1. Introduction
Electronic scrap is considered a peculiar type of waste, containing many different materials such as various metals, fillers, glass fibers, and flame-retardants (FRs) in addition to plastics. Chlorinated and brominated organic compounds or polymers are mixed with the plastics used in electric and electronic equipment (EEE) to prevent fire damage [
1]. On average, 30% of the plastics in EEE contain FRs and halogenated compounds are the most used among them because of their efficiency and suitability with many types of plastics [
2,
3]. Bromine is generally preferred over chlorine because it requires lower quantities of FRs and minimizes the impact of the additive on the polymer’s performance, even if chlorine is present as polyvinyl chloride (PVC) in the electrical wires. Directive 2011/65/EC (RoHS 2 Directive) restricted the use of polybrominated biphenyls (PBB) and polybrominated diphenyl ethers (PBDE) in concentrations lower than 0.1 w/w% in homogenous materials in newly marketed EEE. However, old EEE manufactured before 1 July 2006 could be still in use, as they are long life products and then higher concentrations of brominated compounds can be found to date in WEEE plastics Furthermore, Wäger et al. [
4] have shown in their study on hazardous substances regulated under RoHS that, in the sampling campaigns, no fraction of mixed plastic from WEEE is completely free and that other brominated flame retardants (BFRs), which are not regulated by RoHS, were present as well.
In 2014, a world production of 41.8 Mt of WEEE was estimated, 11.6 Mt of which in Europe, and it is expected to grow up to 50 Mt in 2018 [
5]. The magnitude of this waste stream poses a serious challenge, which has been thrown down by the European Commission. With the entry into force of the so-called WEEE Directive 2012/19/EU, a recovery target of 70%–80% of material and energy recovery has been set and subordinated to all WEEE materials, including the less valuable plastic fraction, which accounts for 30 w/w% of electronic wastes [
6] in order to reduce their landfill disposal. The main environmental and health issue problem arises from the high halogen content in these plastics with respect to their end of life management. In fact, halogenated plastics under heating treatment can release irritant and corrosive gases, such as HCl or HBr. Moreover, in the presence of oxygen, metal oxides in the ashes, and temperatures between 200 and 500 °C, HCl and HBr can be possible precursors of extremely toxic substances such as polyhalogenated dioxins and furans and various other organic halogen compounds [
7]. Even if they undergo pyrolysis, a thermal treatment at low temperatures under inert atmosphere, dehalogenation of pyrolysis oil and gas is essential to make them commercially acceptable. The potential toxicological risk of PVC undergoing a mechanical recycling is due to the presence of additives such as heavy metal stabilizers, phthalates, and some chlorinated paraffins that are used as plasticizers [
8], while the recyclability of polymers treated with BFRs is potentially hampered due to the possible formation of polybrominated dioxins and furans [
9]. For these reasons, the concentrations of bromine and chlorine are quality parameters of WEEE plastics subjected to material recycling or recovery processes. Therefore, they need to be quickly and economically analyzed, especially in thermal treatment, regardless of the knowledge of their specific chemical structures, possibly achieving a satisfying level of precision and accuracy. For example, to consider a waste or a mixture of wastes as a secondary solid fuel, the chlorine content must be lower than 3%, calculated on a dry basis (CEN/TC 15359:2006). Anyway, thermal plants usually cannot accept a feed with more than 2–5 w/w% of chlorine and bromine, being one of the most important criterion for their acceptance [
10].
Sample pre-treatment followed by gas chromatography-mass spectrometry (GC-MS) is perhaps the most widely used instrumental analysis technique used to distinguish restricted from non-restricted BFRs and for their quantification [
9]. Pre-treatment allows a complete dissolution of the polymer in an appropriate solvent, possibly at room temperature, in order to avoid the thermal degradation of BFR [
11,
12]. Alternatively, a solid-liquid extraction is often adopted along with a series of extraction procedures, such as those performed by shaking, ultrasonication, soxhlet, microwave extraction, pressurized solvent, and supercritical CO
2 [
13,
14,
15,
16,
17]. In addition to GC-MS, other analytical techniques used after the sample pretreatment are high performance liquid chromatography coupled with MS or UV detector, infrared analysis, and GC with an electron capture detector [
13,
18,
19,
20]. When the BFRs are copolymerized into the backbone of the polymer, their separation and quantification by the above-cited methods can be very difficult to achieve. Then, pyrolysis–gas chromatography is one of the techniques used in these circumstances [
21,
22]. Unfortunately, as the sample amount to be pyrolyzed is very small (less than 1 mg), this technique does not seem to be suitable for heterogeneous waste mixture samples [
23]. Furthermore, the large number of pyrolysis products detected from the analysis hampers the interpretation and identification of all the compounds. However, all these methods are time consuming (complex and extensive sample preparation procedures), destructive, have a relatively high analysis cost associated to the purchase and maintenance of the equipment, and need significant technical expertise. Furthermore, a quantitative recovery of analytes is an essential requirement in all samples’ pre-treatment.
Other destructive but simpler analysis that could be similarly employed for plastic materials is the elemental analysis of bromine and chlorine by ion chromatography (IC) or titration. These methods include high-pressure oxygen bomb combustion of the sample followed by halogens recovery in a basic solution [
24,
25].
Analysis based upon non–destructive methods provides a useful screening to check the eventual presence of bromine or chlorine. They include energy dispersive X-ray fluorescence spectroscopy (ED-XRF), infrared equipped with an attenuated total reflectance accessory, and Raman spectroscopy [
9,
13,
26,
27]. ED-XRF displays analytical results for bromine and chlorine under elemental form, while infrared and Raman spectroscopy may identify the specific halogenated compounds. Although Kikuchi et al. suggested in their study about BFRs in acrylonitrile butadiene styrene (ABS) [
27] that Raman spectroscopy is less prone to matrix interference, these spectroscopic techniques are affected strongly by this problem that could mask positive findings, and are not appropriate for low concentrations [
9]. Morf et al. carried out a quantitative XRF analysis [
28] preparing the grained samples mixed with a matrix modifier (silicates and/or lime), a matrix similar to that used for calibration (based on a calibration for geological samples with standard reference material). Aldrian et al. tested a handled XRF (HXRF) [
29], equipped with the manufacturer’s software, for a quantitative analysis on WEEE with promising results providing that the device must be checked every working day with reference materials or control standard. Many authors highlighted the importance of routine calibration of the HXRF analyzer [
25], as in some cases factory installed elemental rates are significantly different from calibration standards. In any case, the lack of standards with matrices as similar as possible to those of the samples can lead to less accurate results. Therefore, it might be advisable to confirm the results obtained by using more reliable laboratory analysis methods, and consider XRF as a very useful screening test for determining elemental concentration [
25,
30].
The aim of this work is to compare the performance of four analytical techniques for the quantitative determination of bromine and chlorine on several different real plastic samples extracted from WEEE material recovery facilities (MRF). The instruments used were IC, ion-selective electrodes (ISEs) as simple alternatives to IC, elemental analysis for chlorine and ED-XRF. In order to achieve quantitative analysis with ED-XRF, standard plastic materials with different concentrations of bromine and chlorine have been preliminarily prepared in our laboratory: ABS-Br and polyethylene-Cl (PE-Cl). The home-made plastic standard materials are used for calibration of the ED-XRF and to test the other techniques before the samples analysis. According to our knowledge, no paper is available in literature on XRF calibration that resorts to home-made plastic standards doped by Br or Cl, in order to overcome any matrix effect in waste plastic samples. The need to prepare our standard materials derives from the fact that XRF calibration standards are currently limited to metals doped in homogenous matrices, such as polyethylene (PE) and that no suitable standards exist for use on EEE plastics or complex electric or electronic components; no standard plastic materials with different concentrations of Br and Cl were available in commerce. The European Reference Materials prepared the reference materials, consisting of commercial PE and polypropylene (PP) in granular form, which have been reinforced with technical mixtures of polybrominated diphenylethers and polybrominated biphenyls [
31]. These references are known as ERM-EC590 (PE) and ERM-EC591 (PP) and have a total bromine content of 2.13 ± 0.09 g·kg
−1 and 2.08 ± 0.07 g·kg
−1, respectively. ERM-EC590 and ERM-EC591 are available for sale at the certified reference materials catalog of European Commission [
32] and, even if the matrices are not identical, could be used as control standard for the applied methods.
2. Materials and Methods
2.1. Reference Materials and Investigated Samples
PVC and DecaBDE reference materials were purchased from Sigma Aldrich (both with purities declared of 98%), while virgin polymers ABS and PE derived by Sirmax SpA (Cittadella, Italy).
The four investigated waste plastic samples were collected from various WEEE recovery facilities set in various Italian regions, in particular:
ABS IT: this sample came from a MRF located in the Marche Region, and was composed of grey plastics originating from housing of information technology (IT) equipment. It is made up principally by ABS and was in form of scraps with size of 5–10 mm.
WEEE Residue: residue coming from a MRF located in the Campania Region and originating from the mechanical treatment of printed circuited boards (PCBs) and small WEEE for metals recovery. This residue is made up principally by thermoplastic such as ABS, HIPS, PS, but also includes themosettings and, to a lesser extent, metals, glass, and other non-plastic materials.
Fridge foam: polyurethane (PU) foam for insulation of refrigerators in pressed cylinders (diameter = 100 mm, height = 200 mm), coming from a MRF located in the Lazio Region.
Slot mix: this sample coming from an MRF located in the Lazio Region is made up of edge connector sockets used on PCBs for expansion slots of the peripheral cards. The sockets were intact but separated from metal pins. The most used polymers are polyamides, polybutylene, terephthalate, and polyphenylsulfone (PPS).
Prior to any chemical analysis, real samples and virgin polymers were reduced to particle sizes between 0.50 and 0.25 mm with a Retsch SM 2000 cutting mill. Size reduction is needed to ensure homogeneity of complex real wastes and uniformity among different dimensions of the original samples. To this end, the samples in powder were pelletized in tablets with thickness of 4.0 ± 0.2 mm and about 0.5 g weight for all the following analysis, except proximate and elemental analysis. Each measurement was performed with three or five replicates for each sample.
2.2. Preparation of the Standard Plastic Materials
Calibration of ED-XRF measurements and the check of performances of analytical methods require the use of reference materials with known concentration of the analyte and containing accompanying elements (matrix) with similar physico-chemical characteristics of the sample to be investigated. In particular, the matrix effects, such as absorption and secondary fluorescence are the major sources of errors in the quantitative XRF analysis [
33,
34]. For these reasons, “standard” plastic materials were prepared by mixing in appropriate proportions the virgin ABS or PE powders vigorously, simulating the plastic matrices of the real samples, with PVC or DecaBDE as source of Cl and Br, respectively. The mechanical mixtures were quantitatively picked up from the mixer. The final concentrations of Cl for PE-Cl and Br of ABS-Br were 0.0, 0.5, 2.0, and 5.0 w/w%. The virgin polymers ABS and PE were used as 0.0 w/w% standards having verified by screening with ED-XRF that halogens were not present in the pure products. These concentrations were chosen on the basis of results reported in literature (see
Table 1). The mechanically mixed ABS-Br and PE-Cl samples were also pelletized in tablets, similarly to the samples for the following analysis or calibration.
2.3. Sample Dissolution
The analysis of Cl and Br through IC and ISEs must be performed on a liquid sample. Then, standards and samples were dissolved according to the procedure reported in the CEN/TS 15408:2006. The plastic tablets were combusted with O2 (3 MPa) in the bomb of a C5000 Berthelot-Mahler Calorimeter IKA, previously filled with 10 mL of a 0.2 M KOH solution, in order to absorb the acid gaseous products. After combustion and 10 min of equilibration time, the solution was recovered quantitatively with water in a 100 mL flask. In this study, an alternative method of gas absorption was tested to verify if a better recovery of Cl and Br was obtainable: the combustion bomb was filled with 5 mL of a NaHCO3/Na2CO3 buffer solution (the eluent used in IC) and the flue gas was allowed to gurgle with a low flow rate (30 mL/min) through a trap filled with 50 mL of the same buffer solution. In any case, the resulting solutions were filtered with a 0.20 µm cellulose filter, and finally analyzed.
2.4. Proximate and Elemental Analysis
Before performing the direct measurements of Cl and Br content, both the waste plastic samples and the virgin polymers were thermochemically characterized. Experiments for the determination of humidity, volatile matter, fixed carbon, and ash content were carried out using a TGA 2950 macro thermobalance (TA Instruments) with about 1 g of sample according to the US technical specification ASTM D7582-12 (2012) adopted for coal and coke. The temperature program adopted in this study was described in a previous paper [
35]. The organic matter was the sum of the volatile fraction plus the fixed carbon, while the ash content represents the inorganic matter. Furthermore, proximate analysis provides an estimate of the degradation temperatures of the tested materials.
A Macro VARIO Cube Elemental Analyzer was used to determine simultaneously the weight percentages of carbon, nitrogen, hydrogen, and sulphur on 20–30 mg samples. By introducing specific modules and detectors, it was possible to measure the oxygen and Cl content. In particular, the Cl present in the solid sample was converted into HCl by combustion in a furnace at 1150 °C, and analyzed as chloride in an electrochemical cell (amperometric detection). Two cells were available, one for low amount of Cl (calibration range 0.5 ÷ 20 µg) and the other for high amount (calibration range 15 ÷ 1200 µg). Every working day, the appropriate electrochemical cell in use was calibrated with NH4Cl (Carlo Erba, pure reagent). This module cannot be used for the Br analysis.
2.5. Calorimetric Determination of Low Heating Value
During the sample dissolution procedure, the heat evolved during the burning process of the organic matter was determined using the adiabatic measurement procedure. The low heating value (LHV) represents an estimation of the maximum energetic potentially extractable from a material and can be derived from the high heating value (HHV) directly measured by the calorimeter with the expression: LHV = HHV − 2.5 (9 (HyC) − (HuC)), where HyC and HuC are the hydrogen and the humidity fraction in the sample [
44,
45]. The calibration consisted in assessing the heat capacity of the decomposition vessel by burning tablets of certified benzoic acid, accurately weighed using an analytical balance: masses measured within to 0.0001 g.
2.6. Ion Chromatography
After dissolution and filtration, the sample solutions were analyzed through a 883 Basic IC plus Metrohm Ion Cromatographer, equipped with a Metrosep A Supp 4-250/4.0 anionic column for determining bromide and chloride content. The eluent was a buffer solution NaHCO3/Na2CO3 (Sigma-Aldrich, St. Louis, MO, USA, purity 99.8%) 1.7/1.8 mmol/L with 1 mL/min flow rate. Calibration was performed in the range 1–50 ppm with chloride and bromide standards for IC (Sigma-Aldrich) and, if necessary, home-made standard and samples were diluted with ultrapure water produced by Arium Basic system (Sartorius AG). The Br and Cl concentrations in the blank were subtracted to sample values.
2.7. Ion-Selective Electrodes
The same solutions examined by IC were analyzed by ISEs to determine bromide and chloride content (Electrodes, Van London pHoenix Co., Houston, TX, USA, and Controller, B&C Electronics Srl, Carnate, Italy) by adding them 2 mL of ionic strength 5 M NaNO3 buffer solution. The ISEs were calibrated at least daily with NaCl or NaBr solutions (Panreac, pure reagent) at concentrations of 10, 100, and 1000 ppm. If necessary, sample or standard plastic solutions were diluted with distilled water. The Br and Cl concentrations in the blank were subtracted to sample values.
2.8. Energy Dispersive X-ray Fluorescence
Plastic tablets were investigated through the non-destructive ED-XRF technique, using a EDX-720 Shimadzu Spectrometer equipped with a Rh-lined X-ray tube under a voltage of 15–50 kV in air atmosphere. This technique offers a qualitative multi-element detection in the range from
6C/
11Na to
92U. In order to perform a quantitative analysis, halogenated plastic standards were prepared as described in
Section 2.2. Beyond the weight as for the other techniques, an accurate control of the size distribution and thickness of the tablets is important for an ED-XRF quantitative analysis, since samples with the same element concentration but with different geometry or dimension of the grains can lead to different results.
3. Results
3.1. Thermochemical Characterizations
Table 2 displays the proximate and ultimate analysis, the LHV and the check on the reached conservation of mass referred both to the virgin polymers and the WEEE plastic samples. Humidity of all samples, up to a maximum of 0.4 w/w is a sign of waterproof material quality ascribable to these plastics. The loss of volatile matter corresponding to polymer degradation occurs between 350 and 500 °C and represents the highest amount among the remaining fractions (fixed carbon and ash). The percentage of fixed carbon is quite variable from one sample to the other and is probably connected to the production of conjugated aromatic rings, which present a remarkable thermal stability [
46]. The ash content accounts for the sample inorganic fraction and does not undergo weight loss at these temperatures. As expected, the slot mix showed the highest Ash content (>30 w/w%), being a part of PCBs and in agreement with the result of a previous study on different connectors [
35]. PE and ABS virgin polymers showed an elemental composition very similar to the theoretical one, and they are constituted almost entirely by volatile matter.
As far as elemental analysis of the WEEE plastic samples is concerned, the most abundant element present is carbon, with the maximum content in the ABS IT, which with the fridge foam, is the less contaminated and the most homogenous sample. Actually, its elemental composition is significantly different from that of virgin ABS, especially for the relatively high content of oxygen that should not be present in styrene-based polymers. Probably, the presence of oxygen is due to polycarbonates (PC), as ABS/PC blend used in many different categories of EEE and to a consequent not correct designation during the sorting in the treatment plant. Sulfur content was almost always below the calibration range of the instrument, which is positively considered both in the case of incineration (absence of formation of SO
2 and SO
3) and in the case of pyrolysis (obtaining a low sulfur content oil). The exception is constituted by the slot mix sample, in which the presence of connector in PPS was detected by FTIR-ATR [
47]. Concluding the analysis of the elements content, nitrogen is present as acrylonitrile (ABS IT and WEEE Residue), polyamides (slot mix), and PU (fridge foam). As can be seen from
Table 2, the sum of the percentages of organic (C, N, H, S, O) and inorganic (ash) fractions obtained through ultimate and proximate analysis provides a value slightly lower or equal to 100 w/w% for whichever samples. Thus, unless hypothesizing the presence of other fuel elements, the presence of a not negligible quantity of Cl and/or Br at least in the ABS IT, WEEE Residue, and slot mix samples is extremely probable. Therefore, a further investigation for the measurement of Cl and Br is necessary.
The LHVs are comparable to those of a coal and higher than residue derived fuel for the presence of petroleum derivatives. The high ash and oxygen content strongly contributes to lower the LHV, which does not exceed 16 MJ·kg−1 in the case of slot mix.
3.2. Calibration of ED-XRF
The standard ABS-Br and PE-Cl tablets prepared as described in
Section 2.2 were used in order to calibrate the ED-XRF instrument. To achieve reliable results working directly on solid samples this instrument needs standards as similar as possible to the sample.
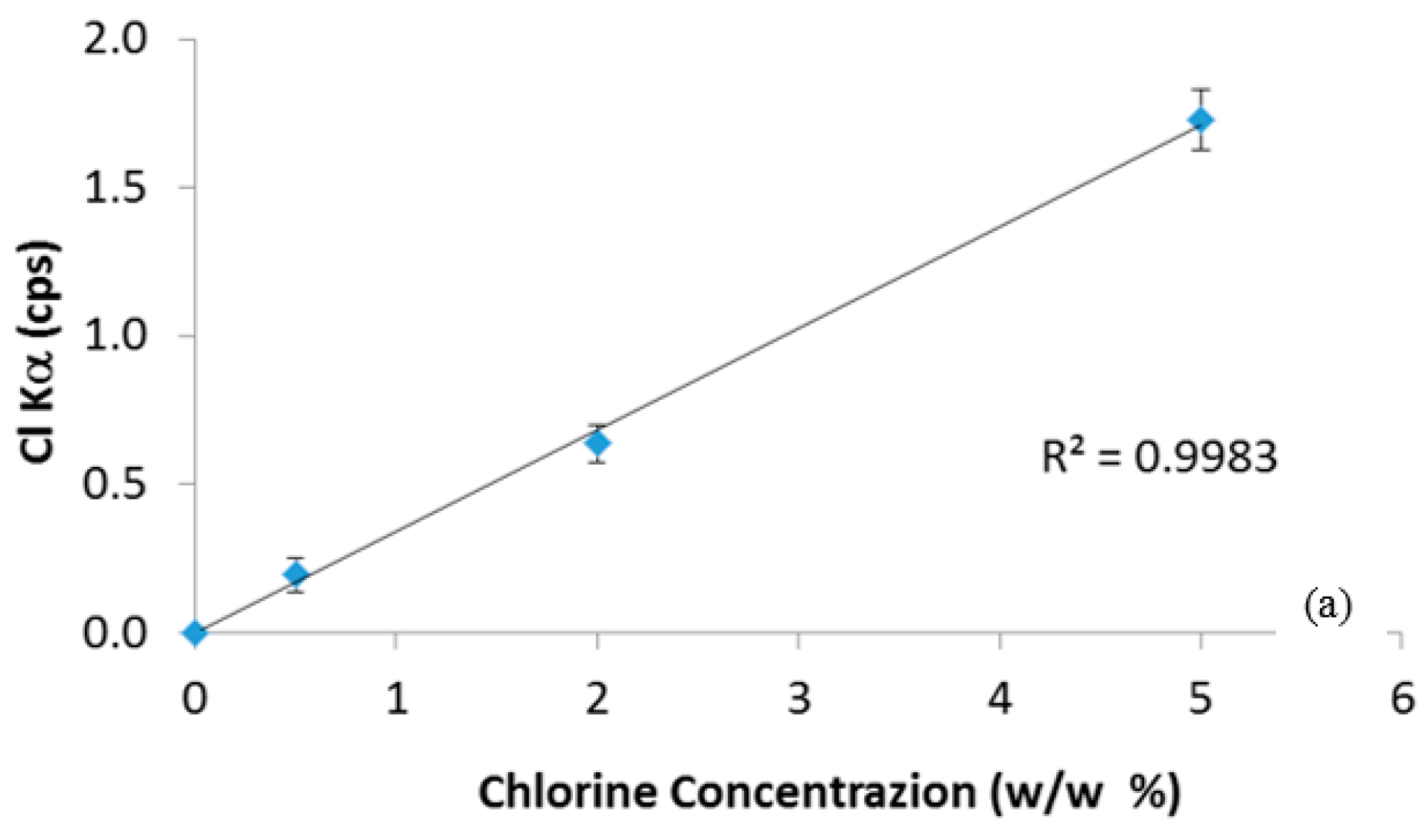
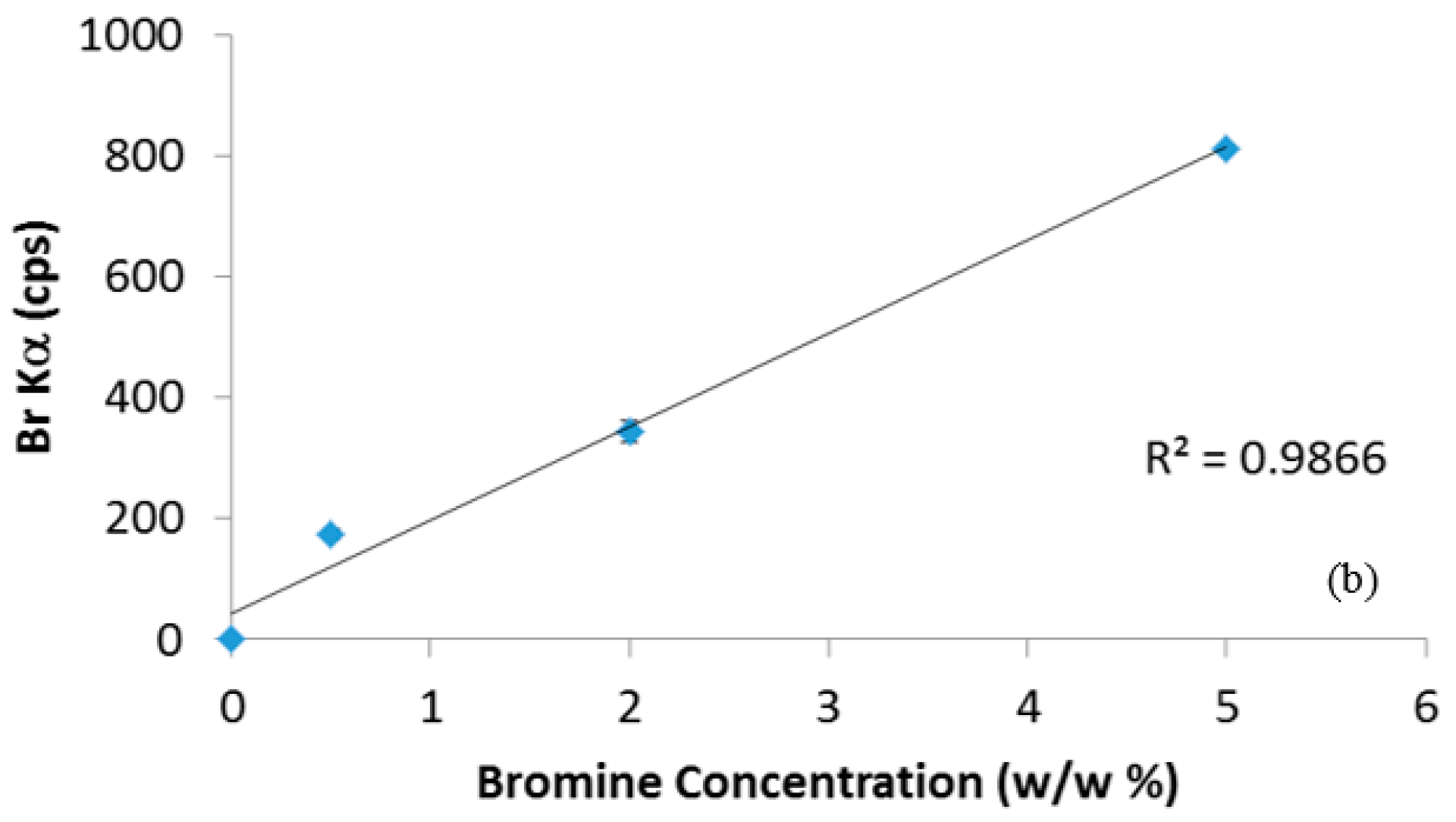
Figure 1 shows the calibration curves for chlorine and bromine determination. The goodness of linear fit (R
2 very close to unity) and the very low standard deviations among the replies show that the standard tablets were prepared properly, without significant loss of material during the tablets preparation (mixing and compression), and that the sources of Cl and Br are homogenously distributed in the plastic matrices. In order to verify the reliability of calibration curves, new standard ABS-Br and PE-Cl tablets with a theoretical concentration of 1 w/w% were prepared and quantitatively analyzed using the regression straight line equations of
Figure 1. The results (0.95 ± 0.06 w/w% for PE-Cl and 1.1 ± 0.1 w/w% for ABS-Br) confirmed that the ED-XRF measurement is adequately accurate and reproducible, since the absence of any pretreatment, reproducibility depends substantially on the homogeneity of the prepared tablets, which is therefore satisfactory. It must be noted that neither the presence of Cl nor that of Br has been revealed in the ABS-Br and PE-Cl samples, respectively, assuring the absence of mutual pollution from chlorine and bromine in these standards. Then, the instrument was ready to quantify the Cl and Br content in the real plastic samples.
3.3. Analysis of Standard Materials by Selected Methods
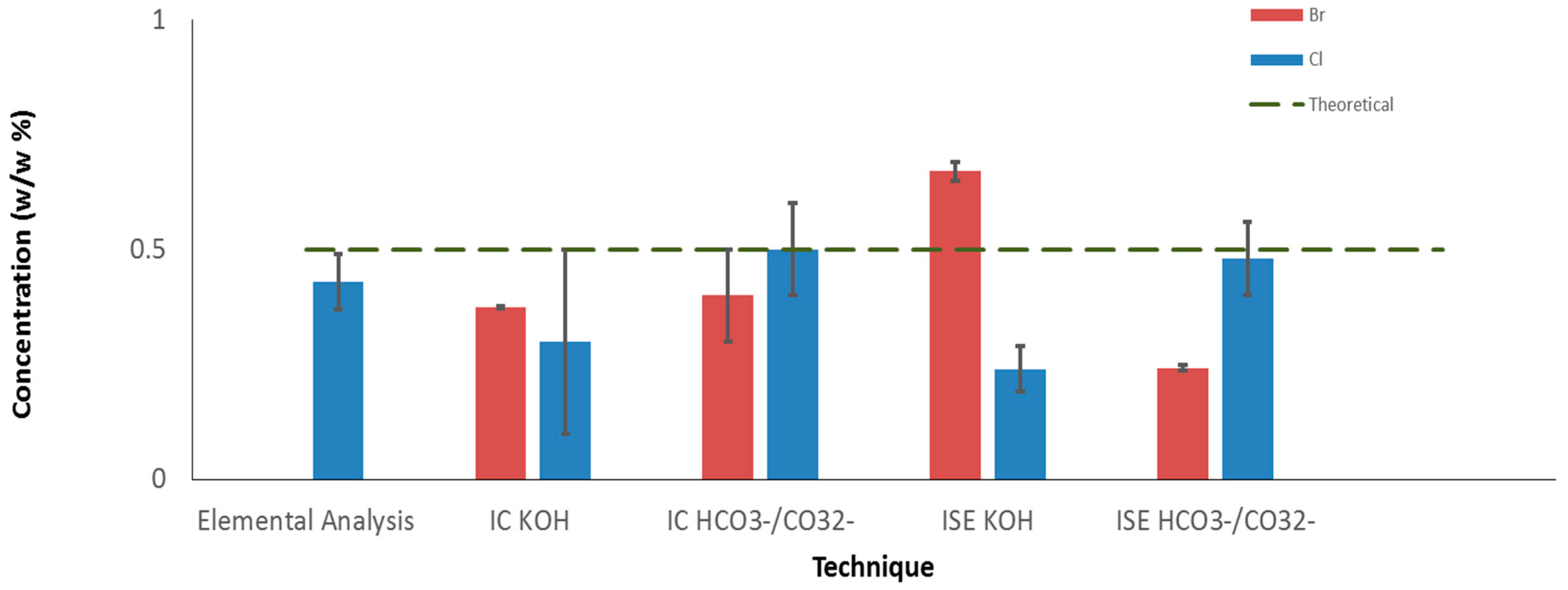
All the instruments and procedure employed in this study were tested, as it was described for ED-XRF measurements, using the ABS-Br and PE-Cl standards (at concentration of 0.5 w/w%) to evaluate their performances. The results obtained from IC, ISEs, and elemental analysis are reported in
Figure 2. For IC both the results with recovery made using KOH as well as NaHCO
3/Na
2CO
3 buffer solution were presented, while the reference material PVC was analyzed only by elemental analysis. The high Cl content in PVC (theoretical concentration, 56.8 w/w%) prevented the analysis with the other techniques since it caused the bomb corrosion during the dissolution procedure and is out of the calibration range of ED-XRF.
Also considering the result on PVC (55 ± 6 w/w%), elemental analysis produced value similar to the theoretical one, but with a lower precision for materials with high Cl content. For IC it was possible to note that the sample dissolution procedure with the low gurgle in the buffer solution seems to achieve a higher recovery in comparison with what was obtained with the KOH method. Furthermore, the Br concentration was lower than the theoretical one since Br is present in several oxidation states after combustion, e.g., BrO4− (CEN/TS 15408:2006). This causes an underestimation, because IC was set only for bromide analysis. The ISEs provided an acceptable result only in the case of chlorine determination in a NaHCO3/Na2CO3 buffer solution but, from a general point of view, their responses did not appear reliable for this type of applications. In addition, ISEs were affected by drift over time and required a new calibration with standard solutions of NaBr and NaCl, approximately every 3 h.
3.4. Analysis of Real WEEE Samples by Selected Methods
Table 3 reports the results of Br and Cl concentrations in the plastic waste samples obtained with the four techniques. Taking into account the results from ED-XRF, the samples showed different amounts of Br and Cl. As expected, the highest amount of Br was present in the slot mix, because these plastics are connected directly to the PCBs, and therefore exposed to combustion risk. Also ABS IT showed a high content of Br, while the ABS used for housing has usually a low content of BFRs [
28,
35,
48]. Then, a big fraction of this ABS came probably from the monitors where high concentrations of BFRs may be present [
39]. Taurino et al. found in gray plastic 4.4 w/w% of Br measured by semi-quantitative XRF analysis [
36], extracted from personal computer monitors constituted by an ABS/PC blend. This evidence seems to be in good agreement with the oxygen content determined by elemental analysis. Detection of chlorine may be symptomatic of the presence of PVC wires and chlorinated FR within the material (WEEE Residue and ABS IT) and the use of chlorofluorocarbons for PU foam expansion in the fridges.
Cl percentage obtained by elemental analysis were always similar or higher than those obtained with ED-XRF, even in the slot mix, where the presence of chlorine may be excluded. This is due to the strong interference of bromide on the electrochemical cell, as it is clearly indicated by the producer. Furthermore, it was difficult to quantify the magnitude of this interference also because the other brominated compounds (with different oxidation states formed during combustion) were not detected. The unique result that should be considered reliable was the Cl content in the fridge foam (the only one where Br is absent), even if the precision is limited. According to our experience, precision of this analysis decreases with the life-time of the electrochemical cell.
Chloride and bromide percentages detected in the samples by IC with KOH and by IC with NaHCO3/Na2CO3 were quite similar and consistent with the ED-XRF analysis, although slightly lower: it was possible that during the dissolution pretreatment an analyte loss occurred (not so for the standard analysis). The difference between the results obtained with both techniques is more pronounced for Br, where there is non-quantitative transformation in Br− during the occurrence of combustion. Furthermore, compared to the results of the analysis on standards, the benefits coming from the use of this procedure with the slow bubbling than the faster described by CEN/TS 15408 appears less evident. The reason for this discrepancy between the results of the standards and those of real samples could be due to a not-negligible matrix complexity, but in this respect more insights are needed. Furthermore, it is worth noting that Br and Cl contained in the fridge foam and in the slot mix, respectively, were under the detection limit.
The concentration values determined by ISEs strongly deviate from those measured by the other techniques and appear unreliable. ISEs failed to detect low concentrations of both halides in the fridge foam, and the Cl content was always significantly overestimated due to the strong interference of Br. On the contrary, the amount of Br is underestimated because of the leakage, and the presence of compounds with different oxidation states. Another source of interference is the presence of metals—such as Cd, Pb, Sn, and Bi—which may form complexes in solution with halogens and have been revealed during the screening analysis with the ED-XRF.
4. Discussion
The main characteristics of the techniques used in this work are reported in
Table 4. With regard to their accuracy and repeatability, the best technique is ED-XRF that along with elemental analysis provided the highest concentration values without the need to dissolve the sample that could lead to a possible loss of analytes. The ED-XRF is commonly used for a qualitative screening, being a fast and non-destructive technique, which requires no (or slightest at least) sample pretreatment. Since, to the best of our knowledge, neither standard plastic materials with different halogen concentrations nor halogens in specific matrixes simulating EEE are commercially available, the main difficulty to use this technique for quantitative analysis is the preparation of standards as similar as possible to the sample matrix. With respect to the other techniques, further attention must be paid to the preparation of the tablets, taking into account that properties such as thickness, grain size, and weight must be the same for the standard and samples, as they influence the instrumental response. Once the calibration is performed, the method can be tested through the analysis of reference materials, such as ERM-EC590 or ERM-EC591, or samples previously analyzed with another technique like GC-MS, taking care to obtain a complete recovery of the analytes. The analysis is fast and both the drawbacks of the initial cost for the purchase of the instrument and the operator training are balanced by low expenses for consumables and maintenance.
IC has generally showed lower values than ED-XRF, and its use for the analysis of Br should be considered with care since, even in case of complete recovery by the pretreatment solution, this halogen content is significantly underestimated. In the near future, it might be interesting to try to estimate the fraction of Br, which under combustion releases at different stages of oxidation, by analyzing a series of standard plastic samples with different concentrations of Br (provided that the complete absorption of the analyte in the basic solution is guaranteed). In this way, a weighing factor directly connected to this fraction could be determined and used to account for a total amount of bromine in the subsequent analysis on samples. IC cannot be used with too concentrated samples because high levels of gaseous HCl and HBr can cause corrosion of the bomb under combustion pretreatment. Finally, precision and accuracy are quite good and IC can be used to have an estimation of Cl and Br content, especially in view of a mandatory threshold value to be complied with.
If IC seems to achieve reliable results for at least Cl determination, by contrast, ISEs have led to useless results in this context, especially when real samples were analyzed. The low cost of the instrumentation and the ease of use cannot offset the huge difficulty to achieve reliable results: probably a complete control of parameters like pH, temperature, ionic strength, and interfering substances may lead to use of adequate standard solutions for calibration of ISEs to be adopted for this application. Finally, ISEs suffer from drift over time and short-term (not more than six months).
Apparently, the simplest and fastest technique would seem to be the elemental analysis that only requires grinding of the sample for homogenization and weighing in a tin boat. Since the instrument is usually equipped with an auto-sampler, this technique would allow analyzing up to 40 samples consecutively. Actually, this instrument requires frequent recalibration to regenerate the measuring cell for 2–3 days by humid atmosphere every 1–2 working days (you must therefore be equipped with at least two identical cells for each concentration range) and a whole series of expensive consumables. Before proceeding with the analysis, it is important to verify the absence of Br in the sample because, as we have seen, this element interferes with the measurement. This method has shown a poor repeatability especially for real samples. To conclude, elemental analysis is an interesting technique with respect to the future, but still not ready for routine use.
The analysis times are fast enough for all techniques, being the slowest IC with about 20 min of waiting for each reply. The true discriminant is, however, the preparation of the sample analysis that for the IC and ISEs can be very long. Moreover, the calorimeter cost needs for the dissolution of solid samples must be added to the “Instrument cost” of these two techniques.
The Cl and Br content regarding analysis on real samples of WEEE taken from different studies reported in literature are compared in
Table 1. Despite the heterogeneity of the investigated WEEE, it can be observed that the values reported in this work fall within the range of those reported by various authors. It must be emphasized that in most studies reported in the literature which deal with processes development rather than analytical methods, XRF used for screening or semi-quantitative analysis [
9], and IC [
35,
39,
40,
41] are the most used techniques.
Taking into account the results concerning the Cl and Br content determined by ED-XRF, now it is possible to complete the mass balance started in
Table 2. For all samples, the balance approaches to close and the difference from 100 w/w% is due to other combustible element not revealed by elemental analysis, if the errors associated with the measurements could be excluded. The first element could be phosphorous, the use of which as FR (primarily as phosphate esters) in EEE is increasing as alternative to BFR [
49] and was qualitatively revealed by ED-XRF screening (data not shown). Due to the high percentage of ash, LHV, and halogens, as well as their nature (fridge foam), a proper end of life management for these specific WEEE plastics seems to be the waste-to-energy or other thermal treatments such as gasification and pyrolysis, providing the adequate treatment of gas upon previous dilution with halogen free matrices.
5. Conclusions
Four analytical and relatively simple techniques were used in this study to determine the Cl and Br content in home-made standard samples and real samples from WEEE treatment plants, and their performances were compared. The results showed that, after appropriate calibration, the most suitable and reliable technique is ED-XRF, due to the highest accuracy and repeatability obtained (considered sufficient enough for this kind of application). In addition, it does not require a complex sample pretreatment. Although it will never be possible to realize home-made standards for reproducing perfectly real plastic waste samples, due to all the possible matrix interferences, the procedure developed in this investigation has allowed to obtain an acceptable estimate of Cl and Br concentrations, taking into account the results of other techniques, the reference literature, and mass balance. Calibration of XRF for elements in different electronic product matrices, still not developed, may lead to more accurate results when this technique is applied.
The presence of Cl and Br in the analyzed waste streams was confirmed by application of the various techniques in all samples tested. Based on the total level of Cl and Br between 0.6–4 w/w% and the high ash content, the effectivity and economic viability of recycling these plastic waste streams is questionable and a more appropriate end of life management seems to be pyrolysis or other thermal treatments.