Environmental Reservoirs of Vibrio cholerae: Challenges and Opportunities for Ocean-Color Remote Sensing
Abstract
1. Introduction
2. Global Distribution of Vibrio cholerae and Cholera Disease Outbreaks
3. Interactions of Vibrio cholerae with the Aquatic Environment
3.1. Vibrio cholerae: Growth, Pathogenicity, Dormancy, and Mortality
3.1.1. Growth and Abundance
3.1.2. Dormancy
3.1.3. Pathogenicity
3.1.4. Mortality
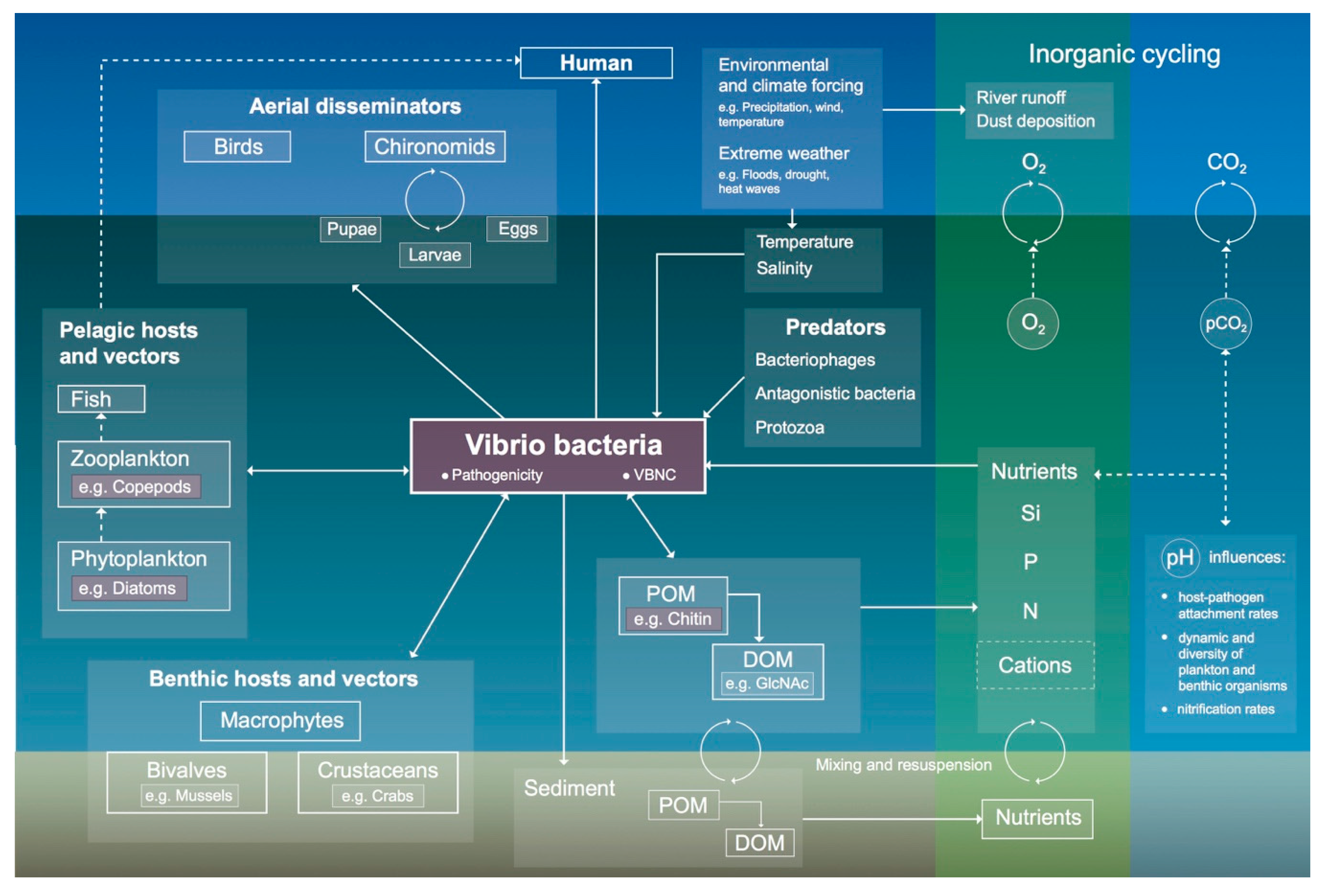
3.2. Environmental Reservoirs and Dissemination of V. cholerae
3.2.1. Host–Pathogen Interactions
3.2.2. Aerial-Dissemination Modes
3.2.3. Aquatic-Dissemination Modes
3.3. Role of Vibrio cholerae in Biogeochemical Cycling
3.4. Climatological Conditions
4. Surveillance and Forecast of Aquatic Reservoirs of Vibrio and Cholera Disease Outbreaks: Existing Approaches and Development Opportunities
4.1. Predictive Models Developed Using Ocean-Color Remote-Sensing Data
4.1.1. Ocean-Color and Cholera Cases
4.1.2. Ocean-Color and V. cholerae Habitat Distribution
4.1.3. Ocean-Color and V. parahaemolyticus in Oysters
4.1.4. Local and Regional Specificity
4.2. Surveillance and Forecast of V. cholerae Reservoirs and Cholera Disease Outbreaks: Development Opportunities from Ocean-Color Remote-Sensing
4.2.1. Sensor-Related Developments
4.2.2. Applications-Related Development
5. Applications of Satellite Remote-Sensing to Cholera Epidemics: Supporting Sustainable Development Goals and Targets
6. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Zhang, X.; Lin, H.; Wang, X.; Austin, B. Significance of Vibrio species in the marine organic carbon cycle—A review. Sci. China Earth Sci. 2018, 61, 1357–1368. [Google Scholar] [CrossRef]
- Huq, A.; Haley, B.J.; Taviani, E.; Chen, A.; Hasan, N.A.; Colwell, R.R. Detection, isolation, and identification of Vibrio cholerae from the environment. Curr. Protoc. Microbiol. 2012, 26, 6A-5. [Google Scholar] [CrossRef] [PubMed]
- Chapman, C.; Henry, M.; Bishop-Lilly, K.A.; Awosika, J.; Briska, A.; Ptashkin, R.N.; Wagner, T.; Rajanna, C.; Tsang, H.; Johnson, S.L.; et al. Scanning the Landscape of Genome Architecture of Non-O1 and Non-O139 Vibrio cholerae by Whole Genome Mapping Reveals Extensive Population Genetic Diversity. PLoS ONE 2015, 10, e0120311. [Google Scholar] [CrossRef] [PubMed]
- World Health Organisation; United Nations Children’s Fund. Wash in the 2030 Agenda: New Global Indicators for Drinking Water, Sanitation and Hygeine; World Health Organisation: Geneva, Switzerland, 2017; p. 8. [Google Scholar]
- Colwell, R.R. Global Climate and Infectious Disease: The Cholera Paradigm*. Science 1996, 274, 2025–2031. [Google Scholar] [CrossRef]
- Jutla, A.S.; Akanda, A.S.; Islam, S. Tracking Cholera in Coastal Regions Using Satellite Observations. JAWRA J. Am. Water Resour. Assoc. 2010, 46, 651–662. [Google Scholar] [CrossRef]
- Hay, S.I.; Battle, K.E.; Pigott, D.M.; Smith, D.L.; Moyes, C.L.; Bhatt, S.; Brownstein, J.S.; Collier, N.; Myers, M.F.; George, D.B.; et al. Global mapping of infectious disease. Philos. Trans. R. Soc. B Biol. Sci. 2013, 368, 20120250. [Google Scholar] [CrossRef]
- Andries, A.; Morse, S.; Murphy, R.; Lynch, J.; Woolliams, E.; Fonweban, J. Translation of Earth observation data into sustainable development indicators: An analytical framework. Sustain. Dev. 2019, 27, 366–376. [Google Scholar] [CrossRef]
- Andries, A.; Morse, S.; Murphy, R.J.; Lynch, J.; Woolliams, E.R. Seeing Sustainability from Space: Using Earth Observation Data to Populate the UN Sustainable Development Goal Indicators. Sustainability 2019, 11, 5062. [Google Scholar] [CrossRef]
- Nilsson, M.; Griggs, D.; Visbeck, M. Policy: Map the interactions between Sustainable Development Goals. Nat. News 2016, 534, 320. [Google Scholar] [CrossRef]
- Kitaoka, M.; Miyata, S.T.; Unterweger, D.; Pukatzki, S. Antibiotic resistance mechanisms of Vibrio cholerae. J. Med. Microbiol. 2011, 60, 397–407. [Google Scholar] [CrossRef]
- Almagro-Moreno, S.; Pruss, K.; Taylor, R.K. Intestinal Colonization Dynamics of Vibrio cholerae. PLoS Pathog. 2015, 11, e1004787. [Google Scholar] [CrossRef] [PubMed]
- Childers, B.M.; Klose, K.E. Regulation of virulence in Vibrio cholerae: The ToxR regulon. Future Med. 2007, 2, 335–344. [Google Scholar]
- Cottingham, K.L.; Chiavelli, D.A.; Taylor, R.K. Environmental microbe and human pathogen: The ecology and microbiology of Vibrio cholerae. Front. Ecol. Environ. 2003, 1, 80–86. [Google Scholar] [CrossRef]
- Lutz, C.; Erken, M.; Noorian, P.; Sun, S.; McDougald, D. Environmental reservoirs and mechanisms of persistence of Vibrio cholerae. Front. Microbiol. 2013, 4, 375. [Google Scholar] [CrossRef] [PubMed]
- Grimes, D.J.; Ford, T.E.; Colwell, R.R.; Baker-Austin, C.; Martinez-Urtaza, J.; Subramaniam, A.; Capone, D.G. Viewing Marine Bacteria, Their Activity and Response to Environmental Drivers from Orbit: Satellite Remote Sensing of Bacteria. Microb. Ecol. 2014, 67, 489–500. [Google Scholar] [CrossRef]
- Jutla, A.S.; Akanda, A.S.; Islam, S. A framework for predicting endemic cholera using satellite derived environmental determinants. Environ. Model. Softw. 2013, 47, 148–158. [Google Scholar] [CrossRef]
- Semenza, J.C.; Trinanes, J.; Lohr, W.; Sudre, B.; Löfdahl, M.; Martinez-Urtaza, J.; Nichols, G.L.; Rocklöv, J. Environmental Suitability of Vibrio Infections in a Warming Climate: An Early Warning System. Environ. Health Perspect. 2017, 125, 107004. [Google Scholar] [CrossRef]
- Colwell, R.; Huq, A. Marine ecosystems and cholera. Hydrobiologia 2001, 460, 141–145. [Google Scholar] [CrossRef]
- Lipp, E.K.; Rose, J.B. The role of seafood in foodborne diseases in the United States of America. Rev. Sci. Tech. Off. Int. Epizoot. 1997, 16, 620–640. [Google Scholar] [CrossRef]
- Feldhusen, F. The role of seafood in bacterialfoodborne diseases. Microbes Infect. 2000, 2, 1651–1660. [Google Scholar] [CrossRef]
- Iwamoto, M.; Ayers, T.; Mahon, B.E.; Swerdlow, D.L. Epidemiology of Seafood-Associated Infections in the United States. Clin. Microbiol. Rev. 2010, 23, 399–411. [Google Scholar] [CrossRef] [PubMed]
- Barange, M. Impacts of Climate Change on Fisheries and Aquaculture: Synthesis of Current Knowledge, Adaptation and Mitigation Options; Food and Agriculture Organization: Rome, Italy, 2018. [Google Scholar]
- Mutreja, A.; Kim, D.W.; Thomson, N.R.; Connor, T.R.; Lee, J.H.; Kariuki, S.; Croucher, N.J.; Choi, S.Y.; Harris, S.R.; Lebens, M.; et al. Evidence for several waves of global transmission in the seventh cholera pandemic. Nature 2011, 477, 462–465. [Google Scholar] [CrossRef] [PubMed]
- Domman, D.; Quilici, M.L.; Dorman, M.J.; Njamkepo, E.; Mutreja, A.; Mather, A.E.; Delgado, G.; Morales-Espinosa, R.; Grimont, P.A.; Lizárraga-Partida, M.L. Integrated view of Vibrio cholerae in the Americas. Science 2017, 358, 789–793. [Google Scholar] [CrossRef] [PubMed]
- Ramamurthy, T.; Mutreja, A.; Weill, F.X.; Das, B.; Ghosh, A.; Nair, G.B. Revisiting the Global Epidemiology of Cholera in Conjunction with the Genomics of Vibrio cholerae. Front. Public Health 2019, 7, 203. [Google Scholar] [CrossRef] [PubMed]
- Thompson, J.R.; Polz, M.F. Dynamics of Vibrio Populations and Their Role in Environmental Nutrient Cycling. Biol. Vibrios 2006, 190–203. [Google Scholar] [CrossRef]
- Böer, S.I.; Heinemeyer, E.A.; Luden, K.; Erler, R.; Gerdts, G.; Janssen, F.; Brennholt, N. Temporal and Spatial Distribution Patterns of Potentially Pathogenic Vibrio spp. at Recreational Beaches of the German North Sea. Microb. Ecol. 2013, 65, 1052–1067. [Google Scholar] [CrossRef]
- Bresnan, E.; Baker-Austin, C.; Campos, C.J.A.; Davidson, K.; Edwards, M.; Hall, A.; Lees, D.; McKinney, A.; Milligan, S.; Silke, J. Human health. MCCIP Sci. Rev. 2017, 2017, 100–107. [Google Scholar]
- Schwartz, K.; Hammerl, J.A.; Göllner, C.; Strauch, E. Environmental and Clinical Strains of Vibrio cholerae Non-O1, Non-O139 From Germany Possess Similar Virulence Gene Profiles. Front. Microbiol. 2019, 10, 733. [Google Scholar] [CrossRef]
- Binsztein, N.; Costagliola, M.C.; Pichel, M.; Jurquiza, V.; Ramírez, F.C.; Akselman, R.; Vacchino, M.; Huq, A.; Colwell, R. Viable but Nonculturable Vibrio cholerae O1 in the Aquatic Environment of Argentina. Appl. Environ. Microbiol. 2004, 70, 7481–7486. [Google Scholar] [CrossRef]
- Martinelli Filho, J.E.; Lopes, R.M.; Rivera, I.N.G.; Colwell, R.R. Vibrio cholerae O1 detection in estuarine and coastal zooplankton. J. Plankton Res. 2011, 33, 51–62. [Google Scholar] [CrossRef]
- Azarian, T.; Ali, A.; Johnson, J.A.; Jubair, M.; Cella, E.; Ciccozzi, M.; Nolan, D.J.; Farmerie, W.; Rashid, M.H.; Sinha-Ray, S.; et al. Non-toxigenic environmental Vibrio cholerae O1 strain from Haiti provides evidence of pre-pandemic cholera in Hispaniola. Sci. Rep. 2016, 6, 36115. [Google Scholar] [CrossRef] [PubMed]
- Chávez, M.D.; Sedas, V.P.; Borunda, E.O.; Reynoso, F.L. Influence of water temperature and salinity on seasonal occurrences of Vibrio cholerae and enteric bacteria in oyster-producing areas of Veracruz, México. Mar. Pollut. Bull. 2005, 50, 1641–1648. [Google Scholar] [CrossRef] [PubMed]
- Lipp, E.K.; Rivera, I.N.G.; Gil, A.I.; Espeland, E.M.; Choopun, N.; Louis, V.R.; Russek-Cohen, E.; Huq, A.; Colwell, R.R. Direct Detection of Vibrio cholerae and ctxA in Peruvian Coastal Water and Plankton by PCR. Appl. Environ. Microbiol. 2003, 69, 3676–3680. [Google Scholar] [CrossRef] [PubMed]
- Gil, A.I.; Louis, V.R.; Rivera, I.N.G.; Lipp, E.; Huq, A.; Lanata, C.F.; Taylor, D.N.; Russek-Cohen, E.; Choopun, N.; Sack, R.B.; et al. Occurrence and distribution of Vibrio cholerae in the coastal environment of Peru. Environ. Microbiol. 2004, 6, 699–706. [Google Scholar] [CrossRef]
- Graü, C.; Barbera, A.L.; Zerpa, A. Isolation of Vibrio spp. And Evaluation of the Sanitary Condition of Bivalve Mollusks Arca zebra and Perna perna Collected on the Northeastern Coast of Sucre State, Venezuela. Rev. Cient. 2004, 14, 513–521. [Google Scholar]
- Louis, V.R.; Russek-Cohen, E.; Choopun, N.; Rivera, I.N.G.; Gangle, B.; Jiang, S.C.; Rubin, A.; Patz, J.A.; Huq, A.; Colwell, R.R. Predictability of Vibrio cholerae in Chesapeake Bay. Appl. Environ. Microbiol. 2003, 69, 2773–2785. [Google Scholar] [CrossRef]
- Dobbs, F.C.; Goodrich, A.L.; Thomson, F.K.; Hynes, W. Pandemic Serotypes of Vibrio cholerae Isolated from Ships’ Ballast Tanks and Coastal Waters: Assessment of Antibiotic Resistance and Virulence Genes (tcpA and ctxA). Microb. Ecol. 2013, 65, 969–974. [Google Scholar] [CrossRef]
- Kiiru, J.; Mutreja, A.; Mohamed, A.A.; Kimani, R.W.; Mwituria, J.; Sanaya, R.O.; Muyodi, J.; Revathi, G.; Parkhill, J.; Thomson, N.; et al. A Study on the Geophylogeny of Clinical and Environmental Vibrio cholerae in Kenya. PLoS ONE 2013, 8, e74829. [Google Scholar] [CrossRef]
- Du Preez, M.; Van der Merwe, M.R.; Cumbana, A.; Le Roux, W. A survey of Vibrio cholerae O1 and O139 in estuarine waters and sediments of Beira, Mozambique. Water SA 2010, 36. [Google Scholar] [CrossRef]
- Mukhopadhyay, A.K.; Basu, A.; Garg, P.; Bag, P.K.; Ghosh, A.; Bhattacharya, S.K.; Takeda, Y.; Nair, G.B. Molecular Epidemiology of Reemergent Vibrio cholerae O139 Bengal in India. J. Clin. Microbiol. 1998, 36, 2149–2152. [Google Scholar]
- Sack, R.B.; Siddique, A.K.; Longini, I.M., Jr.; Nizam, A.; Yunus, M.; Islam, M.S.; Morris, J.G., Jr.; Ali, A.; Huq, A.; Nair, G.B.; et al. A 4-Year Study of the Epidemiology of Vibrio cholerae in Four Rural Areas of Bangladesh. J. Infect. Dis. 2003, 187, 96–101. [Google Scholar] [CrossRef] [PubMed]
- de Magny, G.C.; Murtugudde, R.; Sapiano, M.R.P.; Nizam, A.; Brown, C.W.; Busalacchi, A.J.; Yunus, M.; Nair, G.B.; Gil, A.I.; Lanata, C.F.; et al. Environmental signatures associated with cholera epidemics. Proc. Natl. Acad. Sci. USA 2008, 105, 17676–17681. [Google Scholar] [CrossRef] [PubMed]
- Batabyal, P.; Mookerjee, S.; Einsporn, M.H.; Lara, R.J.; Palit, A. Environmental drivers on seasonal abundance of riverine-estuarine V. cholerae in the Indian Sundarban mangrove. Ecol. Indic. 2016, 69, 59–65. [Google Scholar] [CrossRef]
- Ali, M.; Gupta, S.S.; Arora, N.; Khasnobis, P.; Venkatesh, S.; Sur, D.; Nair, G.B.; Sack, D.A.; Ganguly, N.K. Identification of burden hotspots and risk factors for cholera in India: An observational study. PLoS ONE 2017, 12, e0183100. [Google Scholar] [CrossRef] [PubMed]
- Xu, M.; Cao, C.X.; Wang, D.C.; Kan, B.; Xu, Y.F.; Ni, X.L.; Zhu, Z.C. Environmental factor analysis of cholera in China using remote sensing and geographical information systems. Epidemiol. Infect. 2016, 144, 940–951. [Google Scholar] [CrossRef]
- Emch, M.; Feldacker, C.; Islam, M.S.; Ali, M. Seasonality of cholera from 1974 to 2005: A review of global patterns. Int. J. Health Geogr. 2008, 7, 31. [Google Scholar] [CrossRef]
- Vezzulli, L.; Pruzzo, C.; Huq, A.; Colwell, R.R. Environmental reservoirs of Vibrio cholerae and their role in cholera: Environmental reservoirs of V. cholerae. Environ. Microbiol. Rep. 2010, 2, 27–33. [Google Scholar] [CrossRef]
- Vezzulli, L.; Grande, C.; Reid, P.C.; Hélaouët, P.; Edwards, M.; Höfle, M.G.; Brettar, I.; Colwell, R.R.; Pruzzo, C. Climate influence on Vibrio and associated human diseases during the past half-century in the coastal North Atlantic. Proc. Natl. Acad. Sci. USA 2016, 113, E5062–E5071. [Google Scholar] [CrossRef]
- Baker-Austin, C.; Trinanes, J.A.; Taylor, N.G.H.; Hartnell, R.; Siitonen, A.; Martinez-Urtaza, J. Emerging Vibrio risk at high latitudes in response to ocean warming. Nat. Clim. Chang. 2013, 3, 73–77. [Google Scholar] [CrossRef]
- Huq, A.; Sack, R.B.; Nizam, A.; Longini, I.M.; Nair, G.B.; Ali, A.; Morris, J.G.; Khan, M.N.H.; Siddique, A.K.; Yunus, M.; et al. Critical Factors Influencing the Occurrence of Vibrio cholerae in the Environment of Bangladesh. Appl. Environ. Microbiol. 2005, 71, 4645–4654. [Google Scholar] [CrossRef]
- Takemura, A.F.; Chien, D.M.; Polz, M.F. Associations and dynamics of Vibrionaceae in the environment, from the genus to the population level. Front. Microbiol. 2014, 5, 38. [Google Scholar] [CrossRef] [PubMed]
- Escobar, L.E.; Ryan, S.J.; Stewart-Ibarra, A.M.; Finkelstein, J.L.; King, C.A.; Qiao, H.; Polhemus, M.E. A global map of suitability for coastal Vibrio cholerae under current and future climate conditions. Acta Trop. 2015, 149, 202–211. [Google Scholar] [CrossRef]
- Kopprio, G.A.; Streitenberger, M.E.; Okuno, K.; Baldini, M.; Biancalana, F.; Fricke, A.; Martínez, A.; Neogi, S.B.; Koch, B.P.; Yamasaki, S.; et al. Biogeochemical and hydrological drivers of the dynamics of Vibrio species in two Patagonian estuaries. Sci. Total Environ. 2017, 579, 646–656. [Google Scholar] [CrossRef] [PubMed]
- Beardsley, C.; Pernthaler, J.; Wosniok, W.; Amann, R. Are Readily Culturable Bacteria in Coastal North Sea Waters Suppressed by Selective Grazing Mortality? Appl. Environ. Microbiol. 2003, 69, 2624–2630. [Google Scholar] [CrossRef] [PubMed]
- Worden, A.Z.; Seidel, M.; Smriga, S.; Wick, A.; Malfatti, F.; Bartlett, D.; Azam, F. Trophic regulation of Vibrio cholerae in coastal marine waters. Environ. Microbiol. 2006, 8, 21–29. [Google Scholar] [CrossRef]
- Westrich, J.R.; Ebling, A.M.; Landing, W.M.; Joyner, J.L.; Kemp, K.M.; Griffin, D.W.; Lipp, E.K. Saharan dust nutrients promote Vibrio bloom formation in marine surface waters. Proc. Natl. Acad. Sci. USA 2016, 113, 5964–5969. [Google Scholar] [CrossRef]
- Zhang, R.; Kelly, R.L.; Kauffman, K.M.; Reid, A.K.; Lauderdale, J.M.; Follows, M.J.; John, S.G. Growth of marine Vibrio in oligotrophic environments is not stimulated by the addition of inorganic iron. Earth Planet. Sci. Lett. 2019, 516, 148–155. [Google Scholar] [CrossRef]
- Fernández-Delgado, M.; García-Amado, M.A.; Contreras, M.; Incani, R.N.; Chirinos, H.; Rojas, H.; Suárez, P. SURVIVAL, INDUCTION AND RESUSCITATION OF Vibrio cholerae FROM THE VIABLE BUT NON-CULTURABLE STATE IN THE SOUTHERN CARIBBEAN SEA. Rev. Inst. Med. Trop. São Paulo 2015, 57, 21–26. [Google Scholar] [CrossRef]
- Faruque, S.M.; Mekalanos, J.J. Phage-bacterial interactions in the evolution of toxigenic Vibrio cholerae. Virulence 2012, 3, 556–565. [Google Scholar] [CrossRef]
- Ellison, C.K.; Dalia, T.N.; Vidal Ceballos, A.; Wang, J.C.Y.; Biais, N.; Brun, Y.V.; Dalia, A.B. Retraction of DNA-bound type IV competence pili initiates DNA uptake during natural transformation in Vibrio cholerae. Nat. Microbiol. 2018, 3, 773–780. [Google Scholar] [CrossRef]
- Suzuki, S.; Hoa, P.T.P. Distribution of Quinolones, Sulfonamides, Tetracyclines in Aquatic Environment and Antibiotic Resistance in Indochina. Front. Microbiol. 2012, 3, 67. [Google Scholar] [CrossRef] [PubMed]
- Wilhelm, S.W.; Suttle, C.A. Viruses and Nutrient Cycles in the SeaViruses play critical roles in the structure and function of aquatic food webs. BioScience 1999, 49, 781–788. [Google Scholar] [CrossRef]
- Abd, H.; Saeed, A.; Weintraub, A.; Nair, G.B.; Sandstrom, G. Vibrio cholerae O1 strains are facultative intracellular bacteria, able to survive and multiply symbiotically inside the aquatic free-living amoeba Acanthamoeba castellanii: Vibrio cholerae in amoebae. FEMS Microbiol. Ecol. 2007, 60, 33–39. [Google Scholar] [CrossRef] [PubMed]
- Wei, Y.; Perez, L.J.; Ng, W.L.; Semmelhack, M.F.; Bassler, B.L. Mechanism of Vibrio cholerae Autoinducer-1 Biosynthesis. ACS Chem. Biol. 2011, 6, 356–365. [Google Scholar] [CrossRef] [PubMed]
- Conner, J.G.; Teschler, J.K.; Jones, C.J.; Yildiz, F.H. Staying alive: Vibrio cholerae’s cycle of environmental survival, transmission, and dissemination. Microbiol. Spectr. 2016, 4. [Google Scholar] [CrossRef]
- Madico, G.; Checkley, W.; Gilman, R.H.; Bravo, N.; Cabrera, L.; Calderon, M.; Ceballos, A. Active surveillance for Vibrio cholerae O1 and vibriophages in sewage water as a potential tool to predict cholera outbreaks. J. Clin. Microbiol. 1996, 34, 2968–2972. [Google Scholar]
- Jensen, M.A.; Faruque, S.M.; Mekalanos, J.J.; Levin, B.R. Modeling the role of bacteriophage in the control of cholera outbreaks. Proc. Natl. Acad. Sci. USA 2006, 103, 4652–4657. [Google Scholar] [CrossRef]
- Hood, M.A.; Winter, P.A. Attachment of Vibrio cholerae under various environmental conditions and to selected substrates. FEMS Microbiol. Ecol. 1997, 22, 215–223. [Google Scholar] [CrossRef]
- Paz, S.; Broza, M. Wind Direction and Its Linkage with Vibrio cholerae Dissemination. Environ. Health Perspect. 2007, 115, 195–200. [Google Scholar] [CrossRef][Green Version]
- Paz, S. The cholera epidemic in Yemen—How did it start? The role of El Niño conditions followed by regional winds. Environ. Res. 2019, 176, 108571. [Google Scholar] [CrossRef]
- Halpern, M.; Senderovich, Y.; Izhaki, I. Waterfowl—The Missing Link in Epidemic and Pandemic Cholera Dissemination? PLoS Pathog. 2008, 4, e1000173. [Google Scholar] [CrossRef] [PubMed]
- Senderovich, Y.; Izhaki, I.; Halpern, M. Fish as Reservoirs and Vectors of Vibrio cholerae. PLoS ONE 2010, 5, e8607. [Google Scholar] [CrossRef] [PubMed]
- Halpern, M.; Izhaki, I. Fish as Hosts of Vibrio cholerae. Front. Microbiol. 2017, 8, 282. [Google Scholar] [CrossRef] [PubMed]
- Frischkorn, K.R.; Stojanovski, A.; Paranjpye, R. Vibrio parahaemolyticus type IV pili mediate interactions with diatom-derived chitin and point to an unexplored mechanism of environmental persistence. Environ. Microbiol. 2013, 15, 1416–1427. [Google Scholar] [CrossRef] [PubMed]
- Weiss, I.M.; Schönitzer, V.; Eichner, N.; Sumper, M. The chitin synthase involved in marine bivalve mollusk shell formation contains a myosin domain. FEBS Lett. 2006, 580, 1846–1852. [Google Scholar] [CrossRef]
- Dalusi, L.; Lyimo, T.J.; Lugomela, C.; Hosea, K.M.M.; Sjöling, S. Toxigenic Vibrio cholerae identified in estuaries of Tanzania using PCR techniques. FEMS Microbiol. Lett. 2015, 362, fnv009. [Google Scholar] [CrossRef]
- Meibom, K.L.; Li, X.B.; Nielsen, A.T.; Wu, C.Y.; Roseman, S.; Schoolnik, G.K. The Vibrio cholerae chitin utilization program. Proc. Natl. Acad. Sci. USA 2004, 101, 2524–2529. [Google Scholar] [CrossRef]
- Huq, A.; West, P.A.; Small, E.B.; Huq, M.I.; Colwell, R.R. Influence of water temperature, salinity, and pH on survival and growth of toxigenic Vibrio cholerae serovar 01 associated with live copepods in laboratory microcosms. Appl. Environ. Microbiol. 1984, 48, 420–424. [Google Scholar]
- Colwell, R.R. Polyphasic Taxonomy of the Genus Vibrio: Numerical Taxonomy of Vibrio cholerae, Vibrio parahaemolyticus, and Related Vibrio Species. J. Bacteriol. 1970, 104, 410–433. [Google Scholar]
- Li, X.; Wang, L.X.; Wang, X.; Roseman, S. The chitin catabolic cascade in the marine bacterium Vibrio cholerae: Characterization of a unique chitin oligosaccharide deacetylase. Glycobiology 2007, 17, 1377–1387. [Google Scholar] [CrossRef]
- Pruzzo, C.; Vezzulli, L.; Colwell, R.R. Global impact of Vibrio cholerae interactions with chitin. Environ. Microbiol. 2008, 10, 1400–1410. [Google Scholar] [CrossRef] [PubMed]
- Sison-Mangus, M.P.; Jiang, S.; Kudela, R.M.; Mehic, S. Phytoplankton-Associated Bacterial Community Composition and Succession during Toxic Diatom Bloom and Non-Bloom Events. Front. Microbiol. 2016, 7, 1433. [Google Scholar] [CrossRef] [PubMed]
- Broza, M.; Gancz, H.; Halpern, M.; Kashi, Y. Adult non-biting midges: Possible windborne carriers of Vibrio cholerae non-O1 non-O139. Environ. Microbiol. 2005, 7, 576–585. [Google Scholar] [CrossRef] [PubMed]
- Broza, M.; Halpern, M. Chironomid egg masses and Vibrio cholerae. Nature 2001, 412, 40. [Google Scholar] [CrossRef] [PubMed]
- Ogg, J.E.; Ryder, R.A.; Smith, H.L. Isolation of Vibrio cholerae from aquatic birds in Colorado and Utah. Appl. Environ. Microbiol. 1989, 55, 95–99. [Google Scholar] [PubMed]
- Laviad-Shitrit, S.; Lev-Ari, T.; Katzir, G.; Sharaby, Y.; Izhaki, I.; Halpern, M. Great cormorants (Phalacrocorax carbo) as potential vectors for the dispersal of Vibrio cholerae. Sci. Rep. 2017, 7, 7973. [Google Scholar] [CrossRef] [PubMed]
- Laviad-Shitrit, S.; Izhaki, I.; Arakawa, E.; Halpern, M. Wild waterfowl as potential vectors of Vibrio cholerae and Aeromonas species. Trop. Med. Int. Health 2018, 23, 758–764. [Google Scholar] [CrossRef]
- Spira, W.M.; Huq, A.; Ahmed, Q.S.; Saeed, Y.A. Uptake of Vibrio cholerae Biotype eltor from Contaminated Water by Water Hyacinth (Eichornia crassipes). Appl. Environ. Microbiol. 1981, 42, 550–553. [Google Scholar]
- Feikin, D.R.; Tabu, C.W.; Gichuki, J. Does Water Hyacinth on East African Lakes Promote Cholera Outbreaks? Am. J. Trop. Med. Hyg. 2010, 83, 370–373. [Google Scholar] [CrossRef]
- Wachsmuth, I.K.; Evins, G.M.; Fields, P.I.; Olsvik, Ø.; Popovic, T.; Bopp, C.A.; Wells, J.G.; Carrillo, C.; Blake, P.A. The Molecular Epidemiology of Cholera in Latin America. J. Infect. Dis. 1993, 167, 621–626. [Google Scholar] [CrossRef]
- McCarthy, S.A.; Khambaty, F.M. International dissemination of epidemic Vibrio cholerae by cargo ship ballast and other nonpotable waters. Appl. Environ. Microbiol. 1994, 60, 2597–2601. [Google Scholar] [PubMed]
- Rivera, I.N.G.; Souza, K.M.C.; Souza, C.P.; Lopes, R.M. Free-Living and Plankton-Associated Vibrios: Assessment in Ballast Water, Harbor Areas, and Coastal Ecosystems in Brazil. Front. Microbiol. 2013, 3, 443. [Google Scholar] [CrossRef] [PubMed]
- Ng, C.; Le, T.H.; Goh, S.G.; Liang, L.; Kim, Y.; Rose, J.B.; Yew-Hoong, K.G. A Comparison of Microbial Water Quality and Diversity for Ballast and Tropical Harbor Waters. PLoS ONE 2015, 10, e0143123. [Google Scholar] [CrossRef] [PubMed]
- Kirstein, I.V.; Kirmizi, S.; Wichels, A.; Garin-Fernandez, A.; Erler, R.; Löder, M.; Gerdts, G. Dangerous hitchhikers? Evidence for potentially pathogenic Vibrio spp. on microplastic particles. Mar. Environ. Res. 2016, 120, 1–8. [Google Scholar] [CrossRef]
- Rodrigues, A.; Oliver, D.M.; McCarron, A.; Quilliam, R.S. Colonisation of plastic pellets (nurdles) by E. coli at public bathing beaches. Mar. Pollut. Bull. 2019, 139, 376–380. [Google Scholar] [CrossRef]
- Yokota, K.; Waterfield, H.; Hastings, C.; Davidson, E.; Kwietniewski, E.; Wells, B. Finding the missing piece of the aquatic plastic pollution puzzle: Interaction between primary producers and microplastics. Limnol. Oceanogr. Lett. 2017, 2, 91–104. [Google Scholar] [CrossRef]
- Jeuniaux, C.; Voss-Foucart, M.F. Chitin biomass and production in the marine environment. Biochem. Syst. Ecol. 1991, 19, 347–356. [Google Scholar] [CrossRef]
- Trtanj, J.; Jantarasami, L.; Brunkard, J.; Collier, T.; Jacobs, J.; Lipp, E.; McLellan, S.; Moore, S.; Paerl, H.; Ravenscroft, J.; et al. Ch. 6: Climate Impacts on Water-Related Illness. The Impacts of Climate Change on Human Health in the United States: A Scientific Assessment; MFR, U.S. Global Change Research Program: Washington, DC, USA, 2016. [Google Scholar]
- Parmesan, C.; Attrill, M. Impacts and effects of ocean warming on human health (disease). In Explaining Ocean Warming: Causes, Scale, Effects and Consequences; Laffoley, D., Baxter, J.M., Eds.; IUCN: Gland, Switzerland, 2016; pp. 439–449. [Google Scholar]
- Koelle, K. The impact of climate on the disease dynamics of cholera. Clin. Microbiol. Infect. 2009, 15, 29–31. [Google Scholar] [CrossRef]
- Colwell, R.; Patz, J. Climate, Infectious Disease and Health: An Interdisciplinary Perspective; MFR: Washington, DC, USA, 1998. [Google Scholar]
- Pascual, M.; Bouma, M.J.; Dobson, A.P. Cholera and climate: Revisiting the quantitative evidence. Microbes Infect. 2002, 4, 237–245. [Google Scholar] [CrossRef]
- Pascual, M.; Chaves, L.; Cash, B.; Rodó, X.; Yunus, M. Predicting endemic cholera: The role of climate variability and disease dynamics. Clim. Res. 2008, 36, 131–140. [Google Scholar] [CrossRef]
- Moore, J.K.; Fu, W.; Primeau, F.; Britten, G.L.; Lindsay, K.; Long, M.; Doney, S.C.; Mahowald, N.; Hoffman, F.; Randerson, J.T. Sustained climate warming drives declining marine biological productivity. Science 2018, 359, 1139–1143. [Google Scholar] [CrossRef] [PubMed]
- Martinez, P.P.; Reiner, R.C.; Cash, B.A.; Rodó, X.; Mondal, M.S.; Roy, M.; Yunus, M.; Faruque, A.S.G.; Huq, S.; King, A.A.; et al. Cholera forecast for Dhaka, Bangladesh, with the 2015–2016 El Niño: Lessons learned. PLoS ONE 2017, 12, e0172355. [Google Scholar] [CrossRef] [PubMed]
- Cash, B.A.; Rodó, X.; Kinter, J.L. Links between Tropical Pacific SST and Cholera Incidence in Bangladesh: Role of the Eastern and Central Tropical Pacific. J. Clim. 2008, 21, 4647–4663. [Google Scholar] [CrossRef]
- Hashizume, M.; Armstrong, B.; Hajat, S.; Wagatsuma, Y.; Faruque, A.S.G.; Hayashi, T.; Sack, D.A. The Effect of Rainfall on the Incidence of Cholera in Bangladesh. Epidemiology 2008, 19, 103–110. [Google Scholar] [CrossRef] [PubMed]
- Martinez-Urtaza, J.; Trinanes, J.; Gonzalez-Escalona, N.; Baker-Austin, C. Is El Niño a long-distance corridor for waterborne disease? Nat. Microbiol. 2016, 1, 16018. [Google Scholar] [CrossRef]
- Vezzulli, L.; Brettar, I.; Pezzati, E.; Reid, P.C.; Colwell, R.R.; Höfle, M.G.; Pruzzo, C. Long-term effects of ocean warming on the prokaryotic community: Evidence from the vibrios. ISME J. 2012, 6, 21–30. [Google Scholar] [CrossRef]
- Jacobs, J.; Moore, S.K.; Kunkel, K.E.; Sun, L. A framework for examining climate-driven changes to the seasonality and geographical range of coastal pathogens and harmful algae. Clim. Risk Manag. 2015, 8, 16–27. [Google Scholar] [CrossRef]
- Sperling, M.; Piontek, J.; Gerdts, G.; Wichels, A.; Schunck, H.; Roy, A.S.; Roche, J.L.; Gilbert, J.; Nissimov, J.I.; Bittner, L.; et al. Effect of elevated CO2 on the dynamics of particle-attached and free-living bacterioplankton communities in an Arctic fjord. Biogeosciences 2013, 10, 181–191. [Google Scholar] [CrossRef]
- Riebesell, U.; Aberle-Malzahn, N.; Achterberg, E.P.; Algueró-Muñiz, M.; Alvarez-Fernandez, S.; Arístegui, J.; Bach, L.T.; Boersma, M.; Boxhammer, T.; Guan, W.; et al. Toxic algal bloom induced by ocean acidification disrupts the pelagic food web. Nat. Clim. Chang. 2018, 8, 1082–1086. [Google Scholar] [CrossRef]
- Beck, L.R.; Lobitz, B.M.; Wood, B.L. Remote sensing and human health: New sensors and new opportunities. Emerg. Infect. Dis. 2000, 6, 217. [Google Scholar] [CrossRef]
- Lobitz, B.; Beck, L.; Huq, A.; Wood, B.; Fuchs, G.; Faruque, A.S.G.; Colwell, R. Climate and infectious disease: Use of remote sensing for detection of Vibrio cholerae by indirect measurement. Proc. Natl. Acad. Sci. USA 2000, 97, 1438–1443. [Google Scholar] [CrossRef] [PubMed]
- Tatem, A.J.; Goetz, S.J.; Hay, S.I. Terra and Aqua: New data for epidemiology and public health. Int. J. Appl. Earth Obs. Geoinf. 2004, 6, 33–46. [Google Scholar] [CrossRef] [PubMed]
- de Magny, G.C.; Paroissin, C.; Cazelles, B.; de Lara, M.; Delmas, J.F.; Guégan, J.F. Modeling environmental impacts of plankton reservoirs on cholera population dynamics. ESAIM Proc. 2005, 14, 156–173. [Google Scholar] [CrossRef]
- Finger, F.; Knox, A.; Bertuzzo, E.; Mari, L.; Bompangue, D.; Gatto, M.; Rodriguez-Iturbe, I.; Rinaldo, A. Cholera in the Lake Kivu region (DRC): Integrating remote sensing and spatially explicit epidemiological modeling. Water Resour. Res. 2014, 50, 5624–5637. [Google Scholar] [CrossRef]
- Sathyendranath, S.; Abdulaziz, A.; Menon, N.; Grinson, G.; Evers-King, H.; Kulk, G.; Colwell, R.; Jutla, A.S.; Platt, T. Building capacity and resilience against diseases transmitted via water under climate perturbations and extreme weather stress. In Space Capacity Building in the XXI Century; Ferretti, S., Ed.; Springer Nature: Basel, Switzerland, 2019; Volume 24, p. 14. [Google Scholar]
- Konrad, S.; Paduraru, P.; Romero-Barrios, P.; Henderson, S.B.; Galanis, E. Remote sensing measurements of sea surface temperature as an indicator of Vibrio parahaemolyticus in oyster meat and human illnesses. Environ. Health 2017, 16, 92. [Google Scholar] [CrossRef]
- Xu, M.; Cao, C.; Wang, D.; Kan, B. Identifying Environmental Risk Factors of Cholera in a Coastal Area with Geospatial Technologies. Int. J. Environ. Res. Public Health 2015, 12, 354–370. [Google Scholar] [CrossRef]
- Jutla, A.; Akanda, A.S.; Huq, A.; Faruque, A.S.G.; Colwell, R.; Islam, S. A water marker monitored by satellites to predict seasonal endemic cholera. Remote Sens. Lett. 2013, 4, 822–831. [Google Scholar] [CrossRef]
- Jutla, A.S.; Akanda, A.S.; Islam, S. Satellite Remote Sensing of Space-Time Plankton Variability in the Bay of Bengal: Connections to Cholera Outbreaks. Remote Sens. Environ. 2012, 123, 196–206. [Google Scholar] [CrossRef]
- Nkoko, D.; Giraudoux, P.; Plisnier, P.D.; Tinda, A.; Piarroux, M.; Sudre, B.; Horion, S.; Tamfum, J.J.; Ilunga, B.; Piarroux, R. Dynamics of Cholera Outbreaks in Great Lakes Region of Africa, 1978–2008. Emerg. Infect. Dis. 2011, 17, 2026. [Google Scholar] [CrossRef]
- Emch, M.; Yunus, M.; Escamilla, V.; Feldacker, C.; Ali, M. Local population and regional environmental drivers of cholera in Bangladesh. Environ. Health 2010, 9, 2. [Google Scholar] [CrossRef]
- Ford, T.E. Using Satellite Images of Environmental Changes to Predict Infectious Disease Outbreaks. Emerg. Infect. Dis. 2009, 15, 1341–1346. [Google Scholar] [CrossRef] [PubMed]
- Mendelsohn, J.; Dawson, T. Climate and cholera in KwaZulu-Natal, South Africa: The role of environmental factors and implications for epidemic preparedness. Int. J. Hyg. Environ. Health 2008, 211, 156–162. [Google Scholar] [CrossRef] [PubMed]
- Phillips, A.M.B.; DePaola, A.; Bowers, J.; Ladner, S.; Grimes, D.J. An Evaluation of the Use of Remotely Sensed Parameters for Prediction of Incidence and Risk Associated with Vibrio parahaemolyticus in Gulf Coast Oysters (Crassostrea virginica). J. Food Prot. 2007, 70, 879–884. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Sathyendranath, S.; Brewin, R.J.W.; Brockmann, C.; Brotas, V.; Calton, B.; Chuprin, A.; Cipollini, P.; Couto, A.B.; Dingle, J.; Doerffer, R.; et al. An Ocean-Colour Time Series for Use in Climate Studies: The Experience of the Ocean-Colour Climate Change Initiative (OC-CCI). Sensors 2019, 19, 4285. [Google Scholar] [CrossRef]
- Akanda, A.S.; Jutla, A.S.; Gute, D.M.; Sack, R.B.; Alam, M.; Huq, A.; Colwell, R.R.; Islam, S. Population Vulnerability to Biannual Cholera Outbreaks and Associated Macro-Scale Drivers in the Bengal Delta. Am. J. Trop. Med. Hyg. 2013, 89, 950–959. [Google Scholar] [CrossRef]
- Bouma, M.J.; Pascual, M. Seasonal and interannual cycles of endemic cholera in Bengal 1891–1940 in relation to climate and geography. Hydrobiologia 2001, 460, 147–156. [Google Scholar] [CrossRef]
- CEOS EO HANDBOOK. Available online: http://eohandbook.com/sdg/ (accessed on 30 September 2019).
- Groom, S.; Sathyendranath, S.; Ban, Y.; Bernard, S.; Brewin, R.; Brotas, V.; Brockmann, C.; Chauhan, P.; Choi, J.; Chuprin, A.; et al. Satellite Ocean Colour: Current Status and Future Perspective. Front. Mar. Sci. 2019, 6, 485. [Google Scholar] [CrossRef]
- Donlon, C.; Berruti, B.; Buongiorno, A.; Ferreira, M.H.; Féménias, P.; Frerick, J.; Goryl, P.; Klein, U.; Laur, H.; Mavrocordatos, C.; et al. The Global Monitoring for Environment and Security (GMES) Sentinel-3 mission. Remote Sens. Environ. 2012, 120, 37–57. [Google Scholar] [CrossRef]
- Liardon, J.L.; Hostettler, L.; Zulliger, L.; Kangur, K.; Shaik, N.; Barry, D.A. Lake Imaging and Monitoring Aerial Drone. HardwareX 2018, 3, 146–159. [Google Scholar] [CrossRef]
- Wu, J.L.; Ho, C.R.; Huang, C.C.; Srivastav, A.L.; Tzeng, J.H.; Lin, Y.T. Hyperspectral Sensing for Turbid Water Quality Monitoring in Freshwater Rivers: Empirical Relationship between Reflectance and Turbidity and Total Solids. Sensors 2014, 14, 22670–22688. [Google Scholar] [CrossRef]
- Kisevic, M.; Morovic, M.; Andricevic, R. The use of hyperspectral data for evaluation of water quality parameters in the River Sava. Fresenius Environ. Bull. 2016, 25, 4814–4822. [Google Scholar]
- Bidigare, R.R.; Ondrusek, M.E.; Morrow, J.H.; Kiefer, D.A. In-vivo absorption properties of algal pigments. 1990, Volume 1302, pp. 290–302. Available online: https://www.researchgate.net/publication/252465392_In-vivo_absorption_properties_of_algal_pigments (accessed on 30 September 2019).
- Trees, C.C.; Clark, D.K.; Bidigare, R.R.; Ondrusek, M.E.; Mueller, J.L. Accessory pigments versus chlorophyll a concentrations within the euphotic zone: A ubiquitous relationship. Limnol. Oceanogr. 2000, 45, 1130–1143. [Google Scholar] [CrossRef]
- Sathyendranath, S.; Cota, G.; Stuart, V.; Maass, H.; Platt, T. Remote sensing of phytoplankton pigments: A comparison of empirical and theoretical approaches. Int. J. Remote Sens. 2001, 22, 249–273. [Google Scholar] [CrossRef]
- Brewin, R.J.W.; Sathyendranath, S.; Hirata, T.; Lavender, S.J.; Barciela, R.M.; Hardman-Mountford, N.J. A three-component model of phytoplankton size class for the Atlantic Ocean. Ecol. Model. 2010, 221, 1472–1483. [Google Scholar] [CrossRef]
- Brewin, R.J.W.; Dall’Olmo, G.; Sathyendranath, S.; Hardman-Mountford, N.J. Particle backscattering as a function of chlorophyll and phytoplankton size structure in the open-ocean. Opt. Express 2012, 20, 17632–17652. [Google Scholar] [CrossRef]
- Brewin, R.J.W.; Sathyendranath, S.; Jackson, T.; Barlow, R.; Brotas, V.; Airs, R.; Lamont, T. Influence of light in the mixed-layer on the parameters of a three-component model of phytoplankton size class. Remote Sens. Environ. 2015, 168, 437–450. [Google Scholar] [CrossRef]
- Brewin, R.J.W.; Morán, X.A.G.; Raitsos, D.E.; Gittings, J.A.; Calleja, M.L.; Viegas, M.; Ansari, M.I.; Al-Otaibi, N.; Huete-Stauffer, T.M.; Hoteit, I. Factors Regulating the Relationship Between Total and Size-Fractionated Chlorophyll-a in Coastal Waters of the Red Sea. Front. Microbiol. 2019, 10, 1964. [Google Scholar] [CrossRef]
- Alvain, S.; Moulin, C.; Dandonneau, Y.; Bréon, F.M. Remote sensing of phytoplankton groups in case 1 waters from global SeaWiFS imagery. Deep Sea Res. Part Oceanogr. Res. 2005, 52, 1989–2004. [Google Scholar] [CrossRef]
- Alvain, S.; Moulin, C.; Dandonneau, Y.; Loisel, H. Seasonal distribution and succession of dominant phytoplankton groups in the global ocean: A satellite view. Glob. Biogeochem. Cycles 2008, 22. [Google Scholar] [CrossRef]
- Alvain, S.; Loisel, H.; Dessailly, D. Theoretical analysis of ocean color radiances anomalies and implications for phytoplankton groups detection in case 1 waters. Opt. Express 2012, 20, 1070–1083. [Google Scholar] [CrossRef] [PubMed]
- Rêve-Lamarche, A.H.; Alvain, S.; Racault, M.F.; Dessailly, D.; Guiselin, N.; Jamet, C.; Vantrepotte, V.; Beaugrand, G. Estimation of the Potential Detection of Diatom Assemblages Based on Ocean Color Radiance Anomalies in the North Sea. Front. Mar. Sci. 2017, 4, 408. [Google Scholar] [CrossRef]
- Sathyendranath, S.; Watts, L.; Devred, E.; Platt, T.; Caverhill, C.; Maass, H. Discrimination of diatoms from other phytoplankton using ocean-colour data. Mar. Ecol. Prog. Ser. 2004, 272, 59–68. [Google Scholar] [CrossRef]
- Racault, M.F.; Le Quéré, C.; Buitenhuis, E.; Sathyendranath, S.; Platt, T. Phytoplankton phenology in the global ocean. Ecol. Indic. 2012, 14, 152–163. [Google Scholar] [CrossRef]
- Racault, M.F.; Raitsos, D.E.; Berumen, M.L.; Brewin, R.J.W.; Platt, T.; Sathyendranath, S.; Hoteit, I. Phytoplankton phenology indices in coral reef ecosystems: Application to ocean-color observations in the Red Sea. Remote Sens. Environ. 2015, 160, 222–234. [Google Scholar] [CrossRef]
- Racault, M.F.; Sathyendranath, S.; Menon, N.; Platt, T. Phenological Responses to ENSO in the Global Oceans. Surv. Geophys. 2017, 38, 277–293. [Google Scholar] [CrossRef]
- Cole, H.S.; Henson, S.; Martin, A.P.; Yool, A. Basin-wide mechanisms for spring bloom initiation: How typical is the North Atlantic? ICES J. Mar. Sci. 2015, 72, 2029–2040. [Google Scholar] [CrossRef]
- Sallée, J.B.; Llort, J.; Tagliabue, A.; Lévy, M. Characterization of distinct bloom phenology regimes in the Southern Ocean. ICES J. Mar. Sci. 2015, 72, 1985–1998. [Google Scholar] [CrossRef]
- Platt, T.; Sathyendranath, S. Oceanic Primary Production: Estimation by Remote Sensing at Local and Regional Scales. Science 1988, 241, 1613–1620. [Google Scholar] [CrossRef]
- Sathyendranath, S.; Stuart, V.; Nair, A.; Oka, K.; Nakane, T.; Bouman, H.; Forget, M.H.; Maass, H.; Platt, T. Carbon-to-chlorophyll ratio and growth rate of phytoplankton in the sea. Mar. Ecol. Prog. Ser. 2009, 383, 73–84. [Google Scholar] [CrossRef]
- Martinez-Vicente, V.; Dall’Olmo, G.; Tarran, G.; Boss, E.; Sathyendranath, S. Optical backscattering is correlated with phytoplankton carbon across the Atlantic Ocean. Geophys. Res. Lett. 2013, 40, 1154–1158. [Google Scholar] [CrossRef]
- Kostadinov, T.S.; Milutinović, S.; Marinov, I.; Cabré, A. Carbon-based phytoplankton size classes retrieved via ocean color estimates of the particle size distribution. Ocean Sci. 2016, 12, 561–575. [Google Scholar] [CrossRef]
- Roy, S.; Sathyendranath, S.; Platt, T. Size-partitioned phytoplankton carbon and carbon-to-chlorophyll ratio from ocean colour by an absorption-based bio-optical algorithm. Remote Sens. Environ. 2017, 194, 177–189. [Google Scholar] [CrossRef]
- Dogliotti, A.I.; Ruddick, K.G.; Nechad, B.; Doxaran, D.; Knaeps, E. A single algorithm to retrieve turbidity from remotely-sensed data in all coastal and estuarine waters. Remote Sens. Environ. 2015, 156, 157–168. [Google Scholar] [CrossRef]
- Stramski, D.; Reynolds, R.A.; Babin, M.; Kaczmarek, S.; Lewis, M.R.; Röttgers, R.; Sciandra, A.; Stramska, M.; Twardowski, M.S.; Franz, B.A.; et al. Relationships between the surface concentration of particulate organic carbon and optical properties in the eastern South Pacific and eastern Atlantic Oceans. Biogeosciences 2008, 5, 171–201. [Google Scholar] [CrossRef]
- Evers-King, H.; Martinez-Vicente, V.; Brewin, R.J.W.; Dall’Olmo, G.; Hickman, A.E.; Jackson, T.; Kostadinov, T.S.; Krasemann, H.; Loisel, H.; Röttgers, R.; et al. Validation and Intercomparison of Ocean Color Algorithms for Estimating Particulate Organic Carbon in the Oceans. Front. Mar. Sci. 2017, 4, 251. [Google Scholar] [CrossRef]
- Gordon, H.R.; Boynton, G.C.; Balch, W.M.; Groom, S.B.; Harbour, D.S.; Smyth, T.J. Retrieval of coccolithophore calcite concentration from SeaWiFS Imagery. Geophys. Res. Lett. 2001, 28, 1587–1590. [Google Scholar] [CrossRef]
- Balch, W.M.; Gordon, H.R.; Bowler, B.C.; Drapeau, D.T.; Booth, E.S. Calcium carbonate measurements in the surface global ocean based on Moderate-Resolution Imaging Spectroradiometer data. J. Geophys. Res. Oceans 2005, 110. [Google Scholar] [CrossRef]
- Smyth, T.J.; Tyrrell, T.; Tarrant, B. Time series of coccolithophore activity in the Barents Sea, from twenty years of satellite imagery. Geophys. Res. Lett. 2004, 31. [Google Scholar] [CrossRef]
- Moore, T.S.; Dowell, M.D.; Franz, B.A. Detection of coccolithophore blooms in ocean color satellite imagery: A generalized approach for use with multiple sensors. Remote Sens. Environ. 2012, 117, 249–263. [Google Scholar] [CrossRef]
- Lee, Z.; Carder, K.L.; Arnone, R.A. Deriving inherent optical properties from water color: A multiband quasi-analytical algorithm for optically deep waters. Appl. Opt. 2002, 41, 5755–5772. [Google Scholar] [CrossRef] [PubMed]
- Maritorena, S.; Siegel, D.A.; Peterson, A.R. Optimization of a semianalytical ocean color model for global-scale applications. Appl. Opt. 2002, 41, 2705–2714. [Google Scholar] [CrossRef] [PubMed]
- Werdell, P.J.; Franz, B.A.; Bailey, S.W.; Feldman, G.C.; Boss, E.; Brando, V.E.; Dowell, M.; Hirata, T.; Lavender, S.J.; Lee, Z.; et al. Generalized ocean color inversion model for retrieving marine inherent optical properties. Appl. Opt. 2013, 52, 2019–2037. [Google Scholar] [CrossRef] [PubMed]
- Doney, S.C. Plankton in a warmer world. Nature 2006, 444, 695–696. [Google Scholar] [CrossRef]
- Ali, M.; Nelson, A.R.; Lopez, A.L.; Sack, D.A. Updated Global Burden of Cholera in Endemic Countries. PLoS Negl. Trop. Dis. 2015, 9, e0003832. [Google Scholar] [CrossRef]
- Scharlemann, J.P.W.; Mant, R.C.; Balfour, N.; Brown, C.; Burgess, N.D.; Guth, M.; Ingram, D.J.; Lane, R.; Martin, J.; Wicander, S.; et al. Global Goals Mapping: The Environment-Human Landscape. Available online: http://www.nerc.ac.uk/research/partnerships/international/overseas/tase/mapping/ (accessed on 30 September 2019).
- Brewin, R.J.W.; Brewin, T.G.; Phillips, J.; Rose, S.; Abdulaziz, A.; Wimmer, W.; Sathyendranath, S.; Platt, T. A Printable Device for Measuring Clarity and Colour in Lake and Nearshore Waters. Sensors 2019, 19, 936. [Google Scholar] [CrossRef]
- Akanda, A.S.; Aziz, A.; Jutla, A.; Huq, A.; Alam, M.; Asham, G.U.; Colwell, R.R. Satellites and Cell Phones Form a Cholera Early-Warning System. Eos Trans. Am. Geophys. Union 2018, 99. [Google Scholar] [CrossRef]
- Finger, F.; Genolet, T.; Mari, L.; de Magny, G.C.; Manga, N.M.; Rinaldo, A.; Bertuzzo, E. Mobile phone data highlights the role of mass gatherings in the spreading of cholera outbreaks. Proc. Natl. Acad. Sci. USA 2016, 113, 6421–6426. [Google Scholar] [CrossRef]
- Abubakar, A.; Azman, A.S.; Rumunu, J.; Ciglenecki, I.; Helderman, T.; West, H.; Lessler, J.; Sack, D.A.; Martin, S.; Perea, W.; et al. The First Use of the Global Oral Cholera Vaccine Emergency Stockpile: Lessons from South Sudan. PLoS Med. 2015, 12, e1001901. [Google Scholar] [CrossRef]
| Predicted variable | Covariate | Method | Study Region and Period | Satellite Sensor or Product | Reference |
|---|---|---|---|---|---|
| Vibrio parahaemolyticus densities in oyster meat | RS: SST, Chlorophyll-a Other: salinity | Multilinear regression model | British Columbia, Canada; 2003–2015 | Multi-scale Ultra-high-resolution SST Analysis, MODIS | [121] |
| Cholera incidence | RS: SST, SSH, Chlorophyll-a Other: precipitation, temperature | Macro environment–SIR model and multilinear regression analysis of environmental drivers | Zhejiang province, China; 2001–2008 | AVHRR, TOPEX/Poseidon and Jason-1, SeaWiFS | [47] |
| Cholera incidence | RS: SST, SSH, Chlorophyll-a | Multilinear regression model | China; 1999–2008 | AVHRR TOPEX/Poseidon and Jason-1, SeaWiFS | [122] |
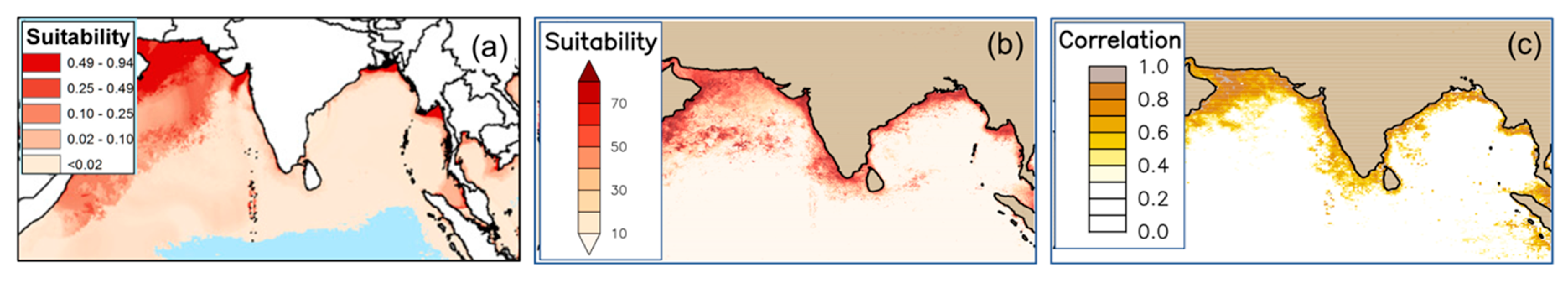
| Vibrio cholerae habitat suitability index | RS: Chlorophyll-a, SST, Photosynthetically Available Radiation Other: salinity, pH, O2, nitrate, phosphate | Ecological niche model | Global oceans; 2005–2010 | MODIS aqua, SeaWiFS | [54] |
| Cholera incidence | RS: Chlorophyll-a, precipitation Other: ENSO and DMI climate indices | Inhomogeneous Markov Chain model | Lake Kivu region, DRC; 2002–2012 | MODIS aqua, TRMM | [119] |
| Cholera incidence | RS: reflectance at 412 and 555 nm | Satellite Water Marker (SWM) model based on RS reflectance | Bay of Bengal, Mozambique Channel; 1998–2009 | SeaWiFS | [123] |
| Cholera incidence | RS: Chlorophyll-a | Correlation analyses | Bay of Bengal coastal and offshore; 1998–2007 | SeaWiFS | [124] |
| Cholera incidence | RS: Chlorophyll-a Other: precipitation, fishing activities | Time-series and correlation analyses | Lake Kivu region, DRC; 2002–2006 | MODIS aqua | [125] |
| Cholera incidence | RS: SST, SSH, Chlorophyll-a Other: socio-economic status | Generalized linear model | Bangladesh, Bay of Bengal; 2003–2007 | AVHRR, Jason-1, SeaWiFS | [126] |
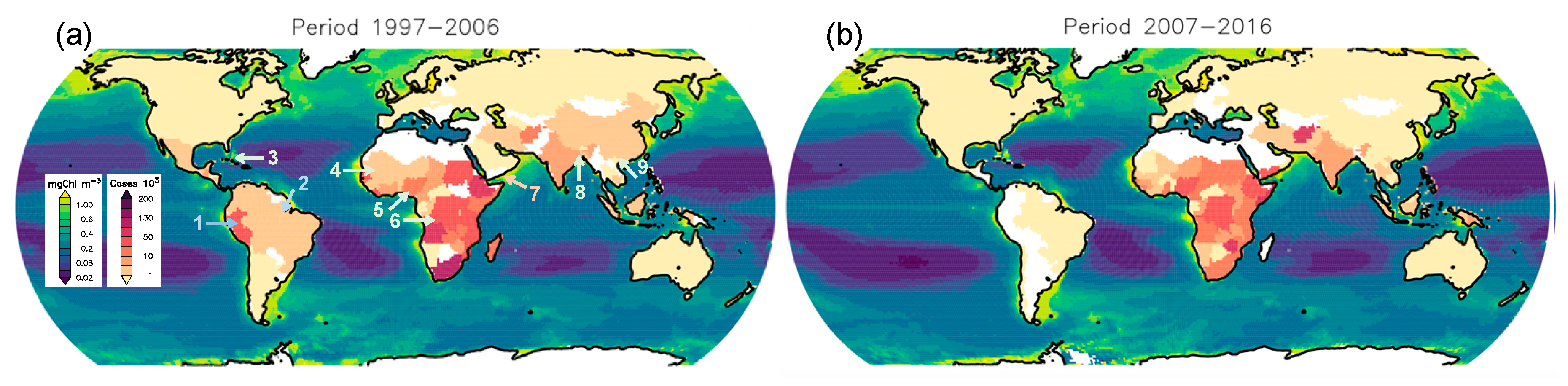
| Cholera incidence | RS: Chlorophyll-a Other: river discharge data | Analysis of annual variations | Bay of Bengal, Mozambique Channel; 1997–2010 | SeaWiFS | [6] |
| Cholera incidence | RS: SST, SSH, (Chlorophyll-a) | Correlation analyses | Bangladesh, Bay of Bengal; 1992–1995 and 1997–1998 | AVHRR, TOPEX/Poseidon, (SeaWiFS) | [116] |
| Cholera incidence | RS: SST, SSH, Chlorophyll-a | Analysis of interannual variations | Bangladesh, Bay of Bengal; 1998–2002 | AVHRR, TOPEX/Poseidon, SeaWiFS | Colwell and Calkins, unpub. data reported in [16,127] |
| Cholera incidence | RS: SST, Chlorophyll-a, precipitation | Generalized linear model | Bangladesh, Matlab, India, Kolkata, Bay of Bengal; 1998–2006 | NOAA Optimally Interpolated product, SeaWiFS, Global Precipitation Climatology Project (GPCP) | [44] |
| Cholera incidence | RS: SST, SSH, Chlorophyll-a, precipitation | Linear regression analyses | KwaZulu-Natal, South Africa; 2000–2001 | AVHRR, Topex/Poseidon, SeaWiFS, Merge of infrared and microwave satellite estimates with rain gauge data (GPCP) | [128] |
| Cholera incidence | RS: SST, SSH, Chlorophyll-a Other: Precipitation, temperature, river discharge or height | Multivariate regression analyses | Matlab, Bangladesh, Hue, Vietnam; 1997–2003 | AVHRR, TOPEX/Poseidon and Jason-1, SeaWiFS | [48] |
| Vibrio parahaemolyticus densities in oyster meat | RS: SST, turbidity, Chlorophyll-a Other: salinity, bottom water temperature | Regression analyses | Alabama coastal region, USA; 1999–2000 | AVHRR, SeaWiFS | [129] |
| Application | Status | Environmental Interaction | Reservoir | Ocean-Color Product | Ocean-Color Product Reference |
|---|---|---|---|---|---|
| Reservoir distribution and cholera outbreaks | Realized (references in Table 1) | Host–pathogen | Phytoplankton biomass | Total chlorophyll-a | [139,140,141] |
| Reservoir distribution and cholera outbreaks | Realized Jutla et al., 2013 | Host–pathogen | Phytoplankton biomass and suspended matter | Satellite Water Marker based on Rrs | [123] |
| Reservoir distribution | Potential | Host–pathogen | Phytoplankton size structure | Micro-, nano-, picohytoplankton chlorophyll-a | [142,143,144,145] |
| Reservoir distribution | Potential | Host–pathogen | Phytoplankton functional groups | Diatoms, Coccolithophores, Phaeocystis, Prochlorococcus, Synechoccus, Nano-Eukaryotes | [146,147,148,149] |
| Reservoir distribution | Potential | Host–pathogen | Chitin-containing phytoplankton | Diatoms | [150] |
| Reservoir temporal dynamics | Potential | Host–pathogen | Phytoplankton phenology | Timings of initiation, peak, termination, and duration | [151,152,153,154,155] |
| Biogeochemical cycles regulation | Potential | Carbon cycle | Phytoplankton organic carbon productivity | Primary production | [156] |
| Biogeochemical cycles regulation | Potential | Carbon cycle | Phytoplankton carbon | Total phytoplankton carbon | [157,158,159,160] |
| Biogeochemical cycles regulation | Potential | Carbon cycle | Phytoplankton carbon | Micro-, nano-, picohytoplankton carbon | [159,160] |
| Reservoir distribution | Potential | Host–pathogen | Suspended matter | Turbidity, total suspended matter | [161] |
| Biogeochemical cycles regulation | Potential | Nutrients, Carbon cycle | Particulate organic matter | Particulate organic carbon | [162,163] |
| Biogeochemical cycles regulation | Potential | Carbon cycle | Particulate inorganic matter | Particulate inorganic carbon | [164,165] |
| Biogeochemical cycles regulation | Potential | Carbon cycle | Carbon export, acidification | Coccolithophores | [166,167] |
| Biogeochemical cycles regulation | Potential | Nutrients, Carbon cycle | Dissolved organic matter | Colored dissolved organic matter | [168,169,170] |
| Goal | Target | Support from Satellite Applications | Indicator of Impact |
|---|---|---|---|
| 3-Health | [3.3] Combat epidemics of water-borne diseases; [3.d] Strengthen capacity for early warning, risk reduction and management of national and global health risks | - Improve knowledge on the transmission patterns of the Vibrio cholerae pathogens in affected countries → the information will be useful for health services to help provide the most adapted and efficient treatment for the affected local populations, and to increase chances of recovery; - Improve surveillance systems and strengthen the capacity of affected countries to produce early warning and risk maps for cholera outbreaks → the improved/local/regional forecast systems may be delivered to national agencies for operational use and the risk warning may be placed in the public domain to reduce public health risks. | [3.3.5] a reduction in the number of people requiring treatment against Vibrio disease; [3.9.2] a reduction in mortality rate associated with exposure to contaminated water; [3.d.1] an increase in capacity for disaster mitigation in affected areas. |
| 6-Water quality | [6.3] Improve water quality by reducing pollution, halving the proportion of untreated wastewater; [6.5] Implement integrated water resource management | - Provide evidence on Vibrio disease hotspots → this information will help government authorities to identify areas where microbial and antibiotic pollution should be treated as a priority. - Improve knowledge of the transmission routes and dynamics of cholera pathogens in the lacustrine and coastal ecosystems in relation to climate variability and disease outbreaks → this information will support increased preparedness along the transmission routes, and integration of locally-targeted water sanitation control measures that will help to interrupt the transmission routes. | [6.b.1] an increase in the number of actions taken by local administrations to treat sources of pollution, and to engage the local communities in water and sanitation management. |
| 13-Climate | [13.1] Strengthen resilience and adaptive capacity to climate-related hazards and natural disasters | - Improve knowledge of the influence of extreme weather events and climate variability on the incidence of cholera outbreaks and the contamination routes of Vibrio pathogens → this will help to prioritize policy measures for health service preparedness and population awareness when high-risk climate events occur, and hence improve cholera-disaster risk mitigation. | [13.3.1] an increase in the number of countries that recognize the need to integrate climate-related risk in their early warning systems for cholera outbreaks, and in their adaptation and mitigation plans (to reduce impact and risk of cholera outbreaks). |
| 14-Marine Life | [14.1] Prevent and significantly reduce marine pollution of all kinds; [14.2] Sustainably manage and protect marine and coastal ecosystems to avoid significant adverse impacts and achieve healthy and productive oceans | - Provide evidence on the extent to which lacustrine and coastal communities are suffering from Vibrio diseases → this information can be used to support the development of sustainable management plans. | [14.c.1] an increase in the number of countries that incorporate ecosystem-based management in their stewardship of coastal ecosystems and their resources. |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Racault, M.-F.; Abdulaziz, A.; George, G.; Menon, N.; C, J.; Punathil, M.; McConville, K.; Loveday, B.; Platt, T.; Sathyendranath, S.; et al. Environmental Reservoirs of Vibrio cholerae: Challenges and Opportunities for Ocean-Color Remote Sensing. Remote Sens. 2019, 11, 2763. https://doi.org/10.3390/rs11232763
Racault M-F, Abdulaziz A, George G, Menon N, C J, Punathil M, McConville K, Loveday B, Platt T, Sathyendranath S, et al. Environmental Reservoirs of Vibrio cholerae: Challenges and Opportunities for Ocean-Color Remote Sensing. Remote Sensing. 2019; 11(23):2763. https://doi.org/10.3390/rs11232763
Chicago/Turabian StyleRacault, Marie-Fanny, Anas Abdulaziz, Grinson George, Nandini Menon, Jasmin C, Minu Punathil, Kristian McConville, Ben Loveday, Trevor Platt, Shubha Sathyendranath, and et al. 2019. "Environmental Reservoirs of Vibrio cholerae: Challenges and Opportunities for Ocean-Color Remote Sensing" Remote Sensing 11, no. 23: 2763. https://doi.org/10.3390/rs11232763
APA StyleRacault, M.-F., Abdulaziz, A., George, G., Menon, N., C, J., Punathil, M., McConville, K., Loveday, B., Platt, T., Sathyendranath, S., & Vijayan, V. (2019). Environmental Reservoirs of Vibrio cholerae: Challenges and Opportunities for Ocean-Color Remote Sensing. Remote Sensing, 11(23), 2763. https://doi.org/10.3390/rs11232763










