Biome-Scale Forest Properties in Amazonia Based on Field and Satellite Observations
Abstract
:1. Introduction
2. Forest Biophysical Properties
2.1. Wood Density
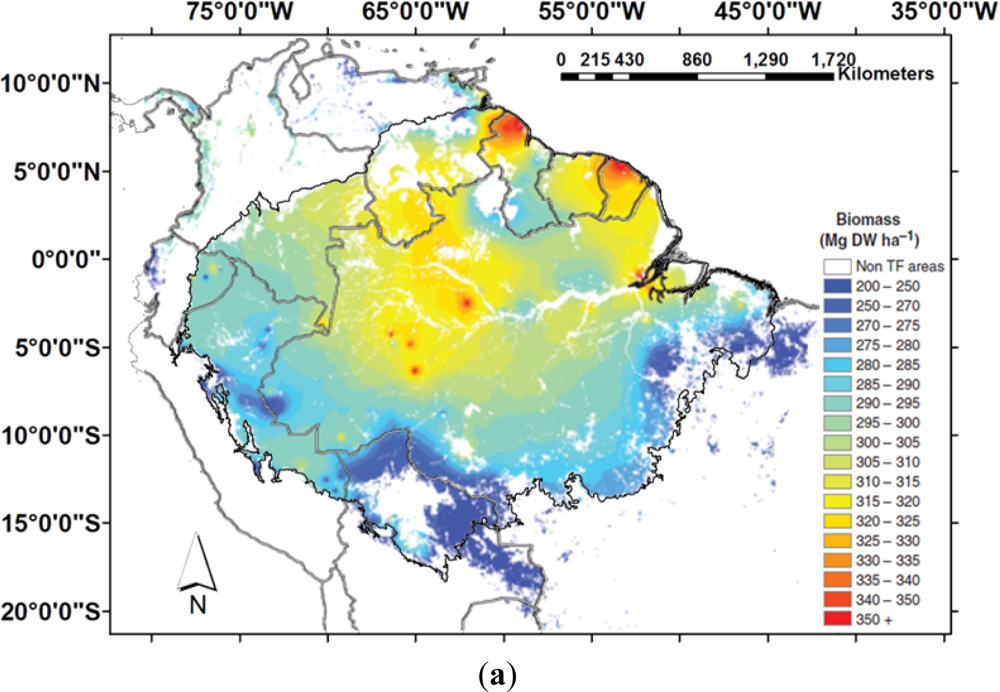
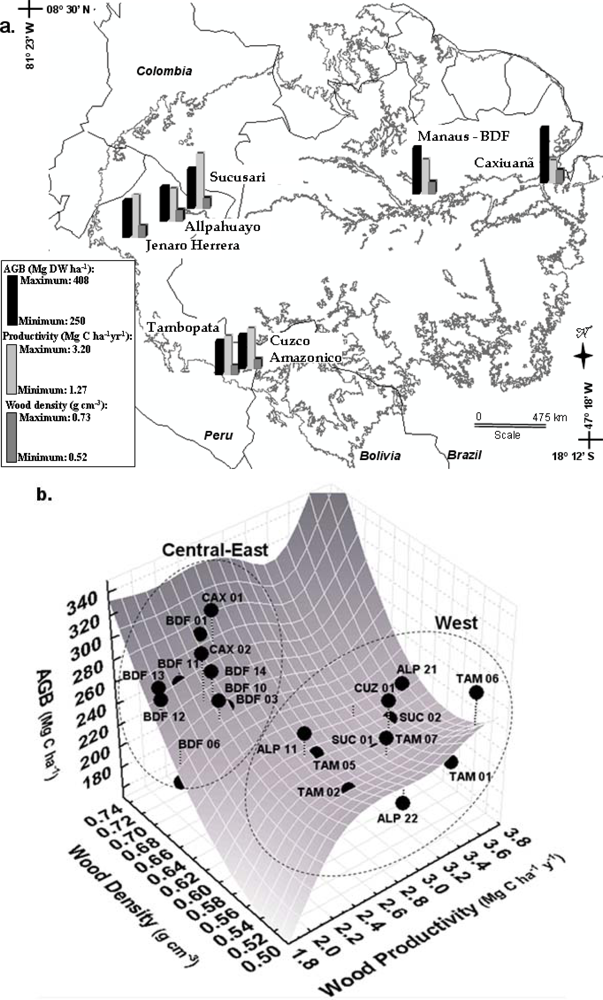
2.2. Above Ground Biomass
2.3. Canopy Height
2.4. Crown Size and Leaf Size Patterns
2.5. Forest Productivity
3. Vegetation Phenology
3.1. Phenological Patterns Observed on the Ground
3.2. Phenological Patterns Derived from Remote Sensing
3.2.1. Background
3.2.2. Monitoring Forest Phenology
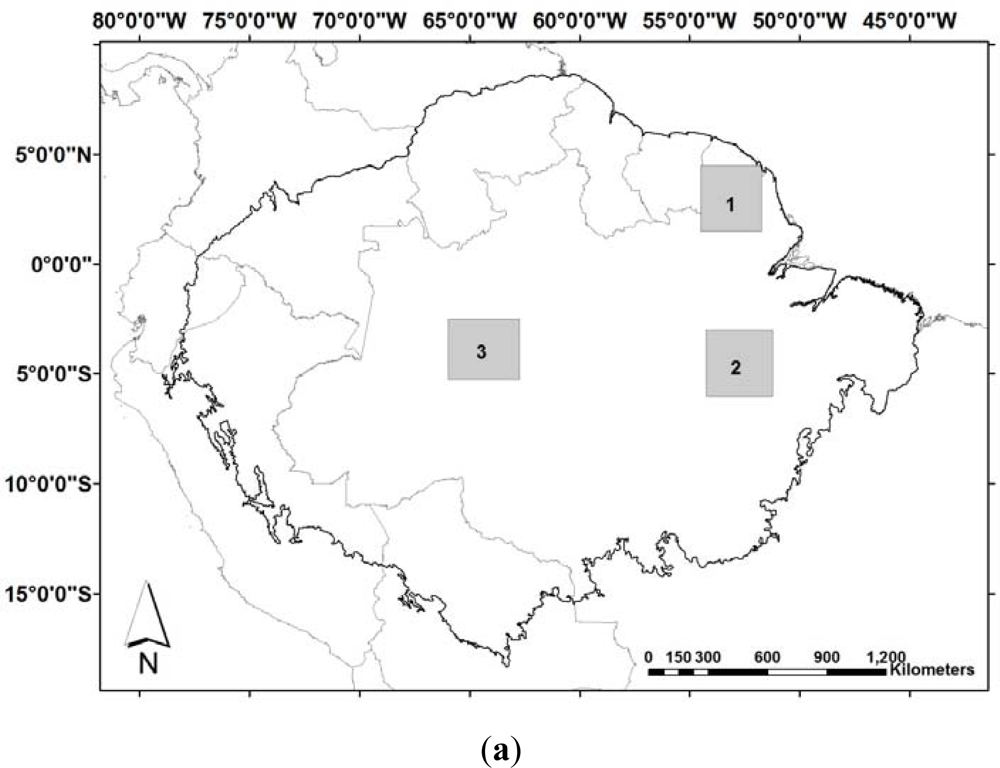
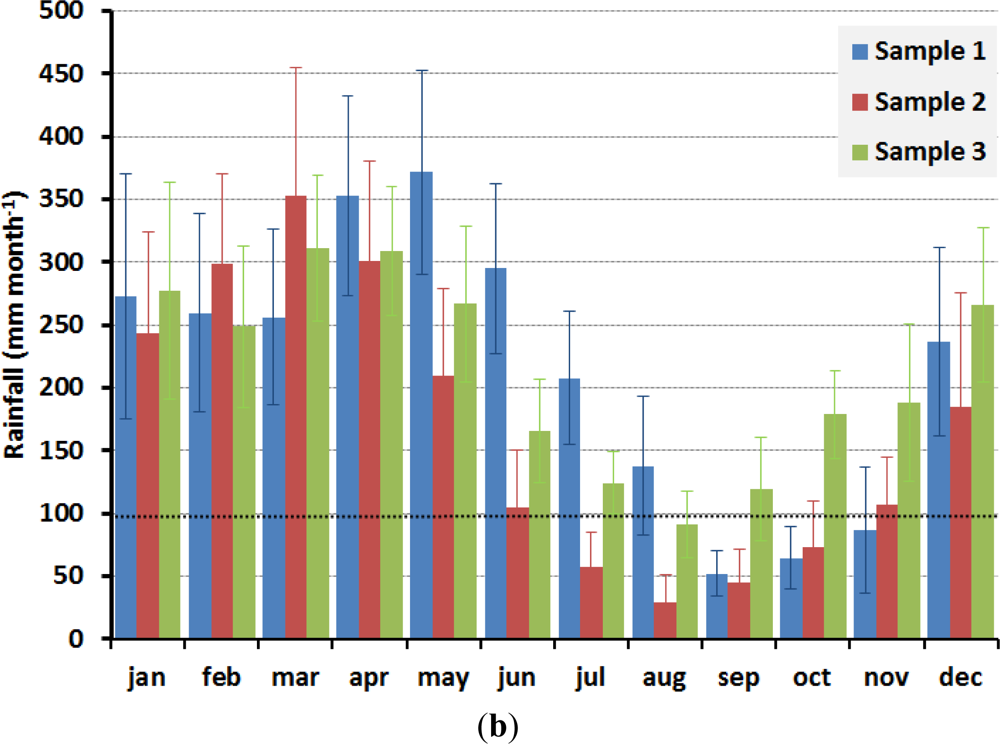
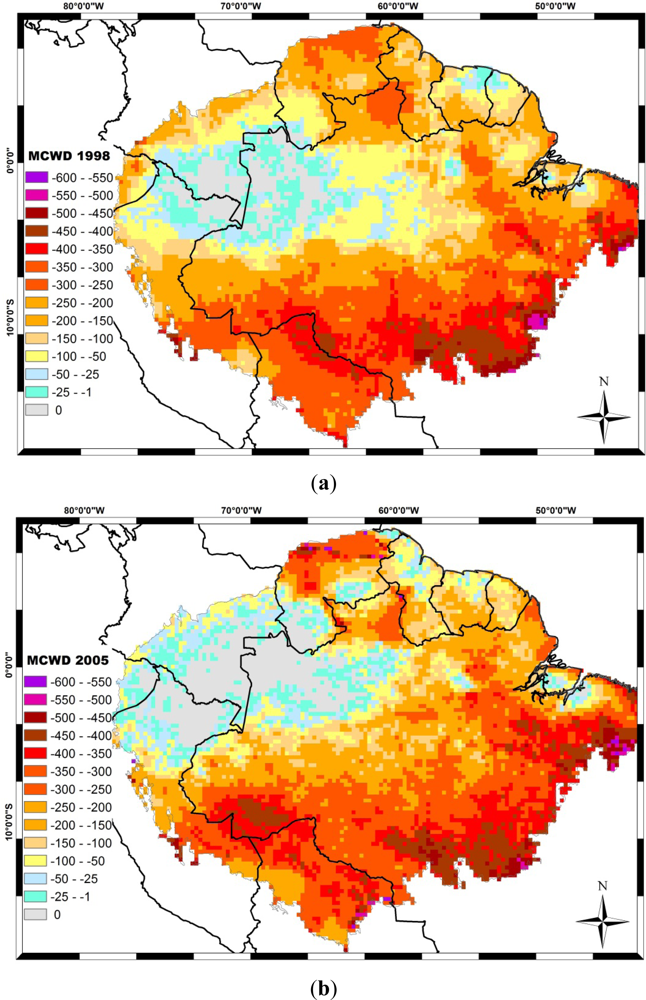
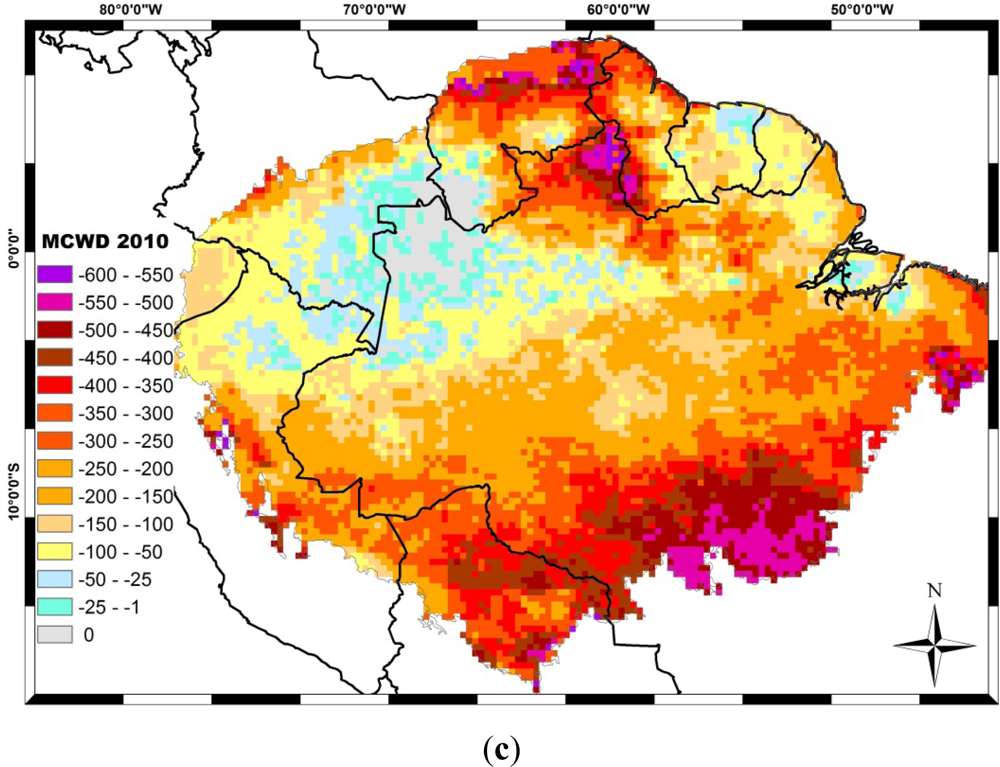
4. Drought Effects
5. Future Directions
Acknowledgments
References
- Malhi, Y.; Wood, D.; Baker, T.R.; Wright, J.; Phillips, O.L.; Cochrane, T.; Meir, P.; Chave, J.; Almeida, S.; Arroyo, L.; et al. Regional variation of above-ground live biomass in old-growth Amazonian forests. Glob. Change Biol 2006, 12, 1–32. [Google Scholar]
- Saatchi, S.S.; Houghton, R.A.; Dos Santos Alvala, R.C.; Soares, J.V.; Yu, Y. Distribution of aboveground live biomass in the Amazon basin. Glob. Change Biol 2007, 13, 816–837. [Google Scholar]
- Field, C.B.; Behrenfeld, M.J.; Randerson, J.T.; Falkowski, P. Primary production of the biosphere: Integrating terrestrial and oceanic components. Science 1998, 281, 237–240. [Google Scholar]
- Malhi, Y.; Grace, J. Tropical forests and atmospheric carbon dioxide. Trends Ecol. Evol 2000, 15, 332–337. [Google Scholar]
- Grace, J. Understanding and managing the global carbon cycle. J. Ecol 2004, 92, 189–202. [Google Scholar]
- Keller, M.; Alencar, A.; Asner, G.P.; Braswell, B.; Bustamente, M.; Davidson, E.; Feldpausch, T.; Fernándes, E.; Goulden, M.; Kabat, P.; et al. Ecological research in the Large-scale Biosphere–Atmosphere experiment in Amazonia: Early results. Ecol. Appl 2004, 14, 3–16. [Google Scholar]
- Magnusson, W.E.; Costa, F.; Lima, A.; Baccaro, F.; Braga-Neto, R.; Laerte Romero, R.; Menin, M.; Penha, J.; Hero, J.-M.; Lawson, B.E. A program for monitoring biological diversity in the Amazon: An alternative perspective to threat-based monitoring. Biotropica 2008, 40, 409–411. [Google Scholar]
- Gedney, N.; Valdes, P.J. The effect of Amazonian deforestation on the northern hemisphere circulation and climate. Geophys. Res. Lett 2000, 27, 3053–3056. [Google Scholar]
- Werth, D.; Avissar, R. The local and global effects of Amazon deforestation. J. Geophys. Res 2002, 107, 8087. [Google Scholar]
- Freitas, S.; Longo, K.; Silva Dias, M.; Silva Dias, P.; Chatfield, R.; Prins, E.; Artaxo, P.; Grell, G.; Recuero, F. Monitoring the transport of biomass burning emissions in South America. Environ. Fluid Mech 2005, 5, 135–167. [Google Scholar]
- ter Steege, H.; Pitman, N.C.A.; Phillips, O.L.; Chave, J.; Sabatier, D.; Duque, A.; Molino, J.-F.; Prevost, M.-F.; Spichiger, R.; Castellanos, H.; et al. Continental-scale patterns of canopy tree composition and function across Amazonia. Nature 2006, 443, 444–447. [Google Scholar]
- Malhado, A.C.M.; Whittaker, R.J.; Malhi, Y.; Ladle, R.J.; ter Steege, H.; Butt, N.; Aragão, L.E.O.C.; Quesada, C.A.; Murakami-Araujo, A.; Phillips, O.L.; et al. Spatial distribution and functional significance of leaf lamina shape in Amazonian forest trees. Biogeosciences 2009, 6, 1577–1590. [Google Scholar]
- Fyllas, N.M.; Patiño, S.; Baker, T.R.; Bielefeld Nardoto, G.; Martinelli, L.A.; Quesada, C.A.; Paiva, R.; Schwarz, M.; Horna, V.; Mercado, L.M.; et al. Basin-wide variations in foliar properties of Amazonian forest: Phylogeny, soils and climate. Biogeosciences 2009, 6, 2677–2708. [Google Scholar]
- Graham, E.A.; Mulkey, S.S.; Kitajima, K.; Phillips, N.G.; Wright, S.J. Cloud cover limits net CO2 uptake and growth of a rainforest tree during tropical rainy seasons. Proc. Natl. Acad. Sci. USA 2003, 100, 572–576. [Google Scholar]
- Oliveira, P.H.F.; Artaxo, P.; Pires, C.; De Lucca, S.; Procopio, A.; Holben, B.; Schafer, J.; Cardoso, L.F.; Wofsy, S.C.; Rocha, H.R. The effects of biomass burning aerosols and clouds on the CO2 flux in Amazonia. Tellus 2007, 59B, 338–349. [Google Scholar]
- Quesada, C.A.; Lloyd, J.; Schwarz, M.; Baker, T.R.; Phillips, O.L.; Patiño, S.; Czimczik, C.; Hodnett, M.G.; Herrera, R.; Arneth, A.; et al. Regional and large-scale patterns in Amazon forest structure and function are mediated by variations in soil physical and chemical properties. Biogeosci. Discuss 2009, 6, 3993–4057. [Google Scholar] [Green Version]
- Clark, D.A. Detecting tropical forests’ responses to global climatic and atmospheric change: Current challenges and a way forward. Biotropica 2007, 39, 4–19. [Google Scholar]
- Fearnside, P.M. Greenhouse gases from deforestation in Brazilian Amazonia: Net committed emissions. Climate Change 1997, 35, 321–360. [Google Scholar]
- Brown, S.; Lugo, A.E. Biomass of tropical forests: A new estimate based on forest volumes. Science 1984, 223, 1290–1293. [Google Scholar]
- Chave, J.; Condit, R.; Aguilar, S.; Hernandez, A.; Lao, S.; Perez, R. Error propagation and scaling for tropical forest biomass estimates. Philos. Trans. R. Soc. Lond. Ser. B 2004, 359, 409–420. [Google Scholar]
- Enquist, B.; West, G.; Charnov, E.; Brown, J. Allometric scaling of production and life-history variation in vascular plants. Nature 1999, 401, 907–911. [Google Scholar]
- Roderick, M.L.; Berry, S.L. Linking wood density with tree growth and environment: A theoretical analysis based on the motion of water. New Phytol 2001, 149, 473–485. [Google Scholar]
- Baker, T.R.; Phillips, O.L.; Malhi, Y.; Almeida, S.; Arroyo, L.; Di Fiore, A.; Erwin, T.; Higuchi, N.; Killeen, T.J.; Laurance, S.G.; et al. Increasing biomass in Amazonian forest plots. Philos. Trans. R. Soc. Lond. Ser. B 2004, 359, 353–365. [Google Scholar]
- Muller-Landau, H.C. Interspecific and inter-site variation in wood specific gravity of tropical trees. Biotropica 2004, 36, 20–32. [Google Scholar]
- Nogueira, E.M.; Fearnside, P.M.; Nelson, B.W.; Barbosa, R.I.; Keizer, E.W.H. Estimates of forest biomass in the Brazilian Amazon: New allometric equations and adjustments to biomass from wood-volume inventories. For. Ecol. Manage 2008, 256, 1853–1867. [Google Scholar]
- Chao, K.-J.; Phillips, O.L.; Gloor, E.; Monteagudo, A.; Torres-Lezama, A.; Martínez, R.V. Growth and wood density predict tree mortality in Amazon forests. J. Ecol 2008, 96, 281–292. [Google Scholar]
- Baker, T.R.; Phillips, O.L.; Malhi, Y.; Almeida, S.; Arroyo, L.; Di Fiore, A.; Erwin, T.; Killeen, T.J.; Laurance, S.G.; Laurance, W.F.; et al. Variation in wood density determines spatial patterns in Amazonian forest biomass. Glob. Change Biol 2004, 10, 545–562. [Google Scholar]
- ter Steege, H.; Hammond, D.S. Character convergence, diversity, and disturbance in tropical rain forest in Guyana. Ecology 2001, 82, 3197–3212. [Google Scholar]
- Keller, M.; Palace, M.; Hurtt, G. Biomass estimation in the Tapajos National Forest, Brazil: Examination of sampling and allometric uncertainties. For. Ecol. Manage 2001, 154, 371–382. [Google Scholar]
- Chave, J.; Andalo, C.; Brown, S.; Cairns, M.; Chambers, J.; Eamus, D.; Fölster, H.; Fromard, F.; Higuchi, N.; Kira, T.; et al. Tree allometry and improved estimation of carbon stocks and balance in tropical forests. Oecologia 2005, 145, 87–99. [Google Scholar]
- Brown, S.; Lugo, A.E. Aboveground biomass estimates for tropical moist forests of the Brazilian Amazon. Interciencia 1992, 17, 8–18. [Google Scholar]
- Chave, J.; Riera, B.; Dubois, M.-A. Estimation of biomass in a Neotropical forest of French Guiana: Spatial and temporal variability. J. Trop. Ecol 2001, 17, 79–96. [Google Scholar]
- Cummings, D.L.; Boone Kauffman, J.; Perry, D.A.; Flint Hughes, R. Aboveground biomass and structure of rainforests in the southwestern Brazilian Amazon. For. Ecol. Manage 2002, 163, 293–307. [Google Scholar]
- de Castilho, C.V.; Magnusson, W.E.; de Araújo, R.N.O.; Luizão, R.C.C.; Luizão, F.J.; Lima, A.P.; Higuchi, N. Variation in aboveground tree live biomass in a central Amazonian Forest: Effects of soil and topography. For. Ecol. Manage 2006, 234, 85–96. [Google Scholar]
- DeFries, R.S.; Townshend, J.R.G.; Hansen, M.C. Continuous fields of vegetation characteristics at the global scale at 1-km resolution. J. Geophys. Res 1999, 104, 16911–16923. [Google Scholar]
- Potter, C.; Brooks Genovese, V.; Klooster, S.; Bobo, M.; Torregrosa, A. Biomass burning losses of carbon estimated from ecosystem modelling and satellite data analysis for the Brazilian Amazon region. Atmos. Environ 2001, 35, 1773–1781. [Google Scholar]
- Lu, D. Aboveground biomass estimation using Landsat TM data in the Brazilian Amazon. Int. J. Remote Sens 2005, 26, 2509–2525. [Google Scholar]
- Houghton, R.A.; Lawrence, K.T.; Hackler, J.L.; Brown, S. The spatial distribution of forest biomass in the Brazilian Amazon: A comparison of estimates. Glob. Change Biol 2001, 7, 731–746. [Google Scholar]
- Ramankutty, N.; Gibbs, H.K.; Achard, F.; Defries, R.; Foley, J.A.; Houghton, R.A. Challenges to estimating carbon emissions from tropical deforestation. Glob. Change Biol 2007, 13, 51–66. [Google Scholar]
- Nelson, R.F.; Kimes, D.S.; Salas, W.A.; Routhier, M. Secondary forest age and tropical forest biomass estimation using thematic mapper imagery. BioScience 2000, 50, 419–431. [Google Scholar]
- Foody, G.M.; Boyd, D.S.; Cutler, M.E.J. Predictive relations of tropical forest biomass from Landsat TM data and their transferability between regions. Remote Sens. Environ 2003, 85, 463–474. [Google Scholar]
- Defries, R.S.; Hansen, M.C.; Townshend, J.R.G.; Janetos, A.C.; Loveland, T.R. A new global 1-km dataset of percentage tree cover derived from remote sensing. Glob. Change Biol 2000, 6, 247–254. [Google Scholar]
- Le Toan, T.; Quegan, S.; Davidson, M.W.J.; Balzter, H.; Paillou, P.; Plummer, S.; Papathanassiou, K.; Rocca, F.; Saatchi, S.; Shugart, H.; et al. The BIOMASS mission: Mapping global forest biomass to better understand the terrestrial carbon cycle. Remote Sens. Environ 2011, 115, 2850–2860. [Google Scholar]
- Saatchi, S.; Harris, N.L.; Brown, S.; Lefsky, M.; Mitchard, E.T.A.; Salas, W.; Zutta, B.R.; Buermann, W.; Lewis, S.L.; Hagen, S.; et al. Benchmark map of forest carbon stocks in tropical regions across three continents. Proc. Natl. Acad. Sci. USA 2011. [Google Scholar] [CrossRef]
- Anderson, L.O.; Malhi, Y.; Ladle, R.J.; Aragão, L.E.O.C.; Shimabukuro, Y.; Phillips, O.L.; Baker, T.; Costa, A.C.L.; Espejo, J.S.; Higuchi, N.; et al. Influence of landscape heterogeneity on spatial patterns of wood productivity, wood specific density and above ground biomass in Amazonia. Biogeosciences 2009, 6, 1883–1902. [Google Scholar]
- Foody, G.M.; Green, R.M.; Lucas, R.M.; Curran, P.J.; Honzak, M.; Amaral, I.D. Observations on the relationship between SIR-C radar backscatter and the biomass of regenerating tropical forests. Int. J. Remote Sens 1997, 18, 687–694. [Google Scholar]
- Luckman, A.; Baker, J.; Honzak, M.; Lucas, R. Tropical forest biomass density estimation using JERS-1 SAR: Seasonal variation, confidence limits, and application to image mosaics. Remote Sens. Environ 1998, 63, 126–139. [Google Scholar]
- Santos, J.R.; Freitas, C.C.; Araujo, L.S.; Dutra, L.V.; Mura, J.C.; Gama, F.F.; Soler, L.S.; Sant’Anna, S.J.S. Airborne P-band SAR applied to the aboveground biomass studies in the Brazilian tropical rainforest. Remote Sens. Environ 2003, 87, 482–493. [Google Scholar]
- Gonçalves, F.G.; Santos, J.R.; Treuhaft, R.N. Stem volume of tropical forests from polarimetric radar. Int. J. Remote Sens 2011. [Google Scholar] [CrossRef]
- Goetz, S.; Baccini, A.; Laporte, N.; Johns, T.; Walker, W.; Kellndorfer, J.; Houghton, R.; Sun, M. Mapping and monitoring carbon stocks with satellite observations: A comparison of methods. Carbon Balance Manage 2009, 4, 2. [Google Scholar]
- Midgley, J.J. Is bigger better in plants? The hydraulic costs of increasing size in trees. Trends Evol. Ecol 2003, 18, 5–6. [Google Scholar]
- Lefsky, M.A. A global forest canopy height map from the moderate resolution imaging spectroradiometer and the geoscience laser altimeter system. Geophys. Res. Lett 2010, 37, L15401. [Google Scholar]
- Simard, M.; Pinto, N.; Fisher, J.B.; Baccini, A. Mapping forest canopy height globally with spaceborne lidar. J. Geophys. Res 2011. [Google Scholar] [CrossRef]
- Dubayah, R.O.; Sheldon, S.L.; Clark, D.B.; Hofton, M.A.; Blair, J.B.; Hurtt, G.C.; Chazdon, R.L. Estimation of tropical forest height and biomass dynamics using lidar remote sensing at La Selva, Costa Rica. J. Geophys. Res 2010. [Google Scholar] [CrossRef]
- Barbier, N.; Couteron, P.; Proisy, C.; Malhi, Y.; Gastellu-Etchegorry, J.-P. The variation of apparent crown size and canopy heterogeneity across lowland Amazonian forests. Glob. Ecol. Biogeogr 2010, 19, 72–84. [Google Scholar]
- Malhado, A.C.M.; Malhi, Y.; Whittaker, R.J.; Ladle, R.J.; ter Steege, H.; Phillips, O.L.; Butt, N.; Aragão, L.E.O.C.; Quesada, C.A.; Araujo-Murakami, A.; et al. Spatial trends in leaf size of Amazonian rainforest trees. Biogeosciences 2009, 6, 1563–1576. [Google Scholar]
- Kerkhoff, A.J.; Enquist, B.J. Ecosystem allometry: The scaling of nutrient stocks and primary productivity across plant communities. Ecol. Lett 2006, 9, 419–427. [Google Scholar]
- Clark, D.A.; Brown, S.; Kicklighter, D.W.; Chambers, J.Q.; Thomlinson, J.R.; Ni, J.; Holland, E.A. Net primary production in tropical forests: An evaluation and synthesis of existing field data. Ecol. Appl 2001, 11, 371–384. [Google Scholar]
- Nepstad, D.C.; Moutinho, P.; Dias-Filho, M.B.; Davidson, E.; Cardinot, G.; Markewitz, D.; Figueiredo, R.; Vianna, N.; Chambers, J.; Ray, D.; et al. The effects of partial throughfall exclusion on canopy processes, aboveground production, and biogeochemistry of an Amazon forest. J. Geophys. Res.-Atmos 2002, 107, 8085:1–8085:18. [Google Scholar]
- Malhi, Y.; Baker, T.R.; Phillips, O.L.; Almeida, S.; Alvarez, E.; Arroyo, L.; Chave, J.; Czimczik, C.I.; Fiore, A.D.; Higuchi, N.; et al. The above-ground coarse wood productivity of 104 Neotropical forest plots. Glob. Change Biol 2004, 10, 563–591. [Google Scholar]
- Aragão, L.E.O.C.; Malhi, Y.; Metcalfe, D.B.; Silva-Espejo, J.E.; Jiménez, E.; Navarrete, D.; Almeida, S.; Costa, A.C.L.; Salinas, N.; Phillips, O.L.; et al. Above- and below-ground net primary productivity across ten Amazonian forests on contrasting soils. Biogeosciences 2009, 6, 2759–2778. [Google Scholar]
- Chave, J.; Navarrete, D.; Almeida, S.; Álvarez, E.; Aragão, L.E.O.C.; Bonal, D.; Châtelet, P.; Silva Espejo, J.; Goret, J.Y.; von Hildebrand, P.; et al. Regional and temporal patterns of litterfall in tropical South America. Biogeosciences 2010, 7, 43–55. [Google Scholar]
- Metcalfe, D.; Meir, P.; Aragão, L.; da Costa, A.; Braga, A.; Gonçalves, P.; de Athaydes Silva Junior, J.; de Almeida, S.; Dawson, L.; Malhi, Y.; et al. The effects of water availability on root growth and morphology in an Amazon rainforest. Plant Soil 2008, 311, 189–199. [Google Scholar]
- Ichii, K.; Maruyama, M.; Yamaguchi, Y. Multi-temporal analysis of deforestation in Rondonia state in Brazil using Landsat MSS, TM, ETM plus and NOAA AVHRR imagery and its relationship to changes in the local hydrological environment. Int. J. Remote Sens 2003, 24, 4467–4479. [Google Scholar]
- Nemani, R.R.; Keeling, C.D.; Hashimoto, H.; Jolly, W.M.; Piper, S.C.; Tucker, C.J.; Myneni, R.B.; Running, S.W. Climate-driven Increases in global terrestrial net primary production from 1982 to 1999. Science 2003, 300, 1560–1563. [Google Scholar]
- Myneni, R.B.; Yang, W.; Nemani, R.R.; Huete, A.R.; Dickinson, R.E.; Knyazikhin, Y.; Didan, K.; Fu, R.; Negrón Juárez, R.I.; Saatchi, S.S.; et al. Large seasonal swings in leaf area of Amazon rainforests. Proc. Natl. Acad. Sci. USA 2007, 104, 4820–4823. [Google Scholar]
- Running, S.W.; Nemani, R.R.; Heinsch, F.A.; Zhao, M.; Reeves, M.; Hashimoto, H. A continuous satellite-derived measure of global terrestrial primary production. BioScience 2008, 54, 547–560. [Google Scholar]
- Poulter, B.; Cramer, W. Satellite remote sensing of tropical forest canopies and their seasonal dynamics. Int. J. Remote Sens 2009, 30, 6575–6590. [Google Scholar]
- Malhi, Y.; Aragão, L.E.O.C.; Metcalfe, D.B.; Paiva, R.; Quesada, C.A.; Almeida, S.; Anderson, L.; Brando, P.; Chambers, J.Q.; Da Costa, A.C.L.; et al. Comprehensive assessment of carbon productivity, allocation and storage in three Amazonian forests. Glob. Change Biol 2009, 15, 1255–1274. [Google Scholar]
- Potter, C.; Klooster, S.; de Carvalho, C.R.; Genovese, V.B.; Torregrosa, A.; Dungan, J.; Bobo, M.; Coughlan, J. Modelling seasonal and interannual variability in ecosystem carbon cycling for the Brazilian Amazon region. J. Geophys. Res 2001, 106, 10423–10446. [Google Scholar]
- Tian, H.; Melillo, J.M.; Kicklighter, D.W.; McGuire, A.D.; Helfrich, J.V.K.; Moore, B.; Vorosmarty, C.J. Effect of interannual climate variability on carbon storage in Amazonian ecosystems. Nature 1998, 396, 664–667. [Google Scholar]
- Sommer, R.; Fölster, H.; Vielhauer, K.; Carvalho, E.J.M.; Vlek, P.L.G. Deep soil water dynamics and depletion by secondary vegetation in the Eastern Amazon. Soil Sci. Soc. Am. J 2003, 67, 1672–1686. [Google Scholar]
- Rivera, G.; Elliott, S.; Caldas, L.; Nicolossi, G.; Coradin, V.; Borchert, R. Increasing day-length induces spring flushing of tropical dry forest trees in the absence of rain. Trees Struct. Funct 2002, 16, 445–456. [Google Scholar]
- Huete, A.R.; Didan, K.; Shimabukuro, Y.E.; Ratana, P.; Saleska, S.R.; Hutyra, L.R.; Yang, W.; Nemani, R.R.; Myneni, R. Amazon rainforests green-up with sunlight in dry season. Geophys. Res. Lett 2006, 33. [Google Scholar] [CrossRef]
- Rathcke, B.; Lacey, E.P. Phenological patterns of terrestrial plants. Annu. Rev. Ecol. Syst 1985, 16, 179–214. [Google Scholar]
- Chabot, B.F.; Hicks, D.J. The ecology of leaf life spans. Annu. Rev. Ecol. Syst 1982, 13, 229–259. [Google Scholar]
- Aide, T.M. Herbivory as a selective agent on the timing of leaf production in a tropical understory community. Nature 1988, 336, 574–575. [Google Scholar]
- Prance, G. Notes on the vegetation of Amazonia III. The terminology of Amazonian forest types subject to inundation. Brittonia 1979, 31, 26–38. [Google Scholar]
- Peres, C.A. Primate responses to phenological changes in an Amazonian terra firme forest. Biotropica 1994, 26, 98–112. [Google Scholar]
- Ferreira, L.; Parolin, P. Tree phenology in central Amazonian Floodplain forests: Effects of water level Fluctuation and precipitation at community and population level. Pesquisas Botânica 2007, 58, 139–156. [Google Scholar]
- Gribel, R.; Gibbs, P.E.; Queiroz, A.L. Flowering phenology and pollination biology of Ceiba pentandra (Bombacaceae) in Central Amazonia. J. Trop. Ecol 1999, 15, 247–263. [Google Scholar]
- Parolin, P. Phenology and CO2-assimilation of trees in Central Amazonian floodplains. J. Trop. Ecol 2000, 16, 465–473. [Google Scholar]
- Haugaasen, T.; Peres, C.A. Mammal assemblage structure in Amazonian flooded and unflooded forests. J. Trop. Ecol 2005, 21, 133–145. [Google Scholar]
- Parolin, P.; Armbruster, N.; Junk, W.J. Two Amazonian floodplain trees react differently to periodical flooding. Trop. Ecol 2006, 47, 243–250. [Google Scholar]
- Basset, Y. Insect herbivores foraging on seedlings in an unlogged rain forest in Guyana: Spatial and temporal considerations. Stud. Neotrop. Fauna E 2000, 35, 115–129. [Google Scholar]
- Brienen, R.; Zuidema, P. Relating tree growth to rainfall in Bolivian rain forests: A test for six species using tree ring analysis. Oecologia 2005, 146, 1–12. [Google Scholar]
- Borchert, R. Responses of tropical trees to rainfall seasonality and its long-term changes. Climate Change 1998, 39, 381–393. [Google Scholar]
- Aragão, L.E.O.C.; Malhi, Y.; Roman-Cuesta, R.M.; Saatchi, S.; Anderson, L.O.; Shimabukuro, Y.E. Spatial patterns and fire response of recent Amazonian droughts. Geophys. Res. Lett 2007, 34, L07701. [Google Scholar] [CrossRef]
- Stubblebine, W.; Langenheim, J.H.; Lincoln, D. Vegetative response to photoperiod in the tropical leguminous tree hymenaea courbaril L. Biotropica 1978, 10, 18–29. [Google Scholar]
- Wright, S.J.; van Schaik, C.P. Light and the phenology of tropical trees. Am. Nat 1994, 143, 192–199. [Google Scholar]
- Mulkey, S.S.; Kitajima, K.; Wright, S.J. Plant physiological ecology of tropical forest canopies. Trends Ecol. Evol 1996, 11, 408–412. [Google Scholar]
- Brando, P.; Ray, D.; Nepstad, D.; Cardinot, G.; Curran, L.; Oliveira, R. Effects of partial throughfall exclusion on the phenology of Coussarea racemosa (Rubiaceae) in an east-central Amazon rainforest. Oecologia 2006, 150, 181–189. [Google Scholar]
- Defler, T.; Defler, S. Diet of a group of Lagothrix Lagothricha Lagothricha in southeastern Colombia. Int. J. Primatol 1996, 17, 161–190. [Google Scholar]
- Kitajima, K.; Mulkey, S.S.; Wright, S.J. Seasonal leaf phenotypes in the canopy of a tropical dry forest: Photosynthetic characteristics and associated traits. Oecologia 1997, 109, 490–498. [Google Scholar]
- Reich, P.B.; Uhl, C.; Walters, M.B.; Prugh, L.; Ellsworth, D.S. Leaf demography and phenology in Amazonian rain forest: A census of 40 000 leaves of 23 tree species. Ecol. Monogr 2004, 74, 3–23. [Google Scholar]
- Ruiz, J.E.A.; da Cruz Alencar, J. Interpretação fenológica de cinco espécies de Chrysobalanaceae na Reserva Florestal Adolpho Ducke, Manaus, Amazonas, Brasil. Acta Amazonica 1999, 29, 223–242. [Google Scholar]
- Miura, T.; Huete, A.R.; van Leeuwen, W.J.D.; Didan, K. Vegetation detection through smoke-filled AVIRIS images: An assessment using MODIS band passes. J. Geophys. Res 1998, 103, 32001–32011. [Google Scholar]
- Kobayashi, H.; Dye, D.G. Atmospheric conditions for monitoring the long-term vegetation dynamics in the Amazon using normalized difference vegetation index. Remote Sens. Environ 2005, 97, 519–525. [Google Scholar]
- Asner, G.P. Cloud cover in Landsat observations of the Brazilian Amazon. Int. J. Remote Sens 2001, 22, 3855–3862. [Google Scholar]
- Justice, C.O.; Townshend, J.R.G.; Vermote, E.F.; Masuoka, E.; Wolfe, R.E.; Saleous, N.; Roy, D.P.; Morisette, J.T. An overview of MODIS Land data processing and product status. Remote Sens. Environ 2002, 83, 3–15. [Google Scholar]
- Myneni, R.B.; Hall, F.G.; Sellers, P.J.; Marshak, A.L. Marshak.. The interpretation of spectral vegetation indexes. IEEE Trans. Geosci. Remote Sens 1995, 33, 481–486. [Google Scholar]
- Spanner, M.A.; Pierce, L.L.; Running, S.W.; Peterson, D.L. The seasonality of AVHRR data of temperate coniferous forests: Relationship with leaf area index. Remote Sens. Environ 1990, 33, 97–112. [Google Scholar]
- Reed, B.C.; Brown, J.F.; VanderZee, D.; Loveland, T.R.; Merchant, J.W.; Ohlen, D.O. Measuring phenological variability from satellite imagery. J. Veg. Sci 1994, 5, 703–714. [Google Scholar]
- Batista, G.T.; Shimabukuro, Y.E.; Lawrence, W.T. The Long-term monitoring of vegetation cover in the Amazon region in Northern Brazil using NOAA-AVHRR data. Int. J. Remote Sens 1997, 18, 3195–3210. [Google Scholar]
- Duchemin, B.; Guyon, D.; Lagouarde, J.P. Potential and limits of NOAA-AVHRR temporal composite data for phenology and water stress monitoring of temperate forest ecosystems. Int. J. Remote Sens 1999, 20, 895–917. [Google Scholar]
- Asner, G.P.; Townsend, A.R.; Braswell, B.H. Satellite observation of el niño effects on amazon forest phenology and productivity. Geophys. Res. Lett 2000, 27, 981–984. [Google Scholar]
- Azzali, S.; Menenti, M. Mapping vegetation-soil-climate complexes in southern Africa using temporal Fourier analysis of NOAA-AVHRR NDVI data. Int. J. Remote Sens 2000, 21, 973–996. [Google Scholar]
- Lu, H.; Raupach, M.R.; McVicar, T.R.; Barrett, D.J. Decomposition of vegetation cover into woody and herbaceous components using AVHRR NDVI time series. Remote Sens. Environ 2003, 86, 1–18. [Google Scholar]
- Huete, A. A soil-adjusted vegetation index (SAVI). Remote Sens. Environ 1988, 25, 295–309. [Google Scholar]
- Kaufman, Y.J.; Tanré, D. Atmospherically resistant vegetation index (ARVI) for EOS-MODIS. IEEE Trans. Geosci. Remote Sens 1992, 30, 261–270. [Google Scholar]
- Huete, A.; Justice, C.; Liu, H. Development of vegetation and soil indices for MODIS-EOS. Remote Sens. Environ 1994, 49, 224–234. [Google Scholar]
- Huete, A.R.; Liu, H.Q.; Batchily, K.; van Leeuwen, W. Comparison of vegetation indices over a global set of TM images for EOS-MODIS. Remote Sens. Environ 1997, 59, 440–451. [Google Scholar]
- Tanre, D.; Holben, B.N.; Kaufman, Y.J. Atmospheric correction against algorithm for NOAA-AVHRR products: Theory and application. IEEE Trans. Geosci. Remote Sens 1992, 30, 231–248. [Google Scholar]
- Privette, J.L.; Fowler, C.; Wick, G.A.; Baldwin, D.; Emery, W.J. Effects of orbital drift on advanced very high resolution radiometer products: Normalized difference vegetation index and sea surface temperature. Remote Sens. Environ 1995, 53, 164–171. [Google Scholar]
- Rouse, J.; Haas, R.; Schell, J.; Deering, D. Monitoring Vegetation Systems in the Great Plains with ERTS. In Third Earth Resources Technology Satellite-1 Symposium; NASA: Greenbelt, MD, USA, 1974; pp. 301–317. [Google Scholar]
- Tucker, C.J. Red and photographic infrared linear combinations for monitoring vegetation. Remote Sens. Environ 1979, 8, 127–150. [Google Scholar]
- Justice, C.O.; Eck, T.F.; Tanré, D.; Holben, B.N. The effect of water vapour on the normalized difference vegetation index derived for the Sahelian region from NOAA AVHRR data. Int. J. Remote Sens 1991, 12, 1165–1187. [Google Scholar]
- Kogan, F.N. Remote sensing of weather impacts on vegetation in non-homogeneous areas. Int. J. Remote Sens 1990, 11, 1405–1419. [Google Scholar]
- Kogan, F.N. Application of vegetation index and brightness temperature for drought detection. Adv. Space Res 1995, 15, 91–100. [Google Scholar]
- Kogan, F.N. Global drought watch from space. Bull. Am. Meteorol. Soc 1997, 78, 621–636. [Google Scholar]
- Kogan, F.; Stark, R.; Gitelson, A.; Jargalsaikhan, L.; Dugrajav, C.; Tsooj, S. Derivation of pasture biomass in Mongolia from AVHRR-based vegetation health indices. Int. J. Remote Sens 2004, 25, 2889–2896. [Google Scholar]
- Karnieli, A.; Gabai, A.; Ichoku, C.; Zaady, E.; Shachak, M. Temporal dynamics of soil and vegetation spectral responses in a semi-arid environment. Int. J. Remote Sens 2002, 23, 4073–4087. [Google Scholar]
- Palmer, W.C. Meteorological Drought; US Department of Commerce, Weather Bureau: Washington, DC, USA, 1965; pp. 1–58. [Google Scholar]
- Walsh, S. Comparison of NOAA AVHRR data to meteorologic drought indices. Photogramm. Eng. Remote. Sens 1987, 53, 1069–1074. [Google Scholar]
- Di, L. Regional-Scale Soil Moisture Monitoring Using NOAA/AVHRR Data; ETD Collection for University of Nebraska-Lincoln Paper AAI9129546; University of Nebraska-Lincoln: Lincoln, NE, USA, 1991. [Google Scholar]
- Gao, B.C. NDWI: A Normalized Difference Water Index for remote sensing of vegetation liquid water from space. Remote Sens. Environ 1996, 58, 257–266. [Google Scholar]
- Zarco-Tejada, P.J.; Rueda, C.A.; Ustin, S.L. Water content estimation in vegetation with MODIS reflectance data and model inversion methods. Remote Sens. Environ 2003, 85, 109–124. [Google Scholar]
- Xiao, X.; Boles, S.; Liu, J.; Zhuang, D.; Liu, M. Characterization of forest types in Northeastern China, using multitemporal SPOT-4 VEGETATION sensor data. Remote Sens. Environ 2002, 82, 335–348. [Google Scholar]
- Asner, G.P.; Carlson, K.M.; Martin, R.E. Substrate age and precipitation effects on Hawaiian forest canopies from spaceborne imaging spectroscopy. Remote Sens. Environ 2005, 98, 457–467. [Google Scholar]
- D’Souza, G.; Malingreau, J.P. Malingreau. NOAA-AVHRR studies of vegetation characteristics and deforestation mapping in the Amazon Basin. Remote Sens. Reviron 1994, 10, 5–34. [Google Scholar]
- Liu, W.T.; Kogan, F.N. Monitoring regional drought using the Vegetation Condition Index. Int. J. Remote Sens 1996, 14, 2761–2782. [Google Scholar]
- Santos, P.; Negri, A.J. A comparison of the normalized difference vegetation index and rainfall for the Amazon and Northeastern Brazil. J. App. Meteorol 1997, 36, 958–965. [Google Scholar]
- Xiao, X.; Hagen, S.; Zhang, Q.; Keller, M.; Moore, B., III. Detecting leaf phenology of seasonally moist tropical forests in South America with multi-temporal MODIS images. Remote Sens. Environ 2006, 103, 465–473. [Google Scholar]
- Brando, P.M.; Goetz, S.J.; Baccini, A.; Nepstad, D.C.; Beck, P.S.A.; Christman, M.C. Seasonal and interannual variability of climate and vegetation indices across the Amazon. Proc. Natl. Acad. Sci. USA 2010, 107, 14685–14690. [Google Scholar]
- Galvão, L.S.; Santos, J.R.; Roberts, D.A.; Breuning, F.M.; Tomey, M.; Moura, Y.M. On the intra-annual EVI variability in the dry season of tropical forest: A case study with MODIS and hyperspectral data. Remote Sens. Environ 2011, 115, 2350–2359. [Google Scholar]
- Liebmann, B.; Camargo, S.J.; Seth, A.; Marengo, J.A.; Carvalho, L.M.V.; Allured, D.; Fu, R.; Vera, C.S. Onset and end of the rainy season in South America in observations and the ECHAM 4.5 atmospheric general circulation model. J. Clim 2007, 20, 2037–2050. [Google Scholar]
- Marengo, J.A.; Nobre, C.A. The hydroclimatological Framework in Amazonia. In Biogeochemistry of Amazonia; Richey, J., McClaine, M., Victoria, R., Eds.; Oxford University Press: Oxford, UK, 2001; pp. 17–42. [Google Scholar]
- Marengo, J.A.; Nobre, C.A.; Tomasella, J.; Oyama, M.; Sampaio, G.; Camargo, H.; Alves, L.M. The drought of Amazonia in 2005. J. Clim 2008, 21, 495–516. [Google Scholar]
- Williamson, G.B.; Laurance, W.F.; Oliveira, A.A.; Delamônica, P.; Gascon, C.; Lovejoy, T.E.; Pohl, L. Amazonian tree mortality during the 1997 El Niño Drought. Conserv. Biol 2000, 14, 1538–1542. [Google Scholar]
- Saleska, S.R.; Didan, K.; Huete, A.R.; da Rocha, H.R. Amazon forests green-up during 2005 drought. Science 2007, 318, 612. [Google Scholar]
- Phillips, O.L.; Aragão, L.E.O.C.; Lewis, S.L.; Fisher, J.B.; Lloyd, J.; Lopez-Gonzalez, G.; Malhi, Y.; Monteagudo, A.; Peacock, J.; Quesada, C.A.; et al. Drought sensitivity of the Amazon rainforest. Science 2009, 323, 1344–1347. [Google Scholar]
- Samanta, A.; Ganguly, S.; Myneni, R.B. MODIS Enhanced Vegetation Index data do not show greening of Amazon forests during the 2005 drought. New Phytol 2011, 189, 11–15. [Google Scholar]
- Anderson, L.O.; Malhi, Y.; Aragão, L.E.O.C.; Ladle, R.J.; Arai, E.; Barbier, N.; Phullips, O. Remote sensing detection of droughts in Amazonian forest canopies. New Phytol 2010, 187, 733–750. [Google Scholar]
- Anderson, L.O.; Shimabukuro, Y.E.; Aragão, L.E.O.; Huete, A. Fraction images derived from Terra/MODIS data: Monitoring intra-annual phenology in Amazonia. Int. J. Remote Sens 2011, 32, 387–408. [Google Scholar]
- Lewis, L.S.; Brando, P.M.; Phillips, O.L.; van der Heijden, G.M.F.; Nepstad, D. The 2010 Amazon Drought. Science 2011, 331, 554. [Google Scholar]
- Xu, L.; Samanta, A.; Costa, M.H.; Ganguly, S.; Nemani, R.R.; Myneni, R.B. Widespread decline in greenness of Amazonian vegetation due to the 2010 drought. Geophys. Res. Lett 2011, 38. [Google Scholar] [CrossRef]
- Aragão, L.E.O.C.; Shimabukuro, Y.E. The incidence of fire in Amazonian Forests with implications for REDD. Science 2010, 328, 1275–1278. [Google Scholar]
- Cardozo, F.S.; Shimabukuro, Y.E.; Pereira, G.; Silva, F.B. Using remote sensing products for environmental analysis in South America. Remote Sens 2011, 3, 2110–2127. [Google Scholar]
- Shimabukuro, Y.E.; Duarte, V.; Arai, E.; Freitas, R.M.; Lima, A.; Valeriano, D.M.; Brown, I.F.; Maldonado, M.L.R. Fraction images derived from Terra Modis data for mapping burnt areas in Brazilian Amazonia. Int. J. Remote Sens 2009, 30, 1537–1546. [Google Scholar]
- Aragão, L.E.O.C.; Shimabukuro, Y.E.; Espírito Santo, F.D.B.; Williams, M. Landscape pattern and spatial variability of leaf area index in eastern Amazonia. For. Ecol. Manage 2005, 211, 240–256. [Google Scholar]
- Salovaara, K.J.; Thessler, S.; Malik, R.N.; Tuomisto, H. Classification of Amazonian primary rain forest vegetation using Landsat ETM+ satellite imagery. Remote Sens. Environ 2005, 97, 39–51. [Google Scholar]
- Barlow, J.; Ewers, R.M.; Anderson, L.; Aragao, L.E.O.C.; Baker, T.R.; Boyd, E.; Feldpausch, T.R.; Gloor, E.; Hall, A.; Malhi, Y.; et al. Using learning networks to understand complex systems: A case study of biological, geophysical and social research in the Amazon. Biol. Rev 2010. [Google Scholar] [CrossRef] [Green Version]

Share and Cite
Anderson, L.O. Biome-Scale Forest Properties in Amazonia Based on Field and Satellite Observations. Remote Sens. 2012, 4, 1245-1271. https://doi.org/10.3390/rs4051245
Anderson LO. Biome-Scale Forest Properties in Amazonia Based on Field and Satellite Observations. Remote Sensing. 2012; 4(5):1245-1271. https://doi.org/10.3390/rs4051245
Chicago/Turabian StyleAnderson, Liana O. 2012. "Biome-Scale Forest Properties in Amazonia Based on Field and Satellite Observations" Remote Sensing 4, no. 5: 1245-1271. https://doi.org/10.3390/rs4051245
APA StyleAnderson, L. O. (2012). Biome-Scale Forest Properties in Amazonia Based on Field and Satellite Observations. Remote Sensing, 4(5), 1245-1271. https://doi.org/10.3390/rs4051245



