Using Visual Ozone Damage Scores and Spectroscopy to Quantify Soybean Responses to Background Ozone
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Site
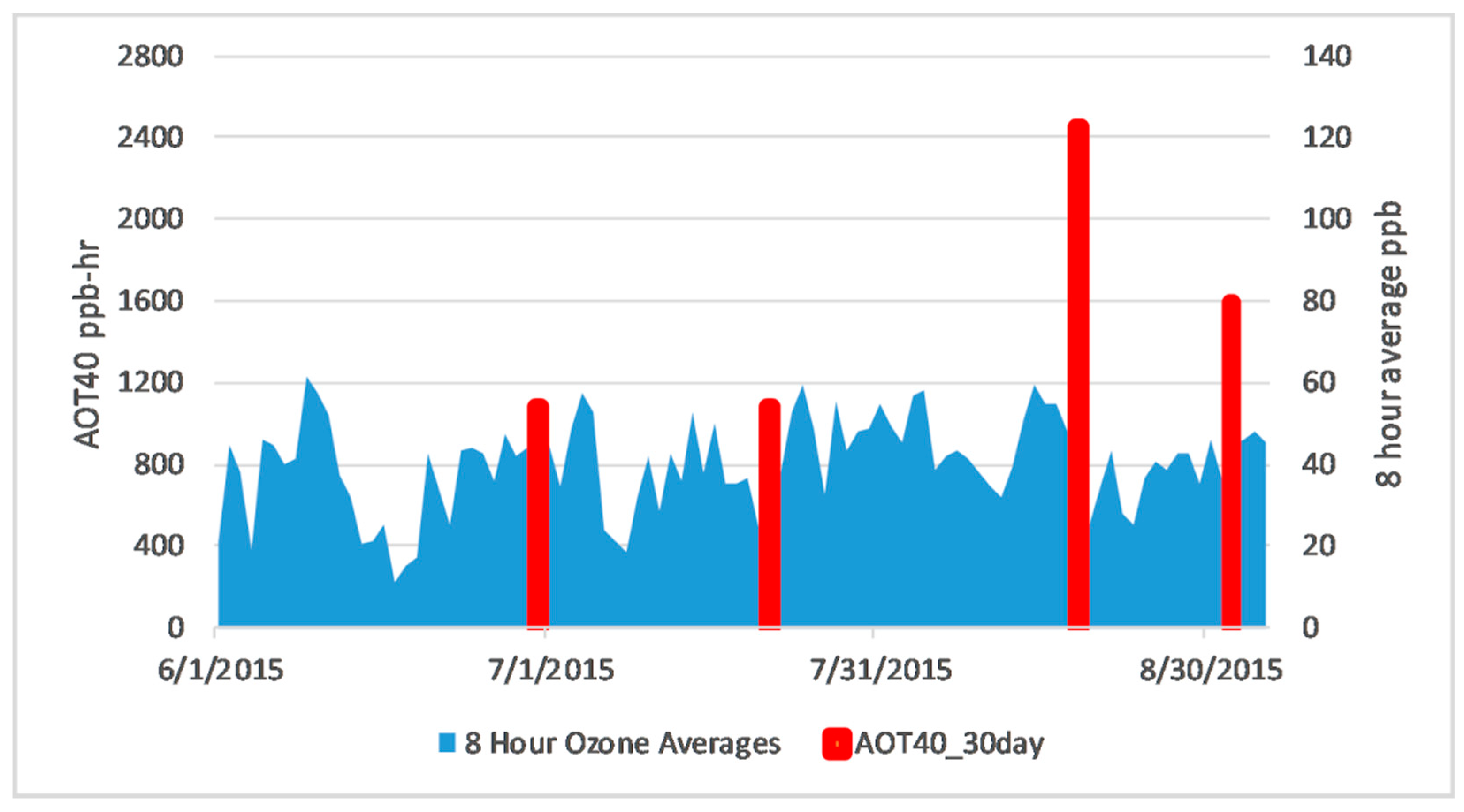
2.2. Background Ozone
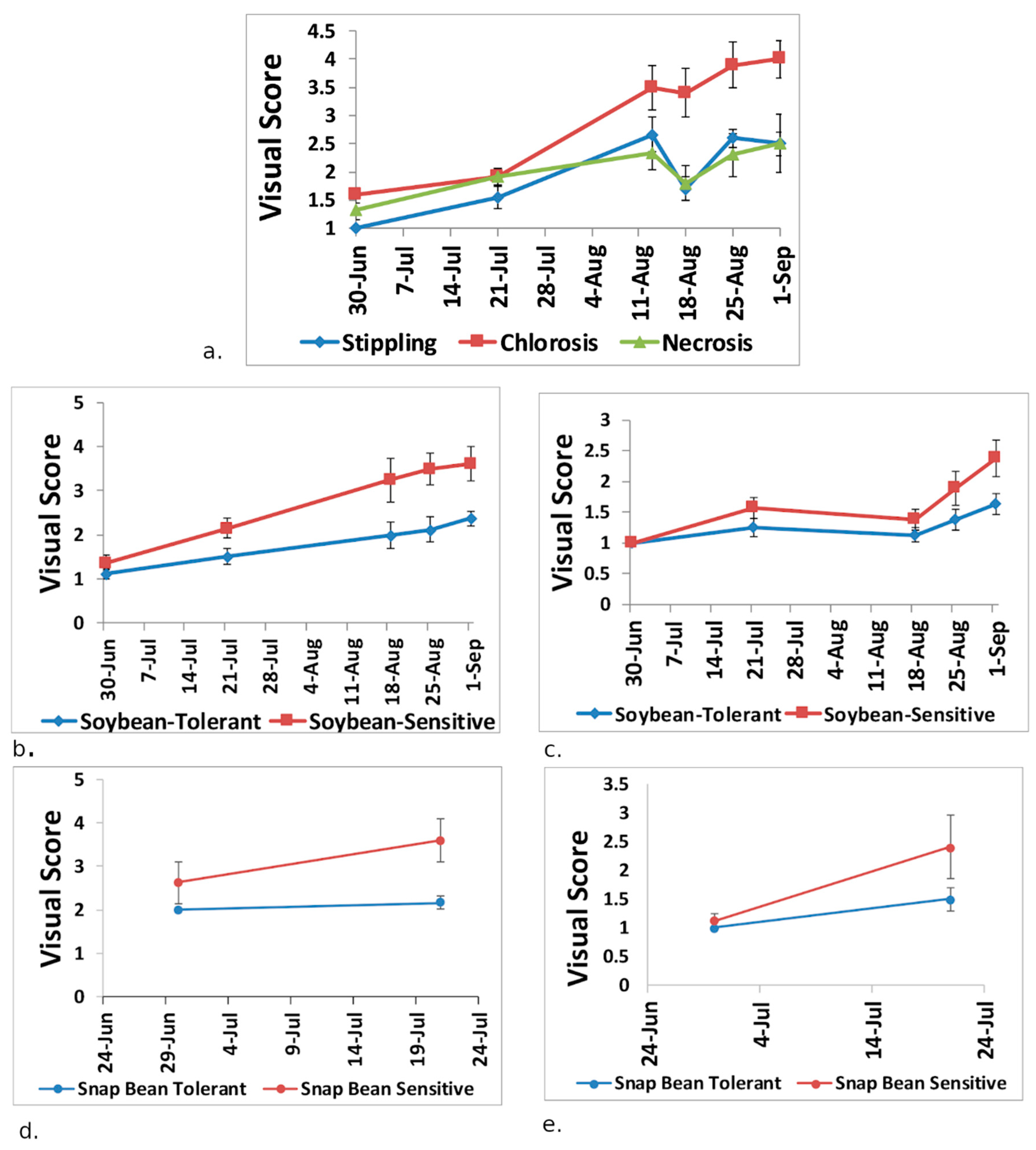
2.3. Visual Scoring of Ozone Damage
2.4. Leaf Spectral Data
2.5. Leaf Gas Exchange, Photosynthetic Rate, and Chlorophyll Content
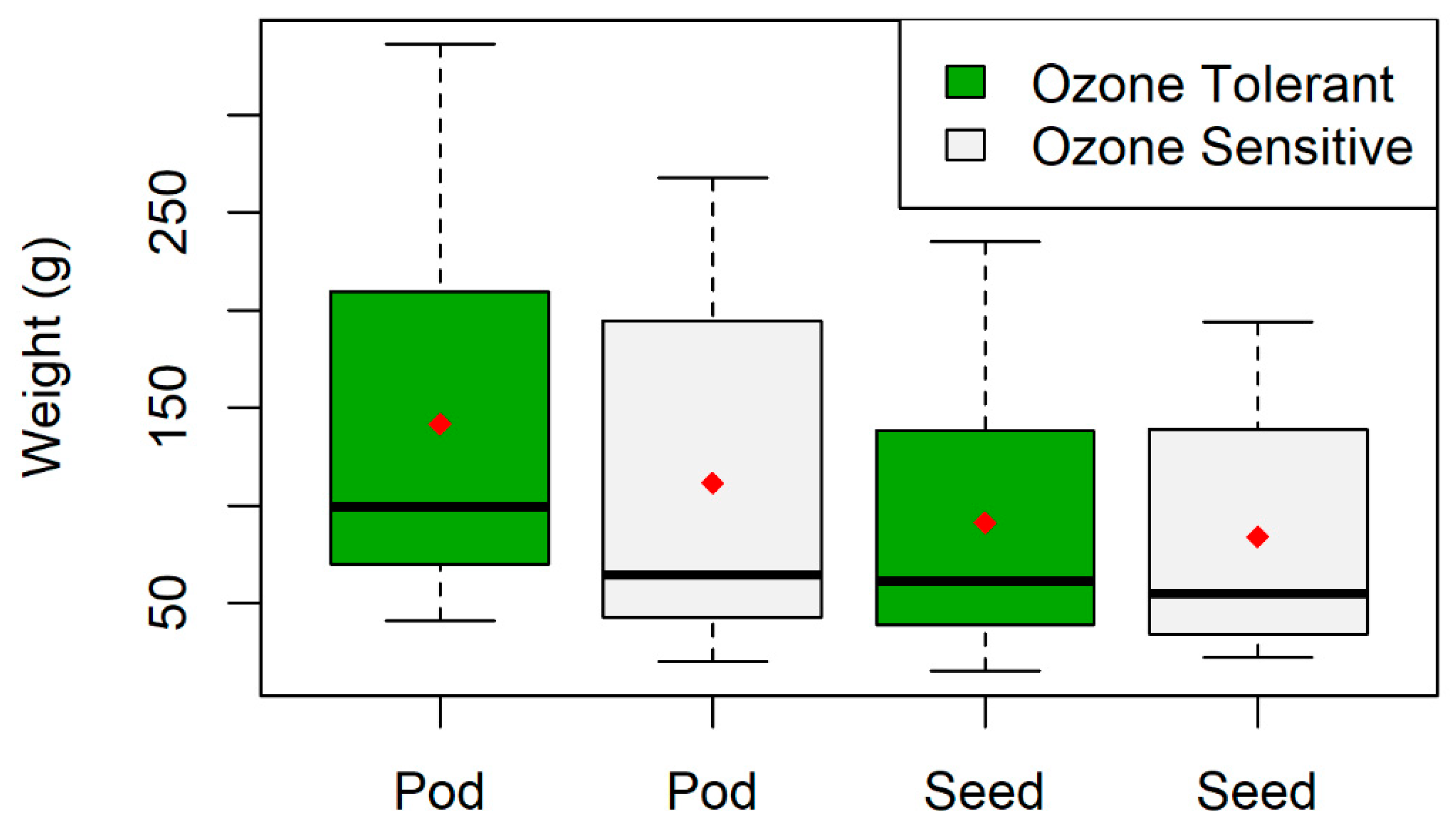
2.6. Pod and Seed Weight Data
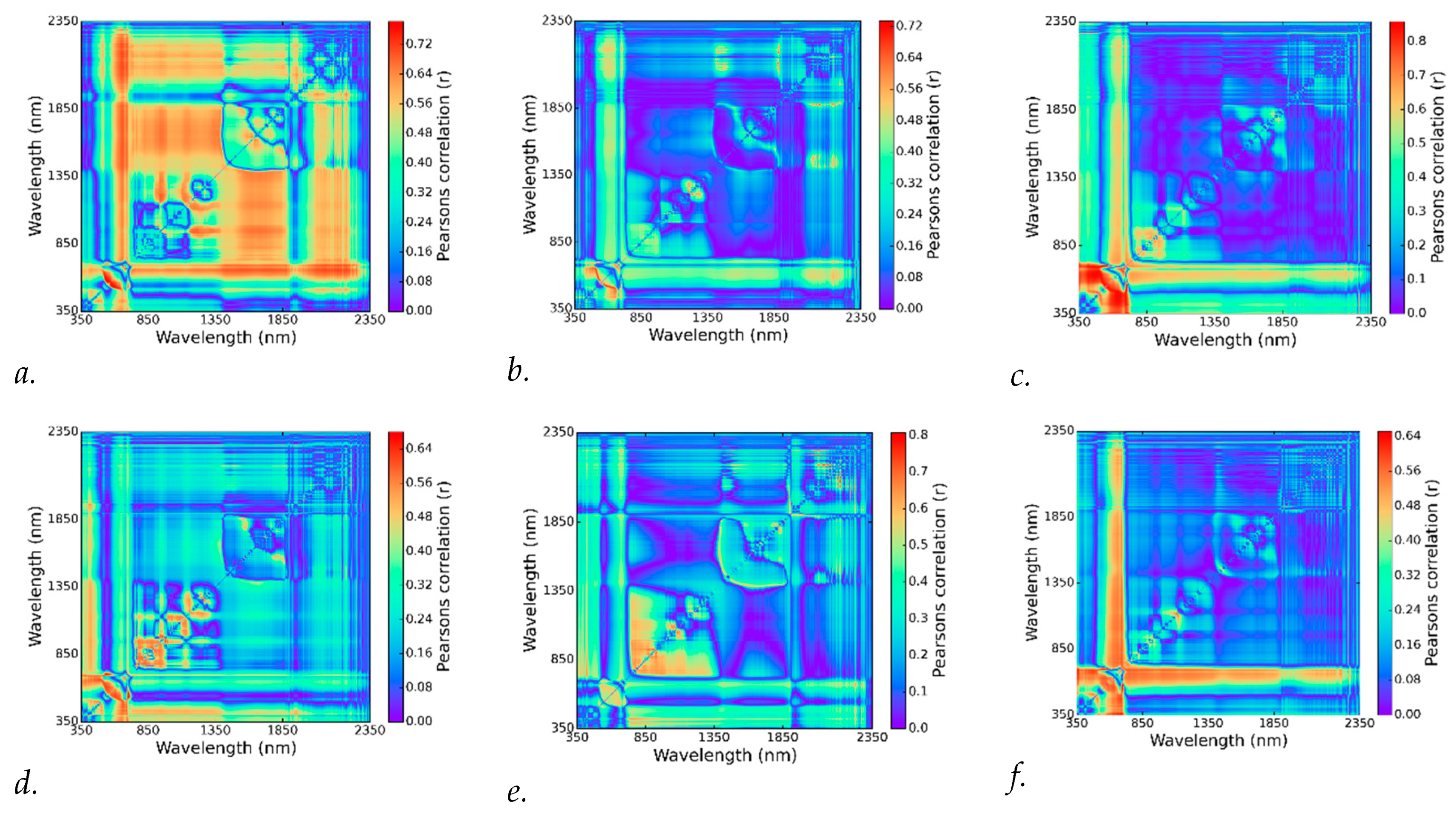
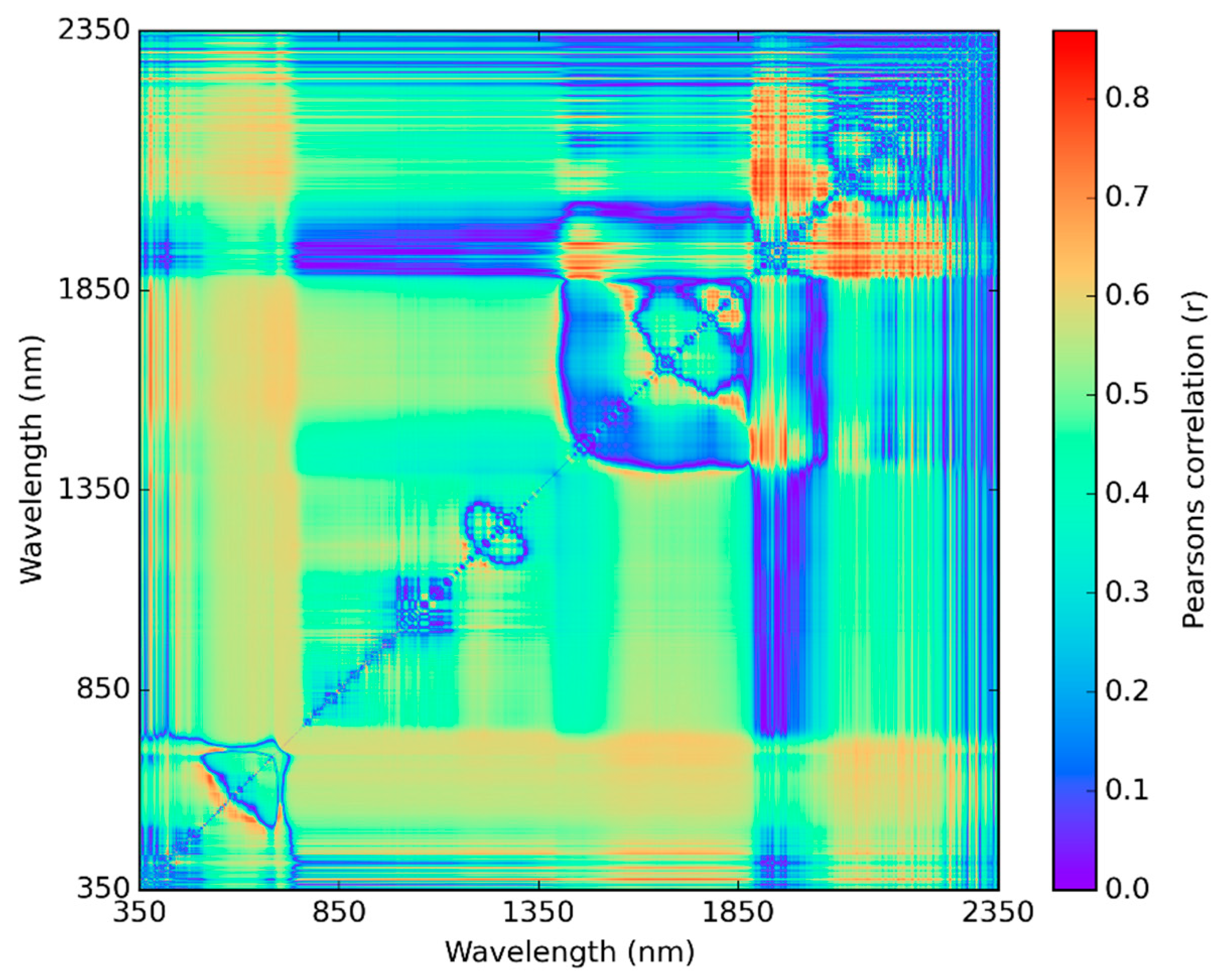
2.7. Statistical Analysis Using NDSI Spectral Correlation Mapping
3. Results
3.1. Plant Visual Scores and Seed/Pod Weight
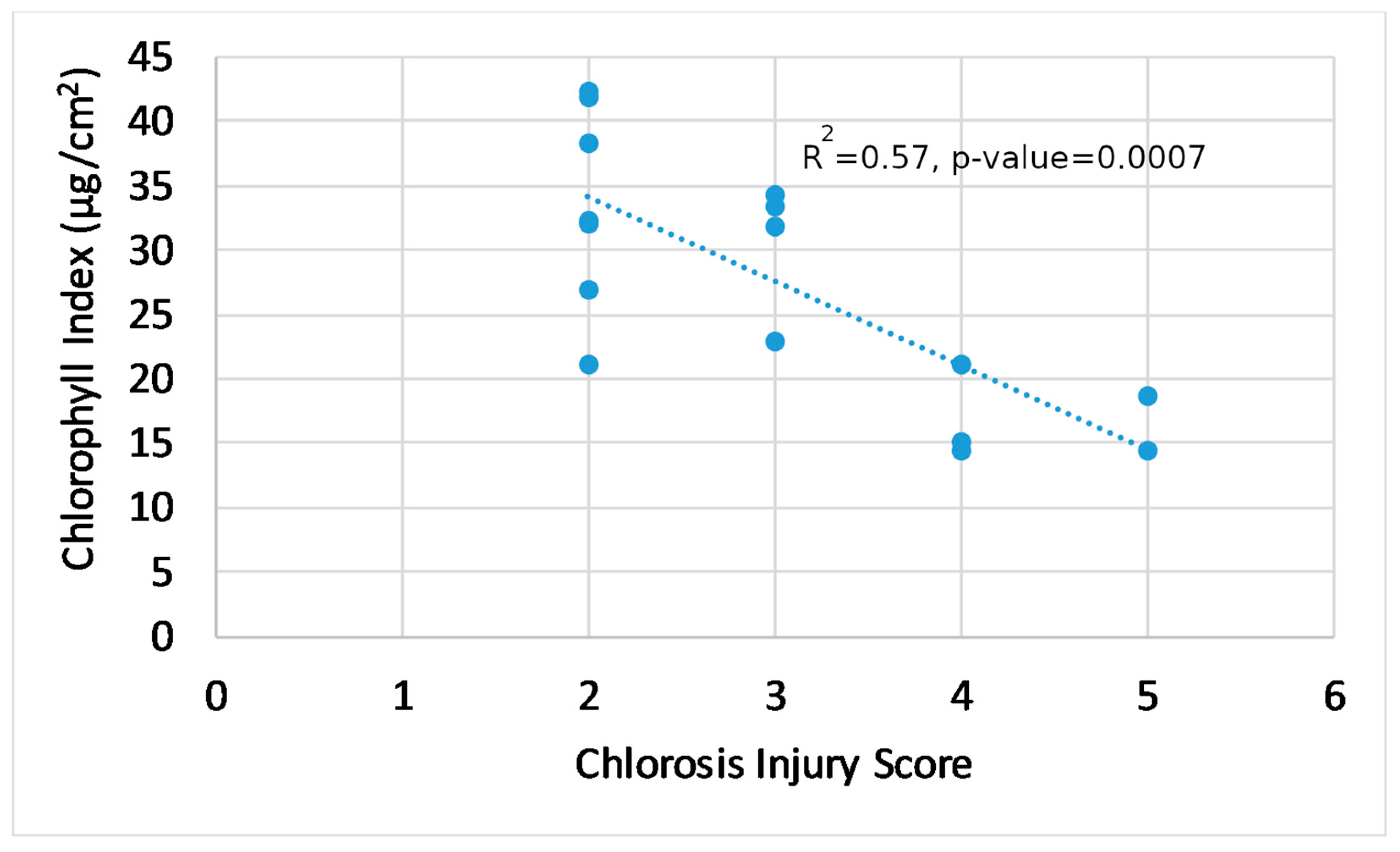
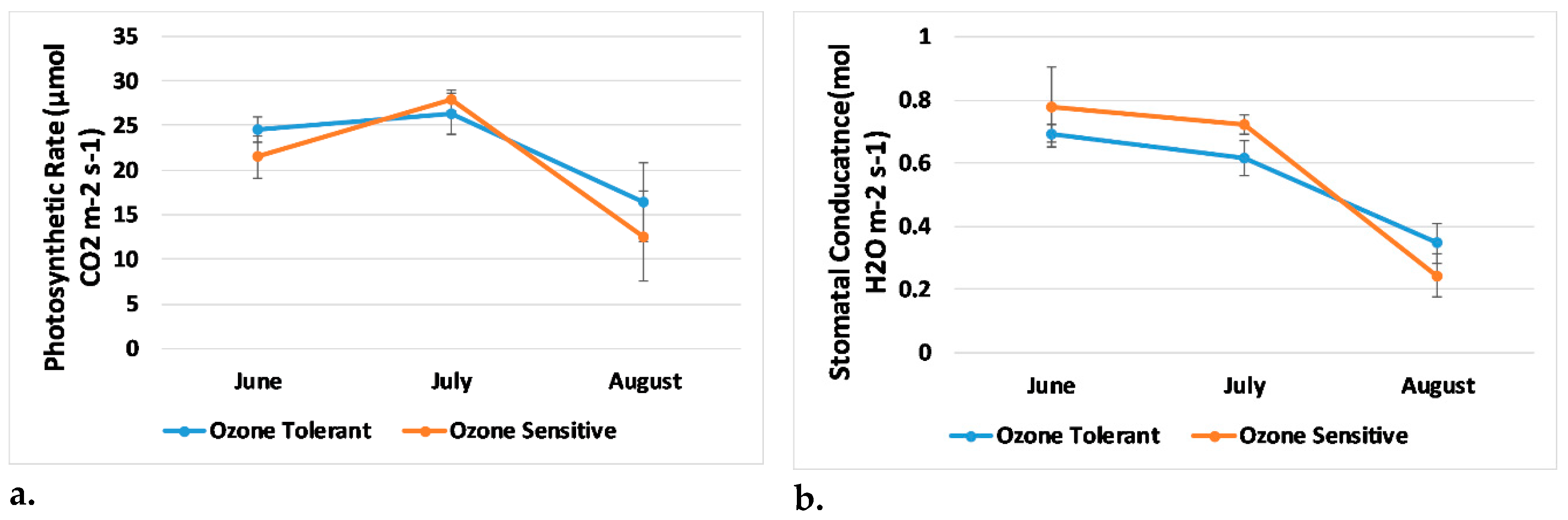
3.2. Plant Physiology and Visual Damage
3.3. Best Spectral Regions for NDSI Correlated with Visual Scores
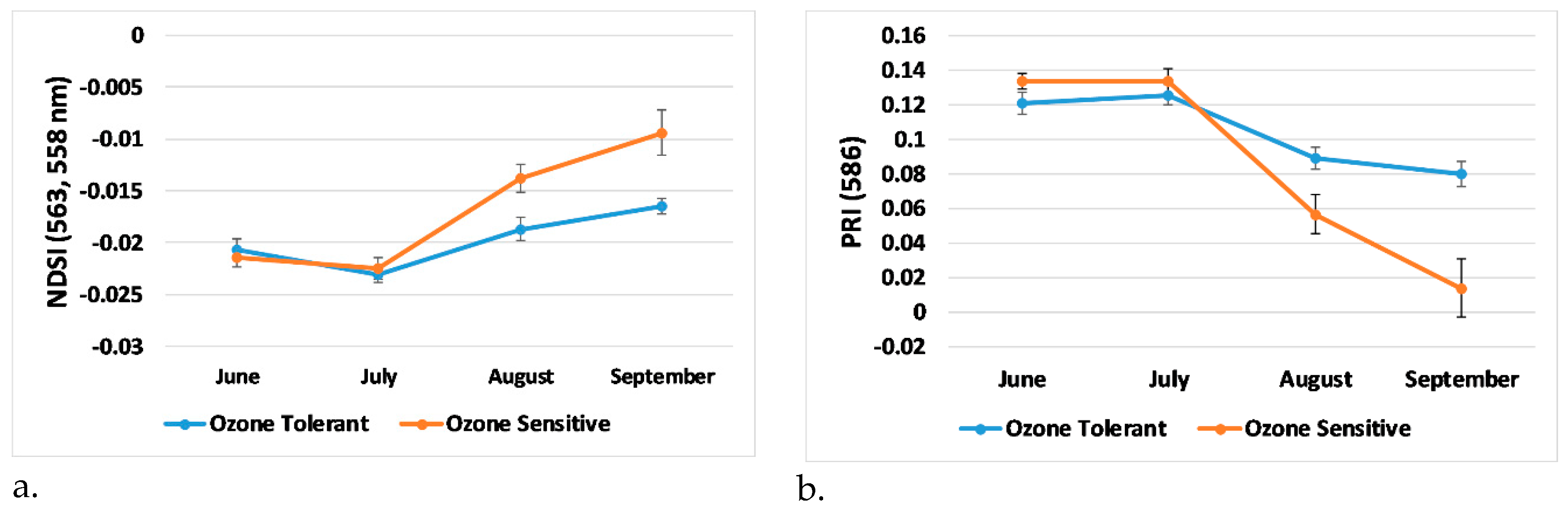
3.4. Trends in Generated Indices, Visual Score, and Plant Physiology
3.5. Comparison of Generated Indices with Existing Indices for Seed/Pod Weight Correlation
4. Discussion
5. Conclusions
- (1)
- A visual scoring system developed for bio-indicator plants was applied to soybeans to investigate the crop’s ozone damage. Correlations between foliar damage scores and physiological plant properties along with end-of-season seed and pod weight indicate this method as having potential for an ozone damage metric in soybeans.
- (2)
- NDSI [R563, R558] was identified as having the strongest correlation with soybean ozone damage chlorosis visual scores. Similar wavelengths were identified for common milkweed (NDSI [R558, R554]) and when data was evaluated for only the month of August (NDSI [R563, R560]) when there was a large range in chlorosis visual scores. The newly identified NDSI most sensitive to visible scores in August also had the highest correlation with soybean seed and pod weight when compared to multiple relevant indices well-established in the literature.
- (3)
- When evaluating the spectral bands with 3 and 9 nm bandwidth for use in an NDSI, longer wavelengths in the SWIR correlated best to chlorosis visual scores for soybeans in August and September. These bands may also indicate ozone sensitivity in soybeans due to ozone-induced changes in foliar lignin content.
- (4)
- Trends in newly developed NDSI showed separation between the ozone tolerant and sensitive genotypes after the July observation date. This agreed with ozone 8-h average observations along with analysis of time spent above 40 ppb for thirty days prior to each observation date, indicating that ozone had a greater effect on the soybean plants after July.
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Kirtman, B.; Power, S.; Adedoyin, A.; Boer, G.; Bojariu, R.; Camilloni, I.; Doblas-Reyes, F.; Fiore, A.; Kimoto, M.; Meehl, G. Near-Term Climate Change: Projections and Predictability; Cambridge University Press: Cambridge, UK; New York, NY, USA, 2013. [Google Scholar]
- Fowler, D.; Amann, M.; Anderson, F.; Ashmore, M.; Cox, P.; Depledge, M.; Derwent, D.; Grennfelt, P.; Hewitt, N.; Hov, O. Ground-Level Ozone in the 21st Century: Future Trends, Impacts and Policy Implications; Royal Society Science Policy Report; Royal Society Science: London, UK, 2008; Volume 15. [Google Scholar]
- Xu, J.; Ma, J.; Zhang, X.; Xu, X.; Xu, X.; Lin, W.; Wang, Y.; Meng, W.; Ma, Z. Measurements of ozone and its precursors in Beijing during summertime: Impact of urban plumes on ozone pollution in downwind rural areas. Atmos. Chem. Phys. 2011, 11, 12241–12252. [Google Scholar] [CrossRef]
- Emberson, L.D.; Pleijel, H.; Ainsworth, E.A.; Van den Berg, M.; Ren, W.; Osborne, S.; Mills, G.; Pandey, D.; Dentener, F.; Büker, P. Ozone effects on crops and consideration in crop models. Eur. J. Agron. 2018, 100, 19–34. [Google Scholar] [CrossRef]
- Van Dingenen, R.; Dentener, F.J.; Raes, F.; Krol, M.C.; Emberson, L.; Cofala, J. The global impact of ozone on agricultural crop yields under current and future air quality legislation. Atmos. Environ. 2009, 43, 604–618. [Google Scholar] [CrossRef]
- Backlund, P.; Janetos, A.; Schimel, D. The Effects of Climate Change on Agriculture, Land Resources, Water Resources, and Biodiversity in the United States; Synthesis and Assessment Product 4.3; US Environmental Protection Agency, Climate Change Science Program: Washington, DC, USA, 2008; 240p. [Google Scholar]
- Fishman, J.; Creilson, J.K.; Parker, P.A.; Ainsworth, E.A.; Vining, G.G.; Szarka, J.; Booker, F.L.; Xu, X. An investigation of widespread ozone damage to the soybean crop in the upper Midwest determined from ground-based and satellite measurements. Atmos. Environ. 2010, 44, 2248–2256. [Google Scholar] [CrossRef]
- Avnery, S.; Mauzerall, D.L.; Liu, J.; Horowitz, L.W. Global crop yield reductions due to surface ozone exposure: 2. Year 2030 potential crop production losses and economic damage under two scenarios of O3 pollution. Atmos. Environ. 2011, 45, 2297–2309. [Google Scholar] [CrossRef]
- Miller, P.R.; Stolte, K.W.; Duriscoe, D.M.; Pronos, J. Evaluating Ozone Air Pollution Effects on Pines in the Western United States; Gen. Tech. Rep. PSW-GTR-155; Pacific Southwest Research Station, Forest Service, US Department of Agriculture: Albany, CA, USA, 1996; Volume 155, 79p. [Google Scholar]
- Kefauver, S.C.; Penuelas, J.; Ribas, A.; Díaz-de-Quijano, M.; Ustin, S. Using Pinus uncinata to monitor tropospheric ozone in the Pyrenees. Ecol. Indic. 2014, 36, 262–271. [Google Scholar] [CrossRef]
- Matyssek, R.; Sandermann, H. Impact of ozone on trees: An ecophysiological perspective. In Progress in Botany; Springer: Berlin/Heidelberg, Germany, 2003; pp. 349–404. [Google Scholar]
- Klumpp, A.; Ansel, W.; Klumpp, G.; Vergne, P.; Sifakis, N.; Sanz, M.J.; Rasmussen, S.; Ro-Poulsen, H.; Ribas, A.; Penuelas, J. Ozone pollution and ozone biomonitoring in European cities Part II. Ozone-induced plant injury and its relationship with descriptors of ozone pollution. Atmos. Environ. 2006, 40, 7437–7448. [Google Scholar] [CrossRef]
- Burkey, K.O.; Miller, J.E.; Fiscus, E.L. Assessment of ambient ozone effects on vegetation using snap bean as a bioindicator species. J. Environ. Qual. 2005, 34, 1081–1086. [Google Scholar] [CrossRef]
- Klumpp, A.; Ansel, W.; Klumpp, G.; Belluzzo, N.; Calatayud, V.; Chaplin, N.; Garrec, J.; Gutsche, H.; Hayes, M.; Hentze, H. EuroBionet: A pan-European biomonitoring network for urban air quality assessment. Environ. Sci. Pollut. Res. 2002, 9, 199–203. [Google Scholar] [CrossRef]
- Mills, G.; Sharps, K.; Simpson, D.; Pleijel, H.; Broberg, M.; Uddling, J.; Jaramillo, F.; Davies, W.J.; Dentener, F.; Van den Berg, M. Ozone pollution will compromise efforts to increase global wheat production. Glob. Chang. Biol. 2018, 24, 3560–3574. [Google Scholar] [CrossRef]
- Heagle, A.; Miller, J.; Pursley, W. Influence of ozone stress on soybean response to carbon dioxide enrichment: III. Yield and seed quality. Crop Sci. 1998, 38, 128–134. [Google Scholar] [CrossRef]
- Sandermann, H., Jr. Ozone and plant health. Annu. Rev. Phytopathol. 1996, 34, 347–366. [Google Scholar] [CrossRef] [PubMed]
- Mills, G.; Hayes, F.; Simpson, D.; Emberson, L.; Norris, D.; Harmens, H.; Büker, P. Evidence of widespread effects of ozone on crops and (semi-) natural vegetation in Europe (1990–2006) in relation to AOT40-and flux-based risk maps. Glob. Chang. Biol. 2011, 17, 592–613. [Google Scholar] [CrossRef]
- Chappelka, A.; Neufeld, H.; Davison, A.; Somers, G.; Renfro, J. Ozone injury on cutleaf coneflower (Rudbeckia laciniata) and crown-beard (Verbesina occidentalis) in Great Smoky Mountains National Park. Environ. Pollut. 2003, 125, 53–59. [Google Scholar] [CrossRef]
- Duchelle, S.; Skelly, J. The response of Asclepias syriaca to oxidant air pollution in the Shenandoah National Park of Virginia. Plant Dis. 1981, 65, 661–663. [Google Scholar] [CrossRef]
- Ladd, I.; Skelly, J.; Pippin, M.; Fishman, J. Ozone Induced Foliar Injury Field Guide; NASA Langley Research Center: Hampton, VA, USA, 2011. [Google Scholar]
- Keen, N.T.; Taylor, O. Ozone injury in soybeans: Isoflavonoid accumulation is related to necrosis. Plant Physiol. 1975, 55, 731–733. [Google Scholar] [CrossRef]
- Heagle, A.S.; Miller, J.E.; Rawlings, J.O.; Vozzo, S.F. Effect of growth stage on soybean response to chronic ozone exposure. J. Environ. Qual. 1991, 20, 562–570. [Google Scholar] [CrossRef]
- Cure, W.; Nusser, S.; Heagle, A. Canopy Reflectance of Soybean as Affected by Chronic Doses of Ozone in Open-Top Field Chambers; North Carolina State Univ.: Raleigh, NC, USA, 1988. [Google Scholar]
- Williams, J.; Ashenden, T. Differences in the spectral characteristics of white clover exposed to gaseous pollutants and acid mist. New Phytol. 1992, 120, 69–75. [Google Scholar] [CrossRef]
- Ustin, S.L.; Curtiss, B. Spectral characteristics of ozone-treated conifers. Environ. Exp. Bot. 1990, 30, 293–308. [Google Scholar] [CrossRef]
- Gamon, J.; Penuelas, J.; Field, C. A narrow-waveband spectral index that tracks diurnal changes in photosynthetic efficiency. Remote Sens. Environ. 1992, 41, 35–44. [Google Scholar] [CrossRef]
- Gamon, J.; Serrano, L.; Surfus, J. The photochemical reflectance index: An optical indicator of photosynthetic radiation use efficiency across species, functional types, and nutrient levels. Oecologia 1997, 112, 492–501. [Google Scholar] [CrossRef] [PubMed]
- Campbell, P.; Middleton, E.; McMurtrey, J.; Chappelle, E. Assessment of vegetation stress using reflectance or fluorescence measurements. J. Environ. Qual. 2007, 36, 832–845. [Google Scholar] [CrossRef] [PubMed]
- Castagna, A.; Nali, C.; Ciompi, S.; Lorenzini, G.; Soldatini, G.; Ranieri, A. Ozone exposure affects photosynthesis of pumpkin (Cucurbita pepo) plants. New Phytol. 2001, 152, 223–229. [Google Scholar] [CrossRef]
- Ranieri, A.; Giuntini, D.; Ferraro, F.; Nali, C.; Baldan, B.; Lorenzini, G.; Soldatini, G.F. Chronic ozone fumigation induces alterations in thylakoid functionality and composition in two poplar clones. Plant Physiol. Biochem. 2001, 39, 999–1008. [Google Scholar] [CrossRef]
- Ainsworth, E.A.; Serbin, S.P.; Skoneczka, J.A.; Townsend, P.A. Using leaf optical properties to detect ozone effects on foliar biochemistry. Photosynth. Res. 2014, 119, 65–76. [Google Scholar] [CrossRef] [PubMed]
- Aparicio, N.; Villegas, D.; Casadesus, J.; Araus, J.L.; Royo, C. Spectral vegetation indices as nondestructive tools for determining durum wheat yield. Agron. J. 2000, 92, 83–91. [Google Scholar] [CrossRef]
- Panda, S.S.; Ames, D.P.; Panigrahi, S. Application of vegetation indices for agricultural crop yield prediction using neural network techniques. Remote Sens. 2010, 2, 673–696. [Google Scholar] [CrossRef]
- Bolton, D.K.; Friedl, M.A. Forecasting crop yield using remotely sensed vegetation indices and crop phenology metrics. Agric. For. Meteorol. 2013, 173, 74–84. [Google Scholar] [CrossRef]
- Ma, B.; Dwyer, L.M.; Costa, C.; Cober, E.R.; Morrison, M.J. Early prediction of soybean yield from canopy reflectance measurements. Agron. J. 2001, 93, 1227–1234. [Google Scholar] [CrossRef]
- Ashmore, M. Assessing the future global impacts of ozone on vegetation. Plant Cell Environ. 2005, 28, 949–964. [Google Scholar] [CrossRef]
- Fiscus, E.L.; Booker, F.L.; Burkey, K.O. Crop responses to ozone: Uptake, modes of action, carbon assimilation and partitioning. Plant Cell Environ. 2005, 28, 997–1011. [Google Scholar] [CrossRef]
- Biswas, D.; Xu, H.; Li, Y.; Liu, M.; Chen, Y.; Sun, J.; Jiang, G. Assessing the genetic relatedness of higher ozone sensitivity of modern wheat to its wild and cultivated progenitors/relatives. J. Exp. Bot. 2008, 59, 951–963. [Google Scholar] [CrossRef] [PubMed]
- Morgan, P.B.; Bernacchi, C.J.; Ort, D.R.; Long, S.P. An in vivo analysis of the effect of season-long open-air elevation of ozone to anticipated 2050 levels on photosynthesis in soybean. Plant Physiol. 2004, 135, 2348–2357. [Google Scholar] [CrossRef] [PubMed]
- Mills, G.; Pleijel, H.; Braun, S.; Büker, P.; Bermejo, V.; Calvo, E.; Danielsson, H.; Emberson, L.; Fernández, I.G.; Grünhage, L. New stomatal flux-based critical levels for ozone effects on vegetation. Atmos. Environ. 2011, 45, 5064–5068. [Google Scholar] [CrossRef]
- Bernacchi, C.J.; Leakey, A.D.; Heady, L.E.; Morgan, P.B.; Dohleman, F.G.; McGrath, J.M.; Gillespie, K.M.; Wittig, V.E.; Rogers, A.; Long, S.P. Hourly and seasonal variation in photosynthesis and stomatal conductance of soybean grown at future CO2 and ozone concentrations for 3 years under fully open-air field conditions. Plant Cell Environ. 2006, 29, 2077–2090. [Google Scholar] [CrossRef]
- Betzelberger, A.M.; Yendrek, C.R.; Sun, J.; Leisner, C.P.; Nelson, R.L.; Ort, D.R.; Ainsworth, E.A. Ozone exposure response for US soybean cultivars: Linear reductions in photosynthetic potential, biomass, and yield. Plant Physiol. 2012, 160, 1827–1839. [Google Scholar] [CrossRef]
- Singh, E.; Tiwari, S.; Agrawal, M. Effects of elevated ozone on photosynthesis and stomatal conductance of two soybean varieties: A case study to assess impacts of one component of predicted global climate change. Plant Biol. 2009, 11, 101–108. [Google Scholar] [CrossRef]
- Mulchi, C.L.; Lee, E.; Tuthill, K.; Olinick, E. Influence of ozone stress on growth processes, yields and grain quality characteristics among soybean cultivars. Environ. Pollut. 1988, 53, 151–169. [Google Scholar] [CrossRef]
- Ghude, S.D.; Jena, C.; Chate, D.; Beig, G.; Pfister, G.; Kumar, R.; Ramanathan, V. Reductions in India’s crop yield due to ozone. Geophys. Res. Lett. 2014, 41, 5685–5691. [Google Scholar] [CrossRef]
- McGrath, J.M.; Betzelberger, A.M.; Wang, S.; Shook, E.; Zhu, X.-G.; Long, S.P.; Ainsworth, E.A. An analysis of ozone damage to historical maize and soybean yields in the United States. Proc. Natl. Acad. Sci. USA 2015, 112, 14390–14395. [Google Scholar] [CrossRef]
- Betzelberger, A.M.; Gillespie, K.M.; Mcgrath, J.M.; Koester, R.P.; Nelson, R.L.; Ainsworth, E.A. Effects of chronic elevated ozone concentration on antioxidant capacity, photosynthesis and seed yield of 10 soybean cultivars. Plant Cell Environ. 2010, 33, 1569–1581. [Google Scholar] [CrossRef] [PubMed]
- University of Kuopio; Department of Ecology; Environmental Science; Lauri Kärenlampi; Lena Skärby; Workshop on Long-Range Transboundary Air Pollution. Critical Levels for Ozone in Europe: Testing and Finalizing the Concepts: UN-ECE Workshop Report: UN-ECE Convention on Long-Range Transboundary Air Pollution Workshop in Kuopio, Finland, 15–17 April, 1996, Organized by University of Kuopio, Department of Ecology and Environmental Science, Swedish Environmental Research Institute (IVL), Gothenburg, Sponsored by Nordic Council of Ministers (NMR); Kuopio University Printing Office: Kuopio, Finland, 1996. [Google Scholar]
- Fuhrer, J.; Skärby, L.; Ashmore, M.R. Critical levels for ozone effects on vegetation in Europe. Environ. Pollut. 1997, 97, 91–106. [Google Scholar] [CrossRef]
- Mills, G.; Buse, A.; Gimeno, B.; Bermejo, V.; Holland, M.; Emberson, L.; Pleijel, H. A synthesis of AOT40-based response functions and critical levels of ozone for agricultural and horticultural crops. Atmos. Environ. 2007, 41, 2630–2643. [Google Scholar] [CrossRef]
- Naeve, S.; Soybean Growth Stages. UMN Extension. Available online: https://extension.umn.edu/growing-soybean/soybean-growth-stages#days-between-stages-539862 (accessed on 10 December 2019).
- Van Der Walt, S.; Colbert, S.C.; Varoquaux, G. The NumPy array: A structure for efficient numerical computation. Comput. Sci. Eng. 2011, 13, 22. [Google Scholar] [CrossRef]
- Hunter, J.D. Matplotlib: A 2D graphics environment. Comput. Sci. Eng. 2007, 9, 90. [Google Scholar] [CrossRef]
- Panigada, C.; Rossini, M.; Meroni, M.; Cilia, C.; Busetto, L.; Amaducci, S.; Boschetti, M.; Cogliati, S.; Picchi, V.; Pinto, F. Fluorescence, PRI and canopy temperature for water stress detection in cereal crops. Int. J. Appl. Earth Obs. Geoinf. 2014, 30, 167–178. [Google Scholar] [CrossRef]
- Haboudane, D.; Miller, J.R.; Pattey, E.; Zarco-Tejada, P.J.; Strachan, I.B. Hyperspectral vegetation indices and novel algorithms for predicting green LAI of crop canopies: Modeling and validation in the context of precision agriculture. Remote Sens. Environ. 2004, 90, 337–352. [Google Scholar] [CrossRef]
- Merzlyak, M.N.; Gitelson, A.A.; Chivkunova, O.B.; Rakitin, V.Y. Non-destructive optical detection of pigment changes during leaf senescence and fruit ripening. Physiol. Plant. 1999, 106, 135–141. [Google Scholar] [CrossRef]
- Rouse, J.W., Jr.; Haas, R.; Schell, J.; Deering, D. Monitoring Vegetation Systems in the Great Plains with ERTS; NASA, Goddard Space Flight Center 3d ERTS-1 Symp. Sect. A; United States Texas A&M Univ.: College Station, TX, USA, 1974; Volume 1, pp. 309–317. [Google Scholar]
- Sims, D.A.; Gamon, J.A. Relationships between leaf pigment content and spectral reflectance across a wide range of species, leaf structures and developmental stages. Remote Sens. Environ. 2002, 81, 337–354. [Google Scholar] [CrossRef]
- Penuelas, J.; Baret, F.; Filella, I. Semi-empirical indices to assess carotenoids/chlorophyll a ratio from leaf spectral reflectance. Photosynthetica 1995, 31, 221–230. [Google Scholar]
- Gitelson, A.A.; Zur, Y.; Chivkunova, O.B.; Merzlyak, M.N. Assessing Carotenoid Content in Plant Leaves with Reflectance Spectroscopy. Photochem. Photobiol. 2002, 75, 272–281. [Google Scholar] [CrossRef]
- Gitelson, A.A.; Merzlyak, M.N.; Chivkunova, O.B. Optical properties and nondestructive estimation of anthocyanin content in plant leaves. Photochem. Photobiol. 2001, 74, 38–45. [Google Scholar] [CrossRef]
- Gitelson, A.; Merzlyak, M.N. Spectral reflectance changes associated with autumn senescence of Aesculus hippocastanum L. and Acer platanoides L. leaves. Spectral features and relation to chlorophyll estimation. J. Plant Physiol. 1994, 143, 286–292. [Google Scholar] [CrossRef]
- Gamon, J.; Surfus, J. Assessing leaf pigment content and activity with a reflectometer. New Phytol. 1999, 143, 105–117. [Google Scholar] [CrossRef]
- Gitelson, A.A.; Kaufman, Y.J.; Merzlyak, M.N. Use of a green channel in remote sensing of global vegetation from EOS-MODIS. Remote Sens. Environ. 1996, 58, 289–298. [Google Scholar] [CrossRef]
- Haboudane, D.; Miller, J.R.; Tremblay, N.; Zarco-Tejada, P.J.; Dextraze, L. Integrated narrow-band vegetation indices for prediction of crop chlorophyll content for application to precision agriculture. Remote Sens. Environ. 2002, 81, 416–426. [Google Scholar] [CrossRef]
- Sagan, V.; Maimaitiyiming, M.; Fishman, J. Effects of Ambient Ozone on Soybean Biophysical Variables and Mineral Nutrient Accumulation. Remote Sens. 2018, 10, 562. [Google Scholar] [CrossRef]
- Daughtry, C.; Walthall, C.; Kim, M.; De Colstoun, E.B.; McMurtrey Iii, J. Estimating corn leaf chlorophyll concentration from leaf and canopy reflectance. Remote Sens. Environ. 2000, 74, 229–239. [Google Scholar] [CrossRef]
- Guyot, G.; Baret, F. Utilisation de la haute resolution spectrale pour suivre l’etat des couverts vegetaux. In Proceedings of the Spectral Signatures of Objects in Remote Sensing, Aussois, France, 18–22 January 1988; p. 279. [Google Scholar]
- Zarco-Tejada, P.J.; Miller, J.R.; Noland, T.L.; Mohammed, G.H.; Sampson, P.H. Scaling-up and model inversion methods with narrowband optical indices for chlorophyll content estimation in closed forest canopies with hyperspectral data. IEEE Trans. Geosci. Remote Sens. 2001, 39, 1491–1507. [Google Scholar] [CrossRef]
- Daughtry, C.S. Discriminating crop residues from soil by shortwave infrared reflectance. Agron. J. 2001, 93, 125–131. [Google Scholar] [CrossRef]
- Broge, N.H.; Leblanc, E. Comparing prediction power and stability of broadband and hyperspectral vegetation indices for estimation of green leaf area index and canopy chlorophyll density. Remote Sens. Environ. 2001, 76, 156–172. [Google Scholar] [CrossRef]
- Cheng, T.; Rivard, B.; Sanchez-Azofeifa, A. Spectroscopic determination of leaf water content using continuous wavelet analysis. Remote Sens. Environ. 2011, 115, 659–670. [Google Scholar] [CrossRef]
- Pell, E.J.; Schlagnhaufer, C.D.; Arteca, R.N. Ozone-induced oxidative stress: Mechanisms of action and reaction. Physiol. Plant. 1997, 100, 264–273. [Google Scholar] [CrossRef]
- Booker, F.L.; Miller, J.E. Phenylpropanoid metabolism and phenolic composition of soybean [Glycine max (L.) Merr.] leaves following exposure to ozone. J. Exp. Bot. 1998, 49, 1191–1202. [Google Scholar] [CrossRef]
- Cabané, M.; Pireaux, J.-C.; Léger, E.; Weber, E.; Dizengremel, P.; Pollet, B.; Lapierre, C. Condensed lignins are synthesized in poplar leaves exposed to ozone. Plant Physiol. 2004, 134, 586–594. [Google Scholar] [CrossRef]

| Plant Species | Spectral Observation Dates |
|---|---|
| Soybean (Glycine max) [2 varieties: Dwight (O3 sensitive) and Pana (O3 tolerant)] | 30 June 2015 28 July 2015 18 August 2015 1 September 2015 |
| Common Milkweed (Asclepias syriaca) | 30 June 2015 28 July 2015 18 August 2015 1 September 2015 |
| Snap Beans (Phaseolus vulgaris) [2 varieties: S156 (O3 sensitive) and R123 (O3 tolerant)] | 30 June 2015 21 July 2015 |
| Chlorosis Score | Necrosis Score | ||||||
|---|---|---|---|---|---|---|---|
| Month | R2 | p-value | MAE (g) | R2 | p-value | MAE (g) | |
| Pod Weight (g) | Aug | 0.60 | 0.0004 | 45.95 | 0.21 | 0.0750 | 69.73 |
| Sept | 0.37 | 0.0119 | 60.75 | 0.20 | 0.0797 | 68.32 | |
| Seed Weight (g) | Aug | 0.52 | 0.0016 | 37.00 | 0.18 | 0.1012 | 48.75 |
| Sept | 0.28 | 0.0341 | 45.59 | 0.14 | 0.1588 | 49.81 | |
| August Only | Overall (June–August) | |||
|---|---|---|---|---|
| r | p-value | r | p-value | |
| Photosynthetic Rate | −0.685 | 0.0034 | −0.585 | 0.00002 |
| Transpiration | −0.641 | 0.0074 | −0.534 | 0.00011 |
| Stomatal Conductance | −0.694 | 0.0029 | −0.537 | 0.00010 |
| Chlorosis | Necrosis | |||||
|---|---|---|---|---|---|---|
| Crop | Month(s) | Band-width | R2 | Wavelength of Max R2 (nm) | R2 | Wavelength of Max R2 (nm) |
| Soybeans | June–Sept | 1 nm | 0.61 | 563, 558 | 0.46 | 962, 978 |
| 3 nm | 0.59 | 533–535, 590–592 | 0.42 | 530–532, 693–695 | ||
| 9 nm | 0.58 | 530–538, 584–592 | 0.42 | 530–538, 692–700 | ||
| August | 1 nm | 0.76 | 563, 560 | 0.65 | 927, 917 | |
| 3 nm | 0.75 | 1907–1909, 2378–2380 | 0.60 | 1949–1951, 1955–1957 | ||
| 9 nm | 0.68 | 1889–1897, 2141–2149 | 0.55 | 2141–2149, 2357–2365 | ||
| September | 1 nm | 0.79 | 571, 568 | 0.79 | 2218, 2326 | |
| 3 nm | 0.80 | 2156–2158, 2183–2185 | 0.71 | 662–664, 665–667 | ||
| 9 nm | 0.80 | 2150–2158, 2177–2185 | 0.67 | 674–682, 692–700 | ||
| Snap Beans | June–July | 1 nm | 0.54 | 1876, 2187 | 0.65 | 926, 917 |
| 3 nm | 0.54 | 1874–1876, 2184–2186 | 0.59 | 911–913, 926–928 | ||
| 9 nm | 0.53 | 1871–1880, 2186–2194 | 0.52 | 872–880, 881–889 | ||
| Milkweed | June–Sept | 1 nm | 0.74 | 558, 554 | 0.42 | 697, 581 |
| 3 nm | 0.73 | 530–533, 701–703 | 0.42 | 572–574, 698–700 | ||
| 9 nm | 0.71 | 476–484, 638–646 | 0.41 | 575–583, 692–700 | ||
| Aug–Sept | 1 nm | 0.88 | 743, 745 | 0.77 | 1993, 2036 | |
| 3 nm | 0.87 | 740–742, 743–745 | 0.72 | 1994–1996, 2075–2077 | ||
| 9 nm | 0.87 | 737–745, 746–754 | 0.64 | 1988–1996, 2033–2041 | ||
| Spectral Index | Acronym | Equation | R2 (Chlorosis Visual Score) | R2 (Pod Weight) | R2 (Seed Weight) | Ref |
|---|---|---|---|---|---|---|
| Plant Senescence Reflectance Index | PSRI | (R680 − R500)/R750 | 0.00 | 0.03 | 0.07 | [57] |
| Normalized Difference Vegetation Index | NDVI | (R800 − R680)/(R800 + R680) | 0.09 | 0.05 | 0.02 | [58] |
| Modified Simple Ratio | mSR705 | (R750 − R445)/(R705 − R445) | 0.19 | 0.13 | 0.09 | [59] |
| Structure Insensitive Pigment Index | SIPI | (R800 − R445)/(R800 − R680) | 0.21 | 0.13 | 0.08 | [60] |
| Carotenoid Index (Gitelson) | CarGtln | 1/R510 − 1/R550 | 0.21 | 0.13 | 0.11 | [61] |
| Anthocyanin (Gitelson) | ANTGtln | (1/R550 − 1/R700)xR800 | 0.22 | 0.13 | 0.07 | [62] |
| Chlorophyll Index | CI | (R750 − R705)/(R750 + R705) | 0.23 | 0.12 | 0.05 | [63] |
| Anthocyanin (Gamon) | ANTGmn | R650/R550 | 0.23 | 0.08 | 0.03 | [64] |
| Photochemical Reflectance Index (570) | PRI570 | (R531 − R570)/(R531 + R570) | 0.23 | 0.21 | 0.14 | [27] |
| Green Normalized Difference Vegetation Index | GNDVI | (R750 − R540 + R570)/(R750 + R540 − R570) | 0.23 | 0.20 | 0.13 | [65] |
| Red Edge Ratio Index | RERI | R700/R670 | 0.26 | 0.07 | 0.02 | [66] |
| Photochemical Reflectance Index (519) | PRI519 | (R531 − R519)/(R531 + R519) | 0.27 | 0.14 | 0.09 | [67] |
| Photochemical Reflectance Index (525) | PRI525 | (R531 − R525)/(R531 + R525) | 0.29 | 0.20 | 0.15 | [67] |
| Transformed Chlorophyll Absorption in Reflectance Index | TCARI | 3 × ((R700 − R670) − 0.2 × (R700 − R550) × (R700/R670)) | 0.29 | 0.13 | 0.06 | [66] |
| Modified Chlorophyll Absorption in Reflectance Index | MCARI | [(R700 − R670) − 0.2(R700 − R550)] × (R700/R670) | 0.29 | 0.13 | 0.06 | [68] |
| Red Edge Position | REP | 700 + 40 × ([R670 + R780)/2 − R700]/(R740 − R700)) | 0.31 | 0.11 | 0.05 | [69] |
| Red Edge | ZM | R750/R710 | 0.33 | 0.11 | 0.06 | [70] |
| Cellulose Absorption Index | CAI | 0.5(R2000 + R2200) − R2100 | 0.34 | 0.03 | 0.01 | [71] |
| Photochemical Reflectance Index (586) | PRI586 | (R531 − R586)/(R531 + R586) | 0.38 | 0.16 | 0.10 | [55] |
| Triangular Vegetation Index | TVI | 0.5 × (120 × (R750 − R550) − 200 × (R670 − R550)) | 0.39 | 0.20 | 0.15 | [72] |
| Modified Triangular Vegetation Index | MTVI | 1.2 × [1.2(R800 − R550) − 2.5(R670 − R550)] | 0.40 | 0.21 | 0.16 | [56] |
| NDSI Band | NDSI | (R1907-1909 − R2378-2380)/(R1907-1909 + R2378-2380) | 0.75 | 0.48 | 0.39 | This study |
| Normalized Difference Spectral Index (563 nm, 560 nm) | NDSI | (R563 − R560)/(R563 + R560) | 0.76 | 0.64 | 0.54 | This study |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gosselin, N.; Sagan, V.; Maimaitiyiming, M.; Fishman, J.; Belina, K.; Podleski, A.; Maimaitijiang, M.; Bashir, A.; Balakrishna, J.; Dixon, A. Using Visual Ozone Damage Scores and Spectroscopy to Quantify Soybean Responses to Background Ozone. Remote Sens. 2020, 12, 93. https://doi.org/10.3390/rs12010093
Gosselin N, Sagan V, Maimaitiyiming M, Fishman J, Belina K, Podleski A, Maimaitijiang M, Bashir A, Balakrishna J, Dixon A. Using Visual Ozone Damage Scores and Spectroscopy to Quantify Soybean Responses to Background Ozone. Remote Sensing. 2020; 12(1):93. https://doi.org/10.3390/rs12010093
Chicago/Turabian StyleGosselin, Nichole, Vasit Sagan, Matthew Maimaitiyiming, Jack Fishman, Kelley Belina, Ann Podleski, Maitiniyazi Maimaitijiang, Anbreen Bashir, Jayashree Balakrishna, and Austin Dixon. 2020. "Using Visual Ozone Damage Scores and Spectroscopy to Quantify Soybean Responses to Background Ozone" Remote Sensing 12, no. 1: 93. https://doi.org/10.3390/rs12010093
APA StyleGosselin, N., Sagan, V., Maimaitiyiming, M., Fishman, J., Belina, K., Podleski, A., Maimaitijiang, M., Bashir, A., Balakrishna, J., & Dixon, A. (2020). Using Visual Ozone Damage Scores and Spectroscopy to Quantify Soybean Responses to Background Ozone. Remote Sensing, 12(1), 93. https://doi.org/10.3390/rs12010093









