Plant Breeding to Mitigate Climate Change—Present Status and Opportunities with an Assessment of Winter Wheat Cultivation in Northern Europe as an Example
Abstract
1. Introduction—Target and Aims of the Review
2. Plant Breeding—History and Traditional Aims
2.1. The Historical Start of Plant Breeding and Its Current Status
2.2. Traits of Focus in Traditional Plant Breeding
3. Climate Change and Plant Breeding
3.1. Impact of Climate Change on Agricultural Crops and Implications for Plant Breeding
3.2. Opportunities of Using Plant Breeding of Agricultural Crops as a Tool to Mitigate Climate Change
3.3. Opportunities to Use Plant Breeding as a Tool for Carbon Sequestration by Agricultural Crops
3.4. Opportunities to Breed for Nutrient Use Efficiency in Agricultural Crops
4. Assessment of Estimated Climate Mitigation Effects from Plant Breeding
4.1. Baselines for the Assessment
4.2. Impact on Climate Change Mitigation from Plant Breeding Derived Measures
5. Long-Term Sustainability Effects of the Use of Plant Breeding Strategies in Agricultural Crops
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Bernardo, R. Essentials of Plant Breeding; Stemma Press: Woodbury, MN, USA, 2014. [Google Scholar]
- Cowling, W.A. Sustainable plant breeding. Plant Breed. 2013, 132, 1–9. [Google Scholar] [CrossRef]
- Acquuah, G. Principles of Plant Genetics and Breeding, 2nd ed.; John Wiley and Sons, Ltd.: Hoboken, NJ, USA, 2012. [Google Scholar]
- Heisey, P.W.; Srinivasan, C.S.; Thirtle, C.G. Public sector plant breeding in a privatizing world. In Agriculture Information: Resource Economics Division USDA-ERS; Vol. Bulletin No. 772; Economic Research Service, United States Department of Agriculture: Washington, DC, USA, 2001. [Google Scholar]
- Luby, J.J.; Shaw, D.V. Plant breeders’ perspectives on improving yield and quality traits in horticultural food crops. HortScience 2009, 44, 20–22. [Google Scholar]
- Shiferaw, B.; Smale, M.; Braun, H.-J.; Duveiller, E.; Reynolds, M.; Muricho, G. Crops that feed the world 10. Past successes and future challenges to the role played by wheat in global food security. Food Secur. 2013, 5, 291–317. [Google Scholar]
- Gaunt, R. The relationship between plant disease severity and yield. Annu. Rev. Phytopathol. 1995, 33, 119–144. [Google Scholar] [CrossRef]
- Johansson, E.; Prieto-Linde, M.L.; Jönsson, J.Ö. Effects of Wheat Cultivar and Nitrogen Application on Storage Protein Composition and Breadmaking Quality. Cereal Chem. 2001, 78, 19–25. [Google Scholar] [CrossRef]
- Holm, L.; Malik, A.H.; Johansson, E. Optimizing yield and quality in malting barley by the governance of field cultivation conditions. J. Cereal Sci. 2018, 82, 230–242. [Google Scholar]
- Lammerts van Bueren, E.T.; Struik, P.C.; van Eekeren, N.; Nuijten, E. Towards resilience through systems-based plant breeding. A review. Agron. Sustain. Dev. 2018, 38, 42. [Google Scholar]
- Purugganan, M.D.; Fuller, D.Q. The nature of selection during plant domestication. Nature 2009, 457, 843–848. [Google Scholar] [CrossRef]
- Breseghello, F.; Coelho, A.S.G. Traditional and Modern Plant Breeding Methods with Examples in Rice (Oryza sativa L.). J. Agric. Food Chem. 2013, 61, 8277–8286. [Google Scholar] [CrossRef]
- Kerridge, E. The Agricultural Revolution; Routledge, Taylor and Francis: London, UK, 2006. [Google Scholar]
- Overton, M. Agricultural Revolution in England: The Transformation of the Agrarian Economy 1500–1850; Cambridge University Press: Cambridge, UK, 1996. [Google Scholar]
- Vega, L. Fundamentals of Genetics; Scientific e-Resources; ED-Tech Press: London, UK, 2019. [Google Scholar]
- Labroo, M.R.; Studer, A.J.; Rutkoski, J.E. Heterosis and hybrid crop breeding: A multidisciplinary review. Front. Genet. 2021, 12, 643761. [Google Scholar]
- Lee, E.; Tracy, W. Modern Maize Breeding. In Handbook of Maize; Genetics and Genomics; Bennetzen, J., Hake, S., Eds.; Springer: New York, NY, USA, 2009. [Google Scholar]
- Varshney, R.K.; Dubey, A. Novel Genomic Tools and Modern Genetic and Breeding Approaches for Crop Improvement. J. Plant Biochem. Biotechnol. 2009, 18, 127–138. [Google Scholar]
- Lenaerts, B.; Collard, B.C.Y.; Demont, M. Review: Improving global food security through accelerated plant breeding. Plant Sci. 2019, 287, 110207. [Google Scholar] [PubMed]
- Zhang, H.; Li, Y.; Zhu, J.-K. Developing naturally stress-resistant crops for a sustainable agriculture. Nat. Plants 2018, 4, 989–996. [Google Scholar] [CrossRef] [PubMed]
- Rahmatov, M.; Otambekova, M.; Muminjanov, H.; Rouse, M.N.; Hovmøller, M.S.; Nazari, K.; Steffenson, B.J.; Johansson, E. Characterization of stem, stripe and leaf rust resistance in Tajik bread wheat accessions. Euphytica 2019, 215, 55. [Google Scholar] [CrossRef]
- Kaur, S.; Kaur, R.; Chauhan, B.S. Understanding crop-weed-fertilizer-water interactions and their implications for weed management in agricultural systems. Crop Protect. 2018, 103, 65–72. [Google Scholar] [CrossRef]
- Pimentel, D. Environmental and Economic Costs of the Application of Pesticides Primarily in the United States. Environ. Dev. Sustain. 2005, 7, 229–252. [Google Scholar] [CrossRef]
- Yazdani, M. Inducing Novel Resistance Gene in Wheat towards Stem Rust to Improve Food Security; Series 2022:1; Swedish University of Agricultural Sciences (SLU): Alnarp, Sweden, 2022. [Google Scholar]
- Ando, K.; Grumet, R.; Terpstra, K.; Kelly, J.D. Manipulation of plant architecture to enhance crop disease control. CABI Rev. 2007, 2007. [Google Scholar] [CrossRef]
- Shabbir, R.; Singhal, R.K.; Mishra, U.N.; Chauhan, J.; Javed, T.; Hussain, S.; Kumar, S.; Anuragi, H.; Lal, D.; Chen, P. Combined Abiotic Stresses: Challenges and Potential for Crop Improvement. Agronomy 2022, 12, 2795. [Google Scholar]
- Snowdon, R.J.; Wittkop, B.; Chen, T.-W.; Stahl, A. Crop adaptation to climate change as a consequence of long-term breeding. Theor. Appl. Genet. 2021, 134, 1613–1623. [Google Scholar] [CrossRef]
- Peng, J.; Richards, D.E.; Hartley, N.M.; Murphy, G.P.; Devos, K.M.; Flintham, J.E.; Beales, J.; Fish, L.J.; Worland, A.J.; Pelica, F.; et al. ‘Green revolution’ genes encode mutant gibberellin response modulators. Nature 1999, 400, 256–261. [Google Scholar] [CrossRef]
- Berry, P.M.; Kendall, S.; Rutterford, Z.; Orford, S.; Griffiths, S. Historical analysis of the effects of breeding on the height of winter wheat (Triticum aestivum) and consequences for lodging. Euphytica 2015, 203, 375–383. [Google Scholar] [CrossRef]
- Guzmán, C.; Autrique, E.; Mondal, S.; Huerta-Espino, J.; Singh, R.P.; Vargas, M.; Crossa, J.; Amaya, A.; Peña, R.J. Genetic improvement of grain quality traits for CIMMYT semi-dwarf spring bread wheat varieties developed during 1965–2015: 50 years of breeding. Field Crops Res. 2017, 210, 192–196. [Google Scholar]
- Fan, M.-S.; Zhao, F.-J.; Fairweather-Tait, S.J.; Poulton, P.R.; Dunham, S.J.; McGrath, S.P. Evidence of decreasing mineral density in wheat grain over the last 160 years. J. Trace Elem. Med. Biol. 2008, 22, 315–324. [Google Scholar] [CrossRef] [PubMed]
- Bektas, H.; Hohn, C.E.; Waines, J.G. Root and shoot traits of bread wheat (Triticum aestivum L.) landraces and cultivars. Euphytica 2016, 212, 297–311. [Google Scholar] [CrossRef]
- Bertholdsson, N.O.; Kolodinska Brantestam, A. A century of Nordic barley breeding—Effects on early vigour root and shoot growth, straw length, harvest index and grain weight. Eur. J. Agron. 2009, 30, 266–274. [Google Scholar]
- Lan, Y.; Chawade, A.; Kuktaite, R.; Johansson, E. Climate Change Impact on Wheat Performance-Effects on Vigour, Plant Traits and Yield from Early and Late Drought Stress in Diverse Lines. Int. J. Mol. Sci. 2022, 23, 3333. [Google Scholar]
- Fufa, H.; Baenziger, P.S.; Beecher, B.S.; Graybosch, R.A.; Eskridge, K.M.; Nelson, L.A. Genetic improvement trends in agronomic performances and end-use quality characteristics among hard red winter wheat cultivars in Nebraska. Euphytica 2005, 144, 187–198. [Google Scholar] [CrossRef]
- Austin, R.B.; Bingham, J.; Blackwell, R.D.; Evans, L.T.; Ford, M.A.; Morgan, C.L.; Taylor, M. Genetic improvements in winter wheat yields since 1900 and associated physiological changes. J. Agric. Sci. 1980, 94, 675–689. [Google Scholar]
- Chapman, S.C.; Chakraborty, S.; Dreccer, M.F.; Howden, S.M. Plant adaptation to climate change—Opportunities and priorities in breeding. Crop Pasture Sci. 2012, 63, 251–268. [Google Scholar] [CrossRef]
- Atkinson, M.D.; Kettlewell, P.S.; Poulton, P.R.; Hollins, P.D. Grain quality in the Broadbalk Wheat Experiment and the winter North Atlantic Oscillation. J. Agric. Sci. 2008, 146, 541–549. [Google Scholar] [CrossRef]
- Mendelsohn, R. The Impact of Climate Change on Agriculture in Asia. J. Integr. Agric. 2014, 13, 660–665. [Google Scholar] [CrossRef]
- Ceccarelli, S.; Grando, S.; Maatougui, M.; Michael, M.; Slash, M.; Haghparast, R.; Rahmanian, M.; Taheri, A.; Al-Yassin, A.; Benbelkacem, A. Climate Change and Agriculture Paper. Plant Breed. Clim. Chang. J. Agric. Sci. 2010, 148, 627–637. [Google Scholar]
- Mukamuhirwa, A.; Persson Hovmalm, H.; Ortiz, R.; Nyamangyoku, O.; Prieto–Linde, M.L.; Ekholm, A.; Johansson, E. Effect of intermittent drought on grain yield and quality of rice (Oryza sativa L.) grown in Rwanda. J. Agron. Crop Sci. 2020, 206, 252–262. [Google Scholar] [CrossRef]
- Cooper, M.; Messina, C.D.; Podlich, D.; Totir, L.R.; Baumgarten, A.; Hausmann, N.J.; Wright, D.; Graham, G. Predicting the future of plant breeding: Complementing empirical evaluation with genetic prediction. Crop Pasture Sci. 2014, 65, 311–336. [Google Scholar]
- Pilbeam, D.J. Breeding crops for improved mineral nutrition under climate change conditions. J. Exp. Bot. 2015, 66, 3511–3521. [Google Scholar]
- Döring, T.F.; Knapp, S.; Kovacs, G.; Murphy, K.; Wolfe, M.S. Evolutionary Plant Breeding in Cereals—Into a New Era. Sustainability 2011, 3, 1944–1971. [Google Scholar]
- Dwivedi, S.L.; Ceccarelli, S.; Blair, M.W.; Upadhyaya, H.D.; Are, A.K.; Ortiz, R. Landrace Germplasm for Improving Yield and Abiotic Stress Adaptation. Trends Plant Sci. 2016, 21, 31–42. [Google Scholar] [PubMed]
- Raffo, M.A.; Jensen, J. Gene × gene and genotype × environment interactions in wheat. Crop Sci. 2023, 63, 1779–1793. [Google Scholar]
- Crews, T.E.; Rumsey, B.E. What Agriculture Can Learn from Native Ecosystems in Building Soil Organic Matter: A Review. Sustainability 2017, 9, 578. [Google Scholar]
- Cormier, F.; Foulkes, J.; Hirel, B.; Gouache, D.; Moënne-Loccoz, Y.; Le Gouis, J. Breeding for increased nitrogen-use efficiency: A review for wheat (T. aestivum L.). Plant Breed. 2016, 135, 255–278. [Google Scholar]
- Boote, K.J.; Ibrahim, A.M.H.; Lafitte, R.; McCulley, R.; Messina, C.; Murray, S.C.; Specht, J.E.; Taylor, S.; Westgate, M.E.; Glasener, K.; et al. Position Statement on Crop Adaptation to Climate Change. Crop Sci. 2011, 51, 2337–2343. [Google Scholar]
- Atlin, G.N.; Cairns, J.E.; Das, B. Rapid breeding and varietal replacement are critical to adaptation of cropping systems in the developing world to climate change. Glob. Food Sec. 2017, 12, 31–37. [Google Scholar] [PubMed]
- Ferrante, A.; Nocito, F.F.; Morgutti, S.; Sacchi, G.A. Plant Breeding for Improving Nutrient Uptake and Utilization Efficiency. In Advances in Research on Fertilization Management of Vegetable Crops; Tei, F., Nicola, S., Benincasa, P., Eds.; Springer International Publishing: Cham, Switzerland, 2017; pp. 221–246. [Google Scholar]
- Kumar, R.; Pandey, S.; Pandey, A. Plant roots and carbon sequestration. Curr. Sci. 2006, 91, 885–890. [Google Scholar]
- Lal, R. Global Potential of Soil Carbon Sequestration to Mitigate the Greenhouse Effect. Crit. Rev. Plant Sci. 2003, 22, 151–184. [Google Scholar] [CrossRef]
- Kätterer, T.; Bolinder, M.A.; Andrén, O.; Kirchmann, H.; Menichetti, L. Roots contribute more to refractory soil organic matter than above-ground crop residues, as revealed by a long-term field experiment. Agric. Ecosyst. Environ. 2011, 141, 184–192. [Google Scholar] [CrossRef]
- Poeplau, C.; Kätterer, T.; Bolinder, M.A.; Börjesson, G.; Berti, A.; Lugato, E. Low stabilization of aboveground crop residue carbon in sandy soils of Swedish long-term experiments. Geoderma 2015, 237–238, 246–255. [Google Scholar] [CrossRef]
- Cormier, F.; Le Gouis, J.; Dubreuil, P.; Lafarge, S.; Praud, S. A genome-wide identification of chromosomal regions determining nitrogen use efficiency components in wheat (Triticum aestivum L.). Theor. Appl. Genet. 2014, 127, 2679–2693. [Google Scholar]
- Chawade, A.; Armoniené, R.; Berg, G.; Brazauskas, G.; Frostgård, G.; Geleta, M.; Gorash, A.; Henriksson, T.; Himanen, K.; Ingver, A.; et al. A transnational and holistic breeding approach is needed for sustainable wheat production in the Baltic Sea region. Physiol. Plant. 2018, 164, 442–451. [Google Scholar]
- Wissuwa, M.; Mazzola, M.; Picard, C. Novel approaches in plant breeding for rhizosphere-related traits. Plant Soil 2008, 321, 409. [Google Scholar]
- Hetrick, B.A.D.; Wilson, G.W.T.; Cox, T.S. Mycorrhizal dependence of modern wheat cultivars and ancestors: A synthesis. Can. J. Bot. 1993, 71, 512–518. [Google Scholar]
- Hetrick, B.A.D.; Wilson, G.W.T.; Cox, T.S. Mycorrhizal dependence of modern wheat varieties, landraces, and ancestors. Can. J. Bot. 1992, 70, 2032–2040. [Google Scholar] [CrossRef]
- Egle, K.; Manske, G.; Römer, W.; Vlek, P.L.G. Improved phosphorus efficiency of three new wheat genotypes from CIMMYT in comparison with an older Mexican variety. J. Plant Nutr. Soil Sci. 1999, 162, 353–358. [Google Scholar] [CrossRef]
- Fageria, N.K.; Baligar, V.C. Phosphorus-use efficiency in wheat genotypes. J. Plant Nutr. 1999, 22, 331–340. [Google Scholar] [CrossRef]
- Campos, P.; Borie, F.; Cornejo, P.; López-Ráez, J.A.; López-García, Á.; Seguel, A. Phosphorus Acquisition Efficiency Related to Root Traits: Is Mycorrhizal Symbiosis a Key Factor to Wheat and Barley Cropping? Front. Plant Sci. 2018, 9, 1–21. [Google Scholar]
- van de Wiel, C.C.M.; van der Linden, C.G.; Scholten, O.E. Improving phosphorus use efficiency in agriculture: Opportunities for breeding. Euphytica 2016, 207, 1–22. [Google Scholar]
- SS-EN ISO 14044; Environmental Management-Lifecycle Assessment-Requirements and Guidelines. International Standard Organization: Geneva, Switzerland, 2006.
- Lantz, M.; Prade, T.; Ahlgren, S.; Björnsson, L. Biogas and ethanol from wheat grain or straw: Is there a trade-off between climate impact, avoidance of iLUC and production cost? Energies 2018, 11, 2633. [Google Scholar]
- Hansson, P.; Saltzman, I.-L.; Bååth Jacobsson, S.; Petersson, P. Produktionsgrenskalkyler för Växtodling-Efterkalkyler för år 2014-Södra Sverige; Hushållningssällskapen Kalmar-Kronoberg-Blekinge, Krisitanstad, Malmöhus och Halland: Kristianstad, Sweden, 2014. [Google Scholar]
- Gissén, C.; Prade, T.; Kreuger, E.; Nges, I.A.; Rosenqvist, H.; Svensson, S.-E.; Lantz, M.; Mattsson, J.E.; Börjesson, P.; Björnsson, L. Comparing energy crops for biogas production–Yields, energy input and costs in cultivation using digestate and mineral fertilisation. Biomass Bioenergy 2014, 64, 199–210. [Google Scholar] [CrossRef]
- Makinkalkylgruppen. Maskinkostnader 2022; Swedish Rural Economy and Agricultural Societies Malmöhus: Bjärred, Sweden, 2022. [Google Scholar]
- Börjesson, P. Energianalyser av Biobränsleproduktion i Svenskt Jord-och Skogsbruk-Idag och Kring 2015; Department of Technology and Society, Lund University: Lund, Sweden, 1994. [Google Scholar]
- European Commission. Directive on the Promotion of the Use of Energy from Renewable Sources (2009/28/EC); European Parliament and Council of the European Union: Brussels, Belgium, 2009.
- Börjesson, P.; Tufvesson, L.; Lantz, M. Life Cycle Assessment of Biofuels in Sweden; 9188360962; Lund University: Lund, Sweden, 2010; p. 88. [Google Scholar]
- Green, M. Energy in pesticide manufacture, distribution and use. Energy World Agric. 1987, 2, 166–177. [Google Scholar]
- Audsley, E.; Stacey, K.; Parsons, D.J.; Williams, A.G. Estimation of the Greenhouse Gas Emissions from Agricultural Pesticide Manufacture and Use; Cranfield University: Bedford, UK, 2009. [Google Scholar]
- Nilsson, D.; Bernesson, S. Halm som Bränsle-Del 1: Tillgångar och Skördetidpunkter; Report/Department of Energy and Tecnology, SLU: Uppsala, Sweden, 2009. [Google Scholar]
- Hakala, K.; Heikkinen, J.; Sinkko, T.; Pahkala, K. Field trial results of straw yield with different harvesting methods, and modelled effects on soil organic carbon. A case study from Southern Finland. Biomass Bioenergy 2016, 95, 8–18. [Google Scholar]
- Ma, S.; He, F.; Tian, D.; Zou, D.; Yan, Z.; Yang, Y.; Zhou, T.; Huang, K.; Shen, H.; Fang, J. Variations and determinants of carbon content in plants: A global synthesis. Biogeosciences 2018, 15, 693–702. [Google Scholar]
- Bolinder, M.; Kätterer, T.; Andrén, O.; Parent, L. Estimating carbon inputs to soil in forage-based crop rotations and modeling the effects on soil carbon dynamics in a Swedish long-term field experiment. Can. J. Soil Sci. 2012, 92, 821–833. [Google Scholar] [CrossRef]
- IPCC. Chapter 11: N2O Emissions from Managed Soils and CO2 Emissions from Lime and Urea Application; IPCC: Geneva, Switzerland, 2006. [Google Scholar]
- Pribyl, D.W. A critical review of the conventional SOC to SOM conversion factor. Geoderma 2010, 156, 75–83. [Google Scholar] [CrossRef]
- IPCC. Climate Change 2013: The Physical Science Basis. Contribution of Working Group I to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change; Cambridge University Press: Cambridge, UK; New York, NY, USA, 2013. [Google Scholar]
- Jonasson, S.; Neuman, L. Alternativ till Spannmålstorkning Med Fossil Energi; LRF Media: Stockholm, Sweden, 2015. [Google Scholar]
- Gode, J.; Martinsson, F.; Hagberg, L.; Öman, A.; Höglund, J.; Palm, D. Miljöfaktaboken 2011-Uppskattade Emissionsfaktorer för Bränslen, el, Värme och Transporter; Värmeforsk: Stockholm, Sweden, 2011. [Google Scholar]
- Mortimer, N.; Elsayed, M.; Horne, R. Energy and Greenhouse Gas Emissions for Bioethanol Production from Wheat Grain and Sugar Beet; Resources Research Unit School of Environment and Development, Sheffield Hallam University: Sheffield, UK, 2004. [Google Scholar]
- Gaju, O.; Allard, V.; Martre, P.; Snape, J.W.; Heumez, E.; LeGouis, J.; Moreau, D.; Bogard, M.; Griffiths, S.; Orford, S.; et al. Identification of traits to improve the nitrogen-use efficiency of wheat genotypes. Field Crops Res. 2011, 123, 139–152. [Google Scholar] [CrossRef]
- Sylvester-Bradley, R.; Kindred, D.R. Analysing nitrogen responses of cereals to prioritize routes to the improvement of nitrogen use efficiency. J. Exp. Bot. 2009, 60, 1939–1951. [Google Scholar] [CrossRef]
- Ritchie, H.; Roser, M. Sweden: CO2 Country Profile. Available online: https://ourworldindata.org/co2-and-other-greenhouse-gas-emissions (accessed on 31 January 2023).
- Sepa Arbetsmaskiner, Utsläpp av Växthusgaser. Available online: https://www.naturvardsverket.se/data-och-statistik/klimat/vaxthusgaser-utslapp-fran-arbetsmaskiner/ (accessed on 24 January 2023).
- Muurinen, S.; Slafer, G.A.; Peltonen-Sainio, P. Breeding Effects on Nitrogen Use Efficiency of Spring Cereals under Northern Conditions. Crop Sci. 2006, 46, 561–568. [Google Scholar] [CrossRef]
- Peng, C.; Zhang, Z.; Li, Y.; Zhang, Y.; Dong, H.; Fang, Y.; Han, L.; Xu, W.; Hu, L. Genetic improvement analysis of nitrogen uptake, utilization, translocation, and distribution in Chinese wheat in Henan Province. Field Crops Res. 2022, 277, 108406. [Google Scholar]
- Shi, H.; Chen, M.; Gao, L.; Wang, Y.; Bai, Y.; Yan, H.; Xu, C.; Zhou, Y.; Xu, Z.; Chen, J.; et al. Genome-wide association study of agronomic traits related to nitrogen use efficiency in wheat. Theor. Appl. Genet. 2022, 135, 4289–4302. [Google Scholar]
- Bell, L.W.; Wade, L.J.; Ewing, M.A. Perennial wheat: A review of environmental and agronomic prospects for development in Australia. Crop Pasture Sci. 2010, 61, 679–690. [Google Scholar]
- Cui, L.; Ren, Y.; Murray, T.D.; Yan, W.; Guo, Q.; Niu, Y.; Sun, Y.; Li, H. Development of Perennial Wheat Through Hybridization Between Wheat and Wheatgrasses: A Review. Engineering 2018, 4, 507–513. [Google Scholar]
- Björnsson, L.; Prade, T. Sustainable Cereal Straw Management: Use as Feedstock for Emerging Biobased Industries or Cropland Soil Incorporation? Waste Biomass Valorization 2021, 12, 5649–5663. [Google Scholar]
- Sheehan, H.; Bentley, A. Changing times: Opportunities for altering winter wheat phenology. Plants People Planet 2021, 3, 113–123. [Google Scholar] [CrossRef]
- Araus, J.L.; Slafer, G.A.; Reynolds, M.P.; Royo, C. Plant Breeding and Drought in C3 Cereals: What Should We Breed For? Ann. Bot. 2002, 89, 925–940. [Google Scholar] [CrossRef]
- Baslam, M.; Mitsui, T.; Hodges, M.; Priesack, E.; Herritt, M.T.; Aranjuelo, I.; Sanz-Sáez, Á. Photosynthesis in a Changing Global Climate: Scaling Up and Scaling Down in Crops. Front. Plant Sci. 2020, 11, 1–29. [Google Scholar]
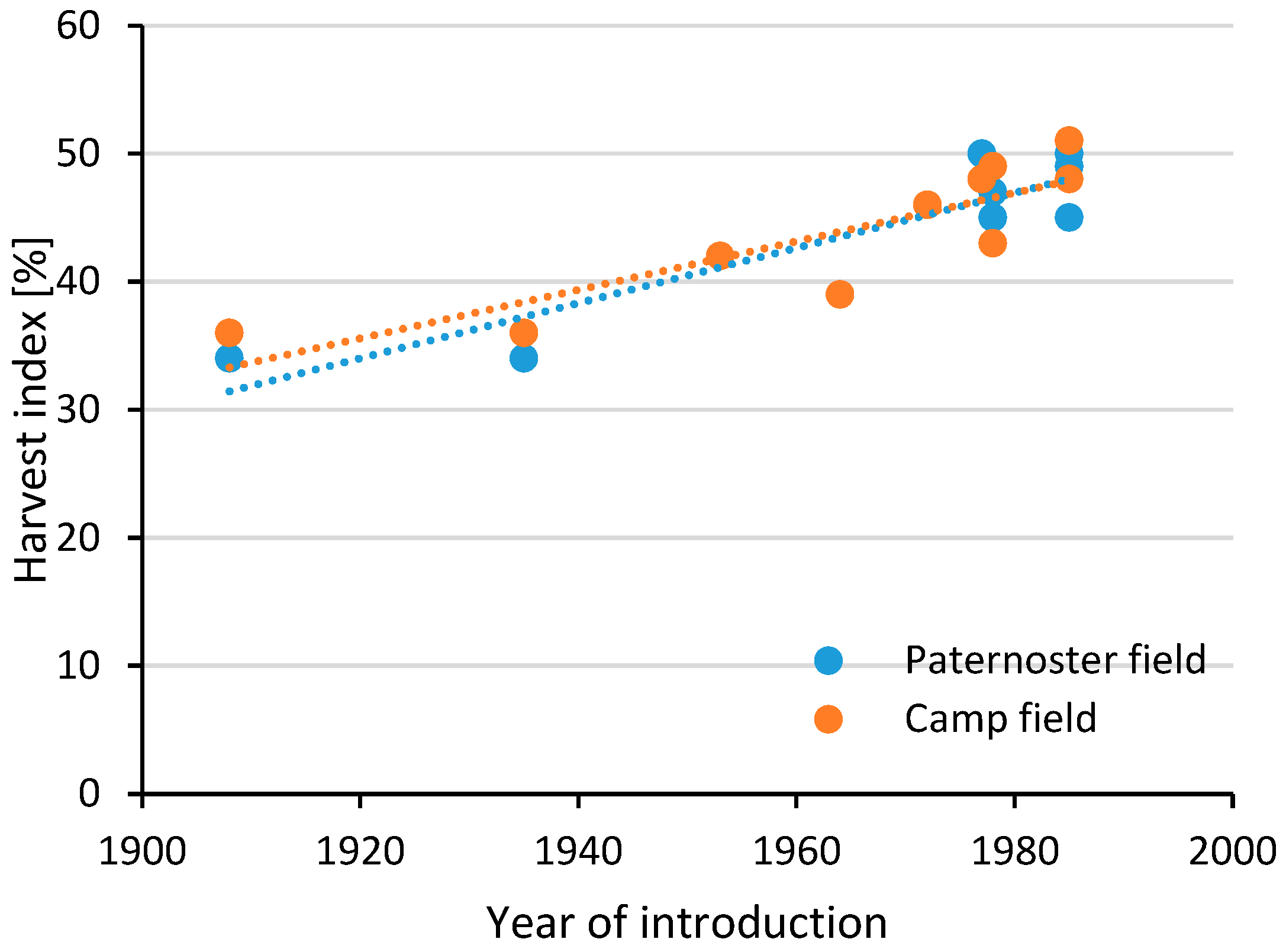
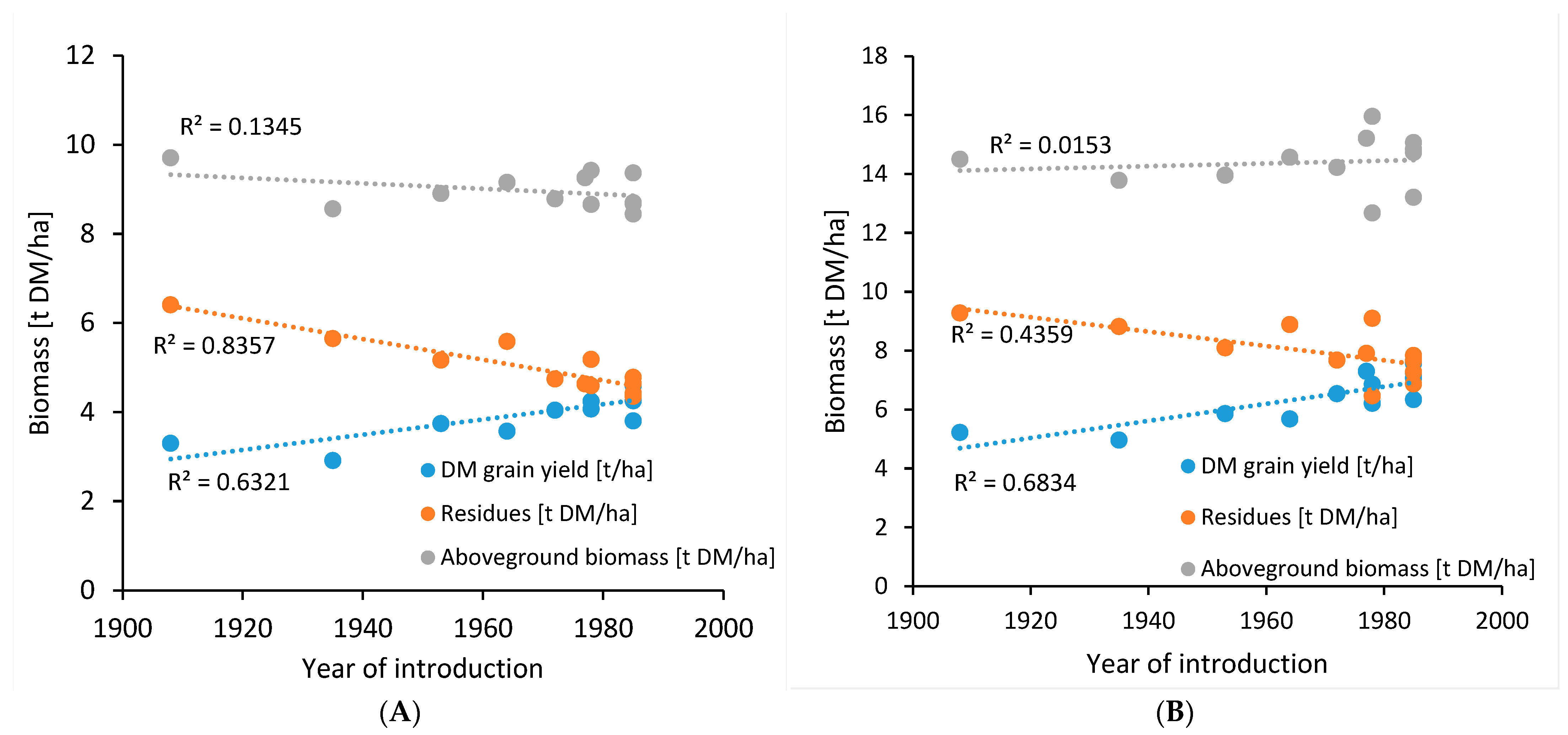

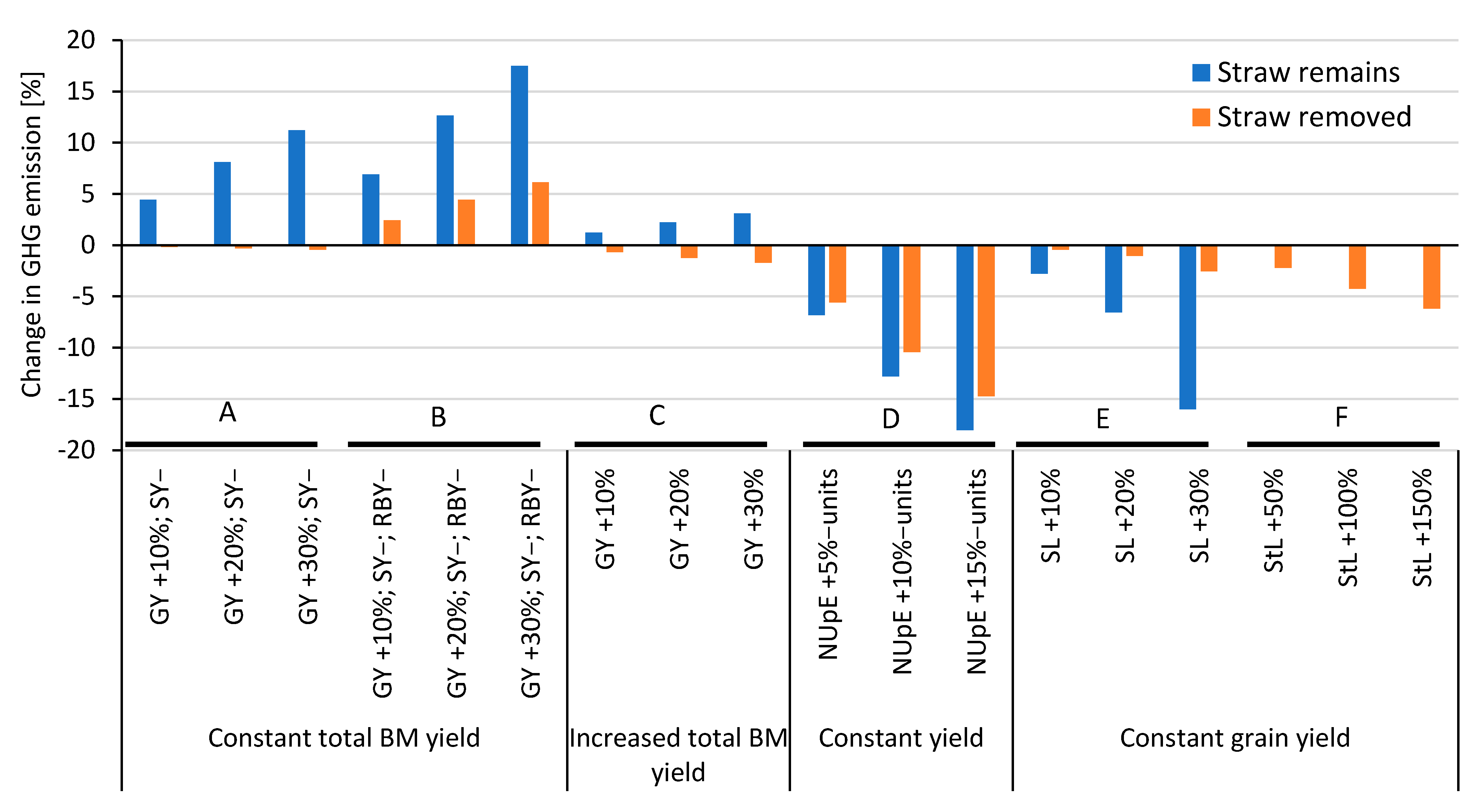
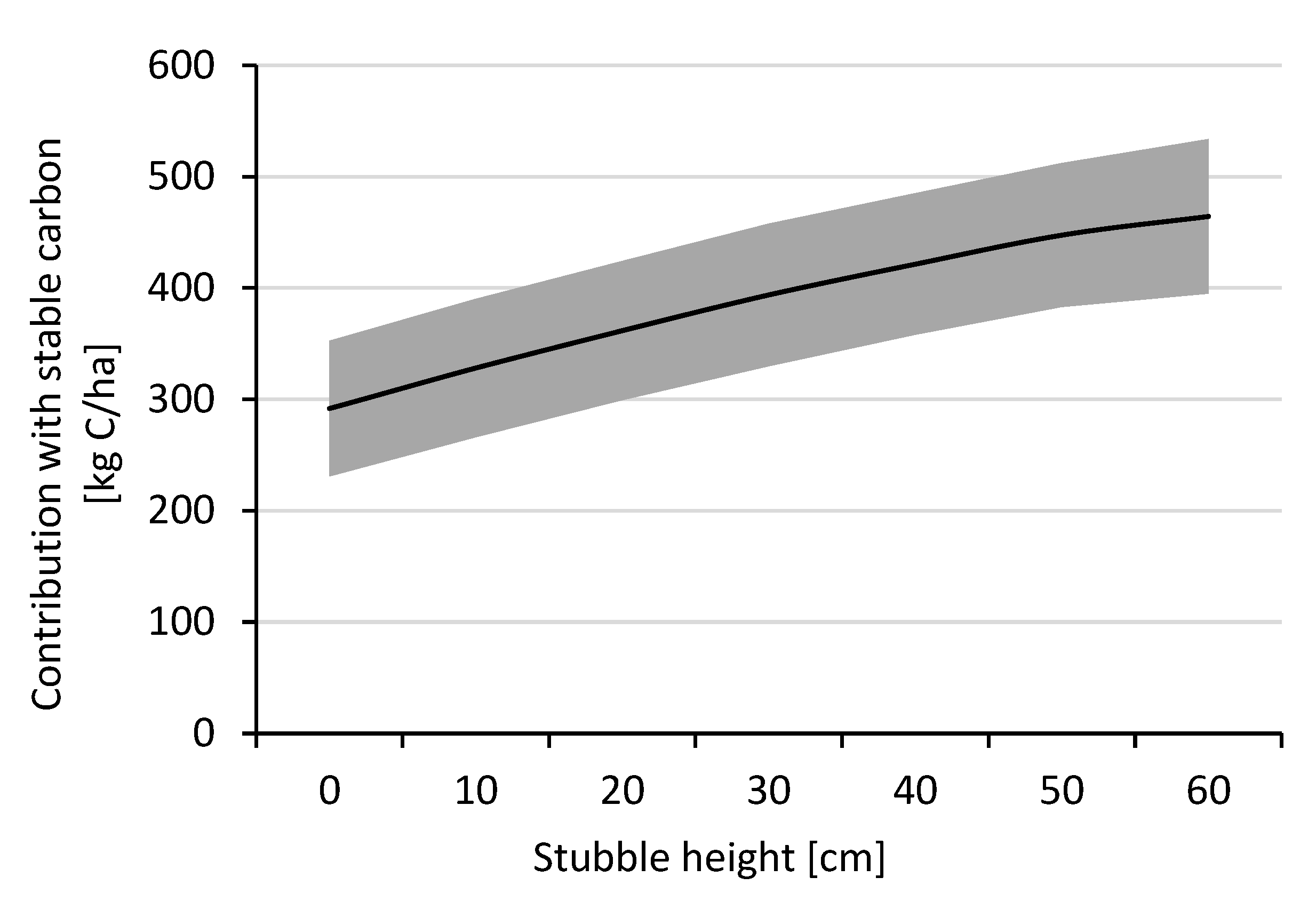
| Operation | Machinery Used | Work Time | Diesel Use | Indirect Energy | GHG Emissions |
|---|---|---|---|---|---|
| [h/ha] | [L/ha] | [MJ/ha] | [kg/ha] | ||
| Stubble treatment (cultivator) | Disc cultivator, 8 m working width | 0.18 | 5.5 | 21 | 19 |
| Ploughing | Plough, 6-bladed | 0.83 | 25.0 | 87 | 87 |
| Harrowing | Harrow, 8 m working width | 0.20 | 12.0 | 61 | 44 |
| Sowing | Combiseeder, 6 m working width, 4600 L capacity | 0.29 | 8.8 | 82 | 36 |
| Rolling | Roller, 12 m working width | 0.17 | 5.0 | 45 | 20 |
| Fertilizer spreading | Fertilizer spreader, 24 m working width, 4000 L capacity | 0.13 | 4.0 | 10 | 14 |
| Spraying | Pesticide sprayer, 24 m working width, 4000 L capacity | 0.13 | 6.5 | 12 | 21 |
| Combine harvest a | Combine, 12 m working width | 0.15–0.19 | 14.1–18.3 | 38–49 | 48–62 |
| Transp. field-dryer (10 km) a | Field trailer, 15 t and 18.5 m3 capacity | 0.15–0.19 | 13.3–17.3 | 44–57 | 46–60 |
| Transp. Dryer-storage (50 km) a | Truck, 37 t and 60 m3 capacity | 0.21–0.27 | 10.3–13.4 | 2.3–3 | 33–42 |
| Drying (ventilator electricity only) a | 8.6–11 | ||||
| Drying (heat production) a | 164–213 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Johansson, E.; Muneer, F.; Prade, T. Plant Breeding to Mitigate Climate Change—Present Status and Opportunities with an Assessment of Winter Wheat Cultivation in Northern Europe as an Example. Sustainability 2023, 15, 12349. https://doi.org/10.3390/su151612349
Johansson E, Muneer F, Prade T. Plant Breeding to Mitigate Climate Change—Present Status and Opportunities with an Assessment of Winter Wheat Cultivation in Northern Europe as an Example. Sustainability. 2023; 15(16):12349. https://doi.org/10.3390/su151612349
Chicago/Turabian StyleJohansson, Eva, Faraz Muneer, and Thomas Prade. 2023. "Plant Breeding to Mitigate Climate Change—Present Status and Opportunities with an Assessment of Winter Wheat Cultivation in Northern Europe as an Example" Sustainability 15, no. 16: 12349. https://doi.org/10.3390/su151612349
APA StyleJohansson, E., Muneer, F., & Prade, T. (2023). Plant Breeding to Mitigate Climate Change—Present Status and Opportunities with an Assessment of Winter Wheat Cultivation in Northern Europe as an Example. Sustainability, 15(16), 12349. https://doi.org/10.3390/su151612349









