Abstract
Chlorophyll fluorescence parameters can provide useful indications of photosynthetic performance in vivo. Coupling appropriate fluorescence measurements with other noninvasive techniques, such as absorption spectroscopy or gas exchange, can provide insights into the limitations to photosynthesis under given conditions. Chlorophyll content is one of the dominant factors influencing the conditions of a vegetation growing season, and can be tested using both fluorescence and remote sensing methods. Hyperspectral remote sensing and recording the narrow range of the spectrum can be used to accurately analyze the parameters and properties of plants. The aim of this study was to analyze the influence of lead ions (Pb, 5 mM Pb(NO3)2) on the growth of pea plants using spectral properties. Hyperspectral remote sensing and chlorophyll fluorescence measurements were used to assess the physiological state of plants seedlings treated by lead ions during the experiment. The plants were growing in hydroponic cultures supplemented with Pb ions under various conditions (control, complete Knop + phosphorus (+P); complete Knop + phosphorus (+P) + Pb; Knop (-P) + Pb, distilled water + Pb) affecting lead uptake via the root system. Spectrometric measurements allowed us to calculate the remote sensing indices of vegetation, which were compared with chlorophyll and carotenoids content and fluorescence parameters. The lead contents in the leaves, roots, and stems were also analyzed. Spectral characteristics and vegetation properties were analyzed using statistical tests. We conclude that: (1) pea seedlings grown in complete Knop (with P) and in the presence of Pb ions were spectrally similar to the control plants because lead was not transported to the shoots of plants; (2) lead most influenced plants that were grown in water, according to the highest lead content in the leaves; and (3) the effects of lead on plant growth were confirmed by remote sensing indices, whereas fluorescence parameters identified physiological changes induced by Pb ions in the plants.
1. Introduction
The use of chlorophyll a fluorescence measurements to examine stress in algae and plants is now widespread in physiological and ecophysiological studies [1]. Fluorescence can be a powerful tool to study photosynthetic performance, especially when coupled with other noninvasive measurements such as absorption spectroscopy, gas analyses, and infrared thermometry. Many environmental stresses affect CO2 assimilation. Changes in fluorescence induced by illumination of dark-adapted leaves are qualitatively correlated with their photosynthetic rates [2,3]. Photosynthetic CO2 fixation is a process significantly affected by heavy metals in a number of plant species [4]. The mechanism(s) of heavy metal toxicity on photosynthesis is still a matter of speculation, but it almost certainly involves electron transport in light reactions [5] and enzyme activity in the dark reactions [6].
The ions determine the proper development and functioning of organisms [2,3], but they can also cause stress in plant cells, which produces disturbances in the functioning of processes including, among others, photosynthesis [4,7]. Burzynski and Kłobus [8] reported the inhibitory effect of metals on growth and biomass of plants as well as changes in the metabolic processes, which reduce photosynthetic activity. The effects are dependent on the operation of the ion, its concentration and duration of action, and the environment in which the plant grows [9]. The most damaging heavy metals (HMs) are cadmium (Cd), lead (Pb), mercury (Hg), nickel (Ni), and arsenic (As). Pb causes a reduction in chlorophyll content [10] and assimilation of CO2, and inhibits the respiratory intensity [10,11]. Pb also affects the absorption of ions from the soil including, among others, iron and magnesium [12,13]. Pb is known to induce a broad range of toxic effects, including those that are morphological, physiological, and biochemical in origin. This metal impairs plant growth, root elongation, seed germination, seedling development, transpiration, chlorophyll production, and cell division [14]. A high Pb content in plants thus contributes to a reduction in the water absorption capacity [12]. Heavy metal ions also affect the formation of reactive oxygen species (ROS), which cause changes in signal transduction, change the properties of the membranes, and affect gene expression, damaging photosystem II (PSII) [15,16]. Stress-induced decreases in stomatal conductance, carbon metabolism, and transport processes can all decrease PSII efficiency [7]. Changes in chlorophyll a fluorescence measured in vivo enables the estimation of the physiological state of the plant and illustrates how absorbed light energy is used.
Stress induces changes in light absorption and can result in loss of excitation energy in the form of heat (non-photochemical quenching, NPQ) [17]. NPQ protects the photosynthetic membrane against photodamage, leading to early senescence and reduced plant growth and fitness [18]. NPQ is assume to be zero in the dark adapted state, because Fv’ = Fv and Fm’ = Fm. When plants are exposed to light, we can estimate the non-photochemical quenching from maximal fluorescence with an adaptation to darkness (Fm) to maximal fluorescence without adaptation to darkness (Fm′) and monitors the apparent rate constant for non-radiative decay (heat loss) from PSII and its antennae for understanding the protective action of NPQ [19]. Plants have various responses to the noxious effects of lead, such as selective metal uptake, metal binding to the root surface, metal binding to the cell wall, and induction of antioxidants. The published findings on the evaluation of photochemical processes and physiological do not allow for the unambiguous determination of the response of photosynthetic apparatus to stress caused by lead [14,20,21]. Pb was found to induce changes in the absorption and dissipation of energy within PSII, whereas small changes were observed in electron transport [22]. Heavy metals, such as cadmium, nickel, and lead, are phytotoxic [23]. The literature also confirms a variable response to Pb as a stress factor, depending on the species. For example, corn and soybean showed varying degrees of photosynthesis sensitivity to lead [24].
Remote and non-invasive detection of changes in vegetation due to contamination with lead is possible using remote sensing techniques (spectrometric measurements, and multispectral and hyperspectral imaging). Using hyperspectral techniques is becoming increasingly important in the study of vegetation stress [25,26]. In response to the HMs presence, remote sensing indices show a decrease in photosynthetic active radiation (PAR), chlorophyll and carotenoids contents, a shift of red-edge spectral bands to the visible part of the spectrum (VIS), and changes in plant-water accumulation in short-wave infrared (SWIR). A limited growth, changes of canopy structures, decrease of chlorophyll content (leaf chlorosis), and changes in cell water content is a consequence of pollution [27].
The development of technologies and retrieval algorithms to evaluate fluorescence has progressed with model developments [28,29,30]. The state of vegetation contaminated with heavy metals may be tested using remote sensing indices [31] and by changes in the spectral reflectance curves, mainly the red edge [32]. The spectral reflectance and thereby the relevant spectral features offer an easy and simple method to assess plant health and metal-concentration. ASD FieldSpec was used for monitoring the phytoaccumulation of arsenic into above-ground parts of barley [33] and paddy plants (measurement of field spectral reflectance was one basis of airborne or spaceborne remote sensing monitoring). Ren et al. [34] also studied these spectral changes to relate them with the phytoaccumulation of metals [35,36]. Most of the studies used the various vegetation indices (ratios or linear combinations of two or more spectral wavelengths [37]) and red-edge position (a sharp transition between red and NIR wavelengths; positively related to chlorophyll concentration [38]) as indications of plant stress. The impact of heavy metals was observed in the spectral reflectance curve, where, under the influence of, for example Cd and Ni [39], the red edge decreased. Zagajewski et al. [39] observed that stressed plants (expressed as lower chlorophyll content) showed an increase in the fluorescence at F690 (red) and a decrease in the intensity of fluorescence at F740 (far-red) with decreasing intensity at 710 nm. Different fluorescence features are affected by leaf chlorophyll content in the antenna and the F690/F740 ratio appears to be one of the most indicative of chlorophyll variations [40]. Changes in the state of vegetation caused by the impact of heavy metals were also observed in multispectral images, where decreases in crop biomass and chlorophyll content were observed, as were changes in the red edge during the measurements [31]. Many observations indicate that the interactions between chlorophyll and carotenoids seem to be crucial for excess energy dissipation [41]. Remote sensing technologies provide complementary capacity for measuring and interpreting fluorescence in the context of physiological processes in field- [42,43], airborne- [44,45], and satellite-level measurements [46].
Despite extensive studies conducted on the effects of various heavy metals on different photosynthetic parameters of plants, our knowledge is still incomplete. Various changes in physiological and biochemical events occurring at different sites during photosynthesis are tightly linked so that even slight inhibition of one may result in a series of processes limiting gas exchange in the plants. Fluorescence assessment in the laboratory or growth chamber involves a suite of measurement devices, including fluorescence microscopes, spectrophotometers spectrofluorometers, fluorometers, and others. These have allowed studies on scales ranging from isolated photosystems to small vegetation canopies. Many researchers underlined that in the future, laboratory-scale or controlled-environment trials can support sun-induced fluorescence (SIF) remote sensing activities, such as by the elucidation of confounding factors for interpreting SIF changes, or by identifying ancillary data types needed for airborne- or space-based missions [47].
The aim of this study was to combine devices, such as a fluorometer and spectrometer, for detection of the influence of lead on photosynthetic parameters in pea plants (Pisum sativum) growing in laboratory conditions. Laboratory and field experiments provide new insights into fluorescence–photosynthesis linkages, stress effects, and relationships between other photosynthetic parameters. We investigated the relationship between fluorescence parameters: the maximum quantum efficiency of PSII in the light, measured as a Fv’/Fm’ ratio (variable fluorescence without adaptation (Fv’) to maximal fluorescence without adaptation to darkness (Fm′)) and non-photochemical quenching of fluorescence (NPQ) and hyperspectral remote sensing indices. We also report the effect of lead ions on the growth of plants in various conditions influencing Pb uptake from the root medium. These data provide a novel insight into the relationships between the methods used for measurements of heavy metal effects on the plants.
2. Materials and Methods
2.1. Plant Material
Pea (P. sativum L.) seedlings were grown hydroponically in aerated:
- (1)
- Knop’s solution [1] containing (g·L−1) 0.8 CaNO3·4H2O, 0.2 KNO3, 0.2 KH2PO4, 0.2 MgSO4·7H2O, and 0.028 EDTA-Fe (EthyleneDiamineTetraacetic) enriched with A–Z microelement nutrients (Control);
- (2)
- Knop solution [1] with 5 mM Pb(NO3)2 (lead-treated plants; Knop + Pb);
- (3)
- Knop solution [1] without phosphorus with 5mM Pb(NO3)2 (described below as Knop (-P) +Pb);
- (4)
- distilled water with 5 mM Pb(NO3)2 (described below as H2O + Pb).
Plants were grown in a growth chamber with a 14 h photoperiod and a day/night regime at 25/20 °C at an irradiance of 50 µmol photons·m−2·s−1 (Figure 1). Pb ions (5 mM Pb(NO3)2) were added to the growth medium (see above) when seedlings were two weeks old. Lead was introduced into the leaves via the transpiration stream. The leaves from three-week-old plants were used for experiments.

Figure 1.
Pea plants used in experiments. From left to right: control plants in Knop’s solution; Knop medium without phosphorus; Knop solution with 5 mM Pb(NO3)2 (lead-treated plants, Pb); and distilled water with 5 mM Pb(NO3)2 (photo: M. Kycko).
2.2. In Vivo Measurements of Chlorophyll a Fluorescence
Chlorophyll a fluorescence of pea leaves was measured at room temperature with a fluorometer (FMS 1, Hansatech, Norfolk U.K.) run by Modfluor software (Hansatech, Norfolk U.K.). Leaves were adapted to darkness for 30 minutes prior to measurements, and then were used in these assays with an actinic radiation of 60–1100 μmol photons·m−2s−1 and the saturation radiation of 4500 μmol photons·m−2s−1. The standard amber modulating beam had a center frequency of 594 nm. The photochemical quenching coefficient (qP), non-photochemical quenching (NPQ), and quantum efficiency of PSII electron transport in the light (Fv’/Fm’) were measured at steady-state photosynthesis. The procedure developed by Genty at al. [48] was followed. All results are represented as means ± standard error (SE) from 8 independent series of experiments (10 measurements each).
2.3. Determination of Lead Content
After harvesting, leaves, stalks, and roots were washed with redistilled water, then samples were oven-dried at 85 °C and ground to powder. The powder was washed in a muffle furnace at 550 °C for 4 h and the residue was brought to a standard volume with 1 M HNO3. The Pb concentration of the extract was determined by an Atomic Absorption Spectrophotometer (AAS; PerkinElmer, model 3300; PerkinElmer, Waltham, MA, USA). The Pb ion content, expressed in mg·g−1 dry weight of the tissue tested, was determined on the basis of indications obtained from the corrected concentration of lead using Equation (1):
where
A = Cs × V/m
- A is the Pb ion content in mg·g−1 dry weight,
- Cs is the corrected concentration Pb (mg·L−1),
- V is the volume of the sample for analysis (L), and
- m is the dry weight of a sample (g).
2.4. Laboratory Measurements
To determine the effect of lead on pea plants, we measured the spectral characteristics of the plant vegetation with a ASD FieldSpec 3 spectrometer (ASD Inc., Longmont, CO, USA) with the ASD PlantProbe (ASD Inc., Longmont, CO, USA), and chlorophyll a fluorescence using a Hansatech FS-1 fluorometer (Hansatech, Norfolk U.K.); and the growth parameters including lengths and fresh and dry weights of the seedlings. On the basis of the dry weight, we determined the water content in plants and the lead content in the leaves by AAS.
Several randomly chosen pea seedlings, including control and plants grown in the presence of lead in different types of hydroponic medium, were used for measurements. The measurements were recorded using the contact probe (ASD PlantProbe) on fragments of the tested pea plant for each growth environment. The spectral characteristics of the plants were registered in the range from 350 to 2500 nm. The study consisted of calibrating the spectrometer (25 independent measurements per calibration) using a white pattern of spectralon (SG 33,151 Zenith Lite Reflectance Target and calibration screen P/N A122634 Leaf clip) and appropriate measurements of vegetation (the reported value of one record is the average of 25 measurements). Table 1 lists the measurements recorded for every group of plants.

Table 1.
ASD FieldSpec spectrometric measurements for control and lead-treated plants.
The data obtained from spectrometric measurements were saved in *.asd format and then exported to the ASD FieldSpec View in *.txt ASCII format and imported into an Excel spreadsheet and the Statistica 13 software (TIBCO Software Inc., Palo Alto, CA, USA) for statistical analyses. The reflectance values were averaged and the standard deviations were computed. For particular research elements, spectral reflectance curves were drawn. The program also calculated the selected remote sensing vegetation indices:
- (1)
- The content and structure of chlorophyll: modified normalized difference vegetation index 705 (mNDVI705) [49], red edge position index (REPI) [50], ratio analysis of reflectance spectra—chlorophyll a (RARSa) [51], RARS—chlorophyll b (RARSb) [51], and RARS—carotenoids (RARSc) [51];
- (2)
- The amount of light used in photosynthesis: structural independent pigment index (SIPI) [52];
- (3)
- Nitrogen content: normalized difference nitrogen index (NDNI) [53];
- (4)
- Amount of carbon: normalized difference lignin index (NDLI) [53] and plant senescence reflectance index (PSRI) [54];
- (5)
- Amount of carotenoids: carotenoid reflectance index (CRI 1) [55], anthocyanin reflectance index (ARI 1) [56], red/green ratio (RGR), and anthocyanins/chlorophyll [57]; and
- (6)
- Water content of the plant cover: normalized difference water index (NDWI) [58], water index (WI) [59], normalized water index-2 (NWI-2) [60], and disease water stress index (DSWI) [61].
Then, to determine the overall physiological activity, changes in the functioning photosynthetic parameters of the plants [62] and their condition [17], the chlorophyll fluorescence measurements were used. This measurement allows for the determination of the physiological state of photosynthetic apparatus on the basis of the energy used in photochemical reactions [62]. The leaf was exposed to a short pulse of high light (4500 µmol photons·m−2s−1): QA is the maximally reduced fluorescence level and the Fm is the maximal fluorescence level observed. On the basis of these two parameters, we calculated the maximum quantum efficiency of PSII (Fv/Fm) in plants adapted to darkness and after actinic light was used. Measurement cycles were performed using light intensities of 60, 120, 180, 240, 420, 600, 820 and 1020 µmol photons·(m−2s−1). We measured the fluorescence maximum (Fm’) for the plant adapted to relevant light conditions and components that decreased in fluorescence (NPQ).
The growth rate of seedlings (the length of roots and the stalk), fresh and dry weights of leaves, and the roots of four randomly selected plants from each of the investigated variants were measured (Figure 2). The dry weight was determined after drying plant material at 85 °C for 24 h until a constant weight was obtained.

Figure 2.
A plant treated with 5 mM Pb(NO3)2 on the left side and control pea plant on the right side (photo: I. Kaźmierczak).
The pigment contents in the leaves were measured in acetone. The leaves (approx. 0.1–0.2 g) were cut into small pieces and then mixed with a pestle in a mortar cooled to about 0 °C with the addition the purified sand and CaCO3 in 2 mL cold pure acetone. The extracts were filtered and washed with 80% acetone in a schott funnel (Schott AG, Mainz, Germany) and refilled with 80% acetone to a constant volume. Measurements were recorded with a UV-VIS 160A Shimadzu spectrophotometer (Shimadzu, Kyoto, Japan) at wavelengths of 663.2, 646.8, and 470 nm relative to 80% acetone as described by Arnon [49]. Pigment content was analyzed based on the fresh weight (FW) of the leaves (mg Chl/FW). The concentrations of the pigments (in μg/mL) was calculated with Equations (2)–(4) [38]:
Chlorophyll a = 12.25 × A663.2 − 2.79 × A646.8
Chlorophyll b = 21.5 × A646.8 − 5.1 × A663.2
Carotenoids = (1000 × A470 − 1.82 × Chl a − 85.02 × Chl b)/198
The statistical analyses were conducted basing on the Statistica 13 software and used included univariate ANOVA and Kruskal–Wallis one-way ANOVA by ranks. Univariate ANOVA for independent groups (called one-way ANOVA), proposed by Fisher [63], was used to verify the hypothesis of equality of means to test variables in several (k > 2) populations. The Kruskal–Wallis ANOVA is used for nonparametric data to study the effect of factors (independent variables) on the dependent variable [64]. To assess significant differences in the studied indices, we used the Mann–Whitney U test. This test is used to verify hypotheses about the insignificance of the differences between the variable medians studied in two populations; in this case, this was used to verify the differences between the controls and those treated with lead. The assessment of the significance of variance was performed at a significance level of 0.001. This analysis allowed the separation of statistically significant intervals of the spectrum and remote sensing vegetation indicators for the plants treated with lead. In the analysis, we considered all the samples and a statement of the trial with the trial of control. The characteristic spectral ranges are marked on the graphs of spectral reflection of plants.
3. Results
Lead toxicity on photosynthesis has often been examined with various species but rarely or not at all with ecotypes or populations all [65]. The generally accepted view is that heavy metals cause a reduction in photosynthesis in leaves primarily through induction of stomatal closure rather than a direct effect on light or dark reactions [20]. The lead treatment of pea plants resulted in statistically significant changes in the condition of the peas. The ANOVA performed for all the samples indicated that the presented ranges of the electromagnetic spectrum (Figure 3 and Figure 4) were statistically significant at the significance level of 0.001 (the most sensitive to the effects of heavy metals in the case of lead). In the figures, the marked grey ranges are the sensitive range for pigments and water content (the statistically significant changes depending on the medium in which the plants were grown: control; Knop + Pb; Knop (-P) + Pb; distilled water + Pb; at the p < 0.001). In the case of plants treated with the lead, the red-edge is noticeably, indicating damage to the cell structures of plants (Figure 3). In the plants growing in distilled water with lead, we perceived that the changes were noticed for 480–515 nm and 540–720 nm (responsible for absorption of light by chlorophyll. For Knop + Pb and control plants the following spectral ranges were significantly important: 710–735, 830–950, 995–1145, 1385–1570, 1760–1902, and 1980–2500 nm (responsible for water absorption). In contrast to ranges obtained for plants from Knop (-P) + Pb and control plants, also 850–1400, 1570–1760 and 1800–1830 nm indicated water absorption features (Figure 4). The differences occurred in the ranges: 714–723, 858–951, 995–1145, 1386–1389, 1570–1579, and 1801–1829 nm (marked with a black dashed line in Figure 4).
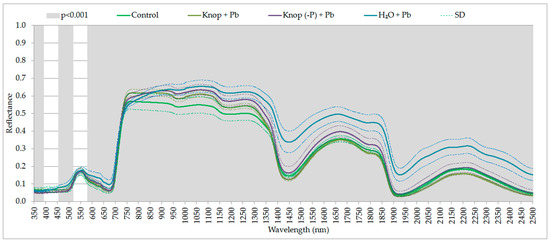
Figure 3.
The statistically significant spectral ranges (p < 0.001) for the different treatments of pea plants at different wavelengths.
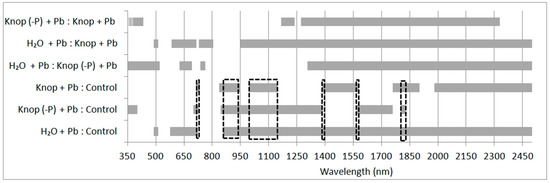
Figure 4.
The statistically significant spectral ranges (p < 0.001) for pairs of the different treatments of the measurements.
The calculated remote sensing indicators of Pb-treated plants showed significant differences in comparison with the reference plants. Statistical analysis was performed using the Mann–Whitney U test at the 0.001 level, which confirmed the significance of the observed changes (marked in red in Table 2). We observed a statistically significant (p < 0.001) decrease in the value of most of the indicators compared with the control. These changes were clearly visible in the indicators describing the contents of chlorophyll in the plants (mNDVI705, REPI1, RARSa, and RARSb), the amount of light used in photosynthesis (SIPI), nitrogen (NDNI), the amount of carbon (NDLI) and carotenoids (CRI1, ARI1), and the water content in vegetation cover (WBI, NDWI, MSI, and NDII). Overall analysis of all samples at a level of significance of 0.001 indicated that the most sensitive indicator to changes caused in the plant due to lead is the water content as indicated by NDWI, WI, NWI-2, and DSWI. The values of these indicators decreased by 5–34%, 1–5%, 23–43%, and 5–27%, respectively, thereby indicating the water stress of the plants. In the H2O + Pb-treated plants, we also observed a significant decrease in the value of chlorophyll b (RARSb) by about 35% in comparison to the control, a statistically significant difference in the indicators describing the dry parts of plants, and an increase of about 57% in the amount of anthocyanins (ARI1) in relation to the control. In the plants from the Knop (-P) + Pb treatments, we also observed significant decreases in the value of water (generally about 1–34% in relation to the control), a statistically significant difference in the indicators describing the dry parts of plants and chlorophyll content (mNDVI705, a difference about 19% in relation to the control).

Table 2.
Mean values of the remote sensing indices with the standard deviation of the different measurements indices comparing Knop + Pb, Knop (-P) + Pb, and H2O + Pb treatments with the control. * indicates a statistical significant difference α = 0.001; ±SD).
The results represent the fluorescence parameter values obtained for the different variants of pea leaves, using light intensities of 60,120, 180, 240, 420, 600, 820, and 1020 µmol photons·m−2s−1 (Figure 5). When leaves of high- or low-light-grown plants are exposed to other environmental stresses, a pronounced and sustained increase in energy dissipation may be induced, which could largely account for the change in photochemical efficiency. It is not clear whether the growth light conditions can modify the response of plants to heavy metals. We measured the quantum efficiency of PSII (Fv’/Fm’) and non-photochemical quenching of fluorescence (NPQ) in the light. The results show that the the maximum quantum efficiency of PSII (Fv’/Fm’) was significantly reduced in the presence of lead (Figure 5). At the higher light intensities above 400 μmol photons·m−2s−1, we observed slightly decreased photosynthetic efficiency (by about 4% to 8%), which was also confirmed by a significant difference in SIPI values, indicating the amount of light used in the photosynthesis process for peas growing in H2O + Pb and Knop (-P) + Pb medium was statistically significantly different compared with the control. Maximal photosynthetic efficiency (Fv’/Fm’ of ~0.78) was observed for control plants. We observed that in the Pb-treated plants in the range of light intensity from 60 to 600 μmol photons·m−2s−1, heat emission (NPQ) significantly reduced, but increased significantly at higher light intensities. This may indicate participation of carotenoids in the NPQ process. This was confirmed by a significant difference in the values for remote sensing CRI1 and ARI1 indices, which indicate the amount of carotenoids and anthocyanins, respectively. For peas grown in H2O + Pb and Knop (-P) + Pb medium, these indices showed a statistically significant change compared with the control. In control plants, NPQ was higher than in Pb-treated plants; thus, heat emission protects photosynthesis components and systems when light absorption is maximal.
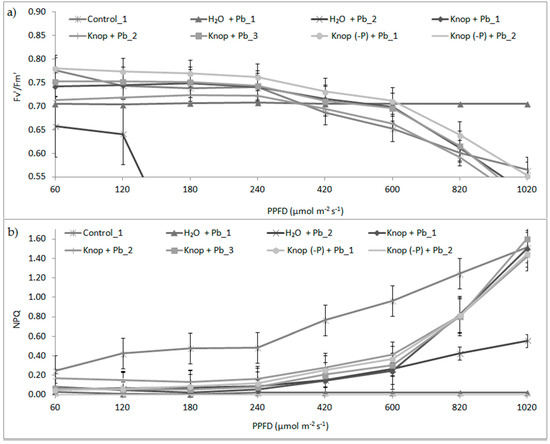
Figure 5.
Relationship between photosynthetic photon flux density (PPFD) and the maximum quantum efficiency of PSII (Fv’/Fm’; (a)) in the light adapted state and non-photochemical fluorescence quenching (NPQ; (b)) for the different variants of the measurement pea leaves. Data are means ± SE (n ≥ 5).
The maximum quantum efficiency of PSII in the light (Fv’/Fm’; Figure 5) for all Pb-treated plants was low and decreased significantly as PPFD increased from 420 to 1020 μmol·photon m−2s−1, When the Pb ions were added to the distilled water, we observed a strong decrease in the F’/Fm’ values, even in lowest PPDF, or no change at all. This result suggests that PSII operating efficiency is inhibited, which demonstrates that the ability to oxidize QA decreases and irreversible damage of PSII occurs. Generally, decreases in quantum efficiency of PSII is indicated by increases in NPQ. NPQ was measured after illuminating the leaf at increasing light intensities to investigate the light responses of this parameter. NPQ increased for control plants from the lowest PPDF, which demonstrates that both the energy processing and the dissipating capacities increased, in turn indicating the better performance of photosynthetic apparatus. In Pb-treated plants, the NPQ was suppressed by Pb ions, even at low PPDF; thus, the photoprotective mechanisms was not present in these plants. The changes caused by lead, as well as impeding the proper course of photosynthesis, were visible in the leaves, Deposits of lead were visible in the wall of the vascular bundles (Figure 6). The data also indicate that the water thermal dissipation of excessive excitation energy was at the lowest level in Pb-plants.

Figure 6.
Leaves of pea plants grown in Knop medium with addition of 5mM Pb(NO3)2. Deposits of lead are visible in the wall of vascular bundles (photo: I. Kaźmierczak).
The plants under stress conditions related to the presence of lead ions in the medium were characterized by a much lower height, a longer but less branched root system, a smaller number of trichomes, and clearly visible deposits of lead in the vascular tissues of the leaves (Figure 6). Also, the fresh and dry masses of Pb-treated plants were lower than those of the control (Table 3). The lead contents (median value) in various parts of the tested plants (30 plants from each environment) are presented in Table 3.

Table 3.
The content of lead in the roots, stalks, and leaves of peas for different treatments (μg·g−1 dry weight).
The content of lead in control plants was very low, which might indicate the minor contamination of reagents used (Table 3). The highest amounts of lead content (an average of 15.3 mg Pb g−1 dry weight) was found in the roots of plants growing in complete Knop medium supplemented with 5 mM Pb(NO3)2, whereas the lowest content was found in the leaves of these plants, resulting from the reduced transport of the lead ions to the leaves. The addition of Pb to the water increased this metal concentration in the leaves approximately 200 times competed to that in Knop + Pb plants. This may be related to a lack of nutrients (salts present in Knop medium) in distilled water, which resulted in increased transport of lead ions from the medium. The Pb concentration in the leaves from plants grown in the Knop medium without phosphate and with 5 mM Pb(NO3)2 was increased approximately 25 times compared to that in the leaves of plants grown with the water with Pb ions. The results indicate that the lead content in the leaves can be decreased by phosphorus in the growth medium. Plants grown on Knop (-P) + Pb accumulated more lead in the leaves than the plants grown in complete medium with Pb, which creates less insoluble salts, which facilitate the transport of metal ions to the leaves. The obtained results indicate the importance of the selected growth media on degree of accumulation of heavy metals in the plant leaves. The leaves and roots of vegetables have a particular predisposition for accumulating toxic metals such as lead and therefore can be used for biomonitoring of the environment, mainly as a tool for assessing the extent of soil contamination. The presence of phosphorus in Knop media reduces the uptake of Pb ions by pea leaves. This indicates that P ions help reduce Pb toxicity by limiting its availability for plants. This result is also confirmed by the differences in the values of the remote sensing indicators.
The lead had negative effects on plant vegetative growth. Root and stalk growth was sensitive to lead and was inhibited approximately 30–40% for all investigated plants compared to control plants (Table 4). Our results show that although the accumulation of lead was not uniform among organs, sensitivity to heavy metal of growth processes was similar. Lead may induce unknown signal(s) that are transmitted from root to shoot. Table 5 presents the effect of lead on the fresh weight of stalks, leaves, and roots of the investigated plants. The fresh weight of the organs markedly decreased in all plants treated with lead compared to the control. The largest decrease (approximately 70%) was observed for all organs in pea seedlings grown in distilled H2O + Pb. For plants grown in Knop + Pb, the decrease was highest in the roots (about 50%) according to highest accumulation of lead. Thus, changes in the growth and biomass accumulation depends on the photochemical activity and thus on the rate of photosynthesis.

Table 4.
The effect of lead on the length (cm) of stalks and roots. The length of control plants was set to 100%.

Table 5.
The effect of lead on a fresh mass (g) of selected plant organs (calculated for one plant). The mass of control plants was set to 100%.
We also observed that the reduction in plant fresh mass and inhibition of growth elongation by Pb ions was accompanied by water loss of plants organs (data not presented). The greatest depletion of water content was noted in the leaves of plants grown in water with Pb ions (approximately 40%), whereas the smallest occurred in the leaves of plants grown in Knop + Pb compared to the control plants. This was also confirmed by the highest difference in the indicators from the group describing water content in vegetation cover (WBI, NDWI, MSI, and NDII); these differences ranged from 1% to 42%.
Leaves of pea seedlings grown in Knop (with and without P) with Pb ions exhibited reduction in chlorophyll a content compared with control seedlings (Figure 7). Growth in water and Pb(NO3)2 treatment increased the chlorophyll content significantly (about 70%, Figure 7). The plants treated with Pb ions showed lower chlorophyll a/b ratios than control plants (2.7) and was lowest (1.3) in plants grown in water with Pb. The plants grown in hydroponic cultures with Knop medium, in both variants (with and without phosphorus), contained less carotenoids relative to the control plants (Figure 7). Carotenoids (Car) were less affected by lead compared to chlorophyll (Chl); however, the Chl/Car ratio increased significantly after Pb treatment: 5.9 for control plants but approximately 10 for plants grown in water with Pb. Remote sensing indicators describing the content of chlorophyll (such as RARSa and RARSb; Table 6) also confirmed a decrease of about 35% (in the case of both indicators) between the growth environment, H2O + Pb, and control (Table 6). However, the amount of dyes, such as anthocyanins or carotenoids reflected by remote sensing vegetation indicators ARI1 and CRI1, also confirmed the fluorescence results. For the H2O + Pb growth environment, the difference in CRI1 was 47% and 575 for ARI1 compared with the control.
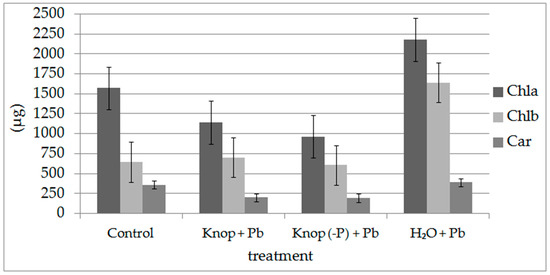
Figure 7.
The photosynthetic pigments (chlorophyll a and b and carotenoids) contents in the pea leaves, expressed as micrograms per gram fresh weight.

Table 6.
The ratio of Chla/b and chlorophyll to carotenoids (Chl/Car) in the pea leaves contaminated with lead ion, Pb(NO3)2, calculated as in Arnon [49] and indicators of remote sensing (RARS, RARSb, and RARSc).
In summary, the plants grown in distilled water with Pb ions were most sensitive to the toxic effects of lead. This result was also confirmed by the differences in the remote sensing indicator values, where the effects in peas could be ranked depending on the medium in which they were grown as follows from the worst to the best condition: H2O + Pb, Knop (-P) + Pb, Knop + Pb, control. Although under these conditions lead is accumulated to a large degree in the leaves with only a small amount in the roots, both the elongation growth and mass the organs were inhibited. The maximum quantum efficiency of PSII in the light and the Chl a/b ratio were very low for these plants, indicating the inhibition of photosynthesis. The Fv/Fm ratio is used for monitoring in vivo effects of stress because it is linearly correlated with the quantum yield of light limited O2 evolution and the number of PSII centers. In contrast, the plants grown on complete Knop + Pb ions showed the least response to the presence of the toxic metal due to the accumulation of Pb in the roots and the formation of insoluble phosphate salts. The indicators that showed statistically significant differences and thereby damage to plant cells caused by the action of lead were indicators determining the chlorophyll content: mNDVI705, RARSa, and RARSb; the amount of light used in photosynthesis: SIPI; the amount of carotenoids and other pigments: CRI1 and ARI1; and water content: NDWI, WI, NWI-2, and DSWI. These results indicate that the type of growth medium has a considerable influence on the absorption and accumulation of toxic metals in different parts of the plant, thereby affecting their function and modifying growth.
4. Discussion
Coupling fluorescence measurements with other non-invasive techniques, such as absorption spectroscopy in model laboratory conditions, we obtained more comprehensive results showing changes in the rate of growth, water content, photosynthetic activity, and others induced by lead under given conditions that change the transport of Pb ions to the leaves. Hence, irradiance during growth is responsible for changes in the antioxidant enzymes activities by affecting the rate of photosynthesis and related processes. The presented results confirm that the effect of Pb ions on photosynthetic parameters in pea seedlings can be detected using hyperspectral and fluorescence techniques. Despite the visual differences and accumulation of lead in the leaves, we confirm that the chlorosis of leaves may be a consequence of the inhibition of chlorophyll synthesis, as well as the increase in its degradation [66,67]. We previously observed [14] that Pb ions stimulated the respiration rate in pea leaves in plants grown in high light conditions, which was accompanied by an increase in ATP production in mitochondria. We also found that the inhibition of photosynthesis by Pb ions was higher in low- than in high-light-grown plants as a consequence of decreasing electron transport in the photosynthesis light reaction [68]. Lead is absorbed by plants mainly through the root system and in minor amounts through the leaves. At lethal concentrations, this barrier is broken and lead may enter vascular tissues. Lead in plants may form deposits of various sizes, present mainly in intercellular spaces, cell walls, and vacuoles. The negative effects of lead on plant vegetative growth mainly result from the following factors: distortion of chloroplast ultrastructure, obstructed electron transport, inhibition of Calvin cycle enzymes, impaired uptake of essential elements such as Mg and Fe, and induced deficiency of CO2 resulting from stomatal closure [69,70].
Important for research on the impact of lead and heavy metals on peas is the environment in which the plant grows, both in terms of the soil and the nutrient medium. In our study, pea seedlings had the lowest values in comparison to the control when grown in H2O + Pb medium. In the literature, we found an example where detached leaves of 14-day-old dark-grown pea seedlings were immersed with their cut ends either in water (control) or in 20 mM Pb(NO3)2 solution [71]. They were exposed to continuous illumination for 24 and 48 h. Values of Fv, Fm, and Fv/Fm were reduced by Pb2+. Here, with H2O + Pb treatment, the Fv’/Fm’ value decreased about two-fold. The decrease in the chlorophyll a fluorescence parameters occurred in parallel with the strong inhibition of the biosynthesis of chlorophyll a and b but less of the carotenoids by this metal [71]. We also confirmed that the difference in the chlorophyll value between the H2O + Pb and control treatments was significant (Chl a was 700 μg/g FW; Chl b was 1000 μg/g FW), whereas the difference in the carotenoids value was similar or less by only 20 μg/g FW. Pb2+ drastically reduced photosynthesis but had a stimulatory action on evolution in darkness (DR) after 24 h and only somewhat inhibited DR after 48 h exposure of leaves to this metal [71].
Fluorescence indicators confirmed the negative effect of lead ions on pea seedlings through a decline in the value of Fv/Fm [72]. Fluctuations were observed in PSII electron transport during photosynthesis induction. When the light intensity increased, the maximum quantum efficiency of PSII (Fv/Fm) decreased, as was also observed by Baker et al. [19]. The decrease in the Fv/Fm value was also noted for oats plants (Avena sativa) growing on contaminated places with excess Cu and Pb ions where the half-rise time (t1/2) of Chl a fluorescence decreased, suggesting that the amount of active pigments decreased and the functional Chl antennae size of the photosynthetic apparatus was smaller compared with control plants [73]. In pea plants, after adding lead to the water, we observed that the greatest amount of lead was transported to the leaves, which was also noted for spinach plants treated with lead acetate for 15 days; 45% of Pb ions were accumulated in roots and 55% in the leaves [74]. More than 90% of heavy metals are generally adsorbed into cell walls. The oldest leaves accumulated the highest concentrations of heavy metals. Heavy metals have different mobility in plant tissues and translocation is probably regulated by the carrier proteins of the vascular tissues [75,76]. Decreases in the protein content in pea roots was observed after the addition of cadmium ions [67,77]. Roots accumulate larger amounts of metals than shoots [78]. Permanent stomatal closure, structural damage of chloroplasts, increased synthesis of ethylene (induce senescence), and imbalance in water relationships might be acting synergistically on heavy metal exposed plants, resulting in impairment of photosynthetic functions [79].
Carotenoids were less affected by lead compared to chlorophyll; however, the Chl/Car ratio increased significantly after Pb treatment: 5.9 for control plants 5.9 but and about 10 for plants grown in water with Pb. An increase in the Chl/Car ratio is characteristic for stress conditions because carotenoids are involved in photoprotective mechanisms. This is particularly important when photochemical quenching activity is exceeded, leading to photoinhibition [80]. Carotenoids protect chloroplasts (1) by modulating NPQ, (2) by mediating direct quenching of chlorophyll triples, and (3) by scavenging the ROS generated during photosynthesis. Our results indicate that the main function of carotenoids is scavenging the ROS because the NPQ values were low in Pb-treated plants. The reduced chlorophyll content due to heavy metals toxicity in different plant species has been well documented [81]. The observed increases in chlorophyll content in the leaves of plants grown in water + Pb may be related to the lead transported to the young plants inhibiting the activity of chlorophyllase, so the content of chlorophyll may be even higher in plants treated with lead [10].
The obtained results can be compared with previously reported data of tests of plants under stress conditions. Zagajewski [82], using multispectral spectrometer SPZ5 (24 spectral bands in the visible and near-infrared range from 400 to 1025 nm; Research Space Centre, PAS, Warsaw, Poland), noted the presence of lead in the contaminated grasses with 128 ppm Pb2+. He observed not only a decrease in the light absorption used in the process of photosynthesis (VIS), but also changes in cellular structures. Heavy metals reduced the chlorophyll content, which is closely related to the inhibition of photosynthesis and, as a consequence, causes a decrease in biomass production [51]. Here, this was confirmed by the reduced LAI index value, and the roots and the leaf index determined by the ratio of fresh to dry mass of the leaves [51]. A hyperspectral method was used to assess concentrations of heavy metals in common cane growing along the Le An River in China [83], where the spatial distribution of metal concentration was examined. The stress status of plants was determined by using three indicators: NDVI, red edge position (REP), and continuum removal normalized band depth (CRNBD).
The correlation of remote sensing indices with chlorophyll content has enabled the creation of chlorophyll content models. On the basis of the proposed model, significant differences were observed in the level of heavy metals between the top and bottom of the plant stand corresponding to the chlorophyll gradients within. The concentrations of copper, zinc, and lead were highest in the lower parts of plants, but the coefficient of variation revealed differences in the values between heavy metals used. For Cu ions, the highest value attained was 0.61, 0.53 for Pb, and Zn was lowest at 0.22. The results also showed that the HMs content correlates negatively with chlorophyll level, whereas the vegetation indices showed a positive correlation [83]. Remote sensing of vegetation indices in the research areas contaminated with heavy metals were used, among others, by Elbe in Germany [84]. The aim of this study was to assess the pollution of the river floodplain ecosystems using ground spectrometric measurements (ASD FieldSpec Pro FR) and laboratory measurements. Remote sensing vegetation indices were able to assess the vitality of the plants, including biomass, chlorophyll, water, cellulose, and lignin contents. The water status of plants was excluded as a stressor because the coefficient of variation was small for WBI and MSI. The indicators NPCI, PRI, NDVI, and SIPI showed higher variability over time. In the case of the nitrogen content (NDNI) and lignin (NDLI), small changes were identified. Especially indicators NPCI, PRI, REP, and CR1730 were proven to be sensitive to the stress caused by heavy metals.
The acquired results are also confirmed by the differences in the values of the remote sensing indicators, where the effect on peas can be ranked from the worst to best condition depending on the medium in which they were grown as follows: H2O + Pb, Knop + Pb, Knop (-P) + Pb, and control. Different environments significantly affect the impact of stress on plant growth caused by lead on its vital functions. The single-metal sorption uptake capacity of the biomass for Pb was slightly inhibited by the presence of the other heavy metals in the system. The presence of Cu and Cd cations in separate systems reduced the biomass uptake capacity of Pb by only 6% [85]. Two plant species, spinach (Spinacia oleracea) and wheat (Triticum aestivum), were grown under hydroponic conditions and stressed with lead nitrate, Pb(NO3)2, at three concentrations (1.5, 3, and 15 mM) [86]. Lead was accumulated in a dose-dependent manner in both plant species, which resulted in reduced growth and lower uptake of all mineral ions tested. Total amounts and concentrations of most mineral ions (Na, K, Ca, P, Mg, Fe, Cu, and Zn) decreased, although Mn concentrations increased, as its uptake decreased less relative to the whole plant’s growth. The deficiency in mineral nutrients was correlated with a strong decrease in the chlorophyll a and b and proline contents in both species, but these effects were less pronounced in spinach than in wheat. In contrast, the effects of lead on soluble proteins differed between species; they reduced in wheat at all lead concentrations, whereas they increased in spinach, where their value peaked at 3 mM Pb. In wheat seedlings, concentrations of chlorophylls a and b were already significantly lowered at 1.5 mM Pb, and this effect was even more pronounced at 3 and 15 mM Pb. Lead stress resulted in a heavy reduction in chlorophyll due to both chloroplast disorganization, the reduction in the amount of thylakoids and grana, direct inhibition of chlorophyll synthesis, as well as changes of chlorophyll structure due to replacement of key nutrients (Mg, Fe, and Cu) by lead [87,88]. Lead exposure resulted in dose-dependent damage to both plant species. In wheat plants exposed to 15 mM Pb, growth inhibition was clear, whereas spinach fresh and dry weights decreased by only 28% and 29%, respectively, at 15 mM Pb, when compared with controls.
The spectral reflectance curves obtained for pea plants in the near and mid-infrared indicate a high sensitivity to Pb. Plants treated with lead ions are the most sensitive to light in the red edge range, which also has been confirmed for other metals as Ni, Cd, and Cu [31,40]. An experimental design was established with three Pb concentrations (0, 2.5 × 207.2, 5.0 × 207.2 mg Pb per 1000 g soil) for 30, 50, 65, 80, and 90 days. The differences in the shape of the spectral reflection curve were visible mainly in the red edge range, which was found here (e.g., 714–723 nm). The highest impact of lead (the difference between the spectral characteristics of the concentrations tested) was observed on day 50 and 80 by a flattening of the edges of red [34]. The derivative reflectance calculated from canopy-level CASI airborne imagery showed a peak at the 700–730 nm region, which was experimentally shown to be related to stress conditions and potentially caused by fluorescence emission and chlorophyll content changes in vegetation under stress [89,90]. Here, the ranges in the electromagnetic spectrum were statistically significant for condition, stress influence was the range: 714–723 nm (plant stress), 858–951 nm (biomass, plant density), 995–1145 (biophysical quantity and yield), and 1386–1389, 1570–1579, and 1801–1829 nm (lignin, cellulose, plant litter and water). Similar results were also observed by Zagajewski [82] for grasses 6 and 11 weeks after application of lead. Spectral reflectance curves can also indicate disturbances in the cell structures. The largest differences in reflectance, especially in near-infrared, were shown for the grasses 11 weeks after application of lead.
Lead causes large changes in plant vitality, inducing cellular structures by creating deposits in the cell walls. This may cause developmental disorders, as well as impact the water status of plants and then photosynthesis. In analyzing the spectrum bands for pea and common cane plants growing in the presence of lead, lead induced changes in the spectrum differently for both plants. In the case of the pea plants, the range of the electromagnetic spectrum, describing the structure of the cells and the water content in the plants, indicated the high sensitivity of the pea plants to lead ions. Whereas for cane plants grown on the reservoir flotation [91], changes occurred in the edge of the red (400–800 nm), the near infrared (from 1100 to 1400 nm), and in the mid-infrared.
Numerous studies that have tested the physiological responses to excess levels of heavy metal ions indicate that plants have evolved various mechanisms to cope with heavy-metal-induced stress. Lead is highly reactive and inactivates various enzymes; hence, it impairs plant growth and development [71]. Thus, using noninvasive techniques, such as fluorescence, absorption spectroscopy, or gas exchange, is important for monitoring plant photosynthesis under given conditions even for a long time. The transferability of laboratory-based results to field situations is a subject of discussion. Laboratory results might not mirror in situ behavior due to differences in growing environments, sampling protocols, and operating conditions, but they can provide information about the correlation between investigated processes under controlled conditions. Thus, further comparative work is warranted, which will allow for further development, such as the novel ground-based spectrometer system, PhotoSpec, for measuring SIF in the red (670–732 nm) and far-red (729–784 nm) wavelength range as well as canopy reflectance (400–900 nm) to calculate vegetation indices, such as the normalized difference vegetation index (NDVI), the enhanced vegetation index (EVI), and the photochemical reflectance index (PRI) [92].
5. Conclusions
The obtained results confirm a significant decrease in the vitality of the pea plants treated with lead. Using fluorescence techniques to assess the degree of inhibition of plant metabolism by lead ions provides useful information about the actual physiological state of vegetation in the presence of this heavy metal. These changes are visible in both the spectral reflection curve shape and the remote sensing vegetation indices. Combined with remote sensing measurements and the quantitative survey of selected parameters (e.g., photosynthetic pigments), they are one of the most accurate techniques for describing photosynthesis and evaluating the function of the photosynthetic apparatus in the presence of stress factors. Uptake of heavy metals, their distribution, and accumulation result in permanent structural changes in the plants, which was confirmed by the results obtained from spectrometric measurements. Their effect changes in terms of the content of plant components such as water and photosynthetically active pigments, confirming an adverse effect on the photosynthetic apparatus of the plant (fluorescence measurements). One of the main symptoms induced by lead on the plants is accelerated aging, visible in the decrease in chlorophyll content and the lowering of the water content in tissues (the drying of plants). Lead exposure in plants strongly limits the development and sprouting of seedlings. Plant biomass can also be restricted by high doses of lead exposure.
The disruption of plant water status after lead treatment has been addressed in many studies. Results of such exposures show a decrease in transpiration as well as reduction in the moisture content. Lead reduces plant cell wall plasticity, and thereby influences the cell turgor pressure. The obtained results confirm that hyperspectral techniques can be used to detect the effect of lead as a stress factor on plant vegetation. The contribution of this research is the identification of noticeable changes in the physical properties of pea plants treated with Pb ions and grown in the laboratory conditions, where visible changes were observed in the cell structure and water content as well as changes leading to the death of plants. This information provides the basis for the assumption that the hyperspectral data and fluorescence measurements are useful for the analysis of vegetation contaminated with heavy metals and monitoring of contaminated areas. The determination of the content of heavy metals accumulated in vegetables and physiological effects on the plants can provide information on the source and extent of the impact of pollution emissions.
Author Contributions
The experiment was designed and conducted by E.R. and M.K.; both researchers analyzed the data. All authors prepared the manuscript.
Funding
The publishing costs were financed from by grant No. 500-D119-12-1190000 awarded by the Polish Ministry of Science and Higher Education.
Acknowledgments
The authors express thanks to Anna Robak and Ignacy Kaźmierczak for their participation in laboratory research. The analyses were conducted in the frame of the COST Action: “Optical synergies for spatiotemporal SENsing of Scalable ECOphysiological traits” (SENSECO). Sincere thanks to Echo Chi (editor), Kelly O’Keefe (English editor) and anonymous Reviewer, who allowed us to improve the manuscript.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Mohr, H.; Schopfer, P. Physiology of Movement. In Plant Physiology; Springer: Berlin/Heidelberg, Germany, 1995; pp. 497–538. [Google Scholar] [CrossRef]
- Fryer, M.J.; Andrews, J.R.; Oxborough, K.; Blowers, D.A.; Baker, N.R. Relationship between CO2 Assimilation, Photosynthetic Electron Transport, and Active O2 Metabolism in Leaves of Maize in the Field during Periods of Low Temperature. Plant Physiol. 1998, 116, 571–580. [Google Scholar] [CrossRef] [PubMed]
- Hendrickson, L.; Furbank, R.T.; Chow, W.S. A Simple Alternative Approach to Assessing the Fate of Absorbed Light Energy Using Chlorophyll Fluorescence. Photosynth. Res. 2004, 82, 73–81. [Google Scholar] [CrossRef] [PubMed]
- Romanowska, E. Gas Exchange Functions in Heavy Metal Stressed Plants. In Physiology and Biochemistry of Metal Toxicity and Tolerance in Plants; Springer: Dordrecht, The Netherlands, 2002; pp. 257–285. [Google Scholar] [CrossRef]
- Rashid, A.; Camm, E.L.; Ekramoddoullah, A.K.M. Molecular mechanism of action of Pb2+ and Zn2+ on water oxidizing complex of photosystem II. FEBS Lett. 1994, 350, 296–298. [Google Scholar] [CrossRef]
- Yadav, S.K. Heavy metals toxicity in plants: An overview on the role of glutathione and phytochelatins in heavy metal stress tolerance of plants. S. Afr. J. Bot. 2010, 76, 167–179. [Google Scholar] [CrossRef]
- Schreiber, U. Pulse-Amplitude-Modulation (PAM) Fluorometry and Saturation Pulse Method: An Overview. In Chlorophyll a Fluorescence; Springer: Dordrecht, The Netherlands, 2007; pp. 279–319. [Google Scholar] [CrossRef]
- Burzyński, M.; Kłobus, G. Changes of photosynthetic parameters in cucumber leaves under Cu, Cd, and Pb stress. Photosynthetica 2004, 42, 505–510. [Google Scholar] [CrossRef]
- Woźniak, A.; Drzewiecka, K.; Kęsy, J.; Marczak, Ł.; Narożna, D.; Grobela, M.; Motała, R.; Bocianowski, J.; Morkunas, I. The Influence of Lead on Generation of Signalling Molecules and Accumulation of Flavonoids in Pea Seedlings in Response to Pea Aphid Infestation. Molecules 2017, 22, 1404. [Google Scholar] [CrossRef] [PubMed]
- Parys, E.; Romanowska, E.; Siedlecka, M.; Poskuta, J.W. The effect of lead on photosynthesis and respiration in detached leaves and in mesophyll protoplasts of Pisum sativum. Acta Physiol. Plant. 1998, 20, 313. [Google Scholar] [CrossRef]
- Romanowska, E.; Igamberdiev, A.U.; Parys, E.; Gardestrom, P. Stimulation of respiration by Pb2+ in detached leaves and mitochondria of C3 and C4 plants. Physiol. Plant. 2002, 116, 148–154. [Google Scholar] [CrossRef]
- Nas, F.S.; Ali, M. The effect of lead on plants in terms of growing and biochemical parameters: A review. MOJ Ecol. Environ. Sci. 2018, 3, 265–268. [Google Scholar] [CrossRef]
- Romanowska, E.; Wasilewska, W.; Fristedt, R.; Vener, A.V.; Zienkiewicz, M. Phosphorylation of PSII proteins in maize thylakoids in the presence of Pb ions. J. Plant Physiol. 2012, 169, 345–352. [Google Scholar] [CrossRef]
- Romanowska, E.; Wróblewska, B.; Droƶak, A.; Siedlecka, M. High light intensity protects photosynthetic apparatus of pea plants against exposure to lead. Plant Physiol. Biochem. 2006, 44, 387–394. [Google Scholar] [CrossRef] [PubMed]
- Aro, E.-M.; Suorsa, M.; Rokka, A.; Allahverdiyeva, Y.; Paakkarinen, V.; Saleem, A.; Battchikova, N.; Rintamäki, E. Dynamics of photosystem II: A proteomic approach to thylakoid protein complexes. J. Exp. Bot. 2005, 56, 347–356. [Google Scholar] [CrossRef] [PubMed]
- Miles, C.D.; Brandle, J.R.; Daniel, D.J.; Chu-Der, O.; Schnare, P.D.; Uhlik, D.J. Inhibition of Photosystem II in Isolated Chloroplasts by Lead. Plant Physiol. 1972, 49, 820–825. [Google Scholar] [CrossRef] [PubMed]
- Baker, N.R. Chlorophyll Fluorescence: A Probe of Photosynthesis in vivo. Annu. Rev. Plant Biol. 2008, 59, 89–113. [Google Scholar] [CrossRef] [PubMed]
- Ruban, A.V. Nonphotochemical Chlorophyll Fluorescence Quenching: Mechanism and Effectiveness in Protecting Plants from Photodamage. Plant Physiol. 2016, 170, 1903–1916. [Google Scholar] [CrossRef] [PubMed]
- Baker, N.R.; Oxborough, K. Chlorophyll Fluorescence as a Probe of Photosynthetic Productivity. In Chlorophyll a Fluorescence; Springer: Dordrecht, The Netherlands, 2007; pp. 65–82. [Google Scholar] [CrossRef]
- Clijsters, H.; Van Assche, F. Inhibition of photosynthesis by heavy metals. Photosynth. Res. 1985, 7, 31–40. [Google Scholar] [CrossRef]
- Kalaji, H.M.; Loboda, T. Photosystem II of Barley seedlings under cadmium and lead stress. Plant Soil Environ. 2007, 53, 511–516. [Google Scholar] [CrossRef]
- Lazár, D.; Jablonsky, J. On the approaches applied in formulation of a kinetic model of photosystem II: Different approaches lead to different simulations of the chlorophyll a fluorescence transients. J. Theor. Biol. 2009, 257, 260–269. [Google Scholar] [CrossRef]
- Shamshad, S.; Shahid, M.; Rafiq, M.; Khalid, S.; Dumat, C.; Sabir, M.; Murtaza, B.; Farooq, A.B.U.; Shah, N.S. Effect of organic amendments on cadmium stress to pea: A multivariate comparison of germinating vs. young seedlings and younger vs. older leaves. Ecotoxicol. Environ. Saf. 2018, 151, 91–97. [Google Scholar] [CrossRef]
- Bazzaz, F.A.; Rolfe, G.L.; Carlson, R.W. Effect of cadmium on photosynthesis and transpiration of excised leaves of corn and sunflower. Physiol. Plant. 1974, 32, 373–376. [Google Scholar] [CrossRef]
- Zagajewski, B.; Kycko, M.; Tømmervik, H.; Bochenek, Z.; Wojtuń, B.; Bjerke, J.W.; Kłos, A. Feasibility of hyperspectral vegetation indices for the detection of chlorophyll concentration in three high Arctic plants: Salix polaris, Bistorta vivipara, and Dryas octopetala. Acta Soc. Bot. Pol. 2018, 87, 3604. [Google Scholar] [CrossRef]
- Kycko, M.; Zagajewski, B.; Lavender, S.; Romanowska, E.; Zwijacz-Kozica, M. The Impact of Tourist Traffic on the Condition and Cell Structures of Alpine Swards. Remote Sens. 2018, 10, 220. [Google Scholar] [CrossRef]
- Clevers, J.G.P.W. Beyond NDVI: Extraction of Biophysical Variables from Remote Sensing Imagery. In Remote Sensing and Digital Image Processing; Springer: Dordrecht, The Netherlands, 2014; pp. 363–381. [Google Scholar] [CrossRef]
- Farquhar, G.D.; von Caemmerer, S.; Berry, J.A. A biochemical model of photosynthetic CO2 assimilation in leaves of C3 species. Planta 1980, 149, 78–90. [Google Scholar] [CrossRef]
- Guanter, L.; Zhang, Y.; Jung, M.; Joiner, J.; Voigt, M.; Berry, J.A.; Frankenberg, C.; Huete, A.R.; Zarco-Tejada, P.; Lee, J.-E.; et al. Global and time-resolved monitoring of crop photosynthesis with chlorophyll fluorescence. Proc. Natl. Acad. Sci. USA 2014, 111, E1327–E1333. [Google Scholar] [CrossRef]
- Norton, A.J.; Rayner, P.J.; Koffi, E.N.; Scholze, M. Assimilating solar-induced chlorophyll fluorescence into the terrestrial biosphere model BETHY-SCOPE v1.0: Model description and information content. Geosci. Model Dev. 2018, 11, 1517–1536. [Google Scholar] [CrossRef]
- Kancheva, R.; Georgiev, G. Spectrally-based quantification of plant heavy metal-induced stress. In Remote Sensing for Agriculture, Ecosystems, and Hydrology XIV; SPIE: Bellingham, WA, USA, 2012; Volume 8531, p. 85311D. [Google Scholar] [CrossRef]
- Rajewicz, P.A.; Atherton, J.; Alonso, L.; Porcar-Castell, A. Leaf-level spectral fluorescence measurements: Comparing methodologies for broadleaves and needles. Remote Sens. 2019, 11, 532. [Google Scholar] [CrossRef]
- Rathod, P.H.; Brackhage, C.; Van der Meer, F.D.; Müller, I.; Noomen, M.F.; Rossiter, D.G.; Dudel, G.E. Spectral changes in the leaves of barley plant due to phytoremediation of metals—Results from a pot study. Eur. J. Remote Sens. 2015, 48, 283–302. [Google Scholar] [CrossRef]
- Ren, H.-Y.; Zhuang, D.-F.; Pan, J.-J.; Shi, X.-Z.; Wang, H.-J. Hyper-spectral remote sensing to monitor vegetation stress. J. Soils Sediments 2008, 8, 323–326. [Google Scholar] [CrossRef]
- Dunagan, S.C.; Gilmore, M.S.; Varekamp, J.C. Effects of mercury on visible/nearinfrared reflectance spectra of mustard spinach plants (Brassica rapa P.). Environ. Pollut. 2007, 148, 301–311. [Google Scholar] [CrossRef]
- Su, Y.; Sridhar, B.B.M.; Han, F.X.; Monts, D.L.; Diehl, S.V. Effect of bioaccumulation of Cs and Sr natural isotopes on foliar structure and plant spectral reflectance of Indian mustard (Brassica juncea). Water Air Soil Pollut. 2007, 180, 65–74. [Google Scholar] [CrossRef]
- Bannari, A.; Morin, D.; Bonn, F.; Huete, A. A review of vegetation indices. Remote Sens. Rev. 1995, 13, 95–120. [Google Scholar] [CrossRef]
- Kycko, M.; Zagajewski, B.; Lavender, S.; Dabija, A. In Situ Hyperspectral Remote Sensing for Monitoring of Alpine Trampled and Recultivated Species. Remote Sens. 2019, 11, 1296. [Google Scholar] [CrossRef]
- Zagajewski, B.; Tømmervik, H.; Bjerke, J.; Raczko, E.; Bochenek, Z.; Kłos, A.; Jarocińska, A.; Lavender, S.; Ziółkowski, D. Intraspecific Differences in Spectral Reflectance Curves as Indicators of Reduced Vitality in High-Arctic Plants. Remote Sens. 2017, 9, 1289. [Google Scholar] [CrossRef]
- Kancheva, R.; Borisova, D.; Iliev, I. Chlorophyll fluorescence as a quantitative measure of plant stress. In New Developments and Challenges in Remote Sensing; Bochenek, Z., Ed.; Millpress: Rotterdam, The Netherlands, 2007; ISBN 978-90-5966-053-3. [Google Scholar]
- Holleboom, C.-P.; Walla, P.J. The back and forth of energy transfer between carotenoids and chlorophylls and its role in the regulation of light harvesting. Photosynth. Res. 2014, 119, 215–221. [Google Scholar] [CrossRef]
- Rossini, M.; Fava, F.; Cogliati, S.; Meroni, M.; Marchesi, A.; Panigada, C.; Giardino, C.; Busetto, L.; Migliavacca, M.; Amaducci, S.; et al. Assessing canopy PRI from airborne imagery to map water stress in maize. ISPRS J. Photogramm. Remote Sens. 2013, 86, 168–177. [Google Scholar] [CrossRef]
- Cierniewski, J.; Kazmierowski, C.; Krolewicz, S.; Piekarczyk, J.; Wrobel, M.; Zagajewski, B. Effects of different illumination and observation techniques of cultivated soils on their hyperspectral bidirectional measurements under field and laboratory conditions. IEEE J. Sel. Top. Appl. Earth Obs. Remote Sens. 2014, 7, 2525–2530. [Google Scholar] [CrossRef]
- Pinto, F.; Damm, A.; Schickling, A.; Panigada, C.; Cogliati, S.; Müller-Linow, M.; Balvora, A.; Rascher, U. Sun-induced chlorophyll fluorescence from high-resolution imaging spectroscopy data to quantify spatio-temporal patterns of photosynthetic function in crop canopies. Plant. Cell Environ. 2016, 39, 1500–1512. [Google Scholar] [CrossRef]
- Colombo, R.; Celesti, M.; Bianchi, R.; Campbell, P.K.E.; Cogliati, S.; Cook, B.D.; Corp, L.A.; Damm, A.; Domec, J.-C.; Guanter, L.; et al. Variability of sun-induced chlorophyll fluorescence according to stand age-related processes in a managed loblolly pine forest. Glob. Change Biol. 2018, 24, 2980–2996. [Google Scholar] [CrossRef]
- Drusch, M.; Moreno, J.; Del Bello, U.; Franco, R.; Goulas, Y.; Huth, A.; Kraft, S.; Middleton, E.M.; Miglietta, F.; Mohammed, G.; et al. The Fluorescence Explorer Mission Concept—ESA’s Earth Explorer 8. IEEE Trans. Geosci. Remote Sens. 2017, 55, 1273–1284. [Google Scholar] [CrossRef]
- Rascher, U.; Alonso, L.; Burkart, A.; Cilia, C.; Cogliati, S.; Colombo, R.; Damm, A.; Drusch, M.; Guanter, L.; Hanus, J.; et al. Sun-induced fluorescence—A new probe of photosynthesis: First maps from the imaging spectrometer HyPlant. Glob. Change Biol. 2015, 21, 4673–4684. [Google Scholar] [CrossRef]
- Genty, B.; Briantais, J.-M.; Baker, N.R. The relationship between the quantum yield of photosynthetic electron transport and quenching of chlorophyll fluorescence. Biochim. Biophys. Acta Gen. Subj. 1989, 990, 87–92. [Google Scholar] [CrossRef]
- Arnon, D.I. Cooper enzymes in isolated chloroplasts. Polypenyloxidase in Beta vulgaris. Plant Physiol. 1949, 24, 1–15. [Google Scholar] [CrossRef]
- Dawson, T.P.; Curran, P.J. Technical note A new technique for interpolating the reflectance red edge position. Int. J. Remote Sens. 1998, 19, 2133–2139. [Google Scholar] [CrossRef]
- Chappelle, E.W.; Kim, M.S.; McMurtrey, J.E. Ratio analysis of reflectance spectra (RARS): An algorithm for the remote estimation of the concentrations of chlorophyll A, chlorophyll B, and carotenoids in soybean leaves. Remote Sens. Environ. 1992, 39, 239–247. [Google Scholar] [CrossRef]
- Peñuelas, J.; Baret, F.; Filella, I. Semi-Empirical Indices to Assess Carotenoids/Chlorophyll-a Ratio from Leaf Spectral Reflectance. Photosynthetica 1995, 31, 221–230. [Google Scholar]
- Fourty, T.; Baret, F. On spectral estimates of fresh leaf biochemistry. Int. J. Remote Sens. 1998, 19, 1283–1297. [Google Scholar] [CrossRef]
- Merzlyak, M.N.; Gitelson, A.A.; Chivkunova, O.B.; Rakitin, V.Y. Non-destructive optical detection of pigment changes during leaf senescence and fruit ripening. Physiol. Plant. 1999, 106, 135–141. [Google Scholar] [CrossRef]
- Gitelson, A.A.; Zur, Y.; Chivkunova, O.B.; Merzlyak, M.N. Assessing Carotenoid Content in Plant Leaves with Reflectance Spectroscopy. Photochem. Photobiol. 2002, 75, 272–281. [Google Scholar] [CrossRef]
- Gitelson, A.A.; Merzlyak, M.N.; Chivkunova, O.B. Optical Properties and Nondestructive Estimation of Anthocyanin Content in Plant Leaves. Photochem. Photobiol. 2001, 74, 38. [Google Scholar] [CrossRef]
- Fuentes, D.A.; Gamon, J.A.; Qiu, H.; Sims, D.A.; Roberts, D.A. Mapping Canadian boreal forest vegetation using pigment and water absorption features derived from the AVIRIS sensor. J. Geophys. Res. Atmos. 2001, 106, 33565–33577. [Google Scholar] [CrossRef]
- Gao, B. NDWI—A normalized difference water index for remote sensing of vegetation liquid water from space. Remote Sens. Environ. 1996, 58, 257–266. [Google Scholar] [CrossRef]
- Sims, D.A.; Gamon, J.A. Relationships between leaf pigment content and spectral reflectance across a wide range of species, leaf structures and developmental stages. Remote Sens. Environ. 2002, 81, 337–354. [Google Scholar] [CrossRef]
- Babar, M.A.; Reynolds, M.P.; van Ginkel, M.; Klatt, A.R.; Raun, W.R.; Stone, M.L. Spectral Reflectance Indices as a Potential Indirect Selection Criteria for Wheat Yield under Irrigation. Crop Sci. 2006, 46, 578. [Google Scholar] [CrossRef]
- Galvão, L.S.; Epiphanio, J.C.N.; Breunig, F.M.; Formaggio, A.R. Biophysical and Biochemical Characterization and Plant Species Studies; Thenkabail, P.S., Lyon, J.G., Huete, A., Eds.; CRC Press: Boca Raton, FL, USA, 2018; ISBN 9780429431180. [Google Scholar] [CrossRef]
- Evans, E.H.; Brown, R.G. New trends in photobiology. J. Photochem. Photobiol. B Biol. 1994, 22, 95–104. [Google Scholar] [CrossRef]
- Fisher, R.A. Statistical Methods for Research Workers; Landmark Writings in Western Mathematics: Case Studies, 11th ed.; Grattan-Guinness, I., Ed.; Elsevier: Amsterdam, The Netherlands, 1925; pp. 1640–1940. [Google Scholar]
- StatSoft. StatSoft Manual, Internetowy Podręcznik Statystyki; Statistica: Krakow, Poland, 2012. [Google Scholar]
- Sharma, P.; Dubey, R.S. Lead Toxicity in Plants. Braz. J. Plant Physiol. 2005, 17, 1–19. [Google Scholar] [CrossRef]
- Myśliwa-Kurdziel, B.; Prasad, M.N.V.; Strzałtka, K. Photosynthesis in heavy metal stressed plants. In Heavy Metal Stress in Plants; Springer: Berlin/Heidelberg, Germany, 2004; pp. 146–181. [Google Scholar] [CrossRef]
- Bavi, K.; Kholdebarin, B.; Moradshahi, A. Effect of cadmium on growth, protein content and peroxidase activity in pea plants. Pak. J. Bot. 2011, 43, 1467–1470. [Google Scholar]
- Romanowska, E.; Wróblewska, B.; Drożak, A.; Zienkiewicz, M.; Siedlecka, M. Effect of Pb ions on superoxide dismutase and catalase activities in leaves of pea plants grown in high and low irradiance. Biol. Plant. 2008, 52, 80–86. [Google Scholar] [CrossRef]
- Sengar, R.S.; Gautam, M.; Sengar, R.S.; Sengar, R.S.; Garg, S.K.; Sengar, K.; Chaudhary, R. Lead Stress Effects on Physiobiochemical Activities of Higher Plants. In Reviews of Environmental Contamination and Toxicology; Springer: New York, NY, USA, 2008; pp. 73–93. ISBN 9780387784434. [Google Scholar] [CrossRef]
- Pourrut, B.; Shahid, M.; Dumat, C.; Winterton, P.; Pinelli, E. Lead Uptake, Toxicity, and Detoxification in Plants. In Reviews of Environmental Contamination and Toxicology; Springer: New York, NY, USA, 2011; pp. 113–136. ISBN 9781441998590. [Google Scholar] [CrossRef]
- Łukaszek, M.; Poskuta, J.W. Development of photosynthetic apparatus and respiration in pea seedlings during as influenced by toxic concentration of lead. Acta Physiol. Plant. 1998, 20, 35. [Google Scholar] [CrossRef]
- Joshi, M.K.; Mohanty, P. Chlorophyll a Fluorescence as a Probe of Heavy Metal Ion Toxicity in Plants. In Chlorophyll a Fluorescence; Springer: Dordrecht, The Netherlands, 2007; pp. 637–661. [Google Scholar] [CrossRef]
- Moustakas, M.; Lanaras, T.; Symeonidis, L.; Karataglis, S. Growth and some photosynthesis characteristics of field grown Avena sativa under copper and lead stress. Photosynthetica 1994, 30, 389–396. [Google Scholar]
- Ernst, W.H.O.; Verkleij, J.A.C.; Schat, H. Metal tolerance in plants. Acta Bot. Neerl. 1992, 41, 229–248. [Google Scholar] [CrossRef]
- Gallego, S.M.; Pena, L.B.; Barcia, R.A.; Azpilicueta, C.E.; Iannone, M.F.; Rosales, E.P.; Zawoznik, M.S.; Groppa, M.D.; Benavides, M.P. Unravelling cadmium toxicity and tolerance in plants: Insight into regulatory mechanisms. Environ. Exp. Bot. 2012, 83, 33–46. [Google Scholar] [CrossRef]
- Kłos, A.; Ziembik, Z.; Rajfur, M.; Dołhańczuk-Śródka, A.; Bochenek, Z.; Bjerke, J.W.; Tømmervik, H.; Zagajewski, B.; Ziółkowski, D.; Jerz, D.; et al. Using moss and lichens in biomonitoring of heavy-metal contamination of forest areas in southern and north-eastern Poland. Sci. Total Environ. 2018, 627, 438–449. [Google Scholar] [CrossRef]
- Usman, K.; Al-Ghouti, M.A.; Abu-Dieyeh, M.H. The assessment of cadmium, chromium, copper, and nickel tolerance and bioaccumulation by shrub plant Tetraena qataranse. Sci. Rep. 2019, 9, 5658. [Google Scholar] [CrossRef]
- Cannata, M.G.; Carvalho, R.; Bertoli, A.C.; Augusto, A.S.; Bastos, A.R.R.; Carvalho, J.G.; Freitas, M.P. Effects of Cadmium and Lead on Plant Growth and Content of Heavy Metals in Arugula Cultivated in Nutritive Solution. Commun. Soil Sci. Plant Anal. 2013, 44, 952–961. [Google Scholar] [CrossRef]
- Prasad, M.N.V.; Strzałka, K. Impact of heavy metals on photosynthesis. In Heavy Metal Stress in Plants; Springer: Berlin/Heidelberg, Germany, 1999; pp. 117–138. [Google Scholar] [CrossRef]
- Külheim, C.; Ågren, J.; Jansson, S. Rapid regulation of light harvesting and plant fitness in the field. Science 2002, 297, 91–93. [Google Scholar] [CrossRef]
- Pandey, N.; Pathak, G.C. Nickel alters antioxidative defense and water status in green gram. Ind. J. Plant Physiol. 2006, 11, 113–118. [Google Scholar]
- Zagajewski, B. Remote Sensing Measurements of Lead Concentration in Plants. Misc. Geogr. 2000, 9, 267–282. [Google Scholar] [CrossRef]
- Chen, H. The Possibility of Assessing Heavy Metal Concentrations in Reed along le an River (China) Using Hyperspectral Data; International Institute for Geo-information Science and Earth Observation ITC: Enschede, The Netherlands, 2008; pp. 1–54. [Google Scholar]
- Götze, C.; Jung, A.; Henrich, V.; Merbach, I.; Gläßer, C. Spectrometric analyses in comparison to the physiological condition of heavy metal stressed floodplain vegetation in a standardised experiment. In Proceedings of the 6th EARSeL Workshop on Imaging Spectroscopy SIG, Tel Aviv, Israel, 16–19 March 2009. [Google Scholar]
- Hawari, A.H.; Mulligan, C.N. Effect of the presence of lead on the biosorption of copper, cadmium and nickel by anaerobic biomass. Process Biochem. 2007, 42, 1546–1552. [Google Scholar] [CrossRef]
- Lamhamdi, M.; El Galiou, O.; Bakrim, A.; Nóvoa-Muñoz, J.C.; Arias-Estévez, M.; Aarab, A.; Lafont, R. Effect of lead stress on mineral content and growth of wheat (Triticum aestivum) and spinach (Spinacia oleracea) seedlings. Saudi J. Biol. Sci. 2013, 20, 29–36. [Google Scholar] [CrossRef]
- Haider, S.; Kanwal, S.; Uddin, F.; Azmat, R. Phytotoxicity of Pb: II. Changes in Chlorophyll Absorption Spectrum due to Toxic Metal Pb Stress on Phaseolus mungo and Lens culinaris. Pak. J. Biol. Sci. 2006, 9, 2062–2068. [Google Scholar] [CrossRef]
- Akinci, I.E.; Akinci, S.; Yilmaz, K. Response of tomato (Solanum lycopersicum L.) to lead toxicity: Growth, element uptake, chlorophyll and water content. Afr. J. Agric. Res. 2010, 5, 416–423. [Google Scholar]
- Zarco-Tejada, P.J.; Miller, J.R.; Mohammed, G.H.; Noland, T.L.; Sampson, P.H. Estimation of chlorophyll fluorescence under natural illumination from hyperspectral data. Int. J. Appl. Earth Obs. Geoinf. 2001, 3, 321–327. [Google Scholar] [CrossRef]
- Zarco-Tejada, P.J.; Miller, J.R.; Mohammed, G.H.; Noland, T.L.; Sampson, P.H. Vegetation Stress Detection through Chlorophyll + Estimation and Fluorescence Effects on Hyperspectral Imagery. J. Environ. Qual. 2002, 31, 1433. [Google Scholar] [CrossRef]
- Zagajewski, B.; Lechnio, J.; Sobczak, M. Wykorzystanie Teledetekcji Hiperspektralnej w Analizie Roślinności Zanieczyszczonej Metalami Ciężkimi; Teledetekcja Środowiska: Warszawa, Poland, 2007; Volume 37, pp. 82–100. [Google Scholar]
- Grossmann, K.; Frankenberg, C.; Magney, T.S.; Hurlock, S.C.; Seibt, U.; Stutz, J. PhotoSpec: A new instrument to measure spatially distributed red and far-red solar-induced chlorophyll fluorescence. Remote Sens. Environ. 2018, 216, 311–327. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).