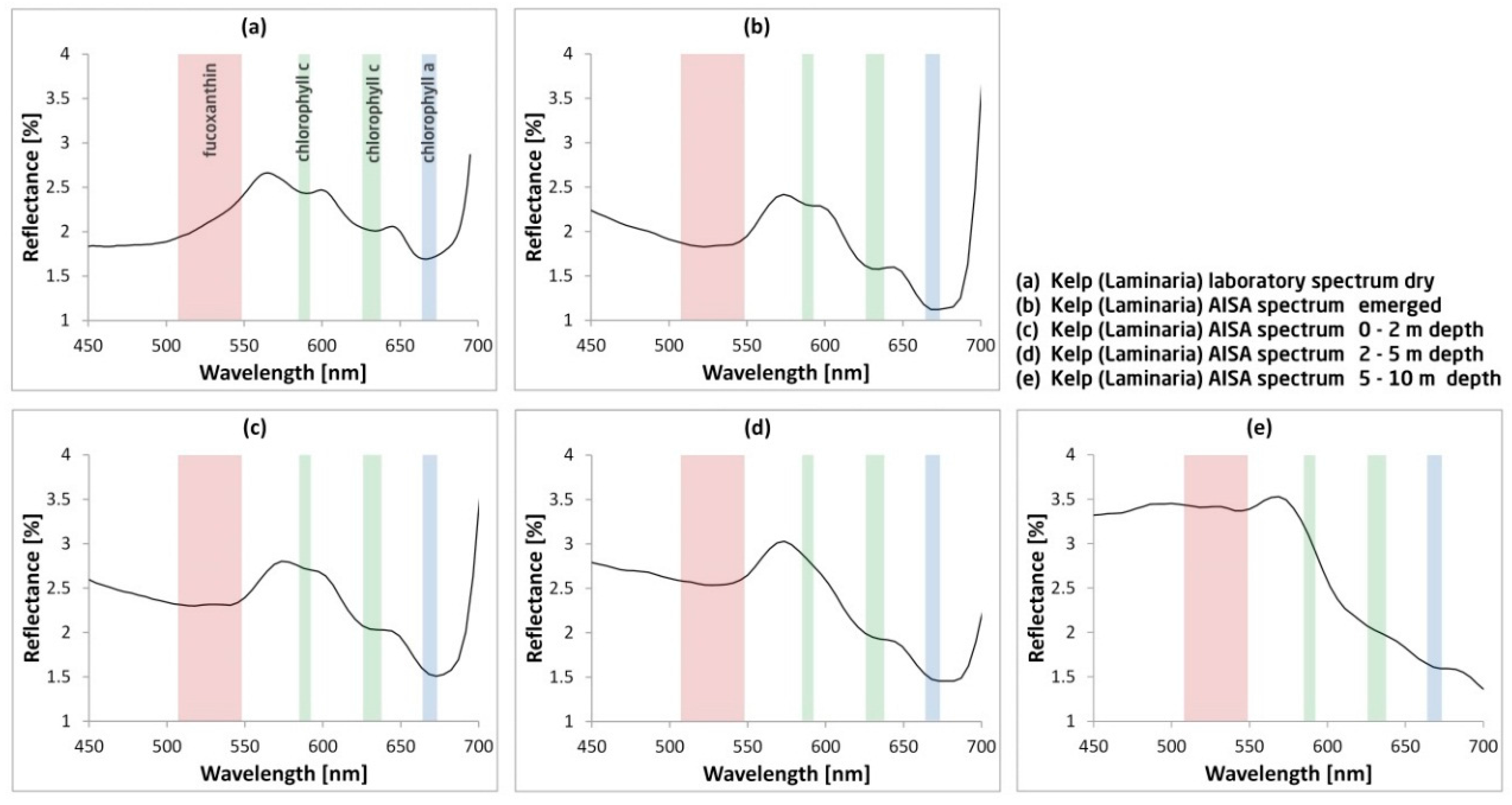
3.1. Wavelength Range for Deep Kelp Detection
3.2. WAF Performance
3.3. Kelp Detection Results Validated with Diving Transects
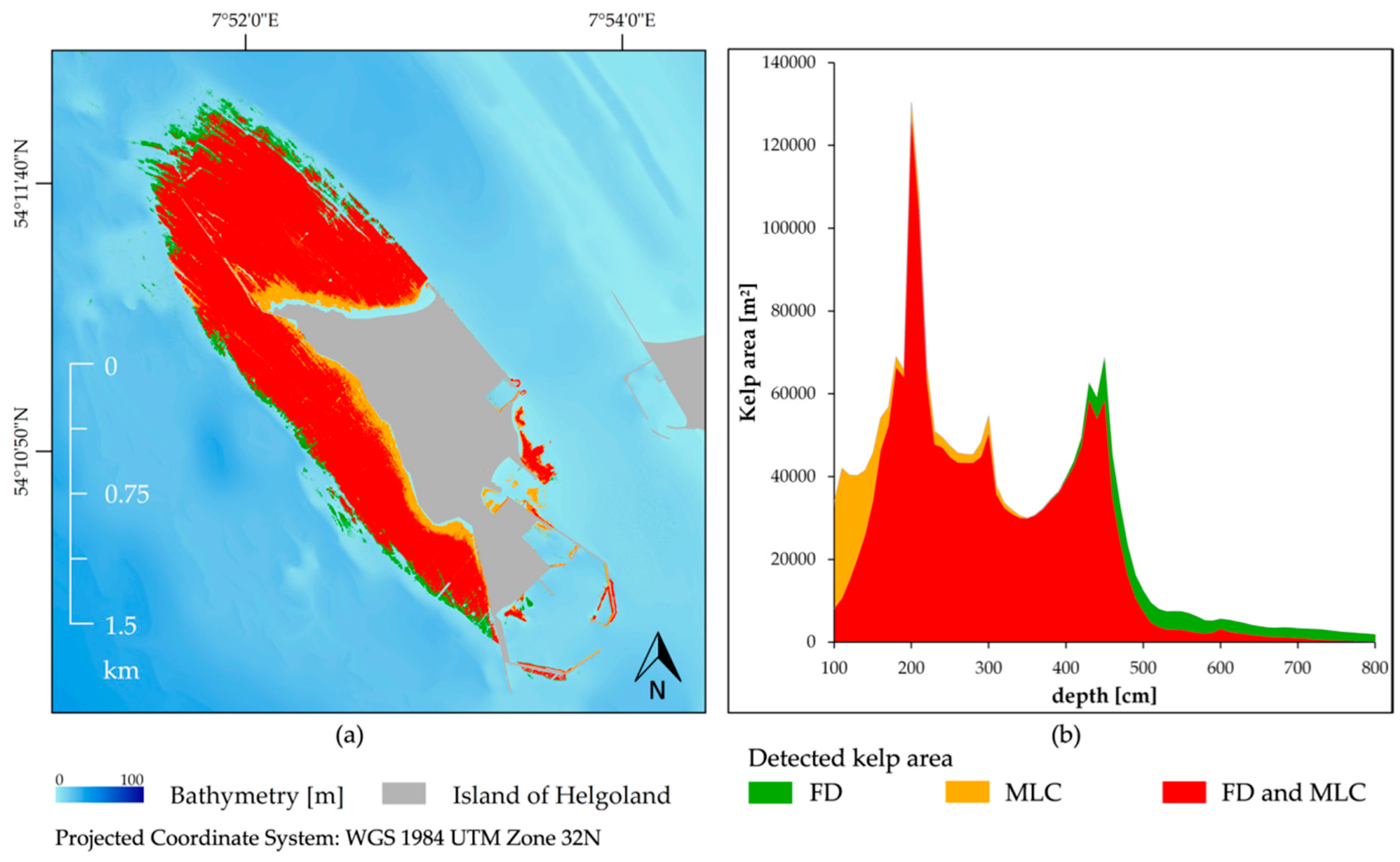
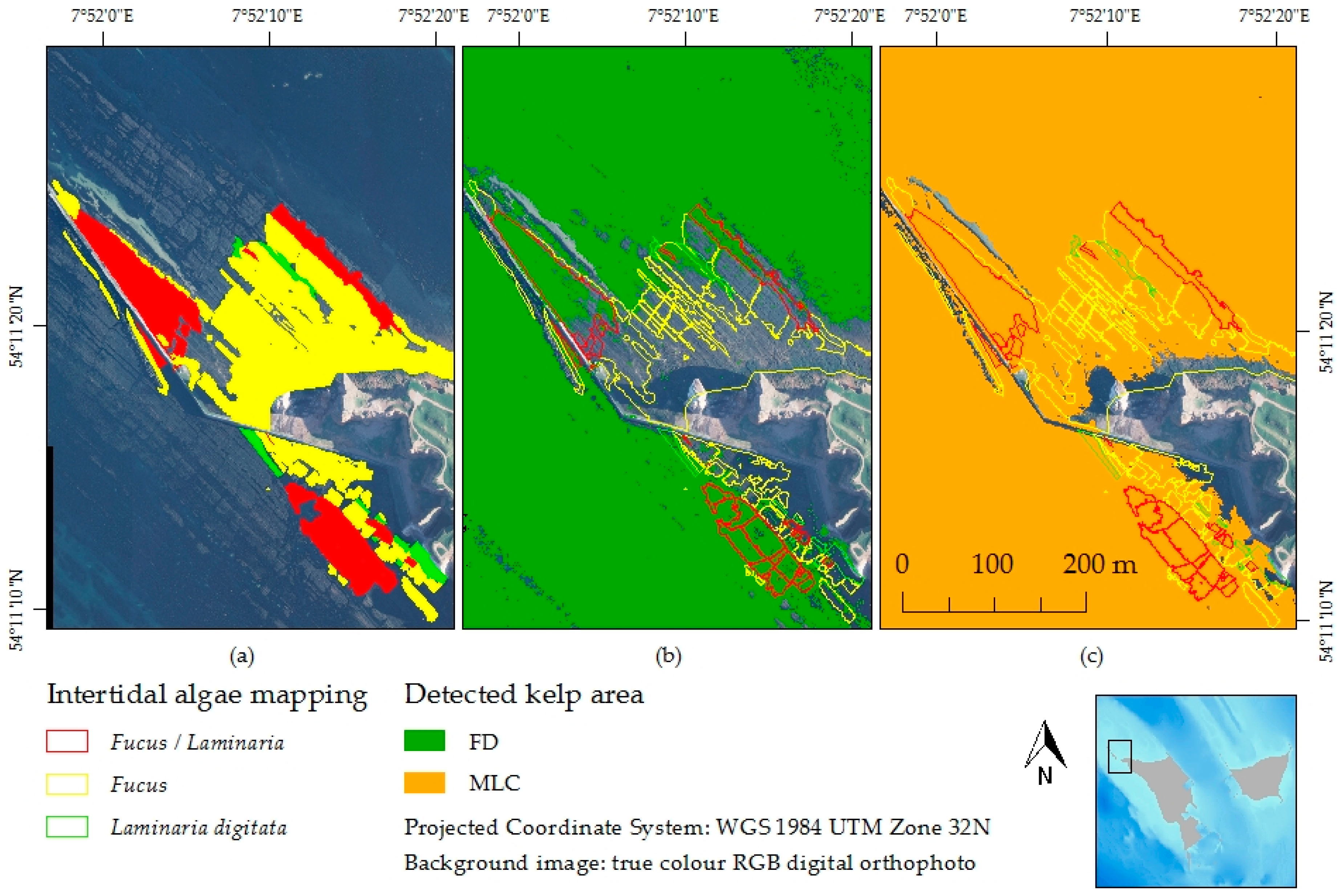
3.4. Feature Detection Results Validated with Maximum Likelihood Classifier
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| SAV | Submerged aquatic vegetation |
| FD | Feature detection |
| FS | Flight stripe |
| MLC | Maximum likelihood classifier |
| FAI | Floating algae index |
| MCI | Maximum chlorophyll index |
| NDVI | Normalized differenced vegetation index |
| RMSE | Root-mean-square error |
| R2 | Pearson product-moment correlation coefficient |
| NSE | Nash–Sutcliffe model efficiency coefficient |
Appendix

References
- Halpern, B.S.; Walbridge, S.; Selkoe, K.A.; Kappel, C.V.; Micheli, F.; D’Agrosa, C.; Bruno, J.F.; Casey, K.S.; Ebert, C.; Fox, H.E.; et al. A Global Map of Human Impact on Marine Ecosystems. Science 2008, 319, 948–952. [Google Scholar] [CrossRef] [PubMed]
- Wiencke, C.; Bischof, K. (Eds.) Seaweed Biology; Springer Berlin Heidelberg: Berlin, Heidelberg, 2012.
- Hafting, J.T.; Craigie, J.S.; Stengel, D.B.; Loureiro, R.R.; Buschmann, A.H.; Yarish, C.; Edwards, M.D.; Critchley, A.T.; Graham, M. Prospects and challenges for industrial production of seaweed bioactives. J. Phycol. 2015, 51, 821–837. [Google Scholar] [CrossRef] [PubMed]
- Levin, P.S. Small-scale recruitment variation in a temperate fish: The roles of macrophytes and food supply. Environ. Biol. Fishes 1994, 40, 271–281. [Google Scholar] [CrossRef]
- Holbrook, S.J.; Carr, M.H.; Schmitt, R.J.; Coyer, J.A. Effect of Giant Kelp on Local Abundance of Reef Fishes: The Importance of Ontogenetic Resource Requirements. Bull. Mar. Sci. 1990, 47, 104–114. [Google Scholar]
- Van Iersel, S.; Flammini, A. Algae-Based Biofuels: Applications and Co-Products. Available online: http://www.fao.org/docrep/012/i1704e/i1704e.pdf (accessed on 25 March 2016).
- Yue, H.; Sun, Y.; Jing, H.; Zeng, S.; Ouyang, H. The Analysis of Laminaria japonica Industry and International Trade Situation in China. In Proceedings of Selected Articles of 2013 World Agricultural Outlook Conference; Xu, S., Ed.; Springer: Heidelberg, Germany, 2014; pp. 39–51. [Google Scholar]
- Kerrison, P.D.; Stanley, M.S.; Edwards, M.D.; Black, K.D.; Hughes, A.D. The cultivation of European kelp for bioenergy: Site and species selection. Biomass Bioenergy 2015, 80, 229–242. [Google Scholar] [CrossRef]
- Lüning, K.; Dring, M.J. Continuous underwater light measurement near Helgoland (North Sea) and its significance for characteristic light limits in the sublittoral region. Helgoländer Wiss. Meeresunters. (Helgoländer Wissenschaftliche Meeresuntersuchungen) 1979, 32, 403–424. [Google Scholar] [CrossRef]
- Mann, K.H. Seaweeds: Their Productivity and Strategy for Growth: The role of large marine algae in coastal productivity is far more important than has been suspected. Science 1973, 182, 975–981. [Google Scholar] [CrossRef] [PubMed]
- Dean, T.A.; Jacobsen, F.R. Growth of juvenile Macrocystis pyrifera (Laminariales) in relation to environmental factors. Mar. Biol. 1984, 83, 301–311. [Google Scholar] [CrossRef]
- Lüning, K. Seaweeds: Their Environment, Biogeography, and Ecophysiology; Wiley: New York, NY, USA, 1990. [Google Scholar]
- Harley, C.D.G.; Randall Hughes, A.; Hultgren, K.M.; Miner, B.G.; Sorte, C.J.B.; Thornber, C.S.; Rodriguez, L.F.; Tomanek, L.; Williams, S.L. The impacts of climate change in coastal marine systems. Ecol. Lett. 2006, 9, 228–241. [Google Scholar] [CrossRef] [PubMed]
- Kirihara, S.; Nakamura, T.; Kon, N.; Fujita, D.; Notoya, M. Recent fluctuations in distribution and biomass of cold and warm temperature species of Laminarialean algae at Cape Ohma, northern Honshu, Japan. In Eighteenth International Seaweed Symposium; Anderson, R., Brodie, J., Onsøyen, E., Critchley, A.T., Eds.; Springer: Dordrecht, The Netherlands, 2007; pp. 295–301. [Google Scholar]
- Johnson, C.R.; Banks, S.C.; Barrett, N.S.; Cazassus, F.; Dunstan, P.K.; Edgar, G.J.; Frusher, S.D.; Gardner, C.; Haddon, M.; Helidoniotis, F.; et al. Climate change cascades: Shifts in oceanography, species’ ranges and subtidal marine community dynamics in eastern Tasmania. J. Exp. Mar. Biol. Ecol. 2011, 400, 17–32. [Google Scholar] [CrossRef]
- Bartsch, I.; Paar, M.; Fredriksen, S.; Schwanitz, M.; Daniel, C.; Hop, H.; Wiencke, C. Changes in kelp forest biomass and depth distribution in Kongsfjorden, Svalbard, between 1996–1998 and 2012–2014 reflect Arctic warming. Polar Biol. 2016. [Google Scholar] [CrossRef]
- Sogn Andersen, G.; Steen, H.; Christie, H.; Fredriksen, S.; Moy, F.E. Seasonal Patterns of Sporophyte Growth, Fertility, Fouling, and Mortality of Saccharina latissima in Skagerrak, Norway: Implications for Forest Recovery. J. Mar. Biol. 2011. [Google Scholar] [CrossRef]
- Díez, I.; Muguerza, N.; Santolaria, A.; Ganzedo, U.; Gorostiaga, J.M. Seaweed assemblage changes in the eastern Cantabrian Sea and their potential relationship to climate change. Estuar. Coast. Shelf Sci. 2012, 99, 108–120. [Google Scholar] [CrossRef]
- Wernberg, T.; Russell, B.D.; Thomsen, M.S.; Gurgel, C.F.D.; Bradshaw, C.J.; Poloczanska, E.S.; Connell, S.D. Seaweed Communities in Retreat from Ocean Warming. Curr. Biol. 2011, 21, 1828–1832. [Google Scholar] [CrossRef] [PubMed]
- Voerman, S.E.; Llera, E.; Rico, J.M. Climate driven changes in subtidal kelp forest communities in NW Spain. Mar. Environ. Res. 2013, 90, 119–127. [Google Scholar] [CrossRef] [PubMed]
- Müller, R.; Laepple, T.; Bartsch, I.; Wiencke, C. Impact of oceanic warming on the distribution of seaweeds in polar and cold-temperate waters. Bot. Mar. 2009, 52, 617–638. [Google Scholar] [CrossRef]
- Jueterbock, A.; Tyberghein, L.; Verbruggen, H.; Coyer, J.A.; Olsen, J.L.; Hoarau, G. Climate change impact on seaweed meadow distribution in the North Atlantic rocky intertidal. Ecol. Evol. 2013, 3, 1356–1373. [Google Scholar] [CrossRef] [PubMed]
- Krause-Jensen, D.; Duarte, C.M. Expansion of vegetated coastal ecosystems in the future Arctic. Front. Mar. Sci. 2014, 1. [Google Scholar] [CrossRef]
- European Parliament, Council of the European Union. Directive 2008/56/EC of the European Parliament and of the Council of 17 June 2008 establishing a framework for community action in the field of marine environmental policy (Marine Strategy Framework Directive). Off. J. Eur. Union 2008, 51, 19–40. [Google Scholar]
- Reichert, K.; Buchholz, F. Changes in the macrozoobenthos of the intertidal zone at Helgoland (German Bight, North Sea): A survey of 1984 repeated in 2002. Helgol. Mar. Res. 2006, 60, 213–223. [Google Scholar] [CrossRef]
- Merzouk, A.; Johnson, L.E. Kelp distribution in the northwest Atlantic Ocean under a changing climate. J. Exp. Mar. Biol. Ecol. 2011, 400, 90–98. [Google Scholar] [CrossRef]
- Van Rein, H.B.; Brown, C.J.; Quinn, R.; Breen, J. A review of sublittoral monitoring methods in temperate waters: A focus on scale. Underw. Technol. Int. J. Soc. Underw. Technol. 2009, 28, 99–113. [Google Scholar] [CrossRef]
- Zhang, X. On the estimation of biomass of submerged vegetation using Landsat thematic mapper (TM) imagery: A case study of the Honghu Lake, PR China. Int. J. Remote Sens. 1998, 19, 11–20. [Google Scholar] [CrossRef]
- Kutser, T.; Miller, I.; Jupp, D.L. Mapping coral reef benthic substrates using hyperspectral space-borne images and spectral libraries. Estuar. Coast. Shelf Sci. 2006, 70, 449–460. [Google Scholar] [CrossRef]
- Bertels, L.; Vanderstraete, T.; van Coillie, S.; Knaeps, E.; Sterckx, S.; Goossens, R.; Deronde, B. Mapping of coral reefs using hyperspectral CASI data; a case study: Fordata, Tanimbar, Indonesia. Int. J. Remote Sens. 2008, 29, 2359–2391. [Google Scholar] [CrossRef]
- Silva, T.S.F.; Costa, M.P.F.; Melack, J.M.; Novo Evlyn, M.L.M. Remote sensing of aquatic vegetation: Theory and applications. Environ. Monit. Assess. 2008, 140, 131–145. [Google Scholar] [CrossRef] [PubMed]
- Dekker, A.G.; Brando, V.E.; Anstee, J.M.; Pinnel, N.; Kutser, T.; Hoogenboom, E.J.; Peters, S.; Pasterkamp, R.; Vos, R.; Olbert, C.; et al. Imaging Spectrometry of Water. In Imaging Spectrometry: Basic Principles and Prospective Applications; van der Meer, F.D., Jong, S.M.D., Eds.; Kluwer Academic Publishers: Dordrecht, The Netherland, 2011; Volume 4, pp. 307–359. [Google Scholar]
- Simms, É.; Dubois, J.-M.M. Satellite remote sensing of submerged kelp beds on the Atlantic coast of Canada. Int. J. Remote Sens. 2001, 22, 2083–2094. [Google Scholar] [CrossRef]
- Vahtmäe, E.; Kutser, T. Mapping Bottom Type and Water Depth in Shallow Coastal Waters with Satellite Remote Sensing. J. Coast. Res. 2007, 50, 185–189. [Google Scholar]
- Fyfe, S.K. Spatial and temporal variation in spectral reflectance: Are seagrass species spectrally distinct? Limnol. Oceanogr. 2003, 48, 464–479. [Google Scholar] [CrossRef]
- Pinnel, N.; Heege, T.; Zimmermann, S. Spectral Discrimination of Submerged Macrophytes in Lakes Using Hyperspectral Remote Sensing Data. SPIE Proc. Ocean Opt. XVII 2004, 1, 1–16. [Google Scholar]
- Han, L.; Rundquist, D.C. The spectral responses of Ceratophyllum demersum at varying depths in an experimental tank. Int. J. Remote Sens. 2003, 24, 859–864. [Google Scholar] [CrossRef]
- Uhl, F.; Oppelt, N.; Bartsch, I. Spectral mixture of intertidal marine macroalgae around the island of Helgoland (Germany, North Sea). Aquat. Bot. 2013, 111, 112–124. [Google Scholar] [CrossRef]
- Malthus, T.J.; George, D.G. Airborne remote sensing of macrophytes in Cefni Reservoir, Anglesey, UK. Aquat. Bot. 1997, 58, 317–332. [Google Scholar] [CrossRef]
- Gitelson, A. The peak near 700 nm on radiance spectra of algae and water: Relationships of its magnitude and position with chlorophyll concentration. Int. J. Remote Sens. 1992, 13, 3367–3373. [Google Scholar] [CrossRef]
- Oppelt, N.; Schulze, F.; Bartsch, I.; Doernhoefer, K.; Eisenhardt, I. Hyperspectral classification approaches for intertidal macroalgae habitat mapping: A case study in Heligoland. Opt. Eng. 2012, 51. [Google Scholar] [CrossRef]
- Kutser, T.; Vahtmäe, E.; Martin, G. Assessing suitability of multispectral satellites for mapping benthic macroalgal cover in turbid coastal waters by means of model simulations. Estuar. Coast. Shelf Sci. 2006, 67, 521–529. [Google Scholar] [CrossRef]
- Roessler, S.; Wolf, P.; Schneider, T.; Melzer, A. Multispectral Remote Sensing of Invasive Aquatic Plants Using RapidEye. In Earth Observation of Global Changes (EOGC); [EOGC2011—3rd Earth Observation and Global Changes Conference, which was organized by Technische Universität München (TUM) in Germany, Peking University (China), and University of Waterloo (Canada)]; Krisp, J.M., Ed.; Springer: Berlin, Germany, 2013; pp. 109–123. [Google Scholar]
- Roelfsema, C.M.; Lyons, M.; Kovacs, E.M.; Maxwell, P.; Saunders, M.I.; Samper-Villarreal, J.; Phinn, S.R. Multi-temporal mapping of seagrass cover, species and biomass: A semi-automated object based image analysis approach. Remote Sens. Environ. 2014, 150, 172–187. [Google Scholar] [CrossRef]
- Hedley, J.; Roelfsema, C.; Chollett, I.; Harborne, A.; Heron, S.; Weeks, S.; Skirving, W.; Strong, A.; Eakin, C.; Christensen, T.; et al. Remote sensing of coral reefs for monitoring and management: A review. Remote Sens. 2016. [Google Scholar] [CrossRef]
- Kaufmann, H.; Segl, K.; Chabrillat, S.; Hofer, S.; Stuffler, T.; Mueller, A.; Richter, R.; Schreier, G.; Haydn, R.; Bach, H. EnMAP A Hyperspectral Sensor for Environmental Mapping and Analysis. In Proceedings of the 2006 IEEE International Symposium on Geoscience and Remote Sensing, Denver, CO, USA, 31 July–4 August 2006; pp. 1617–1619.
- Galeazzi, C.; Sacchetti, A.; Cisbani, A.; Babini, G. The PRISMA Program. In Proceedings of the IGARSS 2008—2008 IEEE International Geoscience and Remote Sensing Symposium, Boston, MA, USA, 7–11 July 2008; pp. 105–108.
- Schmidt, K.S.; Skidmore, A.K. Spectral discrimination of vegetation types in a coastal wetland. Remote Sens. Environ. 2003, 85, 92–108. [Google Scholar] [CrossRef]
- Phinn, S.; Roelfsema, C.; Dekker, A.; Brando, V.; Anstee, J. Mapping seagrass species, cover and biomass in shallow waters: An assessment of satellite multi-spectral and airborne hyper-spectral imaging systems in Moreton Bay (Australia). Remote Sens. Environ. 2008, 112, 3413–3425. [Google Scholar] [CrossRef]
- Dierssen, H.M.; Chlus, A.; Russell, B. Hyperspectral discrimination of floating mats of seagrass wrack and the macroalgae Sargassum in coastal waters of Greater Florida Bay using airborne remote sensing. Remote Sens. Environ. 2015, 167, 247–258. [Google Scholar] [CrossRef]
- Kluijver, M.J. Sublittoral hard substrate communities off Helgoland. Helgol. Meeresunters 1991, 45, 317–344. [Google Scholar] [CrossRef]
- Beermann, J.; Franke, H.-D. A supplement to the amphipod (Crustacea) species inventory of Helgoland (German Bight, North Sea): Indication of rapid recent change. Mar. Biodivers. Rec. 2011, 4. [Google Scholar] [CrossRef]
- DWD Climate Data Center (CDC). Downloadarchiv der Monats—Und Tageswerte von 78 Messstationen in Deutschland; Deutscher Wetterdienst: Offenbach, Germany, 2016. [Google Scholar]
- Franke, H.-D.; Gutow, L. Long-term changes in the macrozoobenthos around the rocky island of Helgoland (German Bight, North Sea). Helgol. Mar. Res. 2004, 58, 303–310. [Google Scholar] [CrossRef]
- Janke, K. Lebensgemeinschaften und ihre Besiedlungsstrukturen in der Gezeitenzone felsiger Meeresküsten: Die Bedeutung Biologischer Wechselwirkungen für die Entstehung und Erhaltung der Biozönose im Nordost-Felswatt von Helgoland; Dissertation: Kiel, Germany, 1989. [Google Scholar]
- Pehlke, C.; Bartsch, I. Changes in depth distribution and biomass of sublittoral seaweeds at Helgoland (North Sea) between 1970 and 2005. Clim. Res. 2008, 37, 135–147. [Google Scholar] [CrossRef]
- Bartsch, I.; Kuhlenkamp, R. The marine macroalgae of Helgoland (North Sea): An annotated list of records between 1845 and 1999. Helgol. Mar. Res. 2000, 54, 160–189. [Google Scholar] [CrossRef]
- Lüning, K. Tauchuntersuchungen zur Vertikalverteilung der sublitoralen Helgoländer Algenvegetation; Biologische Anstalt Helgoland: List auf Sylt, Germany, 1970. [Google Scholar]
- Bartsch, I.; Tittley, I. The rocky intertidal biotopes of Helgoland: Present and past. Helgol. Mar. Res. 2004, 58, 289–302. [Google Scholar] [CrossRef]
- Wasser- und Schifffahrtsverwaltung des Bundes (WSV) im Geschäftsbereich des Bundesministeriums für Verkehr und Digitale Infrastruktur. Available online: https://www.pegelonline.wsv.de/gast/start (accessed on 10 October 2015).
- Uhl, F.; Oppelt, N.; Bartsch, I. Mapping marine macroalgae in case 2 waters using CHRIS PROBA. In Proceedings of the ESA Living Planet Symposium, ESA Special Proceedings SP-722 (CD-ROM), Edinburgh, UK, 9–13 September 2013; p. CD-ROM.
- SPECIM Images. Aisa EAGLE Hyperspectral Sensor. Available online: http://cdn.metricmarketing.ca/www.spectralcameras.com/files/AISA/AisaEAGLE_datasheet_ver1–2013.pdf?this=that (accessed on 11 January 2016).
- Oxford Technical Solutions Limited. RTv2 GNSS-Aided Inertial Measurement Systems; Oxford Technical Solutions Limited: Oxford, UK, 2015. [Google Scholar]
- Richter, R.; Schlaepfer, D. Status of Model ATCOR4 on Atmospheric Topographic Correction for Airborne Hyperspectral Imagery, 2003. Available online: http://www.earsel.org/workshops/imaging-spectroscopy-2003/papers/data_enhancement/richter.pdf (accessed on 11 September 2013).
- Tec5 AG. HandySpec Field: A Portable Spectrometer System. In Proceedings of the 3rd EARSeL Workshop on Imaging Spectroscopy, Herrsching, Germany, 13–16 May 2003.
- Van der Linden, S.; Rabe, A.; Held, M.; Jakimow, B.; Leitão, P.; Okujeni, A.; Schwieder, M.; Suess, S.; Hostert, P. The EnMAP-Box—A Toolbox and Application Programming Interface for EnMAP Data Processing. Remote Sens. 2015, 7, 11249–11266. [Google Scholar] [CrossRef]
- Uhl, F.; Oppelt, N.; Bartsch, I. Einfaches Verfahren zur Bestimmung von marinen Algengemeinschaften mit Hilfe von hyperspektraler Fernerkundung. In Geoinformationen für die Küstenzone, Band 4: Beiträge des 4. Hamburger Symposiums zur Küstenzone und Beiträge des 9. Workshops zur Nutzung der Fernerkundung im Bereich der Bundesanstalt für Gewässerkunde/Wasser- und Schifffahrtsverwaltung des Bundes, 1st ed.; Traub, K.-P., Kohlus, J., Lüllwitz, T., Eds.; Sokrates & Freunde: Koblenz, Germany, 2013; pp. 233–245. [Google Scholar]
- Butler, W.L.; Hopkins, D.W. Higher derivative analysis of complex absorption spectra. Photochem. Photobiol. 1970, 12, 439–450. [Google Scholar] [CrossRef]
- Demetriades-Shah, T.H.; Steven, M.D.; Clark, J.A. High resolution derivative spectra in remote sensing. Remote Sens. Environ. 1990, 33, 55–64. [Google Scholar] [CrossRef]
- Tsai, F.; Philpot, W. Derivative Analysis of Hyperspectral Data. Remote Sens. Environ. 1998, 66, 41–51. [Google Scholar] [CrossRef]
- Savitzky, A.; Golay, M.J.E. Smoothing and Differentiation of Data by Simplified Least Squares Procedures. Anal. Chem. 1964, 36, 1627–1639. [Google Scholar] [CrossRef]
- Noiraksar, T.; Sawayama, S.; Phauk, S.; Komatsu, T. Mapping Sargassum beds off the coast of Chon Buri Province, Thailand, using ALOS AVNIR-2 satellite imagery. Bot. Mar. 2014, 57, 367–377. [Google Scholar] [CrossRef]
- Hoang, T.C.; O’Leary, M.J.; Fotedar, R.K. Remote-Sensed Mapping of Sargassum spp. Distribution around Rottnest Island, Western Australia, Using High-Spatial Resolution WorldView-2 Satellite Data. J. Coast. Res. 2015. [Google Scholar] [CrossRef]
- Lillesand, T.M.; Kiefer, R.W.; Chipman, J.W. Remote Sensing and Image Interpretation, 6th ed.; Wiley: Hoboken, NJ, USA, 2008. [Google Scholar]
- Pasqualini, V.; Pergent-MARTINI, C.; Fernandez, C.; Pergent, G. The use of airborne remote sensing for benthic cartography: Advantages and reliability. Int. J. Remote Sens. 1997, 18, 1167–1177. [Google Scholar] [CrossRef]
- Sawayama, S.; Nurdin, N.; Akbar, A.S.M.; Sakamoto, S.X.; Komatsu, T. Introduction of geospatial perspective to the ecology of fish-habitat relationships in Indonesian coral reefs: A remote sensing approach. Ocean Sci. J. 2015, 50, 343–352. [Google Scholar] [CrossRef]
- Nash, J.E.; Sutcliffe, J.V. River flow forecasting through conceptual models part I—A discussion of principles. J. Hydrol. 1970, 10, 282–290. [Google Scholar] [CrossRef]
- Campbell, J.B.; Wynne, R.H. Introduction to Remote Sensing, 5th ed.; Guilford Press: New York, NY, USA, 2011. [Google Scholar]
- Wiltshire, K.H.; Malzahn, A.M.; Wirtz, K.; Greve, W.; Janisch, S.; Mangelsdorf, P.; Manly, B.F.J.; Boersma, M. Resilience of North Sea phytoplankton spring bloom dynamics: An analysis of long-term data at Helgoland Roads. Limnol. Oceangr. 2008, 53, 1294–1302. [Google Scholar] [CrossRef]
- Hochberg, E.J.; Andrefouet, S.; Tyler, M.R. Sea surface correction of high spatial resolution ikonos images to improve bottom mapping in near-shore environments. IEEE Trans. Geosci. Remote Sens. 2003, 41, 1724–1729. [Google Scholar] [CrossRef]
- Hedley, J.D.; Harborne, A.R.; Mumby, P.J. Technical note: Simple and robust removal of sun glint for mapping shallow-water benthos. Int. J. Remote Sens. 2005, 26, 2107–2112. [Google Scholar] [CrossRef]
- Kutser, T.; Vahtmäe, E.; Praks, J. A sun glint correction method for hyperspectral imagery containing areas with non-negligible water leaving NIR signal. Remote Sens. Environ. 2009, 113, 2267–2274. [Google Scholar] [CrossRef]
- Hu, C. A novel ocean color index to detect floating algae in the global oceans. Remote Sens. Environ. 2009, 113, 2118–2129. [Google Scholar] [CrossRef]
- Gower, J.; King, S.; Borstad, G.; Brown, L. Detection of intense plankton blooms using the 709 nm band of the MERIS imaging spectrometer. Int. J. Remote Sens. 2005, 26, 2005–2012. [Google Scholar] [CrossRef]
- Hu, C.; Feng, L.; Hardy, R.F.; Hochberg, E.J. Spectral and spatial requirements of remote measurements of pelagic Sargassum macroalgae. Remote Sens. Environ. 2015, 167, 229–246. [Google Scholar] [CrossRef]

| FS | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Start time | 10:08 | 10:20 | 10:27 | 10:34 | 10:41 | 10:48 | 10:53 | 10:59 | 11:05 | 11:19 | 11:27 | 11:36 | 11:42 |
| Flight Stripe | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| corrected pixels (%) | 2.91 | 3.89 | 2.24 | 2.11 | 3.06 | 4.15 | 11.65 | 11.88 | 5.51 | 4.42 | 4.17 | 4.92 | 6.56 |
| FS | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| RMSE | 40.45 | 42.61 | 36.28 | 41.27 | 17.65 | 18.54 | 46.85 | 45.27 | 69.83 | 59.11 | 62.58 | 57.32 | 57.14 |
| R2 | 0.43 | 0.43 | 0.40 | 0.38 | 0.70 | 0.72 | 0.39 | 0.33 | 0.06 | 0.16 | 0.20 | 0.29 | 0.03 |
| NSE | −1.42 | −1.28 | −1.02 | −0.93 | 0.57 | 0.65 | −0.81 | −0.94 | −4.90 | −2.05 | −2.29 | −1.93 | −1.25 |
| FS | 1 | 2 | 3 | 4 | 5 | 6 | 10 | 11 | 12 |
|---|---|---|---|---|---|---|---|---|---|
| RMSE | 66.20 | 45.50 | 48.62 | 62.04 | 49.04 | 57.62 | 60.22 | 59.44 | 47.90 |
| R2 | 0.12 | 0.39 | 0.19 | 0.20 | 0.27 | 0.25 | 0.13 | 0.21 | 0.39 |
| NSE | −5.44 | −1.59 | −2.61 | −3.33 | −2.33 | −2.37 | −2.16 | −1.95 | −1.03 |
| Depth Limit (m) | 1 | 2 | 3 | 4 | 5 | 6 |
|---|---|---|---|---|---|---|
| MLC overall accuracy (%) | 100 | 92.65 | 80.53 | 65.66 | 58.5 | 57.66 |
| FD overall accuracy (%) | 100 | 98.53 | 96.46 | 87.95 | 82 | 80.18 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Uhl, F.; Bartsch, I.; Oppelt, N. Submerged Kelp Detection with Hyperspectral Data. Remote Sens. 2016, 8, 487. https://doi.org/10.3390/rs8060487
Uhl F, Bartsch I, Oppelt N. Submerged Kelp Detection with Hyperspectral Data. Remote Sensing. 2016; 8(6):487. https://doi.org/10.3390/rs8060487
Chicago/Turabian StyleUhl, Florian, Inka Bartsch, and Natascha Oppelt. 2016. "Submerged Kelp Detection with Hyperspectral Data" Remote Sensing 8, no. 6: 487. https://doi.org/10.3390/rs8060487
APA StyleUhl, F., Bartsch, I., & Oppelt, N. (2016). Submerged Kelp Detection with Hyperspectral Data. Remote Sensing, 8(6), 487. https://doi.org/10.3390/rs8060487







