Assessing the Feasibility of Using Sentinel-2 Imagery to Quantify the Impact of Heatwaves on Irrigated Vineyards
Abstract
1. Introduction
2. Materials and Methods
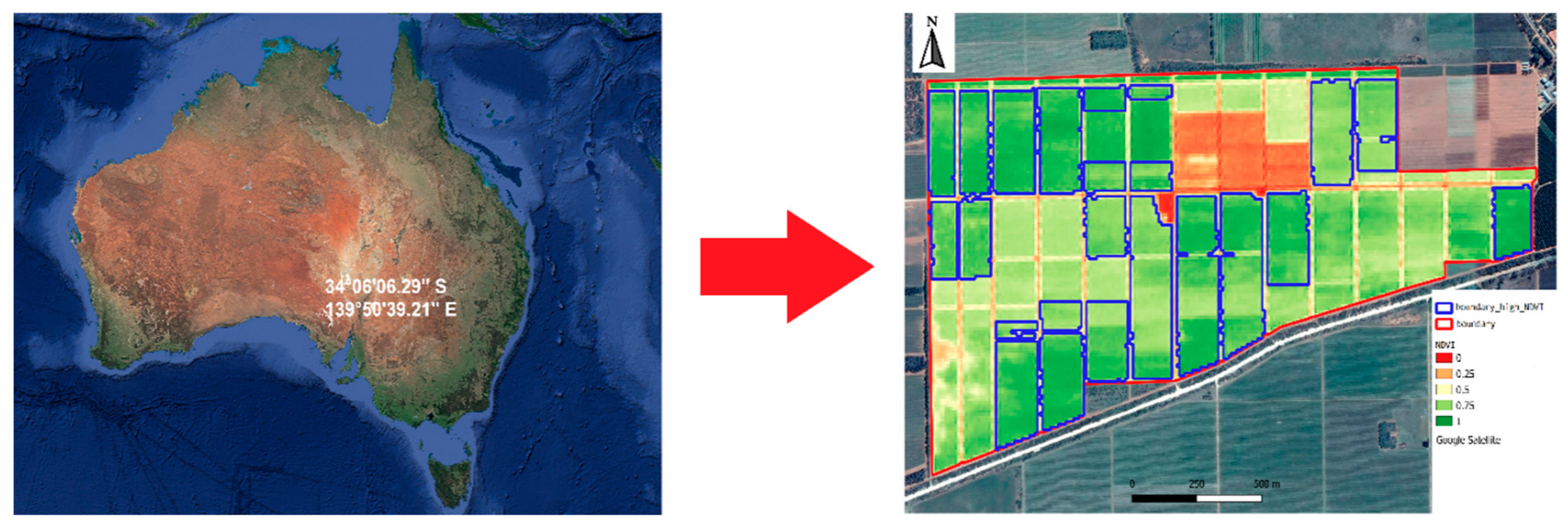
2.1. Study Area
2.2. Meteorological Data
2.3. Data Acquisition and Processing
2.4. Spectral Bands and Vegetation Indices
2.5. Analysis of Remote Sensing Data
2.6. Statistical Analysis
3. Results
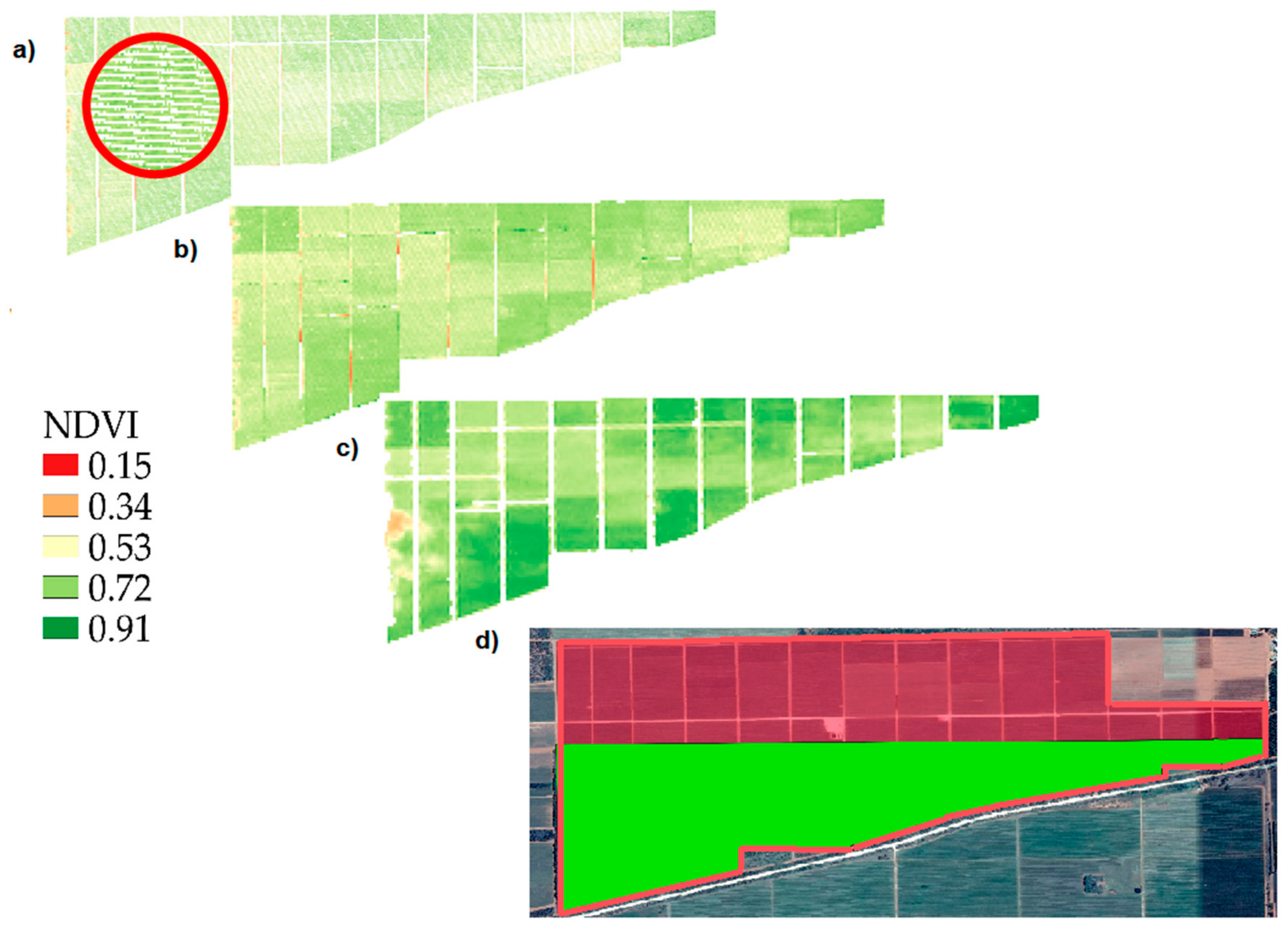
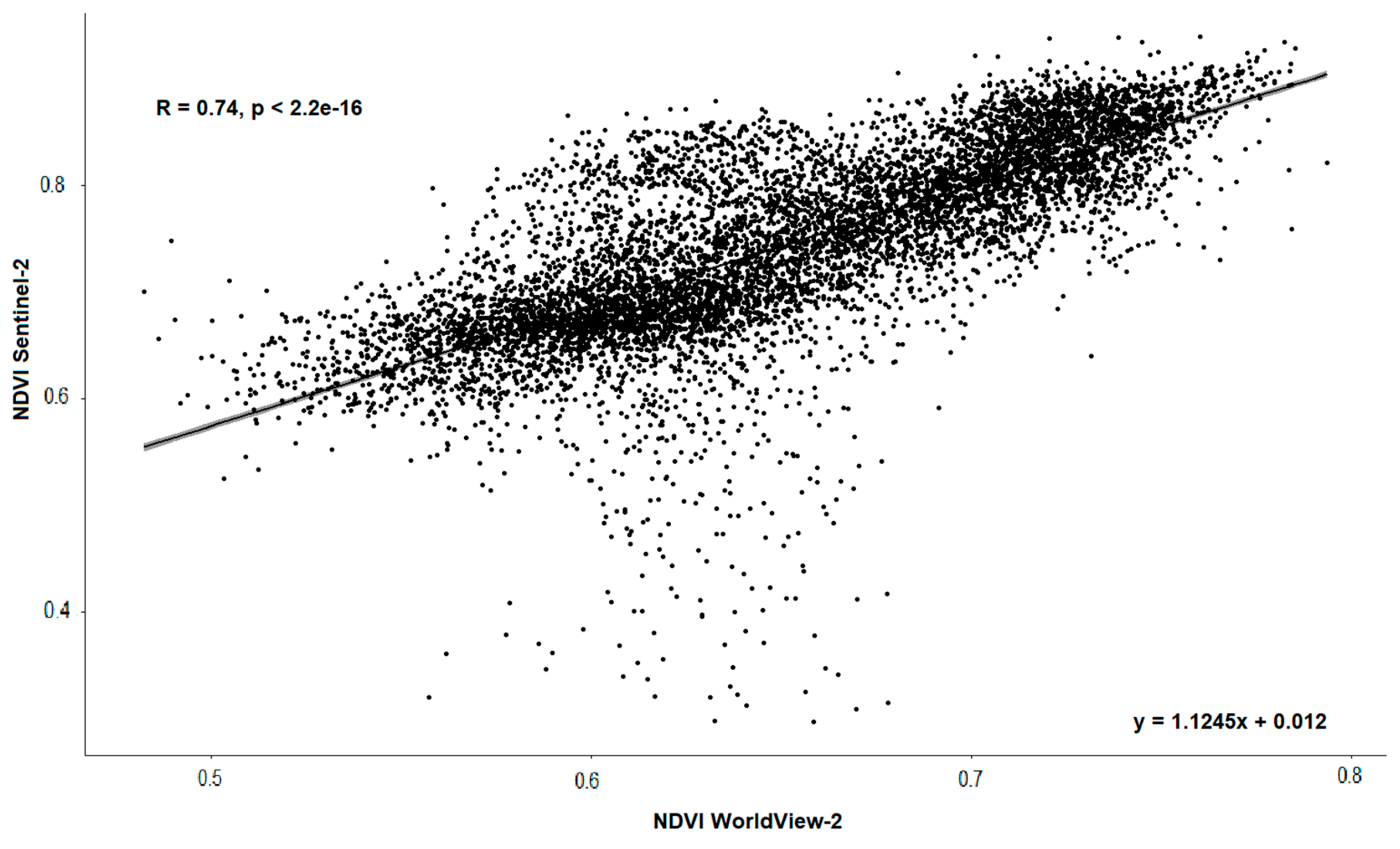
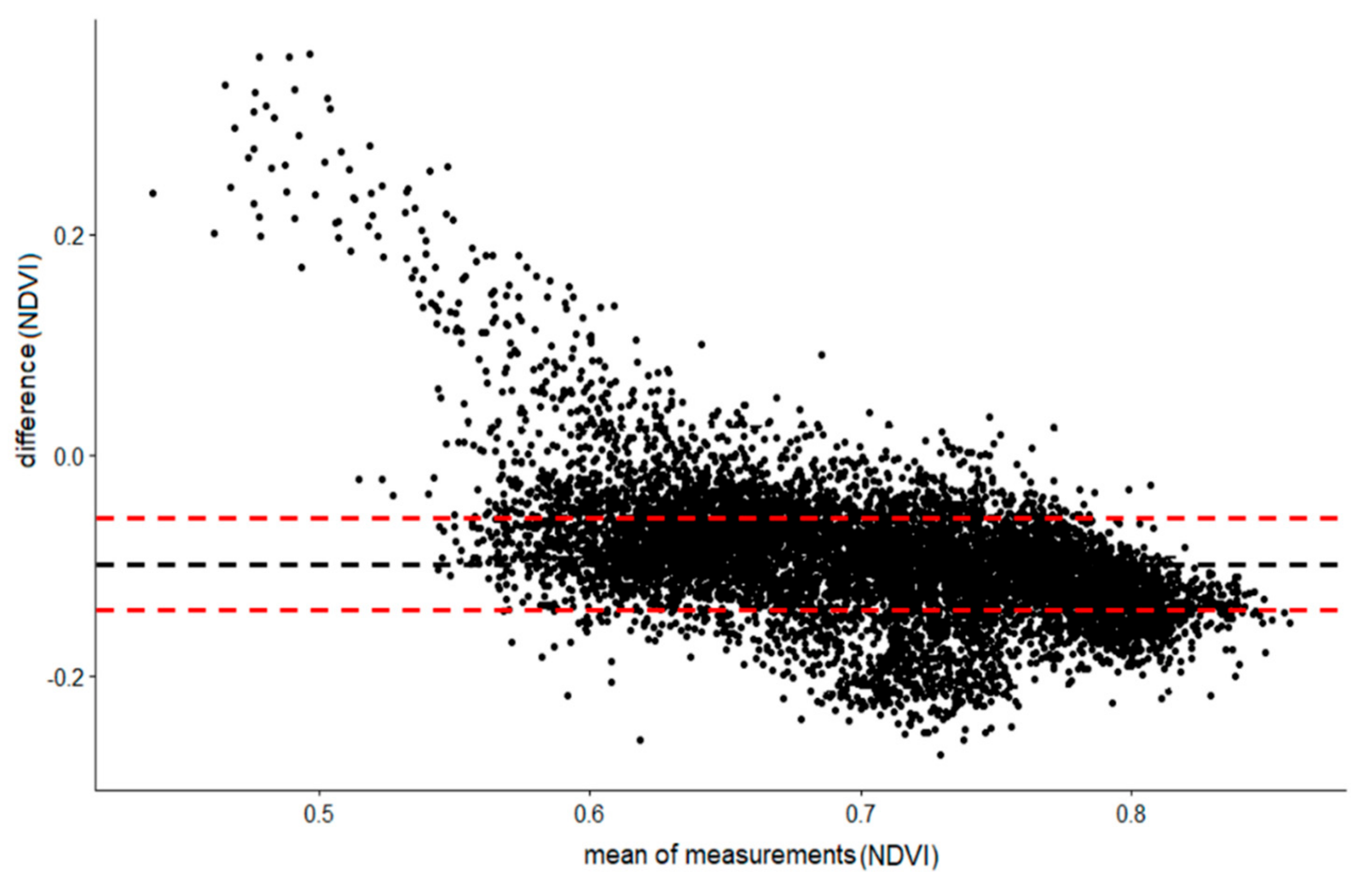
3.1. Comparison between Sentinel-2 and WorldView-2 Images
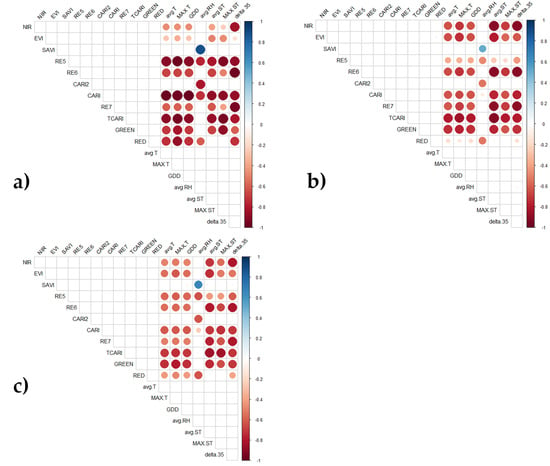
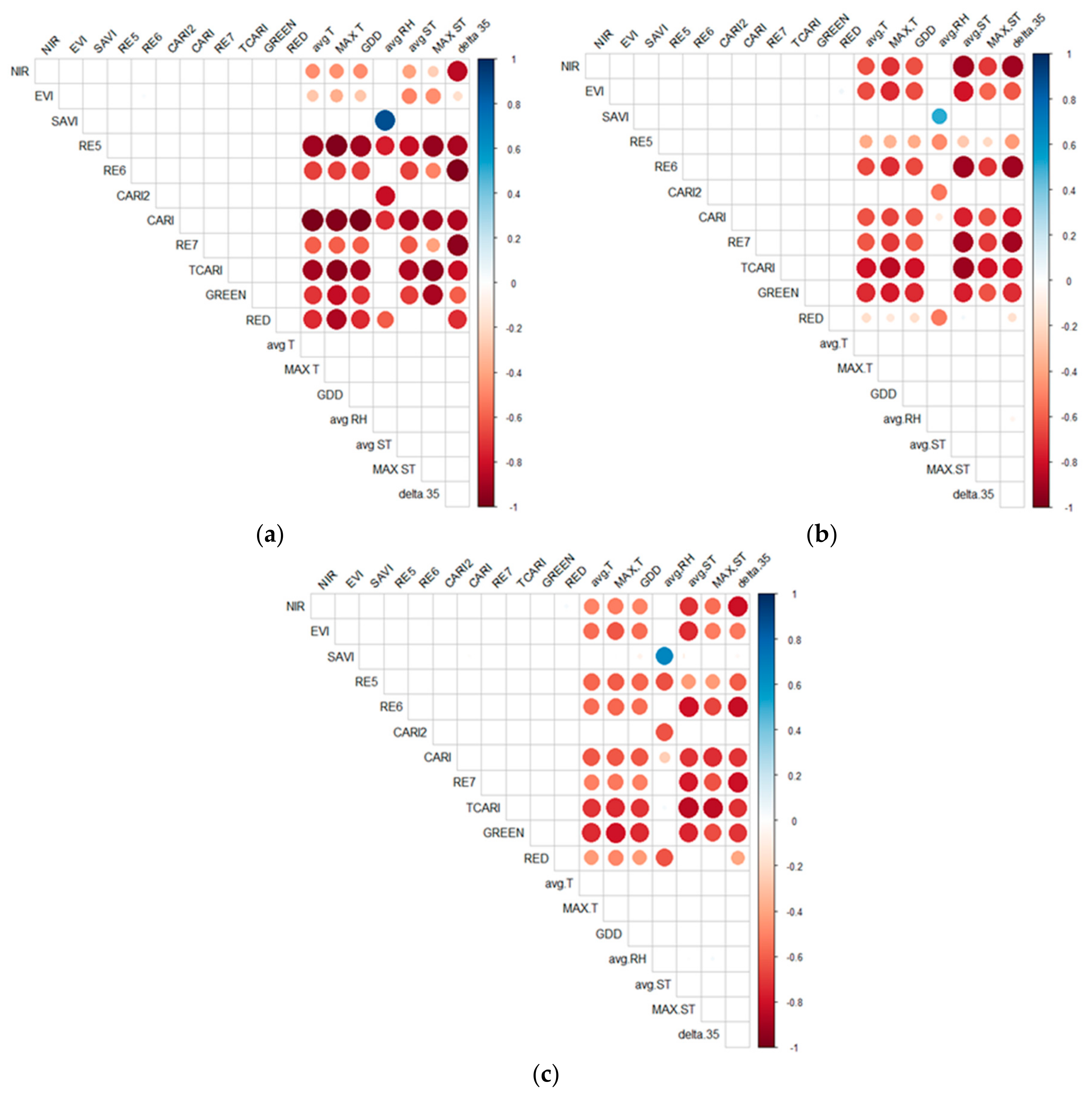
3.2. Analysis 1: Correlations between Environmental Conditions and Spectral Data
3.3. Analysis 2: Spectral Features of the Heatwaves
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Teixeira, A.; Eiras-Dias, J.; Castellarin, S.D.; Gerós, H. Berry phenolics of grapevine under challenging environments. Int. J. Mol. Sci. 2013, 14, 18711–18739. [Google Scholar] [CrossRef] [PubMed]
- Greer, D.H.; Weston, C. Heat stress affects flowering, berry growth, sugar accumulation and photosynthesis of Vitis vinifera cv. Semillon grapevines grown in a controlled environment. Funct. Plant Biol. 2010, 37, 206–214. [Google Scholar] [CrossRef]
- Webb, L.; Whiting, J.; Watt, A.; Hill, T.; Wigg, F.; Dunn, G.; Needs, S.; Barlow, E.W.R. Managing grapevines through severe heat: A survey of growers after the 2009 summer heatwave in south-eastern Australia. J. Wine Res. 2010, 21, 147–165. [Google Scholar] [CrossRef]
- Moriondo, M.; Giannakopoulos, C.; Bindi, M. Climate change impact assessment: The role of climate extremes in crop yield simulation. Clim. Chang. 2011, 104, 679–701. [Google Scholar] [CrossRef]
- Hayman, P.; McCarthy, M.; Thomas, D.; Longbottom, M. Managing Grapevines during Heatwaves. What Is a Heatwave? What Causes Hot Days in Australain Managing Grapevines during Heatwaves What Damage to Grapevines Can Be Caused by Heatwave Events? Wine Australia: Adelaide, Australia, 2014; pp. 1–6. [Google Scholar]
- Carvalho, L.C.; Coito, J.L.; Gonçalves, E.F.; Lopes, C.; Amâncio, S. Physiological and agronomical responses to environmental fluctuations of two Portuguese grapevine varieties during three field seasons. Ciênc. Téc. Vitiviníc. 2018, 33, 1–14. [Google Scholar] [CrossRef]
- Carvalho, L.C.; Gonçalves, E.F.; Coito, J.L.; Amâncio, S.; Chaves, M.M. Differential physiological response of the grapevine varieties Touriga Nacional and Trincadeira to combined heat, drought and light stresses. Plant Biol. 2015, 18, 101–111. [Google Scholar] [CrossRef]
- Zulini, L.; Rubinigg, M.; Zorer, R.; Bertamini, M. Effects of drought stress on chlorophyll fluorescence and photosynthetic pigments in grapevine leaves (Vitis vinifera cv. ’White Riesling’). Acta Hortic. 2007, 754, 289–294. [Google Scholar] [CrossRef]
- Chaves, M.M.; Zarrouk, O.; Francisco, R.; Costa, J.M.; Santos, T.; Regalado, A.P.; Rodrigues, M.L.; Lopes, C.M. Grapevine under deficit irrigation: Hints from physiological and molecular data. Ann. Bot. 2010, 105, 661–676. [Google Scholar] [CrossRef]
- Bergqvist, J.; Dokoozlian, N.; Ebisuda, N. Sunlight exposure and temperature effects on berry growth and composition of Cabernet Sauvignon and Grenache in the central San Joaquin Valley of California. Am. J. Enol. Vitic. 2001, 52, 1–7. [Google Scholar]
- Nicholas, K.A.; Durham, W.H. Farm-scale adaptation and vulnerability to environmental stresses: Insights from winegrowing in Northern California. Glob. Environ. Chang. 2012, 22, 483–494. [Google Scholar] [CrossRef]
- Sepulveda, G.; Kliewer, W.; Ryugo, K. Effect of high temperature on grapevines (Vitis vinifera L.). I: Translocation of 14C-Photosynthates. Am. J. Enol. Vitic. 1986, 37, 13–19. [Google Scholar]
- Sadras, V.O.; Moran, M.A.; Bonada, M. Effects of elevated temperature in grapevine. I Berry sensory traits. Aust. J. Grape Wine Res. 2013, 19, 95–106. [Google Scholar] [CrossRef]
- Spayd, S.E.; Tarara, J.M.; Mee, D.L.; Ferguson, J.C. Separation of Sunlight and Temperature Effects on the Composition of Berries. Am. J. Enol. Vitic. 2002, 3, 171–182. [Google Scholar]
- Zhang, P.; Howell, K.; Krstic, M.; Herderich, M.; Barlow, E.W.R.; Fuentes, S. Environmental factors and seasonality affect the concentration of rotundone in vitis vinifera L. Cv. shiraz wine. PLoS ONE 2015, 10, e0133137. [Google Scholar] [CrossRef] [PubMed]
- Walter, A.; Liebisch, F.; Hund, A. Plant phenotyping: From bean weighing to image analysis. Plant Methods 2015, 11, 14. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Zhang, R.; Song, W.; Han, L.; Liu, X.; Sun, X.; Luo, M.; Chen, K.; Zhang, Y.; Yang, H.; et al. Dynamic plant height QTL revealed in maize through remote sensing phenotyping using a high-throughput unmanned aerial vehicle (UAV). Sci. Rep. 2019, 9, 1–10. [Google Scholar] [CrossRef]
- Bramley, R.G.V.; Trought, M.C.T.; Praat, J.-P. Vineyard variability in Marlborough, New Zealand: Characterising variation in vineyard performance and options for the implementation of Precision Viticulture. Aust. J. Grape Wine Res. 2011, 17, 72–82. [Google Scholar] [CrossRef]
- Ledderhof, D.; Brown, R.; Reynolds, A.; Jollineau, M. Using remote sensing to understand Pinot noir vineyard variability in Ontario. Can. J. Plant Sci. 2016, 96, 89–108. [Google Scholar] [CrossRef]
- Ferrer, M.; Echeverría, G.; Pereyra, G.; Gonzalez-Neves, G.; Pan, D.; Mirás-Avalos, J.M. Mapping vineyard vigor using airborne remote sensing: Relations with yield, berry composition and sanitary status under humid climate conditions. In Precision Agriculture; Springer: Berlin/Heidelberg, Germany, 2019; pp. 1–20. [Google Scholar]
- Hillnhutter, C.; Mahlein, A.-K.; Sikora, R.A.; Oerke, E.-C. Remote sensing to detect plant stress induced by Heterodera schachtii and Rhizoctonia solani in sugar beet fields. Field Crops Res. 2011, 122, 70–77. [Google Scholar] [CrossRef]
- Tirelli, P.; Marchi, M.; Calcante, A.; Vitalini, S.; Iriti, M.; Borghese, N.A.; Oberti, R. Multispectral image analysis for grapevine diseases automatic detection in field conditions. In Proceedings of the International Conference of Agricultural Engineering CIGR-AgEng, Valencia, Spain, 8–12 July 2012; pp. 1–6. [Google Scholar]
- Anderson, M.C.; Hain, C.R.; Jurecka, F.; Trnka, M.; Hlavinka, P.; Dulaney, W.; Otkin, J.A.; Johnson, D.; Gao, F. Relationships between the evaporative stress index and winter wheat and spring barley yield anomalies in the Czech Republic. Clim. Res. 2016, 70, 215–230. [Google Scholar] [CrossRef]
- Gago, J.; Fernie, A.R.; Nikoloski, Z.; Tohge, T.; Martorell, S.; Escalona, J.M.; Ribas-Carbó, M.; Flexas, J.; Medrano, H. Integrative field scale phenotyping for investigating metabolic components of water stress within a vineyard. Plant Methods 2017, 13, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Rao, K.; Anderegg, W.R.L.; Sala, A.; Martínez-Vilalta, J.; Konings, A.G. Satellite-based vegetation optical depth as an indicator of drought-driven tree mortality. Remote Sens. Environ. 2019, 227, 125–136. [Google Scholar] [CrossRef]
- Cogato, A.; Meggio, F.; De Antoni Migliorati, M.; Marinello, F. Extreme weather events in agriculture: A systematic review. Sustainability 2019, 11, 2547. [Google Scholar] [CrossRef]
- Idso, S.B.; Jackson, R.D.; Pinter, P.J.; Reginato, R.J.; Hatfield, J.L. Normalizing the stres-degree-day parameter for environmental variability. Agric. Meteorol. 1981, 24, 45–55. [Google Scholar] [CrossRef]
- Matese, A.; Baraldi, R.; Berton, A.; Cesaraccio, C.; Di Gennaro, S.F.; Duce, P.; Facini, O.; Mameli, M.G.; Piga, A.; Zaldei, A. Estimation of Water Stress in grapevines using proximal and remote sensing methods. Remote Sens. 2018, 10, 114. [Google Scholar] [CrossRef]
- Matese, A.; Di Gennaro, S. Practical Applications of a Multisensor UAV Platform Based on Multispectral, Thermal and RGB High Resolution Images in Precision Viticulture. Agriculture 2018, 8, 116. [Google Scholar] [CrossRef]
- Santesteban, L.G.; Di Gennaro, S.F.; Herrero-Langreo, A.; Miranda, C.; Royo, J.B.; Matese, A. High-resolution UAV-based thermal imaging to estimate the instantaneous and seasonal variability of plant water status within a vineyard. Agric. Water Manag. 2017, 183, 49–59. [Google Scholar] [CrossRef]
- Zarco-Tejada, P.J.; González-Dugo, V.; Williams, L.E.; Suárez, L.; Berni, J.A.J.; Goldhamer, D.; Fereres, E. A PRI-based water stress index combining structural and chlorophyll effects: Assessment using diurnal narrow-band airborne imagery and the CWSI thermal index. Remote Sens. Environ. 2013, 138, 38–50. [Google Scholar] [CrossRef]
- Rouse, J.; Haas, R.; Schell, J.; Deering, D.; Harlan, J. Monitoring the Vernal Advancement and Retrogradation (Greenwave Effect) of Natural Vegetation; RS Center, A Texas, GSF Center Texas A&M University, Remote Sensing Center: Bryan, TX, USA, 1974. [Google Scholar]
- Helman, D.; Bahat, I.; Netzer, Y.; Ben-Gal, A.; Alchanatis, V.; Peeters, A.; Cohen, Y. Using time series of high-resolution planet satellite images to monitor grapevine stem water potential in commercial vineyards. Remote Sens. 2018, 10, 1615. [Google Scholar] [CrossRef]
- Acevedo-Opazo, C.; Tisseyre, B.; Guillaume, S.; Ojeda, H. Test of NDVI information for a relevant vineyard zoning related to vine water status. In Proceedings of the VI European Conference on Precision Agriculture (ECPA), Skiathos, Greece, 3–6 June 2007; pp. 1–9. [Google Scholar]
- Acevedo-Opazo, C.; Tisseyre, B.; Guillaume, S.; Ojeda, H. The potential of high spatial resolution information to define within-vineyard zones related to vine water status. Precis. Agric. 2008, 9, 285–302. [Google Scholar] [CrossRef]
- Haboudane, D.; Miller, J.R.; Tremblay, N.; Zarco-Tejada, P.J.; Dextraze, L. Integrated narrow-band vegetation indices for prediction of crop chlorophyll content for application to precision agriculture. Remote Sens. Environ. 2002, 81, 416–426. [Google Scholar] [CrossRef]
- Baluja, J.; Diago, M.P.; Balda, P.; Zorer, R.; Meggio, F.; Morales, F.; Tardaguila, J. Assessment of vineyard water status variability by thermal and multispectral imagery using an unmanned aerial vehicle (UAV). Irrig. Sci. 2012, 30, 511–522. [Google Scholar] [CrossRef]
- Espinoza, C.Z.; Khot, L.R.; Sankaran, S.; Jacoby, P.W. High resolution multispectral and thermal remote sensing-based water stress assessment in subsurface irrigated grapevines. Remote Sens. 2017, 9, 961. [Google Scholar] [CrossRef]
- Gitelson, A.A.; Merzlyak, M.N. Remote sensing of chlorophyll concentration in higher plant leaves. Adv. Sp. Res. 1998, 22, 689–692. [Google Scholar] [CrossRef]
- Gitelson, A.A.; Stark, R.; Grits, U.; Rundquist, D.; Kaufman, Y.; Derry, D. Vegetation and soil lines in visible spectral space: A concept and technique for remote estimation of vegetation fraction. Int. J. Remote Sens. 2002, 23, 2537–2562. [Google Scholar] [CrossRef]
- Chamard, P.; Courel, M.F.; Ducousso, M.; Guenegou, M.C. Utilisation des bandes spectrales du vert et du rouge pour une meilleure evaluation des formations vegetales actives. In Proceedings of the Journees Scientifiques 4, Reseau Teledetection: Teledetection Appliquee a la Cartographie Thematique et Topographique, Montreal, QC, Canada, 21–23 October 1991; pp. 203–210. [Google Scholar]
- Pôças, I.; Rodrigues, A.; Gonçalves, S.; Costa, P.; Gonçalves, I.; Pereira, L.; Cunha, M. Predicting Grapevine Water Status Based on Hyperspectral Reflectance Vegetation Indices. Remote Sens. 2015, 7, 16460–16479. [Google Scholar] [CrossRef]
- Rondeaux, G.; Steven, M.; Baret, F. Optimization of soil-adjusted vegetation indices. Remote Sens. Environ. 1996, 55, 95–107. [Google Scholar] [CrossRef]
- Romero, M.; Luo, Y.; Su, B.; Fuentes, S. Vineyard water status estimation using multispectral imagery from an UAV platform and machine learning algorithms for irrigation scheduling management. Comput. Electron. Agric. 2018, 147, 109–117. [Google Scholar] [CrossRef]
- Xiang, H.; Tian, L. Development of a low-cost agricultural remote sensing system based on an autonomous unmanned aerial vehicle (UAV). Biosyst. Eng. 2011, 108, 174–190. [Google Scholar] [CrossRef]
- Sozzi, M.; Marinello, F.; Pezzuolo, A.; Sartori, L. Benchmark of Satellites Image Services for Precision Agricultural use. In Proceedings of the AgEng Conference, Wageningen, The Netherlands, 8–12 July 2018; pp. 8–11. [Google Scholar]
- Ciraolo, G.; Capodici, F.; D’Urso, G.; La Loggia, G.; Maltese, A. Mapping Evapotranspiration on Vineyards: The Sentinel-2 Potentiality; Special Publication ESA SP; Eur. Sp. Agency: Harwell, UK, 2012; Volume 707 SP. [Google Scholar]
- Cunha, M.; Marçal, A.R.S.; Silva, L. Very early prediction of wine yield based on satellite data from vegetation. Int. J. Remote Sens. 2010, 31, 3125–3142. [Google Scholar] [CrossRef]
- Sun, L.; Gao, F.; Anderson, M.C.; Kustas, W.P.; Alsina, M.M.; Sanchez, L.; Sams, B.; McKee, L.; Dulaney, W.; White, W.A.; et al. Daily mapping of 30 m LAI and NDVI for grape yield prediction in California vineyards. Remote Sens. 2017, 9, 317. [Google Scholar] [CrossRef]
- Anderson, M.C.; Neale, C.M.U.; Li, F.; Norman, J.M.; Kustas, W.P.; Jayanthi, H.; Chavez, J. Upscaling ground observations of vegetation water content, canopy height, and leaf area index during SMEX02 using aircraft and Landsat imagery. Remote Sens. Environ. 2004, 92, 447–464. [Google Scholar] [CrossRef]
- Richter, K.; Vuolo, F.; D’Urso, G. Leaf area index and surface albedo estimation: Comparative analysis from vegetation indexes to radiative transfer models. Int. Geosci. Remote Sens. Symp. 2008, 3, 4779453. [Google Scholar]
- Mazzetto, F.; Calcante, A.; Mena, A.; Vercesi, A. Integration of optical and analogue sensors for monitoring canopy health and vigour in precision viticulture. Precis. Agric. 2010, 11, 636–649. [Google Scholar] [CrossRef]
- King, P.D.; Smart, R.E.; Mcclellan, D.J. Within-vineyard variability in vine vegetative growth, yield, and fruit and wine composition of Cabernet Sauvignon in Hawke’s Bay, New Zealand. Aust. J. Grape Wine Res. 2014, 20, 234–246. [Google Scholar] [CrossRef]
- Bureau of Meteorology. Available online: http://www.bom.gov.au (accessed on 9 December 2018).
- Greer, D.H.; Weedon, M.M. Modelling photosynthetic responses to temperature of grapevine (Vitis vinifera cv. Semillon) leaves on vines grown in a hot climate. Plant Cell Environ. 2012, 35, 1050–1064. [Google Scholar] [CrossRef]
- Ji, L.; Peters, A.J. Forecasting Vegetation Greenness With Satellite and Climate Data. IEEE Geosci. Remote Sens. Lett. 2004, 1, 3–6. [Google Scholar] [CrossRef]
- Wu, D.; Zhao, X.; Liang, S.; Zhou, T.; Huang, K.; Tang, B.; Zhao, W. Time-lag effects of global vegetation responses to climate change. Glob. Chang. Biol. 2015, 21, 3520–3531. [Google Scholar] [CrossRef]
- Drusch, M.; Del Bello, U.; Carlier, S.; Colin, O.; Fernandez, V.; Gascon, F.; Hoersch, B.; Isola, C.; Laberinti, P.; Martimort, P.; et al. Sentinel-2: ESA’s Optical High-Resolution Mission for GMES Operational Services. Remote Sens. Environ. 2012, 120, 25–36. [Google Scholar] [CrossRef]
- Louis, J.; Debaecker, V.; Pflug, B.; Main-Knorn, M.; Bieniarz, J.; Mueller-Wilm, U.; Cadau, E.; Gascon, F. Sentinel-2 SEN2COR: L2A Processor for Users; Special Publication ESA SP; Eur. Sp. Agency: Harwell, UK, 2016; Volume SP-740, pp. 9–13. [Google Scholar]
- Zuhlke, M.; Fomferra, N.; Brockmann, C.; Peters, M.; Veci, L.; Malik, J.; Regner, P. SNAP (Sentinel Application Platform) and the ESA Sentinel 3 Toolbox. In Proceedings of the Sentinel-3 for Science Workshop, Venice, Italy, 2–5 June 2015; pp. 1–4. [Google Scholar]
- Bivand, R. Implementing Spatial Data Analysis Software Tools in R. Geogr. Anal. 2006, 38, 23–40. [Google Scholar] [CrossRef]
- DigitalGlobe. Available online: http://digitalglobe.com (accessed on 11 November 2018).
- Muja, M.; Lowe, D.G. Scalable nearest neighbor algorithms for high dimensional data. IEEE Trans. Pattern Anal. Mach. Intell. 2014, 36, 2227–2240. [Google Scholar] [CrossRef] [PubMed]
- Bland, J.M.; Altman, D.G. Statistical methods for assessing agreement between two methods of clinical measurement. Lancet 1986, 1, 307–310. [Google Scholar] [CrossRef]
- Kim, M.S.; Daughtry, C.S.T.; Chappelle, E.W.; McMurtrey, J.E. The use of high spectral resolution bands for estimating absorbed photosynthetically active radiation (APAR). In Proceedings of the ISPRS’94, Val d’Isere, France, 17–21 January 1994. [Google Scholar]
- Gitelson, A.A.; Keydan, G.P.; Merzlyak, M.N. Three-band model for noninvasive estimation of chlorophyll, carotenoids, and anthocyanin contents in higher plant leaves. Geophys. Res. Lett. 2006, 33, 2–6. [Google Scholar] [CrossRef]
- Huete, A.; Justice, C.; Van Leeuwen, W. MODIS Vegetation Index (MOD 13) Algorithm Theoretical Basis Document Version 3. 1999. Available online: http://modis.gsfc.nasa.gov/data/atbd/atbd_mod13.pdf (accessed on 15 September 2019).
- Guyot, G.; Baret, F.; Jacquemond, S. Imaging Spectroscopy for Vegetation Studies. In Imaging Spectroscopy: Fundamentals and Prospective Applications. Imaging Spectroscopy: Fundamentals and Prospective Applications; Toselli, F., Bodechtel, J., Eds.; Kluwer Academic Publishers: Dordrecht, The Netherlands, 1992; pp. 145–165. ISBN 0-7923-1535-9. [Google Scholar]
- Daughtry, C.S.T.; Walthall, C.L.; Kim, M.S.; Brown de Colstoun, E.; McMurtrey, J.E. Estimating Corn Leaf Chlorophyll Concentration from Leaf and Canopy Reflectance. Remote Sens. Environ. 2000, 74, 229–239. [Google Scholar] [CrossRef]
- Chen, J.M. Evaluation of vegetation indices and a modified simple ratio for boreal applications. Can. J. Remote Sens. 1996, 22, 229–242. [Google Scholar] [CrossRef]
- Broge, N.H.; Leblanc, E. Comparing prediction power and stability of broadband and hyperspectral vegetation indices for estimation of green leaf area index and canopy chlorophyll density. Remote Sens. Environ. 2001, 76, 156–172. [Google Scholar] [CrossRef]
- Baret, F.; Guyot, G. Potentials and limits of vegetation indices for LAI and APAR assessment. Remote Sens. Environ. 1991, 35, 161–173. [Google Scholar] [CrossRef]
- Di Gennaro, S.F.; Matese, A.; Gioli, B.; Toscano, P.; Zaldei, A.; Palliotti, A.; Genesio, L. Multisensor approach to assess vineyard thermal dynamics combining high-resolution unmanned aerial vehicle (UAV) remote sensing and wireless sensor network (WSN) proximal sensing. Sci. Hortic. 2017, 221, 83–87. [Google Scholar] [CrossRef]
- Hazratkulova, S.; Sharma, R.C.; Alikulov, S.; Islomov, S.; Yuldashev, T.; Ziyaev, Z.; Khalikulov, Z.; Ziyadullaev, Z.; Turok, J. Analysis of genotypic variation for normalized difference vegetation index and its relationship with grain yield in winter wheat under terminal heat stress. Plant Breed. 2012, 131, 716–721. [Google Scholar] [CrossRef]
- Ceccato, P.; Flasse, S.; Tarantola, S.; Jacquemoud, S.; Grégoire, J.M. Detecting vegetation leaf water content using reflectance in the optical domain. Remote Sens. Environ. 2001, 77, 22–33. [Google Scholar] [CrossRef]
- Song, L.; Guanter, L.; Guan, K.; You, L.; Huete, A.; Ju, W.; Zhang, Y. Satellite sun-induced chlorophyll fluorescence detects early response of winter wheat to heat stress in the Indian Indo-Gangetic Plains. Glob. Chang. Biol. 2018, 24, 4023–4037. [Google Scholar] [CrossRef] [PubMed]
- Khaliq, A.; Comba, L.; Biglia, A.; Aimonino, D.R.; Chiaberge, M.; Gay, P. Comparison of satellite and UAV-based multispectral imagery for vineyard variability assessment. Remote Sens. 2019, 11, 436. [Google Scholar] [CrossRef]
- Borgogno-Mondino, E.; Lessio, A.; Tarricone, L.; Novello, V.; de Palma, L. A comparison between multispectral aerial and satellite imagery in precision viticulture. Precis. Agric. 2018, 19, 195–217. [Google Scholar] [CrossRef]
- Reed, B.C. Trend analysis of time-series phenology of North America derived from satellite data. GISci. Remote Sens. 2006, 43, 24–38. [Google Scholar] [CrossRef]
- Bonilla, I.; De Toda, F.M.; Martínez-Casasnovas, J.A. Vine vigor, yield and grape quality assessment by airborne remote sensing over three years: Analysis of unexpected relationships in cv. Tempranillo. Span. J. Agric. Res. 2015, 13, 1–8. [Google Scholar] [CrossRef]
- Mathews, A.J. Object-based spatiotemporal analysis of vine canopy vigor using an inexpensive unmanned aerial vehicle remote sensing system. J. Appl. Remote Sens. 2014, 8, 085199. [Google Scholar] [CrossRef]
- Ghashghaie, J.; Brenckmann, F.; Saugier, B. Water relations and growth of rose plants cultured in vitro under various relative humidities. Plant Cell. Tissue Organ Cult. 1992, 30, 51–57. [Google Scholar] [CrossRef]
- Beadle, C.L.; Ludlow, M.M.; Honeysett, J. Water relations. In Photosynthesis and Production in a Changing Environment; Hall, D.O., Scurlock, J.M.O., Bolhàr-Nordenkampf, H.R., Leegood, R.C., Long, S.P., Eds.; Springer: Dordrecht, The Netherlands, 1993; pp. 113–128. ISBN 9780412429101. [Google Scholar]
- Seelig, H.D.; Hoehn, A.; Stodieck, L.S.; Klaus, D.M.; Adams, W.W.; Emery, W.J. Plant water parameters and the remote sensing R 1300/R 1450 leaf water index: Controlled condition dynamics during the development of water deficit stress. Irrig. Sci. 2009, 27, 357–365. [Google Scholar] [CrossRef]
- Liu, L.; Wang, J.; Huang, W.; Zhao, C.; Zhang, B.; Tong, Q. Estimating winter wheat plant water content using red edge parameters. Int. J. Remote Sens. 2004, 25, 3331–3342. [Google Scholar] [CrossRef]
- Filella, I.; Penuelas, J. The red edge position and shape as indicators of plant chlorophyll content, biomass and hydric status. Int. J. Remote Sens. 1994, 15, 1459–1470. [Google Scholar] [CrossRef]
- Cheng, Y.B.; Zarco-Tejada, P.J.; Riaño, D.; Rueda, C.A.; Ustin, S.L. Estimating vegetation water content with hyperspectral data for different canopy scenarios: Relationships between AVIRIS and MODIS indexes. Remote Sens. Environ. 2006, 105, 354–366. [Google Scholar] [CrossRef]
- Bajgain, R.; Xiao, X.; Wagle, P.; Basara, J.; Zhou, Y. Sensitivity analysis of vegetation indices to drought over two tallgrass prairie sites. ISPRS J. Photogramm. Remote Sens. 2015, 108, 151–160. [Google Scholar] [CrossRef]
- Dong, J.; Xiao, X.; Wagle, P.; Zhang, G.; Zhou, Y.; Jin, C.; Torn, M.S.; Meyers, T.P.; Suyker, A.E.; Wang, J.; et al. Comparison of four EVI-based models for estimating gross primary production of maize and soybean croplands and tallgrass prairie under severe drought. Remote Sens. Environ. 2015, 162, 154–168. [Google Scholar] [CrossRef]
- Caccamo, G.; Chisholm, L.A.; Bradstock, R.A.; Puotinen, M.L. Assessing the sensitivity of MODIS to monitor drought in high biomass ecosystems. Remote Sens. Environ. 2011, 115, 2626–2639. [Google Scholar] [CrossRef]
- Sims, D.A.; Gamon, J.A. Estimation of vegetation water content and photosynthetic tissue area from spectral reflectance: A comparison of indices based on liquid water and chlorophyll absorption features. Remote Sens. Environ. 2003, 84, 526–537. [Google Scholar] [CrossRef]
- Nemani, R.; Pierce, L.; Running, S. Developing Satellite-derived Estimates of Surface Moisture Status. J. Appl. Meteorol. 1992, 32, 548–557. [Google Scholar] [CrossRef]
- Gu, Y.; Brown, J.F.; Verdin, J.P.; Wardlow, B. A five-year analysis of MODIS NDVI and NDWI for grassland drought assessment over the central Great Plains of the United States. Geophys. Res. Lett. 2007, 34, 1–6. [Google Scholar] [CrossRef]
- Ma, S.; Zhou, Y.; Gowda, P.H.; Dong, J.; Zhang, G.; Kakani, V.G.; Wagle, P.; Chen, L.; Flynn, K.C.; Jiang, W. Application of the water-related spectral reflectance indices: A review. Ecol. Indic. 2019, 98, 68–79. [Google Scholar] [CrossRef]
- Heber, U. Conformational changes of chloroplasts induced by illumination of leaves in vivo. Biochim. Biophys. Acta Bioenerg. 1969, 180, 302–319. [Google Scholar] [CrossRef][Green Version]
- Tucker, C.J. Remote sensing of leaf water content in the near infrared. Remote Sens. Environ. 1980, 10, 23–32. [Google Scholar] [CrossRef]
- Zarco-Tejada, P.J.; Berjón, A.; López-Lozano, R.; Miller, J.R.; Martín, P.; Cachorro, V.; González, M.R.; De Frutos, A. Assessing vineyard condition with hyperspectral indices: Leaf and canopy reflectance simulation in a row-structured discontinuous canopy. Remote Sens. Environ. 2005, 99, 271–287. [Google Scholar] [CrossRef]
- Hunt, E.R.; Rock, B.N. Detection of changes in leaf water content using Near- and Middle-Infrared reflectances. Remote Sens. Environ. 1989, 30, 43–54. [Google Scholar]
- Carter, G.A.; McCain, D.C. Relationship of leaf spectral reflectance to chloroplast water content determined using NMR microscopy. Remote Sens. Environ. 1993, 46, 305–310. [Google Scholar] [CrossRef]
| Growing Degree Day Accumulation (° C) | Solar Radiation Accumulation (W/m2) | Average Air Temperature (° C) | Rainfall Accumulation (mm) | |
|---|---|---|---|---|
| 2016–2017 | 1942.7 | 50,969 | 20.3 | 136.8 |
| 2017–2018 | 2028.7 | 48,902 | 21.2 | 62.8 |
| Growing Season | Dates | Duration (days) | Maximum Temperature Registered (°C) |
|---|---|---|---|
| 2016–2017 | 23/12–25/12 | 3 | 41.6 |
| 2016–2017 | 04/01–07/01 | 4 | 40.9 |
| 2016–2017 | 27/01–29/01 | 4 | 40.8 |
| 2016–2017 | 08/02–11/02 | 4 | 45.2 |
| 2017–2018 | 13/11–15/11 | 3 | 37.8 |
| 2017–2018 | 29/11–01/12 | 3 | 38.3 |
| 2017–2018 | 18/01–22/01 | 5 | 42.6 |
| 2017–2018 | 05/02–10/02 | 6 | 40.5 |
| Parameter | Abbreviation |
|---|---|
| Avg daily RH* | avg RH0 |
| Min daily RH | min RH0 |
| Daily VPD* | VPD0 |
| Avg daily T* | avg T0 |
| Max daily T | MAX T0 |
| Avg RH day before | avg RH-1 |
| Min RH day before | min RH-1 |
| VPD day before | VPD-1 |
| Avg T day before | avg T-1 |
| Max T day before | MAX T-1 |
| Avg RH last 3 days | avg RH-3 |
| Min RH last 3 days | min RH-3 |
| VPD last 3 days | VPD-3 |
| Avg T last 3 days | avg T-3 |
| Max T last 3 days | MAX T-3 |
| Growing degree day | GDD |
| Parameter | Abbreviation |
|---|---|
| Avg RH last 12 days* | RH-12 |
| Min RH last 12 days | min RH-12 |
| VPD last 12 days* | VPD-12 |
| ET0 last 12 days* | ET-12 |
| Avg T last 12 days* | avg T-12 |
| Max T last 12 days | MAX T-12 |
| Avg soil T last 12 days | avg ST-12 |
| Max soil T last 12 days | MAX ST-12 |
| Growing degree day last 12 days | GDD-12 |
| Cumulative sum (Δ) of max T above 35 °C last 12 days** | Δ 35 °C-12 |
| Sentinel-2 Band | Central Wavelength (nm) | Bandwidth (nm) | Spatial Resolution (m) |
|---|---|---|---|
| Band 2-Blue | 490 | 65 | 10 |
| Band 3-Green | 560 | 35 | 10 |
| Band 4- Red | 665 | 30 | 10 |
| Band 5- Vegetation Red Edge | 705 | 15 | 20 |
| Band 6-Vegetation Red Edge | 740 | 15 | 20 |
| Band 7-Vegetation Red Edge | 783 | 20 | 20 |
| Band 8-NIR | 842 | 115 | 10 |
| Band 11-SWIR | 1610 | 90 | 20 |
| Index | Equation | Reference |
|---|---|---|
| Chlorophyll Absorption Ratio Index (CARI)* | α = (RED EDGE 5-GREEN)/150 b = (GREEN-((RED EDGE 5-GREEN)/150*550)) | [65] |
| Chlorophyll Absorption Ratio Index 2 (CARI2)* | α = (RED EDGE 5-GREEN)/150 b = (GREEN-((RED EDGE 5-GREEN)/150*GREEN)) | [65] |
| Chlorophyll Green (Chlgreen)* | [66] | |
| Chlorophyll Red-Edge (Chlred-edge)* | [66] | |
| Enhanced Vegetation Index (EVI)** | [67] | |
| Green Normalized Difference Vegetation Index (GNDVI)** | [39] | |
| Linear Red-Edge Index (LREI)* | [68] | |
| Modified chlorophyll absorption in reflectance (MCARI)* | [69] | |
| Modified Simple Ratio (MSR)** | [70] | |
| Normalized Difference Vegetation Index (NDVI)** | [32] | |
| Ratio Difference Vegetation Index (RDVI)** | [71] | |
| Soil-Adjusted Vegetation Index (SAVI)** | [72] | |
| Transformed Chlorophyll Absorption Ratio (TCARI)* | [36] |
| Heatwave Dates | Pre-Heatwave Image Date | Post-Heatwave Image Date |
|---|---|---|
| 23/12/2016–25/12/2016 | 18/12/2016 | 28/12/2016 |
| 04/01/2017–07/01/2017 | 28/12/2016 | 17/01/2017 |
| 24/01/2017–27/01/2017 | 17/01/2017 | 27/01/2017 |
| 08/02/2017–11/02/2017 | 27/01/2017 | 16/02/2017 |
| 13/11/2017–15/11/2017 | 13/11/2017 | 28/11/2017 |
| 29/11/2017–01/12/2017 | 28/11/2017 | 08/12/2017 |
| 18/01/2018–21/01/2018 | 17/01/2018 | 06/02/2018 |
| 05/02/2018–10/02/2018 | 06/02/2018 | 26/02/2018 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cogato, A.; Pagay, V.; Marinello, F.; Meggio, F.; Grace, P.; De Antoni Migliorati, M. Assessing the Feasibility of Using Sentinel-2 Imagery to Quantify the Impact of Heatwaves on Irrigated Vineyards. Remote Sens. 2019, 11, 2869. https://doi.org/10.3390/rs11232869
Cogato A, Pagay V, Marinello F, Meggio F, Grace P, De Antoni Migliorati M. Assessing the Feasibility of Using Sentinel-2 Imagery to Quantify the Impact of Heatwaves on Irrigated Vineyards. Remote Sensing. 2019; 11(23):2869. https://doi.org/10.3390/rs11232869
Chicago/Turabian StyleCogato, Alessia, Vinay Pagay, Francesco Marinello, Franco Meggio, Peter Grace, and Massimiliano De Antoni Migliorati. 2019. "Assessing the Feasibility of Using Sentinel-2 Imagery to Quantify the Impact of Heatwaves on Irrigated Vineyards" Remote Sensing 11, no. 23: 2869. https://doi.org/10.3390/rs11232869
APA StyleCogato, A., Pagay, V., Marinello, F., Meggio, F., Grace, P., & De Antoni Migliorati, M. (2019). Assessing the Feasibility of Using Sentinel-2 Imagery to Quantify the Impact of Heatwaves on Irrigated Vineyards. Remote Sensing, 11(23), 2869. https://doi.org/10.3390/rs11232869