The Effects of Food Waste on Wildlife and Humans
Abstract
:1. Introduction
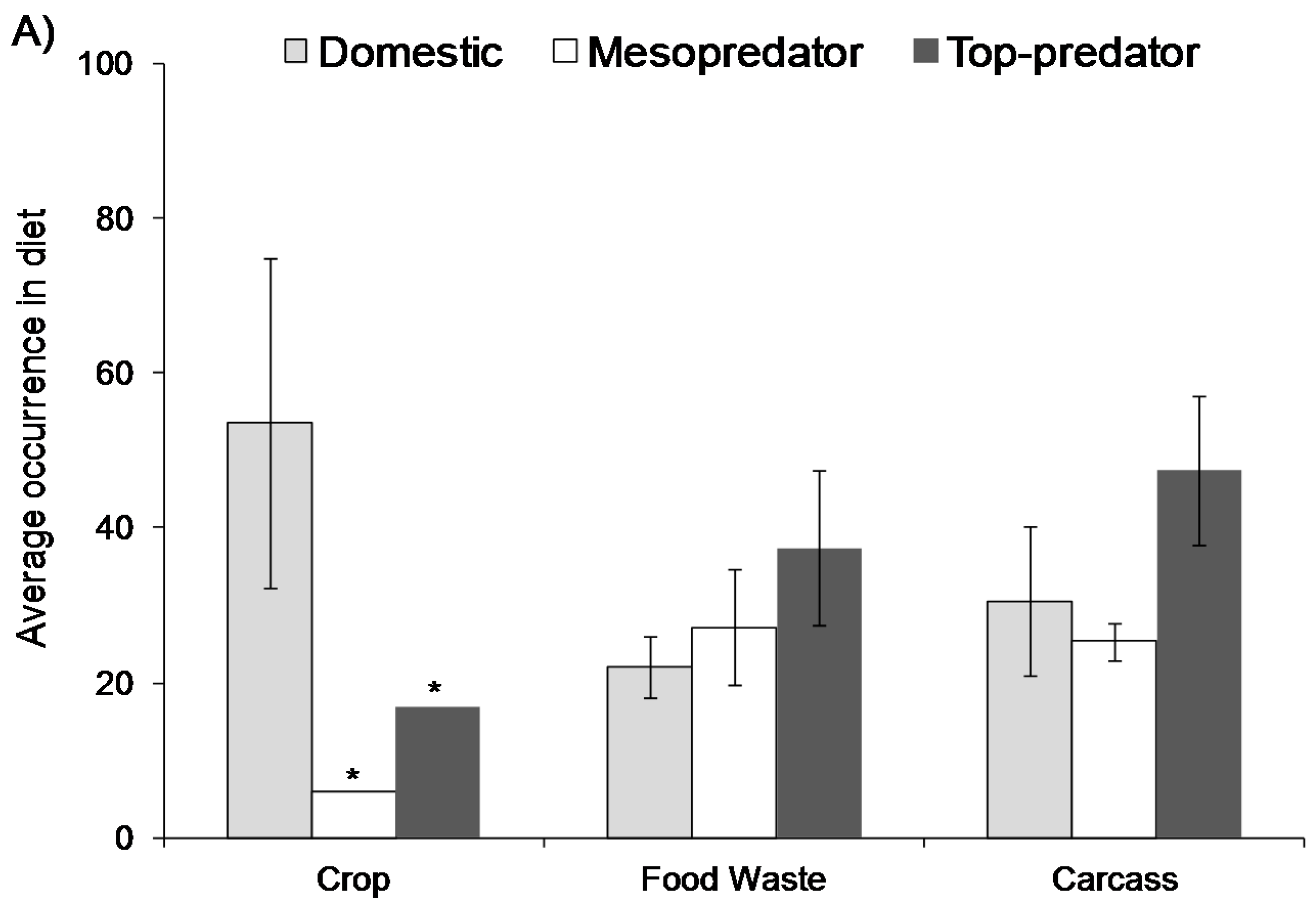
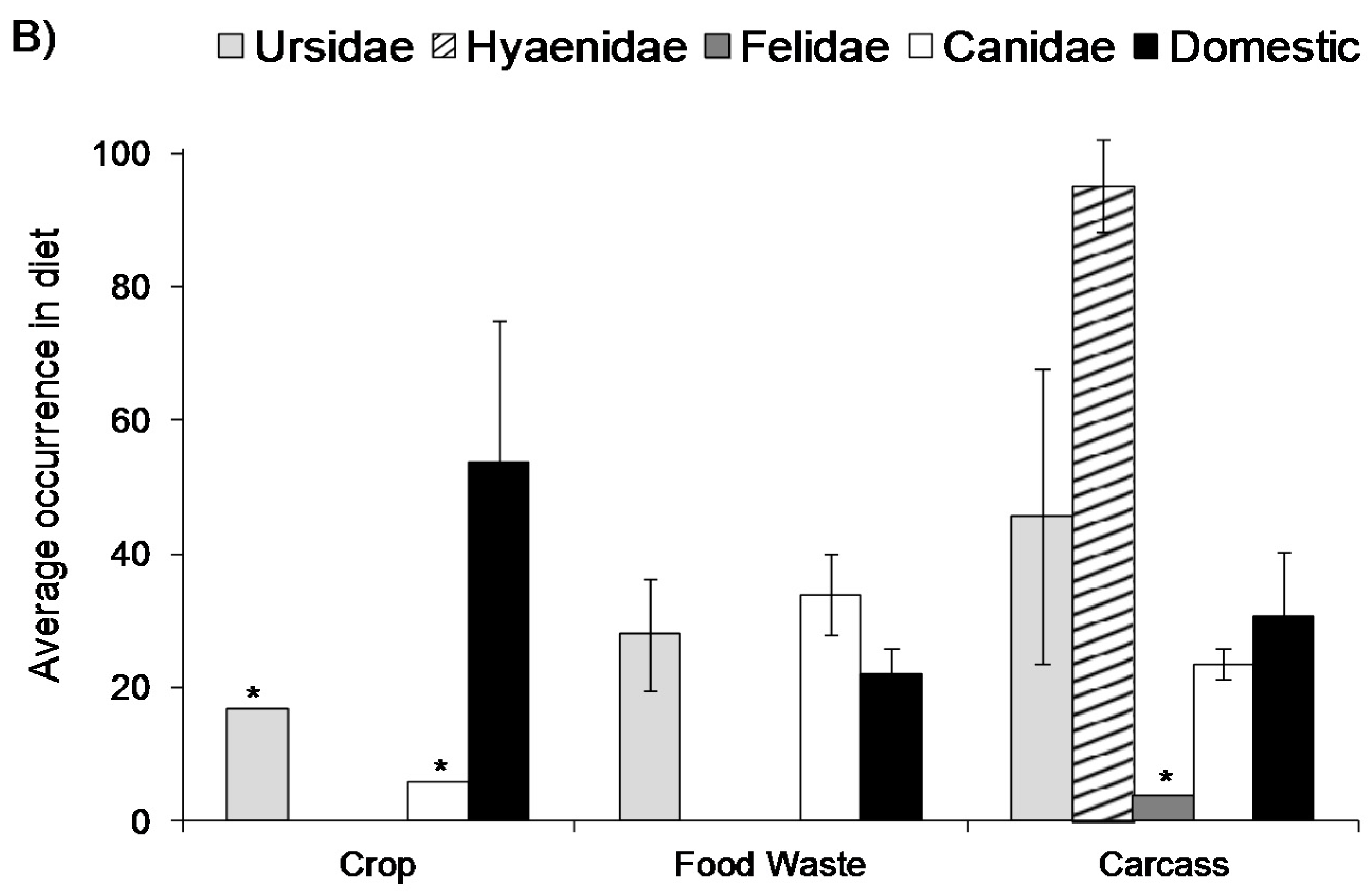
2. Extent of Use
3. Direct Effects on Wildlife Ecology and Behaviour
4. Indirect Effects on Other Species and Ecological Communities
5. Other Impacts to Wildlife Health
6. Human-Wildlife Conflict
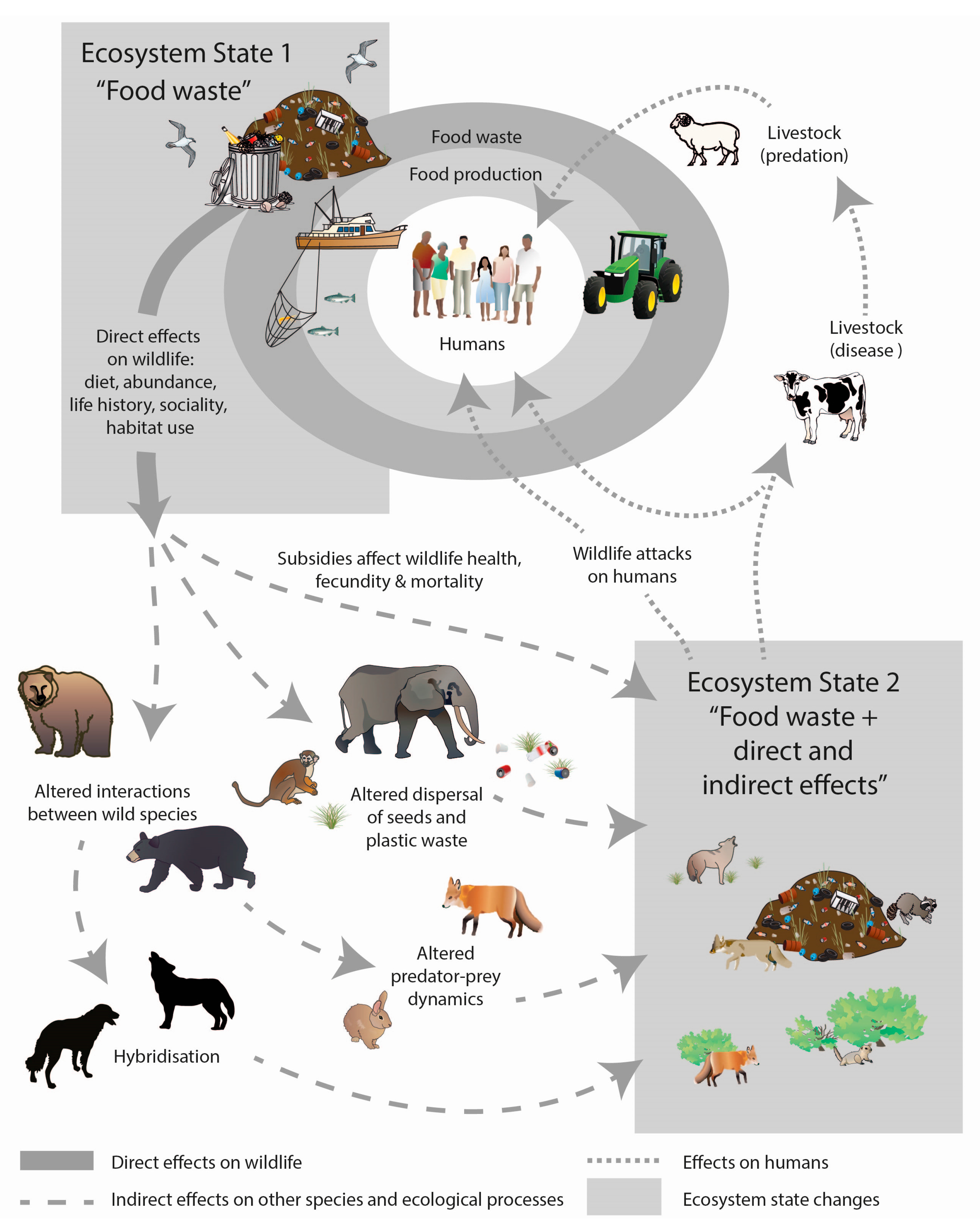
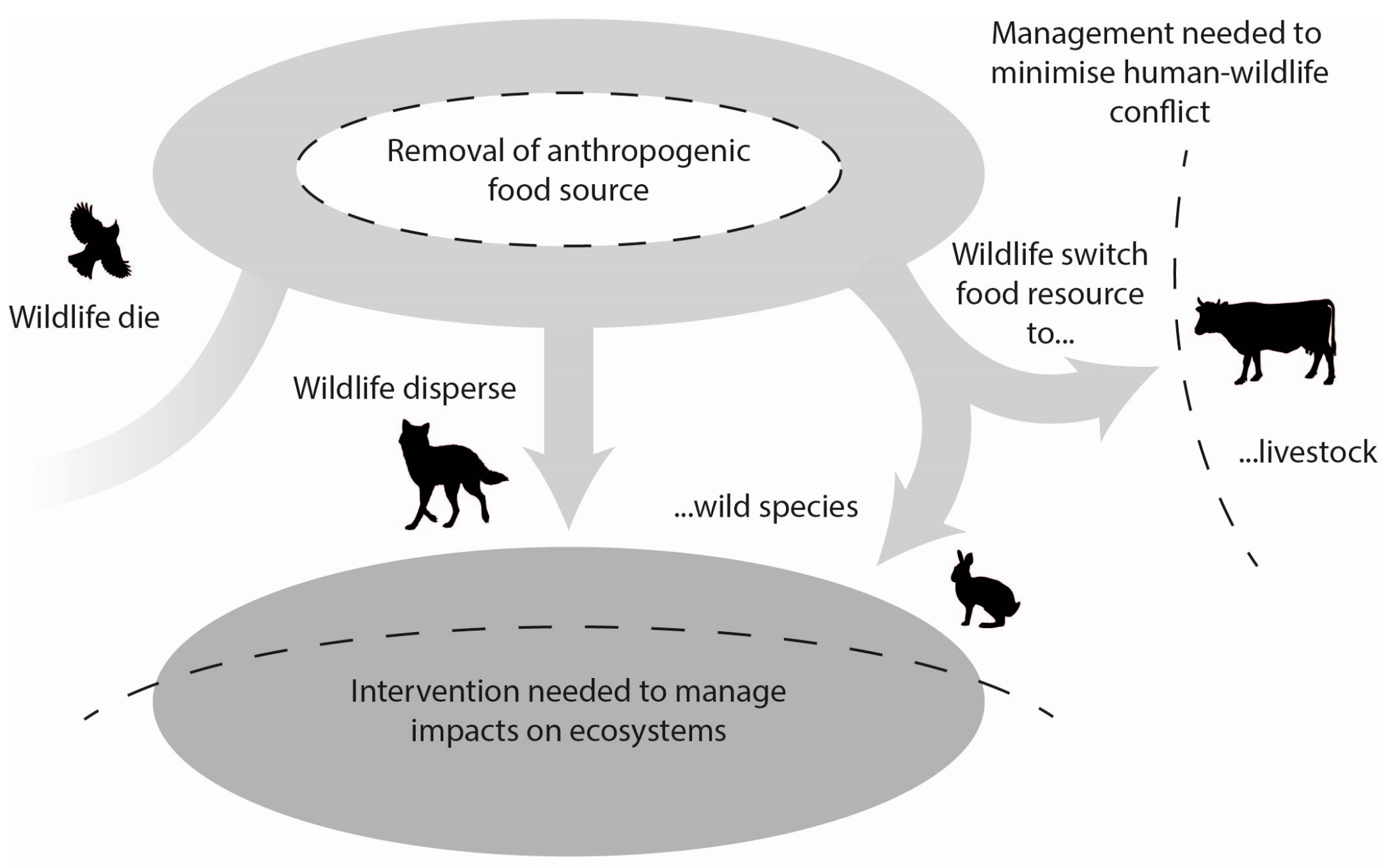
7. Conceptual Models
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Food and Agriculture Organization of the United Nations (FAO). Global Food Losses and Food Waste—Extent, Causes and Prevention; Food and Agriculture Organization of the United Nations: Rome, Italy, 2011. [Google Scholar]
- The World Bank. Food Price Watch, February 2014: Prices Decline at a Slower Pace; Focus on Food Loss and Waste. Available online: http://www.worldbank.org/en/topic/poverty/publication/food-price-watch-february-2014 (accessed on 11 March 2014).
- Machovina, B.; Feeley, K.J. Taking a bite out of biodiversity. Science 2014, 343, 838. [Google Scholar] [CrossRef] [PubMed]
- Godfray, H.C.J.; Beddington, J.R.; Crute, I.R.; Haddad, L.; Lawrence, D.; Muir, J.F.; Pretty, J.; Robinson, S.; Thomas, S.M.; Toulmin, C. Food security: The challenge of feeding 9 billion people. Science 2010, 327, 812–818. [Google Scholar] [CrossRef] [PubMed]
- Newsome, T.M.; Dellinger, J.A.; Pavey, C.R.; Ripple, W.J.; Shores, C.R.; Wirsing, A.J.; Dickman, C.R. The ecological effects of providing resource subsidies to predators. Glob. Ecol. Biogeogr. 2015, 24, 1–11. [Google Scholar] [CrossRef]
- Oro, D.; Genovart, M.; Tavecchia, G.; Fowler, M.S.; Martínez-Abraín, A. Ecological and evolutionary implications of food subsidies from humans. Ecol. Lett. 2013, 16, 1501–1514. [Google Scholar] [CrossRef] [PubMed]
- Kelleher, K. Discards in the World’s Marine Fisheries. An Update; FAO Fisheries Technical Paper, No. 470; Food and Agriculture Organization of the United Nations: Rome, Italy, 2005. [Google Scholar]
- Kolowski, J.M.; Holekamp, K.E. Effects of an open refuse pit on space use patterns of spotted hyenas. Afr. J. Ecol. 2007, 46, 341–349. [Google Scholar] [CrossRef]
- Johnson, H.E.; Breck, S.W.; Baruch-Mordo, S.; Lewis, D.L.; Lackey, C.W.; Wilson, K.R.; Broderick, J.; Mao, J.S.; Beckmann, J.P. Shifting perceptions of risk and reward: Dynamic selection for human development by black bears in the western United States. Biol. Conserv. 2015, 187, 164–172. [Google Scholar] [CrossRef]
- Baruch-Mordo, S.; Wilson, K.R.; Lewis, D.L.; Broderick, J.; Mao, J.S.; Breck, S.W. Stochasticity in natural forage production affects use of urban areas by black bears: Implications to management of human-bear conflicts. PLoS ONE 2014, 9, e85122. [Google Scholar] [CrossRef] [PubMed]
- Beckmann, J.P.; Berger, J. Rapid ecological and behavioural changes in carnivores: The responses of black bears (Ursus americanus) to altered food. J. Zool. 2003, 261, 207–212. [Google Scholar] [CrossRef]
- Camphuysen, C.J.; Calvo, B.; Durinck, J.; Ensor, K.; Follestad, A.; Furness, R.W.; Garthe, S.; Leaper, G.; Skov, H.; Tasker, M.L.; et al. Consumption of Discards by Seabirds in the North Sea; Final Report EC DG XIV Research Contract BIOECO/93/10; Netherlands Institute for Sea Research: Texel, The Netherlands, 1995. [Google Scholar]
- Macdonald, D.W. The ecology of carnivore social behaviour. Nature 1983, 301, 379–384. [Google Scholar] [CrossRef]
- Newsome, T.M.; Ballard, G.-A.; Fleming, P.J.S.; Ven, R.; Story, G.L.; Dickman, C.R. Human-resource subsidies alter the dietary preferences of a mammalian top predator. Oecologia 2014, 175, 139–150. [Google Scholar] [CrossRef] [PubMed]
- Newsome, T.M.; Stephens, D.; Ballard, G.-A.; Dickman, C.R.; Fleming, P.J.S. Genetic profile of dingoes (Canis lupus dingo) and free-roaming domestic dogs (C. l. familiaris) in the Tanami Desert, Australia. Wildl. Res. 2013, 40, 196–206. [Google Scholar] [CrossRef]
- Newsome, T.M.; Ballard, G.-A.; Dickman, C.R.; Fleming, P.J.S.; van de Ven, R. Home range, activity and sociality of a top predator, the dingo: A test of the Resource Dispersion Hypothesis. Ecography 2013, 36, 914–925. [Google Scholar] [CrossRef]
- Newsome, T.M.; Ballard, G.-A.; Dickman, C.R.; Fleming, P.J.S.; Howden, C. Anthropogenic resource subsidies determine space use by Australian arid zone dingoes: An improved resource selection modelling approach. PLoS ONE 2013, 8, e63931. [Google Scholar] [CrossRef] [PubMed]
- Craighead, J.J.; Summer, J.S.; Mitchell, J.A. The Grizzly Bears of Yellowstone: Their Ecology in the Yellowstone Ecosystem, 1959–1992; Island Press: Washington, DC, USA, 1995. [Google Scholar]
- Beckmann, J.P.; Lackey, C.W. Carnivores, urban landscapes, and longitudinal studies: A case history of black bears. Hum.-Wildl. Confl. 2008, 2, 168–174. [Google Scholar]
- Huber, D.; Kusak, J.; Frkovic, A. Traffic kills of brown bears in Gorski Kotar, Croatia. Ursus 1995, 10, 167–171. [Google Scholar]
- Gangadharan, A.; Pollock, S.; Gilhooly, P.; Friesen, A.; Dorsey, B.; St. Clair, C.C. Grain spilled from moving trains create a substantial wildlife attractant in protected areas. Anim. Conserv. 2017, in press. [Google Scholar] [CrossRef]
- Lewis, D.L.; Baruch-Mordo, S.; Wilson, K.R.; Breck, S.W.; Mao, J.S.; Broderick, J. Foraging ecology of black bears in urban environments: Guidance for human-bear conflict mitigation. Ecosphere 2015, 6, Article 141. [Google Scholar] [CrossRef]
- Herrero, S. Social behaviour of black bears at a garbage dump in Jasper National Park. In Bears: Their Biology and Management, Proceedings of the 5th International Conference on Bear Research and Management, Madison, WI, USA, 10–13 February 1980; International Association for Bear Research and Management: West Glacier, MT, USA, 1983; Volume 5, pp. 54–70. [Google Scholar]
- Silva-Rodríguez, E.A.; Sieving, K.E. Domestic dogs shape the landscape-scale distribution of a threatened forest ungulate. Biol. Conserv. 2012, 150, 103–110. [Google Scholar] [CrossRef]
- Lacerda, A.C.R.; Tomas, W.M.; Marinho-Filho, J. Domestic dogs as an edge effect in the Brasília National Park, Brazil: Interactions with native mammals. Anim. Conserv. 2009, 12, 477–487. [Google Scholar] [CrossRef]
- Yirga, G.; De Iongh, H.H.; Leirs, H.; Gebrihiwot, K.; Deckers, J.; Bauer, H. Adaptability of large carnivores to changing anthropogenic food sources: Diet change of spotted hyena (Crocuta crocuta) during Christian fasting period in northern Ethiopia. J. Anim. Ecol. 2012, 81, 1052–1055. [Google Scholar] [CrossRef] [PubMed]
- Osterback, A.-M.K.; Frechette, D.M.; Hayes, S.A.; Shaffer, S.A.; Moore, J.W. Long-term shifts in anthropogenic subsidies to gulls and implications for an imperiled fish. Biol. Conserv. 2015, 191, 606–613. [Google Scholar] [CrossRef]
- Sengupta, A.; McConkey, K.R.; Radhakrishna, S. Primates, provisioning and plants: Impacts of human cultural behaviours on primate ecological functions. PLoS ONE 2015, 10, e0140961. [Google Scholar] [CrossRef] [PubMed]
- Newsome, T.M.; Ripple, W.J. A continental scale trophic cascade from wolves through coyotes to foxes. J. Anim. Ecol. 2015, 84, 49–59. [Google Scholar] [CrossRef] [PubMed]
- VonHoldt, B.M.; Kays, R.; Pollinger, J.P.; Wayne, R.K. Admixture mapping identifies introgressed genomic regions in North American canids. Mol. Ecol. 2016, 25, 2443–2453. [Google Scholar] [CrossRef] [PubMed]
- Newsome, T.M.; Fleming, P.J.S.; Dickman, C.R.; Doherty, T.S.; Ripple, W.J.; Ritchie, E.G.; Wirsing, A.J. Making a new dog? BioScience 2017, 67, 374–381. [Google Scholar] [CrossRef]
- Vilà, C.; Walker, C.; Sundqvist, A.-K.; Flagstad, Ø.; Andersone, Z.; Casulli, A.; Kojola, I.; Valdmann, H.; Halverson, J.; Ellegren, H. Combined use of maternal, paternal and bi-parental genetic markers for the identification of wolf-dog hybrids. Heredity 2003, 90, 17–24. [Google Scholar] [CrossRef] [PubMed]
- Gottelli, D.; Sillero-Zubiri, C.; Applebaum, G.D.; Roy, M.S.; Girman, D.J.; Garcia-Moreno, J.; Ostrander, E.A.; Wayne, R.K. Molecular genetics of the most endangered canid: The Ethiopian wolf Canis simensis. Mol. Ecol. 1994, 3, 301–312. [Google Scholar] [CrossRef] [PubMed]
- Stephens, D.; Wilton, A.N.; Fleming, P.J.S.; Berry, O. Death by sex in an Australian icon: A continent-wide survey reveals extensive hybridization between dingoes and domestic dogs. Mol. Ecol. 2015, 24, 5643–5656. [Google Scholar] [CrossRef] [PubMed]
- Beaumont, M.; Barratt, E.M.; Gottelli, D.; Kitchener, A.C.; Daniels, M.J.; Pritchard, J.K.; Bruford, M.W. Genetic diversity and introgression in the Scottish wildcat. Mol. Ecol. 2001, 10, 319–336. [Google Scholar] [CrossRef] [PubMed]
- Laurenson, K.; Sillero-Zubiri, C.; Thompson, H.; Shiferaw, F.; Thirgood, S.; Malcolm, J. Disease as a threat to endangered species: Ethiopian wolves, domestic dogs and canine pathogens. Anim. Conserv. 1998, 1, 273–280. [Google Scholar] [CrossRef]
- Woodroffe, R.; Ginsberg, J.R. Conserving the African wild dog Lycaon pictus. I. Diagnosing and treating causes of decline. Oryx 1999, 33, 132–142. [Google Scholar] [CrossRef]
- Prange, S.; Gehrt, S.D.; Wiggers, E.P. Demographic factors contributing to high raccoon densities in urban landscapes. J. Wildl. Manag. 2003, 67, 324–333. [Google Scholar] [CrossRef]
- Gall, S.C.; Thompson, R.C. The impact of debris on marine life. Mar. Pollut. Bull. 2015, 92, 170–179. [Google Scholar] [CrossRef] [PubMed]
- Silva, J.A.; Talamoni, S.A. Diet adjustments of maned wolves, Chrysocyon brachyurus (Illiger) (Mammalia, Canidae), subjected to supplemental feeding in a private natural reserve, Southeastern Brazil. Rev. Bras. Zool. 2003, 20, 339–345. [Google Scholar] [CrossRef]
- Aragona, M.; Setz, E.Z.F. Diet of the maned wolf, Chrysocyon brachyurus (Mammalia: Canidae), during wet and dry seasons at Ibitipoca State Park, Brazil. J. Zool. 2001, 254, 131–136. [Google Scholar] [CrossRef]
- Joshi, R. Evaluating the impact of human activities during the Maha–Kumbh 2010 fair on elephants in the Shivalik elephant reserve. Trop. Nat. Hist. 2013, 13, 107–129. [Google Scholar]
- Mendis, R. Elephants Exposed to Rotting Garbage. Available online: http://cea.nsf.ac.lk/bitstream/handle/1/14157/E-2009-Nov-01-Sunday-Leader.pdf?sequence=2&isAllowed=y (accessed on 4 July 2016).
- Home, C.; Pal, R.; Sharma, R.K.; Suryawanshi, K.R.; Bhatnagar, Y.V.; Vanak, A.T. Commensal in conflict: Livestock depredation patterns by free-ranging domestic dogs in the Upper Spiti Landscape, Himachal Pradesh, India. Ambio 2017, in press. [Google Scholar] [CrossRef] [PubMed]
- Cozza, K.; Fico, R.; Battistini, M.-L.; Rogers, E. The damage-conservation interface illustrated by predation on domestic livestock in central Italy. Biol. Conserv. 1996, 78, 329–336. [Google Scholar] [CrossRef]
- Orams, M.B. Feeding wildlife as a tourism attraction: A review of issues and impacts. Tour. Manag. 2002, 23, 281–293. [Google Scholar] [CrossRef]
- Almeida Cunha, A. Negative effects of tourism in a Brazilian Atlantic forest National Park. J. Nat. Conserv. 2010, 18, 291–295. [Google Scholar] [CrossRef]
- Packer, C. Is India’s pride actually African? Science 2013, 340, 1289–1290. [Google Scholar] [CrossRef]
- Burns, G.L.; Howard, P. When wildlife tourism goes wrong: A case study of stakeholder and management issues regarding Dingoes on Fraser Island, Australia. Tour. Manag. 2003, 24, 699–712. [Google Scholar] [CrossRef]
- Herrero, S.; Fleck, S. Injury to people inflicted by black, grizzly or polar bears: Recent trends and new insights. In Bears: Their Biology and Management, Proceedings of the 8th International Conference on Bear Research and Management, Victoria, BC, Canada, 20–25 February 1989; International Association for Bear Research and Management: West Glacier, MT, USA, 1990; Volume 8, pp. 25–32. [Google Scholar]
- Alberti, M.; Correa, C.; Marzluff, J.M.; Hendry, A.P.; Palkovacs, E.P.; Gotanda, K.M.; Hunt, V.M.; Apgar, T.M.; Zhou, Y. Global urban signatures of phenotypic change in animal and plant populations. Proc. Natl. Acad. Sci. USA 2017, in press. [Google Scholar] [CrossRef]
- Lim, H.C.; Sodhi, N.S.; Brook, B.W.; Soh, M.C.K. Undesirable aliens: Factors determining the distribution of three invasive bird species in Singapore. J. Trop. Ecol. 2003, 19, 685–695. [Google Scholar] [CrossRef]
- Bevan, R.D.R. The cost of feral pigeon 1990. In British Ornithological Union, Proceedings of a Symposium; ADAS/British Ornithologists’ Union: London, UK, 1990. [Google Scholar]
- Horton, N.; Brough, T.; Rochard, J.B.A. The importance of refuse tips to gulls wintering in an inland area of south-east England. J. Appl. Ecol. 1983, 20, 751–765. [Google Scholar] [CrossRef]
- Thorpe, J. Fatalities and Destroyed Civil Aircraft Due to Bird Strikes, 1912–2002; International Bird Strike Committee: Warsaw, Poland, 2003. [Google Scholar]
- Bino, G.; Dolev, A.; Yosha, D.; Guter, A.; King, R.; Saltz, D.; Kark, S. Abrupt spatial and numerical responses of overabundant foxes to a reduction in anthropogenic resources. J. Appl. Ecol. 2010, 47, 1262–1271. [Google Scholar] [CrossRef]
- Gordon, I.J.; Altwegg, R.; Evans, D.M.; Ewen, J.G.; Johnson, J.A.; Pettorelli, N.; Young, J.K. Reducing agricultural loss and food waste: How will nature fare? Anim. Conserv. 2016, 19, 305–308. [Google Scholar] [CrossRef]
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Newsome, T.M.; Van Eeden, L.M. The Effects of Food Waste on Wildlife and Humans. Sustainability 2017, 9, 1269. https://doi.org/10.3390/su9071269
Newsome TM, Van Eeden LM. The Effects of Food Waste on Wildlife and Humans. Sustainability. 2017; 9(7):1269. https://doi.org/10.3390/su9071269
Chicago/Turabian StyleNewsome, Thomas M., and Lily M. Van Eeden. 2017. "The Effects of Food Waste on Wildlife and Humans" Sustainability 9, no. 7: 1269. https://doi.org/10.3390/su9071269
APA StyleNewsome, T. M., & Van Eeden, L. M. (2017). The Effects of Food Waste on Wildlife and Humans. Sustainability, 9(7), 1269. https://doi.org/10.3390/su9071269




