Remote Sensing Data-Based Modelling for Analyzing Green Tide Proliferation Drivers in the Yellow Sea
Highlights
- Changes in attachment substrates (e.g., aquaculture rafts) are identified as a significant contributing factor to the 2021 Yellow Sea green tide outbreak, exerting dual effects on green tide dynamics.
- Substrate alterations increase the wind drag coefficient of Ulva prolifera, accelerating northward drift and coastal accumulation, while also enhancing spore germination to boost initial biomass.
- The findings provide a scientific basis for refining green tide prevention strategies, such as strengthening the management of aquaculture raft disposal during extreme weather.
- The findings improve understanding of green tide formation mechanisms, highlighting the need to integrate substrate-related factors into future green tide causal analysis and prediction frameworks.
Abstract
1. Introduction
2. Materials and Methods
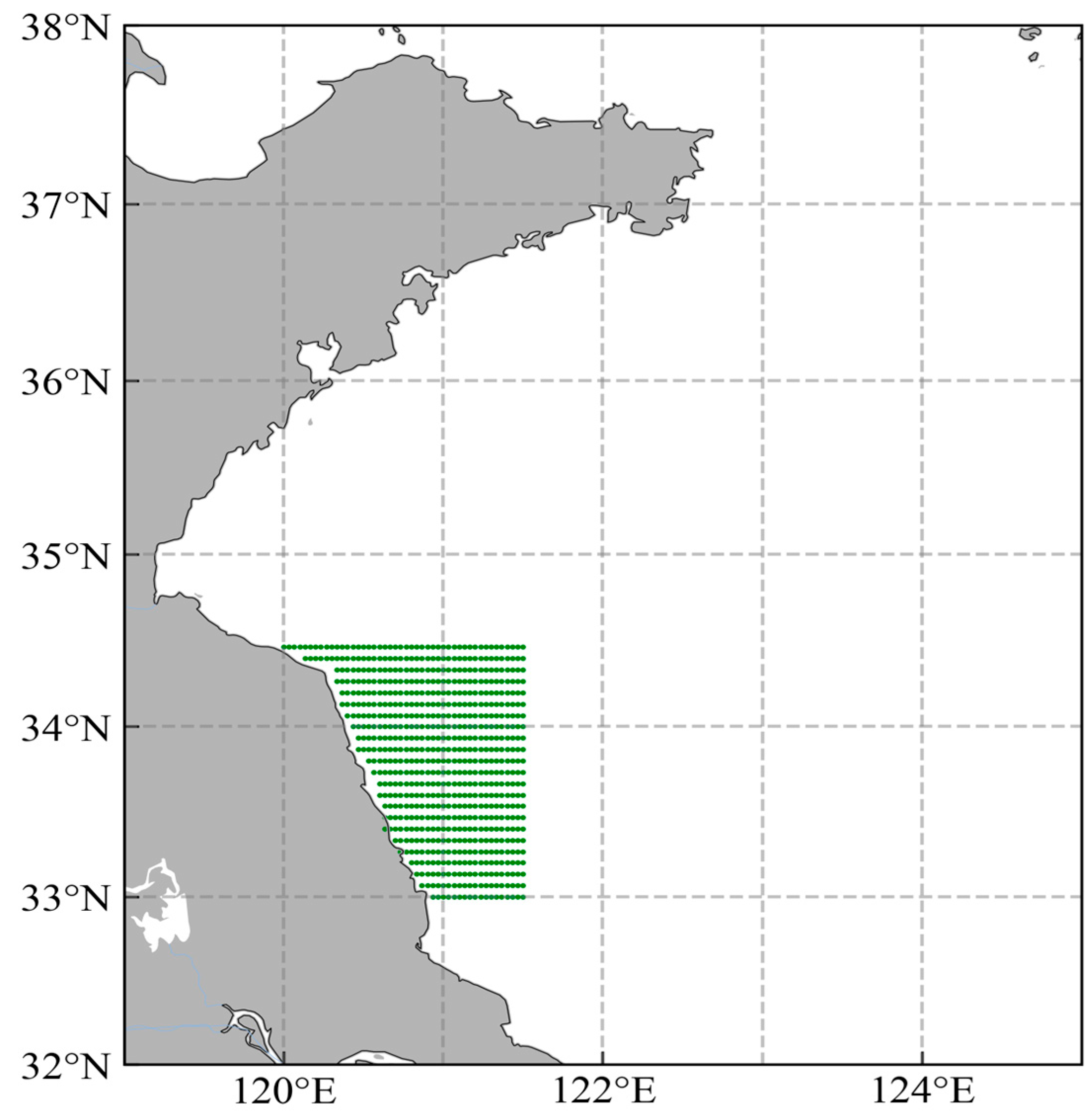
2.1. Model Description
2.2. Data Resources
2.3. Model Setup
2.4. Simulation Design
3. Results
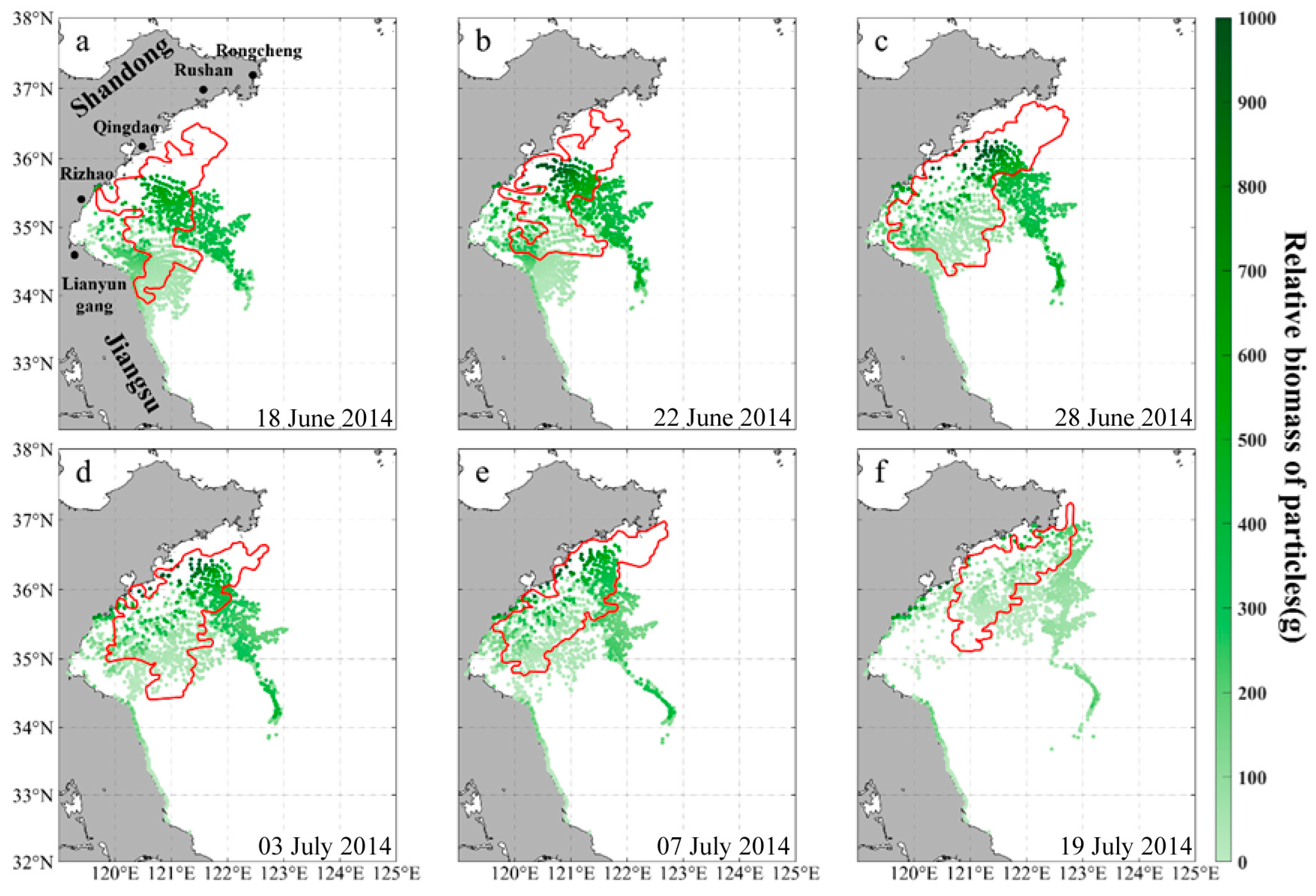
3.1. Validation of Green Tide Development in 2014 and 2015
3.2. The Temporal and Spatial Distribution of the Green Tide in 2021
3.3. Analysis of the Causes of Massive Green Tide Biomass Outbreak in 2021
3.4. Impact of Changes in Substrate on Drift Paths
3.5. Impact of Changes in Substrate on Biomass
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Yang, L.; Zhang, R.; Zhuo, L.; Xie, B.-B.; Lin, J.; Li, S. Stemming Green Tides in the Yellow Sea. Science 2025, 390, 248. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Liu, J.; Xia, J.; Tong, Y.; Li, C.; Zhao, S.; Zhuang, M.; Zhao, X.; Zhang, J.; He, P. Research Development on Resource Utilization of Green Tide Algae from the Southern Yellow Sea. Energy Rep. 2022, 8, 295–303. [Google Scholar] [CrossRef]
- Sun, Y.; Yao, L.; Liu, J.; Tong, Y.; Xia, J.; Zhao, X.; Zhao, S.; Fu, M.; Zhuang, M.; He, P.; et al. Prevention Strategies for Green Tides at Source in the Southern Yellow Sea. Mar. Pollut. Bull. 2022, 178, 113646. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; He, P.; Li, H.; Li, G.; Liu, J.; Jiao, F.; Zhang, J.; Huo, Y.; Shi, X.; Su, R.; et al. Ulva prolifera Green-Tide Outbreaks and Their Environmental Impact in the Yellow Sea, China. Natl. Sci. Rev. 2019, 6, 825–838. [Google Scholar] [CrossRef]
- Zhou, M.-J.; Liu, D.-Y.; Anderson, D.M.; Valiela, I. Introduction to the Special Issue on Green Tides in the Yellow Sea. Estuar. Coast. Shelf Sci. 2015, 163, 3–8. [Google Scholar] [CrossRef]
- Wang, C.; Yu, R.; Zhou, M. Acute toxicity of live and decomposing green alga Ulva (Enteromorpha) prolifera to abalone Haliotis discus hannai. Chin. J. Oceanol. Limnol. 2011, 29, 541–546. [Google Scholar] [CrossRef]
- Ministry of Natural Resources (MNR). The Bulletin of China Marine Disaster; Ministry of Natural Resources of the People’s Republic of China: Beijing, China, 2022. [Google Scholar]
- Yan, T.; Yu, R.; Zhou, M.; Sun, S. Mechanism of Massive Formation and Prevention Strategy against Large-Scale Green Tides in the South Yellow Sea. Oceanol. Limnol. Sin. 2018, 49, 950–958. (In Chinese) [Google Scholar] [CrossRef]
- He, E.; Ji, X.; Gao, S.; Zhao, L.; Wang, Y.; Li, Y.; Yang, J. Numerical Simulation and Forecasting of Drift, Growth, and Death of Enteromorpha in the Yellow Sea. Oceanol. Limnol. Sin. 2021, 52, 39–50. [Google Scholar]
- Hu, L.; Zeng, K.; Hu, C.; He, M.-X. On the Remote Estimation of Ulva prolifera Areal Coverage and Biomass. Remote Sens. Environ. 2019, 223, 194–207. [Google Scholar] [CrossRef]
- Li, D.; Gao, Z.; Wang, Z. Analysis of the Reasons for the Outbreak of Yellow Sea Green Tide in 2021 Based on Long-Term Multi-Source Data. Mar. Environ. Res. 2022, 178, 105649. [Google Scholar] [CrossRef]
- Zheng, L.; Wu, M.; Cui, Y.; Tian, L.; Yang, P.; Zhao, L.; Xue, M.; Liu, J. What Causes the Great Green Tide Disaster in the South Yellow Sea of China in 2021? Ecol. Indic. 2022, 140, 108988. [Google Scholar] [CrossRef]
- Song, M.; Kong, F.; Li, Y.; Zhao, J.; Yu, R.; Zhou, M.; Jiang, P.; Yan, T. A Massive Green Tide in the Yellow Sea in 2021: Field Investigation and Analysis. Int. J. Environ. Res. Public Health 2022, 19, 11753. [Google Scholar] [CrossRef] [PubMed]
- Geng, H.; Yan, T.; Zhou, M.; Liu, Q. Comparative Study of the Germination of Ulva prolifera Gametes on Various Substrates. Estuar. Coast. Shelf Sci. 2015, 163, 89–95. [Google Scholar] [CrossRef]
- Liu, F.; Pang, S.J.; Zhao, X.B.; Hu, C.M. Quantitative, Molecular and Growth Analyses of Ulva Microscopic Propagules in the Coastal Sediment of Jiangsu Province Where Green Tides Initially Occurred. Mar. Environ. Res. 2012, 74, 56–63. [Google Scholar] [CrossRef]
- Geng, H.; Yan, T.; Yu, R.; Zhang, Q.; Kong, F.; Zhou, M. Comparative Study on Germination of Ulva prolifera Spores on Different Substrates. Oceanol. Limnol. Sin. 2018, 49, 1006–1013. (In Chinese) [Google Scholar] [CrossRef]
- Li, S.; Yu, K.; Huo, Y.; Zhang, J.; Wu, H.; Cai, C.; Liu, Y.; Shi, D.; He, P. Effects of Nitrogen and Phosphorus Enrichment on Growth and Photosynthetic Assimilation of Carbon in a Green Tide-Forming Species (Ulva prolifera) in the Yellow Sea. Hydrobiologia 2016, 776, 161–171. [Google Scholar] [CrossRef]
- Xiao, J.; Zhang, X.; Gao, C.; Jiang, M.; Li, R.; Wang, Z.; Li, Y.; Fan, S.; Zhang, X. Effect of Temperature, Salinity and Irradiance on Growth and Photosynthesis of Ulva prolifera. Acta Oceanolog. Sin. 2016, 35, 114–121. [Google Scholar] [CrossRef]
- Feng, L.; Shi, X.; Chen, Y.; Tang, H.; Wang, L. Effects of Temperature on the Nitrate Reductase Activity and Growth of Ulva prolifera. J. Phycol. 2021, 57, 955–966. [Google Scholar] [CrossRef]
- Moisan, J.R.; Moisan, T.A.; Abbott, M.R. Modelling the Effect of Temperature on the Maximum Growth Rates of Phytoplankton Populations. Ecol. Modell. 2002, 153, 197–215. [Google Scholar] [CrossRef]
- Steele, J.H. Environmental Control of Photosynthesis in the Sea. Limnol. Oceanogr. 1962, 7, 137–150. [Google Scholar] [CrossRef]
- Richard, W.E. Temperature and Phytoplankton Growth in the Sea. Fish. Bull. 1971, 70, 1063. [Google Scholar]
- Wang, S.; Zhao, L.; Zhang, H.; Li, F. Green Tide in the Yellow Sea from Generation to Extinction and the Controlling Factor. Oceanol. Limnol. Sin. 2022, 53, 1338–1348. (In Chinese) [Google Scholar] [CrossRef]
- Jiang, X.W.; Liu, J.Q.; Zou, B.; Wang, Q.M.; Zeng, T.; Guo, M.H.; Zhu, H.T.; Zou, Y.R.; Tang, J.W. The Satellite Remote Sensing System Used in Emergency Response Monitoring for Entermorpha Prolifera Disaster and Its Application. Acta Oceanolog. Sin. 2009, 31, 52–64. (In Chinese) [Google Scholar]
- Pan, X.; Cao, M.; Zheng, L.; Xiao, Y.; Qi, L.; Xing, Q.; Kim, K.; Sun, D.; Wang, N.; Guo, M.; et al. Remote Sensing of Ulva prolifera Green Tide in the Yellow Sea Using Multisource Satellite Data: Progress and Prospects. IEEE Geosci. Remote Sens. Mag. 2024, 12, 110–131. [Google Scholar] [CrossRef]
- Zheng, H.Y.; Liu, Z.; Chen, B.; Xu, H. Quantitative Ulva prolifera Bloom Monitoring Based on Multi-Source Satellite Ocean Color Remote Sensing Data. Appl. Ecol. Environ. Res. 2020, 18, 4897–4913. [Google Scholar] [CrossRef]
- Li, Z.; Wang, Z.; Li, Y.; Zhang, Y.; Zheng, J.; Gao, S. Evaluation of Global High-Resolution Reanalysis Products Based on the Chinese Global Oceanography Forecasting System. Atmos. Oceanic Sci. Lett. 2021, 14, 100032. [Google Scholar] [CrossRef]
- Wang, H.; Wan, L.; Qin, Y.; Wang, Y.; Yang, X.; Liu, Y.; Xing, J.; Chen, L.; Wang, Z.; Zhang, T.; et al. Development and Application of the Chinese Global Operational Oceanography Forecasting System. Adv. Earth Sci. 2016, 31, 1090–1104. (In Chinese) [Google Scholar] [CrossRef]
- Ji, X.; Kwon, K.M.; Choi, B.-J.; Liu, G.; Park, K.-S.; Wang, H.; Byun, D.-S.; Li, Y.; Ji, Q.; Zhu, X. Assimilating OSTIA SST into Regional Modeling Systems for the Yellow Sea Using Ensemble Methods. Acta Oceanolog. Sin. 2017, 36, 37–51. [Google Scholar] [CrossRef]
- Liu, D.; Keesing, J.K.; He, P.; Wang, Z.; Shi, Y.; Wang, Y. The World’s Largest Macroalgal Bloom in the Yellow Sea, China: Formation and Implications. Estuar. Coast. Shelf Sci. 2013, 129, 2–10. [Google Scholar] [CrossRef]
- Son, Y.B.; Choi, B.-J.; Kim, Y.H.; Park, Y.-G. Tracing Floating Green Algae Blooms in the Yellow Sea and the East China Sea Using GOCI Satellite Data and Lagrangian Transport Simulations. Remote Sens. Environ. 2015, 156, 21–33. [Google Scholar] [CrossRef]
- Bao, M.; Guan, W.; Yang, Y.; Cao, Z.; Chen, Q. Drifting Trajectories of Green Algae in the Western Yellow Sea during the Spring and Summer of 2012. Estuar. Coast. Shelf Sci. 2015, 163, 9–16. [Google Scholar] [CrossRef]
- Keesing, J.K.; Liu, D.; Fearns, P.; Garcia, R. Inter- and Intra-Annual Patterns of Ulva prolifera Green Tides in the Yellow Sea during 2007–2009, Their Origin and Relationship to the Expansion of Coastal Seaweed Aquaculture in China. Mar. Pollut. Bull. 2011, 62, 1169–1182. [Google Scholar] [CrossRef]
- Zhan, Y.; Qiu, Z.; Wang, Y.; Su, Y.; Li, Y.; Cui, Y.; Qu, S.; Wang, P.; Rong, X. Long-Term Spatiotemporal Characteristics of Ulva prolifera Green Tide and Effects of Environmental Drivers on Its Monitoring by Satellites: A Case Study in the Yellow Sea, China, from 2008 to 2023. J. Mar. Sci. Eng. 2024, 12, 630. [Google Scholar] [CrossRef]
- Huang, J.; Wu, L.; Gao, S.; Li, J. Analysis on the Interannual Distribution Variation of Green Tide in Yellow Sea. Acta Laser Biol. Sin. 2014, 23, 572–578. (In Chinese) [Google Scholar]
- Wang, Z.; Fu, M.; Xiao, J.; Zhang, X.; Song, W. Progress on the Study of the Yellow Sea Green Tides Caused by Ulva prolifera. Acta Oceanologica Sinica 2018, 40, 1–13. (In Chinese) [Google Scholar] [CrossRef]
- Wagner, T.J.W.; Eisenman, I.; Ceroli, A.M.; Constantinou, N.C. How winds and ocean currents influence the drift of floating objects. J. Phys. Oceanogr. 2022, 52, 907–916. [Google Scholar] [CrossRef]
- Rial-Osorio, M.; Pérez-Muñuzuri, V.; Cloux, S. Tracking marine debris in Northwest Spain: Assessing wind influence with a Lagrangian transport model. Ocean Sci. 2026, 22, 777–790. [Google Scholar] [CrossRef]
- Zhang, C.; Lv, X.; Qiao, F. The modelling of Ulva prolifera transport in the yellow sea and its application. Oceanol. Limnol. Sin. 2018, 49, 1019–1028. [Google Scholar] [CrossRef]









| Parameter | Description | Unit | Value | Reference |
|---|---|---|---|---|
| R1 | Empirical coefficient of ocean current | - | 1.0 | [9] |
| R2 | Wind drag coefficient | - | 1% | [9] |
| β | Wind drag deflection Angle | deg | 20 | [9] |
| Gmax | Maximum daily growth rate | - | 45% | [23] |
| Dmax | Maximum daily mortality rate | - | 10% | [9] |
| T1 | The low end of the optimum temperature range for growth | °C | 15 | [9] |
| T2 | The high end of the optimum temperature range for growth | °C | 20 | [9] |
| T3 | The critical temperature for maximum mortality | °C | 25 | [23] |
| θ1 | The coefficient of growth rate affected by temperature | - | 1.10 | [23] |
| θ2 | The coefficient of growth rate affected by temperature | - | 1.80 | [23] |
| θ3 | The coefficient of mortality rate affected by temperature | - | 1.10 | [23] |
| Iopt | Optimum light intensity for growth | μmol E m−2 s−1 | 400 | [9] |
| Case Name | N (≥0 g) | In (≥0 g) | P (≥0 g) | N (≥200 g) | In (≥200 g) | P (≥200 g) | N (≥500 g) | In (≥500 g) | P (≥500 g) |
|---|---|---|---|---|---|---|---|---|---|
| 2014 | 8908 | 3398 | 38.15% | 2678 | 1099 | 41.04% | 581 | 485 | 83.48% |
| 2015 | 9928 | 7086 | 71.37% | 3461 | 1909 | 55.16% | 1586 | 1004 | 63.30% |
| 2021 | 11,855 | 5975 | 50.40% | 4320 | 2042 | 47.27% | 1735 | 693 | 39.94% |
| Average | 30,691 | 16,459 | 53.31% | 10,459 | 5050 | 47.82% | 3902 | 2182 | 62.24% |
| Wind Drag Coefficient | 1% | 1.5% | 2% | 2.5% | 3% |
|---|---|---|---|---|---|
| 16 June | 61.75% | 60.83% | 66.63% | 75.50% | 74.12% |
| 20 June | 48.04% | 48.05% | 57.15% | 65.36% | 65.22% |
| 23 June | 41.92% | 42.32% | 51.66% | 58.60% | 56.95% |
| 1 July | 35.42% | 39.08% | 48.84% | 52.98% | 50.84% |
| Average | 46.78% | 47.58% | 56.07% | 63.11% | 61.78% |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Yang, J.; He, E.; Ji, X.; Guo, Q.; Gao, S.; Jiang, Y. Remote Sensing Data-Based Modelling for Analyzing Green Tide Proliferation Drivers in the Yellow Sea. Remote Sens. 2026, 18, 1014. https://doi.org/10.3390/rs18071014
Yang J, He E, Ji X, Guo Q, Gao S, Jiang Y. Remote Sensing Data-Based Modelling for Analyzing Green Tide Proliferation Drivers in the Yellow Sea. Remote Sensing. 2026; 18(7):1014. https://doi.org/10.3390/rs18071014
Chicago/Turabian StyleYang, Jing, Enye He, Xuanliang Ji, Qianqiu Guo, Shan Gao, and Yuxuan Jiang. 2026. "Remote Sensing Data-Based Modelling for Analyzing Green Tide Proliferation Drivers in the Yellow Sea" Remote Sensing 18, no. 7: 1014. https://doi.org/10.3390/rs18071014
APA StyleYang, J., He, E., Ji, X., Guo, Q., Gao, S., & Jiang, Y. (2026). Remote Sensing Data-Based Modelling for Analyzing Green Tide Proliferation Drivers in the Yellow Sea. Remote Sensing, 18(7), 1014. https://doi.org/10.3390/rs18071014





