Environmental and Socioeconomic Impacts of Shrimp Farming in the Philippines: A Critical Analysis Using PRISMA
Abstract
1. Introduction
2. Materials and Methods
3. Results and Discussion
3.1. Farm Management
3.2. Disease
3.3. Marine Pollution
3.4. Socioeconomic Impacts
3.5. Climate Change
4. Conclusions and Recommendation
- Considering the economic potential to uplift various households from poverty, clearly planned coastal zone use and buffer areas should be delineated, and shrimp pond operators should be educated and informed about land ownership and renting or leasing from the local government and the Department of Environment and Natural Resources (DENR) in the cases that these culture sites are also located in protected areas;
- Aquaculture operators, whether they are small-holders or large-scale operators, should be encouraged and taught how to organize themselves legally, and they should be registered so that they can avail themselves of technical help from the local government, as well as from the Bureau of Fisheries and Aquatic Resources (BFAR);
- Continuous efforts should be extended by the government to organized fish farmers, especially in terms of disease monitoring and water quality monitoring, in order to prevent the spread of disease and eventual economic losses;
- Access to information and educational awareness should be spread within the community organization, as well as to other small-holder farmers, for the uniform implementation of government policies related to shrimp culture, especially with regard to environmental and disease monitoring;
- The development and investments for hatcheries, nurseries, and laboratories should be encouraged among private sector companies, apart from government-led initiatives;
- Shrimp farming should not be allowed on land where it would pose a great threat to cultivated crops, tourism, or recreational areas; proper coastal zoning should be observed by all stakeholders; and unplanned no-consultation aquaculture areas should be discouraged;
- Where there is competition for water resources, such as for crop cultivation and for aquaculture usage, priority is given to rice since it is a staple food. As much as possible, farmlands and shrimp cultivation areas should not be found in the same area because of the possible effluent discharges and seepage;
- In the shrimp farm areas, the proper provision of a network of water supply and drainage canals should be instituted with the observation of better management practices. These should be properly or regularly monitored by the local government, as well as by the BFAR;
- Feeding, fertilizer usage, and the chemicals used should be applied only at the recommended rates to avoid feed wastage and to prevent chemical contamination;
- Food safety, traceability practices, and better farm management practices should be observed, with the corresponding sanctions and with regular monitoring;
- Strong networks among research organizations, local governments, and universities and colleges, as well as NGOs, through forums and extension services, could improve knowledge, the prevention of diseases, and the adoption of sustainable practices by shrimp farmers, which will lead to better management practices.
Author Contributions
Funding
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Islam, M.; Yasmin, R. Impact of aquaculture and contemporary environmental issues in Bangladesh. Int. J. Fish. Aquat. Stud. 2017, 5, 100–107. [Google Scholar]
- Joffre, O.M.; Bosma, R.H. Typology of shrimp farming in Bac Lieu Province, Mekong Delta, using multivariate statistics. Agric. Ecosyst. Environ. 2009, 132, 153–159. [Google Scholar] [CrossRef]
- Henriksson, P.J.G.; Banks, L.K.; Suri, S.K.; Pratiwi, T.Y.; Fatan, N.A.; Troell, M. Indonesian aquaculture futures—Identifying interventions for reducing environmental impacts. Environ. Res. Lett. 2019, 14, 124062. [Google Scholar] [CrossRef]
- Guerrero, R.D. Farmed tilapia production in the Philippines is declining: What has happened and what can be done. Philipp. J. Sci. 2019, 148, XI–XV. [Google Scholar]
- Miao, W.; Mohan, C.V.; Ellis, W.; Brian, D. Adoption of Aquaculture Assessment Tools for Improving the Planning and Management of Aquaculture in Asia and the Pacific; FAO Regional Office for Asia and the Pacific: Bangkok, Thailand, 2013; p. 136. [Google Scholar]
- Largo, D.B.; Diola, A.G.; Marababol, M.S. Development of an integrated multi-trophic aquaculture (IMTA) system for tropical marine species in southern Cebu, Central Philippines. Aquac. Rep. 2016, 3, 67–76. [Google Scholar] [CrossRef]
- Walker, P.J.; Winton, J.R. Emerging viral diseases of fish and shrimp. Vet. Res. 2010, 41, 51. [Google Scholar] [CrossRef]
- Béné, C.; Barange, M.; Subasinghe, R.; Pinstrup-Andersen, P.; Merino, G.; Hemre, G.-I.; Williams, M. Feeding 9 billion by 2050—Putting fish back on the menu. Food Sec. 2015, 7, 261–274. [Google Scholar] [CrossRef]
- Diana, J.S. Aquaculture Production and Biodiversity Conservation. Bioscience 2009, 59, 27–38. [Google Scholar] [CrossRef]
- Béné, C.; Arthur, R.; Norbury, H.; Allison, E.H.; Beveridge, M.; Bush, S.; Campling, L.; Leschen, W.; Little, D.; Squires, D.; et al. Contribution of Fisheries and Aquaculture to Food Security and Poverty Reduction: Assessing the Current Evidence. World Dev. 2016, 79, 177–196. [Google Scholar] [CrossRef]
- FAO. The State of World Fisheries and Aquaculture (SOFIA); Food and Agriculture Organization: Rome, Italy, 2008. [Google Scholar]
- Vo, L.T.T. Quality Management in Shrimp Supply Chain in the Mekong Delta, Vietnam: Problems and Measures; Center for ASEAN Studies: Antwerp, Belgium, 2003; p. 28. [Google Scholar]
- Martinez-Cordova, L.R.; Martinez-Porchas, M. Polyculture of Pacific white shrimp, Litopenaeus vannamei, giant oyster, Crassostrea gigas and black clam, Chione fluctifraga in ponds in Sonora, Mexico. Aquaculture 2006, 258, 321–326. [Google Scholar] [CrossRef]
- FAO. The State of World Fisheries and Aquaculture 2020; Food and Agriculture Organization: Rome, Italy, 2020. [Google Scholar]
- BFAR. Philippine Fisheries Profile 2019; Bureau of Fisheries and Aquatic Resources: Quezon City, Philippines, 2020; p. 76. [Google Scholar]
- Cuvin-Aralar, M.L.A.; Lazartigue, A.G.; Aralar, E.V. Cage culture of the Pacific white shrimp Litopenaeus vannamei (Boone, 1931) at different stocking densities in a shallow eutrophic lake. Aquac. Res. 2009, 40, 181–187. [Google Scholar] [CrossRef]
- Rosario, W.R.; Lopez, N.A. Status of P. vannamei Aquaculture in the Philippines; SEAFDEC Aquaculture Department: Iloilo, Philippines, 2005; pp. 62–68. [Google Scholar]
- Shinn, A.P.; Pratoomyot, J.; Griffiths, D.; Trong, T.Q.; Vu, N.T.; Jiravanichpaisal, P.; Briggs, M. Asian Shrimp Production and the Economic Costs of Disease. Asian Fish. Sci. 2018, 31S, 29–58. [Google Scholar] [CrossRef]
- Primavera, J.H. Overcoming the impacts of aquaculture on the coastal zone. Ocean. Coast. Manag. 2006, 49, 531–545. [Google Scholar] [CrossRef]
- PSA. Fisheries Situationer of the Philippines; Philippine Statistics Authority: Quezon City, Philippines, 2020; p. 321. [Google Scholar]
- Vergel, J.C.V. Current Trends in the Philippines’ Shrimp Aquaculture Industry: A Booming Blue Economy in the Pacific. Oceanogr. Fish. Open Access J. 2017, 5, 555668. [Google Scholar] [CrossRef]
- Tendencia, E.A.; Bosma, R.H.; Verdegem, M.C.J.; Verreth, J.A.J. The potential effect of greenwater technology on water quality in the pond culture of Penaeus monodon Fabricius. Aquac. Res. 2015, 46, 1–13. [Google Scholar] [CrossRef]
- Primavera, J.H. Socioeconomic impacts of shrimp culture. Aquac. Res. 1997, 28, 815–827. [Google Scholar] [CrossRef]
- Primavera, J.H. A critical review of shrimp pond culture in the Philippines. Rev. Fish. Sci. 1993, 1, 151–201. [Google Scholar] [CrossRef]
- PhilStat. Fisheries Statistics of the Philippines 2014–2016; Philippine Statistics Authority: Quezon City, Philippines, 2017; pp. 484–523. [Google Scholar]
- Pudadera, B.J.; Lim, C. Evaluation of milkfish (Chanos chanos Forskal) and prawn (Penaeus monodon Fabricius) in polyculture systems. Fish. Res. J. Philipp. 1980, 7, 51–59. [Google Scholar]
- Eldani, A.; Primavera, J.H. Effect of different stocking combinations on growth, production and survival of milkfish (Chanos chanos) and prawn (Penaeus monodon) in polyculture in brackishwater. Aquaculture 1981, 23, 59–72. [Google Scholar] [CrossRef]
- Flegel, T.W.; Lightner, D.V.; Lo, C.F.; Owens, L. Shrimp disease control: Past, present and future. In Diseases in Asian Aquaculture VI; Fish Health Section, Asian Fisheries Society: Manila, Philippines, 2008; pp. 355–378. [Google Scholar]
- Tendencia, E.A.; dela Peña, M. Investigation of some components of the greenwater system which makes it effective in the initial control of luminous bacteria. Aquaculture 2003, 218, 115–119. [Google Scholar] [CrossRef]
- Jaspe, C.J.; Caipang, C.M.A.; Elle, B.J.G. Polyculture of white shrimp, Litopenaeus vannamei and milkfish, Chanos chanos as a strategy for efficient utilization of natural food production in ponds. ABAH Bioflux 2011, 3, 96–104. [Google Scholar]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G. Preferred Reporting Items for Systematic Reviews and Meta-Analyses: The PRISMA Statement. PLoS Med. 2009, 6, e1000097. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.-M.; Borazon, E.Q.; Muñoz, K.E. Critical problems associated with climate change: A systematic review and meta-analysis of Philippine fisheries research. Environ. Sci. Pollut. Res. 2021, 28, 49425–49433. [Google Scholar] [CrossRef]
- Li, T.; Hua, F.; Dan, S.; Zhong, Y.; Levey, C.; Song, Y. Reporting quality of systematic review abstracts in operative dentistry: An assessment using the PRISMA for Abstracts guidelines. J. Dent. 2020, 102, 103471. [Google Scholar] [CrossRef] [PubMed]
- Sharma, S.; Oremus, M. PRISMA and AMSTAR show systematic reviews on health literacy and cancer screening are of good quality. J. Clin. Epidemiol. 2018, 99, 123–131. [Google Scholar] [CrossRef] [PubMed]
- van Eck, N.J.; Waltman, L. Software survey: VOSviewer, a computer program for bibliometric mapping. Scientometrics 2010, 84, 523–538. [Google Scholar] [CrossRef]
- Snilstveit, B.; Oliver, S.; Vojtkova, M. Narrative approaches to systematic review and synthesis of evidence for international development policy and practice. J. Dev. Eff. 2012, 4, 409–429. [Google Scholar] [CrossRef]
- Schick-Makaroff, K.; MacDonald, M.; Plummer, M.; Burgess, J.; Neander, W. What synthesis methodology should I use? A review and analysis of approaches to research synthesis. AIMS Public Health 2016, 3, 172. [Google Scholar] [CrossRef]
- Ashton, E.C. The impact of shrimp farming on mangrove ecosystems: Perspectives in Agriculture, Veterinary Science, Nutrition and Natural Resources. CAB Rev. 2008, 3, 1–12. [Google Scholar] [CrossRef]
- Primavera, J.H.; Altamirano, J.P.; Lebata, M.J.H.L.; delos Reyes, A.A., Jr.; Pitogo, C.L. Mangroves and shrimp pond culture effluents in Aklan, Panay Is., Central Philippines. Bull. Mar. Sci. 2007, 80, 795–804. [Google Scholar]
- Suh, D.; Pomeroy, R. Projected Economic Impact of Climate Change on Marine Capture Fisheries in the Philippines. Front. Mar. Sci. 2020, 7, 232. [Google Scholar] [CrossRef]
- Macusi, E.D.; Abreo, N.A.S.; Cuenca, G.C.; Ranara, C.T.B.; Cardona, L.T.; Andam, M.B.; Guanzon, G.C.; Katikiro, R.E.; Deepananda, K.H.M.A. The potential impacts of climate change on freshwater fish, fish culture and fishing communities. J. Nat. Stud. 2015, 14, 14–31. [Google Scholar]
- Cruz-Lacierda, E.; Corre, V.; Yamamoto, A.; Koyama, J.; Matsuoka, T. Current Status on the Use of Chemicals and Biological Products and Health Management Practices in Aquaculture Farms in the Philippines. Mem. Fac. Fish. Kagoshima Univ. 2008, 57, 37–45. [Google Scholar]
- Leobert, D.; Lavilla-Pitogo, C.R.; Villar, C.B.R.; Paner, M.G.; Sombito, C.D.; Capulos, G.C. Prevalence of white spot syndrome virus (WSSV) in wild shrimp Penaeus monodon in the Philippines. Dis. Aquat. Organ. 2007, 77, 175–179. [Google Scholar] [CrossRef] [PubMed]
- Lavilla-Pitogo, C.R.; de la Pena, L.D. Bacterial Disease in Shrimp (Penaeus monodon) culture in the Philippines. Fish. Pathol. 1998, 33, 405–411. [Google Scholar] [CrossRef]
- Baticados, M.C.L.; Cruz-Lacierda, E.R.; De la Cruz, M.C.; Duremdez-Fernandez, R.C.; Gacutan, R.Q.; Lavilla-Pitogo, C.R.; Lio-Po, G.D. Diseases of Penaeid Shrimps in the Philippines; Aquaculture Department, Southeast Asian Fisheries Development Center (SEAFDEC): Tigbauan, Iloilo, Philippines, 1990. [Google Scholar]
- Karunasagar, I.; Ababouch, L. Shrimp viral diseases, import risk assessment and international trade. Indian J. Virol. 2012, 23, 141–148. [Google Scholar] [CrossRef]
- Momoyama, K.; Hiraoka, M.; Nakano, H.; Koube, H.; Inouye, K.; Oseko, N. Mass mortalities of cultured Kuruma shrimp, Penaeus japonicus, in Japan in 1993. Fish Pathol. 1994, 29, 141–158. [Google Scholar] [CrossRef]
- Belak, J.; Dhar, A.K.; Primavera, J.H. Prevalence of viral diseases (IHHNV and WSSV) in Penaeus monodon from the Philippines and its association with mangrove status and shrimp culture systems. In Proceedings of the Symposium on Aquaculture and Conservation of Marine Shrimp Biodiversity, North Grafton, MA, USA; 1999. [Google Scholar]
- Briggs, M.; Funge-Smith, S.; Subasinghe, R.; Phillips, M. Introductions and Movement of Penaeus vannamei and Penaeus stylirostris in Asia and the Pacific; Bangkok Food and Agriculture Organization of the United Nations: Bangkok, Thailand, 2004; p. 88. [Google Scholar]
- Vergel, J.C.V.; Cabawatan, L.D.P.; Madrona, V.A.C.; Rosario, A.F.T.; Tare, M.V.R.; Maningas, M.B.B. Detection of Taura Syndrome Virus (TSV) in Litopenaeus vannamei in the Philippines. Philipp. J. Fish. 2019, 26, 8–14. [Google Scholar] [CrossRef]
- Tendencia, E.A.; Bosma, R.H.; Verreth, J.A.J. White spot syndrome virus (WSSV) risk factors associated with shrimp farming practices in polyculture and monoculture farms in the Philippines. Aquaculture 2011, 311, 87–93. [Google Scholar] [CrossRef]
- Moriarty, D.J.W. Disease Control in Shrimp Aquaculture with Probiotic Bacteria. In Proceedings of the 8th International Symposium on Microbial Ecology, Halifax, NS, Canada, 9–14 August 1998; pp. 1–7. [Google Scholar]
- Lavilla-Pitogo, C.R.; Lio-Po, G.D.; Cruz-Lacierda, E.R.; Alapide-Tendencia, E.V.; de la Peña, L.D. Diseases of Penaeid Shrimps in the Philippines; Aquaculture Department, Southeast Asian Fisheries Development Center (SEAFDEC): Tigbauan, Iloilo, Philippines, 2000. [Google Scholar]
- Orosco, F.L.; Lluisma, A.O. Prevalence, diversity and co-occurrence of the white spot syndrome virus, monodon baculovirus and Penaeus stylirostris densovirus in wild populations of Penaeus monodon in the Philippines. Dis. Aquat. Organ. 2017, 125, 199–206. [Google Scholar] [CrossRef]
- De la Peña, L.D. Transboundary Shrimp Viral Diseases with Emphasis on White Spot Syndrome Virus (WSSV) and Taura Syndrome Virus (TSV); Aquaculture Department, SEAFDEC: Tigbauan, Iloilo, Philippines, 2004; pp. 67–69. [Google Scholar]
- Flegel, T.W. Detection of major penaeid shrimp viruses in Asia, a historical perspective with emphasis on Thailand. Aquaculture 2006, 258, 1–33. [Google Scholar] [CrossRef]
- Rajendran, K.V.; Makesh, M.; Karunasagar, I. Monodon baculovirus of shrimp. Indian J. Virol. 2012, 23, 149–160. [Google Scholar] [CrossRef] [PubMed]
- Baticados, M.C.L.; Lavilla-Pitogo, C.R.; Cruz-Lacierda, E.R.; de la Peña, L.D.; Suñaz, N.A. Studies on the chemical control of luminous bacteria Vibrio harveyi and V. splendidus isolated from diseased Penaeus monodon larvae and rearing water. Dis. Aquat. Organ. 1990, 9, 133–139. [Google Scholar] [CrossRef]
- Holmer, M.; Marba, N.; Terrados, J.; Duarte, C.M.; Fortes, M.D. Impacts of milkfish (Chanos chanos) aquaculture on carbon and nutrient fluxes in the Bolinao area, Philippines. Mar. Poll. Bull. 2002, 44, 685–696. [Google Scholar] [CrossRef]
- Azanza, R.; Baula, I.; Fukuyo, Y. Seasonal changes in phytoplankton composition in an extensive fish culture area in Bolinao, Pangasinan Philippines. Coast. Mar. Sci. 2006, 30, 85–87. [Google Scholar]
- Macandog, D.M.; de la Cruz, C.P.P.; Edrial, J.D.; Reblora, M.A.; Pabico, J.P.; Salvacion, A.R.; Marquez, T., Jr.; Macandog, P.B.M.; Perez, D.K.B. Eliciting Local Ecological Knowledge and Community Perception on Fishkill in Taal Lake through Participatory Approaches. J. Environ. Sci. Manag. 2014, 17, 2. [Google Scholar] [CrossRef]
- Vista, A.; Norris, P.; Lupi, F.; Bernsten, R. Nutrient loading and efficiency of tilapia cage culture in Taal Lake, Philippines. Philipp. Agric. Sci. 2006, 89, 48–57. [Google Scholar]
- Primavera, J.H.; Esteban, J. A review of mangrove rehabilitation in the Philippines: Successes, failures and future prospects. Wetl. Ecol. Manag. 2008, 16, 345–358. [Google Scholar] [CrossRef]
- Lee, S.Y.; Primavera, J.H.; Dahdouh-Guebas, F.; Mckee, K.; Bosire, J.O.; Cannicci, S.; Diele, K.; Fromard, F.; Koedam, N.; Marchand, C.; et al. Ecological role and services of tropical mangrove ecosystems: A reassessment. Glob. Ecol. Biogeogr. 2014, 23, 726–743. [Google Scholar] [CrossRef]
- Muallil, R.N.; Mamauag, S.S.; Cababaro, J.T.; Arceo, H.O.; Aliño, P.M. Catch trends in Philippine small-scale fisheries over the last five decades: The fishers’ perspectives. Mar. Policy 2014, 47, 110–117. [Google Scholar] [CrossRef]
- Macusi, E.D.; Liguez, A.K.O.; Macusi, E.S.; Digal, L.N. Factors influencing catch and support for the implementation of the closed fishing season in Davao Gulf, Philippines. Mar. Policy 2021, 130, 104578. [Google Scholar] [CrossRef]
- Cuenca, G.C.; Macusi, E.D.; Abreo, N.A.S.; Ranara, C.T.B.; Andam, M.B.; Cardona, L.T.; Guanzon, G.C. Mangrove Ecosystems and Associated Fauna with Special Reference to Mangrove Crabs in the Philippines: A Review. IAMURE Int. J. Ecol. Conserv. 2015, 15, 60–110. [Google Scholar] [CrossRef]
- Macusi, E.D.; Macusi, E.S.; Jimenez, L.A.; Catam-isan, J.P. Climate change vulnerability and perceived impacts on small-scale fisheries in eastern Mindanao. Ocean Coast. Manag. 2020, 189, 105143. [Google Scholar] [CrossRef]
- Subasinghe, R.P. Network of Aquaculture Centres in Asia-Pacific. Food and Agriculture Organization of the United Nations. In Aquaculture in the Third Millennium; NACA/FAO: Bangkok, Thailand, 2001; p. 471. [Google Scholar]
- Olalo, C. Production, Accessibility and Consumption Patterns of Aquaculture Products in the Philippines; Food and Agriculture Organization: Rome, Italy, 2001. [Google Scholar]
- Miraflor, M.B. PH Shrimp Production to Remain Stagnant, Manila Bulletin; Coloma, H., Jr., Ed.; Sonny Coloma: Manila, Philippines, 2021. [Google Scholar]
- Drury O’Neill, E.; Crona, B.; Ferrer, A.J.G.; Pomeroy, R.; Jiddawi, N.S. Who benefits from seafood trade? A comparison of social and market structures in small-scale fisheries. Ecol. Soc. 2018, 23, 12. [Google Scholar] [CrossRef]
- Macusi, E.D.; Morales, I.D.G.; Macusi, E.S.; Pancho, A.; Digal, L.N. Impact of closed fishing season on supply, catch, price and the fisheries market chain. Mar. Pol. 2022, 138, 105008. [Google Scholar] [CrossRef]
- Islam, M.S.; Islam, M.S.; Wahab, M.A.; Miah, A.A.; Mustafa Kamal, A.H.M. Impacts of Shrimp Farming on the Socioeconomic and Environmental Conditions in the Coastal Regions of Bangladesh. Pak. J. Biol. Sci. 2003, 6, 2058–2067. [Google Scholar] [CrossRef][Green Version]
- Alejos, M.S.; Serrano, A.E.; Jumah, Y.U.; Dela Calzada, R.; Ranara, C.T.B.; Fernandez, J.C. Ecological and social impacts of aquacultural introduction to Philippines waters of Pacific whiteleg shrimp Penaeus vannamei. In Spatial Variability in Environmental Science—Patterns, Processes, and Analyses; IntechOpen: London, UK, 2020. [Google Scholar]
- Bassig, R.A.; Obinque, A.V.; Nebres, V.T.; Delos Santos, V.H.; Peralta, D.M.; Madrid, A.J.J. Utilization of Shrimp Head Wastes into Powder Form as Raw Material for Value-Added Products. Philipp. J. Fish. 2022, 28, 181–190. [Google Scholar] [CrossRef]
- Smith, L.C.; Ramakrishnan, U.; Ndiaye, A.; Haddad, L.; Martorell, R. The Importance of Women’s Status for Child Nutrition in Developing Countries; International Food Policy Research Institute (IFPRI): Washington, DC, USA, 2003; p. 164. [Google Scholar]
- Wandel, M.; Holmboe-Ottesen, G. Women’s work in agriculture and child nutrition in Tanzania. J. Trop. Pediatr. 1992, 38, 252–255. [Google Scholar] [CrossRef]
- Cunningham, K.; Ploubidis, G.B.; Menon, P.; Ruel, M.; Kadiyala, S.; Uauy, R.; Ferguson, E. Women’s empowerment in agriculture and child nutritional status in rural Nepal. Public Health Nutr. 2015, 18, 3134–3145. [Google Scholar] [CrossRef]
- Cruz-Lacierda, E.R.; de la Peña, L.D.; Lumanlan-Mayo, S.C. The Use of Chemicals in Aquaculture in the Philippines. In Proceedings of the Use of Chemicals in Aquaculture in Asia, Tigbauan, Iloilo, Philippines, 20–22 May 1996; pp. 155–184. [Google Scholar]
- Hossain, A.; Habibullah-Al-Mamun, M.; Nagano, I.; Masunaga, S.; Kitazawa, D.; Matsuda, K. Antibiotics, antibiotic-resistant bacteria, and resistance genes in aquaculture: Risks, current concern, and future thinking. Environ. Sci. Pollut. Res. 2022, 29, 11054–11075. [Google Scholar] [CrossRef]
- Monnier, L.; Gascuel, D.; Alava, J.J.; Barragán, M.J.; Gaibor, N.; Hollander, F.A.; Kanstinger, P.; Niedermueller, S.; Ramírez, J.; Cheung, W.W.L. Small-Scale Fisheries in a Warming Ocean: Exploring Adaptation to Climate Change; WWF Germany: Berlin, Germany, 2020. [Google Scholar]
- Eckstein, D.; Künzel, V.; Schäfer, L.; Winges, W. Global Climate Risk Index 2020: Who Suffers Most from Extreme Weather Events? Weather-Related Loss Events in 2018 and 1999 to 2018; Germanwatch e.V.: Bonn, Germany, 2020; p. 44. [Google Scholar]
- Macusi, E.D.; Geronimo, R.C.; Santos, M.D. Vulnerability drivers for small pelagics and milkfish aquaculture value chain determined through online participatory approach. Mar. Policy 2021, 133, 104710. [Google Scholar] [CrossRef]
- Islam, A.M.; Akber, M.A.; Ahmed, M.; Rahman, M.M.; Rahman, M.R. Climate change adaptations of shrimp farmers: A case study from southwest coastal Bangladesh. Clim. Dev. 2018, 11, 459–468. [Google Scholar] [CrossRef]
- Macusi, E.D.; Kezia, L.; Camaso, K.L.; Barboza, A.; Macusi, E.R. Perceived vulnerability and climate change impacts on small-scale fisheries in Davao gulf, Philippines. Front. Mar. Sci. 2021, 8, 597385. [Google Scholar] [CrossRef]
- BFAR. Comprehensive National Fisheries Industry Development Plan (CNFIDP) Medium Term Plan 2016–2020 Department of Agriculture-Bureau of Fisheries and Aquatic Resources (DA-BFAR); Bureau of Fisheries and Aquatic Resources: Quezon City, Philippines, 2016. [Google Scholar]
- Song, A.M.; Dressler, W.H.; Satizábal, P.; Fabinyi, M. From conversion to conservation to carbon: The changing policy discourse on mangrove governance and use in the Philippines. J. Rural Stud. 2021, 82, 184–195. [Google Scholar] [CrossRef]
- Edwards, P. Aquaculture environment interactions: Past, present and likely future. Aquaculture 2015, 447, 2–14. [Google Scholar] [CrossRef]
- Kusumawati, R.; Bush, S.R. Co-producing Better Management Practice standards for shrimp aquaculture in Indonesia. Marit. Stud. 2015, 14, 21. [Google Scholar] [CrossRef]
- Jacinto, G.S. Fish Kill in the Philippines—Déjà Vu. Sci. Diliman 2011, 23, 1–3. [Google Scholar]
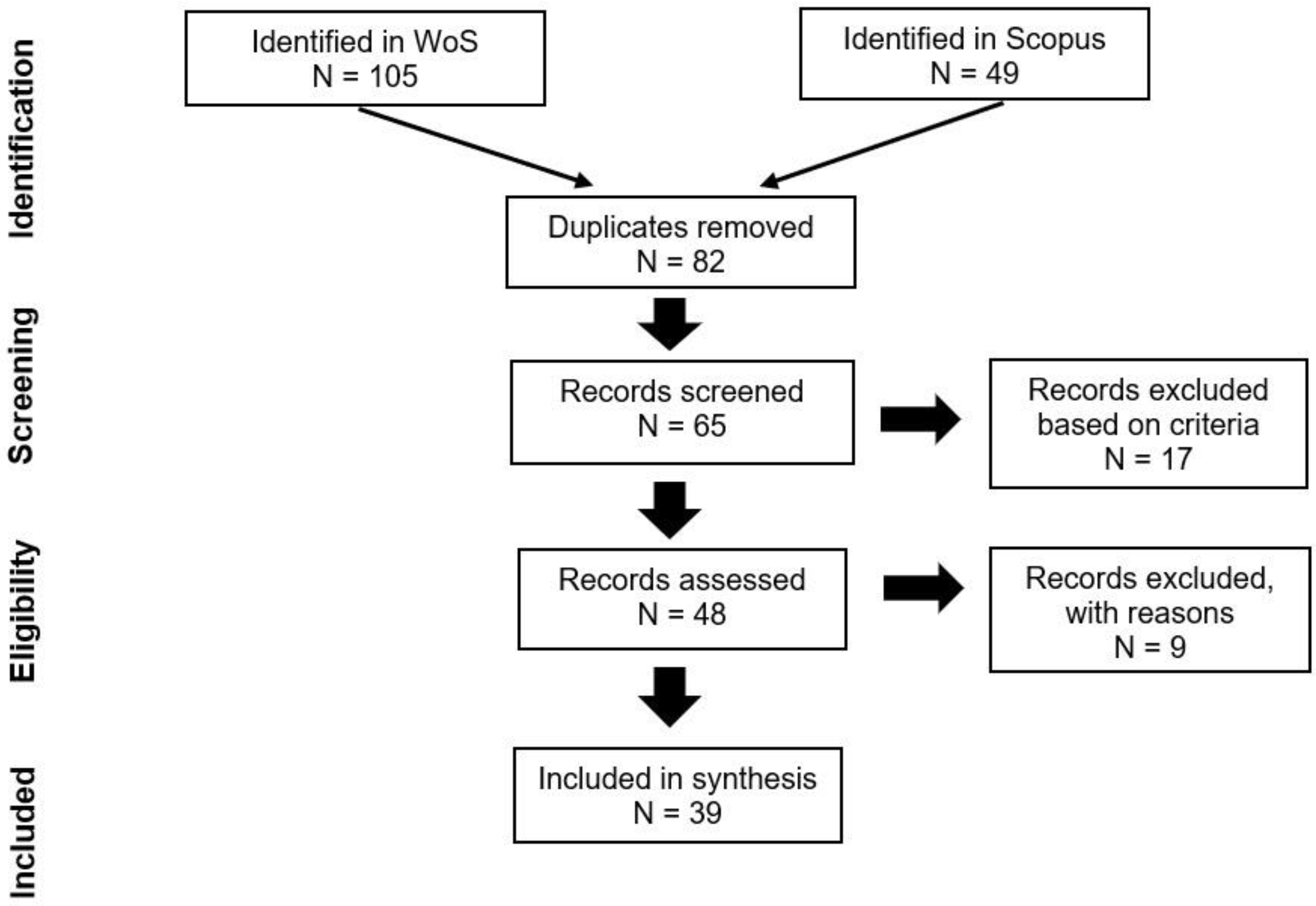

| Year | Milestone | References |
|---|---|---|
| 1960s |
| [17,24] |
| 1970s |
| [17,24] |
| 1980s |
| [17,26,27] |
| 1990s |
| [17,23,24,28] |
| 2000s |
| [17,29] |
| 2010s |
| [15,21,22,30] |
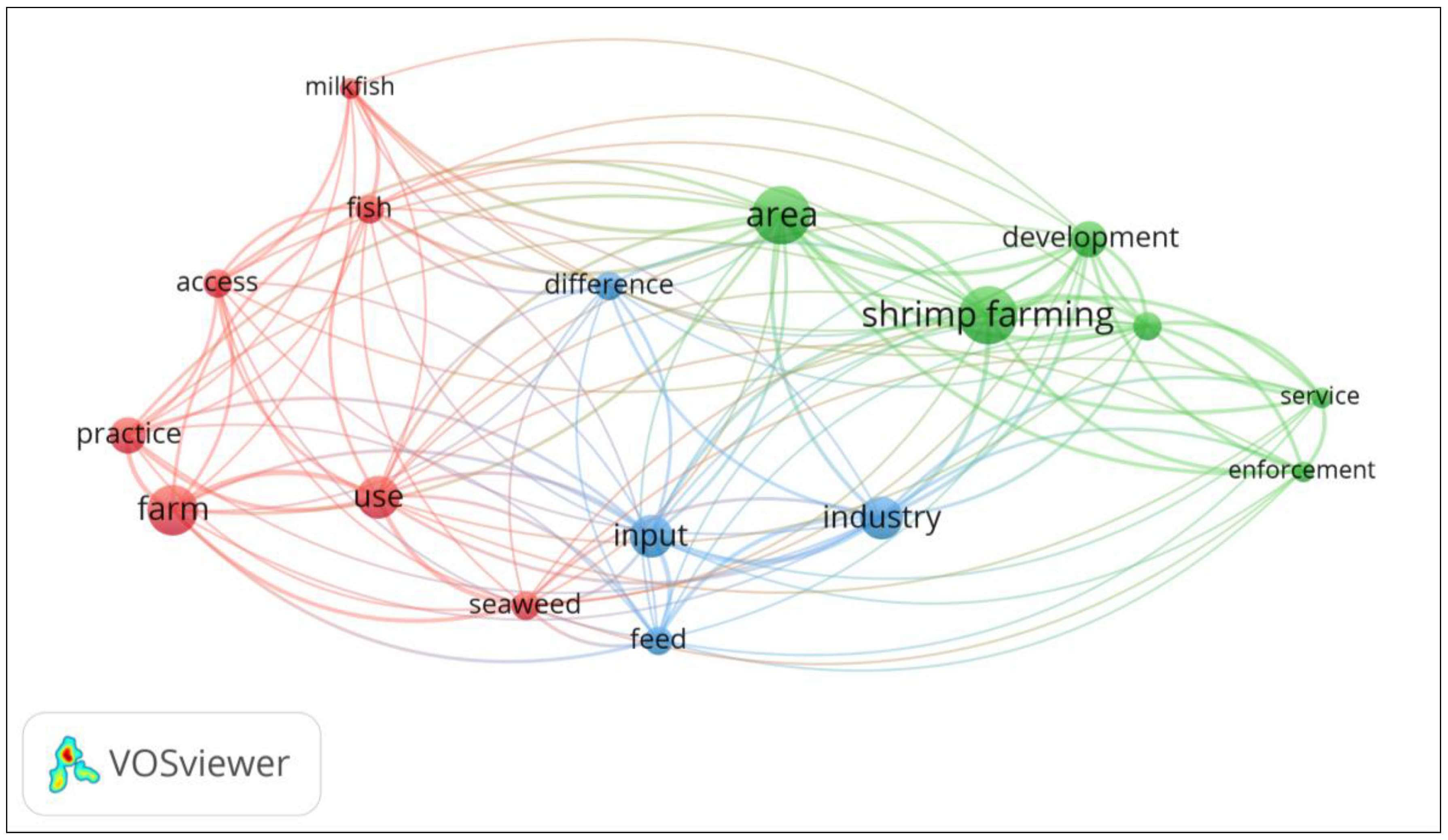
| Keywords | Occurrences | Links | Total Link Strength |
|---|---|---|---|
| Cluster 1 | |||
| Aquaculture | 6 | 9 | 12 |
| Ecological effects | 2 | 6 | 7 |
| Mangroves | 2 | 4 | 4 |
| Penaeus monodon | 3 | 8 | 9 |
| Pollution | 2 | 6 | 6 |
| Shrimp | 2 | 7 | 7 |
| Socioeconomic effects | 2 | 6 | 7 |
| Cluster 2 | |||
| Decapoda (crustacean) | 7 | 7 | 16 |
| Fishery production | 2 | 5 | 8 |
| Philippines | 5 | 8 | 17 |
| Cluster 3 | |||
| Economics | 2 | 2 | 3 |
| Shrimp culture | 6 | 8 | 19 |
| Shrimp farming | 6 | 6 | 13 |
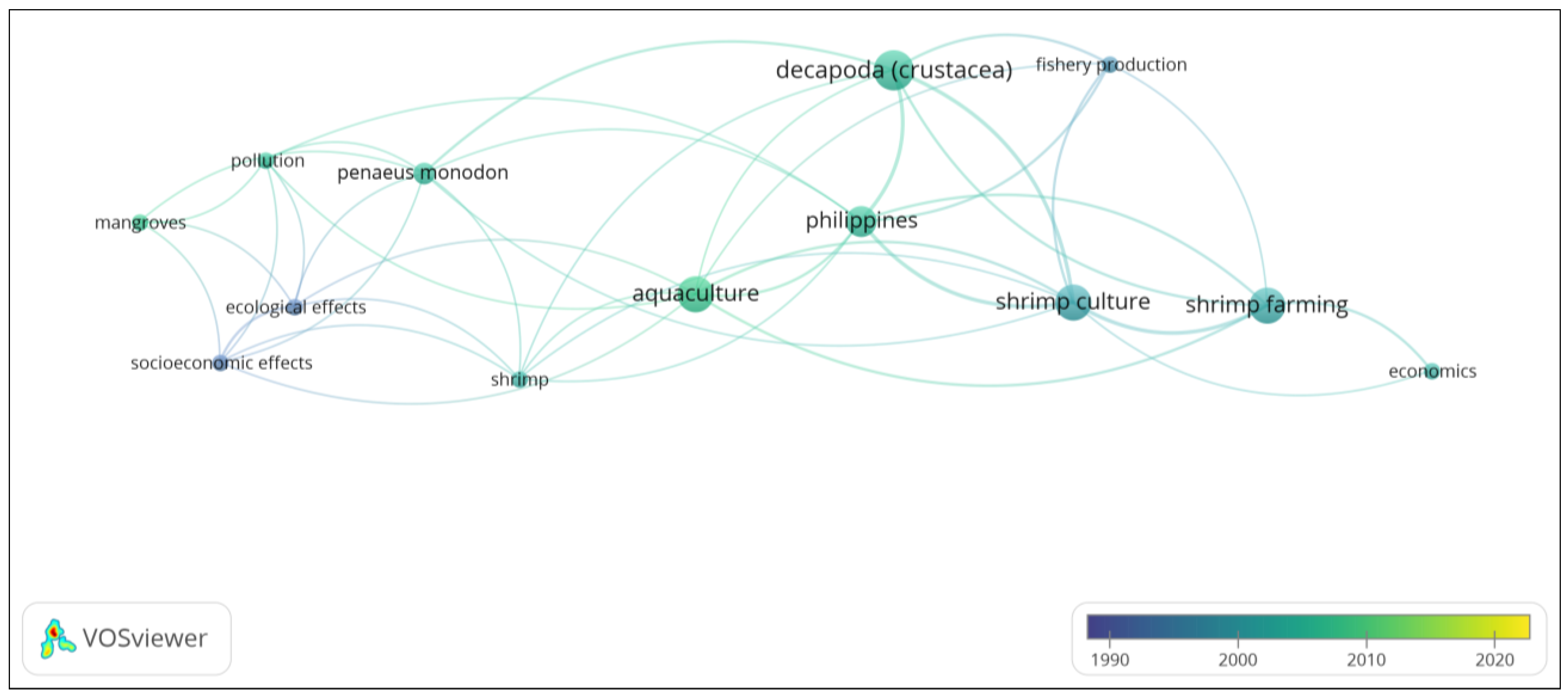
| Abstract | Occurrences | Links | Total Link Strength |
|---|---|---|---|
| Cluster 1 | |||
| Access | 4 | 12 | 18 |
| Farm | 7 | 11 | 25 |
| Fish | 4 | 14 | 22 |
| Milkfish | 3 | 10 | 15 |
| Practice | 5 | 11 | 19 |
| Seaweed | 4 | 15 | 23 |
| Use | 6 | 16 | 31 |
| Cluster 2 | |||
| Area | 8 | 16 | 41 |
| Development | 5 | 14 | 29 |
| Enforcement | 3 | 10 | 21 |
| Mangrove | 4 | 13 | 25 |
| Service | 3 | 10 | 21 |
| Shrimp farming | 8 | 15 | 35 |
| Cluster 3 | |||
| Difference | 4 | 12 | 16 |
| Feed | 4 | 15 | 23 |
| Industry | 6 | 15 | 23 |
| Input | 6 | 15 | 29 |
| Year | No. of Studies | % | Journal Name |
|---|---|---|---|
| 1993 | 1 | 2.56 | Philippine Journal of Science Philippine Agricultural Scientist Fisheries Science Estuarine Coastal and Shelf Science Journal of General and Applied Microbiology Aquaculture Research Journal of Social, Political, And Economic Studies Diseases of Aquatic Organisms Bulletin of Marine Science Environmental Management Fish Pathology Hydrobiologia Journal of General and Applied Microbiology |
| 1995 | 2 | 5.13 | |
| 1997 | 2 | 5.13 | |
| 1998 | 2 | 5.13 | |
| 1999 | 1 | 2.56 | |
| 2000 | 1 | 2.56 | |
| 2001 | 2 | 5.13 | |
| 2002 | 1 | 2.56 | |
| 2003 | 2 | 5.13 | |
| 2004 | 1 | 2.56 | |
| 2005 | 1 | 2.56 | |
| 2006 | 1 | 2.56 | |
| 2007 | 7 | 17.95 | |
| 2008 | 2 | 5.13 | |
| 2009 | 1 | 2.56 | |
| 2011 | 4 | 10.26 | |
| 2012 | 1 | 2.56 | |
| 2014 | 1 | 2.56 | |
| 2015 | 1 | 2.56 | |
| 2017 | 1 | 2.56 | |
| 2018 | 1 | 2.56 | |
| 2020 | 1 | 2.56 | |
| 2021 | 2 | 5.13 | |
| Total | 39 | 100.00 |
| Themes | No. of Studies | % |
|---|---|---|
| Farm management | 12 | 30.77 |
| Disease | 9 | 23.08 |
| Marine pollution | 8 | 20.51 |
| Socioeconomic impacts | 8 | 20.51 |
| Climate change | 2 | 5.13 |
| Total | 39 | 100.00 |
| Disease | Effects | Stage of Culture | References |
|---|---|---|---|
| White Spot Syndrome Virus (WSSV) | First described in Japan, where the initial outbreak occurred in Penaeus japonicus in 1993. Develops rapidly and reaches 100% mortality within 3–10 days. The white inclusion evidently represents the abnormal deposits of calcium salt. | All larval stages of Penaeus monodon | [51,52,53,54,55] |
| Hepatopancreatic Parvo Virus (HPV) | The hepatopancreases of affected shrimp cause abnormal metabolism and eventual death; mortalities may reach 50% within 4–8 weeks. | Juveniles and adults of P. monodon and P. merguiensis | [45] |
| Taura Syndrome Virus (TSV) | First recognized in shrimp farms in Ecuador in 1992 and caused catastrophic losses, with a very high cumulative mortality rate of the affected P. vannamei; includes reddening of the tail fan and visible necrosis in the cuticle. | Postlarvae and broodstock | [28,50,55] |
| Infectious Hypodermal and Hematopoietic Necrosis Virus (IHHNV) | First reported in Hawaii in 1980. The presence of the virus can cause the death of the cell of the cuticle, the blood-forming tissues, and the connective tissues, which causes abnormal metabolism that leads to the mortalities of the shrimp. | All life stages of P. monodon | [45,48,56] |
| Shell Disease | Appearance of brownish-to-black erosion of the carapace, the abdominal segment, and the tail, gills, and appendages. The affected shrimp appendages show a cigarette-butt-like appearance. | All life stages of P. monodon, P. merguiensis, and P. indicus | [28,45] |
| Infectious Myonecrosis Virus (IMNV) | First reported in Hawaii in 1980. Viral occurrences affect the cell nucleus, from the subcuticular epithelium of the mouth appendage, to the gill, the thoracic ganglion, and the nerve fiber of the walking leg, but can also occur sparsely in the cytoplasm. | Postlarvae and broodstock | [21,28] |
| Filamentous Bacterial Disease | Larval shrimp are less prone to infestation than in the postlarval, juvenile, and adult stages because of the rapid succession of molts throughout the different larval stages, while infected eggs show thick mats of filaments on the surfaces that may affect the respiration or hatching. | All life stages of P. monodon, P. merguiensis, and P. indicus | [28,45] |
| Monodon Baculovirus (MBV) | The affected shrimp exhibit pale blue-gray to dark and dark blue-black colorations that cause sluggish and inactive swimming movements, loss of appetite, and retarded growth. The presence of the virus damages the organs, which weakens the shrimp and leads to gradual mortalities. | All life stages of Penaeus monodon and P. merguinsis | [28,57] |
| Luminous Vibriosis | One of the major diseases in grow-out cultured shrimps weakens the larvae and juveniles of the infected shrimp. The larvae become opaque-white, while the juveniles have discolored portions on the body. Systemic infection reaches to 100% of the affected population. | Postlarval stage and broodstock | [58] |
| Environmental Inputs | Chemical Used | Use/Effect |
|---|---|---|
| Pesticide | Saponin (teaseed powder) | Use during pond preparation (broadcast) and rearing phase (periodic); disease control for 30–60 days. |
| Copper compounds | Use during pond preparation (spray) and rearing phase (until phytoplankton bloom). | |
| Potassium Permanganate | Use during pond preparation (spray). | |
| Antibiotics | Tetracycline | Every other day from stocking to harvest. |
| Rifampicin | Disease control daily or until it disappears. | |
| Chloramphenicol | Every other day from stocking to harvest; disease control daily or until it disappears. | |
| Nitrofuran | Every other day from Z1 to harvest; disease control for 3 d (long bath). | |
| Erythromycin | Disease control for 3 d (long bath). | |
| Feeds (Inorganic Fertilizer) | 16-20-0 (monoammonium phosphate) | Use during pond preparation (broadcast) and rearing phase (periodic, broadcast). |
| 18-46-0 (diammonium phosphate) | Use during pond preparation (broadcast). | |
| 14-14-14 (NPK, complete fertilizer) | Rearing phase (periodic, broadcast). | |
| 46-0-0 (urea) | Pond preparation, rearing phase. | |
| 21-0-0 (ammonium sulfate) | Pond preparation, rearing phase. | |
| 0-20-0 (solophos) | Pond preparation, rearing phase. | |
| Organic Fertilizer | Chicken manure | Use during pond preparation, rearing phase (tea bags). |
| Cow manure | Pond preparation, rearing phase (tea bags). | |
| Carabao manure | Pond preparation, rearing phase (tea bags). | |
| VIMACA (chicken/pig manure) | Pond preparation (tea bags). | |
| B-4 | Pond preparation (substitute for manure). | |
| Other Chemicals (Soil and Water Treatment) | Lime | pH control in pond preparation (3–7 days; 20 cm–1.3 m). |
| Dolomite | Pond preparation. | |
| Zeolite | Water quality control, disease control. | |
| Benzalkonium chloride | Water disinfectant. | |
| Oxytetracycline | Control of bacterial diseases. | |
| Teaseed cake | Predator control. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Macusi, E.D.; Estor, D.E.P.; Borazon, E.Q.; Clapano, M.B.; Santos, M.D. Environmental and Socioeconomic Impacts of Shrimp Farming in the Philippines: A Critical Analysis Using PRISMA. Sustainability 2022, 14, 2977. https://doi.org/10.3390/su14052977
Macusi ED, Estor DEP, Borazon EQ, Clapano MB, Santos MD. Environmental and Socioeconomic Impacts of Shrimp Farming in the Philippines: A Critical Analysis Using PRISMA. Sustainability. 2022; 14(5):2977. https://doi.org/10.3390/su14052977
Chicago/Turabian StyleMacusi, Edison D., Darshel Ester P. Estor, Elaine Q. Borazon, Misael B. Clapano, and Mudjekeewis D. Santos. 2022. "Environmental and Socioeconomic Impacts of Shrimp Farming in the Philippines: A Critical Analysis Using PRISMA" Sustainability 14, no. 5: 2977. https://doi.org/10.3390/su14052977
APA StyleMacusi, E. D., Estor, D. E. P., Borazon, E. Q., Clapano, M. B., & Santos, M. D. (2022). Environmental and Socioeconomic Impacts of Shrimp Farming in the Philippines: A Critical Analysis Using PRISMA. Sustainability, 14(5), 2977. https://doi.org/10.3390/su14052977







