Microalgae Potential and Multiple Roles—Current Progress and Future Prospects—An Overview
Abstract
:1. Introduction
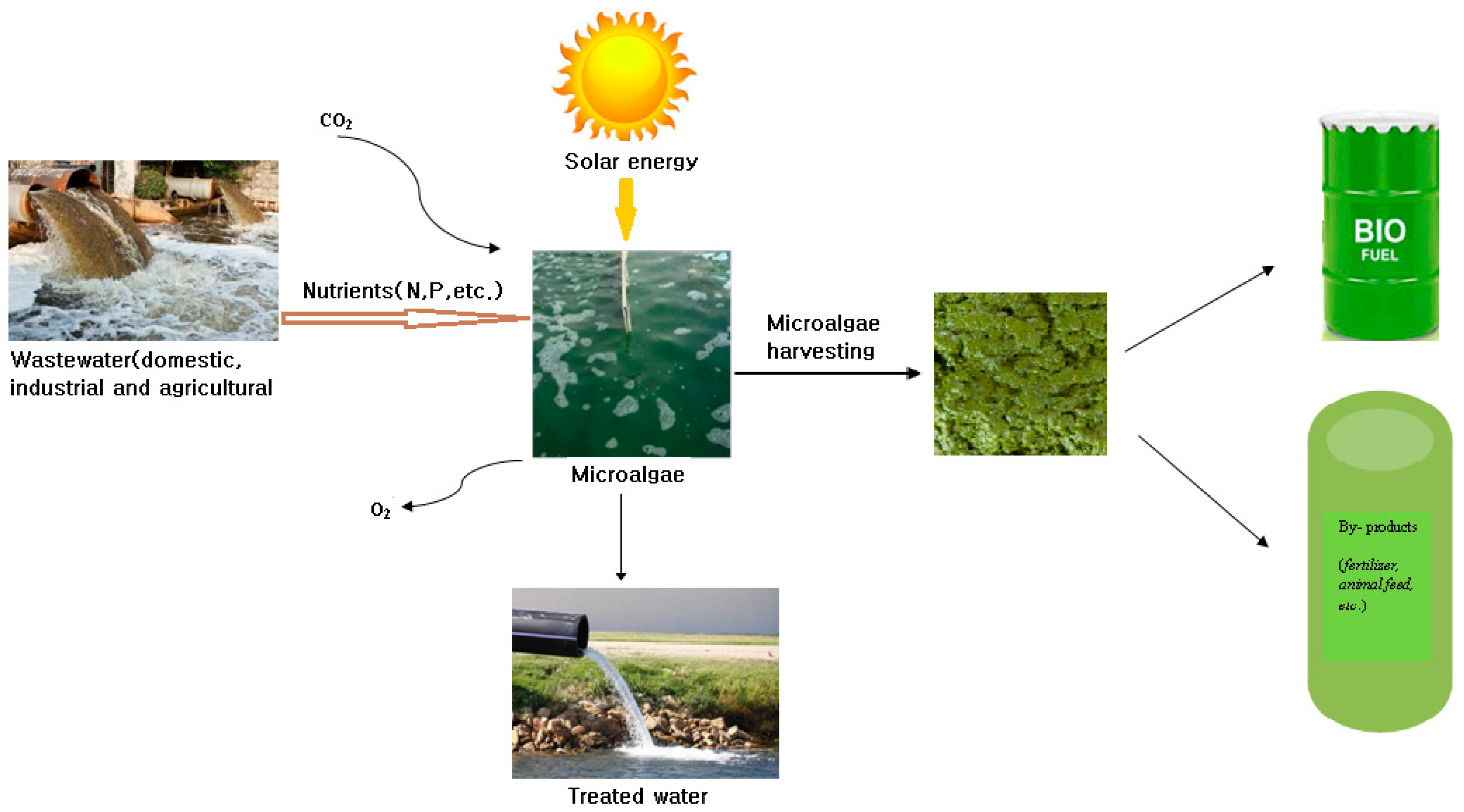
2. Microalgae: As Feedstock for Biofuel
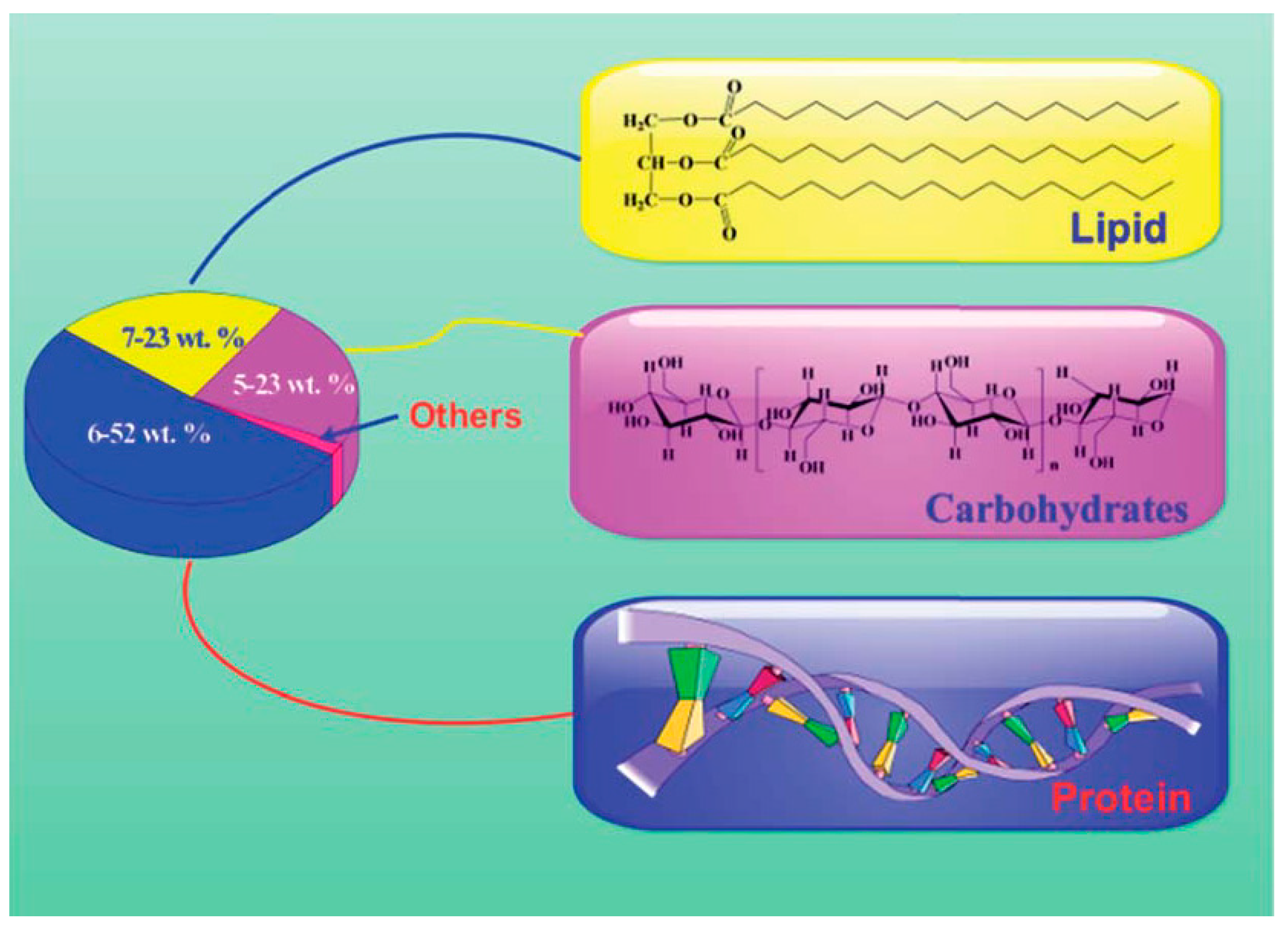
2.1. Biomass Composition of Microalgae
2.1.1. Carbohydrates
2.1.2. Proteins
2.1.3. Lipids
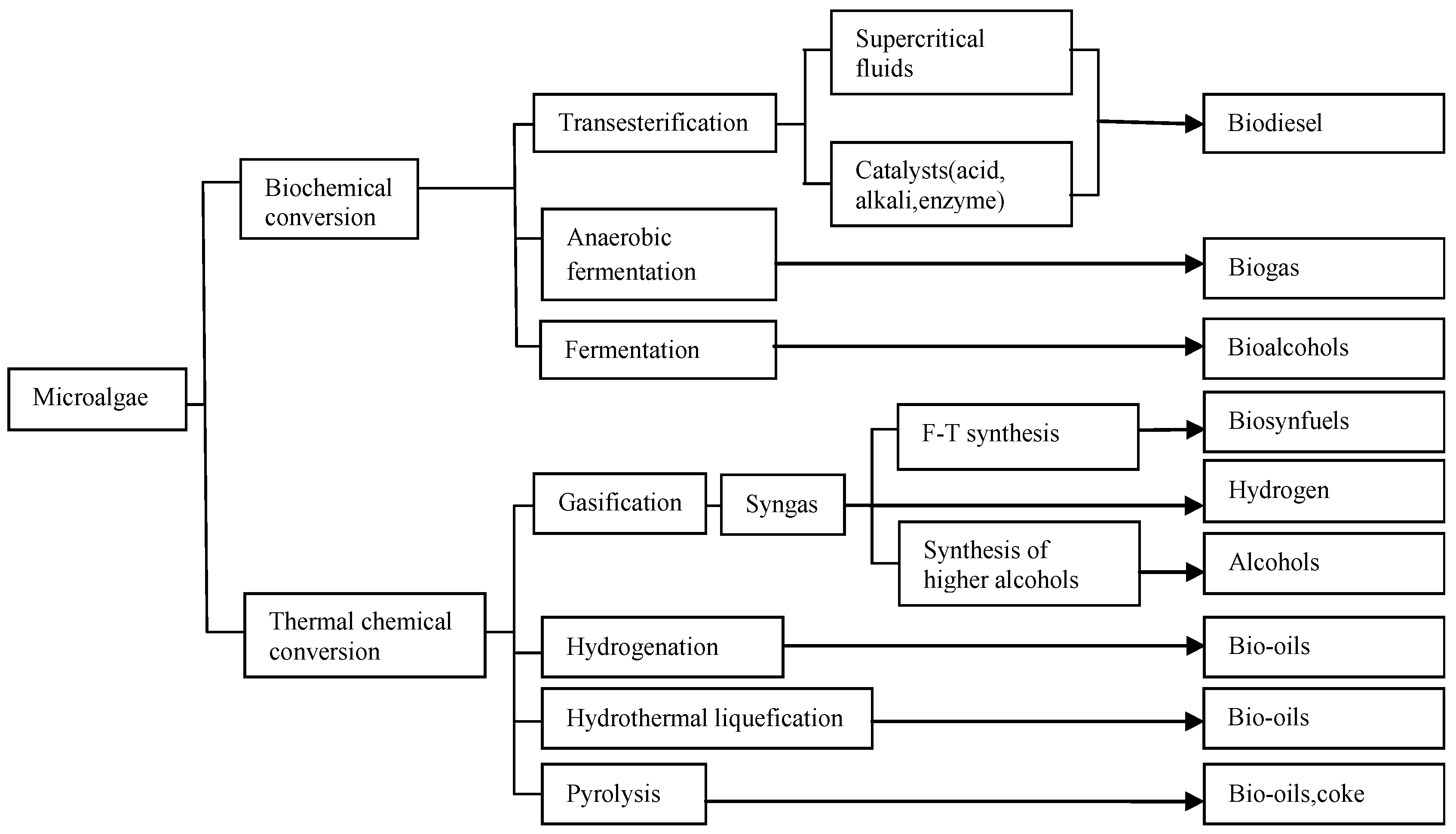
2.2. Biofuel Production from Microalgae
3. Microalgae Cultivation Techniques
3.1. Open Pond Culture Systems
3.2. Closed Photobioreactors
3.3. Culture Using Deep Sea Water
3.4. Co-Culture Methods
4. Novel Approaches for Harvesting of Microalgal Biomass
5. Technologies for Effective Lipid Extraction from Microalgae
6. Microalgae as a Feedstock for Value Added Products
7. Environmental Applications of Microalgae
7.1. Wastewater Treatment and Nutrient Removal
7.2. CO2 Sequestrations
8. Future Prospects
9. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Parsaeimehr, A.; Sun, Z.; Dou, X.; Chen, Y.F. Simultaneous improvement in production of microalgal biodiesel and high-value alpha-linolenic acid by a single regulator acetylcholine. Biotechnol. Biofuels 2015, 8, 11. [Google Scholar] [CrossRef] [PubMed]
- Food and Agriculture Organization of the United Nations (FAO). The State of Food Insecurity in the World; FAO: Rome, Italy, 2003. [Google Scholar]
- Demirbas, M.F. Biorefineries for biofuel upgrading: A critical review. Appl. Energy 2009, 86, S151–S161. [Google Scholar] [CrossRef]
- Lu, J.; Sheahan, C.; Fu, P.C. Metabolic engineering of algae for fourth generation biofuels production. Energy Environ. Sci. 2011, 4, 2451–2466. [Google Scholar] [CrossRef]
- Mooij, P.R.; Stouten, G.R.; Tamis, J.; van Loosdrecht, M.C.M.; Kleerebezem, R. Survival of the fattest. Energy Environ. Sci. 2013, 6, 3404–3406. [Google Scholar] [CrossRef]
- Gerardo, M.L.; Hende, S.V.; Vervaeren, H.; Coward, T.; Skill, S.C. Harvesting of microalgae within a biorefinery approach: A review of the developments and case studies from pilot-plants. Algal Res. 2015, 11, 248–262. [Google Scholar] [CrossRef]
- Barsanti, L.; Gualtieri, P. Algae: Anatomy, Biochemistry and Biotechnology; CRC Press, Taylor and Francis Group: Boca Raton, FL, USA, 2006. [Google Scholar]
- Maity, I.P.; Bundschuh, J.; Chen, C.Y.; Bhattacharaya, P. Microalgae for third generation biofuel production, mitigation of greenhouse gas emissions and wastewater treatment: Present and future perspectives—A mini review. Energy 2014, 78, 1–10. [Google Scholar] [CrossRef]
- Marchetti, J.; Bougaran, G.; Jauffrais, T.; Lefebvre, S.; Rouxel, C.; Saint-Jena, B.; Lukomska, E.; Robert, R.; Cadoret, J.P. Effects of blue light on the biochemical composition and photosynthetic activity of Isochrysis sp. (T-iso). J. Appl. Phycol. 2013, 25, 109–119. [Google Scholar] [CrossRef]
- Chen, Y.; Wu, Y.; Hua, D.; Li, C.; Harold, M.P.; Wang, J.; Wang, M. Thermochemical conversion of low-lipid microalgae for the production of liquid fuels: Challenges and opportunities. RSC Adv. 2015, 5, 18673–18701. [Google Scholar] [CrossRef]
- Gorgônio, C.M.S.; Aranda, D.A.G.; Couri, S. Morphological and chemical aspects of Chlorella pyrenoidosa, Dunaliella tertiolecta, Isochrysis galbana and Tetraselmis gracilis microalgae. Nat. Sci. 2013, 5, 783–791. [Google Scholar]
- Biller, P.; Ross, A.B. Pyrolysis GC–MS as a novel analysis technique to determine the biochemical composition of microalgae. Algal Res. 2014, 6, 91–97. [Google Scholar] [CrossRef]
- Priyadarshani, I.; Rath, B. Commercial and industrial applications of micro algae—A review. J. Algal Biomass Util. 2012, 3, 89–100. [Google Scholar]
- Markou, G.; Angelidaki, I.; Georgakakis, D. Bioethanol production by carbohydrate-enriched Arthrospira (Spirulina) platensis. Energies 2013, 6, 3937–3950. [Google Scholar] [CrossRef]
- Williams, P.J.L.; Laurens, L.M.L. Microalgae as biodiesel and biomass feedstocks: Review and analysis of the biochemistry, energetics and economics. Energy Environ. Sci. 2010, 3, 554–590. [Google Scholar] [CrossRef]
- Lee, J.Y.; Yoo, C.; Jun, S.Y.; Ahn, C.Y.; Oh, H.M. Comparison of several methods for effective lipid extraction from microalgae. Bioresour. Technol. 2010, 101, S75–S77. [Google Scholar] [CrossRef] [PubMed]
- Bi, Z.; He, B.B. Characterization of microalgae for the purpose of biofuel production. Trans. ASABE 2013, 56, 1529–1539. [Google Scholar]
- Hernandez, E. Lipids, Pharmaceutical and Cosmetic Use. In Kirk-Othmer Ecyclopedia of Chemical Technology; Wiley: New York, NY, USA, 2012. [Google Scholar]
- Sharma, Y.C.; Singh, B.; Korstad, J. A critical review on recent methods used for economically viable and eco-friendly development of microalgae as a potential feedstock for synthesis of biodiesel. Green Chem. 2011, 13, 2993–3006. [Google Scholar] [CrossRef]
- Bellou, S.; Aggelis, G. Biochemical activities in Chlorella sp. and Nannochloropsis salina during lipid and sugar synthesis in a lab-scale open pond simulating reactor. J. Biotechnol. 2012, 164, 318–329. [Google Scholar]
- Halim, R.; Danquah, M.K.; Webley, P.A. Extraction of oil from microalgae for biodiesel production: A review. Biotechnol. Adv. 2012, 30, 709–732. [Google Scholar] [CrossRef] [PubMed]
- Ansari, F.A.; Shriwastav, A.; Gupta, S.K.; Rawat, I.; Guldhe, A.; Bux, F. Lipid extracted algae as a source for protein and reduced sugar: A step closer to the biorefinery. Bioresour. Technol. 2015, 179, 559–564. [Google Scholar] [CrossRef]
- Gupta, S.K.; Kumar, N.M.; Guldhe, A.; Ansari, F.A.; Rawat, I.; Kanney, K.; Bux, F. Design and development of polyamine polymer for harvesting microalgae for biofuels. Eng. Conserv. Manag. 2014, 84, 537–544. [Google Scholar] [CrossRef]
- Kim, J.K.; Um, B.; Kim, T.H. Bioethanol production from micro-algae, Schizocytrium sp., using hydrothermal treatment and biological conversion. Korean J. Chem. Eng. 2012, 29, 209–214. [Google Scholar] [CrossRef]
- Biomass Research and Development Technical Advisory Committee. Roadmap for Biomass Technologies in the United States; Department of Energy, Washington State University: Washington, DC, USA, 2002. [Google Scholar]
- Tsukahara, K.; Sawayama, S. Liquid fuel production using microalgae. J. Jpn. Pet. Inst. 2005, 48, 251–259. [Google Scholar] [CrossRef]
- Chisti, Y. Biodiesel from microalgae. Biotechnol. Adv. 2007, 25, 294–306. [Google Scholar] [CrossRef] [PubMed]
- Deng, X.; Li, Y.; Fei, X. Microalgae: A promising feedstock for biodiesel. Afr. J. Microbiol. Res. 2009, 3, 1008–1014. [Google Scholar]
- Hughes, A.D.; Kelly, M.S.; Black, K.D.; Stanley, M.S. Biogas from microalgae: Is it time to revisit the idea. Biotechnol. Biofuel 2012, 5, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Zeng, D.; Li, R.; Yan, T.; Fang, T. Perspectives and advances of microalgal biodiesel production with supercritical fluid technology. RSC Adv. 2014, 4, 39771–39781. [Google Scholar] [CrossRef]
- Ugwu, C.U.; Aoyagi, H.; Uchiyama, H. Photobioreactors for mass cultivation of algae. Bioresour. Technol. 2008, 99, 4021–4028. [Google Scholar] [CrossRef] [PubMed]
- Borowitzka, M.A. Commercial production of microalgae: Ponds, tanks, tubes and fermenters. J. Biotechnol. 1999, 70, 313–321. [Google Scholar] [CrossRef]
- De Godos, I.; Mendoza, J.L.; Acién, F.G.; Molina, E.; Banks, C.J.; Heaven, S. Evaluation of carbon dioxide mass transfer in raceway reactors for microalgae culture using flue gases. Bioresour. Technol. 2014, 153, 307–314. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.H.; Chen, C.J.; Hung, C.I.; Shen, C.H.; Hsu, H.W. A comparison of gasification phenomena among raw biomass, torrefied biomass and coal in an entrained-flow reactor. Appl. Energy 2013, 112, 421–430. [Google Scholar] [CrossRef]
- Tan, C.H.; Show, P.L.; Chang, J.S.; Ling, T.C.; Lan, J.C.W. Novel approaches of producing bioenergies from microalgae: A recent review. Biotechnol. Adv. 2015, 33, 1219–1227. [Google Scholar] [CrossRef] [PubMed]
- Nakasone, K.; Ikegami, A.; Kato, C.; Usami, R.; Horikoshi, K. Mechanisms of gene expression controlled by pressure in deep-sea microorganisms. Extremophiles 1998, 2, 149–154. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez, L.E.; Bashan, Y. Growth promotion of the microalgae Chlorella vulgaris when coimmobilized and cocultured in alginate beads with the plant growth-promoting bacteria Azospirillum brasilense. Appl. Environ. Microbiol. 2000, 66, 1537–1541. [Google Scholar] [CrossRef]
- De-Bashan, L.E.; Bashan, Y. Joint immobilization of plant growth-promoting bacteria and green microalgae in alginate beads as an experimental model for studying plant–bacterium interactions. Appl. Environ. Microbiol. 2008, 74, 6797–6802. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Li, Y.; Hu, X.; Su, W.; Zhong, M. Combined enzymatic and mechanical cell disruption and lipid extraction of green alga Neochloris oleoabundans. Int. J. Mol. Sci. 2015, 16, 7707–7722. [Google Scholar] [CrossRef] [PubMed]
- Wrede, D.; Taha, M.; Miranda, A.F.; Kadali, K.; Stevenson, T.; Ball, A.S.; Mouradov, A. Co-cultivation of fungal and microalgal cells as an efficient system for harvesting microalgal cells, lipid production and wastewater treatment. PLoS ONE 2014, 9, e113497. [Google Scholar] [CrossRef] [PubMed]
- Yeap, K.L.; Teng, T.T.; Poh, B.T.; Morad, N.; Lee, K.E. Preparation and characterization of coagulation/flocculation behavior of a novel inorganic–organic hybrid polymer for reactive and disperse dyes removal. Chem. Eng. J. 2014, 243, 305–314. [Google Scholar] [CrossRef]
- He, Z.; Siripornadulsil, S.; Sayre, R.T.; Traina, S.J.; Weavers, L.K. Removal of mercury from sediment by ultrasound combined with biomass (transgenic Chlamydomonas reinhardtii). Chemosphere 2011, 83, 1249–1254. [Google Scholar] [CrossRef] [PubMed]
- Misra, R.; Guldhe, A.; Singh, P.; Rawat, I.; Bux, F. Electrochemical harvesting process for microalgae by using nonsacrificial carbon electrode: A sustainable approach for biodiesel production. Chem. Eng. J. 2014, 255, 327–333. [Google Scholar] [CrossRef]
- Xia, C.; Zhang, J.; Zhang, W.; Hu, B. A new cultivation method for microbial oil production: Cell pelletization and lipid accumulation by Mucor circinelloides. Biotechnol. Biofuel 2011, 4, 15. [Google Scholar] [CrossRef] [PubMed]
- Johnson, M.B.; Wen, Z. Development of an attached microalgal growth system for biofuel production. Appl. Microbiol. Biotechnol. 2010, 85, 525–534. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Hu, B. A novel method to harvest microalgae via co-culture of filamentous fungi to form cell pellets. Bioresour. Technol. 2012, 114, 529–535. [Google Scholar] [CrossRef] [PubMed]
- King, P.M. The Use of Ultrasound on the Extraction of Microalgal Lipids. Ph.D. Thesis, Coventry University, Coventry, UK, 2014. [Google Scholar]
- Yu, X.; Dong, T.; Zheng, Y.; Miao, C.; Chssen, S. Investigations on cell disruption of oleaginous microorganisms: Hydrochloric acid digestion is an effective method for lipid extraction. Eur. J. Lipid Sci. Technol. 2015, 117, 730–777. [Google Scholar] [CrossRef]
- Wang, Y.; Yang, Y.; Ma, F.; Xuan, L.; Xu, Y.; Huo, H.; Zhou, D.; Dong, S. Optimization of Chlorella vulgaris and bioflocculant producing bacteria co-culture: Enhancing microalgae harvesting and lipid content. Lett. Appl. Microbiol. 2015, 60, 497–503. [Google Scholar] [CrossRef] [PubMed]
- Günerken, E.; D’Hondt, E.; Eppink, M.H.M.; Garcia-Gonzalez, L.; Elst, K.; Wijffels, R.H. Cell disruption for microalgae biorefineries. Biotechnol. Adv. 2015, 33, 243–260. [Google Scholar] [CrossRef] [PubMed]
- Rakesh, S.; Dhar, D.W.; Prasanna, R.; Anil, K.; Saxena, A.K.; Saha, S.; Shukla, M.; Sharma, K. Cell disruption methods for improving lipid extraction efficiency in unicellular microalgae. Eng. Life Sci. 2015, 15, 443–447. [Google Scholar] [CrossRef]
- Balasubramanian, S.; Allen, J.D.; Kanitkar, A.; Boldor, D. Oil extraction from Scenedesmus obliquus using a continuous microwave system-design, optimization, and quality characterization. Bioresour. Technol. 2011, 102, 3396–3403. [Google Scholar] [CrossRef] [PubMed]
- Singh, J.; Gu, S. Commercialization potential of microalgae for biofuels production. Renew. Sustain. Energy Rev. 2010, 14, 2596–2610. [Google Scholar] [CrossRef]
- Koller, M.; Muhr, A.; Braunegg, G. Microalgae as versatile cellular factories for valued products. Algal Res. 2014, 6, 52–63. [Google Scholar] [CrossRef]
- Spolaore, P.; Joannis-Cassan, C.; Duran, E.; Isambert, A. Commercial applications of microalgae. J. Biosci. Bioeng. 2006, 101, 87–96. [Google Scholar] [CrossRef] [PubMed]
- He, M.L.; Hollwich, W.; Rambeck, W.A. Supplementation of algae to the diet of pigs: A new possibility to improve the iodine content in the meat. J. Anim. Physiol. Anim. Nutr. 2002, 86, 97–104. [Google Scholar] [CrossRef]
- Garofalo, R. Algae and aquatic biomass for a sustainable production of 2nd generation fuels. AquaFUELS Taxon. Biol. Biotech. 2009, 1–258. Available online: http://cordis.europa.eu/result/rcn/53073_en.html (accedded on 22 September 2016). [Google Scholar]
- Lum, K.K.; Kim, J.; Lei, X.G. Dual potential of microalgae as a sustainable biofuel feedstock and animal feed. J. Anim. Sci. Biotechnol. 2013, 4, 53. [Google Scholar] [CrossRef] [PubMed]
- Pan, J.F.; Lin, R.G.; Ma, L. A review of heavy metal adsorption by marine algae. Chin. J. Oceanol. Limnol. 2000, 18, 260–264. [Google Scholar]
- Sialve, B.; Bernet, N.; Bernard, O. Anaerobic digestion of microalgae as a necessary step to make microalgal biodiesel sustainable. Biotechnol. Adv. 2009, 27, 409–416. [Google Scholar] [CrossRef] [PubMed]
- World Bank. World Development Indicators; World Bank Publications: Washington, DC, USA, 2012. [Google Scholar]
- Gupta, S.; Ansari, F.; Shriwastav, A.; Sahoo, N.; Rawat, I.; Bux, F. Dual role of Chlorella sorokiniana and Scenedesmus obliquus for comprehensive wastewater treatment and biomass production for bio-fuels. J. Clean. Prod. 2016, 115, 255–264. [Google Scholar] [CrossRef]
- Cho, S.; Luong, T.T.; Lee, D.; Oh, Y.K.; Lee, T. Reuse of effluent water from a municipal wastewater treatment plant in microalgae cultivation for biofuel production. Bioresour. Technol. 2011, 102, 8639–8645. [Google Scholar] [CrossRef] [PubMed]
- Dalrymple, O.K.; Halfhide, T.; Udom, I.; Gilles, B.; Wolan, J.; Zhang, Q.; Ergas, S. Wastewater use in algae production for generation of renewable resources: A review and preliminary results. Aquat. Biosyst. 2013, 9, 2. [Google Scholar] [CrossRef] [PubMed]
- Attasat, S.; Wanichpongpan, P.; Ruenglertpanyakul, W. Cultivation of microalgae (Oscillatoria okeni and Chlorella vulgaris) using tilapia-pond effluent and a comparison of their biomass removal efficiency. Water Sci. Technol. 2013, 67, 271–277. [Google Scholar] [CrossRef] [PubMed]
- Chen, G.; Zhao, L.; Qi, Y. Enhancing the productivity of microalgae cultivated in wastewater toward biofuel production: A critical review. Appl. Energy 2015, 137, 282–291. [Google Scholar] [CrossRef]
- Sriram, S.; Seenivasan, R. Microalgae cultivation in wastewater for nutrient removal. J. Algal Biomass Util. 2012, 3, 9–13. [Google Scholar]
- Perez-Garcia, O.; Escalante, F.M.E.; de-Bashan, L.E.; Bashan, Y. Heterotrophic cultures of microalgae: Metabolism and potential products. Water Res. 2011, 45, 11–36. [Google Scholar] [CrossRef] [PubMed]
- Richmond, A. Handbook of Microalgal Culture: Biotechnology and Applied Phycology; Blackwel Science Ltd.: Oxford, UK, 2004. [Google Scholar]
- Oliver, R.L.; Ganf, G.G. Freshwater blooms. In The Ecology of Cyanobacteria: Their Diversity in Time and Space; Whitton, B.A., Potts, M., Eds.; Kluwer: Dordrecht, The Netherlands, 2002; pp. 149–194. [Google Scholar]
- Powell, N.; Shilton, A.; Chisti, Y.; Pratt, S. Towards a luxury uptake process via microalgae defining the polyphosphate dynamics. Water Res. 2009, 43, 4207–4213. [Google Scholar] [CrossRef] [PubMed]
- Zhou, G.J.; Ying, G.G.; Liu, S.; Zhou, L.J.; Chen, Z.F.; Peng, F.Q. Simultaneous removal of inorganic and organic compounds in wastewater by freshwater green microalgae. Environ. Sci. Process Impacts 2014, 16, 2018–2027. [Google Scholar] [CrossRef] [PubMed]
- Edberg, S.C.; Rice, E.W.; Karlin, R.J.; Allen, M.J. Escherichia coli: The best biological drinking water indicator for public health protection. Symp. Ser. Soc. Appl. Microbiol. 2000, 29, 106S–116S. [Google Scholar] [CrossRef]
- Kiso, Y.; Jung, Y.J.; Park, M.S.; Wang, W.; Shimase, M.; Yamada, T.; Min, K.S. Coupling of sequencing batch reactor and mesh filtration: Operational parameters and wastewater treatment performance. Water Res. 2005, 39, 4887–4898. [Google Scholar] [CrossRef] [PubMed]
- Ansa, E.D.O.; Lubberding, H.J.; Gijzen, H.J. The effect of algal biomass on the removal of faecal coliform from domestic wastewater. Appl. Water Sci. 2012, 37, 317–324. [Google Scholar] [CrossRef]
- Ansa, E.; Lubberding, H.; Ampofo, J.; Gijzen, H. The role of algae in the removal of Escherichia coli in a tropical eutrophic lake. Ecol. Eng. 2011, 37, 317–324. [Google Scholar] [CrossRef]
- Marchello, A.E.; Lombardi, A.T.; Dellamano-Oliveira, M.J.; de Souza, C.W. Microalgae population dynamics in photobioreactors with secondary sewage effluent as culture medium. Braz. J. Microbiol. 2015, 46, 75–84. [Google Scholar] [CrossRef] [PubMed]
- Milano, J.; Ong, H.C.; Masjuki, H.H.; Chong, W.T.; Lam, M.K.; Loh, P.K.; Vellayan, V. Microalgae biofuels as an alternative to fossil fuel for power generation. Renew. Sustain. Energy Rev. 2016, 58, 180–197. [Google Scholar] [CrossRef]
- Bhola, V.; Swalaha, F.; Kumar, R.R.; Singh, M.; Bux, F. Overview of the potential of microalgae for CO2 sequestration. Int. J. Environ. Sci. Technol. 2014, 11, 2103–2118. [Google Scholar] [CrossRef]
- Brennan, L.; Owende, P. Biofuels from microalgae—A review of technologies for production, processing, and extractions of biofuels and co-products. Renew. Sustain. Energy Rev. 2010, 14, 557–577. [Google Scholar] [CrossRef]
- Kao, C.Y.; Chen, T.Y.; Chang, YB.; Chiu, T.W.; Lin, H.Y.; Chen, C.D.; Chang, J.S.; Lin, C.S. Utilization of carbon dioxide in industrial flue gases for the cultivation of microalga Chlorella sp. Bioresour. Technol. 2014, 166, 485–493. [Google Scholar] [CrossRef] [PubMed]

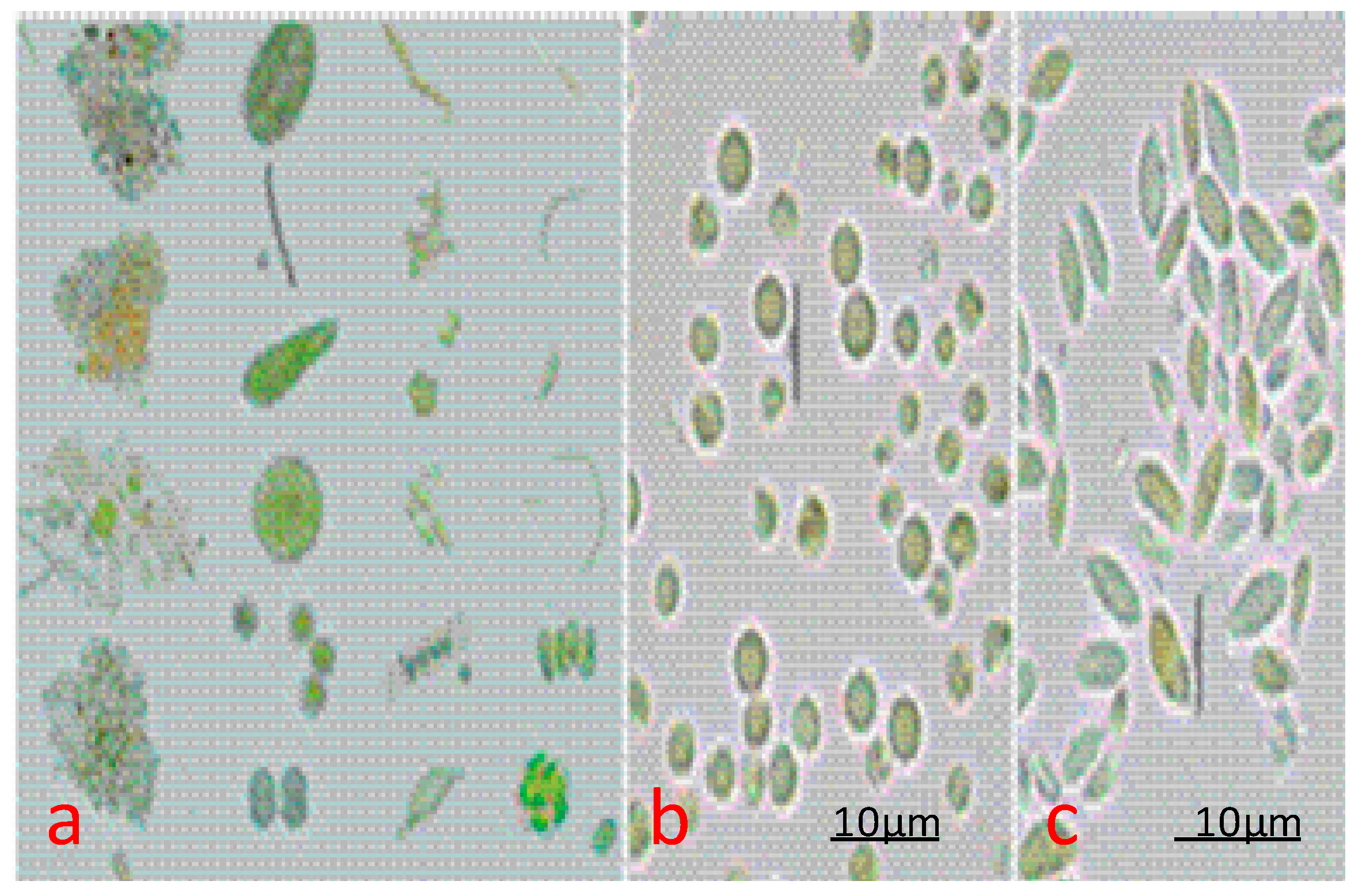
| Strain | Protein (%) | Carbohydrates (%) | Lipid (%) |
|---|---|---|---|
| Anabaena cylindrica | 43–56 | 25–30 | 4–7 |
| Botryococcus braunii | 40 | 2 | 33 |
| Chlamydomonas rheinhardii | 48 | 17 | 21 |
| Chlorella pyrenoidosa | 57 | 26 | 2 |
| Chlorella vulgaris | 41–58 | 12–17 | 10–22 |
| Dunaliella bioculata | 49 | 4 | 8 |
| Dunaliella salina | 57 | 32 | 6 |
| Dunaliella tertiolecta | 29 | 14 | 11 |
| Euglena gracilis | 39–61 | 14–18 | 14–20 |
| Porphyridium cruentum | 28–39 | 40–57 | 9–14 |
| Prymnesium parvum | 28–45 | 25–33 | 22–39 |
| Scenedesmus dimorphus | 8–18 | 21–52 | 16–40 |
| Scenedesmus obliquus | 50–56 | 10–17 | 12–14 |
| Scenedesmus quadricauda | 47 | - | 1.9 |
| Spriogyra sp. | 6–20 | 33–64 | 11–21 |
| Spirulina maxima | 60–71 | 13–16 | 6–7 |
| Spirulina platensis | 42–63 | 8–14 | 4–11 |
| Synechoccus sp. | 63 | 15 | 11 |
| Tetraselmis maculata | 52 | 15 | 3 |
| Pseudochoricystis ellipsoidea | 10.2 | 34 | 38 |
| Chlorogloeopsis fritschii | 41.8 | 37.8 | 8.2 |
| Chlorella emersonii | 9.03 | 37.9 | 29.3 |
| Chlorella zofingiensis | 11.2 | 11.5 | 56.7 |
| Chlorella FC2 IITG | 10.4 | 24.5 | 37.3 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ravindran, B.; Gupta, S.K.; Cho, W.-M.; Kim, J.K.; Lee, S.R.; Jeong, K.-H.; Lee, D.J.; Choi, H.-C. Microalgae Potential and Multiple Roles—Current Progress and Future Prospects—An Overview. Sustainability 2016, 8, 1215. https://doi.org/10.3390/su8121215
Ravindran B, Gupta SK, Cho W-M, Kim JK, Lee SR, Jeong K-H, Lee DJ, Choi H-C. Microalgae Potential and Multiple Roles—Current Progress and Future Prospects—An Overview. Sustainability. 2016; 8(12):1215. https://doi.org/10.3390/su8121215
Chicago/Turabian StyleRavindran, Balasubramani, Sanjay Kumar Gupta, Won-Mo Cho, Jung Kon Kim, Sang Ryong Lee, Kwang-Hwa Jeong, Dong Jun Lee, and Hee-Chul Choi. 2016. "Microalgae Potential and Multiple Roles—Current Progress and Future Prospects—An Overview" Sustainability 8, no. 12: 1215. https://doi.org/10.3390/su8121215
APA StyleRavindran, B., Gupta, S. K., Cho, W.-M., Kim, J. K., Lee, S. R., Jeong, K.-H., Lee, D. J., & Choi, H.-C. (2016). Microalgae Potential and Multiple Roles—Current Progress and Future Prospects—An Overview. Sustainability, 8(12), 1215. https://doi.org/10.3390/su8121215






