Stand Structure Beats Age for Ground Cover Vegetation in Ageing Hemiboreal Scots Pine and Norway Spruce Stands
Abstract
1. Introduction
2. Materials and Methods
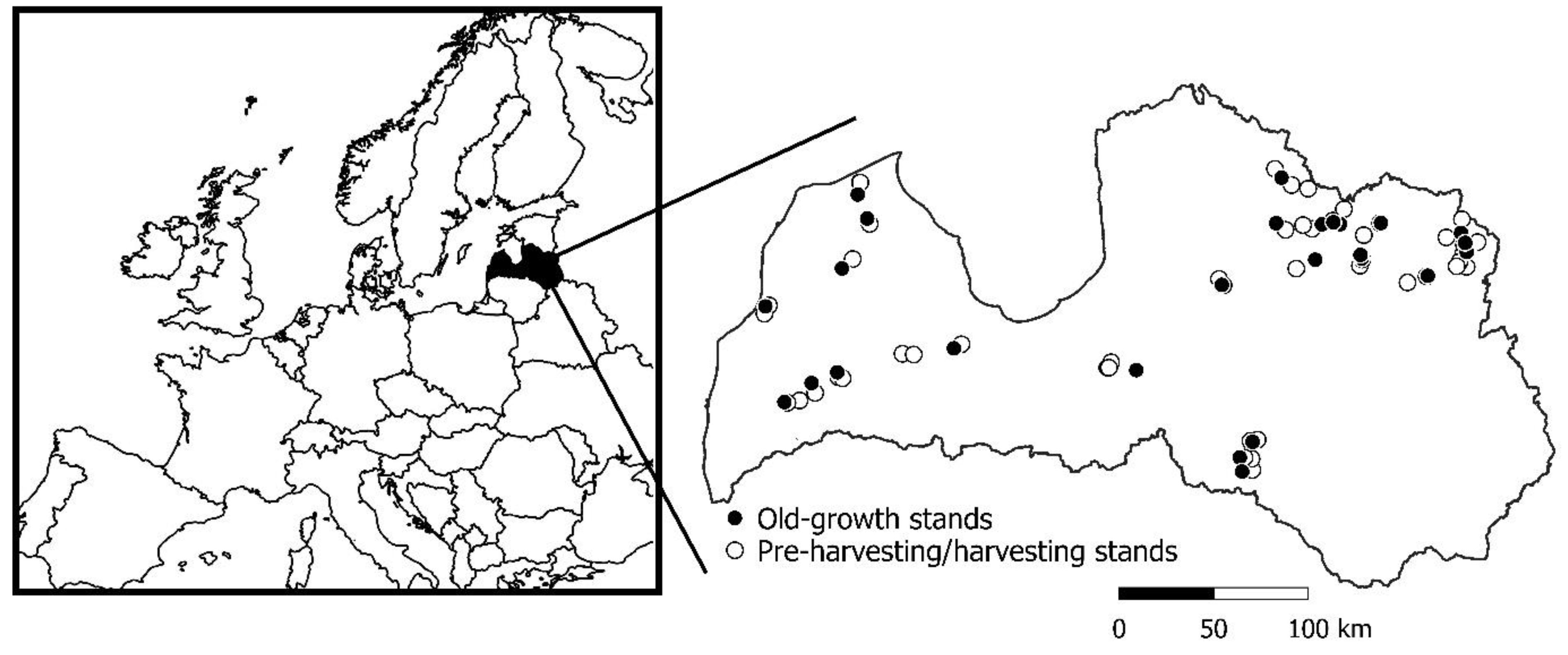
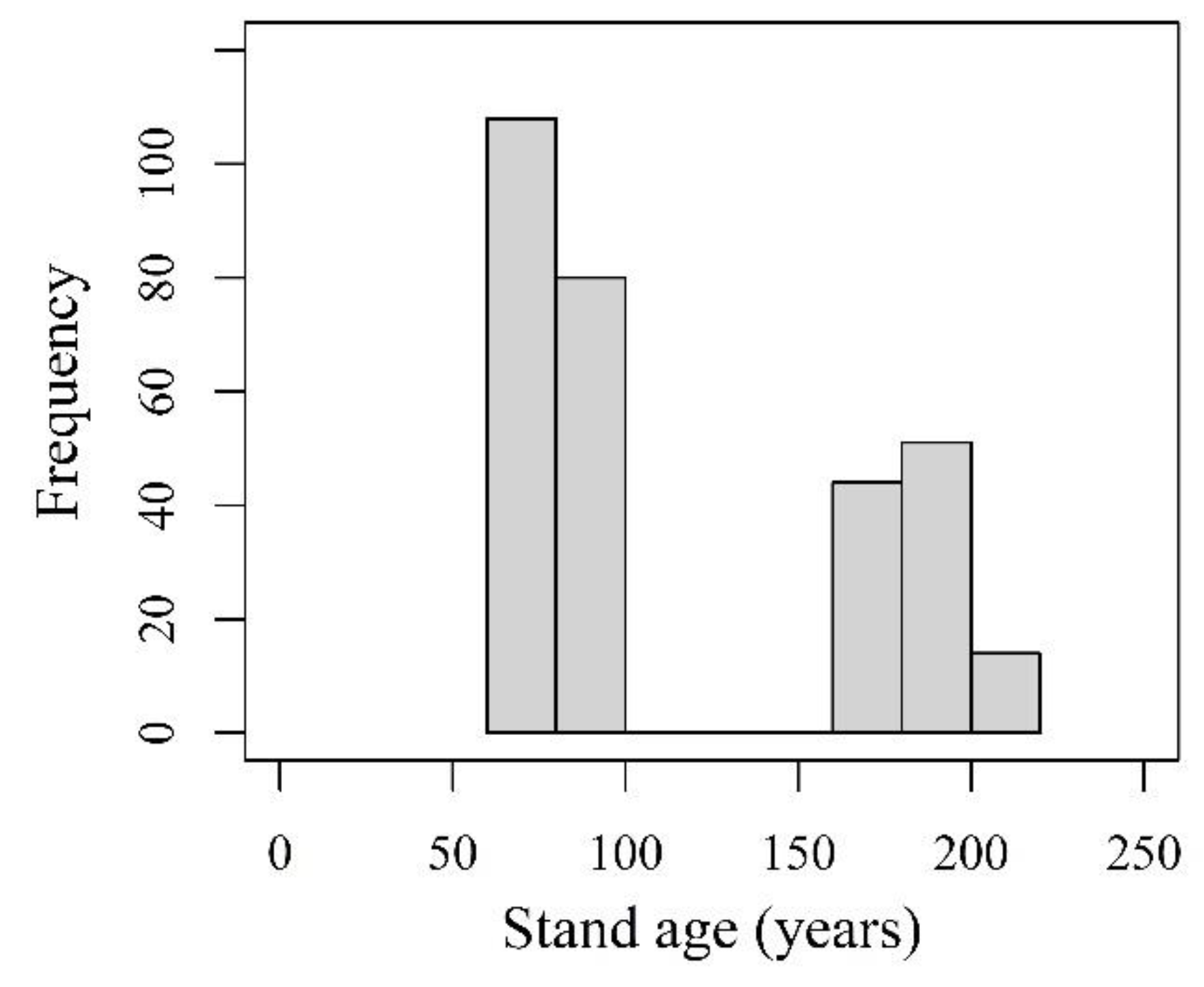
2.1. Study Area and Stand Selection
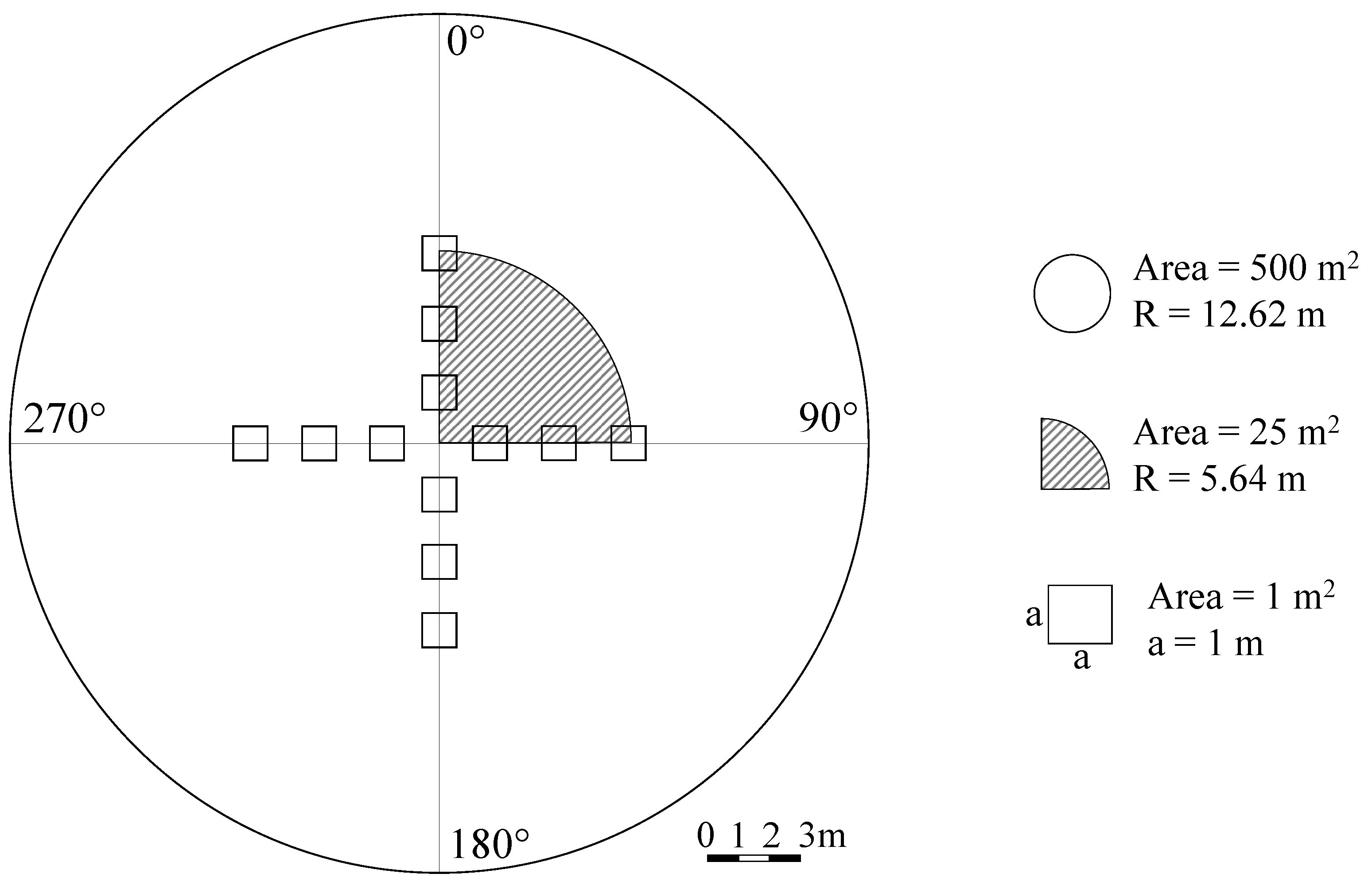
2.2. Measurements and Census
2.3. Statistical Analysis
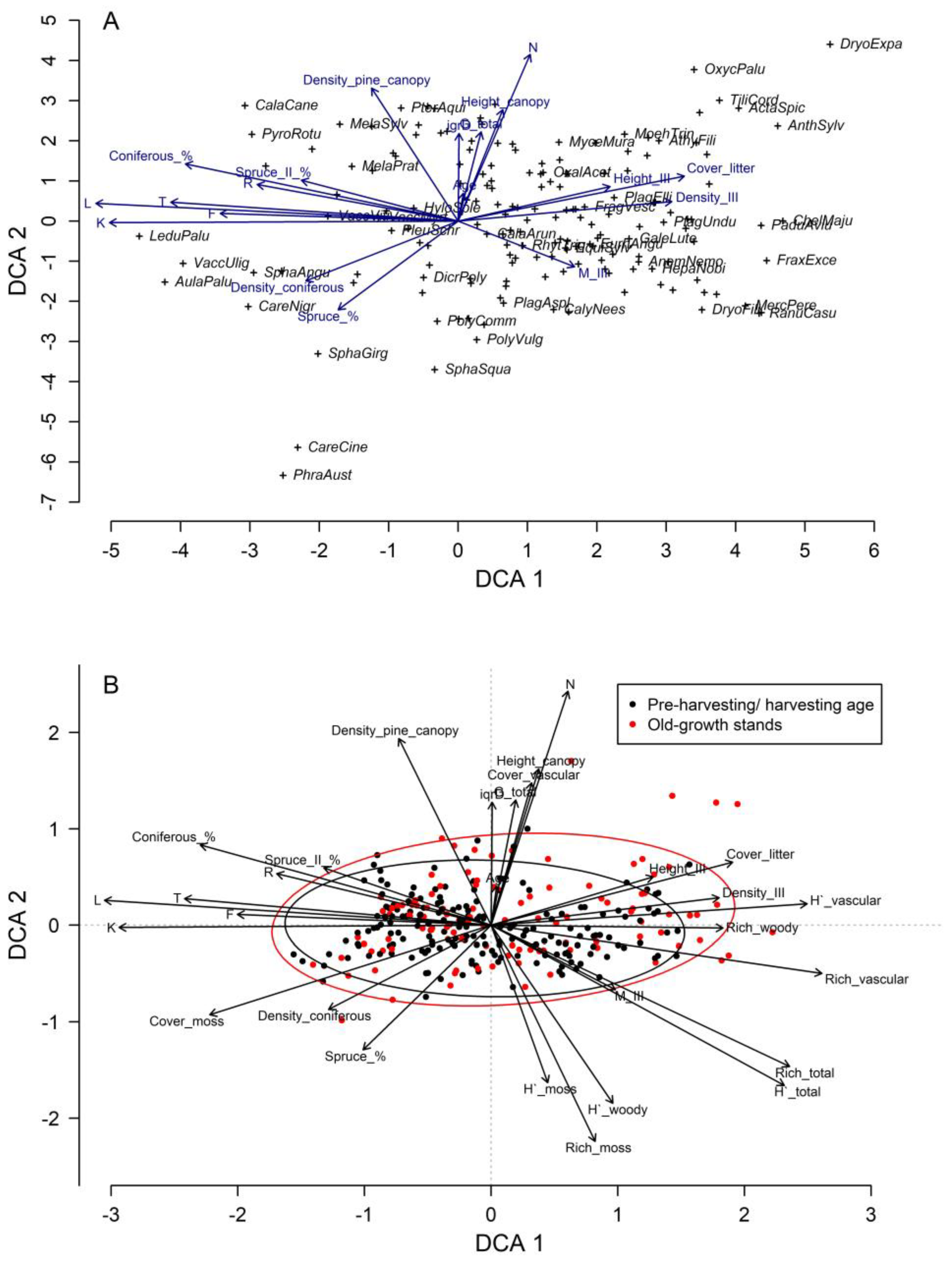
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Routa, J.; Kilpeläinen, A.; Ikonen, V.P.; Asikainen, A.; Venäläinen, A.; Peltola, H. Effects of intensified silviculture on timber production and its economic profitability in boreal Norway spruce and Scots pine stands under changing climatic conditions. For. Int. J. For. Res. 2019, 92, 648–658. [Google Scholar] [CrossRef]
- Temperli, C.; Stadelmann, G.; Thürig, E.; Brang, P. Silvicultural strategies for increased timber harvesting in a Central European mountain landscape. Eur. J. For. Res. 2017, 136, 493–509. [Google Scholar] [CrossRef]
- Socha, J.; Solberg, S.; Tymińska-Czabańska, L.; Tompalski, P.; Vallet, P. Height growth rate of Scots pine in Central Europe increased by 29% between 1900 and 2000 due to changes in site productivity. For. Ecol. Manag. 2021, 490, 119102. [Google Scholar] [CrossRef]
- Lange, M.; Türke, M.; Pašalić, E.; Boch, S.; Hessenmöller, D.; Müller, J.; Prati, D.; Socher, S.A.; Fischer, M.; Weisser, W.W.; et al. Effects of forest management on ground-dwelling beetles (Coleoptera; Carabidae, Staphylinidae) in Central Europe are mainly mediated by changes in forest structure. For. Ecol. Manag. 2014, 329, 166–176. [Google Scholar] [CrossRef]
- Södra Skog, Lönsamt Med Kortare Omloppstid i Granskog. [Shorter Rotations are Profitable in Spruce Forest] (Press Release). 2012. Available online: https://www.sodra.com/sv/se/om-sodra/pressrum/pressmeddelanden/lonsamt-med-kortare-omloppstid-i-granskog/ (accessed on 20 February 2023). (In Swedish).
- Zimová, S.; Dobor, L.; Hlásny, T.; Rammer, W.; Seidl, R. Reducing rotation age to address increasing disturbances in Central Europe: Potential and limitations. For. Ecol. Manag. 2020, 475, 118408. [Google Scholar] [CrossRef] [PubMed]
- Petrokas, R.; Baliuckas, V.; Manton, M. Successional categorization of European hemi-boreal forest tree species. Plants 2020, 9, 1381. [Google Scholar] [CrossRef]
- Hanewinkel, M.; Cullmann, D.A.; Schelhaas, M.-J.; Nabuurs, G.J.; Zimmermann, N.E. Climate change may cause severe loss in the economic value of European forest land. Nat. Clim. Chang. 2013, 3, 203–207. [Google Scholar] [CrossRef]
- Molina-Valero, J.A.; Camarero, J.J.; Alvarez-Gonzalez, J.G.; Cerioni, M.; Hevia, A.; Sanchez-Salguero, R.; Martín-Benito, D.; Perez-Cruzado, C. Mature forests hold maximum live biomass stocks. For. Ecol. Manag. 2021, 480, 118635. [Google Scholar] [CrossRef]
- Roberge, J.-M.; Laudon, H.; Björkman, C.; Ranius, T.; Sandström, C.; Felton, A.; Sténs, A.; Nordin, A.; Granström, A.; Widemo, F.; et al. Socio-ecological implications of modifying rotation lengths in forestry. Ambio 2016, 45, 109–123. [Google Scholar] [CrossRef] [PubMed]
- Gao, T.; Hedblom, M.; Emilsson, T.; Nielsen, A.B. The role of forest stand structure as biodiversity indicator. For. Ecol. Manag. 2014, 330, 82–93. [Google Scholar] [CrossRef]
- Humphrey, J.W. Benefits to biodiversity from developing old-growth conditions in British upland spruce plantations: A review and recommendations. Forestry 2005, 78, 33–53. [Google Scholar] [CrossRef]
- Zanchi, G.; Brady, M.V. Evaluating the contribution of forest ecosystem services to societal welfare through linking dynamic ecosystem modelling with economic valuation. Ecosyst. Serv. 2019, 39, 101011. [Google Scholar] [CrossRef]
- Bauhus, J.; Puettmann, K.; Messier, C. Silviculture for old-growth attributes. For. Ecol. Manag. 2009, 258, 525–537. [Google Scholar] [CrossRef]
- Betts, M.G.; Phalan, B.T.; Wolf, C.; Baker, S.C.; Messier, C.; Puettmann, K.J.; Green, R.; Harris, S.H.; Edwards, D.P.; Lindenmayer, D.B.; et al. Producing wood at least cost to biodiversity: Integrating T riad and sharing–sparing approaches to inform forest landscape management. Biol. Rev. 2021, 96, 1301–1317. [Google Scholar] [CrossRef] [PubMed]
- Himes, A.; Betts, M.; Messier, C.; Seymour, R. Perspectives: Thirty years of triad forestry, a critical clarification of theory and recommendations for implementation and testing. For. Ecol. Manag. 2022, 510, 120103. [Google Scholar] [CrossRef]
- Kraus, D.; Krumm, F. (Eds.) Integrative Approaches as an Opportunity for the Conservation of Forest Biodiversity; European Forest Institute: Freiburg, Germany, 2013; 284p. [Google Scholar]
- Royer-Tardif, S.; Bauhus, J.; Doyon, F.; Nolet, P.; Thiffault, N.; Aubin, I. Revisiting the functional zoning concept under climate change to expand the portfolio of adaptation options. Forests 2021, 12, 273. [Google Scholar] [CrossRef]
- Tittler, R.; Filotas, E.; Kroese, J.; Messier, C. Maximizing conservation and production with intensive forest management: It’s all about location. Environ. Manag. 2015, 56, 1104–1117. [Google Scholar] [CrossRef]
- Burrascano, S.; Keeton, W.S.; Sabatini, F.M.; Blasi, C. Commonality and variability in the structural attributes of moist temperate old-growth forests: A global review. For. Ecol. Manag. 2013, 291, 458–479. [Google Scholar] [CrossRef]
- Crites, S.; Dale, M.R. Diversity and abundance of bryophytes, lichens, and fungi in relation to woody substrate and successional stage in aspen mixedwood boreal forests. Can. J. Bot. 1998, 76, 641–651. [Google Scholar] [CrossRef]
- Torresan, C.; del Río, M.; Hilmers, T.; Notarangelo, M.; Bielak, K.; Binder, F.; Boncina, A.; Bosela, M.; Forrester, D.I.; Hobi, M.L.; et al. Importance of tree species size dominance and heterogeneity on the productivity of spruce-fir-beech mountain forest stands in Europe. For. Ecol. Manag. 2020, 457, 117716. [Google Scholar] [CrossRef]
- Viljur, M.; Abella, S.R.; Adámek, M.; Alencar, J.B.R.; Barber, N.A.; Beudert, B.; Burkle, L.A.; Cagnolo, L.; Campos, B.R.; Chao, A.; et al. The effect of natural disturbances on forest biodiversity: An ecological synthesis. Biol. Rev. 2022, 97, 1930–1947. [Google Scholar] [CrossRef] [PubMed]
- Mayor, S.J.; Cahill, J.F., Jr.; He, F.; Sólymos, P.; Boutin, S. Regional boreal biodiversity peaks at intermediate human disturbance. Nat. Commun. 2012, 3, 1142. [Google Scholar] [CrossRef] [PubMed]
- Paillet, Y.; Bergès, L.; Hjältén, J.; Ódor, P.; Avon, C.; Bernhardt-Römermann, M.; Bijlsma, R.-J.; De Bruyn, L.; Fuhr, M.; Grandin, U.; et al. Biodiversity differences between managed and unmanaged forests: Meta-analysis of species richness in Europe. Conserv. Biol. 2010, 24, 101–112. [Google Scholar] [CrossRef] [PubMed]
- Chernenkova, T.; Kotlov, I.; Belyaeva, N.; Suslova, E.; Morozova, O.; Pesterova, O.; Arkhipova, M. Role of silviculture in the formation of Norway spruce forests along the southern edge of their range in the Central Russian Plain. Forests 2020, 11, 778. [Google Scholar] [CrossRef]
- Marozas, V.; Sasnauskiene, J. Changes of ground vegetation after shelter wood cuttings in pine forests, the hemiboreal zone, Lithuania. Balt. For. 2021, 27, 72–79. [Google Scholar] [CrossRef]
- Oettel, J.; Lapin, K. Linking forest management and biodiversity indicators to strengthen sustainable forest management in Europe. Ecol. Indic. 2021, 122, 107275. [Google Scholar] [CrossRef]
- Smith, G.F.; Gittings, T.; Wilson, M.; French, L.; Oxbrough, A.; O’donoghue, S.; O’halloran, J.; Kelly, D.L.; Mitchell, F.J.G.; Kelly, T.; et al. Identifying practical indicators of biodiversity for stand-level management of plantation forests. In Plantation Forests and Biodiversity: Oxymoron or Opportunity? Topics in Biodiversity and Conservation; Brockerhoff, E.G., Jactel, H., Parrotta, J.A., Quine, C.P., Sayer, J., Hawksworth, D.L., Eds.; Springer: Dordrecht, The Netherlands, 2007; Volume 9. [Google Scholar] [CrossRef]
- Ćosović, M.; Bugalho, M.N.; Thom, D.; Borges, J.G. Stand structural characteristics are the most practical biodiversity indicators for forest management planning in Europe. Forests 2020, 11, 343. [Google Scholar] [CrossRef]
- Barbier, S.; Gosselin, F.; Balandier, P. Influence of tree species on understory vegetation diversity and mechanisms involved: A critical review for temperate and boreal forests. For. Ecol. Manag. 2008, 254, 1–15. [Google Scholar] [CrossRef]
- Coote, L.; Dietzsch, A.C.; Wilson, M.W.; Graham, C.T.; Fuller, L.; Walsh, A.T.; Irwin, S.; Kelly, D.L.; Mitchell, F.J.; Kelly, T.C.; et al. Testing indicators of biodiversity for plantation forests. Ecol. Indic. 2013, 32, 107–115. [Google Scholar] [CrossRef]
- Lassauce, A.; Paillet, Y.; Jactel, H.; Bouget, C. Deadwood as a surrogate for forest biodiversity: Meta-analysis of correlations between deadwood volume and species richness of saproxylic organisms. Ecol. Indic. 2011, 11, 1027–1039. [Google Scholar] [CrossRef]
- Parisi, F.; Lombardi, F.; Sciarretta, A.; Tognetti, R.; Campanaro, A.; Marchetti, M.; Trematerra, P. Spatial patterns of saproxylic beetles in a relic silver fir forest (Central Italy), relationships with forest structure and biodiversity indicators. For. Ecol. Manag. 2016, 381, 217–234. [Google Scholar] [CrossRef]
- Larrieu, L.; Paillet, Y.; Winter, S.; Bütler, R.; Kraus, D.; Krumm, F.; Lachat, T.; Michel, A.K.; Regnery, B.; Vandekerkhove, K. Tree related microhabitats in temperate and Mediterranean European forests: A hierarchical typology for inventory standardization. Ecol. Indic. 2018, 84, 194–207. [Google Scholar] [CrossRef]
- Sever, K.; Nagel, T.A. Patterns of tree microhabitats across a gradient of managed to old-growth conditions: A case study from beech dominated forests of South-Eastern Slovenia. Acta Silvae Ligni 2019, 118, 29–40. [Google Scholar] [CrossRef]
- Commarmot, B.; Bachofen, H.; Bundziak, Y.; Bürgi, A.; Ramp, B.; Shparyk, Y.; Sukhariuk, D.; Viter, R.; Zingg, A. Structures of virgin and managed beech forests in Uholka (Ukraine) and Sihlwald (Switzerland): A comparative study. For. Snow Landsc. Res. 2005, 79, 45–56. [Google Scholar]
- Humphrey, J.W.; Davey, S.; Peace, A.J.; Ferris, R.; Harding, K. Lichens and bryophyte communities of planted and semi-natural forests in Britain: The influence of site type, stand structure and deadwood. Biol. Conserv. 2002, 107, 165–180. [Google Scholar] [CrossRef]
- Felton, A.M.; Wam, H.K.; Stolter, C.; Mathisen, K.M.; Wallgren, M. The complexity of interacting nutritional drivers behind food selection, a review of northern cervids. Ecosphere 2018, 9, e02230. [Google Scholar] [CrossRef]
- Felton, A.; Knight, E.; Wood, J.; Zammit, C.; Lindenmayer, D. A meta-analysis of fauna and flora species richness and abundance in plantations and pasture lands. Biol. Conserv. 2010, 143, 545–554. [Google Scholar] [CrossRef]
- Hart, S.A.; Chen, H.Y.H. Understory vegetation dynamics of North American boreal forests. Crit. Rev. Plant Sci. 2006, 25, 381–397. [Google Scholar] [CrossRef]
- Chavez, V.; Macdonald, S.E. Partitioning vascular understory diversity in mixedwood boreal forests: The importance of mixed canopies for diversity conservation. For. Ecol. Manag. 2012, 271, 19–26. [Google Scholar] [CrossRef]
- Nilsson, M.C.; Wardle, D.A. Understory vegetation as a forest ecosystem driver: Evidence from the northern Swedish boreal forest. Front. Ecol. Environ. 2005, 3, 421–428. [Google Scholar] [CrossRef]
- O’Brien, M.J.; O’Hara, K.L.; Erbilgin, N.; Wood, D.L. Overstory and shrub effects on natural regeneration processes in native Pinus radiata stands. For. Ecol. Manag. 2007, 240, 178–185. [Google Scholar] [CrossRef]
- Kuuluvainen, T.; Angelstam, P.; Frelich, L.; Jõgiste, K.; Koivula, M.; Kubota, Y.; Lafleur, B.; Macdonald, E. Natural disturbance-based forest management: Moving beyond retention and continuous-cover forestry. Front. For. Glob. Chang. 2021, 4, 629020. [Google Scholar] [CrossRef]
- Durrant, T.H.; De Rigo, D.; Caudullo, G. Pinus sylvestris in Europe: Distribution, habitat, usage and threats. Eur. Atlas For. Tree Species 2016, 14, 845–846. [Google Scholar]
- Schlyter, P.; Stjernquist, I.; Barring, L.; Jonsson, A.M.; Nilsson, C. Assessment of the impacts of climate change and weather extremes on boreal forests in northern Europe, focusing on Norway spruce. Clim. Res. 2006, 31, 75–84. [Google Scholar] [CrossRef]
- Buras, A.; Menzel, A. Projecting tree species composition changes of European forests for 2061–2090 under RCP 4.5 and RCP 8.5 scenarios. Front. Plant Sci. 2019, 9, 1986. [Google Scholar] [CrossRef]
- Albert, M.; Nagel, R.V.; Nuske, R.; Sutmöller, J.; Spellmann, H. Tree species selection in the face of drought risk—Uncertainty in forest planning. Forests 2017, 8, 363. [Google Scholar] [CrossRef]
- Jactel, H.; Petit, J.; Desprez-Loustau, M.L.; Delzon, S.; Piou, D.; Battisti, A.; Koricheva, J. Drought effects on damage by forest insects and pathogens: A meta-analysis. Glob. Chang. Biol. 2012, 18, 267–276. [Google Scholar] [CrossRef]
- Krisans, O.; Saleniece, R.; Rust, S.; Elferts, D.; Kapostins, R.; Jansons, A.; Matisons, R. Effect of bark-stripping on mechanical stability of Norway spruce. Forests 2020, 11, 357. [Google Scholar] [CrossRef]
- Seidl, R.; Thom, D.; Kautz, M.; Martin-Benito, D.; Peltoniemi, M.; Vacchiano, G.; Wild, J.; Ascoli, D.; Petr, M.; Honkaniemi, J.; et al. Forest disturbances under climate change. Nat. Clim. Chang. 2017, 7, 395–402. [Google Scholar] [CrossRef]
- Ķēniņa, L.; Elferts, D.; Jaunslaviete, I.; Bāders, E.; Jansons, Ā. Sustaining Carbon Storage: Lessons from Hemiboreal Old-Growth Coniferous and Deciduous Forest Stands. For. Sci. 2023, 69, 158–166. [Google Scholar] [CrossRef]
- Ķēniņa, L.; Elferts, D.; Jaunslaviete, I.; Bāders, E.; Šņepsts, G.; Jansons, Ā. Tree biomass–a fragile carbon storage in old-growth birch and aspen stands in hemiboreal Latvia. Balt. For. 2022, 28, N654. [Google Scholar] [CrossRef]
- Ahti, T.; Hämet-Ahti, L.; Jalas, J. Vegetation zones and their sections in northwestern Europe. Ann. Bot. Fenn. 1968, 5, 169–211. [Google Scholar]
- Kottek, M.; Grieser, J.; Beck, C.; Rudolf, B.; Rubel, F. World map of the Köppen-Geiger climate classification updated. Meteorol. Z. 2006, 15, 259–263. [Google Scholar] [CrossRef]
- Avotniece, Z.; Rodinov, V.; Lizuma, L.; Briede, A.; Kļaviņš, M. Trends in frequency of extreme climate events in Latvia. Baltica 2010, 23, 135–148. [Google Scholar]
- Sandström, F.; Petersson, H.; Kruys, N.; Ståhl, G. Biomass conversion factors (density and carbon concentration) by decay classes for dead wood of Pinus sylvestris, Picea abies and Betula spp. in boreal forests of Sweden. For. Ecol. Manag. 2017, 243, 19–27. [Google Scholar] [CrossRef]
- Liepa, I. Tree Growth Study; LUA: Jelgava, Latvia, 1996; 123p. (In Latvian) [Google Scholar]
- Ellenberg, H.; Weber, H.E.; Düll, R.; Wirth, V.; Werner, W.; Paulissen, D. Zeigerwerte von Pflanzen in Mitteleuropa, 2nd ed.; Scripta Geobotanica: Göttingen, Germany, 1992; pp. 1–248. [Google Scholar]
- Oksanen, J.; Simpson, G.; Blanchet, F.G.; Kindt, R.; Legendre, P.; Minchin, P.; Hara, R.; Solymos, P.; Stevens, H. Vegan: Community Ecology Package, R Package Version 2.6-4. 2022. Available online: https://CRAN.R-project.org/package=vegan (accessed on 31 March 2023).
- Correa-Metrio, A.; Dechnik, Y.; Lozano-García, S.; Caballero, M. Detrended correspondence analysis: A useful tool to quantify ecological changes from fossil data sets. Boletín Soc. Geológica Mex. 2014, 66, 135–143. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2022; Available online: https://www.R-project.org/ (accessed on 15 March 2023).
- Bates, D.; Maechler, M.; Bolker, B.; Walker, S. Fitting Linear Mixed-Effects Models Using lme4. J. Stat. Softw. 2015, 67, 1–48. [Google Scholar] [CrossRef]
- Heinken, T.; Diekmann, M.; Liira, J.; Orczewska, A.; Schmidt, M.; Brunet, J.; Chytrý, M.; Chabrerie, O.; Decocq, G.; De Frenne, P.; et al. The European forest plant species list (EuForPlant): Concept and applications. J. Veg. Sci. 2022, 33, e13132. [Google Scholar] [CrossRef]
- Bušs, K. Forest Ecology and Typology; Zinātne: Rīga, Latvia, 1981; 68p. (In Latvian) [Google Scholar]
- Marozas, V.; Racinskas, J.; Bartkevicius, E. Dynamics of ground vegetation after surface fires in hemiboreal Pinus sylvestris forests. For. Ecol. Manag. 2007, 250, 47–55. [Google Scholar] [CrossRef]
- Busby, J.R.; Bliss, L.C.; Hamilton, C.D. Microclimate control of growth rates and habitats of the boreal forest mosses, Tomenthypnum nitens and Hylocomium splendens. Ecol. Monogr. 1978, 48, 95–110. [Google Scholar] [CrossRef]
- Lõhmus, A.; Remm, L. Disentangling the effects of seminatural forestry on an ecosystem good: Bilberry (Vaccinium myrtillus) in Estonia. For. Ecol. Manag. 2017, 404, 75–83. [Google Scholar] [CrossRef]
- Timoshok, E.E. The ecology of bilberry (Vaccinium myrtillus L.) and cowberry (Vaccinium vitis-idaea L.) in Western Siberia. Russ. J. Ecol. 2000, 31, 8–13. [Google Scholar] [CrossRef]
- Petersson, L.; Holmström, E.; Lindbladh, M.; Felton, A. Tree species impact on understory vegetation: Vascular plant communities of Scots pine and Norway spruce managed stands in northern Europe. For. Ecol. Manag. 2019, 448, 330–345. [Google Scholar] [CrossRef]
- Augusto, L.; De Schrijver, A.; Vesterdal, L.; Smolander, A.; Prescott, C.; Ranger, J. Influences of evergreen gymnosperm and deciduous angiosperm tree species on the functioning of temperate and boreal forests. Biol. Rev. 2015, 90, 444–466. [Google Scholar] [CrossRef] [PubMed]
- Augusto, L.; Dupouey, J.L.; Ranger, J. Effects of tree species on understory vegetation and environmental conditions in temperate forests. Ann. For. Sci. 2003, 60, 823–831. [Google Scholar] [CrossRef]
- Packham, J.R. Biological Flora of the British Isles. No. 141. Oxalis acetosella L. J. Ecol. 1978, 66, 669–693. [Google Scholar] [CrossRef]
- Hitchcock, C.L.; Cronquist, A.; Ownbey, M.; Thompson, J.W. Vascular Plants of the Pacific Northwest; University of Washington Press: Washington, DC, USA, 1969; 914p. [Google Scholar]
- Kovács, B.; Tinya, F.; Ódor, P. Stand structural drivers of microclimate in mature temperate mixed forests. Agric. For. Meteorol. 2017, 234, 11–21. [Google Scholar] [CrossRef]
- French, L.J.; Smith, G.F.; Kelly, D.L.; Mitchell, F.J.; O’Donoghue, S.; Iremonger, S.F.; McKee, A.M. Ground flora communities in temperate oceanic plantation forests and the influence of silvicultural, geographic and edaphic factors. For. Ecol. Manag. 2008, 255, 476–494. [Google Scholar] [CrossRef]
- Coroi, M.; Skeffington, M.S.; Giller, P.; Smith, C.; Gormally, M.; O’Donovan, G. Vegetation diversity and stand structure in streamside forests in the south of Ireland. For. Ecol. Manag. 2004, 202, 39–57. [Google Scholar] [CrossRef]
- Saetre, P.; Saetre, L.S.; Brandtberg, P.O.; Lundkvist, H.; Bengtsson, J. Ground vegetation composition and heterogeneity in pure Norway spruce and mixed Norway spruce–birch stands. Can. J. For. Res. 1997, 27, 2034–2042. [Google Scholar] [CrossRef]
- Eriksson, O. Seedling recruitment in deciduous forest herbs: The effects of litter, soil chemistry and seedbank. Flora 1995, 190, 65–70. [Google Scholar] [CrossRef]
- Esteso-Martínez, J.; Gil-Pelegrín, E. Frost resistance of seeds in Mediterranean oaks and the role of litter in the thermal protection of acorns. Ann. For. Sci. 2004, 61, 481–486. [Google Scholar] [CrossRef]
- Graae, B.J.; Heskjær, V.S. A comparison of understorey vegetation between untouched and managed deciduous forest in Denmark. For. Ecol. Manag. 1997, 96, 111–123. [Google Scholar] [CrossRef]
- Ellsworth, J.; Harrington, R.; Fownes, J. Seedling emergence, growth, and allocation of oriental bittersweet: Effects of seed input, seed bank, and forest floor litter. For. Ecol. Manag. 2004, 190, 255–264. [Google Scholar] [CrossRef]
- Moning, C.; Werth, S.; Dziock, F.; Bässler, C.; Bradtka, J.; Hothorn, T.; Müller, J. Lichen diversity in temperate montane forests is influenced by forest structure more than climate. For. Ecol. Manag. 2009, 258, 745–751. [Google Scholar] [CrossRef]
- Bujoczek, L.; Bujoczek, M.; Zięba, S. How much, why and where? Deadwood in forest ecosystems: The case of Poland. Ecol. Indic. 2021, 121, 107027. [Google Scholar] [CrossRef]
- Andringa, J.I.; Zuo, J.; Berg, M.P.; Klein, R.; Veer, J.V.; de Geus, R.; de Beaumont, M.; Goudzwaard, L.; van Hal, J.; Broekman, R.; et al. Combining tree species and decay stages to increase invertebrate diversity in dead wood. For. Ecol. Manag. 2019, 441, 80–88. [Google Scholar] [CrossRef]
- Dittrich, S.; Jacob, M.; Bade, C.; Leuschner, C.; Hauck, M. The significance of deadwood for total bryophyte, lichen, and vascular plant diversity in an old-growth spruce forest. Plant Ecol. 2014, 215, 1123–1137. [Google Scholar] [CrossRef]
- Fries, C.; Johansson, O.; Pettersson, B.; Simonsson, P. Silvicultural models to maintain and restore natural stand structures in Swedish boreal forests. For. Ecol. Manag. 1997, 94, 89–103. [Google Scholar] [CrossRef]
| Ground Flora | Vascular | Woody | Bryophyte | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Mean | SE | Mean | SE | Mean | SE | Mean | SE | ||
| Number of species | PHH | 21.13 | 2.35 | 11.63 | 1.63 | 3.07 | 0.47 | 6.42 | 0.66 |
| OG | 19.08 | 2.68 | 10.67 | 1.78 | 2.71 | 0.67 | 5.71 | 0.96 | |
| Relative projective cover (%) | PHH | 112.03 | 4.34 | 43.48 | 5.53 | 3.77 | 1.19 | 64.78 | 6.28 |
| OG | 103.19 | 6.06 | 42.57 | 7.08 | 2.61 | 1.19 | 58.01 | 9.90 | |
| Shannon-Wiener index | PHH | 2.45 | 0.14 | 1.89 | 0.17 | 1.25 | 0.11 | 0.48 | 0.13 |
| OG | 2.37 | 0.16 | 1.83 | 0.19 | 1.1 | 0.16 | 0.44 | 0.17 | |
| Pre-Harvesting/Harvesting Age Stands | Old-Growth Stands | ||||
|---|---|---|---|---|---|
| Species | Cover | Occurrence | Species | Cover | Occurrence |
| Vaccinium myrtillus | 18.27 | 89.36 | Vaccinium myrtillus | 14.30 | 79.82 |
| Oxalis acetosella | 9.55 | 78.19 | Oxalis acetosella | 12.63 | 80.73 |
| Calamagrostis arundinacea | 9.54 | 76.60 | Calamagrostis arundinacea | 4.38 | 54.13 |
| Maianthemum bifolium | 2.42 | 79.26 | Maianthemum bifolium | 3.36 | 81.65 |
| Pteridium aquilinum | 2.35 | 28.19 | Luzula pilosa | 2.77 | 77.98 |
| Luzula pilosa | 2.03 | 70.21 | Vaccinium vitis-idaea | 2.58 | 50.46 |
| Vaccinium vitis-idaea | 2.02 | 41.49 | Pteridium aquilinum | 2.07 | 20.18 |
| Carex digitata | 1.79 | 46.81 | Athyrium filix-femina | 1.83 | 21.10 |
| Melampyrum pratense | 1.75 | 37.23 | Trientalis europaea | 1.66 | 57.80 |
| Trientalis europaea | 1.68 | 67.02 | Melampyrum pratense | 1.61 | 31.19 |
| Rubus saxatilis | 1.59 | 32.45 | Rubus saxatilis | 1.70 | 26.61 |
| Dryopteris carthusiana | 1.45 | 43.09 | Dryopteris carthusiana | 1.57 | 37.61 |
| Festuca ovina | 1.25 | 13.30 | Carex digitata | 1.56 | 42.20 |
| DCA1 | ||
| Fixed effects | ||
| χ2 | p-value | |
| Light | 357.7 | <0.001 |
| Reaction | 120.0 | <0.001 |
| Moisture | 15.6 | <0.001 |
| Canopy coniferous, % | 74.6 | <0.001 |
| Understory stock | 5.0 | 0.02 |
| Density of Betula in canopy | 13.0 | <0.001 |
| Model performance | ||
| R2, marginal | 0.77 | |
| R2, conditional | 0.88 | |
| DCA2 | ||
| Fixed effects | ||
| χ2 | p-value | |
| Nitrogen | 36.4 | <0.001 |
| Total standing stock | 6.8 | 0.009 |
| Model performance | ||
| R2, marginal | 0.13 | |
| R2, conditional | 0.70 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Matisone, I.; Jansone, D.; Jaunslaviete, I.; Matisons, R.; Liepiņa, A.A.; Jansons, Ā. Stand Structure Beats Age for Ground Cover Vegetation in Ageing Hemiboreal Scots Pine and Norway Spruce Stands. Sustainability 2023, 15, 7594. https://doi.org/10.3390/su15097594
Matisone I, Jansone D, Jaunslaviete I, Matisons R, Liepiņa AA, Jansons Ā. Stand Structure Beats Age for Ground Cover Vegetation in Ageing Hemiboreal Scots Pine and Norway Spruce Stands. Sustainability. 2023; 15(9):7594. https://doi.org/10.3390/su15097594
Chicago/Turabian StyleMatisone, Ilze, Diāna Jansone, Ieva Jaunslaviete, Roberts Matisons, Agnese Anta Liepiņa, and Āris Jansons. 2023. "Stand Structure Beats Age for Ground Cover Vegetation in Ageing Hemiboreal Scots Pine and Norway Spruce Stands" Sustainability 15, no. 9: 7594. https://doi.org/10.3390/su15097594
APA StyleMatisone, I., Jansone, D., Jaunslaviete, I., Matisons, R., Liepiņa, A. A., & Jansons, Ā. (2023). Stand Structure Beats Age for Ground Cover Vegetation in Ageing Hemiboreal Scots Pine and Norway Spruce Stands. Sustainability, 15(9), 7594. https://doi.org/10.3390/su15097594







