Satellite-Based Ocean Color and Thermal Signatures Defining Habitat Hotspots and the Movement Pattern for Commercial Skipjack Tuna in Indonesia Fisheries Management Area 713, Western Tropical Pacific
Abstract
1. Introduction
2. Materials and Methods
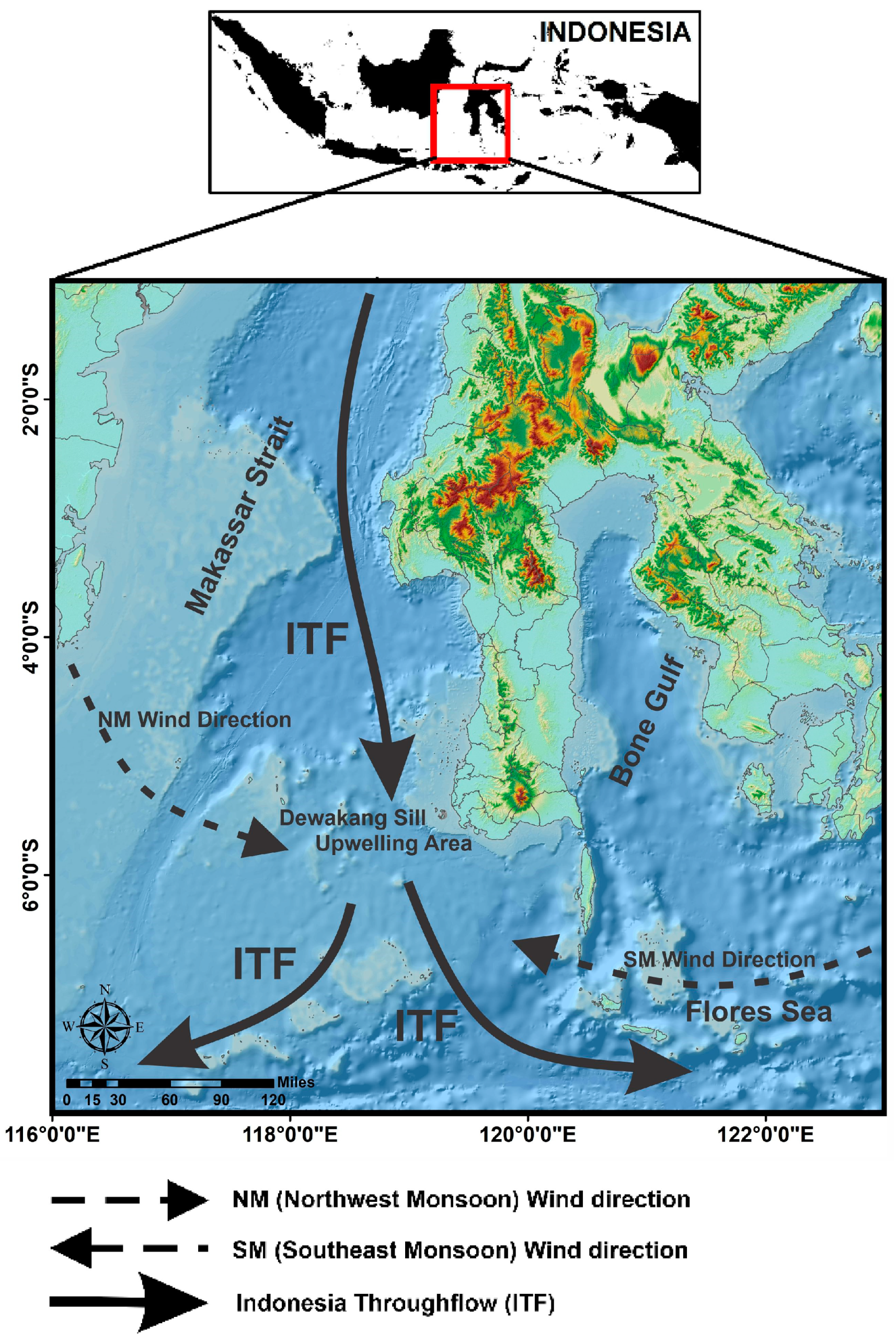
2.1. Study Area
2.2. Skipjack Fishery Data
2.3. Remotely Sensed Oceanographic Data
2.4. Model Construction for Predicting Potential Skipjack Habitat
2.5. Movement Pattern of Skipjack Tuna Fishing Ground
2.6. Mapping and Validating Potential Skipjack Tuna Habitat
3. Results
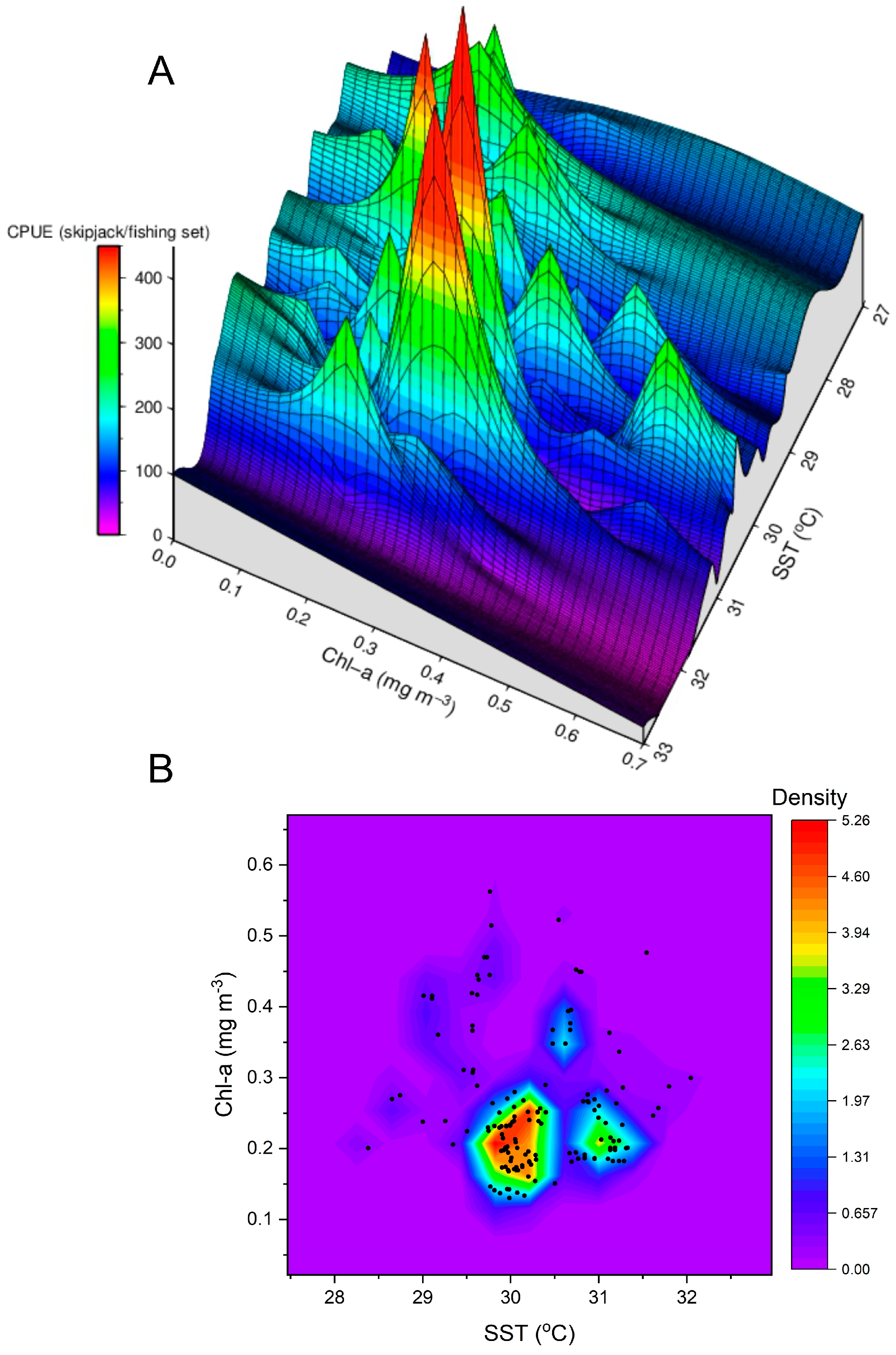
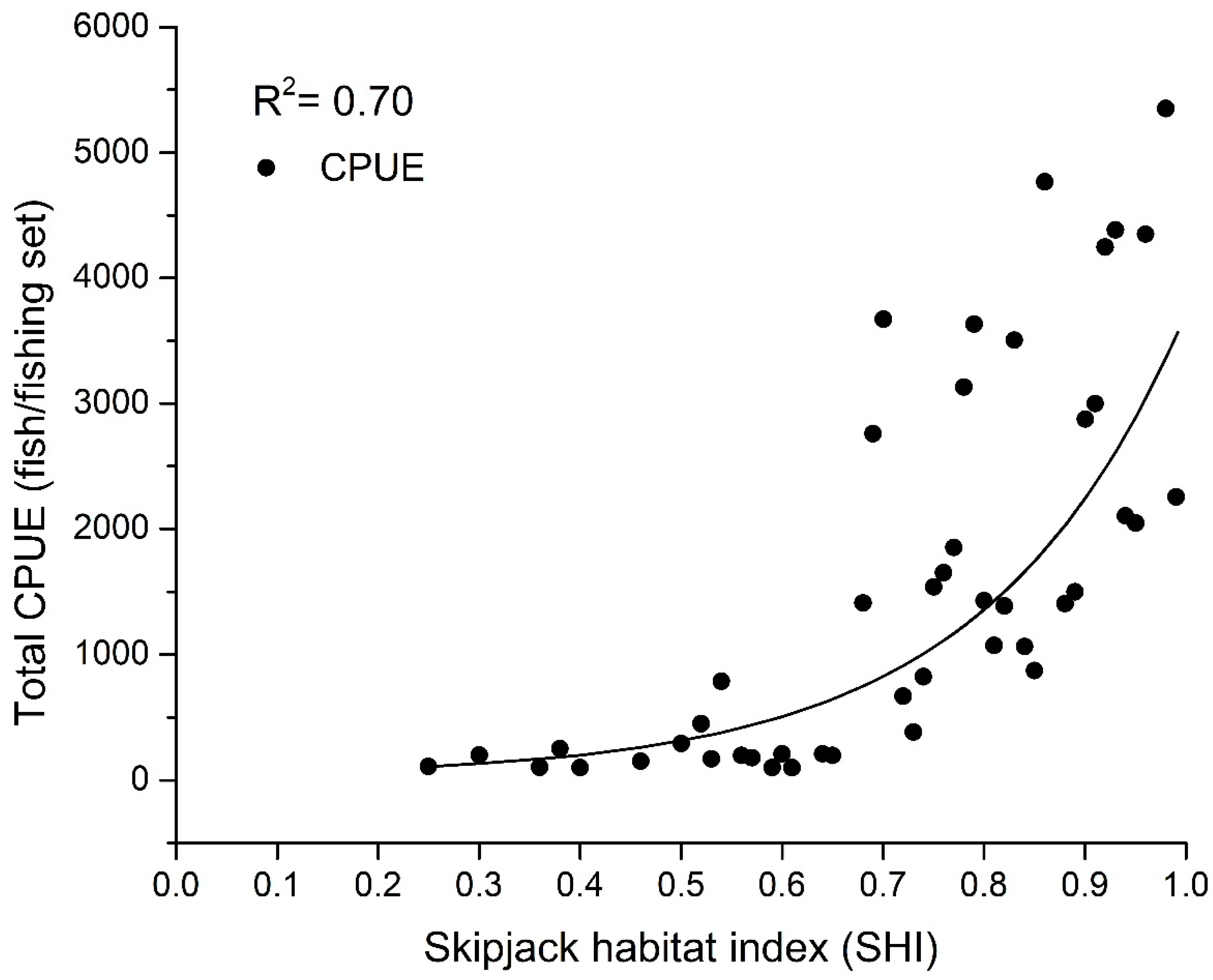
3.1. Skipjack Tuna Fishing Performance versus Oceanographic Factors
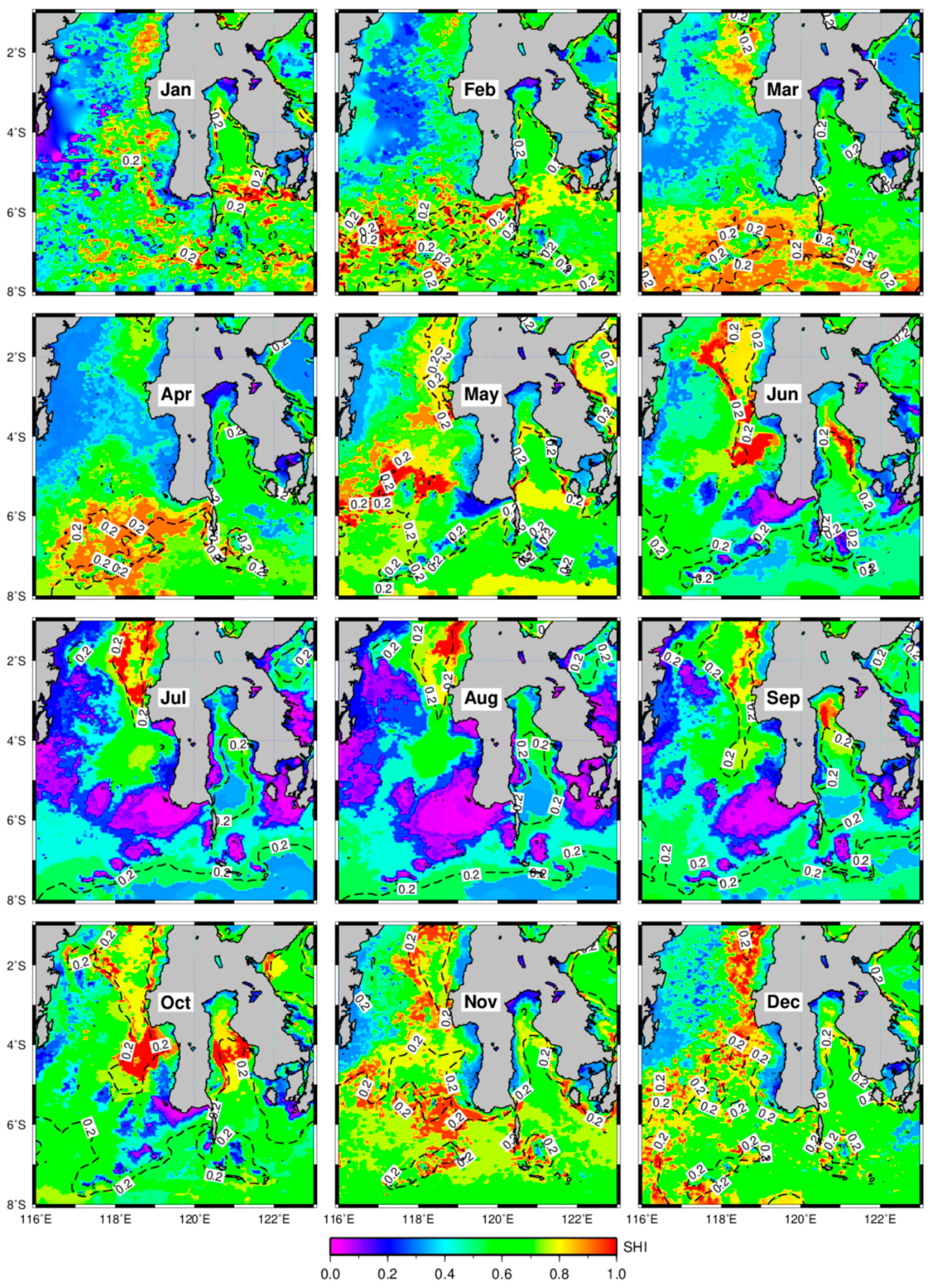
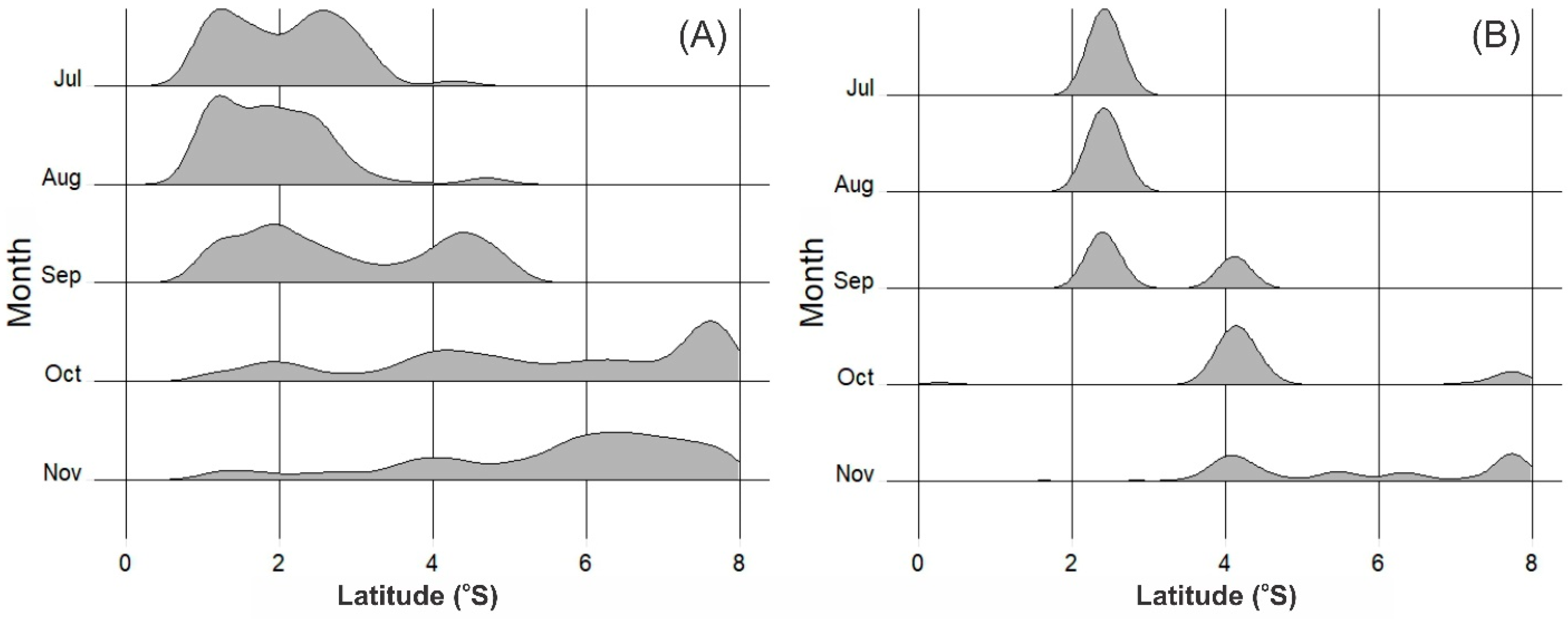
3.2. Distribution of Potential Skipjack Tuna Habitat in Space and Time
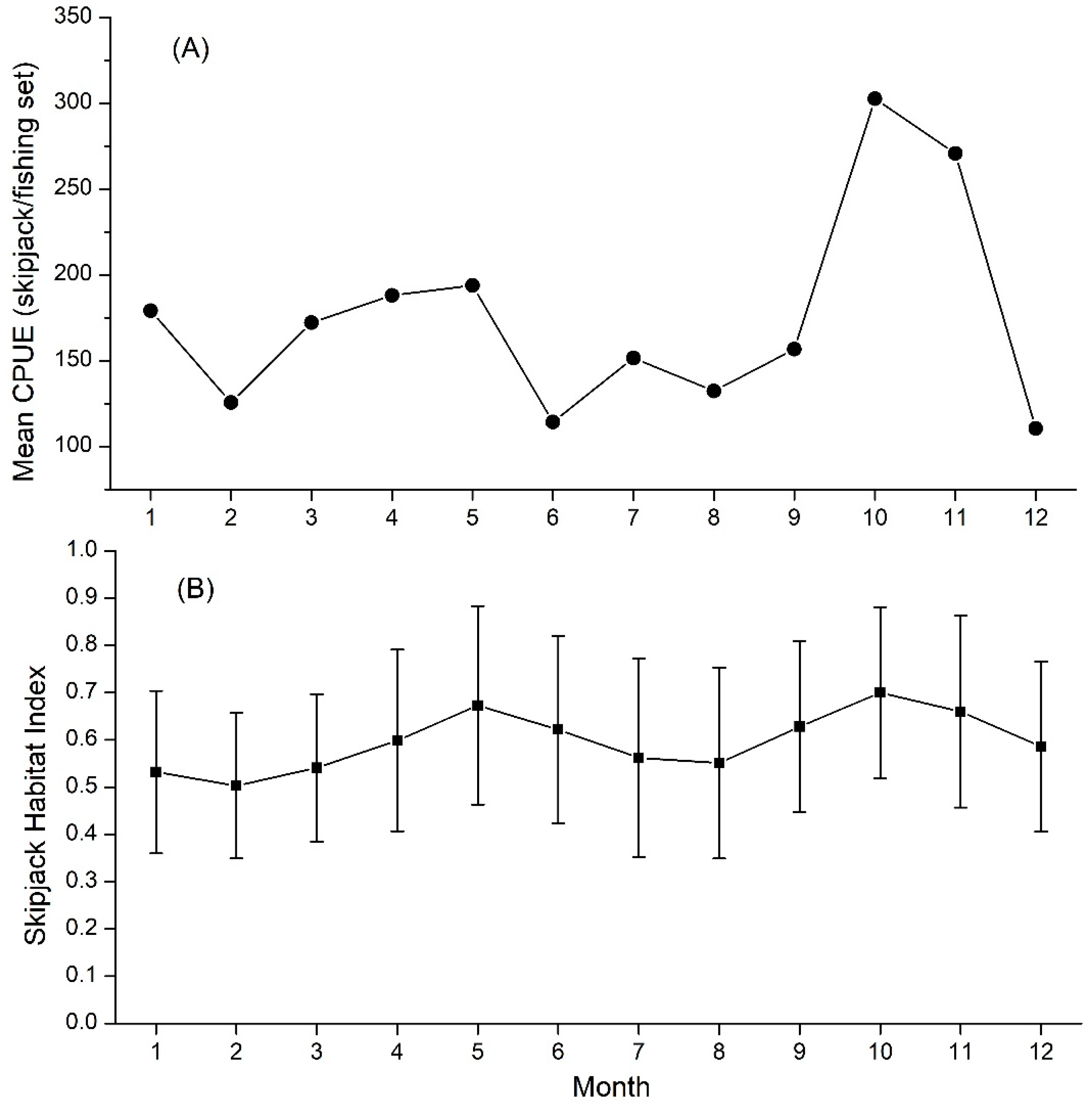
3.3. Validation of Skipjack Habitat Hotspot Prediction
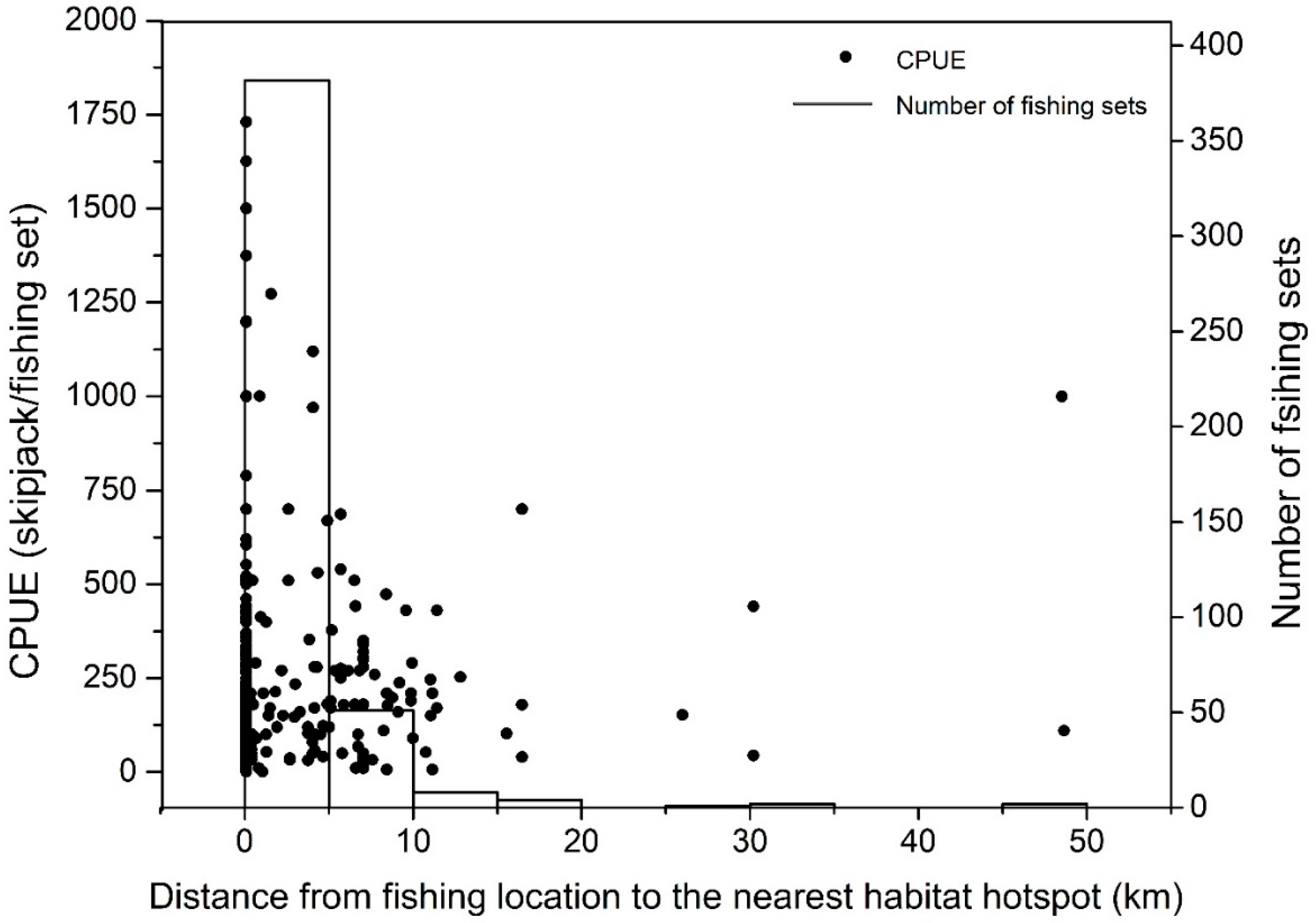
3.4. Movement Pattern of Skipjack Fishing Ground Relative to Habitat Hotspots
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Arai, T.; Kotake, A.; Kayama, S.; Ogura, M.; Watanabe, Y. Movements and Life History Patterns of the Skipjack Tuna Katsuwonus pelamis in the Western Pacific, as Revealed by Otolith Sr: Ca Ratios. J. Mar. Biol. Assoc. UK 2005, 85, 1211–1216. [Google Scholar] [CrossRef]
- ISSF. Status of the World Fisheries for Tuna: Nov. 2022. ISSF Technical Report 2022-15; ISSF: Pittsburgh, PA, USA, 2022. [Google Scholar]
- FAO. The State of World Fisheries and Aquaculture 2020. Sustainability in Action; FAO: Roma, Italy, 2020. [Google Scholar]
- Hare, S.R.; Williams, P.G.; Jordan, C.C.; Hamer, P.A.; Hampton, W.J.; Scott, R.D. The Western and Central Pacific Tuna Fishery: 2020 Overview and Status of Stocks; Pacific Community: Noumea, New Caledonia, 2021; ISBN 9789820009509. [Google Scholar]
- Langley, A.; Hampton, J.; Ogura, M. Stock Assessment of Skipjack Tuna in the Western and Central Pacific Ocean. St Meeting of the Scientific Committee of the Western and Central Pacific Fisheries Commission WCPFC–SC1. 2005, pp. 1–69. Available online: https://www.semanticscholar.org/paper/Stock-assessment-of-skipjack-tuna-in-the-Western-Hoyle-Kleiber/0cc5043b07aa915f9a2ad87996083c7b75854d2a?p2df (accessed on 20 February 2023).
- Krogh-Poulsen, B.; McDonald, S.L.; Woodcock, T. Seafood Social Risk Tool: Identifying Risk of Forced Labor, Human Trafficking, and Hazardous Child Labor in the Seafood Industry; Monterey Bay Aquarium Foundation: Monterey, CA USA, 2022. [Google Scholar]
- Dignan, S.; Gascoigne, J.; Abdul, H. Indonesia Pole-and-Line and Handline, Skipjack and Yellowfin Tuna of Western and Central Pacific Archipelagic Waters. Public Certification Report. 2021. Available online: https://cert.msc.org/FileLoader/FileLinkDownload.asmx/GetFile?encryptedKey=u/HnmumOkSyuGS/2y9U8IyQB9M+H5lrnbvwCrDXpqrnbHwJpI5DlsM2xL9GlVkec (accessed on 20 February 2023).
- Koeshendrajana, S.; Rusastra, W. Potensi Sumber Daya Kelautan Dan Perikanan WPP-NRI 713; AMAFRAD Press: Jakarta, Indonesia, 2019; ISBN 9786025791710. [Google Scholar]
- Loneragan, N.; Wiryawan, B.; Hordyk, A.; Halim, A. Proceedings From Workshops on Management Strategy Evaluation of Data-Limited Fisheries; MurdochUniversity: Murdoch, Australia; IPB University: Bogor, Indonesia, 2021; ISBN 9780646829517. [Google Scholar]
- Lehodey, P.; Bertignac, M.; Hampton, J.; Lewis, A.; Picaut, J. El Niῆo Southern Oscillation and Tuna in the Western Pacific. Nature 1997, 389, 715–718. [Google Scholar] [CrossRef]
- Bertignac, P.; Lehodey, P.; Hampton, J. A Spatial Population Dynamics Simulation Model of Tropical Tunas Using a Habitat Index Based on Environmental Parameters. Fish. Oceanogr. 1998, 7, 326–334. [Google Scholar] [CrossRef]
- Mugo, R.; Saitoh, S.; Nihira, A.; Kuroyama, T. Habitat Characteristics of Skipjack Tuna ( Katsuwonus pelamis) in the Western North Pacific: A Remote Sensing Perspective. Fish. Oceanogr. 2010, 19, 382–396. [Google Scholar] [CrossRef]
- Zainuddin, M.; Farhum, A.; Safruddin, S.; Selamat, M.B.; Sudirman, S.; Nurdin, N.; Syamsuddin, M.; Ridwan, M.; Saitoh, S.-I. Detection of Pelagic Habitat Hotspots for Skipjack Tuna in the Gulf of Bone-Flores Sea, Southwestern Coral Triangle Tuna, Indonesia. PLoS ONE 2017, 12, e0185601. [Google Scholar] [CrossRef]
- Lehodey, P. Impacts of the El Niño Southern Oscillation on Tuna Populations and Fisheries in the Tropical Pacific Ocean. Prog. Oceanogr. 2001, 49, 439–468. [Google Scholar] [CrossRef]
- Brill, R.W. A Review of Temperature and Oxygen Tolerance Studies of Tunas Pertinent to Fisheries Oceanography, Movement Models and Stock Assessments. Fish. Oceanogr. 1994, 3, 204–216. [Google Scholar] [CrossRef]
- Barkley, A.; Neill, W.H.; Gooding, R.M. Skipjack Tuna, Katsuwonus pelamis, Habitat Based on Temperature and Oxygen Requirements. Fish. Bulletin. 1978, 76, 653–662. [Google Scholar]
- Chen, G.; Yang, J.; Han, G. Remote Sensing of Environment Eddy Morphology: Egg-like Shape, Overall Spinning, and Oceanographic Implications. Remote Sens. Environ. 2021, 257, 112348. [Google Scholar] [CrossRef]
- Kiyofuji, H.; Aoki, Y.; Kinoshita, J.; Okamoto, S.; Masujima, M.; Matsumoto, T.; Fujioka, K. Progress in Oceanography Northward Migration Dynamics of Skipjack Tuna (Katsuwonus pelamis) Associated with the Lower Thermal Limit in the Western Pacific Ocean. Prog. Oceanogr. 2019, 175, 55–67. [Google Scholar] [CrossRef]
- Alatorre-Ramirez, V.G.; Galván-Magaña, F.; Torres-Rojas, Y.E.; Olson, R.J. Trophic Segregation of Mixed Schools of Yellowfin Tuna (Thunnus albacares) and Skipjack Tuna (Katsuwonus pelamis) Caught in the Eastern Tropical Pacific Ocean. Fish. Bulletin. 2017, 115, 252–268. [Google Scholar] [CrossRef]
- Venegas, R.; Oliver, T.; Brainard, R.E.; Santos, M.; Geronimo, R.; Widlansky, M. Climate-Induced Vulnerability of Fisheries in the Coral Triangle: Skipjack Tuna Thermal Spawning Habitats. Fish. Oceanogr. 2019, 28, 117–130. [Google Scholar] [CrossRef]
- Sund, P.N.; Balckburn, M.; Williams, F. Tunas and Their Environment in the Pacific Ocean: A Review. Oceanogr. Mar. Biol. Ann. Rev. 1981, 19, 443–512. [Google Scholar]
- Lehodey, P.; Bertignac, M.; Stoens, A.; Memery, L.; Grima, N. Predicting Skipjack Tuna Forage Distributions in the Equatorial Pacific Using a Coupled Dynamical Bio-Geochemical Model. Fish. Oceanogr. 1998, 7, 317–325. [Google Scholar] [CrossRef]
- Evans, R.H.; McLain, D.R.; Bauer, R.A. Atlantic Skipjack Tuna: Their Vulnerability to Surface Fishing Gear. Mar. Fish. Rev. 1981, 43, 1–11. [Google Scholar]
- Ashida, H.; Gosho, T.; Watanabe, K.; Okazaki, M.; Tanabe, T. Reproductive Traits and Seasonal Variations in the Spawning Activity of Female Albacore, Thunnus alalunga, in the Subtropical Western North Pacific Ocean. J. Sea Res. 2020, 160–161, 101902. [Google Scholar] [CrossRef]
- Iizuka, K.; Asano, M.; Naganuma, A. Feeding Habits of Skipjack Tuna Katsuwonus-pelamis Linnaeus Caught by Pole and Line and the State of Young Skipjack Tuna Distribution in the Tropical Seas of the Western Pacific Ocean. Bull. Tohoku Natl. Fish. Res. Inst. 1989, 51, 107–116. [Google Scholar]
- Boehlert, G.W.; Mundy, B.C. Vertical and Onshore-Offshore Distributional Patterns of Tuna Larvae in Relation to Physical Habitat Features. Mar. Ecol. Prog. Ser. 1994, 107, 1–14. [Google Scholar] [CrossRef]
- Azwir, A.; Muchlisin, Z.A.; Ramadhani, I. The Study on the Fish Food Habits of Skipjack (Katsuwonus pelamis) and Frigate Tuna (Auxis thazard). J. Nat. 2004, 4, 20–23. (In Indonesian) [Google Scholar]
- Hany Restiangsih, Y.; Amri, K. Biological Aspects and Food Habits of Skipjack Tuna (Katsuwonus pelamis) in Flores Sea and Adjacent Waters. BAWAL 2018, 10, 187–196. [Google Scholar]
- Hsu, T.Y.; Chang, Y.; Lee, M.A.; Wu, R.F.; Hsiao, S.C. Predicting Skipjack Tuna Fishing Grounds in the Western and Central Pacific Ocean Based on High-spatial-Temporal-resolution Satellite Data. Remote Sens. 2021, 13, 861. [Google Scholar] [CrossRef]
- Arrizabalaga, H.; Dufour, F.; Kell, L.; Merino, G.; Ibaibarriaga, L.; Chust, G.; Irigoien, X.; Santiago, J.; Murua, H.; Fraile, I.; et al. Global Habitat Preferences of Commercially Valuable Tuna. Deep-Sea Res. II 2015, 113, 102–112. [Google Scholar] [CrossRef]
- Graham, J.B.; Dickson, K.A. Tuna Comparative Physiology. J. Exp. Biol. 2004, 207, 4015–4024. [Google Scholar] [CrossRef]
- Madureira, L.S.P.; Coletto, J.L.; Pinho, M.P.; Weigert, S.C.; Varela, C.M.; Campello, M.E.S.; Llopart, A. Skipjack (Katsuwonus pelamis) Fishery Improvement Project: From Satellite and 3D Oceanographic Models to Acoustics, towards Predator-Prey Landscapes. In Proceedings of the 2017 IEEE/OES Acoustics in Underwater Geosciences Symposium, RIO Acoustics 2017, Rio de Janeiro, Brazil, 25–27 January 2018; pp. 1–7. [Google Scholar] [CrossRef]
- Collette, B.B.; Nauen, C.E. FAO Species Catalogue Vol. 2. Scombrids of the World : An Annotated and Illustrated Catalogue of Tunas, Mackerels, Bonitos, and Related Species Known to Date. 1983, Volume 2. Available online: https://www.cabdirect.org/cabdirect/abstract/19841458934 (accessed on 20 February 2023).
- Stéquert, B.; Ramcharrun, B. La Reproduction Du Listao (Katsuwonus pelamis) Dans Le Bassin Ouest de l’ocean Indien. Aquat. Living Resour. 1996, 9, 235–247. [Google Scholar] [CrossRef]
- Andrade, H.A.; Garcia, C.A.E. Skipjack Tuna Fishery in Relation to Sea Surface Temperature off the Southern Brazilian Coast. Fish. Oceanogr. 1999, 8, 245–254. [Google Scholar] [CrossRef]
- Andrade, H.A. The Relationship between the Skipjack Tuna (Katsuwonus pelamis) Fishery and Seasonal Temperature Variability in the South-Western Atlantic. Fish. Oceanogr. 2003, 12, 10–18. [Google Scholar] [CrossRef]
- Ashida, H. Spatial and Temporal Differences in the Reproductive Traits of Skipjack Tuna Katsuwonus pelamis between the Subtropical and Temperate Western Pacific Ocean. Fish. Res. 2020, 221, 105352. [Google Scholar] [CrossRef]
- Druon, J.N.; Chassot, E.; Murua, H.; Soto, M. Preferred Feeding Habitat of Skipjack Tuna in the Eastern Central Atlantic and Western Indian Oceans: Relations with Carrying Capacity and Vulnerability to Purse Seine Fishing. In Proceedings of the IOTC Proceedings, Colombo, Sri Lanka, 6–10 September 2016. [Google Scholar]
- Druon, J.N.; Chassot, E.; Murua, H.; Lopez, J. Skipjack Tuna Availability for Purse Seine Fisheries Is Driven by Suitable Feeding Habitat Dynamics in the Atlantic and Indian Oceans. Front. Mar. Sci. 2017, 4. [Google Scholar] [CrossRef]
- Zainuddin, M.; Saitoh, K.; Saitoh, S.-I. Albacore (Thunnus alalunga) Fishing Ground in Relation to Oceanographic Conditions in the Western North Pacific Ocean Using Remotely Sensed Satellite Data. Fish. Oceanogr. 2008, 17, 61–73. [Google Scholar] [CrossRef]
- Laurs, R.M.; Fiedler, P.C.; Montgomery, D.R. Albacore Tuna Catch Distributions Relative to Environmental Features Observed from Satellites. Deep-Sea Res. 1984, 31, 1085–1099. [Google Scholar] [CrossRef]
- Zainuddin, M.; Kiyofuji, H.; Saitoh, K.; Saitoh, S. Using Multi-Sensor Satellite Remote Sensing and Catch Data to Detect Ocean Hot Spots for Albacore (Thunnus alalunga) in the Northwestern North Pacific. Deep-Sea Res. II 2006, 53, 419–431. [Google Scholar] [CrossRef]
- Polovina, J.J.; Howell, E.; Kobayashi, D.R.; Seki, M.P. The Transition Zone Chlorophyll Front, a Dynamic Global Feature Defining Migration and Forage Habitat for Marine Resources. Prog. Oceanogr. 2001, 49, 469–483. [Google Scholar] [CrossRef]
- Domokos, R.; Seki, M.P.; Polovina, J.J.; Hawn, D.R. Oceanographic Investigation of the American Samoa Albacore (Thunnus alalunga) Habitat and Longline Fishing Grounds. Fish. Oceanogr. 2007, 16, 555–572. [Google Scholar] [CrossRef]
- Harsono, G.; Manurung, D.; Atmadipoera, A.S.; Baskoro, M.S.; Syamsudin, F. Halmahera Eddy Displacement in Relation to Skipjack Productivity in Surrounding Waters. J. Teknol. Perikan. Dan Kelaut. 2014, 5, 147–154. [Google Scholar]
- Istnaeni, Z.D.; Zainuddin, M. The Impact of Oceanographic Parameters Changes on The Distribution and Abundance of Skipjack Tuna Katsuwonus pelamis in Makassar Strait. J. Ilmu Dan Teknol. Kelaut. Trop. 2019, 11, 171–180. [Google Scholar] [CrossRef]
- Hastie, T.; Tibshirani, R. Generalized Additive Models; Chapman and Hall: London, UK, 1990; p. 356. [Google Scholar]
- Drexler, M.; Ainsworth, C.H. Generalized Additive Models Used to Predict Species Abundance in the Gulf of Mexico: An Ecosystem Modeling Tool. PLoS ONE 2013, 8, e0064458. [Google Scholar] [CrossRef] [PubMed]
- Bigelow, K.A.; Boggs, C.H.; He, X. Environmental Effects on Swordfish and Blue Shark Catch Rates in the US North Pacific Longline Fishery. Fish. Oceanogr. 1999, 8, 178–198. [Google Scholar] [CrossRef]
- Moisen, G.G.; Frescino, T.S. Comparing Five Modelling Techniques for Predicting Forest Characteristics. Ecol. Model. 2002, 157, 209–225. [Google Scholar] [CrossRef]
- Mugo, R.; Saitoh, S.-I. Ensemble Modelling of Skipjack Tuna (Katsuwonus pelamis) Habitats in the Western North Pacific Using Satellite Remotely Sensed Data; a Comparative Analysis Using Machine-Learning Models. Remote Sens. 2020, 12, 2591. [Google Scholar] [CrossRef]
- Katsanevakis, S.; Maravelias, C.D. Bathymetric Distribution of Demersal Fish in the Aegean and Ionian Seas Based on Generalized Additive Modeling. Fish. Sci. 2009, 75, 13–23. [Google Scholar] [CrossRef]
- Chen, Q.; Wynne, R.J.; Goulding, P.; Sandoz, D. The Application of Principal Component Analysis and Kernel Density Estimation to Enhance Process Monitoring. Control. Eng. Pr. 2000, 8, 531–543. [Google Scholar] [CrossRef]
- Węglarczyk, S. Kernel Density Estimation and Its Application. ITM Web Conf. 2018, 23, 00037. [Google Scholar] [CrossRef]
- Ramos, A.G.; Santiago, J.; Sangra, P.; Canton, M. An Application of Satellite-Derived Sea Surface Temperature Data to the Skipjack (Katsuwonus pelamis Linnaeus, 1758) and Albacore Tuna (Thunnus alalunga Bonaterre, 1788) Fisheries in the North-East Atlantic. Int. J. Remote Sens. 1996, 17, 749–759. [Google Scholar] [CrossRef]
- Mallawa, A.; Amir, F.; Zainuddin, M. Biological Performance Aspect of Skipjack Tuna (Katsuwonus pelamis) Population Captured by Purse Seine in East Season at Flores Sea. J. IPTEKS PSP 2014, 1, 129–145. [Google Scholar]
- Mallawa, A.; Amir, F.; Mallawa, E. Sustainability of Fishing Technologies of Skipjack Tuna (Katsuwonus pelamis) in Bone Bay Waters, South Sulawesi Oleh. Mar. Fish. 2018, 9, 93–106. [Google Scholar]
- Gordon, A.L. Oceanography of the Indonesian Seas and their Throughflow. Oceanography 2005, 18, 14–27. [Google Scholar] [CrossRef]
- Qu, T.; Du, Y.; Strachan, J.; Meyers, G.; Slingo, J. Sea Surface Temperature and Its Variability in the Indonesian Region. Oceanography 2005, 18, 50–61. [Google Scholar] [CrossRef]
- Sprintall, J.; Liu, T. Ekman Mass and Heat Transport in the Indonesia Seas. Oceanography 2005, 18, 88–97. [Google Scholar] [CrossRef]
- Agus, S. Atmadipoera and Priska Widyastuti A Numerical Modeling Study on Upwelling Mechanism in Southern Makassar Strait. Ilmu Dan Teknol. Kelaut. Trop. 2014, 6, 355–372. [Google Scholar]
- Nuzula, F.; Syamsudin, M.L.; Yuliadi, L.P.S.; Purba, N.P. Martono Eddies Spatial Variability at Makassar Strait—Flores Sea. IOP Conf. Ser. Earth Environ. Sci. PAPER 2017, 54, 1–10. [Google Scholar] [CrossRef]
- Hendiarti, N.; Suwarso; Aldrian, E.; Amri, K.; Andiastuti, R.; Sachoemar, S.; Wahyono, I.B. Seasonal Variation of Pelagic Fish Catch Around Java. Oceanography 2005, 18, 112–123. [Google Scholar] [CrossRef]
- Ashida, H.; Tanabe, T.; Suzuki, N. Recent Progress on Reproductive Biology of Skipjack Tuna in the Tropical Region of the Western and Central Pacific Ocean. In Proceedings of the 5th Regular Session of the Scientific Committee, Port Vila, Vanuatu, 10–21 August 2009. [Google Scholar]
- Jamal, M.; Sondita, F.A.; Wiryawan, B.; Haluan, J.; Pemanfaatan, J.; Perikanan, S.; Perikanan, F.; Kelautan, I.; Pemanfaatan, D. Management Concept of Skipjack Tuna (Katsuwonus pelamis) Fisheries Within Bone Bay Zone in The Perspective of Sustainability. J. IPTEKS PSP 2014, 1, 196–207. [Google Scholar]
- Zainuddin, M.; Saitoh, S.-I.; Saitoh, K. Detection of Potential Fishing Ground for Albacore Tuna Using Synoptic Measurements of Ocean Color and Thermal Remote Sensing in the Northwestern North Pacific. Geophys. Res. Lett. 2004, 31. [Google Scholar] [CrossRef]
- Bertrand, A.; Josse, E.; Bach, P.; Gros, P.; Dagorn, L. Hydrological and Trophic Characteristics of Tuna Habitat: Consequences on Tuna Distribution and Longline Catchability Résumé. Can. J. Fish. Aquat. Sci. 2002, 59, 1002–1013. [Google Scholar] [CrossRef]
- Richards, L.J.; Schnute, J.T. An Experimental and Statistical Approach to the Question: Is CPUE an Index of Abundance? Can. J. Fish. Aquat. Sci. 1986, 43, 1214–1227. [Google Scholar] [CrossRef]
- Wood, S. Generalized Additive Models, An Introduction with R; Chapman and Hall: London, UK, 2006. [Google Scholar]
- Johnson, J.B.; Omland, K.S. Model Selection in Ecology and Evolution. Trends Ecol. Evol. 2004, 19, 101–108. [Google Scholar] [CrossRef]
- Nurdin, S.; Mustapha, M.A.; Lihan, T.; Zainuddin, M. Applicability of Remote Sensing Oceanographic Data in the Detection of Potential Fishing Grounds of Rastrelliger kanagurta in the Archipelagic Waters of Spermonde, Indonesia. Fish. Res. 2017, 196, 1–12. [Google Scholar] [CrossRef]
- Wessel, P.; Smith, W.H.F.; Scharroo, R.; Luis, J.; Wobbe, F. Generic Mapping Tools: Improved Version Released. EOS 2013, 94, 409–410. [Google Scholar] [CrossRef]
- Lan, K.; Shimada, T.; Lee, M.; Su, N.; Chang, Y. Using Remote-Sensing Environmental and Fishery Data to Map Potential Yellowfin Tuna Habitats in the Tropical Pacific Ocean. Remote Sens. 2017, 9, 444. [Google Scholar] [CrossRef]
- Owen, R.W. Fronts and Eddies in the Sea: Mechanisms, Interaction and Biological Effects; Academic Press: New York, NY, USA, 1981; pp. 197–234. [Google Scholar]
- Bakun, A. Fronts and Eddies as Key Structures in the Habitat of Marine Fish Larvae: Opportunity. Adapt. Response 2006, 70, 105–122. [Google Scholar]
- Uda, M. Pulsative Fluctuation of Oceanic Fronts in Association with Tuna Fishing Ground and Fisheries. J. Fac. Mar. Sci. Technol. Tokai Univ. 1973, 7, 245–266. [Google Scholar]
- Wilson, C.; Morales, J.; Nayak, S.; Asanuma, I.; Feldman, G. Ocean-color radiometry and fisheries. In Why OceanColour? The Societal Benefits of Ocean-Color Technology; Platt, T., Hoepffner, N., Stuart, V., Eds.; Reports ofthe International Ocean-Color Coordinating Group, No. 7; IOCCG: Dartmouth, NS, Canada, 2008; pp. 47–58. [Google Scholar]
- Seki, M.P.; Polovina, J.J.; Brainard, R.E.; Bidigare, R.R.; Leonard, C.L.; Foley, D.G. Biological Enhancement at Cyclonic Eddies Tracked with GOES Thermal Imagery in Hawaiian Waters. Geophys. Res. Lett. 2001, 28, 1583–1586. [Google Scholar] [CrossRef]
- Gower, J.F.R. A Survey of the Uses of Remote Sensing from Aircraft and Satellites in Oceanography and Hydrography. Pac. Mar. Sci. Rep. Inst. Ocean. Sci. 1972, 72, 39. [Google Scholar]
- Olson, B.D.B.; Hitchcock, G.L.; Mariano, A.J.; Ashjian, C.J.; Peng, G.; Nero, R.W.; Podest, G.P. Life on the Edge: Marine Life and Fronts. Oceanography 1994, 7, 52–60. [Google Scholar] [CrossRef]
- Kirby, D.S.; Fiksen, Y.; Hart, P.J.B. A Dynamic Optimisation Model for the Behaviour of Tunas at Ocean Fronts. Fish. Oceanogr. 2000, 9, 328–342. [Google Scholar] [CrossRef]
- Tanabe, T. Feeding Habits of Skipjack Tuna Katsuwonus pelamis and Other Tuna Thunnus Spp. Juveniles in the Tropical Western Pacific. Fish. Sci. 2001, 67, 563–570. [Google Scholar] [CrossRef]
- Zhang, J.-Z.; Wanninkhof, R.; Lee, K. Enhanced New Production Observed from the Diurnal Cycle of Nitrate in an Oligotrophic Anticyclonic Eddy. Geophys. Res. Lett. 2001, 28, 1579–1582. [Google Scholar] [CrossRef]
- Logerwell, E.A.; Smith, P.E. Mesoscale Eddies and Survival of Late Stage Pacific Sardine (Sardinops sagax) Larvae. Fish. Oceanogr. 2001, 10, 13–25. [Google Scholar] [CrossRef]
- Duan, W.; He, B.; Yi Wang, K. Climate Change Impacts on Wave Characteristics along the Coast of Japan from 1986 to 2012. J. Coast. Res. 2014, 68, 97104. [Google Scholar] [CrossRef]





| Oceanographic Parameters | Unit | Spatial Footprint | Temporal Resolution |
|---|---|---|---|
| Sea Surface Temperature | °C | 4 × 4 km | Monthly |
| Chl-a concentration | mg m−3 | 4 × 4 km | Monthly |
| Net primary productivity | gr C m−2 month−1 | 9 × 9 km | Monthly |
| Current velocity (U,V) | m s−1 | 8 km × 8 km | Monthly |
| Variable | Edf | Ref.df | F | p-Value | CDE (%) |
|---|---|---|---|---|---|
| s(SST) | 8.591 | 8.947 | 12.688 | <2 × 10−16 | |
| s(Chl-a) | 6.984 | 8.019 | 2.716 | 0.00632 | |
| Total | 30.3 |
| SHI | Average Skipjack CPUE (Fish/Setting) |
|---|---|
| 0.1 | 0 |
| 0.2 | 110 |
| 0.3 | 200 |
| 0.4 | 20 |
| 0.5 | 398 |
| 0.6 | 514 |
| 0.7 | 940 |
| 0.8 | 1456 |
| 0.9 | 2008 |
| 1.0 | 2420 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zainuddin, M.; Safruddin, S.; Farhum, A.; Budimawan, B.; Hidayat, R.; Selamat, M.B.; Wiyono, E.S.; Ridwan, M.; Syamsuddin, M.; Ihsan, Y.N. Satellite-Based Ocean Color and Thermal Signatures Defining Habitat Hotspots and the Movement Pattern for Commercial Skipjack Tuna in Indonesia Fisheries Management Area 713, Western Tropical Pacific. Remote Sens. 2023, 15, 1268. https://doi.org/10.3390/rs15051268
Zainuddin M, Safruddin S, Farhum A, Budimawan B, Hidayat R, Selamat MB, Wiyono ES, Ridwan M, Syamsuddin M, Ihsan YN. Satellite-Based Ocean Color and Thermal Signatures Defining Habitat Hotspots and the Movement Pattern for Commercial Skipjack Tuna in Indonesia Fisheries Management Area 713, Western Tropical Pacific. Remote Sensing. 2023; 15(5):1268. https://doi.org/10.3390/rs15051268
Chicago/Turabian StyleZainuddin, Mukti, Safruddin Safruddin, Aisjah Farhum, Budimawan Budimawan, Rachmat Hidayat, Muhammad Banda Selamat, Eko Sri Wiyono, Muhammad Ridwan, Mega Syamsuddin, and Yudi Nurul Ihsan. 2023. "Satellite-Based Ocean Color and Thermal Signatures Defining Habitat Hotspots and the Movement Pattern for Commercial Skipjack Tuna in Indonesia Fisheries Management Area 713, Western Tropical Pacific" Remote Sensing 15, no. 5: 1268. https://doi.org/10.3390/rs15051268
APA StyleZainuddin, M., Safruddin, S., Farhum, A., Budimawan, B., Hidayat, R., Selamat, M. B., Wiyono, E. S., Ridwan, M., Syamsuddin, M., & Ihsan, Y. N. (2023). Satellite-Based Ocean Color and Thermal Signatures Defining Habitat Hotspots and the Movement Pattern for Commercial Skipjack Tuna in Indonesia Fisheries Management Area 713, Western Tropical Pacific. Remote Sensing, 15(5), 1268. https://doi.org/10.3390/rs15051268








