Pollution Assessment with Persistent Organic Pollutants in Upper Soil of a Series of Rural Roma Communities in Transylvania, Romania, Its Sources Apportionment, and the Associated Risk on Human Health
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals and Reagents
2.2. Instrumentation
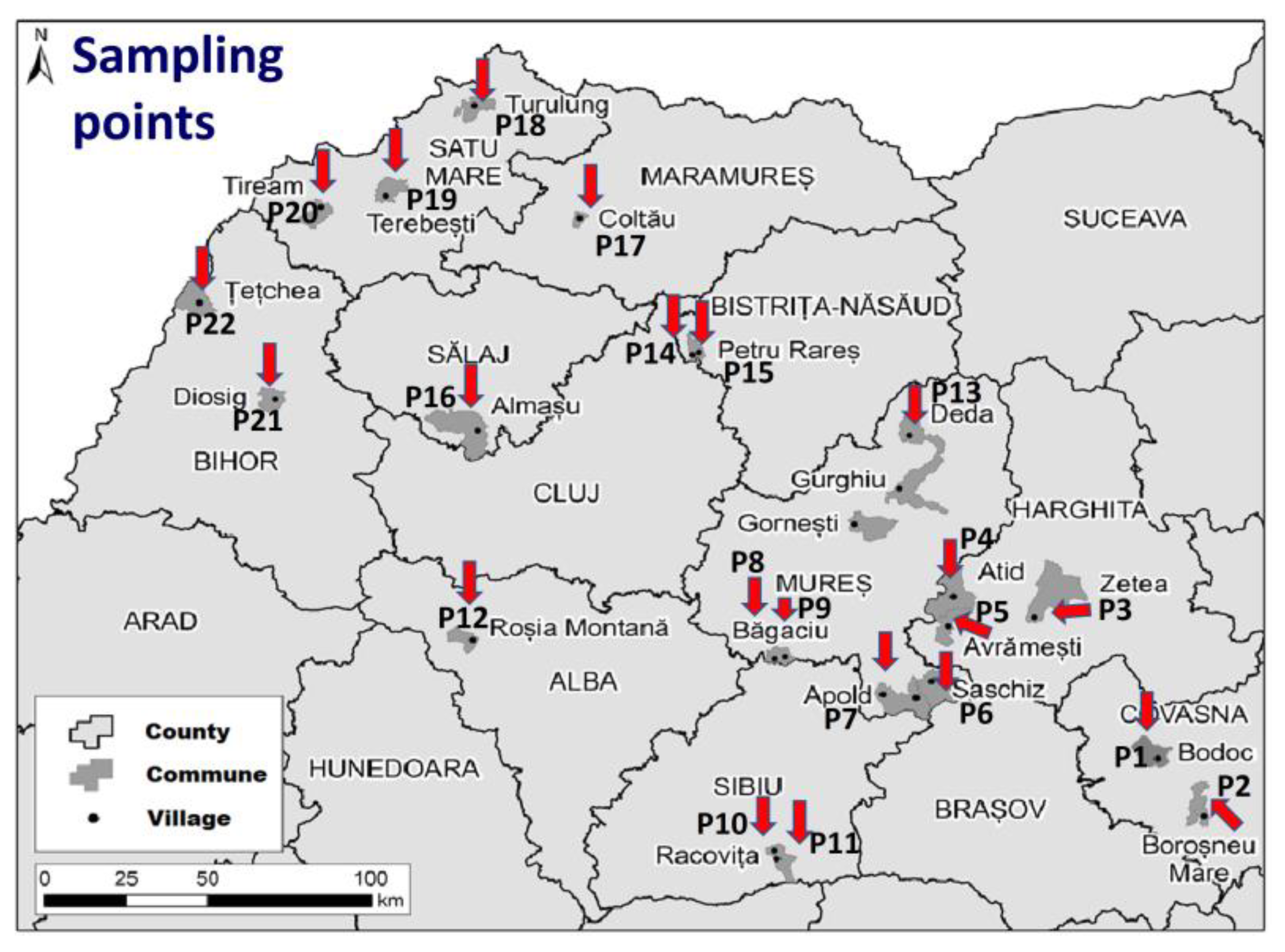
2.3. Sample Collection
2.4. Sample Preparation
2.5. Health Risk Assessment
3. Results and Discussion
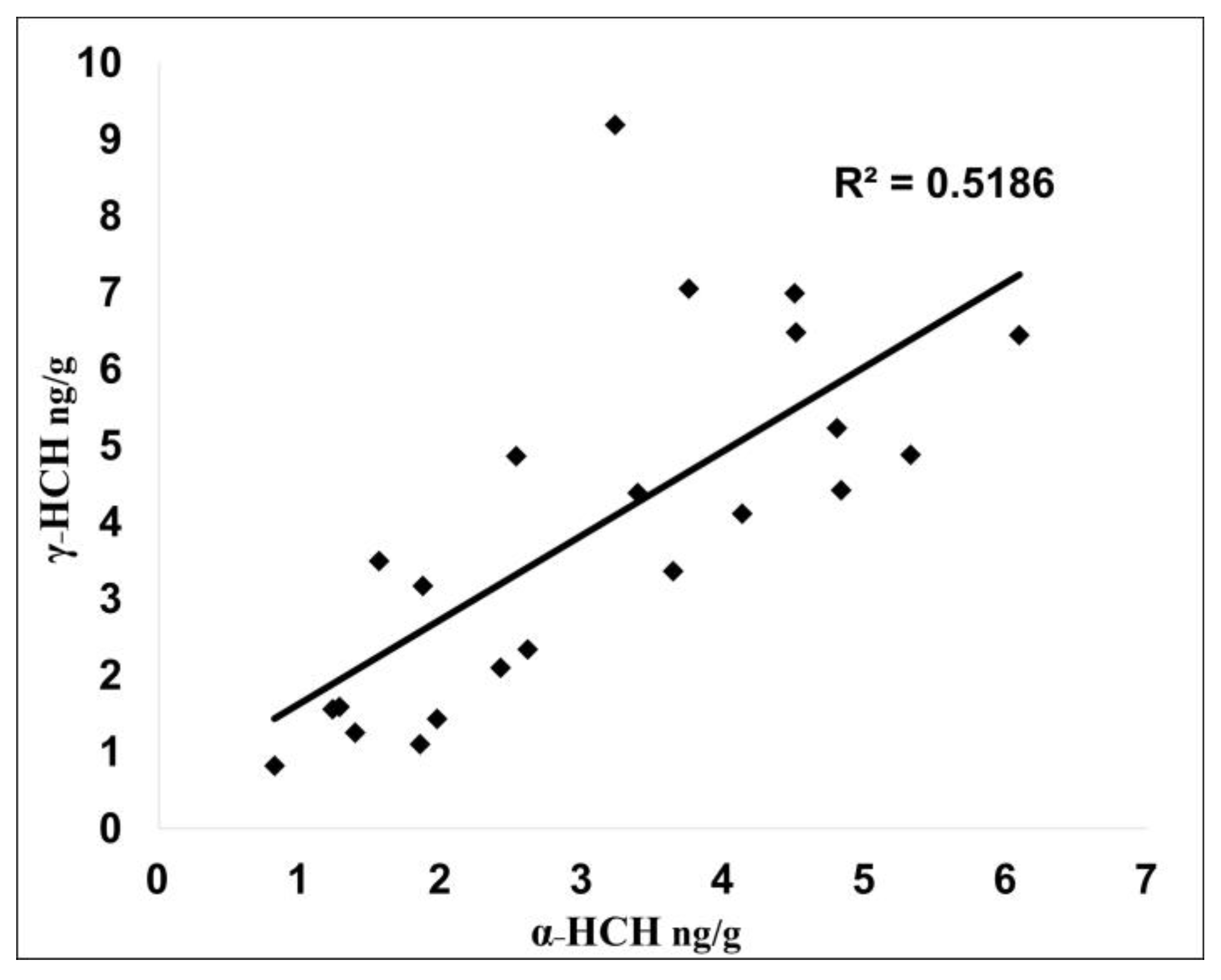
3.1. OCP Distribution and Potential Source Apportionment
3.2. PCBs Concentrations in Soil Samples
3.3. PAHs Distribution and Sources
3.4. Health Risk Assessment
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Grassi, P.; Fattore, E.; Generoso, C.; Fanelli, R.; Arvati, M.; Zuccato, E. Polychlorobiphenyls (PCBs), polychlorinated dibenzo-p-dioxins (PCDDs) and dibenzofurans (PCDFs) in fruit and vegetables from an industrial area in northern Italy. Chemosphere 2010, 79, 292–298. [Google Scholar] [CrossRef] [PubMed]
- Dreyer, A.; Minkos, A. Polychlorinated biphenyls (PCBs) and polychlorinated dibenzo-para-dioxins and dibenzofurans (PCDD/F) in ambient air and depositions in the German background. Environ. Pollut. 2023, 316, 120511. [Google Scholar] [CrossRef] [PubMed]
- Breivik, K.; Sweetman, A.; Pacyna, J.M.; Jones, K.C. Towards a global historical emission inventory for selected PCB congeners—A mass balance approach 3. An update. Sci. Total Environ. 2007, 377, 296–307. [Google Scholar] [CrossRef] [PubMed]
- Polder, A.; Muller, M.B.; Lyche, J.L.; Mdegela, R.H.; Nonga, H.E.; Mabiki, F.P.; Mbise, T.J.; Skaare, J.U.; Sandvik, M.; Skjerve, E.; et al. Levels and patterns of persistent organic pollutants (POPs) in tilapia (Oreochromis sp.) from four different lakes in Tanzania: Geographical differences and implications for human health. Sci. Total Environ. 2014, 488–489, 252–260. [Google Scholar] [CrossRef] [PubMed]
- Deng, Z.; Han, X.; Chen, C.; Wang, H.; Ma, B.; Zhang, D.; Zhang, Z.; Zhang, C. The distribution characteristics of polychlorinated biphenyls (PCBs) in the surface sediments of Ross Sea and Drake Passage, Antarctica: A 192 congeners analysis. Mar. Pollut. Bull. 2020, 154, 111043. [Google Scholar] [CrossRef]
- Pănescu, V.A.; Begy, R.; Roșian, G.; Bruzzoniti, M.C.; Beldean-Galea, M.S. Historical assessment of atmospheric persistent organic pollutants depositions in Muntinu glacial lake, southern Romanian Carpathians, based on radionuclide-dated sediments. Studia UBB Chem. 2022, LXVII, 287–302. [Google Scholar] [CrossRef]
- Albanese, S.; Guarino, A. Assessing contamination sources and environmental hazards for potentially toxic elements and organic compounds in the soils of a heavily anthropized area: The case study of the Acerra plain (Southern Italy). Geosciences 2010, 8, 552–578. [Google Scholar] [CrossRef]
- Wong, M.H.; Leung, A.O.W.; Chan, J.K.Y.; Choi, M.P.K. A review on the usage of POP pesticides in China, with emphasis on DDT loadings in human milk. Chemosphere 2005, 6, 740–752. [Google Scholar] [CrossRef]
- Wolmarans, N.J.; Bervoets, L.; Gerber, R.; Yohannes, Y.B.; Nakayama, S.M.M.; Ikenaka, Y.; Ishizuka, M.; Meire, P.; Smit, N.J.; Wepener, V. Bioaccumulation of DDT and other organochlorine pesticides in amphibians from two conservation areas within malaria risk regions of South Africa. Chemosphere 2021, 274, 129956. [Google Scholar] [CrossRef]
- Drăgan, D.; Cucu-Man, S.; Dirtu, A.C.; Mocanu, R.; Van Vaeck, L.; Covaci, A. Occurrence of organochlorine pesticides and polychlorinated biphenyls in soils and sediments from Eastern Romania. Int. J. Environ. Anal. 2006, 86, 833–842. [Google Scholar] [CrossRef]
- Agency for Toxic Substances and Disease Registry (ATSDR). Public Statement, Polycyclic Aromatic Hydrocarbons (PAHs); ATSDR: Washington, DC, USA, 1995; pp. 1–6. [Google Scholar]
- Giubilato, E.; Radomyski, A.; Critto, A.; Ciffroy, P.; Brochot, C.; Pizzol, L.; Marcomini, A. Modelling ecological and human exposure to POPs in Venice lagoon. Part I—Application of MERLIN-Expo tool for integrated exposure assessment. Sci. Total Environ. 2016, 565, 961–976. [Google Scholar] [CrossRef] [PubMed]
- United Nations Environment Programme (UNEP). Available online: https://chm.pops.int/TheConvention/ThePOPs/ListingofPOPs/tabid/2509/Default.aspx (accessed on 24 October 2023).
- Boehm, P.D. Polycyclic Aromatic Hydrocarbons (PAHs). In Environmental Forensic, 1st ed.; Morrison, R., Murphy, B., Eds.; Elsevier: Amsterdam, The Netherlands, 2005; pp. 313–337. [Google Scholar]
- Covaci, A.; Gheorghe, A.; Voorspoels, S.; Maervoet, J.; Redeker, E.S.; Blust, R.; Schepens, P. Polybrominated diphenyl ethers, polychlorinated biphenyls and organochlorine pesticides in sediment cores from the Western Scheldt river (Belgium): Analytical aspects and depth profiles. Environ. Int. 2005, 31, 367–375. [Google Scholar] [CrossRef] [PubMed]
- Megson, D. Polychlorinated Biphenyls. In Encyclopedia of Analytical Science, 3rd ed.; Worsfold, P., Townshend, A., Poole, C.F., Miro, M., Eds.; Elsevier: Amsterdam, The Netherlands, 2019; pp. 318–328. [Google Scholar]
- Barhoumi, B.; Beldean-Galea, M.S.; Al-Rawabdeh, A.M.; Roba, C.; Martonos, I.M.; Bălc, R.; Kahlaoui, M.; Touil, S.; Tedetti, M.; Driss, M.R.; et al. Occurrence, distribution and ecological risk of trace metals and organic pollutants in surface sediments from a Southeastern European river (Someşu Mic River, Romania). Sci. Total Environ. 2019, 660, 660–676. [Google Scholar] [CrossRef] [PubMed]
- Niu, L.; Mao, S.; Zhou, J.; Zhao, L.; Zhu, Y.; Xu, C.; Sun, X.; Sun, J.; Liu, W. Polychlorinated biphenyls (PCBs) in soils from typical paddy fields of China: Occurrence, influencing factors and human health risks. Environ. Pollut. 2022, 307, 119567. [Google Scholar] [CrossRef] [PubMed]
- Teng, Y.; Li, J.; Wu, J.; Lu, S.; Wang, Y.; Chen, H. Environmental distribution and associated human health risk due to trace elements and organic compounds in soil in Jiangxi province China. Ecotox. Environ. Saf. 2015, 122, 406–416. [Google Scholar] [CrossRef]
- Adesina, O.A.; Ezengwa, I.; Abdulraheem, K.A.; Adewole, A.J.; Oyetunji, O.B. Soil concentrations of Polychlorinated Biphenyl in a typical Nigerian University environment and its risk assessment. Case Stud. Chem. Environ. Eng. 2023, 7, 100343. [Google Scholar] [CrossRef]
- Xu, Y.; Dai, S.; Meng, K.; Wang, Y.; Ren, W.; Zhao, L.; Christie, P.; Teng, Y. Occurrence and risk assessment of potentially toxic elements and typical organic pollutants in contaminated rural soils. Sci. Total Environ. 2018, 630, 618–629. [Google Scholar] [CrossRef]
- Ferencz, L.; Balog, A. Pesticide Survey in Soil, Water and Foodstuffs from Central Romania. Carpathian J. Earth Environ. Sci. 2010, 5, 111–118. [Google Scholar]
- Zhao, W.; Lu, J.; Lai, Y.; Hou, Y.; Zhao, X.; Wei, Q.; Zou, X.; Gou, Z. Occurrences, Possible Sources, and Risk Impacts of Organochlorine Pesticides in Soil of Changchun Central Urban Area, Northeast China. Sustainability 2023, 15, 16801. [Google Scholar] [CrossRef]
- Surenjav, E.; Fiedler, H. POPs in the Mongolian environment. Emerg. Contam. 2023, 9, 100251. [Google Scholar] [CrossRef]
- Zhao, Z.; Jiang, Y.; Li, Q.; Cai, Y.; Yin, H.; Zhang, L.; Zhang, J. Spatial correlation analysis of polycyclic aromatic hydrocarbons (PAHs) and organochlorine pesticides (OCPs) in sediments between Taihu Lake and its tributary rivers. Ecotoxicol. Environ. Saf. 2017, 142, 117–128. [Google Scholar] [CrossRef] [PubMed]
- Eqani, S.A.; Malik, R.N.; Mohammad, A. The levels and distribution of selected organo-chlorine pesticides in sediments from River Chenab, Pakistan. Environ. Geochem. Health. 2011, 33, 33–47. [Google Scholar] [CrossRef] [PubMed]
- Syed, J.H.; Malik, R.N.; Liu, D.; Xu, Y.; Wang, Y.; Li, J.; Zhang, G.; Jones, K.C. Organochlorine pesticides in air and soil and estimated air–soil exchange in Punjab, Pakistan. Sci. Total. Environ. 2013, 444, 491–497. [Google Scholar] [CrossRef] [PubMed]
- Doong, R.A.; Peng, C.K.; Sun, Y.C.; Liao, P.L. Composition and distribution of organochlorine pesticide residues in surface sediments from the Wu-Shi River estuary, Taiwan. Mar. Pollut. Bull. 2002, 45, 246–253. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Yang, H.; Gao, Q.; Pan, H.; Yang, H. Residues of Organochlorine Pesticides in Water and Suspended Particulate Matter from Xiangshan Bay, East China Sea. Bull. Environ. Contam. Toxicol. 2012, 89, 811–815. [Google Scholar] [CrossRef] [PubMed]
- Ali, U.; Bajwa, A.; Chaudhry, M.J.I.; Adeel, M.; Syed, J.H.; Li, J.; Zhang, G.; Jones, K.C.; Malik, R.N. Significance of black carbon in the sediment–water partitioning of organochlorine pesticides (OCPs) in the Indus River, Pakistan. Ecotoxicol. Environ. Saf. 2016, 126, 177–185. [Google Scholar] [CrossRef]
- Covaci, A.; Manirakiza, P.; Schepens, P. Persistent Organochlorine Pollutants in Soils from Belgium, Italy, Greece, and Romania. Bull. Environ. Contam. Toxicol. 2002, 68, 97–103. [Google Scholar] [CrossRef]
- Alamdar, A.; Syed, J.H.; Malik, R.N.; Katsoyiannis, A.; Liu, J.; Li, J.; Zhang, G.; Jones, K.C. Organochlorine pesticides in surface soils from obsolete pesticide dumping ground in Hyderabad City, Pakistan: Contamination levels and their potential for air–soil exchange. Sci. Total Environ. 2014, 470–471, 733–741. [Google Scholar] [CrossRef]
- Sultan, M.; Waheed, S.; Ali, U.; Sweetman, A.J.; Jones, K.C.; Malik, R.N. Insight into occurrence, profile and spatial distribution of organochlorine pesticides in soils of solid waste dumping sites of Pakistan: Influence of soil properties and implications for environmental fate. Ecotoxicol. Environ. Saf. 2019, 170, 195–204. [Google Scholar] [CrossRef]
- Yang, R.Q.; Lv, A.H.; Shi, J.B.; Jaing, G.B. The level and distribution of organochlorine pesticides (OCPs) in sediments from the Haihe River, China. Chemosphere 2005, 61, 347–354. [Google Scholar] [CrossRef]
- Ali, U.; Syed, J.H.; Junwen, L.; Sánchez-García, L.; Malik, R.N.; Chaudhry, M.J.I.; Arshad, M.; Li, J.; Zhang, G.; Jones, K.C. Assessing the relationship and influence of black carbon on distribution status of organochlorines in the coastal sediments from Pakistan. Environ. Pollut. 2014, 190, 82–90. [Google Scholar] [CrossRef]
- Min, C.; Li, C.; Huang, P. Assessment, composition and possible source of organochlorine pesticides in surface soil from Ürümqi, China. Pedosphere 2015, 25, 888–900. [Google Scholar]
- Malik, R.N.; Rauf, S.; Mohammad, A.; Eqani, S.; Ahad, K. Organochlorine residual concentrations in cattle egret from the Punjab Province, Pakistan. Environ. Monit. Assess. 2011, 173, 325–341. [Google Scholar] [CrossRef]
- Jiang, Y.F.; Wang, X.T.; Jia, Y.; Wang, F.; Wu, M.H.; Sheng, G.Y.; Fu, J.M. Occurrence, distribution and possible sources of organochlorine pesticides in agricultural soil of Shanghai, China. J. Hazard. Mater. 2009, 170, 989–997. [Google Scholar] [CrossRef]
- Ene, A.; Bogdevich, O.; Sion, A. Levels and distribution of organochlorine pesticides (OCPs) and polycyclic aromatic hydrocarbons (PAHs) in topsoils from SE Romania. Sci. Total Environ. 2012, 439, 76–86. [Google Scholar] [CrossRef]
- Mao, S.; Liu, S.; Zhou, Y.; An, Q.; Zhou, X.; Mao, Z.; Wu, Y.; Liu, W. The occurrence and sources of polychlorinated biphenyls (PCBs) in agricultural soils across China with an emphasis on unintentionally produced PCBs. Environ. Pollut. 2021, 271, 116171. [Google Scholar] [CrossRef]
- Yu, H.; Liu, Y.; Shu, X.; Ma, L.; Pan, Y. Assessment of the spatial distribution of organochlorine pesticides (OCPs) and polychlorinated biphenyls (PCBs) in urban soil of China. Chemosphere 2020, 243, 125392. [Google Scholar] [CrossRef]
- Vanier, C.; Sylvestre, M.; Planas, D. Persistence and fate of PCBs in sediments of the Saint Lawrence River. Sci. Total Environ. 1996, 192, 229–244. [Google Scholar] [CrossRef]
- Zhou, S.S.; Shao, L.Y.; Yang, H.Y.; Wang, C.; Liu, W.P. Residues and sources recognition of polychlorinated biphenyls in surface sediments of Jiaojiang Estuary, East China Sea. Mar. Pollut. Bull. 2012, 64, 539–545. [Google Scholar] [CrossRef]
- Frame, G.M.; Cochran, J.W.; Bøwadt, S.S. Complete PCB congener distributions for 17 aroclor mixtures determined by 3 HRGC systems optimized for comprehensive, quantitative, congener-specific analysis. J. High Resolut. Chromatogr. 1996, 19, 657–668. [Google Scholar] [CrossRef]
- Zhang, R.; Wang, Y.; Zhang, Y.; Bai, Y. Distribution, sources, and health risk of polycyclic aromatic hydrocarbons in farmland soil of Helan, China. Sustainability 2023, 15, 16667. [Google Scholar] [CrossRef]
- Tobiszewski, M.; Namieśnik, J. PAH diagnostic ratios for the identification of pollution emission sources. Environ. Pollut. 2012, 162, 110–119. [Google Scholar] [CrossRef]
- Zhang, W.; Zhang, S.; Wan, C.; Yue, D.; Ye, Y.; Wang, X. Source diagnostics of polycyclic aromatic hydrocarbons in urban road runoff, dust, rain and canopy throughfall. Environ. Pollut. 2008, 153, 594–601. [Google Scholar] [CrossRef]
- Zhu, L.; Chen, Y.; Zhou, R. Distribution of polycyclic aromatic hydrocarbons in water, sediment and soil in drinking water resource of Zhejiang Province, China. J. Hazard. Mater. 2008, 150, 308–316. [Google Scholar] [CrossRef]
- Cai, Y.; Wang, X.; Wu, Y.; Li, Y.; Ya, M. Over 100-year sedimentary record of polycyclic aromatic hydrocarbons (PAHs) and organochlorine compounds (OCs) in the continental shelf of the East China Sea. Environ. Pollut. 2016, 219, 774–784. [Google Scholar] [CrossRef]


| Body weight | Bw | kg | 70 (adult); 15 (child) |
| Averaging time | AT | days (d) | ED × 365 (non-carcinogenic), 25,550 (cancer) |
| Exposure frequency | EF | d/y | 350 |
| Exposure duration | ED | year (y) | 24 (adult); 6 (child) |
| Soil ingestion rate | IngR | mg/d | 100 (adult); 200 (child) |
| Soil inhalation rate | InhR | m3/d | 20 (adult); 7.6 (child) |
| Conversion factor | CF | kg/mg | 1 × 10−6 |
| Dermal surface exposure | SA | cm2/d | 5700 (adult); 2800 (child) |
| Adherence factor to skin | AF | mg cm2 | 0.2 (organic compound) |
| Dermal absorption factor | ABS | Unitless | 0.1 (organic compound) |
| Gastrointestinal absorption factor | ABSGI | Unitless | 1.0 (p,p′-DDD, p,p′-DDT, p,p′-DDE, α-HCH, γ-HCH, PCBs) |
| Particle emission factor | PEF | m3/kg | 1.36 × 109 |
| Ingestion reference values | RfDing | mg/kg/d | 0.002 (p,p′-DDD); 5 × 10−4 (p,p′-DDT); 0.008 (α-HCH); 3 × 10−4 (γ-HCH); 2.75 (BaP) |
| Dermal reference values | RfDdermal | mg/kg/d | 0.002 (p,p′-DDD); 5 × 10−4 (p,p′-DDT); 0.008 (α-HCH); 3 × 10−4 (γ-HCH), |
| Carcinogenic slope factor via ingestion | CSFing | 1/(mg/kg/d) | 0.24 (p,p′-DDD); 0.34 (p,p′-DDT); 0.34 (p,p′-DDE); 6.3 (α-HCH); 1.1 (γ-HCH); 7.3 (BaP); 2.0 (PCBs) |
| Dermal contact factor | CSFdermal | 1/(mg/kg/d) | 0.24 (p,p′-DDD); 0.34 (p,p′-DDT); 0.34 (p,p′-DDE); 6.3 (α-HCH); 1.1 (γ-HCH); 25 (BaP); 2.22 PCB |
| Inhalation unit risk | CSFinhal | mg/m3 | 0.069 (p,p′-DDD); 0.097 (p,p′-DDT); 0.097 (p,p′-DDE); 1.8 (α-HCH); 0.31 (γ-HCH); 3.85 (BaP); 0.57 (PCBs), |
| Toxic equivalent factor of PAH | TEF BaP | unitless | 0.001 (NAP, ACY, ACE, FL, PHE, FLT, PYR); 0.01 (ANT, CHR, BghiP); 0.1 (BaA, BbF, BkF, IND); 1 (BaP, DahA) |
| Sample Code | Total OCPs | HCH | DDT | Heptachlor | Cyclodiene | ∑DDT/ ∑HCH | α-HCH/ γ-HCH | DDE + DDD /∑DDT | DDE/ DDD | o,p′-DDT/ p,p′-DDT |
|---|---|---|---|---|---|---|---|---|---|---|
| P1 | 25.62 | 3.75 | 4.44 | 14.94 | 2.49 | 2.71 | 1.00 | 3.99 | - | 0.27 |
| P2 | 31.79 | 11.84 | 7.63 | 0.34 | 9.77 | 0.76 | 0.92 | 6.71 | 0.67 | 0.18 |
| P3 | 29.57 | 5.96 | 2.1 | 18.17 | 3.44 | 0.80 | 1.11 | 0.91 | - | 0.28 |
| P4 | 60.66 | 8.90 | 3.88 | 29.4 | 17.27 | 0.77 | 0.59 | 3.97 | - | 0.30 |
| P5 | 53.32 | 15.3 | 3.41 | 28.11 | 0.51 | 0.23 | 1.26 | 0.79 | 1.54 | 0.74 |
| P6 | 106.47 | 18.45 | 19.18 | 41.14 | 22.4 | 1.67 | 0.64 | 2.81 | - | 0.68 |
| P7 | 133.97 | 11.61 | 55.79 | 20.73 | 41.21 | 20.00 | 0.79 | 1.79 | - | 0.73 |
| P8 | 38.59 | 8.26 | 5.04 | 20.24 | 4.32 | 1.00 | 0.45 | 3.27 | 0.18 | 0.46 |
| P9 | 64.15 | 18.25 | 4.98 | 26.74 | 12.95 | 0.45 | 0.70 | 1.10 | 0.11 | 0.39 |
| P10 | 28.22 | 5.02 | 4.02 | 16.35 | 1.88 | 0.89 | 1.15 | 0.61 | - | 0.54 |
| P11 | 28.21 | 5.30 | 2.05 | 16.73 | 3.14 | 0.71 | 0.81 | 0.86 | - | 0.36 |
| P12 | 63.66 | 9.70 | 17.24 | 19.82 | 15.9 | 2.33 | 0.52 | 2.16 | - | 0.81 |
| P13 | 87.65 | 14.11 | 11.89 | 43.99 | 16.11 | 0.37 | 1.09 | 2.52 | - | 0.36 |
| P14 | 125.60 | 9.53 | 32.33 | 27.44 | 52.05 | 6.53 | 1.12 | 1.63 | - | 0.76 |
| P15 | 139.31 | 17.02 | 15.67 | 49.33 | 53.73 | 1.26 | 0.35 | 4.12 | - | 0.28 |
| P16 | 45.12 | 9.70 | 2.07 | 30.75 | nd | 0.25 | 1.00 | 0.29 | - | - |
| P17 | 32.97 | 5.01 | 1.59 | 18.93 | 4.71 | 0.47 | 1.38 | 3.97 | - | - |
| P18 | 79.62 | 19.81 | 14.08 | 36.33 | 9.40 | 1.00 | 0.53 | 3.99 | - | - |
| P19 | 111.29 | 10.48 | 39.37 | 24.22 | 27.77 | 5.62 | 1.08 | 4.15 | 0.17 | - |
| P20 | 74.70 | 23.03 | 10.96 | 28.85 | 10.57 | 1.07 | 1.09 | 3.98 | - | 0.22 |
| P21 | 62.91 | 14.16 | 7.05 | 32.7 | 6.60 | 0.97 | 0.77 | 4.28 | 0.97 | 0.84 |
| P22 | 30.12 | 5.23 | 0.65 | 15.77 | 6.90 | 0.22 | 1.68 | 0.76 | - | - |
| Sample Code | Total PAH | ∑Comb | %∑Comb of PAHs | ∑LMW/ ∑HMW | FL/ (FL + PYR) | ANT/ (ANT + PHEN) | FLT/ (FLT + PYR) |
|---|---|---|---|---|---|---|---|
| P1 | 168.74 | 166.3 | 98.56 | 0.015 | 0.008 | 0.45 | 0.63 |
| P2 | 451.85 | 447 | 98.93 | 0.011 | 0.002 | 0.35 | 0.62 |
| P3 | 9.58 | 8.97 | 93.65 | 0.068 | 0.043 | 0.25 | 0.58 |
| P4 | 18.18 | 17.03 | 93.69 | 0.067 | 0.071 | 0.50 | 0.59 |
| P5 | 26.17 | 25.76 | 98.42 | 0.016 | 0.041 | 0.40 | 0.36 |
| P6 | 62.61 | 60.60 | 92.36 | 0.083 | 0.001 | 0.40 | 0.54 |
| P7 | 6.49 | 4.86 | 74.80 | 0.337 | 0.434 | 0.42 | 0.58 |
| P8 | 87.08 | 84.75 | 97.33 | 0.027 | 0.003 | 0.30 | 0.61 |
| P9 | 5.53 | 4.18 | 75.72 | 0.320 | 0.616 | 0.55 | 0.48 |
| P10 | 12.17 | 12.03 | 98.82 | 0.012 | 0.026 | 0.40 | 0.43 |
| P11 | 4.86 | 4.77 | 98.21 | 0.018 | 0.295 | 0.29 | 0.77 |
| P12 | 94.09 | 85.79 | 91.17 | 0.097 | 0.020 | 0.43 | 0.63 |
| P13 | 52.03 | 50.57 | 97.21 | 0.029 | 0.011 | 0.28 | 0.67 |
| P14 | 15.02 | 13.16 | 87.52 | 0.143 | 0.097 | 0.44 | 0.69 |
| P15 | 16.45 | 15.32 | 93.04 | 0.075 | 0.028 | 0.44 | 0.49 |
| P16 | 2.99 | 2.29 | 76.36 | 0.310 | 0.458 | 0.31 | 0.58 |
| P17 | 6.44 | 5.38 | 83.69 | 0.198 | 0.901 | 0.40 | 0.44 |
| P18 | 24.37 | 21.89 | 89.83 | 0.113 | 0.039 | 0.46 | 0.59 |
| P19 | 34.85 | 32.84 | 94.21 | 0.061 | 0.001 | 0.40 | 0.55 |
| P20 | 108.59 | 106.75 | 98.30 | 0.017 | 0.007 | 0.48 | 0.57 |
| P21 | 26.89 | 25.81 | 96.03 | 0.041 | 0.058 | 0.28 | 0.72 |
| P22 | 28.33 | 27.22 | 96.13 | 0.040 | 0.071 | 0.32 | 0.68 |
| Compound | HIAdult | HIChild | Cancer RiskAdult | Cancer RiskChild | ||||
|---|---|---|---|---|---|---|---|---|
| Min | Max | Min | Max | Min | Max | Min | Max | |
| α-HCH | 0.15 × 10−5 | 1.15 × 10−5 | 0.16 × 10−5 | 1.18 × 10−5 | 7.80 × 10−8 | 57.93 × 10−8 | 8.05 × 10−8 | 59.78 × 10−8 |
| γ-HCH | 4.12 × 10−5 | 4.62 × 10−4 | 4.26 × 10−5 | 4.77 × 10−4 | 1.36 × 10−8 | 15.26 × 10−8 | 1.41 × 10−8 | 1.57 × 10−7 |
| ∑DDD | 0.21 × 10−5 | 2.7 × 10−4 | 0.21 × 10−5 | 2.78 × 10−4 | 1.10 × 10−8 | 1.40 × 10−6 | 0.10 × 10−8 | 1.33 × 10−7 |
| ∑DDE | - | - | - | - | - | 8.78 × 10−8 | - | 0.90 × 10−7 |
| ∑DDT | 0.31 × 10−5 | 1.96 × 10−4 | 042 × 10−5 | 2.63 × 10−4 | 0.17 × 10−8 | 1.02 × 10−7 | 0.17 × 10−8 | 1.06 × 10−7 |
| ∑16PAH | 0.31 × 10−8 | 16.04 × 10−8 | 0.10 × 10−8 | 5.29 × 10−8 | 5.56 × 10−8 | 2.89 × 10−6 | 1.80 × 10−7 | 9.37 × 10−6 |
| ∑12PCB | - | - | - | - | 0.67 × 10−8 | 1.49 × 10−6 | 0.70 × 10−8 | 1.569 × 10−6 |
| Carcinogenic Risk CR | α-HCH | γ-HCH | DDD | DDT | ∑16PAH | ∑12PCB | |
|---|---|---|---|---|---|---|---|
| Ingestion (×10−8) | Adult | 25.02 | 5.68 | 1.11 | 0.94 | 6.25 | 25.15 |
| Child | 22.08 | 5.02 | 0.98 | 0.83 | 24.05 | 22.19 | |
| Dermal (×10−8) | Adult | 2.76 | 0.63 | 0.125 | 0.105 | 4.47 | 3.08 |
| Child | 6.59 | 1.50 | 0.295 | 0.245 | 10.68 | 7.35 | |
| Inhalation (×10−12) | Adult | 1.05 | 0.18 | 0.045 | 0.04 | 0.92 | 1.05 |
| Child | 1.88 | 0.42 | 0.085 | 0.07 | 1.64 | 1.88 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pănescu, V.-A.; Bocoș-Bințințan, V.; Herghelegiu, M.-C.; Coman, R.-T.; Berg, V.; Lyche, J.L.; Beldean-Galea, M.S. Pollution Assessment with Persistent Organic Pollutants in Upper Soil of a Series of Rural Roma Communities in Transylvania, Romania, Its Sources Apportionment, and the Associated Risk on Human Health. Sustainability 2024, 16, 232. https://doi.org/10.3390/su16010232
Pănescu V-A, Bocoș-Bințințan V, Herghelegiu M-C, Coman R-T, Berg V, Lyche JL, Beldean-Galea MS. Pollution Assessment with Persistent Organic Pollutants in Upper Soil of a Series of Rural Roma Communities in Transylvania, Romania, Its Sources Apportionment, and the Associated Risk on Human Health. Sustainability. 2024; 16(1):232. https://doi.org/10.3390/su16010232
Chicago/Turabian StylePănescu, Vlad-Alexandru, Victor Bocoș-Bințințan, Mihaela-Cătălina Herghelegiu, Radu-Tudor Coman, Vidar Berg, Jan Ludvig Lyche, and Mihail Simion Beldean-Galea. 2024. "Pollution Assessment with Persistent Organic Pollutants in Upper Soil of a Series of Rural Roma Communities in Transylvania, Romania, Its Sources Apportionment, and the Associated Risk on Human Health" Sustainability 16, no. 1: 232. https://doi.org/10.3390/su16010232
APA StylePănescu, V.-A., Bocoș-Bințințan, V., Herghelegiu, M.-C., Coman, R.-T., Berg, V., Lyche, J. L., & Beldean-Galea, M. S. (2024). Pollution Assessment with Persistent Organic Pollutants in Upper Soil of a Series of Rural Roma Communities in Transylvania, Romania, Its Sources Apportionment, and the Associated Risk on Human Health. Sustainability, 16(1), 232. https://doi.org/10.3390/su16010232







