Multispectral Approach for Identifying Invasive Plant Species Based on Flowering Phenology Characteristics
Abstract
1. Introduction
2. Materials and Methods
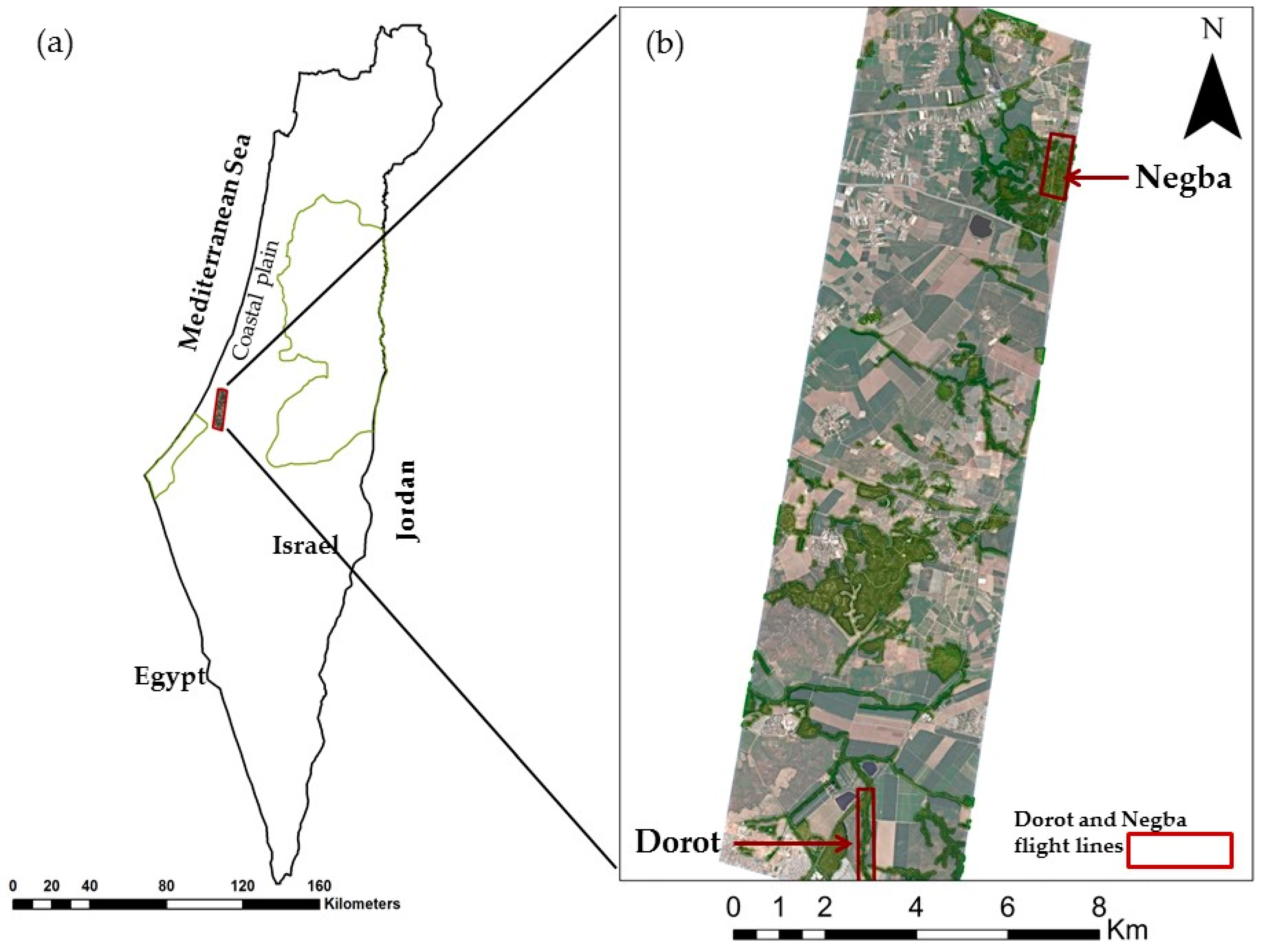
2.1. Study Area
2.2. Study Species
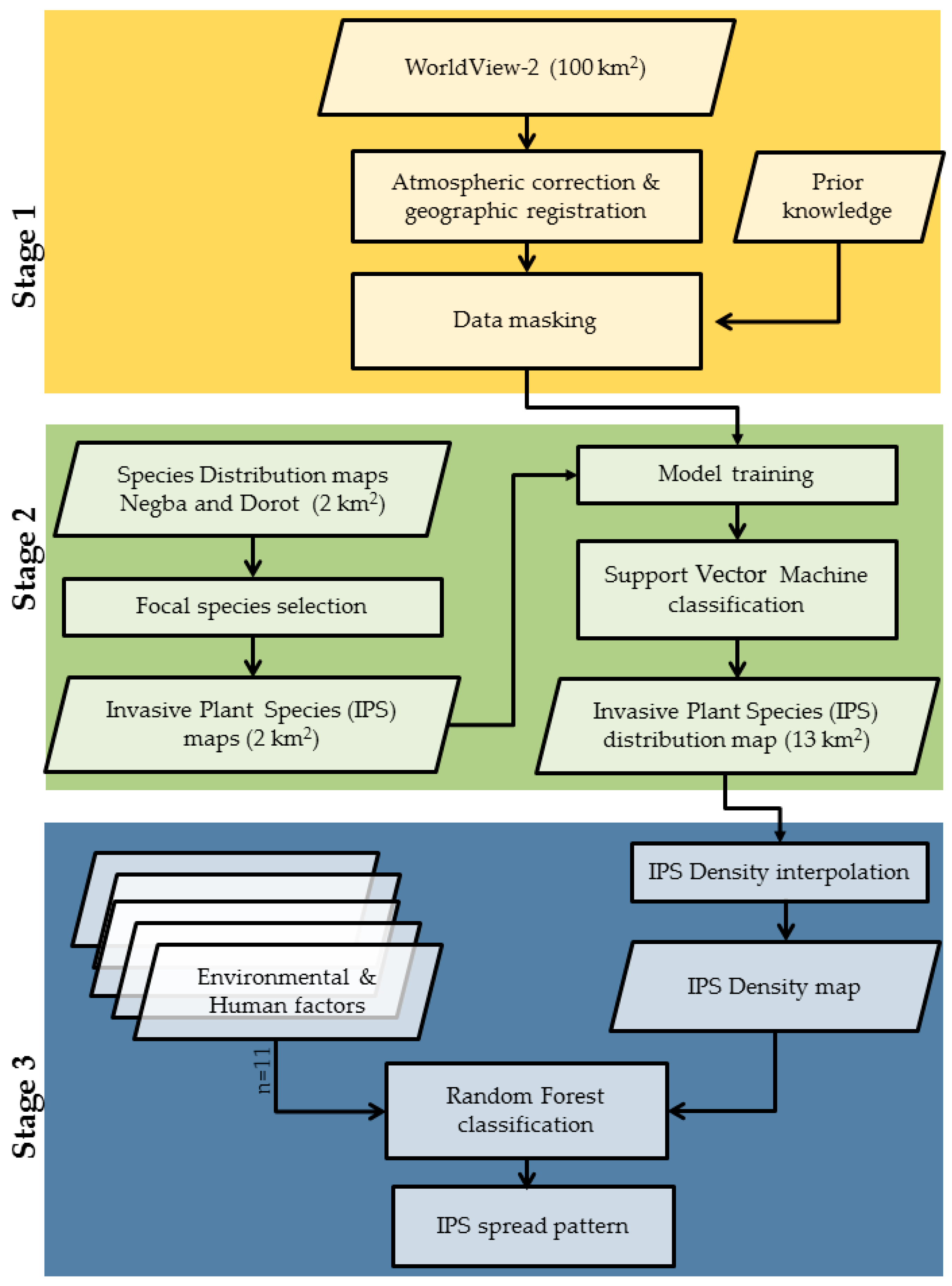
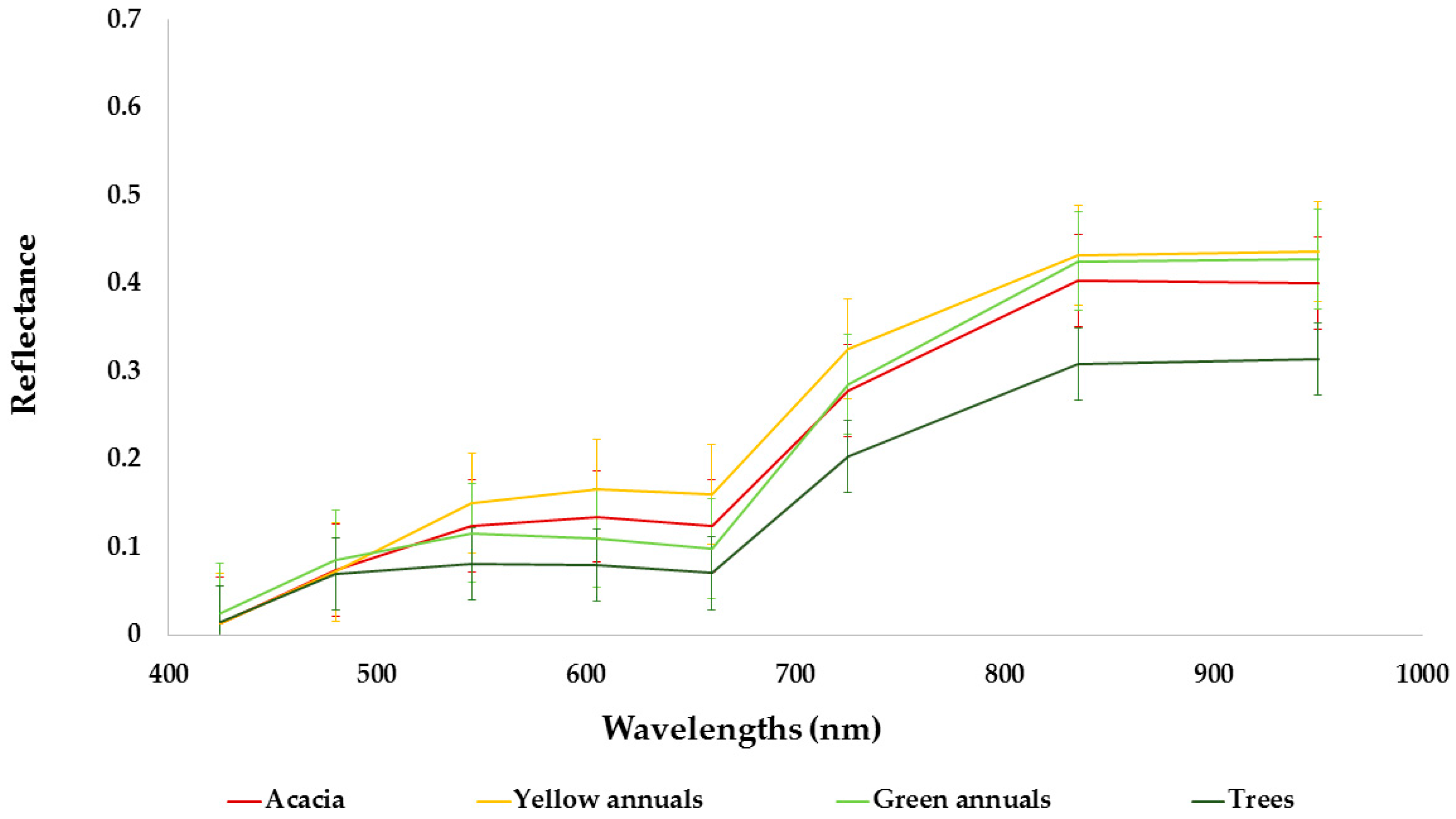
2.3. Remote Sensing Methodology
2.3.1. Potential Area for Invasion
2.3.2. Classification for Detecting Invasive Plant Species
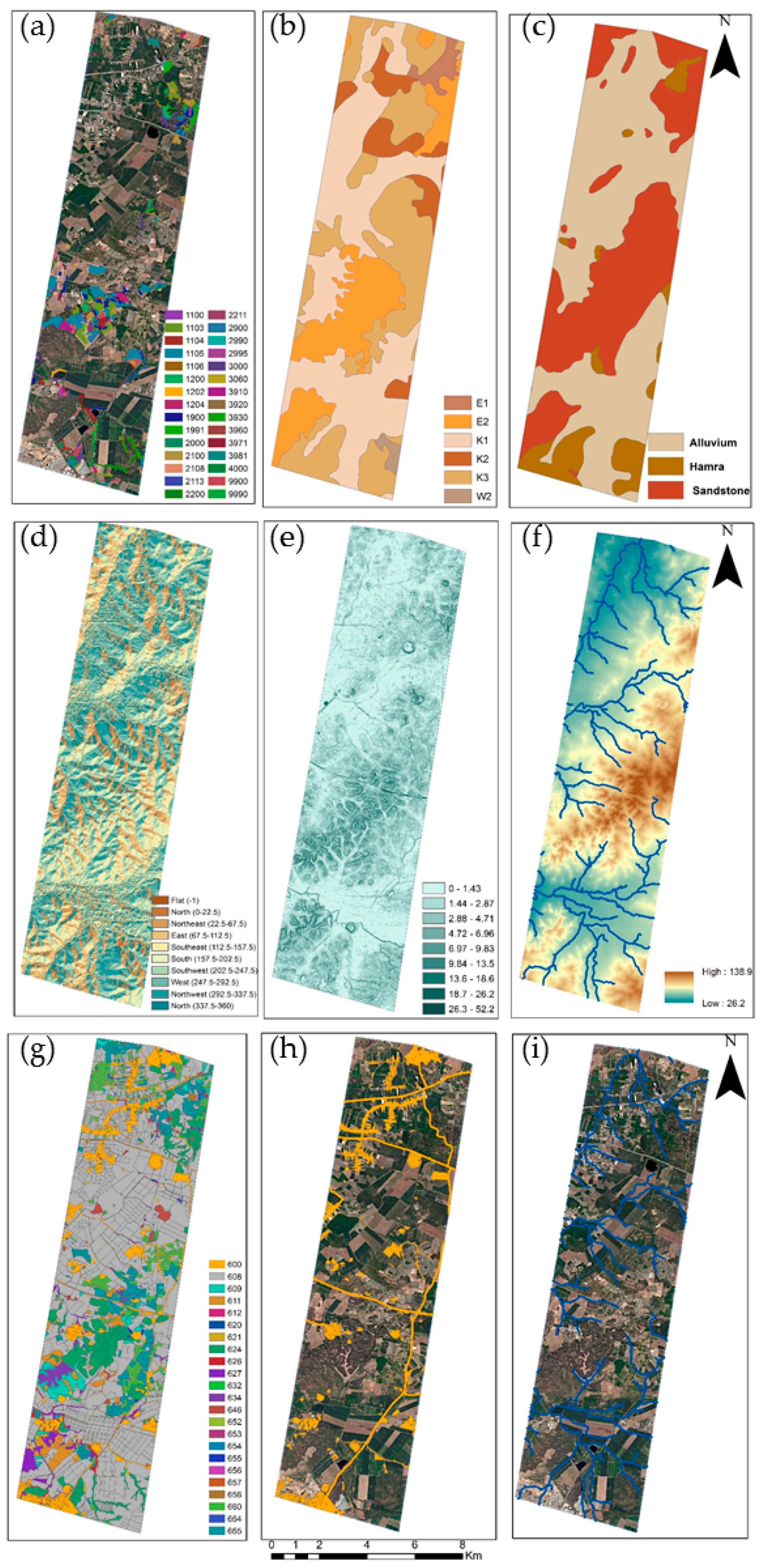
2.4. Environmental and Human Factors
3. Results
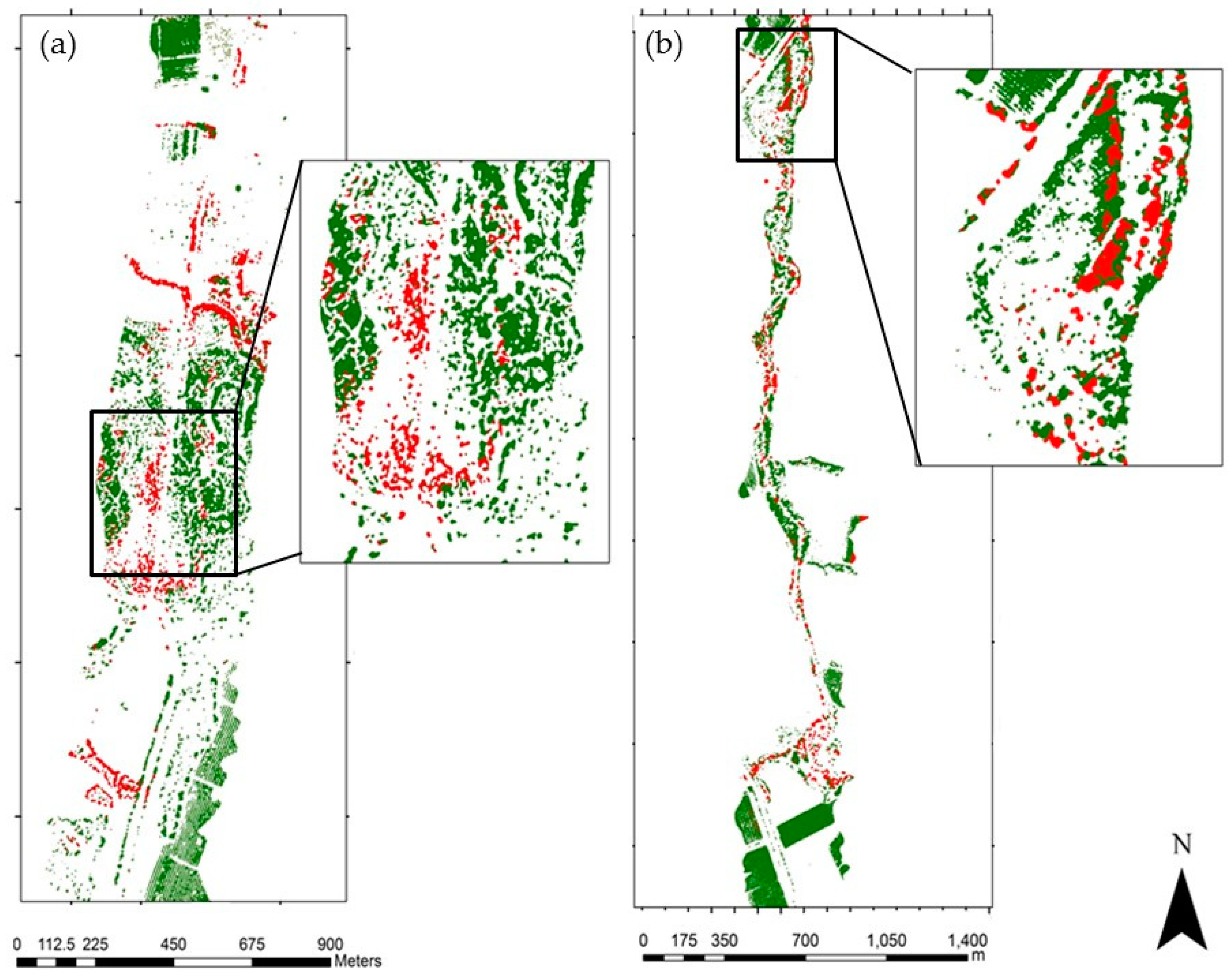
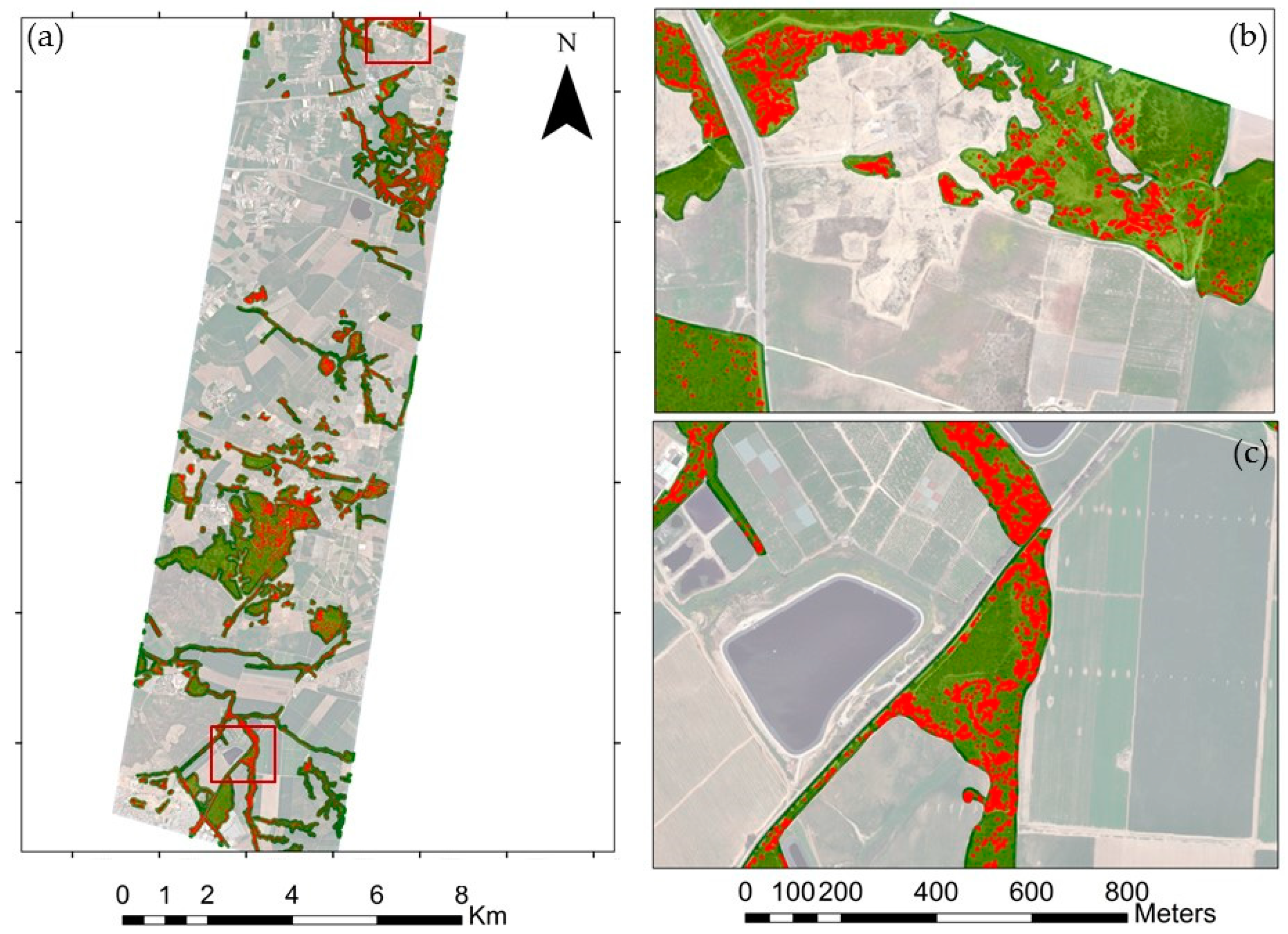
3.1. Species Classification Map
3.2. Invasive Plant Species Classification
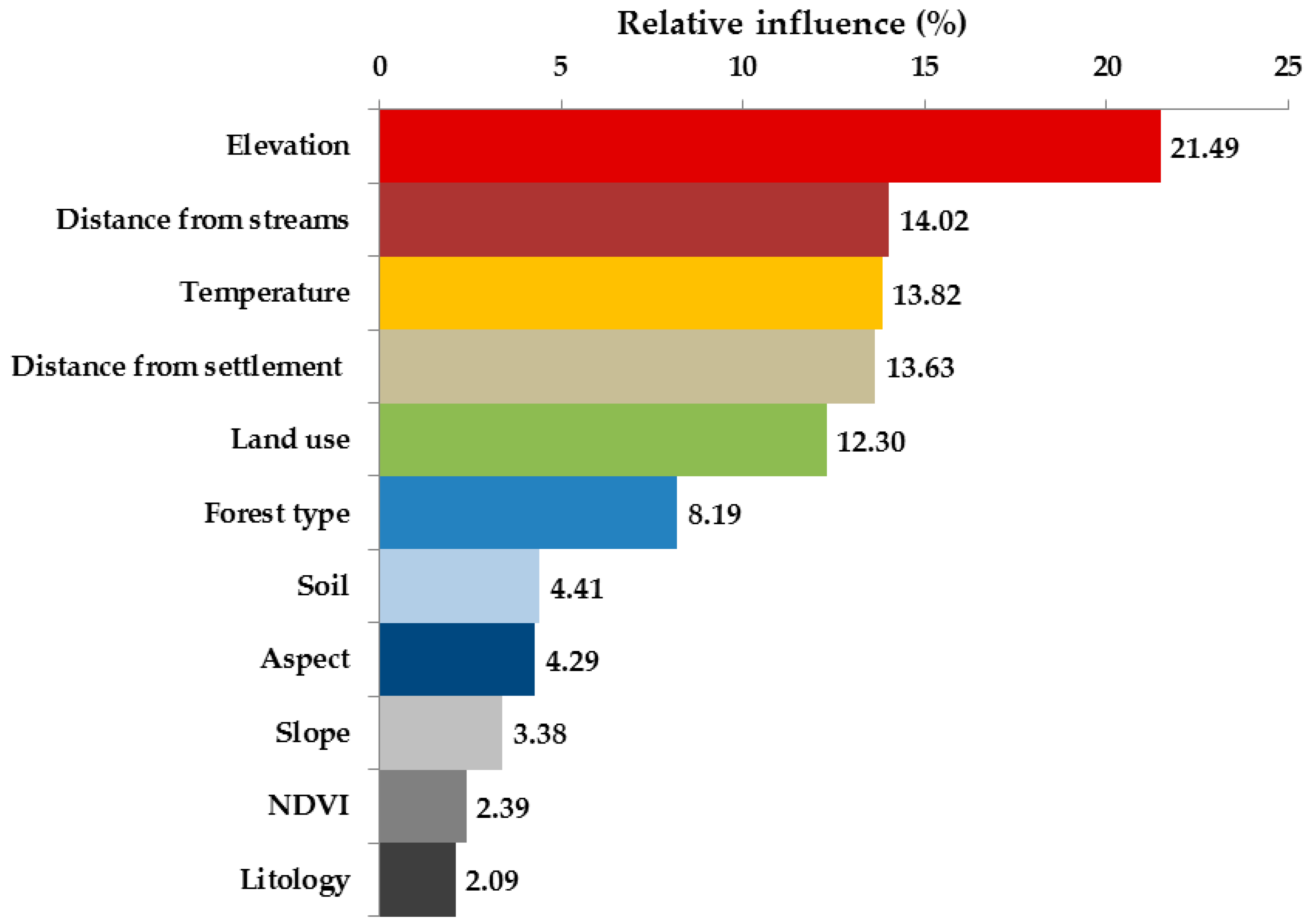
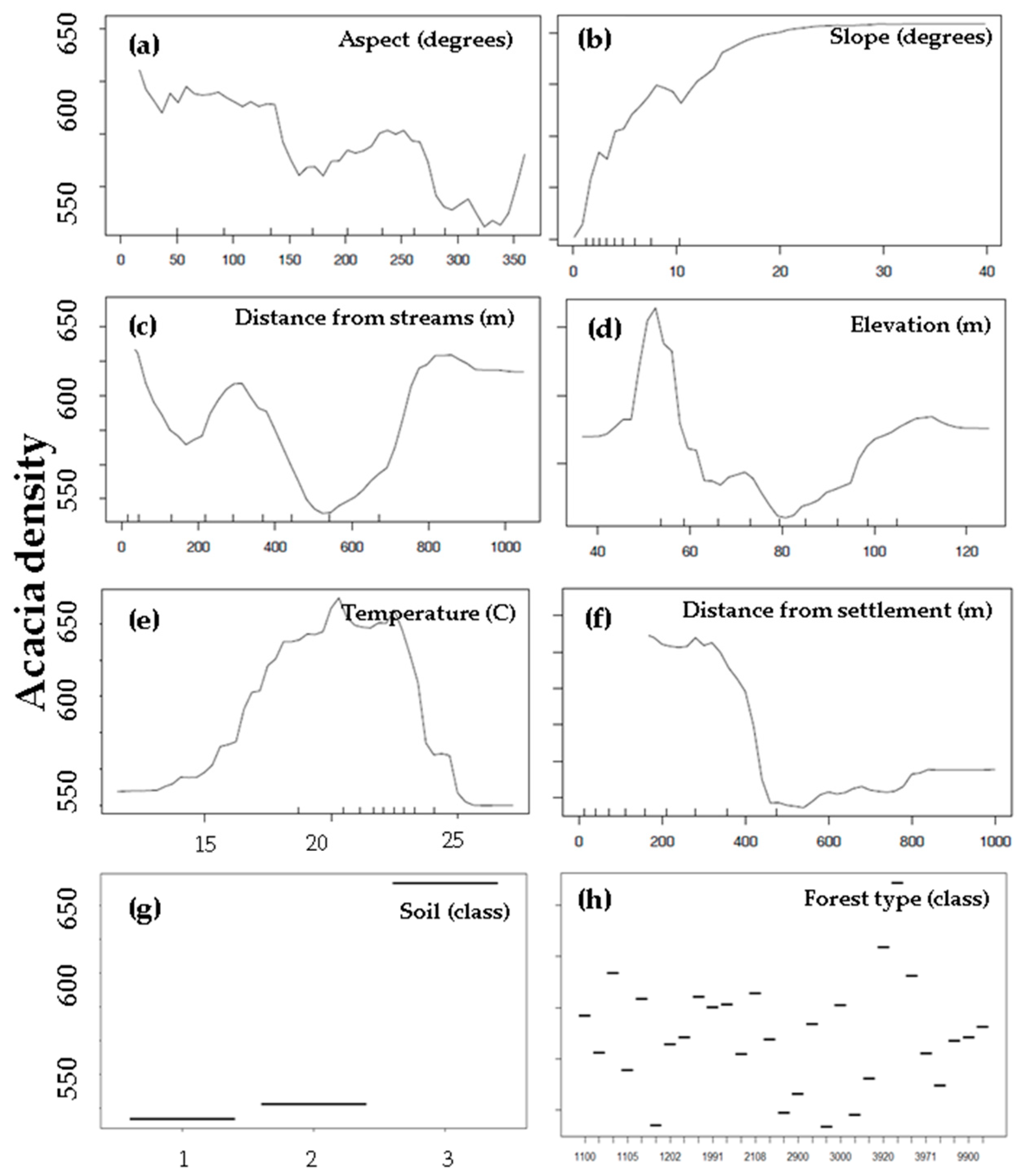
3.3. Environmental and Human Factors
4. Discussion
4.1. Integrated Hyper and Multi-Spectral Data
4.2. Environmental and Human Factors
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
Appendix A
| Elevation | Aspect | Slope | Distance from Settlement | Distance from Streams | NDVI | Temperature | Land-Use | Soil | Lithology | Forest Type | |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Elevation | 1.00 | ||||||||||
| Aspect | −0.01 | 1.00 | |||||||||
| Slope | 0.40 | −0.06 | 1.00 | ||||||||
| Distance from settlement | 0.20 | −0.03 | 0.09 | 1.00 | |||||||
| Distance from Streams | 0.60 | 0.10 | 0.23 | 0.09 | 1.00 | ||||||
| NDVI | −0.24 | 0.02 | −0.14 | −0.11 | −0.23 | 1.00 | |||||
| Temperature | 0.00 | −0.03 | −0.03 | 0.01 | 0.06 | −0.08 | 1.00 | ||||
| Land-use | 0.07 | −0.04 | 0.03 | 0.29 | 0.05 | −0.05 | −0.12 | 1.00 | |||
| Soil | 0.60 | 0.02 | 0.28 | 0.11 | 0.50 | −0.26 | −0.02 | 0.03 | 1.00 | ||
| Lithology | −0.07 | 0.01 | −0.11 | −0.14 | −0.04 | 0.08 | 0.07 | −0.07 | 0.01 | 1.00 | |
| Forest type | −0.18 | 0.05 | −0.03 | −0.20 | −0.19 | 0.07 | 0.01 | −0.24 | −0.10 | 0.12 | 1.00 |
| Pedology Class | Lithology Class | Forest Type | Land Use | ||||
|---|---|---|---|---|---|---|---|
| E1 | Alkaline soils | 1 | Clay, silt, sand, gravel (Alluvium soil) | 1103 | Pinus brutia | 600 | Settlements and roads |
| E2 | Pre-Randzina | 6 | Red silty sandstone loam | 1104 | Stone pine | 608 | Cultivated -agriculture |
| K1 | Dark brown sandy land | 5 | Sandstone | 1105 | Pinus halepensis | 609 | Abandoned cultivated area |
| K2 | Brown soils | 1106 | Pinus canariensis C. Smith | 611 | Stony area covered with vegetation | ||
| K3 | Ranzina | 1100 | Pinus | 612 | Sandy soils covered with vegetation | ||
| W3 | Sandy soils | 1200 | Cupressus | 620 | Forest | ||
| 1204 | Mediterranean cypress | 621 | Forest | ||||
| 1900 | Mixed coniferous forest | 624 | Planted Forest | ||||
| 1991 | Mixed coniferous forest | 626 | Grove | ||||
| 2000 | Broad-leafed trees | 627 | Shrubland | ||||
| 2100 | Eucalyptus | 632 | Inactive floodplain | ||||
| 2108 | Eucalyptus omphocephala | 634 | Badlands | ||||
| 2113 | Eucalyptus camaldulensis | 646 | Disturbed area | ||||
| 2200 | Acacia | 652 | Garden or park | ||||
| 2211 | Acacia saligna | 653 | Sports ground | ||||
| 2900 | Broad-leafed Mixed forest | 654 | Playground | ||||
| 2990 | Fruit trees | 655 | Open area | ||||
| 2995 | Ceratonia siliqua | 656 | Plastered surface | ||||
| 3000 | Flora Palaestina | 657 | Junkyard | ||||
| 3060 | Tamarisk | 658 | Garbage dump area | ||||
| 3910 | Natural Mediterranean forest | 660 | Orchards | ||||
| 3920 | Natural Mediterranean forest | 664 | Grapevine | ||||
| 3930 | Natural Mediterranean forest | 665 | Citrus orchard | ||||
| 3960 | Natural Mediterranean forest | ||||||
| 3971 | Natural Mediterranean forest | ||||||
| 3981 | Natural Mediterranean forest | ||||||
| 4000 | Natural Mediterranean forest | ||||||
| 9900 | open landscape | ||||||
| 9990 | open landscape | ||||||
References
- Pimentel, D.; McNair, S.; Janecka, J.; Wightman, J.; Simmonds, C.; O’connell, C.; Wong, E.; Russel, L.; Zern, J.; Aquino, T. Economic and environmental threats of alien plant, animal, and microbe invasions. Agric. Ecosyst. Environ. 2001, 84, 1–20. [Google Scholar] [CrossRef]
- Hobbs, R.J. Invasive Species in a Changing World; Island Press: Washington, DC, USA; Covelo, CA, USA, 2000. [Google Scholar]
- Vilà, M.; Espinar, J.L.; Hejda, M.; Hulme, P.E.; Jarošík, V.; Maron, J.L.; Pergl, J.; Schaffner, U.; Sun, Y.; Pyšek, P. Ecological impacts of invasive alien plants: A meta-analysis of their effects on species, communities and ecosystems. Ecol. Lett. 2011, 14, 702–708. [Google Scholar] [CrossRef] [PubMed]
- Bartz, R.; Kowarik, I. Assessing the environmental impacts of invasive alien plants: A review of assessment approaches. NeoBiota 2019, 43, 69. [Google Scholar] [CrossRef]
- MacDougall, A.S.; Turkington, R. Are invasive species the drivers or passengers of change in degraded ecosystems? Ecology 2005, 86, 42–55. [Google Scholar] [CrossRef]
- Theoharides, K.A.; Dukes, J.S. Plant invasion across space and time: Factors affecting nonindigenous species success during four stages of invasion. New Phytol. 2007, 176, 256–273. [Google Scholar] [CrossRef]
- Gallien, L.; Münkemüller, T.; Albert, C.H.; Boulangeat, I.; Thuiller, W. Predicting potential distributions of invasive species: Where to go from here? Divers. Distrib. 2010, 16, 331–342. [Google Scholar] [CrossRef]
- With, K.A. The landscape ecology of invasive spread. Conserv. Biol. 2002, 16, 1192–1203. [Google Scholar] [CrossRef]
- Mata, T.; Haddad, N.; Holyoak, M. How invader traits interact with resident communities and resource availability to determine invasion success. Oikos 2013, 122, 149–160. [Google Scholar] [CrossRef]
- DeGasperis, B.G.; Motzkin, G. Windows of opportunity: Historical and ecological controls on berberis thunbergii invasions. Ecology 2007, 88, 3115–3125. [Google Scholar] [CrossRef]
- Alpert, P.; Bone, E.; Holzapfel, C. Invasiveness, invasibility and the role of environmental stress in the spread of non-native plants. Perspect. Plant Ecol. Evol. Syst. 2000, 3, 52–66. [Google Scholar] [CrossRef]
- Barbosa, J.M.; Asner, G.P.; Hughes, R.F.; Johnson, M.T. Landscape-scale gpp and carbon density inform patterns and impacts of an invasive tree across wet forests of hawaii. Ecol. Appl. 2017, 27, 403–415. [Google Scholar] [CrossRef]
- Simberloff, D.; Martin, J.-L.; Genovesi, P.; Maris, V.; Wardle, D.A.; Aronson, J.; Courchamp, F.; Galil, B.; García-Berthou, E.; Pascal, M.; et al. Impacts of biological invasions: What’s what and the way forward. Trends Ecol. Evol. 2013, 28, 58–66. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.-Y.; Asner, G.P. Applications of remote sensing to alien invasive plant studies. Sensors 2009, 9, 4869–4889. [Google Scholar] [CrossRef]
- Niphadkar, M.; Nagendra, H. Remote sensing of invasive plants: Incorporating functional traits into the picture. Int. J. Remote Sens. 2016, 37, 3074–3085. [Google Scholar] [CrossRef]
- Rocchini, D.; Andreo, V.; Förster, M.; Garzon-Lopez, C.X.; Gutierrez, A.P.; Gillespie, T.W.; Hauffe, H.C.; He, K.S.; Kleinschmit, B.; Mairota, P.; et al. Potential of remote sensing to predict species invasions: A modelling perspective. Prog. Phys. Geogr. 2015, 39, 283–309. [Google Scholar] [CrossRef]
- Skowronek, S.; Ewald, M.; Isermann, M.; Van De Kerchove, R.; Lenoir, J.; Aerts, R.; Warrie, J.; Hattab, T.; Honnay, O.; Schmidtlein, S.; et al. Mapping an invasive bryophyte species using hyperspectral remote sensing data. Biol. Invasions 2017, 19, 239–254. [Google Scholar] [CrossRef]
- Thenkabail, P.S.; Lyon, J.G. Hyperspectral Remote Sensing of Vegetation; CRC Press: London, NY, USA, 2016. [Google Scholar]
- Ustin, S.L.; Gamon, J.A. Remote sensing of plant functional types. New Phytol. 2010, 186, 795–816. [Google Scholar] [PubMed]
- Yelenik, S.G.; DiManno, N.; D’Antonio, C.M. Evaluating nurse plants for restoring native woody species to degraded subtropical woodlands. Ecol. Evol. 2015, 5, 300–313. [Google Scholar] [CrossRef] [PubMed]
- Féret, J.-B.; Asner, G.P. Mapping tropical forest canopy diversity using high-fidelity imaging spectroscopy. Ecol. Appl. 2014, 24, 1289–1296. [Google Scholar] [CrossRef] [PubMed]
- Somers, B.; Asner, G.P. Tree species mapping in tropical forests using multi-temporal imaging spectroscopy: Wavelength adaptive spectral mixture analysis. Int. J. Appl. Earth Observ. Geoinf. 2014, 31, 57–66. [Google Scholar] [CrossRef]
- Ustin, S.L.; Roberts, D.A.; Gamon, J.A.; Asner, G.P.; Green, R.O. Using imaging spectroscopy to study ecosystem processes and properties. BioScience 2004, 54, 523–534. [Google Scholar] [CrossRef]
- He, K.S.; Rocchini, D.; Neteler, M.; Nagendra, H. Benefits of hyperspectral remote sensing for tracking plant invasions. Divers. Distrib. 2011, 17, 381–392. [Google Scholar] [CrossRef]
- Narumalani, S.; Mishra, D.R.; Wilson, R.; Reece, P.; Kohler, A. Detecting and mapping four invasive species along the floodplain of north platte river, nebraska. Weed Technol. 2009, 23, 99–107. [Google Scholar] [CrossRef]
- Weil, G.; Lensky, I.M.; Resheff, Y.S.; Levin, N. Optimizing the timing of unmanned aerial vehicle image acquisition for applied mapping of woody vegetation species using feature selection. Remote Sens. 2017, 9, 1130. [Google Scholar] [CrossRef]
- Adam, E.; Mutanga, O.; Rugege, D. Multispectral and hyperspectral remote sensing for identification and mapping of wetland vegetation: A review. Wetl. Ecol. Manag. 2010, 18, 281–296. [Google Scholar] [CrossRef]
- Dewey, S.A.; Price, K.P.; Ramsey, D. Satellite remote sensing to predict potential distribution of dyers woad (isatis tinctoria). Weed Technol. 1991, 5, 479–484. [Google Scholar] [CrossRef]
- Lawrence, R.L.; Wood, S.D.; Sheley, R.L. Mapping invasive plants using hyperspectral imagery and breiman cutler classifications (randomforest). Remote Sens. Environ. 2006, 100, 356–362. [Google Scholar] [CrossRef]
- Fernandes, M.R.; Aguiar, F.C.; Silva, J.M.; Ferreira, M.T.; Pereira, J.M. Optimal attributes for the object based detection of giant reed in riparian habitats: A comparative study between airborne high spatial resolution and worldview-2 imagery. Int. J. Appl. Earth Observ. Geoinf. 2014, 32, 79–91. [Google Scholar] [CrossRef]
- Everitt, J.H.; Anderson, G.L.; Escobar, D.E.; Davis, M.R.; Spencer, N.R.; Andrascik, R.J. Use of remote sensing for detecting and mapping leafy spurge (euphorbia esula). Weed Technol. 1995, 9, 599–609. [Google Scholar] [CrossRef]
- Andrew, M.E.; Ustin, S.L. The role of environmental context in mapping invasive plants with hyperspectral image data. Remote Sens. Environ. 2008, 112, 4301–4317. [Google Scholar] [CrossRef]
- Andrew, M.E.; Ustin, S.L. Habitat suitability modelling of an invasive plant with advanced remote sensing data. Divers. Distrib. 2009, 15, 627–640. [Google Scholar] [CrossRef]
- Belluco, E.; Camuffo, M.; Ferrari, S.; Modenese, L.; Silvestri, S.; Marani, A.; Marani, M. Mapping salt-marsh vegetation by multispectral and hyperspectral remote sensing. Remote Sens. Environ. 2006, 105, 54–67. [Google Scholar] [CrossRef]
- Paz-Kagan, T.; Caras, T.; Herrmann, I.; Shachak, M.; Karnieli, A. Multiscale mapping of species diversity under changed land use using imaging spectroscopy. Ecol. Appl. 2017, 27, 1466–1484. [Google Scholar] [CrossRef] [PubMed]
- Rozenstein, O.; Karnieli, A. Comparison of methods for land-use classification incorporating remote sensing and gis inputs. Appl. Geogr. 2011, 31, 533–544. [Google Scholar] [CrossRef]
- Mussery, A.; Leu, S.; Lensky, I.; Budovsky, A. The effect of planting techniques on arid ecosystems in the northern negev. Arid Land Res. Manag. 2013, 27, 90–100. [Google Scholar] [CrossRef]
- Bar, P.; Cohen, O.; Shoshany, M. Invasion rate of the alien species acacia saligna within coastal sand dune habitats in israel. Israel J. Plant Sci. 2004, 52, 115–124. [Google Scholar]
- Thompson, G.D.; Bellstedt, D.U.; Richardson, D.M.; Wilson, J.R.; Le Roux, J.J. A tree well travelled: Global genetic structure of the invasive tree acacia saligna. J. Biogeogr. 2015, 42, 305–314. [Google Scholar] [CrossRef]
- Witkowski, E. Growth and competition between seedlings of protea repens (l.) l. And the alien invasive, acacia saligna (labill.) wendl. In relation to nutrient availability. Funct. Ecol. 1991, 5, 101–110. [Google Scholar] [CrossRef]
- Richter, R.; Schläpfer, D. Atmospheric/Topographic Correction for Satellite Imagery; DLR Report DLR-IB; ATCOR 2/3 User Gide, Version 9.3; DLR—German Aerospace Center: Wessling, Germany; ReSe Applications: Wil, Switzerland, 2005. [Google Scholar]
- Lu, D.; Weng, Q. A survey of image classification methods and techniques for improving classification performance. Int. J. Remote Sens. 2007, 28, 823–870. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Core Team: Vienna, Austria, 2018. [Google Scholar]
- Paz-Kagan, T.; Vaughn, N.R.; Martin, R.E.; Brodrick, P.G.; Stephenson, N.L.; Das, A.J.; Nydick, K.R.; Asner, G.P. Landscape-scale variation in canopy water content of giant sequoias during drought. For. Ecol. Manag. 2017, 419, 291–304. [Google Scholar] [CrossRef]
- Evans, J.S.; Murphy, M.A.; Holden, Z.A.; Cushman, S.A. Modeling species distribution and change using random forest. In Predictive Species and Habitat Modeling in Landscape Ecology; Springer: London, NY, USA, 2011; pp. 139–159. [Google Scholar]
- Maidment, D.R.; Morehouse, S. Arc Hydro: Gis for Water Resources; ESRI, Inc.: Redland, CA, USA, 2002; Volume 1. [Google Scholar]
- Zuur, A.F.; Ieno, E.N.; Elphick, C.S. A protocol for data exploration to avoid common statistical problems. Methods Ecol. Evol. 2010, 1, 3–14. [Google Scholar] [CrossRef]
- Ai, F.-F.; Bin, J.; Zhang, Z.-M.; Huang, J.-H.; Wang, J.-B.; Liang, Y.-Z.; Yu, L.; Yang, Z.-Y. Application of random forests to select premium quality vegetable oils by their fatty acid composition. Food Chem. 2014, 143, 472–478. [Google Scholar] [CrossRef] [PubMed]
- Biau, G.; Devroye, L.; Lugosi, G. Consistency of random forests and other averaging classifiers. J. Mach. Learn. Res. 2008, 9, 2015–2033. [Google Scholar]
- Breiman, L. Random forests. Mach. Learn. 2001, 45, 5–32. [Google Scholar] [CrossRef]
- Friedman, J.H. Greedy function approximation: A gradient boosting machine. Ann. Stat. 2001, 29, 1189–1232. [Google Scholar] [CrossRef]
- Cutler, D.R.; Edwards, T.C., Jr.; Beard, K.H.; Cutler, A.; Hess, K.T.; Gibson, J.; Lawler, J.J. Random forests for classification in ecology. Ecology 2017, 88, 2783–2792. [Google Scholar] [CrossRef]
- Paz-Kagan, T.; Brodrick, P.G.; Vaughn, N.R.; Das, A.J.; Stephenson, N.L.; Nydick, K.R.; Asner, G.P. What mediates tree mortality during drought in the southern sierra nevada? Ecol. Appl. 2017, 27, 2443–2457. [Google Scholar] [CrossRef]
- Pu, R.; Landry, S. A comparative analysis of high spatial resolution ikonos and worldview-2 imagery for mapping urban tree species. Remote Sens. Environ. 2012, 124, 516–533. [Google Scholar] [CrossRef]
- Richardson, D.M.; Kluge, R.L. Seed banks of invasive australian acacia species in south africa: Role in invasiveness and options for management. Perspect. Plant Ecol. Evol. Syst. 2008, 10, 161–177. [Google Scholar] [CrossRef]
- Brooks, W.R.; Jordan, R.C. Propagule pressure and native species richness effects drive invasibility in tropical dry forest seedling layers. Perspect. Plant Ecol., Evol. Syst. 2013, 15, 162–170. [Google Scholar] [CrossRef]
- Barbosa, J.M.; Asner, G.P.; Martin, R.E.; Baldeck, C.A.; Hughes, F.; Johnson, T. Determining subcanopy psidium cattleianum invasion in hawaiian forests using imaging spectroscopy. Remote Sens. 2016, 8, 33. [Google Scholar] [CrossRef]
- Morais, M.; Freitas, H. Phenological dynamics of the invasive plant acacia longifolia in portugal. Weed Res. 2015, 55, 555–564. [Google Scholar] [CrossRef]
- Buermann, W.; Saatchi, S.; Smith, T.B.; Zutta, B.R.; Chaves, J.A.; Milá, B.; Graham, C.H. Predicting species distributions across the amazonian and andean regions using remote sensing data. J. Biogeogr. 2008, 35, 1160–1176. [Google Scholar] [CrossRef]
- Bobrowski, M.; Bechtel, B.; Böhner, J.; Oldeland, J.; Weidinger, J.; Schickhoff, U. Application of thermal and phenological land surface parameters for improving ecological niche models of betula utilis in the himalayan region. Remote Sens. 2018, 10, 814. [Google Scholar] [CrossRef]
- Yelenik, S.; Stock, W.; Richardson, D. Ecosystem level impacts of invasive acacia saligna in the south african fynbos. Restor. Ecol. 2004, 12, 44–51. [Google Scholar] [CrossRef]
- Hastings, A.; Cuddington, K.; Davies, K.F.; Dugaw, C.J.; Elmendorf, S.; Freestone, A.; Harrison, S.; Holland, M.; Lambrinos, J.; Malvadkar, U.; et al. The spatial spread of invasions: New developments in theory and evidence. Ecol. Lett. 2005, 8, 91–101. [Google Scholar] [CrossRef]
- Walker, L.R. Ecosystems of Disturbed Ground; Elsevier: Amsterdam, The Netherlands, 1999; Volume 16. [Google Scholar]
- Trombulak, S.C.; Frissell, C.A. Review of ecological effects of roads on terrestrial and aquatic communities. Conserv. Biol. 2000, 14, 18–30. [Google Scholar] [CrossRef]
- Harris, L.D. Edge effects and conservation of biotic diversity. Conserv. Biol. 1988, 2, 330–332. [Google Scholar] [CrossRef]
- Cerboncini, R.A.S.; Roper, J.J.; Passos, F.C. Edge effects without habitat fragmentation? Small mammals and a railway in the atlantic forest of southern brazil. Oryx 2016, 50, 460–467. [Google Scholar] [CrossRef]
- Holway, D.A. Edge effects of an invasive species across a natural ecological boundary. Biol. Conserv. 2005, 121, 561–567. [Google Scholar] [CrossRef]
- Pardini, R. Effects of forest fragmentation on small mammals in an atlantic forest landscape. Biodivers. Conserv. 2004, 13, 2567–2586. [Google Scholar] [CrossRef]
- Le Maitre, D.C.; Gaertner, M.; Marchante, E.; Ens, E.J.; Holmes, P.M.; Pauchard, A.; O’Farrell, P.J.; Rogers, A.M.; Blanchard, R.; Blignaut, J.; et al. Impacts of invasive australian acacias: Implications for management and restoration. Divers. Distrib. 2011, 17, 1015–1029. [Google Scholar] [CrossRef]

| Categories Enveromantal | Factors | Units |
|---|---|---|
| Topography | Elevation | m |
| Slope | degree | |
| Aspect | degree | |
| Distance from rivers or streams | m | |
| Substrate | Pedology | Class |
| Lithology | Class | |
| Land Surface | Temperature | °C |
| Productivity | NDVI | unitless |
| Categories/human | ||
| Forest structure | Forest type | Class |
| Land-use | Land-use | Class |
| Distance from roads and settlements | m |
| Classes | Acacia | Annual Green | Dark Soil | Bright Soil | Trees | Yellow Annuals | Total Pixels |
|---|---|---|---|---|---|---|---|
| Acacia | 548 | 5 | 0 | 1 | 1 | 23 | 578 |
| Green annual | 15 | 375 | 1 | 3 | 2 | 2 | 398 |
| Dark soil | 3 | 0 | 300 | 32 | 2 | 0 | 337 |
| Bright soil | 1 | 0 | 24 | 145 | 0 | 2 | 172 |
| Trees | 6 | 1 | 0 | 0 | 995 | 0 | 1002 |
| Yellow annuals | 12 | 0 | 0 | 6 | 0 | 79 | 97 |
| Total pixels | 585 | 381 | 325 | 187 | 1000 | 106 | 2584 |
| Total accuracy | 93.68 | 95.91 | 92.02 | 77.54 | 98.71 | 74.53 | 89.32 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Paz-Kagan, T.; Silver, M.; Panov, N.; Karnieli, A. Multispectral Approach for Identifying Invasive Plant Species Based on Flowering Phenology Characteristics. Remote Sens. 2019, 11, 953. https://doi.org/10.3390/rs11080953
Paz-Kagan T, Silver M, Panov N, Karnieli A. Multispectral Approach for Identifying Invasive Plant Species Based on Flowering Phenology Characteristics. Remote Sensing. 2019; 11(8):953. https://doi.org/10.3390/rs11080953
Chicago/Turabian StylePaz-Kagan, Tarin, Micha Silver, Natalya Panov, and Arnon Karnieli. 2019. "Multispectral Approach for Identifying Invasive Plant Species Based on Flowering Phenology Characteristics" Remote Sensing 11, no. 8: 953. https://doi.org/10.3390/rs11080953
APA StylePaz-Kagan, T., Silver, M., Panov, N., & Karnieli, A. (2019). Multispectral Approach for Identifying Invasive Plant Species Based on Flowering Phenology Characteristics. Remote Sensing, 11(8), 953. https://doi.org/10.3390/rs11080953




