Cultivating the Mediterranean Wild Edible Species Cichorium spinosum L. in Aquaponics: Functional and Growth Responses to Minimal Nutrient Supplementation
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Design, Growth Conditions and Aquaponic Systems
2.2. Monitoring of the Water Physicochemical Parameters
2.3. Tilapia Rearing Conditions and Growth Performance
- Survival rate, S (%) = (Final number of fish/Initial number of fish) × 100
- Weight gain, WG (g) = Final weight − Initial weight
- Specific growth rate, SGR (% day−1) = ((ln Final weight − ln Initial weight)/∆t) × 100
- Feed conversion ratio, FCR = Food offered (g)/weight gain (g)
2.4. Stamnagathi Cultivation
2.5. Plant Growth, Physiology and Biochemical Analysis
2.5.1. Growth Performance
2.5.2. Total Chlorophyll Content
2.5.3. Photochemical Reflectance Index (PRI)
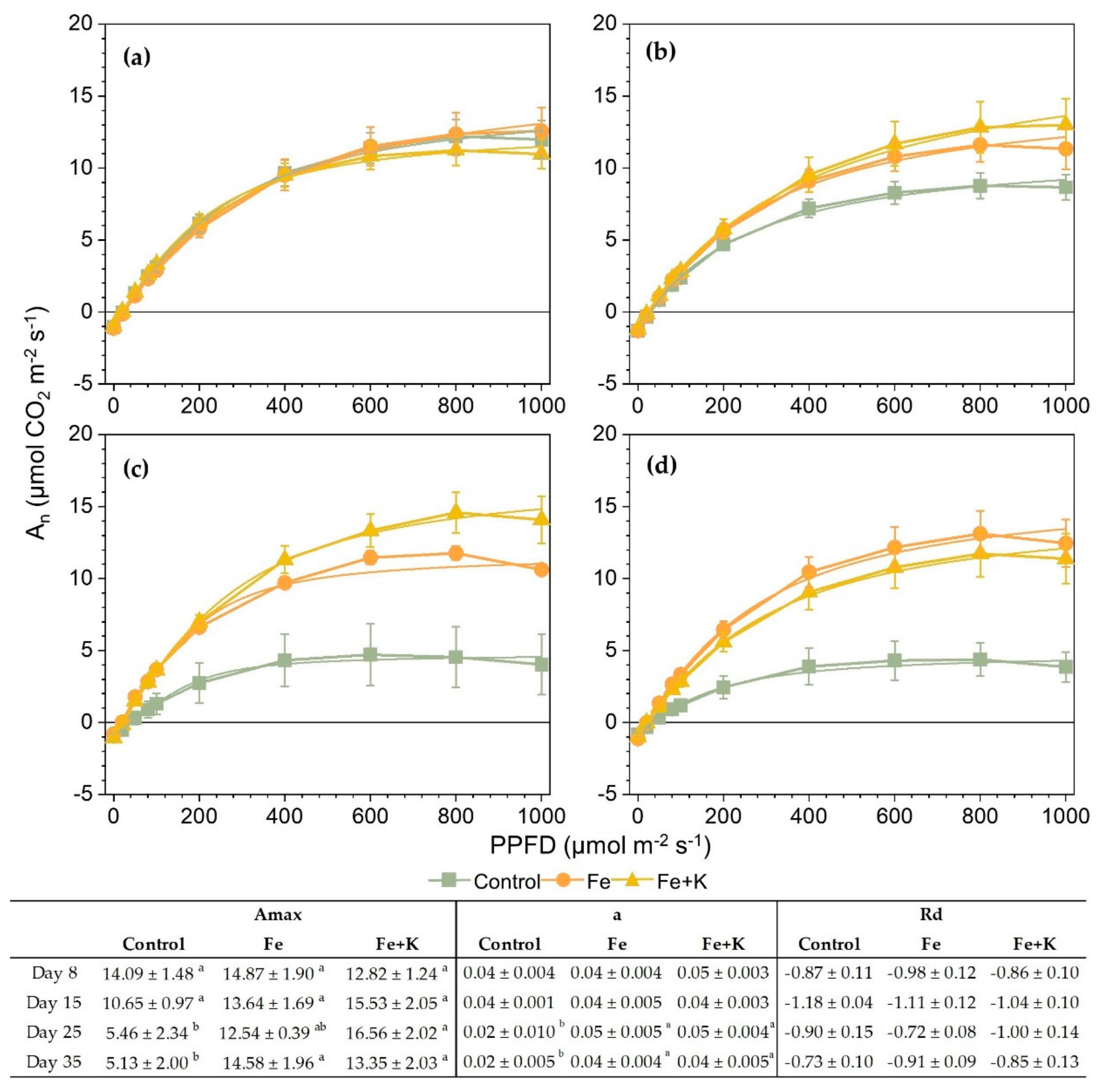
2.5.4. Photosynthesis and Light Response Curves
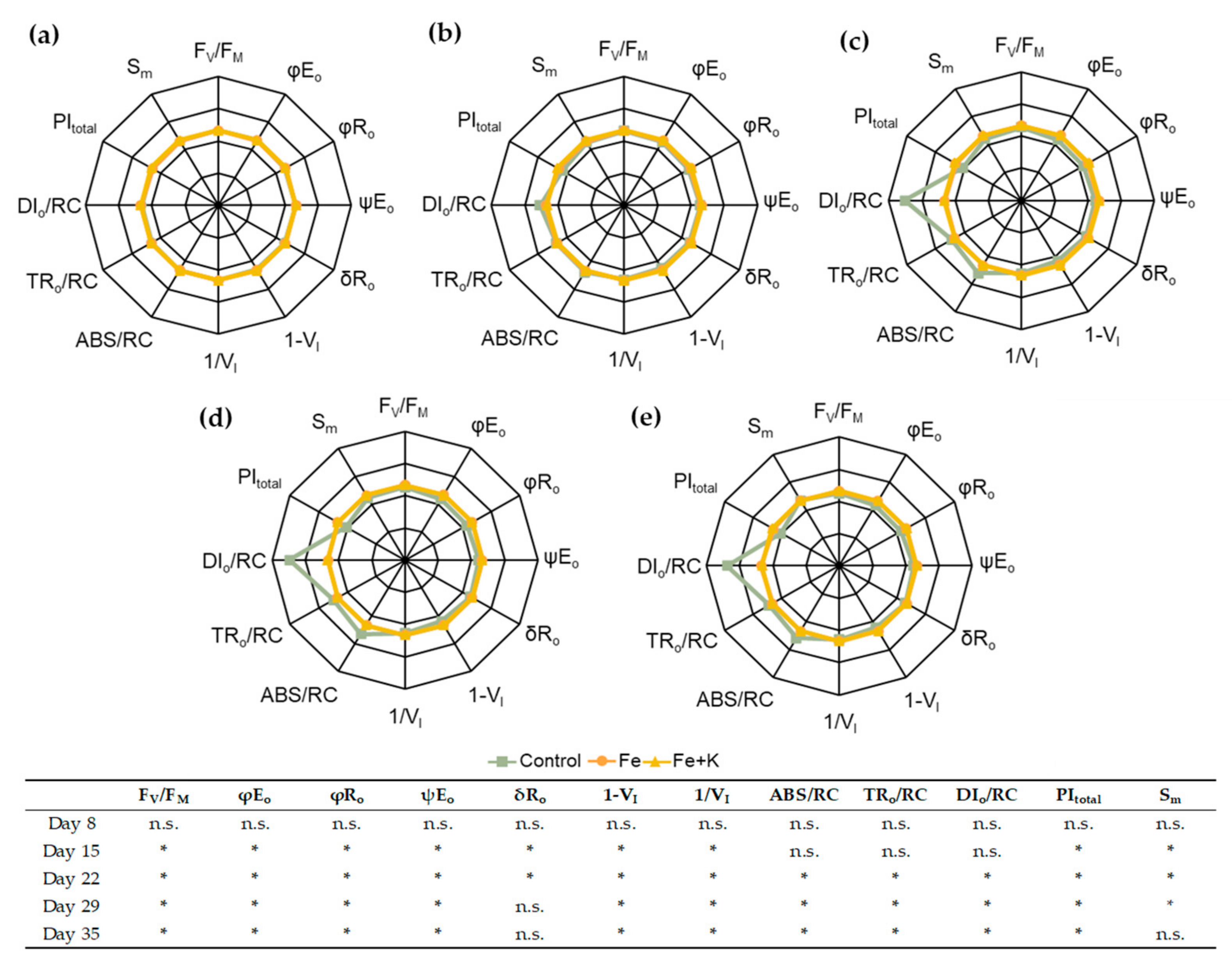
2.5.5. In Vivo Fluorescence of Chlorophyll a
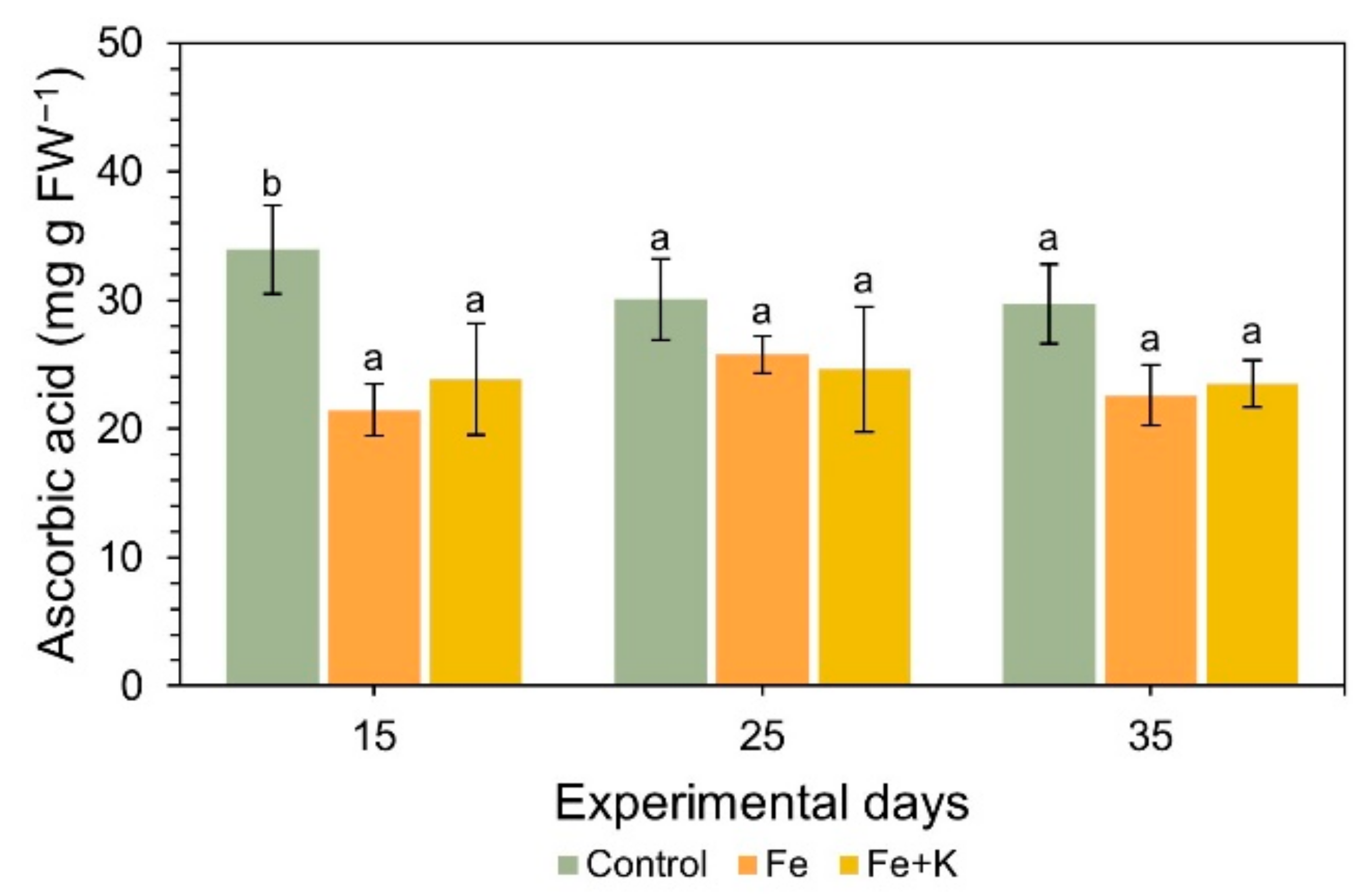
2.5.6. Antioxidant Activity
2.5.7. Elemental Tissue Analysis
2.6. Statistical Analysis
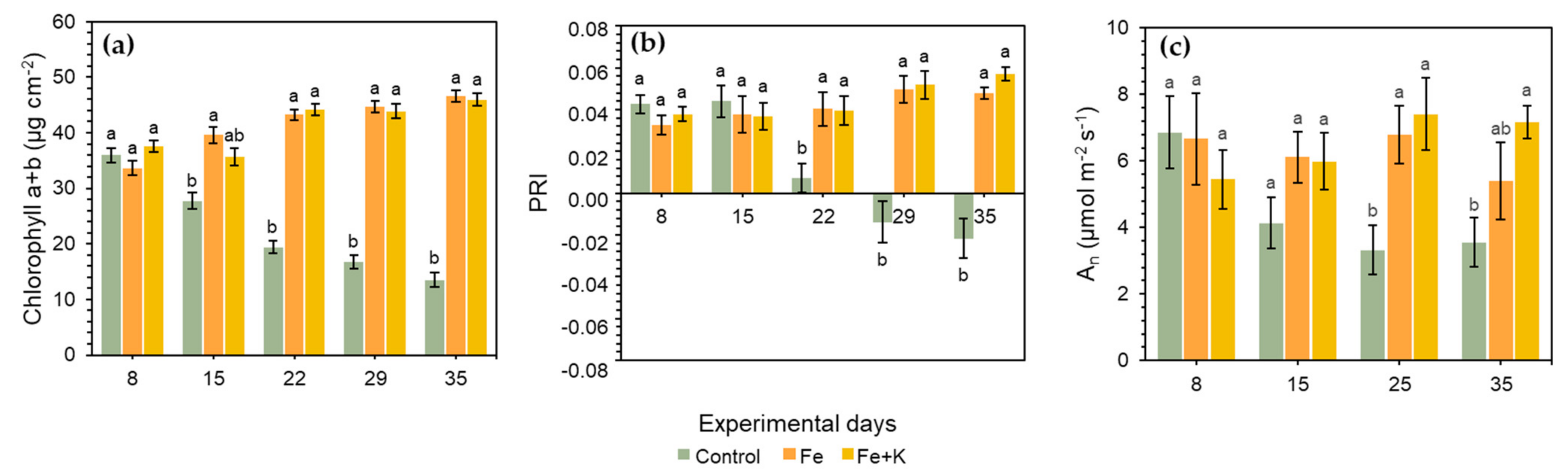
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Aquaponics Food Production Systems: Combined Aquaculture and Hydroponic Production Technologies for the Future; Goddek, S., Joyce, A., Kotzen, B., Burnell, G.M., Eds.; Springer International Publishing: Cham, Switzerland, 2019; ISBN 978-3-030-15942-9. [Google Scholar]
- Junge, R.; König, B.; Villarroel, M.; Komives, T.; Jijakli, M. Strategic Points in Aquaponics. Water 2017, 9, 182. [Google Scholar] [CrossRef]
- Baganz, G.F.M.; Junge, R.; Portella, M.C.; Goddek, S.; Keesman, K.J.; Baganz, D.; Staaks, G.; Shaw, C.; Lohrberg, F.; Kloas, W. The Aquaponic Principle—It Is All about Coupling. Rev. Aquacult. 2022, 14, 252–264. [Google Scholar] [CrossRef]
- Maucieri, C.; Nicoletto, C.; Schmautz, Z.; Sambo, P.; Komives, T.; Borin, M.; Junge, R. Vegetable Intercropping in a Small-Scale Aquaponic System. Agronomy 2017, 7, 63. [Google Scholar] [CrossRef]
- Palm, H.W.; Knaus, U.; Appelbaum, S.; Goddek, S.; Strauch, S.M.; Vermeulen, T.; Haïssam Jijakli, M.; Kotzen, B. Towards Commercial Aquaponics: A Review of Systems, Designs, Scales and Nomenclature. Aquacult. Int. 2018, 26, 813–842. [Google Scholar] [CrossRef]
- Van der Esch, S.; Brink, B.; Stehfest, E.; Bakkenes, M.; Sewell, A.; Bouwman, A.; Meijer, J.; Westhoek, H.; van den Berg, M.; Jan van den Born, G.; et al. Exploring Future Changes in Land Use and Land Condition and the Impacts on Food, Water, Climate Change and Biodiversity: Scenarios for the UNCCD Global Land Outlook; PBL; Netherlands Environmental Assessment Agency: The Hague, The Netherlands, 2017. [Google Scholar]
- Joyce, A.; Goddek, S.; Kotzen, B.; Wuertz, S. Aquaponics: Closing the Cycle on Limited Water, Land and Nutrient Resources. In Aquaponics Food Production Systems: Combined Aquaculture and Hydroponic Production Technologies for the Future; Goddek, S., Joyce, A., Kotzen, B., Burnell, G.M., Eds.; Springer International Publishing: Cham, Switzerland, 2019; pp. 19–34. ISBN 978-3-030-15943-6. [Google Scholar]
- Roosta, H.R.; Hamidpour, M. Effects of Foliar Application of Some Macro- and Micro-Nutrients on Tomato Plants in Aquaponic and Hydroponic Systems. Sci. Hortic. 2011, 129, 396–402. [Google Scholar] [CrossRef]
- Tsoumalakou, E.; Mente, E.; Kormas, K.A.; Katsoulas, N.; Vlahos, N.; Kapsis, P.; Levizou, E. Precise Monitoring of Lettuce Functional Responses to Minimal Nutrient Supplementation Identifies Aquaponic System’s Nutrient Limitations and Their Time-Course. Agriculture 2022, 12, 1278. [Google Scholar] [CrossRef]
- Bittsánszky, A.; Uzinger, N.; Gyulai, G.; Mathis, A.; Junge, R.; Villarroel, M.; Kotzen, B.; Kőmíves, T. Nutrient Supply of Plants in Aquaponic Systems. Ecocycles 2016, 2, 17–20. [Google Scholar] [CrossRef]
- Ru, D.; Liu, J.; Hu, Z.; Zou, Y.; Jiang, L.; Cheng, X.; Lv, Z. Improvement of Aquaponic Performance through Micro- and Macro-Nutrient Addition. Environ. Sci. Pollut. Res. 2017, 24, 16328–16335. [Google Scholar] [CrossRef]
- Tsoumalakou, E.; Mente, E.; Vlahos, N.; Levizou, E. Spinach Responds to Minimal Nutrient Supplementation in Aquaponics by Up-Regulating Light Use Efficiency, Photochemistry, and Carboxylation. Horticulturae 2023, 9, 291. [Google Scholar] [CrossRef]
- Krigas, N.; Tsoktouridis, G.; Anestis, I.; Khabbach, A.; Libiad, M.; Megdiche-Ksouri, W.; Ghrabi-Gammar, Z.; Lamchouri, F.; Tsiripidis, I.; Tsiafouli, M.A.; et al. Exploring the Potential of Neglected Local Endemic Plants of Three Mediterranean Regions in the Ornamental Sector: Value Chain Feasibility and Readiness Timescale for Their Sustainable Exploitation. Sustainability 2021, 13, 2539. [Google Scholar] [CrossRef]
- Fanourakis, D.; Paschalidis, K.; Tsaniklidis, G.; Tzanakakis, V.A.; Bilias, F.; Samara, E.; Liapaki, E.; Jouini, M.; Ipsilantis, I.; Maloupa, E.; et al. Pilot Cultivation of the Local Endemic Cretan Marjoram Origanum microphyllum (Benth.) Vogel (Lamiaceae): Effect of Fertilizers on Growth and Herbal Quality Features. Agronomy 2021, 12, 94. [Google Scholar] [CrossRef]
- Kopittke, P.M.; Menzies, N.W.; Wang, P.; McKenna, B.A.; Lombi, E. Soil and the Intensification of Agriculture for Global Food Security. Environ. Int. 2019, 132, 105078. [Google Scholar] [CrossRef]
- Psaroudaki, A.; Dimitropoulakis, P.; Constantinidis, T.; Katsiotis, A.; Skaracis, G.N. Ten Indigenous Edible Plants: Contemporary Use in Eastern Crete, Greece. CAFÉ 2012, 34, 172–177. [Google Scholar] [CrossRef]
- Petropoulos, S.A.; Fernandes, Â.; Ntatsi, G.; Levizou, E.; Barros, L.; Ferreira, I.C.F.R. Nutritional Profile and Chemical Composition of Cichorium Spinosum Ecotypes. LWT 2016, 73, 95–101. [Google Scholar] [CrossRef]
- Petropoulos, S.; Fernandes, Â.; Karkanis, A.; Antoniadis, V.; Barros, L.; Ferreira, I.C.F.R. Nutrient Solution Composition and Growing Season Affect Yield and Chemical Composition of Cichorium Spinosum Plants. Sci. Hortic. 2018, 231, 97–107. [Google Scholar] [CrossRef]
- Chatzigianni, M.; Ntatsi, G.; Theodorou, M.; Stamatakis, A.; Livieratos, I.; Rouphael, Y.; Savvas, D. Functional Quality, Mineral Composition and Biomass Production in Hydroponic Spiny Chicory (Cichorium spinosum L.) Are Modulated Interactively by Ecotype, Salinity and Nitrogen Supply. Front. Plant Sci. 2019, 10, 1040. [Google Scholar] [CrossRef] [PubMed]
- Hirayama, K. Water Control by Filtration in Closed Culture Systems. Aquaculture 1974, 4, 369–385. [Google Scholar] [CrossRef]
- Da Silva, M.A.; de Alvarenga, É.R.; da Costa, F.F.B.; Turra, E.M.; de Oliveira Alves, G.F.; Manduca, L.G.; de Sales, S.C.M.; Leite, N.R.; Bezerra, V.M.; da Silva Moraes, S.G.; et al. Feeding Management Strategies to Optimize the Use of Suspended Feed for Nile Tilapia (Oreochromis niloticus) Cultivated in Bioflocs. Aquac. Res. 2020, 51, 605–615. [Google Scholar] [CrossRef]
- Lichtenthaler, H.K.; Wellburn, A.R. Determinations of Total Carotenoids and Chlorophylls a and b of Leaf Extracts in Different Solvents. Biochem. Soc. Trans. 1983, 11, 591–592. [Google Scholar] [CrossRef]
- Vanikiotis, T.; Stagakis, S.; Kyparissis, A. MODIS PRI Performance to Track Light Use Efficiency of a Mediterranean Coniferous Forest: Determinants, Restrictions and the Role of LUE Range. Agric. For. Meteorol. 2021, 307, 108518. [Google Scholar] [CrossRef]
- Markos, N.; Kyparissis, A. Ecophysiological Modelling of Leaf Level Photosynthetic Performance for Three Mediterranean Species with Different Growth Forms. Functional. Plant Biol. 2011, 38, 314. [Google Scholar] [CrossRef]
- Goupy, P.; Hugues, M.; Boivin, P.; Amiot, M.J. Antioxidant Composition and Activity of Barley (Hordeum Vulgare) and Malt Extracts and of Isolated Phenolic Compounds. J. Sci. Food Agric. 1999, 79, 1625–1634. [Google Scholar] [CrossRef]
- Hayes, J.E.; Allen, P.; Brunton, N.; O’Grady, M.N.; Kerry, J.P. Phenolic Composition and in Vitro Antioxidant Capacity of Four Commercial Phytochemical Products: Olive Leaf Extract (Olea europaea L.), Lutein, Sesamol and Ellagic Acid. Food Chem. 2011, 126, 948–955. [Google Scholar] [CrossRef]
- Avdouli, D.; Max, J.F.J.; Katsoulas, N.; Levizou, E. Basil as Secondary Crop in Cascade Hydroponics: Exploring Salinity Tolerance Limits in Terms of Growth, Amino Acid Profile, and Nutrient Composition. Horticulturae 2021, 7, 203. [Google Scholar] [CrossRef]
- Kasozi, N.; Abraham, B.; Kaiser, H.; Wilhelmi, B. The Complex Microbiome in Aquaponics: Significance of the Bacterial Ecosystem. Ann. Microbiol. 2021, 71, 1. [Google Scholar] [CrossRef]
- Petropoulos, S.A.; Levizou, E.; Ntatsi, G.; Fernandes, Â.; Petrotos, K.; Akoumianakis, K.; Barros, L.; Ferreira, I.C.F.R. Salinity Effect on Nutritional Value, Chemical Composition and Bioactive Compounds Content of Cichorium spinosum L. Food Chem. 2017, 214, 129–136. [Google Scholar] [CrossRef] [PubMed]
- Vlahos, N.; Levizou, E.; Stathopoulou, P.; Berillis, P.; Antonopoulou, E.; Bekiari, V.; Krigas, N.; Kormas, K.; Mente, E. An Experimental Brackish Aquaponic System Using Juvenile Gilthead Sea Bream (Sparus aurata) and Rock Samphire (Crithmum maritimum). Sustainability 2019, 11, 4820. [Google Scholar] [CrossRef]
- Paschalidis, K.; Fanourakis, D.; Tsaniklidis, G.; Tzanakakis, V.A.; Bilias, F.; Samara, E.; Kalogiannakis, K.; Debouba, F.J.; Ipsilantis, I.; Tsoktouridis, G.; et al. Pilot Cultivation of the Vulnerable Cretan Endemic Verbascum arcturus L. (Scrophulariaceae): Effect of Fertilization on Growth and Quality Features. Sustainability 2021, 13, 14030. [Google Scholar] [CrossRef]
- Sonneveld, C.; Voogt, W. Plant Nutrition of Greenhouse Crops; Springer: Dordrecht, The Netherlands; New York, NY, USA, 2009; ISBN 978-90-481-2531-9. [Google Scholar]
- Murchie, E.H.; Lawson, T. Chlorophyll Fluorescence Analysis: A Guide to Good Practice and Understanding Some New Applications. J. Exp. Bot. 2013, 64, 3983–3998. [Google Scholar] [CrossRef]
- Maxwell, K.; Johnson, G.N. Chlorophyll Fluorescence—A Practical Guide. J. Exp. Bot. 2000, 51, 659–668. [Google Scholar] [CrossRef] [PubMed]
- Kalaji, H.M.; Bąba, W.; Gediga, K.; Goltsev, V.; Samborska, I.A.; Cetner, M.D.; Dimitrova, S.; Piszcz, U.; Bielecki, K.; Karmowska, K.; et al. Chlorophyll Fluorescence as a Tool for Nutrient Status Identification in Rapeseed Plants. Photosynth. Res. 2018, 136, 329–343. [Google Scholar] [CrossRef]
- Roosta, H.R. Comparison of the Vegetative Growth, Eco-Physiological Characteristics and Mineral Nutrient Content of Basil Plants in Different Irrigation Ratios of Hydroponic:Aquaponic Solutions. J. Plant Nutr. 2014, 37, 1782–1803. [Google Scholar] [CrossRef]
- Roosta, H.R.; Estaji, A.; Niknam, F. Effect of Iron, Zinc and Manganese Shortage-Induced Change on Photosynthetic Pigments, Some Osmoregulators and Chlorophyll Fluorescence Parameters in Lettuce. Photosynthetica 2018, 56, 606–615. [Google Scholar] [CrossRef]
- Samborska-Skutnik, I.A.; Kalaji, H.M.; Sieczko, L.; Bąba, W. Special Issue in Honour of Prof. Reto J. Strasser—Structural and Functional Response of Photosynthetic Apparatus of Radish Plants to Iron Deficiency. Photosynthetica 2020, 58, 205–213. [Google Scholar] [CrossRef]
- Kroh, G.E.; Pilon, M. Regulation of Iron Homeostasis and Use in Chloroplasts. Int. J. Mol. Sci. 2020, 21, 3395. [Google Scholar] [CrossRef]
- Larbi, A.; Abadía, A.; Abadía, J.; Morales, F. Down Co-Regulation of Light Absorption, Photochemistry, and Carboxylation in Fe-Deficient Plants Growing in Different Environments. Photosynth. Res. 2006, 89, 113–126. [Google Scholar] [CrossRef] [PubMed]
- Stathopoulou, P.; Tsoumalakou, E.; Levizou, E.; Vanikiotis, T.; Zaoutsos, S.; Berillis, P. Iron and Potassium Fertilization Improve Rocket Growth without Affecting Tilapia Growth and Histomorphology Characteristics in Aquaponics. Appl. Sci. 2021, 11, 5681. [Google Scholar] [CrossRef]
- Jin, C.-W.; Liu, Y.; Mao, Q.-Q.; Wang, Q.; Du, S.-T. Mild Fe-Deficiency Improves Biomass Production and Quality of Hydroponic-Cultivated Spinach Plants (Spinacia oleracea L.). Food Chem. 2013, 138, 2188–2194. [Google Scholar] [CrossRef]
- Patel, M.; Fatnani, D.; Parida, A.K. Potassium Deficiency Stress Tolerance in Peanut (Arachis hypogaea) through Ion Homeostasis, Activation of Antioxidant Defense, and Metabolic Dynamics: Alleviatory Role of Silicon Supplementation. Plant Physiol. Biochem. 2022, 182, 55–75. [Google Scholar] [CrossRef] [PubMed]
| Control | Fe | Fe+K | |
|---|---|---|---|
| pH | 6.99 ± 0.05 b | 7.16 ± 0.04 a | 7.18 ± 0.04 a |
| O2 (mg L−1) | 8.35 ± 0.05 a | 8.32 ± 0.05 a | 8.33 ± 0.05 a |
| EC (μS cm−1) | 694.67 ± 4.02 b | 669.50 ± 3.26 c | 1319.94 ± 39.32 a |
| NH3 (mg L−1) | 0.13 ± 0.04 a | 0.10 ± 0.03 a | 0.13 ± 0.04 a |
| NO2− (mg L−1) | 0.12 ± 0.02 a | 0.11 ± 0.02 a | 0.16 ± 0.03 a |
| NO3− (mg L−1) | 99.43 ± 6.70 a | 77.69 ± 4.14 b | 77.02 ± 3.22 b |
| PO43− (mg L−1) | 32.36 ± 1.88 a | 27.98 ± 2.41 a | 34.88 ± 2.20 a |
| SO42− (mg L−1) | 22.83 ± 0.33 b | 20.72 ± 0.39 c | 226.39 ± 25.41 a |
| Fe (mg L−1) | 0.05 ± 0.01 b | 1.22 ± 0.16 a | 1.22 ± 0.16 a |
| K (mg L−1) | 5.32 ± 0.52 b | 3.62 ± 0.69 b | 224.75 ± 25.78 a |
| Ca2+ (mg L−1) | 29.60 ± 2.46 a | 28.63 ± 2.33 a | 34.45 ± 1.83 a |
| Na (mg L−1) | 47.05 ± 1.08 b | 47.42 ± 0.93 b | 52.18 ± 0.89 a |
| Control | Fe | Fe+K | |
|---|---|---|---|
| Initial weight (g) | 5.64 ± 0.28 | 5.77 ± 0.28 | 5.68 ± 0.28 |
| Final weight (g) | 27.55 ± 0.99 | 27.36 ± 1.21 | 27.68 ± 1.15 |
| Initial length (cm) | 6.98 ± 0.12 | 7.02 ± 0.11 | 6.98 ± 0.13 |
| Final length (cm) | 11.30 ± 0.13 | 11.26 ± 0.16 | 11.28 ± 0.15 |
| SGR (% day−1) | 4.58 ± 0.06 | 4.49 ± 0.05 | 4.54 ± 0.07 |
| Daily feed consumption (g day−1) | 5.36 ± 0.26 | 5.26 ± 1.16 | 5.55 ± 1.21 |
| FCR | 0.78 ± 0.02 | 0.81 ± 0.03 | 0.79 ± 0.03 |
| Survival (%) | 100 | 93.33 | 96.67 |
| Control | Fe | Fe+K | |
|---|---|---|---|
| Leaves fresh weight (g) | 13.83 ± 3.30 b | 59.42 ± 6.56 a | 50.61 ± 5.77 a |
| Leaves dry weight (g) | 1.33 ± 0.23 b | 4.89 ± 0.50 a | 4.25 ± 0.40 a |
| Root dry weight (g) | 1.82 ± 0.45 b | 2.77 ± 0.33 a | 2.77 ± 1.01 a |
| Root-to-shoot ratio | 1.41 ± 0.20 a | 0.59 ± 0.05 b | 0.68 ± 0.06 b |
| Yield (g m−2) | 376.90 ± 50.01 b | 1683.79 ± 168.28 a | 1515.22 ± 202.35 a |
| Number of leaves | 32.53 ± 7.25 b | 94.93 ± 12.83 a | 89.33 ± 12.05 a |
| Leaf area (cm2) | 127.60 ± 30.96 b | 528.74 ± 62.20 a | 491.07 ± 58.81 a |
| Diameter of rosette (cm) | 20.12 ± 2.70 b | 31.27 ± 1.69 a | 32.04 ± 1.52 a |
| Control | Fe | Fe+K | ||
|---|---|---|---|---|
| % | N | 2.62 ± 0.10 b | 3.09 ± 0.16 ab | 3.22 ± 0.08 a |
| P | 0.43 ± 0.02 a | 0.76 ± 0.07 a | 0.76 ± 0.02 a | |
| K | 3.39 ± 0.07 b | 3.51 ± 0.12 b | 5.73 ± 0.22 a | |
| Ca | 2.43 ± 0.20 a | 1.67 ± 0.04 ab | 1.36 ± 0.01 b | |
| Mg | 0.60 ± 0.03 a | 0.42 ± 0.03 b | 0.29 ± 0.01 c | |
| ppm | Fe | 40.11 ± 6.83 b | 70.34 ± 6.97 a | 78.78 ± 1.77 a |
| Zn | 102.53 ± 13.74 a | 26.43 ± 0.66 b | 26.34 ± 0.76 b | |
| Mn | 63.46 ± 18.89 a | 39.85 ± 11.37 a | 41.38 ± 4.88 a | |
| Cu | 6.73 ± 0.21 a | 6.16 ± 0.61 a | 6.04 ± 0.46 a | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tsoumalakou, E.; Mente, E.; Vlahos, N.; Levizou, E. Cultivating the Mediterranean Wild Edible Species Cichorium spinosum L. in Aquaponics: Functional and Growth Responses to Minimal Nutrient Supplementation. Sustainability 2023, 15, 5572. https://doi.org/10.3390/su15065572
Tsoumalakou E, Mente E, Vlahos N, Levizou E. Cultivating the Mediterranean Wild Edible Species Cichorium spinosum L. in Aquaponics: Functional and Growth Responses to Minimal Nutrient Supplementation. Sustainability. 2023; 15(6):5572. https://doi.org/10.3390/su15065572
Chicago/Turabian StyleTsoumalakou, Evangelia, Eleni Mente, Nikolaos Vlahos, and Efi Levizou. 2023. "Cultivating the Mediterranean Wild Edible Species Cichorium spinosum L. in Aquaponics: Functional and Growth Responses to Minimal Nutrient Supplementation" Sustainability 15, no. 6: 5572. https://doi.org/10.3390/su15065572
APA StyleTsoumalakou, E., Mente, E., Vlahos, N., & Levizou, E. (2023). Cultivating the Mediterranean Wild Edible Species Cichorium spinosum L. in Aquaponics: Functional and Growth Responses to Minimal Nutrient Supplementation. Sustainability, 15(6), 5572. https://doi.org/10.3390/su15065572







