Potential Use of Cow Manure for Poly(Lactic Acid) Production
Abstract
1. Introduction
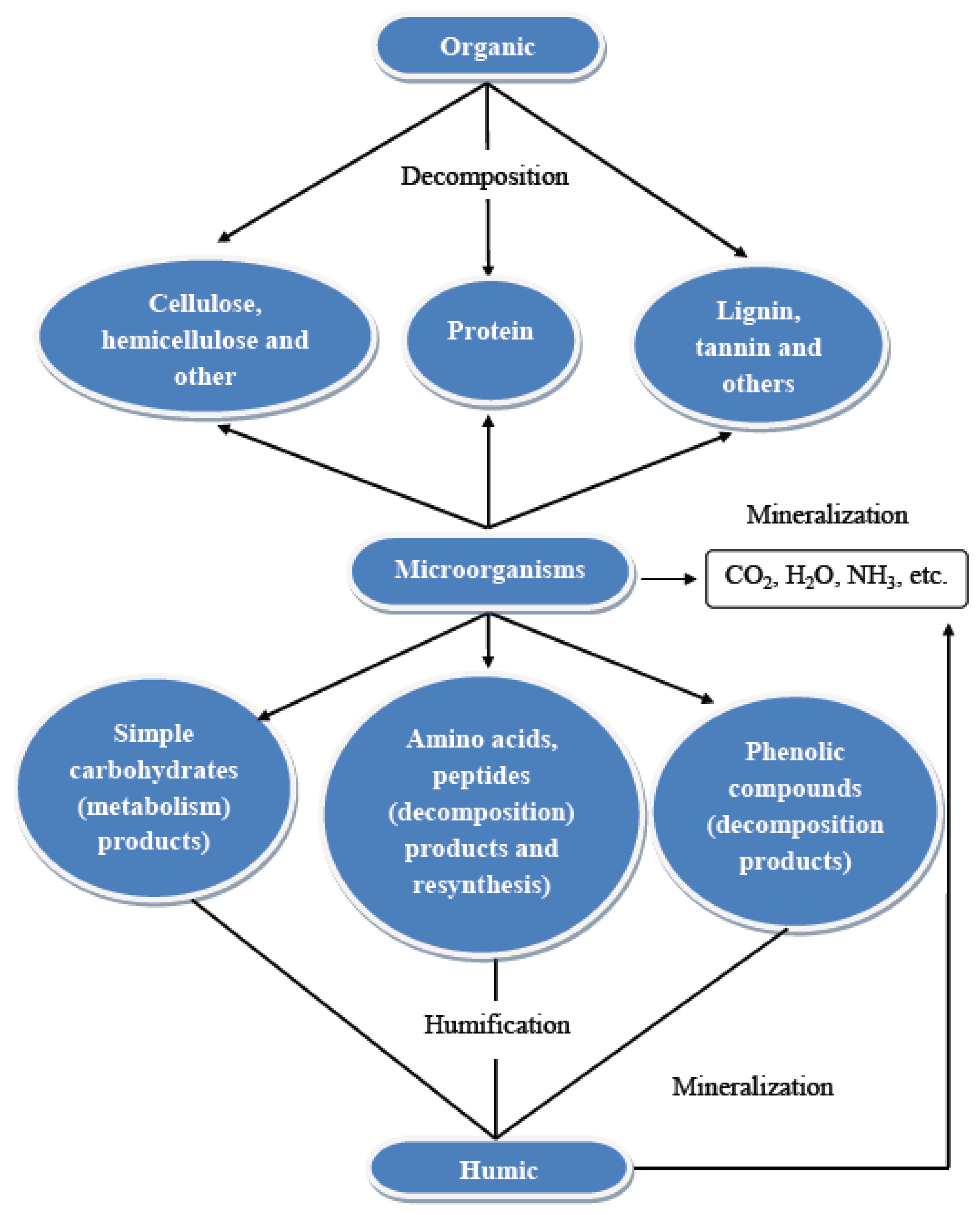
2. Cow Manure Composition
| Dry Matter | Total Nitrogen | N-Ammonia | Phosphorous | |||||
|---|---|---|---|---|---|---|---|---|
| Mean | Range | Mean | Range | Mean | Range | Mean | Range | |
| Liquid manure/slurry | ||||||||
| Cow | 65 | 15–123 | 3.9 | 2.0–7.2 | 2.3 | 1.0–4.9 | 2.3 | 0.2–6.0 |
| Solid manure | ||||||||
| Cow | 223 | 160–430 | 4.8 | 2.0–7.7 | 1.3 | 0.5–2.5 | 3 | 1.0–3.9 |
| Parameter | Farm A | Farm B |
|---|---|---|
| Type of farm | Fattening Cow | Dairy cow |
| Capacity (places) | 850 | 400 |
| Cycles/year | 1.22 | 1 |
| Produced dejections (ton/year) | 1700 | 8525 |
| Dejections | Stable/manure heap | Pit/Pond |
| Nitrogen concentration (kg N/ton) | 11 | 4.8 |
| Cellulose (%) | Hemicellulose (%) | Lignin (%) | Reference |
|---|---|---|---|
| 21.2 | 30.4 | 11.6 | [34] |
| 23.5 | 12.8 | 8.0 | [31] |
| 17.9 | 15.7 | 18.2 | [35] |
| 22.9 | 22.9 | 8.1 | [36] |
| 26.59 | 11.27 | 11.24 | [37] |
| 23.51 | 12.82 | 7.95 | [38] |
| 21.89 | 12.47 | 13.91 | [39] |
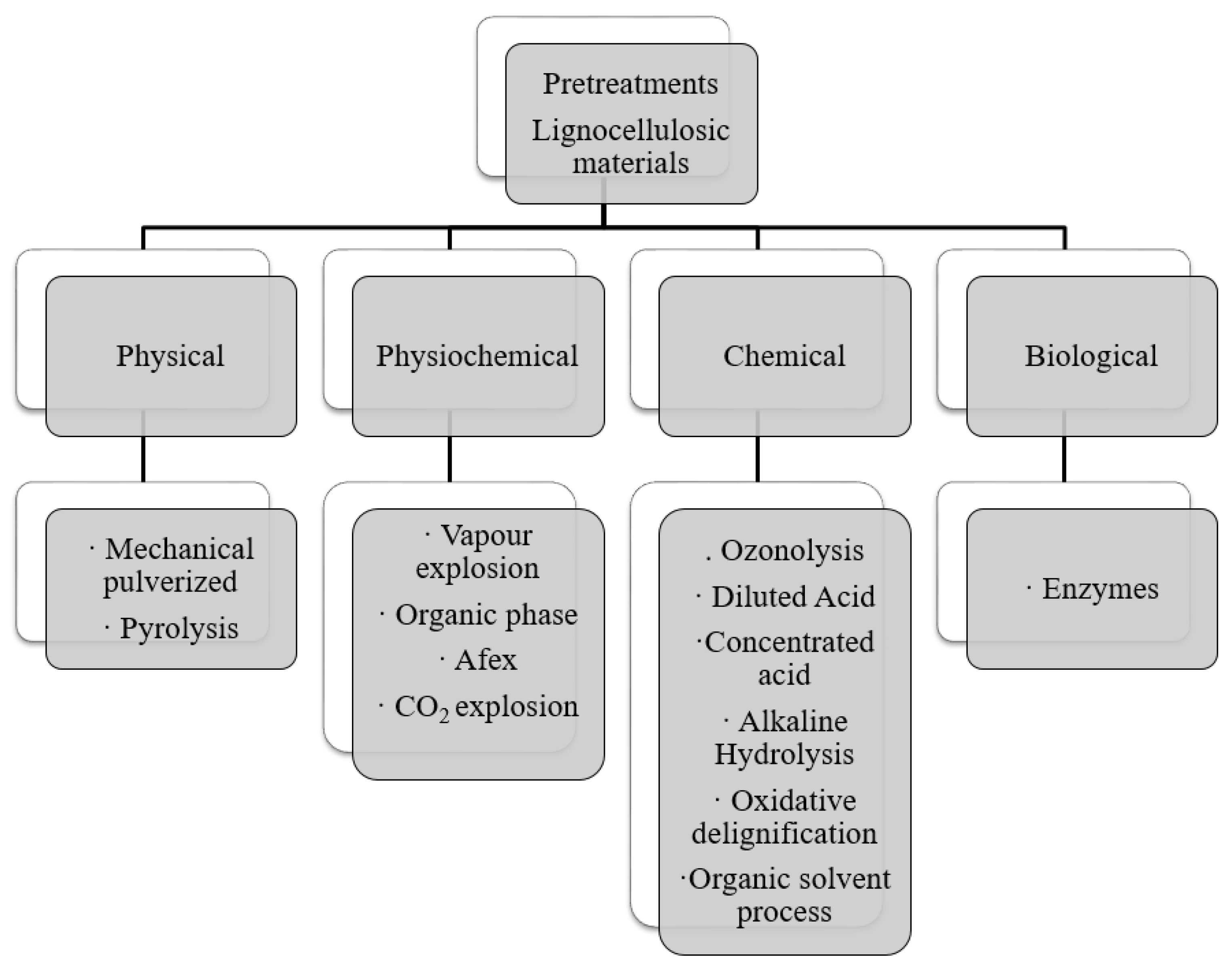
3. Sugar Production from the Lignocellulosic Portion of Cow Manure
3.1. Pretreatment
3.2. Hydrolysis
3.2.1. Acid Hydrolysis
3.2.2. Enzymatic Hydrolysis
4. Microbial Fermented Lactic Acid Production
4.1. Simultaneous Saccharification and Fermentation
4.2. Lactic Acid Recovery
4.3. Purification of Lactic Acid
4.4. Byproduct Formation
5. PLA Production by Lactic Acid Polymerization
PLA Processing
6. Discussion
7. Conclusions
Author Contributions
Funding
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Singhvi, M.; Gokhale, D. Biomass to biodegradable polymer (PLA). RSC Adv. 2013, 3, 13558–13568. [Google Scholar] [CrossRef]
- Jambeck, J.R.; Geyer, R.; Wilcox, C.; Siegler, T.R.; Perryman, M.; Andrady, A.; Narayan, R.; Law, K.L. Plastic waste inputs from land into the ocean. Science 2015, 347, 768–771. [Google Scholar] [CrossRef] [PubMed]
- Lebreton, L.C.M.; van der Zwet, J.; Damsteeg, J.W.; Slat, B.; Andrady, A.; Reisser, J. River plastic emissions to the world’s oceans. Nat. Commun. 2017, 8, 15611. [Google Scholar] [CrossRef]
- Chheda, J.N.; Huber, G.W.; Dumesic, J.A. Liquid-Phase Catalytic Processing of Biomass-Derived Oxygenated Hydrocarbons to Fuels and Chemicals. Angew. Chem. Int. Ed. 2007, 46, 7164–7183. [Google Scholar] [CrossRef] [PubMed]
- Röper, H. Renewable Raw Materials in Europe-Industrial Utilisation of Starch and Sugar. Starch-Stärke 2002, 54, 89–99. [Google Scholar] [CrossRef]
- Maki, M.; Leung, K.T.; Qin, W. The prospects of cellulase-producing bacteria for the bioconversion of lignocellulosic biomass. Int. J. Biol. Sci. 2009, 5, 500–516. [Google Scholar] [CrossRef]
- FAOSTAT. Statistical Yearbook of 2012: Europe and Central Asia; FAO: Rome, Italy, 2012. [Google Scholar]
- Beef2Live. World Cattle Inventory Rankng; Beef2live: Overland Park, AR, USA, 2020. [Google Scholar]
- Boldú, F.P.; Pous, J.P. Guia de les Tecnologies de Tractament de les Dejeccions Ramaderes a Catalunya; Departament d’Agricultura, Ramaderia, Pesca i Alimentació (DARP): Barcelona, Spain, 2020. [Google Scholar]
- Datta, R.; Henry, M. Lactic acid: Recent advances in products, processes and technologies—A review. J. Chem. Technol. Biotechnol. 2006, 81, 1119–1129. [Google Scholar] [CrossRef]
- Datta, R.; Tsai, S.-P.; Bonsignore, P.; Moon, S.-H.; Frank, J.R. Technological and economic potential of poly(lactic acid) and lactic acid derivatives. FEMS Microbiol. Rev. 1995, 16, 221–231. [Google Scholar] [CrossRef]
- Wee, Y.J.; Kim, J.N.; Ryu, H.W. Biotechnological production of lactic acid and its recent applications. Food Technol. Biotechnol. 2006, 44, 163–172. [Google Scholar]
- Joglekar, H.G.; Rahman, I.; Babu, S.; Kulkarni, B.D.; Joshi, A. Comparative assessment of downstream processing options for lactic acid. Sep. Purif. Technol. 2006, 52, 1–17. [Google Scholar] [CrossRef]
- Wang, L.; Zhao, B.; Liu, B.; Yu, B.; Ma, C.; Su, F.; Hua, D.; Li, Q.; Ma, Y.; Xu, P. Efficient production of l-lactic acid from corncob molasses, a waste by-product in xylitol production, by a newly isolated xylose utilizing Bacillus sp. strain. Bioresour. Technol. 2010, 101, 7908–7915. [Google Scholar] [CrossRef]
- Anderson, J.M.; Shive, M.S. Biodegradation and biocompatibility of PLA and PLGA microspheres. Adv. Drug Deliv. Rev. 2012, 64, 72–82. [Google Scholar] [CrossRef]
- Jem, K.J.; Tan, B. The development and challenges of poly (lactic acid) and poly (glycolic acid). Adv. Ind. Eng. Polym. Res. 2020, 3, 60–70. [Google Scholar] [CrossRef]
- Henton, D.E.; Gruber, P.; Lunt, J.; Randall, J. Polylactic Acid Technology. In Natural Fibers, Biopolymers, and Biocomposites; CRC Press: Boca Raton, FL, USA, 2005; pp. 527–578. [Google Scholar]
- Hartmann, M.H. High Molecular Weight Polylactic Acid Polymers. In Biopolymers from Renewable Resources; Springer: Berlin/Heidelberg, Germany, 1998; pp. 367–411. [Google Scholar] [CrossRef]
- D’Adamo, I.; Gastaldi, M.; Morone, P.; Rosa, P.; Sassanelli, C.; Settembre-Blundo, D.; Shen, Y. Bioeconomy of Sustainability: Drivers, Opportunities and Policy Implications. Sustainability 2022, 14, 200. [Google Scholar] [CrossRef]
- Ministerio de la Presidencia, Real Decreto 506, de 28 de Junio, Sobre Productos Fertilizantes, Boletín Oficial del Estado. 2013; pp. 1–24. Available online: https://www.boe.es/boe/dias/2013/07/10/pdfs/BOE-A-2013-7540.pdf (accessed on 7 May 2021).
- Seppälä, M.; Laine, A.; Rintala, J. Screening of novel plants for biogas production in northern conditions. Bioresour. Technol. 2013, 139, 355–362. [Google Scholar] [CrossRef] [PubMed]
- Holm-Nielsen, J.B.; Al Seadi, T.; Oleskowicz-Popiel, P. The future of anaerobic digestion and biogas utilization. Bioresour. Technol. 2009, 100, 5478–5484. [Google Scholar] [CrossRef]
- Burton, C.H. Manure Management–Treatment Strategies for Sustainable Agriculture, second edition. Livest. Sci. 2006, 102, 256–257. [Google Scholar] [CrossRef]
- Burton, C.H.; Beck, J.; Bloxham, P.F.; Derikx, P.J.L.; Martinez, J. Manure Management. Treatment Strategies for Sustainable Agriculture; Silsoe Research Institute: Bedford, UK, 1997; Available online: https://research.wur.nl/en/publications/manure-management-treatment-strategies-for-sustainable-agricultur (accessed on 7 January 2021).
- Rufete Sáez, A.B. Caracterización de Residuos Ganaderos del Sureste Español: Implicaciones Agronómicas y Medioambientales, Universidad Miguel Hernández de Elche. 2015. Available online: http://dspace.umh.es/bitstream/11000/2099/1/TD.pdf (accessed on 6 August 2021).
- Valmaseda, M.; Martínez, A.T.; Almendros, G. Contribution by pigmented fungi to P-type humic acid formation in two forest soils. Soil Biol. Biochem. 1989, 21, 23–28. [Google Scholar] [CrossRef]
- Hassanat, F.; Gervais, R.; Benchaar, C. Methane production, ruminal fermentation characteristics, nutrient digestibility, nitrogen excretion, and milk production of dairy cows fed conventional or brown midrib corn silage. J. Dairy Sci. 2017, 100, 2625–2636. [Google Scholar] [CrossRef]
- Ashekuzzaman, S.M.; Poulsen, T.G. Optimizing feed composition for improved methane yield during anaerobic digestion of cow manure based waste mixtures. Bioresour. Technol. 2011, 102, 2213–2218. [Google Scholar] [CrossRef]
- Díaz, I.; Figueroa-González, I.; Miguel, J.Á.; Bonilla-Morte, L.; Quijano, G. Enhancing the biomethane potential of liquid dairy cow manure by addition of solid manure fractions. Biotechnol. Lett. 2016, 38, 2097–2102. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.-Q.; Zi, L.-H.; Bai, F.-W.; Lin, H.-L.; Hao, X.-M.; Yue, G.-J.; Ho, N.W.Y. Bioethanol from lignocellulosic biomass. Adv. Biochem. Eng. Biotechnol. 2015, 128, 25–51. [Google Scholar] [CrossRef]
- Li, K.; Liu, R.; Sun, C. Comparison of anaerobic digestion characteristics and kinetics of four livestock manures with different substrate concentrations. Bioresour. Technol. 2015, 198, 133–140. [Google Scholar] [CrossRef] [PubMed]
- Casanellas, J.P. Edafología: Uso y Protección de Suelos. 2019. Available online: https://www.mundiprensa.com/catalogo/9788484767503/edafologia--uso-y-proteccion-de-suelos (accessed on 7 January 2021).
- Burton, C.H.; Turner, C. (Eds.) Treatment strategies for livestock manure for sustainable livestock agriculture. In Manure Management: Treatment Strategies for Sustainable Agriculture, 2nd ed.; International Society for Animal Hygiene: Bedford, UK, 2003; p. 490. [Google Scholar]
- Li, R.; Chen, S.; Li, X. Anaerobic co-digestion of kitchen waste and cattle manure for methane Production. Energy Sources Part A Recover. Util. Environ. Eff. 2009, 31, 1848–1856. [Google Scholar] [CrossRef]
- Shen, J.; Zhao, C.; Liu, Y.; Zhang, R.; Liu, G.; Chen, C. Biogas production from anaerobic co-digestion of durian shell with chicken, dairy, and pig manures. Energy Convers. Manag. 2019, 198, 110535. [Google Scholar] [CrossRef]
- Zhao, Y.; Sun, F.; Yu, J.; Cai, Y.; Luo, X.; Cui, Z.; Hu, Y.; Wang, X. Co-digestion of oat straw and cow manure during anaerobic digestion: Stimulative and inhibitory effects on fermentation. Bioresour. Technol. 2018, 269, 143–152. [Google Scholar] [CrossRef]
- Kumar, P.; Barrett, D.M.; Delwiche, M.J.; Stroeve, P. Methods for pretreatment of lignocellulosic biomass for efficient hydrolysis and biofuel production. Ind. Eng. Chem. Res. 2009, 48, 3713–3729. [Google Scholar] [CrossRef]
- Liao, W.; Liu, Y.; Liu, C.; Wen, Z.; Chen, S. Acid hydrolysis of fibers from dairy manure. Bioresour. Technol. 2006, 97, 1687–1695. [Google Scholar] [CrossRef]
- Wen, Z.; Liao, W.; Chen, S. Hydrolysis of animal manure lignocellulosics for reducing sugar production. Bioresour. Technol. 2004, 91, 31–39. [Google Scholar] [CrossRef]
- Yan, Q.; Liu, X.; Wang, Y.; Li, H.; Li, Z.; Zhou, L.; Qu, Y.; Li, Z.; Bao, X. Cow manure as a lignocellulosic substrate for fungal cellulase expression and bioethanol production. AMB Express 2018, 8, 190. [Google Scholar] [CrossRef]
- van Dyk, J.S.; Pletschke, B.I. A review of lignocellulose bioconversion using enzymatic hydrolysis and synergistic cooperation between enzymes-Factors affecting enzymes, conversion and synergy. Biotechnol. Adv. 2012, 30, 1458–1480. [Google Scholar] [CrossRef] [PubMed]
- Howard, R.L.; Abotsi, E.; van Rensburg, E.L.J.; Howard, S. Lignocellulose biotechnology: Issues of bioconversion and enzyme production. Afr. J. Biotechnol. 2003, 2, 602–619. [Google Scholar] [CrossRef]
- Gosselink, R.J.A.; Teunissen, W.; van Dam, J.E.G.; de Jong, E.; Gellerstedt, G.; Scott, E.L.; Sanders, J.P.M. Lignin depolymerisation in supercritical carbon dioxide/acetone/water fluid for the production of aromatic chemicals. Bioresour. Technol. 2012, 106, 173–177. [Google Scholar] [CrossRef] [PubMed]
- Czernik, S.; Bridgwater, A.V. Overview of applications of biomass fast pyrolysis oil. Energy Fuels 2004, 18, 590–598. [Google Scholar] [CrossRef]
- Castro, Y.P. Aprovechamiento de Biomasa Lignocelulósica: Algunas Experiencias de Investigación en Colombia; UTadeo: Bogotá, Colombia, 2014. [Google Scholar]
- Miller, C.; Fosmer, A.; Rush, B.; McMullin, T.; Beacom, D.; Suominen, P. Industrial Production of Lactic Acid, 2nd ed.; Elsevier B.V.: Amsterdam, The Netherlands, 2011. [Google Scholar] [CrossRef]
- Maican, E.; Coz, A.; Ferdeş, M. Continuous Pretreatment Process For Bioethanol Production. In Proceedings of the 4th International Conference on Thermal Equipment, Renewable Energy and Rural Development, Arge County, Romania, 6 June 2015. [Google Scholar]
- Kabel, M.A.; Bos, G.; Zeevalking, J.; Voragen, A.G.J.; Schols, H.A. Effect of pretreatment severity on xylan solubility and enzymatic breakdown of the remaining cellulose from wheat straw. Bioresour. Technol. 2007, 98, 2034–2042. [Google Scholar] [CrossRef] [PubMed]
- Palmqvist, E.; Hahn-Hägerdal, B. Fermentation of lignocellulosic hydrolysates. II: Inhibitors and mechanisms of inhibition. Bioresour. Technol. 2000, 74, 25–33. [Google Scholar] [CrossRef]
- Alvira, P.; Tomás-Pejó, E.; Ballesteros, M.; Negro, M.J. Pretreatment technologies for an efficient bioethanol production process based on enzymatic hydrolysis: A review. Bioresour. Technol. 2010, 101, 4851–4861. [Google Scholar] [CrossRef]
- Taherzadeh, M.J.; Karimi, K. Enzyme-based hydrolysis processes for ethanol from lignocellulosic materials: A review. BioResources 2007, 2, 707–738. [Google Scholar] [CrossRef]
- Taherzadeh, M.J.; Karimi, K. Acid-based hydrolysis processes for ethanol from lignocellulosic materials: A review. BioResources 2007, 2, 472–499. [Google Scholar]
- Yang, B.; Wyman, C.E. Effect of Xylan and Lignin Removal by Batch and Flowthrough Pretreatment on the Enzymatic Digestibility of Corn Stover Cellulose. Biotechnol. Bioeng. 2004, 86, 88–98. [Google Scholar] [CrossRef]
- Wojtusik, M.; Villar, J.C.; Zurita, M.; Ladero, M.; Garcia-Ochoa, F. Study of the enzymatic activity inhibition on the saccharification of acid pretreated corn stover. Biomass Bioenergy 2017, 98, 1–7. [Google Scholar] [CrossRef]
- Taherzadeh, M.; Karimi, K. Pretreatment of Lignocellulosic Wastes to Improve Ethanol and Biogas Production: A Review. Int. J. Mol. Sci. 2008, 9, 1621–1651. [Google Scholar] [CrossRef] [PubMed]
- Garzón, H.; Orozco, D. Hidrólisis Enzimática del Material Lignocelulósico de la Planta de Banano y su Fruto. Bachelor’s Thesis, Escuela de Procesos y Energía, Universidad Nacional de Colombia-Sede Medellín, Medellín, Colombia, 2006. [Google Scholar]
- Hamelinck, C.N.; van Hooijdonk, G.; Faaij, A.P.C. Ethanol from lignocellulosic biomass: Techno-economic performance in short-, middle- and long-term. Biomass Bioenergy 2005, 28, 384–410. [Google Scholar] [CrossRef]
- Pi, Y.; Lozano, J.T. Aprovechamiento de Biomasa Lignocelulósica, Algunas Experiencias de Investigación en Colombia, 2014th ed.; Fundación Universidad de Bogotá Jorge Tadeo Lozano Carrera: Bogotá, Colombia, 2016; Available online: https://www.researchgate.net/publication/279448880%0AAprovechamiento (accessed on 7 April 2021).
- Jung, Y.H.; Kim, I.J.; Kim, J.J.; Oh, K.K.; Han, J.-I.; Choi, I.-G.; Kim, K.H. Ethanol production from oil palm trunks treated with aqueous ammonia and cellulase. Bioresour. Technol. 2011, 102, 7307–7312. [Google Scholar] [CrossRef]
- Kahar, P.; Taku, K.; Tanaka, S. Enzymatic digestion of corncobs pretreated with low strength of sulfuric acid for bioethanol production. J. Biosci. Bioeng. 2010, 110, 453–458. [Google Scholar] [CrossRef]
- Mesa, L.; González, E.; Romero, I.; Ruiz, E.; Cara, C.; Castro, E. Comparison of process configurations for ethanol production from two-step pretreated sugarcane bagasse. Chem. Eng. J. 2011, 175, 185–191. [Google Scholar] [CrossRef]
- Geddes, C.C.; Peterson, J.J.; Roslander, C.; Zacchi, G.; Mullinnix, M.T.; Shanmugam, K.T.; Ingram, L.O. Optimizing the saccharification of sugar cane bagasse using dilute phosphoric acid followed by fungal cellulases. Bioresour. Technol. 2010, 101, 1851–1857. [Google Scholar] [CrossRef]
- Oberoi, H.S.; Sandhu, S.K.; Vadlani, P.V. Statistical optimization of hydrolysis process for banana peels using cellulolytic and pectinolytic enzymes. Food Bioprod. Process. 2012, 90, 257–265. [Google Scholar] [CrossRef]
- Wojtusik, M.; Zurita, M.; Villar, J.C.; Ladero, M.; Garcia-Ochoa, F. Influence of fluid dynamic conditions on enzymatic hydrolysis of lignocellulosic biomass: Effect of mass transfer rate. Bioresour. Technol. 2016, 216, 28–35. [Google Scholar] [CrossRef]
- Djukić-Vuković, A.; Mladenović, D.; Ivanović, J.; Pejin, J.; Mojović, L. Towards sustainability of lactic acid and poly-lactic acid polymers production. Renew. Sustain. Energy Rev. 2019, 108, 238–252. [Google Scholar] [CrossRef]
- Gao, C.; Ma, C.; Xu, P. Biotechnological routes based on lactic acid production from biomass. Biotechnol. Adv. 2011, 29, 930–939. [Google Scholar] [CrossRef] [PubMed]
- Wilhelm, B.J.B.W.; Holzapfel, H. Lactic Acid Bacteria: Biodiversity and Taxonomy; Wiley: New York, NY, USA, 2014. [Google Scholar]
- Chaisu, K.; Charles, A.L.; Guu, Y.-K.; Yen, T.-B.; Chiu, C.-H. Optimization Lactic Acid Production from Molasses Renewable Raw Material through Response Surface Methodology with Lactobacillus Casei M-15. APCBEE Procedia 2014, 8, 194–198. [Google Scholar] [CrossRef]
- Keshwani, D.R.; Cheng, J.J. Switchgrass for bioethanol and other value-added applications: A review. Bioresour. Technol. 2009, 100, 1515–1523. [Google Scholar] [CrossRef]
- Budhavaram, N.K.; Fan, Z. Production of lactic acid from paper sludge using acid-tolerant, thermophilic Bacillus coagulan strains. Bioresour. Technol. 2009, 100, 5966–5972. [Google Scholar] [CrossRef]
- Maas, R.H.W.; Bakker, R.R.; Jansen, M.L.A.; Visser, D.; de Jong, E.; Eggink, G.; Weusthuis, R.A. Lactic acid production from lime-treated wheat straw by Bacillus coagulans: Neutralization of acid by fed-batch addition of alkaline substrate. Appl. Microbiol. Biotechnol. 2008, 78, 751–758. [Google Scholar] [CrossRef]
- van der Pol, E.C.; Eggink, G.; Weusthuis, R.A. Production of l(+)-lactic acid from acid pretreated sugarcane bagasse using Bacillus coagulans DSM2314 in a simultaneous saccharification and fermentation strategy. Biotechnol. Biofuels 2016, 9, 248. [Google Scholar] [CrossRef]
- Wang, Y.; Cao, W.; Luo, J.; Wan, Y. Exploring the potential of lactic acid production from lignocellulosic hydrolysates with various ratios of hexose versus pentose by Bacillus coagulans IPE22. Bioresour. Technol. 2018, 261, 342–349. [Google Scholar] [CrossRef]
- Pleissner, D.; Neu, A.K.; Mehlmann, K.; Schneider, R.; Puerta-Quintero, G.I.; Venus, J. Fermentative lactic acid production from coffee pulp hydrolysate using Bacillus coagulans at laboratory and pilot scales. Bioresour. Technol. 2016, 218, 167–173. [Google Scholar] [CrossRef]
- Åkerberg, C.; Hofvendahl, K.; Zacchi, G.; Hahn-Hägerdal, B. Modelling the influence of pH, temperature, glucose and lactic acid concentrations on the kinetics of lactic acid production by Lactococcus lactis ssp. lactis ATCC 19435 in whole-wheat flour. Appl. Microbiol. Biotechnol. 1998, 49, 682–690. [Google Scholar] [CrossRef]
- Soccol, C.R.; Stonoga, V.I.; Raimbault, M. Production of l-lactic acid by Rhizopus species. World J. Microbiol. Biotechnol. 1994, 10, 433–435. [Google Scholar] [CrossRef] [PubMed]
- Tay, A.; Yang, S.T. Production of L(+)-lactic acid from glucose and starch by immobilized cells of Rhizopus oryzae in a rotating fibrous bed bioreactor. Biotechnol. Bioeng. 2002, 80, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Yun, J.S.; Wee, Y.J.; Ryu, H.W. Production of optically pure L(+)-lactic acid from various carbohydrates by batch fermentation of Enterococcus faecalis RKY1. Enzym. Microb. Technol. 2003, 33, 416–423. [Google Scholar] [CrossRef]
- Dumbrepatil, A.; Adsul, M.; Chaudhari, S.; Khire, J.; Gokhale, D. Utilization of molasses sugar for lactic acid production by Lactobacillus delbrueckii subsp. delbrueckii mutant Uc-3 in batch fermentation. Appl. Environ. Microbiol. 2008, 74, 333–335. [Google Scholar] [CrossRef]
- Wee, Y.-J.; Reddy, L.V.A.; Ryu, H.-W. Fermentative production of L(+)-lactic acid from starch hydrolyzate and corn steep liquor as inexpensive nutrients by batch culture of Enterococcus faecalis RKY1. J. Chem. Technol. Biotechnol. 2008, 83, 1387–1393. [Google Scholar] [CrossRef]
- Wee, Y.J.; Yun, J.S.; Park, D.H.; Ryu, H.W. Biotechnological production of L(+)-lactic acid from wood hydrolyzate by batch fermentation of Enterococcus faecalis. Biotechnol. Lett. 2004, 26, 71–74. [Google Scholar] [CrossRef]
- Abdel-Rahman, M.A.; Tashiro, Y.; Zendo, T.; Shibata, K.; Sonomoto, K. Isolation and characterisation of lactic acid bacterium for effective fermentation of cellobiose into optically pure homo l-(+)-lactic acid. Appl. Microbiol. Biotechnol. 2011, 89, 1039–1049. [Google Scholar] [CrossRef]
- Abdel-Rahman, M.A.; Tashiro, Y.; Zendo, T.; Sonomoto, K. Effective (+)-Lactic Acid Production by Co-fermentation of Mixed Sugars. J. Biotechnol. 2010, 150, 347–348. [Google Scholar] [CrossRef]
- Abdel-Rahman, M.A.; Tashiro, Y.; Zendo, T.; Sonomoto, K. Isolation and characterization of novel lactic acid bacterium for efficient production of l (+)-lactic acid from xylose. J. Biotechnol. 2010, 150, 347. [Google Scholar] [CrossRef]
- Berry, A.R.; Franco, C.M.M.; Zhang, W.; Middelberg, A.P.J. Growth and lactic acid production in batch culture of Lactobacillus rhamnosus in a defined medium. Biotechnol. Lett. 1999, 21, 163–167. [Google Scholar] [CrossRef]
- Marques, S.; Santos, J.A.L.; Gírio, F.M.; Roseiro, J.C. Lactic acid production from recycled paper sludge by simultaneous saccharification and fermentation. Biochem. Eng. J. 2008, 41, 210–216. [Google Scholar] [CrossRef]
- Gullón, B.; Yáñez, R.; Alonso, J.L.; Parajó, J.C. l-Lactic acid production from apple pomace by sequential hydrolysis and fermentation. Bioresour. Technol. 2008, 99, 308–319. [Google Scholar] [CrossRef] [PubMed]
- Romaní, A.; Yáñez, R.; Garrote, G.; Alonso, J.L. SSF production of lactic acid from cellulosic biosludges. Bioresour. Technol. 2008, 99, 4247–4254. [Google Scholar] [CrossRef]
- Iyer, P.V.; Thomas, S.; Lee, Y.Y. High-Yield Fermentation of Pentoses into Lactic Acid. Appl. Biochem. Biotechnol. 2000, 84–86, 665–678. [Google Scholar] [CrossRef] [PubMed]
- Cui, F.; Li, Y.; Wan, C. Lactic acid production from corn stover using mixed cultures of Lactobacillus rhamnosus and Lactobacillus brevis. Bioresour. Technol. 2011, 102, 1831–1836. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Zhao, B.; Liu, B.; Yang, C.; Yu, B.; Li, Q.; Ma, C.; Xu, P.; Ma, Y. Efficient production of l-lactic acid from cassava powder by Lactobacillus rhamnosus. Bioresour. Technol. 2010, 101, 7895–7901. [Google Scholar] [CrossRef]
- Djukić-Vuković, A.P.; Mojović, L.V.; Vukašinović-Sekulić, M.S.; Nikolić, S.B.; Pejin, J.D. Integrated production of lactic acid and biomass on distillery stillage. Bioprocess Biosyst. Eng. 2013, 36, 1157–1164. [Google Scholar] [CrossRef]
- Pejin, J.; Radosavljević, M.; Pribić, M.; Kocić-Tanackov, S.; Mladenović, D.; Djukić-Vuković, A.; Mojović, L. Possibility of L-(+)-lactic acid fermentation using malting, brewing, and oil production by-products. Waste Manag. 2018, 79, 153–163. [Google Scholar] [CrossRef]
- Schepers, A.W.; Thibault, J.; Lacroix, C. Lactobacillus helveticus growth and lactic acid production during pH-controlled batch cultures in whey permeate/yeast extract medium. Part I. Multiple factor kinetic analysis. Enzym. Microb. Technol. 2002, 30, 176–186. [Google Scholar] [CrossRef]
- Burgos-Rubio, C.N.; Okos, M.R.; Wankat, P.C. Kinetic Study of the Conversion of Different Substrates to Lactic Acid Using Lactobacillus bulgaricus. Biotechnol. Prog. 2000, 16, 305–314. [Google Scholar] [CrossRef]
- Liu, P.; Zheng, Z.; Xu, Q.; Qian, Z.; Liu, J.; Ouyang, J. Valorization of dairy waste for enhanced D-lactic acid production at low cost. Process. Biochem. 2018, 71, 18–22. [Google Scholar] [CrossRef]
- Hujanen, M.; Linko, Y.-Y. Effect of temperature and various nitrogen sources on L (+)-lactic acid production by Lactobacillus casei. Appl. Microbiol. Biotechnol. 1996, 45, 307–313. [Google Scholar] [CrossRef]
- Taniguchi, M.; Tokunaga, T.; Horiuchi, K.; Hoshino, K.; Sakai, K.; Tanaka, T. Production of l-lactic acid from a mixture of xylose and glucose by co-cultivation of lactic acid bacteria. Appl. Microbiol. Biotechnol. 2004, 66, 160–165. [Google Scholar] [CrossRef] [PubMed]
- John, R.P.; Madhavan Nampoothiri, K.; Pandey, A. Simultaneous Saccharification and Fermentation of Cassava Bagasse for L-(+)-Lactic Acid Production Using Lactobacilli. Appl. Biochem. Biotechnol. 2006, 134, 263–272. [Google Scholar] [CrossRef] [PubMed]
- Nancib, A.; Nancib, N.; Boudrant, J. Production of lactic acid from date juice extract with free cells of single and mixed cultures of Lactobacillus casei and Lactococcus lactis. World J. Microbiol. Biotechnol. 2009, 25, 1423–1429. [Google Scholar] [CrossRef]
- Kurbanoglu, E.B.; Kurbanoglu, N.I. Utilization for lactic acid production with a new acid hydrolysis of ram horn waste. FEMS Microbiol. Lett. 2003, 225, 29–34. [Google Scholar] [CrossRef]
- Oonkhanond, B.; Jonglertjunya, W.; Srimarut, N.; Bunpachart, P.; Tantinukul, S.; Nasongkla, N.; Sakdaronnarong, C. Lactic acid production from sugarcane bagasse by an integrated system of lignocellulose fractionation, saccharification, fermentation, and ex-situ nanofiltration. J. Environ. Chem. Eng. 2017, 5, 2533–2541. [Google Scholar] [CrossRef]
- Bustos, G.; Moldes, A.B.; Cruz, J.M.; Domínguez, J.M. Production of fermentable media from vine-trimming wastes and bioconversion into lactic acid by Lactobacillus pentosus. J. Sci. Food Agric. 2004, 84, 2105–2112. [Google Scholar] [CrossRef]
- Moldes, A.B.; Torrado, A.; Converti, A.; Domínguez, J.M. Complete bioconversion of hemicellulosic sugars from agricultural residues into lactic acid by Lactobacillus pentosus. Appl. Biochem. Biotechnol. 2006, 135, 219–228. [Google Scholar] [CrossRef]
- Garde, A.; Jonsson, G.; Schmidt, A.S.; Ahring, B.K. Lactic acid production from wheat straw hemicellulose hydrolysate by Lactobacillus pentosus and Lactobacillus brevis. Bioresour. Technol. 2002, 81, 217–223. [Google Scholar] [CrossRef]
- Zhu, Y.; Lee, Y.Y.; Elander, R.T. Conversion of aqueous ammonia-treated corn stover to lactic acid by simultaneous saccharification and cofermentation. Appl. Biochem. Biotechnol. 2007, 137–140, 721–738. [Google Scholar] [CrossRef]
- Vishnu, C.; Seenayya, G.; Reddy, G. Direct fermentation of various pure and crude starchy substrates to L(+) lactic acid using Lactobacillus amylophilus GV6. World J. Microbiol. Biotechnol. 2002, 18, 429–433. [Google Scholar] [CrossRef]
- Wee, Y.-J.; Ryu, H.-W. Lactic acid production by Lactobacillus sp. RKY2 in a cell-recycle continuous fermentation using lignocellulosic hydrolyzates as inexpensive raw materials. Bioresour. Technol. 2009, 100, 4262–4270. [Google Scholar] [CrossRef] [PubMed]
- Yun, J.-S.; Wee, Y.-J.; Kim, J.-N.; Ryu, H.-W. Fermentative production of dl-lactic acid from amylase-treated rice and wheat brans hydrolyzate by a novel lactic acid bacterium, Lactobacillus sp. Biotechnol. Lett. 2004, 26, 1613–1616. [Google Scholar] [CrossRef] [PubMed]
- Givry, S.; Prevot, V.; Duchiron, F. Lactic acid production from hemicellulosic hydrolyzate by cells of Lactobacillus bifermentans immobilized in Ca-alginate using response surface methodology. World J. Microbiol. Biotechnol. 2008, 24, 745–752. [Google Scholar] [CrossRef]
- Singhvi, M.; Joshi, D.; Adsul, M.; Varma, A.; Gokhale, D. d-(−)-Lactic acid production from cellobiose and cellulose by Lactobacillus lactis mutant RM2-24. Green Chem. 2010, 12, 1106–1109. [Google Scholar] [CrossRef]
- Joshi, D.S.; Singhvi, M.S.; Khire, J.M.; Gokhale, D.V. Strain improvement of Lactobacillus lactis for d-lactic acid production. Biotechnol. Lett. 2010, 32, 517–520. [Google Scholar] [CrossRef]
- Laopaiboon, P.; Thani, A.; Leelavatcharamas, V.; Laopaiboon, L. Acid hydrolysis of sugarcane bagasse for lactic acid production. Bioresour. Technol. 2010, 101, 1036–1043. [Google Scholar] [CrossRef]
- Roble, N.D.; Ogbonna, J.C.; Tanaka, H. L-lactic acid production from raw cassava starch in a circulating loop bioreactor with cells immobilized in loofa (Luffa cylindrica). Biotechnol. Lett. 2003, 25, 1093–1098. [Google Scholar] [CrossRef]
- Tanaka, K.; Komiyama, A.; Sonomoto, K.; Ishizaki, A.; Hall, S.; Stanbury, P. Two different pathways for D-xylose metabolism and the effect of xylose concentration on the yield coefficient of L-lactate in mixed-acid fermentation by the lactic acid bacterium Lactococcus lactis IO-1. Appl. Microbiol. Biotechnol. 2002, 60, 160–167. [Google Scholar] [CrossRef]
- Hofvendahl, K.; Hahn-Hägerdal, B. l-lactic acid production from whole wheat flour hydrolysate using strains of Lactobacilli and Lactococci. Enzym. Microb. Technol. 1997, 20, 301–307. [Google Scholar] [CrossRef]
- Hofvendahl, K.; Hahn-Hägerdal, B.; Åkerberg, C.; Zacchi, G. Simultaneous enzymatic wheat starch saccharification and fermentation to lactic acid by Lactococcus lactis. Appl. Microbiol. Biotechnol. 1999, 52, 163–169. [Google Scholar] [CrossRef]
- Wang, L.; Zhao, B.; Li, F.; Xu, K.; Ma, C.; Tao, F.; Li, Q.; Xu, P. Highly efficient production of d-lactate by Sporolactobacillus sp. CASD with simultaneous enzymatic hydrolysis of peanut meal. Appl. Microbiol. Biotechnol. 2011, 89, 1009–1017. [Google Scholar] [CrossRef]
- Li, Y.; Wang, L.; Ju, J.; Yu, B.; Ma, Y. Efficient production of polymer-grade d-lactate by Sporolactobacillus laevolacticus DSM442 with agricultural waste cottonseed as the sole nitrogen source. Bioresour. Technol. 2013, 142, 186–191. [Google Scholar] [CrossRef] [PubMed]
- Guo, W.; Jia, W.; Li, Y.; Chen, S. Performances of Lactobacillus brevis for producing lactic acid from hydrolysate of Lignocellulosics. Appl. Biochem. Biotechnol. 2010, 161, 124–136. [Google Scholar] [CrossRef] [PubMed]
- Yáñez, R.; Moldes, A.B.; Alonso, J.L.; Parajó, J.C. Production of D(-)-lactic acid from cellulose by simultaneous saccharification and fermentation using Lactobacillus coryniformis subsp. torquens. Biotechnol. Lett. 2003, 25, 1161–1164. [Google Scholar] [CrossRef]
- Yáñez, R.; Alonso, J.L.; Parajó, J.C. D-Lactic acid production from waste cardboard. J. Chem. Technol. Biotechnol. 2005, 80, 76–84. [Google Scholar] [CrossRef]
- Berlowska, J.; Cieciura-Wloch, W.; Kalinowska, H.; Kregiel, D.; Borowski, S.; Pawlikowska, E.; Binczarski, M.; Witonska, I. Enzymatic Conversion of Sugar Beet Pulp: A Comparison of Simultaneous Saccharification and Fermentation and Separate Hydrolysis and Fermentation for Lactic Acid Production. Food Technol. Biotechnol. 2018, 56, 188–196. [Google Scholar] [CrossRef]
- de Oliveira Moraes, A.; Ramirez, N.I.B.; Pereira, N. Evaluation of the Fermentation Potential of Pulp Mill Residue to Produce d(−)-Lactic Acid by Separate Hydrolysis and Fermentation Using Lactobacillus coryniformis subsp. torquens. Appl. Biochem. Biotechnol. 2016, 180, 1574–1585. [Google Scholar] [CrossRef]
- Fu, W.; Mathews, A.P. Lactic acid production from lactose by Lactobacillus plantarum: Kinetic model and effects of pH, substrate, and oxygen. Biochem. Eng. J. 1999, 3, 163–170. [Google Scholar] [CrossRef]
- Sreenath, H.K.; Moldes, A.B.; Koegel, R.G.; Straub, R.J. Lactic acid production by simultaneous saccharification and fermentation of alfalfa fiber. J. Biosci. Bioeng. 2001, 92, 518–523. [Google Scholar] [CrossRef] [PubMed]
- Okano, K.; Zhang, Q.; Yoshida, S.; Tanaka, T.; Ogino, C.; Fukuda, H.; Kondo, A. D-lactic acid production from cellooligosaccharides and β-glucan using l-LDH gene-deficient and endoglucanase-secreting Lactobacillus plantarum. Appl. Microbiol. Biotechnol. 2010, 85, 643–650. [Google Scholar] [CrossRef] [PubMed]
- Okano, K.; Yoshida, S.; Tanaka, T.; Ogino, C.; Fukuda, H.; Kondo, A. Homo-D-Lactic Acid Fermentation from Arabinose by Redirection of the Phosphoketolase Pathway to the Pentose Phosphate Pathway in L-Lactate Dehydrogenase Gene-Deficient Lactobacillus plantarum. Appl. Environ. Microbiol. 2009, 75, 5175–5178. [Google Scholar] [CrossRef] [PubMed]
- Okano, K.; Yoshida, S.; Yamada, R.; Tanaka, T.; Ogino, C.; Fukuda, H.; Kondo, A. Improved production of homo-D-lactic acid via xylose fermentation by introduction of xylose assimilation genes and redirection of the phosphoketolase pathway to the pentose phosphate pathway in L-lactate dehydrogenase gene-deficient Lactobacillus plantarum. Appl. Environ. Microbiol. 2009, 75, 7858–7861. [Google Scholar] [CrossRef] [PubMed]
- Ohara, H.; Owaki, M.; Sonomoto, K. Xylooligosaccharide fermentation with Leuconostoc lactis. J. Biosci. Bioeng. 2006, 101, 415–420. [Google Scholar] [CrossRef] [PubMed]
- Patel, M.A.; Ou, M.S.; Ingram, L.O.; Shanmugam, K.T. Simultaneous saccharification and co-fermentation of crystalline cellulose and sugar cane bagasse hemicellulose hydrolysate to lactate by a thermotolerant acidophilic Bacillus sp. Biotechnol. Prog. 2005, 21, 1453–1460. [Google Scholar] [CrossRef] [PubMed]
- Montelongo, J.-L.; Chassy, B.M.; McCORD, J.D. Lactobacillus salivarius for Conversion of Soy Molasses into Lactic Acid. J. Food Sci. 1993, 58, 863–866. [Google Scholar] [CrossRef]
- Timbuntam, W.; Sriroth, K.; Tokiwa, Y. Lactic acid production from sugar-cane juice by a newly isolated Lactobacillus sp. Biotechnol. Lett. 2006, 28, 811–814. [Google Scholar] [CrossRef]
- Xiaodong, W.; Xuan, G.; Rakshit, S.K. Direct fermentative production of lactic acid on cassava and other starch substrates. Biotechnol. Lett. 1997, 19, 841–843. [Google Scholar] [CrossRef]
- Coelho, L.F.; de Lima, C.J.B.; Bernardo, M.P.; Contiero, J. d(−)-Lactic Acid Production by Leuconostoc mesenteroides B512 Using Different Carbon and Nitrogen Sources. Appl. Biochem. Biotechnol. 2011, 164, 1160–1171. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Chen, C.; Cai, D.; Wang, Z.; Qin, P.; Tan, T. The optimization of L-lactic acid production from sweet sorghum juice by mixed fermentation of Bacillus coagulans and Lactobacillus rhamnosus under unsterile conditions. Bioresour. Technol. 2016, 218, 1098–1105. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Li, K.; Huang, F.; Wang, J.; Zhao, J.; Zhao, X.; Garza, E.; Manow, R.; Grayburn, S.; Zhou, S. Engineering and adaptive evolution of Escherichia coli W for l-lactic acid fermentation from molasses and corn steep liquor without additional nutrients. Bioresour. Technol. 2013, 148, 394–400. [Google Scholar] [CrossRef] [PubMed]
- Richter, K.; Träger, A. L(+)-Lactic acid from sweet sorghum by submerged and solid-state fermentations. Acta Biotechnol. 1994, 14, 367–378. [Google Scholar] [CrossRef]
- Mladenović, D.; Pejin, J.; Kocić-Tanackov, S.; Djukić-Vuković, A.; Mojović, L. Enhanced Lactic Acid Production by Adaptive Evolution of Lactobacillus paracasei on Agro-industrial Substrate. Appl. Biochem. Biotechnol. 2019, 187, 753–769. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, C.M.; Kim, J.S.; Nguyen, T.N.; Kim, S.K.; Choi, G.J.; Choi, Y.H.; Jang, K.S.; Kim, J.C. Production of l- and d-lactic acid from waste Curcuma longa biomass through simultaneous saccharification and cofermentation. Bioresour. Technol. 2013, 146, 35–43. [Google Scholar] [CrossRef]
- Flores-Albino, B.; Arias, L.; Gómez, J.; Castillo, A.; Gimeno, M.; Shirai, K. Chitin and L(+)-lactic acid production from crab (Callinectes bellicosus) wastes by fermentation of Lactobacillus sp. B2 using sugar cane molasses as carbon source. Bioprocess Biosyst. Eng. 2012, 35, 1193–1200. [Google Scholar] [CrossRef]
- Taskin, M.; Esim, N.; Ortucu, S. Efficient production of l-lactic acid from chicken feather protein hydrolysate and sugar beet molasses by the newly isolated Rhizopus oryzae TS-61. Food Bioprod. Process. 2012, 90, 773–779. [Google Scholar] [CrossRef]
- Zhang, L.; Li, X.; Yong, Q.; Yang, S.T.; Ouyang, J.; Yu, S. Simultaneous saccharification and fermentation of xylo-oligosaccharides manufacturing waste residue for l-lactic acid production by Rhizopus oryzae. Biochem. Eng. J. 2015, 94, 92–99. [Google Scholar] [CrossRef]
- Zheng, Y.; Wang, Y.; Zhang, J.; Pan, J. Using tobacco waste extract in pre-culture medium to improve xylose utilization for l-lactic acid production from cellulosic waste by Rhizopus oryzae. Bioresour. Technol. 2016, 218, 344–350. [Google Scholar] [CrossRef]
- Tanaka, T.; Hoshina, M.; Tanabe, S.; Sakai, K.; Ohtsubo, S.; Taniguchi, M. Production of d-lactic acid from defatted rice bran by simultaneous saccharification and fermentation. Bioresour. Technol. 2006, 97, 211–217. [Google Scholar] [CrossRef]
- Kotzamanidis, C.; Roukas, T.; Skaracis, G. Optimization of lactic acid production from beet molasses by Lactobacillus delbrueckii NCIMB 8130. World J. Microbiol. Biotechnol. 2002, 18, 441–448. [Google Scholar] [CrossRef]
- Adsul, M.; Khire, J.; Bastawde, K.; Gokhale, D. Production of Lactic Acid from Cellobiose and Cellotriose by Lactobacillus delbrueckii Mutant Uc-3. Appl. Environ. Microbiol. 2007, 73, 5055–5057. [Google Scholar] [CrossRef] [PubMed]
- Adsul, M.G.; Varma, A.J.; Gokhale, D.V. Lactic acid production from waste sugarcane bagasse derived cellulose. Green Chem. 2007, 9, 58–62. [Google Scholar] [CrossRef]
- Calabia, B.P.; Tokiwa, Y. Production of d-lactic acid from sugarcane molasses, sugarcane juice and sugar beet juice by Lactobacillus delbrueckii. Biotechnol. Lett. 2007, 29, 1329–1332. [Google Scholar] [CrossRef] [PubMed]
- Iyer, P.V.; Lee, Y.Y. Product inhibition in simultaneous saccharification and fermentation of cellulose into lactic acid. Biotechnol. Lett. 1999, 21, 371–373. [Google Scholar] [CrossRef]
- Iyer, P.V.; Lee, Y.Y. Simultaneous Saccharification and Extractive Fermentation of Lignocellulosic Materials into Lactic Acid in a Two-Zone Fermentor-Extractor System. In Twentieth Symposium on Biotechnology for Fuels and Chemicals; Humana Press: Totowa, NJ, USA, 1999; pp. 409–419. [Google Scholar] [CrossRef]
- Mussatto, S.I.; Fernandes, M.; Mancilha, I.M.; Roberto, I.C. Effects of medium supplementation and pH control on lactic acid production from brewer’s spent grain. Biochem. Eng. J. 2008, 40, 437–444. [Google Scholar] [CrossRef]
- Shen, X.; Xia, L. Lactic acid production from cellulosic material by synergetic hydrolysis and Fermentation. Appl. Biochem. Biotechnol. 2006, 133, 251–262. [Google Scholar] [CrossRef]
- Lu, Z.; Lu, M.; He, F.; Yu, L. An economical approach for d-lactic acid production utilizing unpolished rice from aging paddy as major nutrient source. Bioresour. Technol. 2009, 100, 2026–2031. [Google Scholar] [CrossRef]
- Tsai, T.S.; Millard, C.S. Improved Pre-treatment Process for Lactic Acid Production. WO/1994/013826, 23 June 1994. Available online: https://patentscope.wipo.int/search/en/detail.jsf?docId=WO1994013826 (accessed on 6 April 2021).
- Aksu, Z.; Kutsal, T. Lactic acid production from molasses utilizing Lactobacillus delbrueckii and invertase together. Biotechnol. Lett. 1986, 8, 157–160. [Google Scholar] [CrossRef]
- Balakrishnan, R.; Tadi, S.R.R.; Sivaprakasam, S.; Rajaram, S. Optimization of acid and enzymatic hydrolysis of kodo millet (Paspalum scrobiculatum) bran residue to obtain fermentable sugars for the production of optically pure D (−) lactic acid. Ind. Crops Prod. 2018, 111, 731–742. [Google Scholar] [CrossRef]
- Qiu, Z.; Gao, Q.; Bao, J. Engineering Pediococcus acidilactici with xylose assimilation pathway for high titer cellulosic L-lactic acid fermentation. Bioresour. Technol. 2018, 249, 9–15. [Google Scholar] [CrossRef] [PubMed]
- Bai, Z.; Gao, Z.; He, B.; Wu, B. Effect of lignocellulose-derived inhibitors on the growth and d-lactic acid production of Sporolactobacillus inulinus YBS1-5. Bioprocess Biosyst. Eng. 2015, 38, 1993–2001. [Google Scholar] [CrossRef] [PubMed]
- John, R.P.; Nampoothiri, K.M.; Pandey, A. Fermentative production of lactic acid from biomass: An overview on process developments and future perspectives. Appl. Microbiol. Biotechnol. 2007, 74, 524–534. [Google Scholar] [CrossRef] [PubMed]
- Van Der Pol, E.C. Development of a Lactic Acid Production Process Using Lignocellulosic Biomass as Feedstock. 2016. Available online: https://edepot.wur.nl/374060 (accessed on 5 March 2021).
- Balat, M. Production of bioethanol from lignocellulosic materials via the biochemical pathway: A review. Energy Convers. Manag. 2011, 52, 858–875. [Google Scholar] [CrossRef]
- Barre, P. Identification of Thermobacteria and Homofermentative, Thermophilic, Pentose-utilizing Lactobacilli from High Temperature Fermenting Grape Musts. J. Appl. Bacteriol. 1978, 44, 125–129. [Google Scholar] [CrossRef]
- Hemme, D.; Raibaud, P.; Ducluzeau, R.; Galpin, J.V.; Sicard, P.; van Heyenoort, J. Lactobacillus murinus’ n.sp., a new species of the autochthonous dominant flora of the digestive tract of rat and mouse. Ann. Inst. Pasteur 1980, 131, 297–308. Available online: https://hal.inrae.fr/hal-02732519 (accessed on 11 September 2021).
- Olofsson, K.; Bertilsson, M.; Lidén, G. A short review on SSF—An interesting process option for ethanol production from lignocellulosic feedstocks. Biotechnol. Biofuels 2008, 1, 7. [Google Scholar] [CrossRef]
- Abdel-Rahman, M.A.; Tashiro, Y.; Sonomoto, K. Lactic acid production from lignocellulose-derived sugars using lactic acid bacteria: Overview and limits. J. Biotechnol. 2011, 156, 286–301. [Google Scholar] [CrossRef]
- Biddy, M.J.; Scarlata, C.; Kinchin, C. Chemicals from Biomass: A Market Assessment of Bioproducts with Near-Term Potential; NREL: Golden, CO, USA, 2016. [Google Scholar] [CrossRef]
- Komesu, A.; Martins, P.F.; Lunelli, B.H.; Rocha, J.O.; Filho, R.M.; Maciel, M.R.W. The Effect of Evaporator Temperature on Lactic Acid Purity and Recovery by Short Path Evaporation. Sep. Sci. Technol. 2014, 50, 1548–1553. [Google Scholar] [CrossRef]
- Järvinen, M.; Myllykoski, L.; Keiski, R.; Sohlo, J. Separation of lactic acid from fermented broth by reactive extraction. Bioseparation 2000, 9, 163–166. [Google Scholar] [CrossRef]
- Komesu, A.; Maciel, M.R.W.; Filho, R.M. Separation and Purification Technologies for Lactic Acid—A Brief Review. BioResources 2017, 12, 6885–6901. [Google Scholar] [CrossRef]
- Komesu, A.; Maciel, M.R.W.; de Oliveira, J.A.R.; da Silva Martins, L.H.; Maciel Filho, R. Purification of Lactic Acid Produced by Fermentation: Focus on Non-traditional Distillation Processes. Sep. Purif. Rev. 2017, 46, 241–254. [Google Scholar] [CrossRef]
- Din, N.A.S.; Lim, S.J.; Maskat, M.Y.; Mutalib, S.A.; Zaini, N.A.M. Lactic acid separation and recovery from fermentation broth by ion-exchange resin: A review. Bioresour. Bioprocess. 2021, 8, 31. [Google Scholar] [CrossRef]
- Yankov, D.; Molinier, J.; Kyuchoukov, G.; Albet, J.; Malmary, G. Improvement of the lactic acid extraction. Extraction from aqueous solutions and simulated fermentation broth by means of mixed extractant and TOA, partially loaded with HCI. Chem. Biochem. Eng. Q. 2005, 19, 17–24. [Google Scholar]
- Daful, A.G.; Haigh, K.; Vaskan, P.; Görgens, J.F. Environmental impact assessment of lignocellulosic lactic acid production: Integrated with existing sugar mills. Food Bioprod. Process. 2016, 99, 58–70. [Google Scholar] [CrossRef]
- Wasewar, K.L.; Pangarkar, V.G.; Heesink, A.B.M.; Versteeg, G.F. Intensification of enzymatic conversion of glucose to lactic acid by reactive extraction. Chem. Eng. Sci. 2003, 58, 3385–3393. [Google Scholar] [CrossRef]
- Henczka, M.; Djas, M. Reactive extraction of acetic acid and propionic acid using supercritical carbon dioxide. J. Supercrit. Fluids 2016, 110, 154–160. [Google Scholar] [CrossRef]
- López-Garzón, C.S.; Straathof, A.J.J. Recovery of carboxylic acids produced by fermentation. Biotechnol. Adv. 2014, 32, 873–904. [Google Scholar] [CrossRef]
- Kumar, A.; Thakur, A.; Panesar, P.S. Lactic acid and its separation and purification techniques: A review. Rev. Environ. Sci. Bio/Technology 2019, 18, 823–853. [Google Scholar] [CrossRef]
- Wojtyniak, B.; Kołodziejczyk, J.; Szaniawska, D. Production of lactic acid by ultrafiltration of fermented whey obtained in bioreactor equipped with ZOSS membrane. Chem. Eng. J. 2016, 305, 28–36. [Google Scholar] [CrossRef]
- Aqar, D.Y.; Rahmanian, N.; Mujtaba, I.M. Integrated batch reactive distillation column configurations for optimal synthesis of methyl lactate. Chem. Eng. Process. Process Intensif. 2016, 108, 197–211. [Google Scholar] [CrossRef]
- Nielsen, D.R.; Amarasiriwardena, G.S.; Prather, K.L. Predicting the adsorption of second generation biofuels by polymeric resins with applications for in situ product recovery (ISPR). Bioresour. Technol. 2010, 101, 2762–2769. [Google Scholar] [CrossRef] [PubMed]
- Pradhan, N.; Rene, E.R.; Lens, P.N.L.; Dipasquale, L.; D’Ippolito, G.; Fontana, A.; Panico, A.; Esposito, G. Adsorption behaviour of lactic acid on granular activated carbon and anionic resins: Thermodynamics, isotherms and kinetic Studies. Energies 2017, 10, 665. [Google Scholar] [CrossRef]
- Seeber, G.; Buchmeiser, M.R.; Bonn, G.K.; Bertsch, T. Determination of airborne, volatile amines from polyurethane foams by sorption onto a high-capacity cation-exchange resin based on poly(succinic acid). J. Chromatogr. A 1998, 809, 121–129. [Google Scholar] [CrossRef]
- Kumar, S.; Jain, S. History, introduction, and kinetics of ion exchange Materials. J. Chem. 2013, 2013, 957647. [Google Scholar] [CrossRef]
- Aljundi, I.H.; Belovich, J.M.; Talu, O. Adsorption of lactic acid from fermentation broth and aqueous solutions on Zeolite molecular sieves. Chem. Eng. Sci. 2005, 60, 5004–5009. [Google Scholar] [CrossRef]
- Boonmee, M.; Cotano, O.; Amnuaypanich, S.; Grisadanurak, N. Improved Lactic Acid Production by In Situ Removal of Lactic Acid During Fermentation and a Proposed Scheme for Its Recovery. Arab. J. Sci. Eng. 2016, 41, 2067–2075. [Google Scholar] [CrossRef]
- Zhang, Y.; Qian, Z.; Liu, P.; Liu, L.; Zheng, Z.; Ouyang, J. Efficient in situ separation and production of l-lactic acid by Bacillus coagulans using weak basic anion-exchange resin. Bioprocess Biosyst. Eng. 2018, 41, 205–212. [Google Scholar] [CrossRef]
- Quintero, J.; Acosta, A.; Mejía, C.; Ríos, R.; Torres, A.M. Purification of lactic acid obtained from a fermentative process of cassava syrup using ion exchange resins. Rev. Fac. Ing. Univ. Antioq. 2012, 65, 139–151. [Google Scholar]
- Kuo, Y.C.; Yuan, S.F.; Wang, C.A.; Huang, Y.J.; Guo, G.L.; Hwang, W.S. Production of optically pure l-lactic acid from lignocellulosic hydrolysate by using a newly isolated and d-lactate dehydrogenase gene-deficient Lactobacillus paracasei strain. Bioresour. Technol. 2015, 198, 651–657. [Google Scholar] [CrossRef]
- Peckham, G.T. The Commercial Manufacture of Lactic Acid. Chem. Eng. News 1944, 22, 440–443, 469. [Google Scholar] [CrossRef]
- Inskeep, G.C.; Taylor, G.G.; Breitzke, W.C. LACTIC ACID FROM CORN SUGAR. Ind. Eng. Chem. 1952, 44, 1955–1966. [Google Scholar] [CrossRef]
- Qin, J.; Wang, X.; Zheng, Z.; Ma, C.; Tang, H.; Xu, P. Production of L-lactic acid by a thermophilic Bacillus mutant using sodium hydroxide as neutralizing agent. Bioresour. Technol. 2010, 101, 7570–7576. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.-C.; Ju, L.-K. Adsorption Characteristics of Polyvinylpyridine and Activated Carbon for Lactic Acid Recovery from Fermentation of Lactobacillus delbrueckii. Sep. Sci. Technol. 1998, 33, 1423–1437. [Google Scholar] [CrossRef]
- Kumar, R.; Nanavati, H.; Noronha, S.B.; Mahajani, S.M. A continuous process for the recovery of lactic acid by reactive distillation. J. Chem. Technol. Biotechnol. 2006, 81, 1767–1777. [Google Scholar] [CrossRef]
- Madzingaidzo, L.; Danner, H.; Braun, R. Process development and optimisation of lactic acid purification using electrodialysis. J. Biotechnol. 2002, 96, 223–239. [Google Scholar] [CrossRef]
- Choi, J.H.; Kim, S.H.; Moon, S.H. Recovery of lactic acid from sodium lactate by ion substitution using ion-exchange membrane. Sep. Purif. Technol. 2002, 28, 69–79. [Google Scholar] [CrossRef]
- Hábová, V.; Melzoch, K.; Rychtera, M.; Přibyl, L.; Mejta, V. Application of electrodialysis for lactic acid recovery. Czech J. Food Sci. 2013, 19, 73–80. [Google Scholar] [CrossRef]
- Kim, Y.H.; Moon, S.H. Lactic acid recovery from fermentation broth using one-stage electrodialysis. J. Chem. Technol. Biotechnol. 2001, 76, 169–178. [Google Scholar] [CrossRef]
- Yebo Li, A.S. Lactic Acid Recovery From Cheese Whey Fermentation Broth Using Combined Ultrafiltration and Nanofiltration Membranes. Appl. Biochem. Biotechnol. 2006, 132, 985–996. [Google Scholar] [CrossRef]
- González, M.I.; Alvarez, S.; Riera, F.A.; Álvarez, R. Lactic acid recovery from whey ultrafiltrate fermentation broths and artificial solutions by nanofiltration. Desalination 2008, 228, 84–96. [Google Scholar] [CrossRef]
- Li, Y.; Shahbazi, A.; Williams, K.; Wan, C. Separate and Concentrate Lactic Acid Using Combination of Nanofiltration and Reverse Osmosis Membranes. Appl. Biochem. Biotechnol. 2007, 147, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Zhao, K.; Qiao, Q.; Chu, D.; Gu, H.; Dao, T.H.; Zhang, J.; Bao, J. Simultaneous saccharification and high titer lactic acid fermentation of corn stover using a newly isolated lactic acid bacterium Pediococcus acidilactici DQ2. Bioresour. Technol. 2013, 135, 481–489. [Google Scholar] [CrossRef] [PubMed]
- van der Pol, E.; Bakker, R.; van Zeeland, A.; Garcia, D.S.; Punt, A.; Eggink, G. Analysis of by-product formation and sugar monomerization in sugarcane bagasse pretreated at pilot plant scale: Differences between autohydrolysis, alkaline and acid pretreatment. Bioresour. Technol. 2015, 181, 114–123. [Google Scholar] [CrossRef] [PubMed]
- Kaščak, J.; Gašpár, Š.; Paško, J.; Husár, J.; Knapčíková, L. Polylactic Acid and Its Cellulose Based Composite as a Significant Tool for the Production of Optimized Models Modified for Additive Manufacturing. Sustainability 2021, 13, 1256. [Google Scholar] [CrossRef]
- Nassajfar, M.N.; Deviatkin, I.; Leminen, V.; Horttanainen, M. Alternative Materials for Printed Circuit Board Production: An Environmental Perspective. Sustainability 2021, 13, 12126. [Google Scholar] [CrossRef]
- Castro-Aguirre, E.; Iñiguez-Franco, F.; Samsudin, H.; Fang, X.; Auras, R. Poly(lactic acid)—Mass production, processing, industrial applications, and end of life. Adv. Drug Deliv. Rev. 2016, 107, 333–366. [Google Scholar] [CrossRef]
- Groot, W.; van Krieken, J.; Sliekersl, O.; de Vos, S. Production and Purification of Lactic Acid and Lactide. In Poly(Lactic Acid): Synthesis, Structures, Properties, Processing, and Applications; Auras, R., Lim, L.-T., Selke, S.E.M., Tsuji, H., Eds.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2010; pp. 1–18. [Google Scholar] [CrossRef]
- Vink, E.T.H.; Rábago, K.R.; Glassner, D.A.; Gruber, P.R. Applications of life cycle assessment to NatureWorksTM polylactide (PLA) production. Polym. Degrad. Stab. 2003, 80, 403–419. [Google Scholar] [CrossRef]
- Hormnirun, P.; Marshall, E.L.; Gibson, V.C.; Pugh, R.I.; White, A.J. Study of ligand substituent effects on the rate and stereoselectivity of lactide polymerization using aluminum salen-type initiators. Proc. Natl. Acad. Sci. USA 2006, 103, 15343–15348. [Google Scholar] [CrossRef]
- Masutani, K.; Kimura, Y. PLA Synthesis and Polymerization; The Royal Society of Chemistry: London, UK, 2014. [Google Scholar]
- Yang, Y.; Wang, H.; Ma, H. Stereoselective Polymerization of rac -Lactide Catalyzed by Zinc Complexes with Tetradentate Aminophenolate Ligands in Different Coordination Patterns: Kinetics and Mechanism. Inorg. Chem. 2015, 54, 5839–5854. [Google Scholar] [CrossRef]
- Jamshidian, M.; Tehrany, E.A.; Imran, M.; Jacquot, M.; Desobry, S. Poly-Lactic Acid: Production, Applications, Nanocomposites, and Release Studies. Compr. Rev. Food Sci. Food Saf. 2010, 9, 552–571. [Google Scholar] [CrossRef] [PubMed]
- Michaliszyn-Gabryś, B.; Krupanek, J.; Kalisz, M.; Smith, J. Challenges for Sustainability in Packaging of Fresh Vegetables in Organic Farming. Sustainability 2022, 14, 5346. [Google Scholar] [CrossRef]
- Lim, L.-T.; Vanyo, T.; Randall, J.; Cink, K.; Agrawal, A.K. PROCESSING OF POLY(LACTIC ACID). In Poly(Lactic Acid): Synthesis, Structures, Properties, Processing, and Applications, and End of Life, 2nd ed.; John Wiley & Sons, Ltd.: New York, NY, USA, 2022; pp. 231–270. [Google Scholar] [CrossRef]
- Siddiqui, M.N.; Redhwi, H.H.; Al-Arfaj, A.A.; Achilias, D.S. Chemical Recycling of PET in the Presence of the Bio-Based Polymers, PLA, PHB and PEF: A Review. Sustainability 2021, 13, 10528. [Google Scholar] [CrossRef]
- Li, S.M.; Garreau, H.; Vert, M. Structure-property relationships in the case of the degradation of massive aliphatic poly-(α-hydroxy acids) in aqueous media. J. Mater. Sci. Mater. Med. 1990, 1, 123–130. [Google Scholar] [CrossRef]
- Hoshino, A.; Isono, Y. Degradation of aliphatic polyester films by commercially available lipases with special reference to rapid and complete degradation of poly(L-lactide) film by lipase PL derived from Alcaligenes sp. Biogeochemistry 2002, 13, 141–147. [Google Scholar] [CrossRef]



| Biomass Components | Cellulose (%) | Hemicellulose (%) |
|---|---|---|
| Cow manure | 16.62 | 15.26 |
| Alkali-treated cow manure | 35.34 | 15.48 |
| Acid-treated cow manure | 26.62 | 7.61 |
| Anaerobically digested cow manure | 14.5 | 12.56 |
| Alkali-treated anaerobically digested cow manure | 28.94 | 15.98 |
| Acid-treated anaerobically digested cow manure | 22.56 | 3.22 |
| Type of Acid Hydrolysis | Advantages | Disadvantages |
|---|---|---|
| Concentrated acid process | Low temperature operation; high sugar performance. | High acid consumption; high energetic cost; long reaction time (2–6 h). |
| Diluted acid process | Low acid consumption; lower permanence time. | High temperatures; low sugar performance; equipment corrosion; formation of non-desired products (degradation). |
| Raw Material | Pretreatment | Enzymes | Hydrolysis Conditions | % | Reference |
|---|---|---|---|---|---|
| Corn bran | AFEX | Specyme Cp Accellerase 1000 | T = 50, t = 48 h, 15 mg/g glucose | 40% | [41] |
| Cassava bagasse | –– | Termamyl 120 L AMG 200 L | T = 90 °C, t = 1 h, pH = 6.5 T = 60 °C, t = 24 h, pH = 4.5 T = 50 °C, t = 96 h, pH = 4.8 | 97.3% | [58] |
| Palm oil logs | AFEX | Accellerase 1000 | N = 170 rpm | 95.4% | [59] |
| Corn | Celluclast 1.5 L Novozyme 188 | T = 50 °C, t = 72h, N = 150 rpm, pH = 5 | 80% | [60] | |
| Cane bagasse | Diluted organosolv acid | Celluclast 1.5 L Novozyme 188 Xilanasa | T = 50 °C, t = 24 h, pH = 4.8 N = 150 rpm | 48–76% | [61] |
| Cane bagasse | Diluted phosphoric acid | Biocellulase W Novozyme-188 | T = 50 °C, t = 96 h, pH = 5 N = 100rpm | 74% | [62] |
| Banana tree | Gelatinization | Celluclast 1.5 L Novozyme-188 Pectinasa (P-2611) | T = 50 °C, t = 9 h, pH = 5 | 80% | [63] |
| Organism | Substrate | Lactic Acid Production [g/L] | Yield [g/g] | Productivity [g/(L·h)] | Reference |
|---|---|---|---|---|---|
| Enterococcus faecalis RKY1 | Glucose | 144 | 0.96 | 5.1 | [78] |
| Molasses | 95.7 | 0.95 | 4.0 | [79] | |
| Corn starch, potato, and wheat | 129.9 | 1.04 | 1.5 | [80] | |
| Wood hydrolyzate | 93 | 0.93 | 1.7 | [81] | |
| E. mundtii QU 25 | Modified Rogosa and Sharpe (mMRS) | 119 | 0.83 | 1.12 | [82] |
| Xylose | 86.7 | 0.84 | 0.9 | [83,84] | |
| Lactobacillus rhamnosus ATCC 10863 | Biomass pellets and glucose | 67 | 0.84 | 2.5 | [85] |
| Lactobacillus rhamnosus ATCC 7469 | Paper sludge | 73 | 0.97 | 2.9 | [86] |
| Lactobacillus rhamnosus CECT-288 | Yeast and meat extract, peptone | 32.5 | 0.88 | 5.4 | [87] |
| Cellulosic biosludges | 42 | 0.38 | 0.87 | [88] | |
| L. casei subsprhamnosus | Softwood | 21.1–23.75 | 0.74–0.83 | 0.15–0.23 | [89] |
| L. rhamnosus and L. brevis | Cornstover | 20.95 | 0.7 | 0.58 | [90] |
| Lactobacillus rhamnosus strain CASL | Cassava powder | 175.4 | 0.71 | 1.8 | [91] |
| L. rhamnosus ATCC 7469 | Distillery stillage | 97.1 | – | 1.80 | [92] |
| L. rhamnosus ATCC 7469 | Malting, brewing, and oil production byproducts | 58.01 | – | 1.19 | [93] |
| Lactobacillus helveticus ATCC 15009 | Whey permeate and yeast extract | 65.5 | 0.66 | 2.7 | [94] |
| Lactobacillus bulgaricus NRRL B-548 | Lactose, glucose, and galactose | 38.7 | 0.9 | 3.5 | [95] |
| L. bulgaricus CGMCC 1.6970 | Dairy waste | 113.18 | – | 2.36 | [96] |
| Lactobacillus casei NRRL B-441 | Barley malt sprouts | 82 | 0.91 | 5.6 | [97] |
| E. casseli flavus and L. casei | Xylose and glucose by co-cultivation | 95 | – | – | [98] |
| L. casei NCIMB 3254 | Cassava bagasse | 83.8 | 0.96 | 1.4 | [99] |
| L. casei and L. lactis | Date juice extract | 60.3 | – | 3.2 | [100] |
| L. casei ATCC 10863 | Ram horn waste | – | 0.08 | – | [101] |
| L. casei TISTR 390 | Sugarcane bagasse | 21.3 | – | 0.18 | [102] |
| Lactobacillus pentosus ATCC 8041 | Trimmings of vine shoots | 21.8 | 0.77 | 0.8 | [103] |
| L. pentosus CECT-4023T (ATCC-8041) | Corncobs | 24 | 0.76 | 0.51 | [104] |
| L. brevis and L. pentosus | Wheat straw | 7.1 | 0.95 | – | [105] |
| L. pentosus | L. pentosus | 74.8 | 0.65 | – | [106] |
| Lactobacillus amylophilus GV6 | Wheat flour | 76.2 | 0.7 | 0.8 | [107] |
| Bacillus sp. strain | Corncob molasses | 74.5 | 0.5 | 0.38 | [14] |
| Bacillus coagulans strains 36D1 | Paper sludge | 92 | 0.77 | 0.96 | [70] |
| Bacillus coagulans DSM 2314 | Lime-treated straw | 40.7 | 0.43 | – | [71] |
| Sugarcane bagasse | 58.7 | 0.73 | 1.81 | [72] | |
| B. coagulans IPE22 | Lignocellulosic hydrolysates | 50.48 | – | 3.16 | [73] |
| B. coagulans | Coffee pulp hydrolysate | 48.0 | – | 1.20 | [74] |
| Lactobacillus sp. RKY2 | Glucose, corn steep liquor, and yeast extract | 27 | 0.9 | 6.7 | [108] |
| Amylase-treated rice and wheat brans | 129 | 0.95 | 2.9 | [109] | |
| Lactobacillus bifermentans DSM 20003 | Wheat bran syrup | 62.8 | 0.83 | 1.2 | [110] |
| L. lactis RM2-24 | α-cellulose | 73 | 0.73 | 1.52 | [111] |
| Molasses and cellbiose | 70 | 0.88 | 1.45 | [112] | |
| Lactococcus lactis IO-1 | Sugarcane bagasse | 10.9 | 0.36 | 0.17 | [113] |
| Lactococcus lactis sp. lactis IFO 12007 | Raw cassava starch | 90 | 0.76 | 1.6 | [114] |
| Lc. Lactis IO-1 | Yeast extract, polypeptone, and xylose | 33.26 | 0.68 | – | [115] |
| L. lactis sub sp. lactis AS211 | Wheat flour | – | 0.77 | – | [116] |
| L. lactis sub sp. lactis ATCC 19435 | Wheat starch | – | – | 1.5 | [117] |
| Sporolactobacillus sp. CASD | Peanut meal | 207 | 0.93 | 3.8 | [118] |
| Sporolactobacillus laevolacticus DSM442 | Agricultural waste cottonseed | 144.4 | – | 4.13 | [119] |
| L. brevis | Hydrolysate of lignocellulosic | 39.1 | 0.7 | 0.81 | [120] |
| L. coryniformis ATCC 25600 | Yeast and meat extract | 54 | 0.89 | 0.5 | [121] |
| L. coryniformis spp. Torquens ATCC 25600 | Waste cardboard | 23.4 | 0.56 | 0.48 | [122] |
| Lactobacillus coryniformis ATCC 25 600 | Pretreated cardboard | 23 | 0.56 | 0.49 | [123] |
| L. coryniformis sub sp. Torquens ATCC 25600 | Pulp mill residue | 55.7 | – | 2.80 | [124] |
| Lactobacillus plantarum ATCC 21028 | Lactobacilli Man–Rogosa–Sharpe (MRS) broth (ATCC formula 416, DIFCO 0881) | 41 | 0.97 | 1 | [125] |
| L. plantarum | Alfalfa fiber | 46.4 | 0.46 | 0.64 | [126] |
| L. plantarum (Recombinant) | Cellooligosaccharides and β-glucan | 1.47 | – | – | [127] |
| Arabinose | 38.6 | 0.82 | 3.78 | [128] | |
| Xylose | 41.2 | 0.89 | 1.6 | [129] | |
| Leuconostoc lactis SHO-47 and SHO-54 | Xyloo-ligosaccharide | 2.3 | – | – | [130] |
| Bacillus sp. Strain 36D1 | Solka floc | 40 | 0. 65 | 0.22 | [131] |
| L. salivarious NRRL B-1950 | Soy molasses | – | 0.76 | – | [132] |
| Lactobacillus sp. | Sugarcane juice | 8.1 | – | – | [133] |
| L. amylovorus | Cassava starch substrate | 4.8 | – | – | [134] |
| Leuconostoc mesenteroides NRRL B 512 | Yeast extract and sugarcane juice | 60.2 | – | 1.25 | [135] |
| Mixed culture of B. coagulans LA1507 and engineered and adapted E. coli WYZ-L | Sweet sorghum juice | 118.0 | – | 1.84 | [136] |
| Molasses and corn steep liquor | 75.0 | – | 0.48 | [137] | |
| Bacillus sp. XZL9 | Corncob molasses | 74.7 | – | 0.38 | [14] |
| L. paracasei | Sweet sorghum | 88 | 0.79 | – | [138] |
| Adapted L. paracasei A-22 | Agro-industrial substrate | 169.9 | – | 1.42 | [139] |
| L. paracasei LA104/L. coryniformis ATCC 25600 | Waste Curcuma longa biomass | 97.1 | – | 2.70 | [140] |
| Lactobacillus sp. B2 | Crab (Callinectes bellicosus) wastes | 19.5 | – | 0.81 | [141] |
| R. oryzae TS-61 | Chicken feather protein hydrolysate and sugar beet molasses | 38.5 | – | 0.92 | [142] |
| R. oryzae NLX-M-1 | Xylo-oligosaccharides manufacturing waste residue | 60.3 | – | 1.0 | [143] |
| R. oryzae As 3.819 | Tobacco waste extract | 173.5 | – | 1.45 | [144] |
| Delbrueckii IFO 3202 | De-fatted rice bran | 28 | 0.78 | 0.28 | [145] |
| Delbrueckii NCIMB 8130 | Molasses | 90 | 0.97 | 3.8 | [146] |
| Lactobacillus delbrueckii Uc-3 | Cellobiose and cellotriose | 90 | 0.9 | 2.3 | [147] |
| L. delbrueckii mutant Uc-3 | Molasses | 166 | 0.87 | 4.2 | [79] |
| Waste sugarcane bagasse | 90 | 0.9 | 2.25 | [148] | |
| Molasses | 166 | 0.87 | 4.2 | [79] | |
| Delbrueckii | Sugarcane juice | 118 | 0.95 | 1.7 | [149] |
| L. delbreuckii | Alfalfa fiber | 35.4 | 0.35 | 0.75 | [126] |
| L. delbreuckii NRRL-B445 | Cellulosic material | 65 | 0.18 | – | [150,151] |
| L. delbrueckii NCIM 2025 | Cassava bagasse | 81.9 | 0.94 | 1.36 | [99] |
| L. delbrueckii UFV H2B20 | Brewer’s spent grain | 35.5 | 0.99 | 0.59 | [152] |
| L. delbrueckii ZU-S2 | Cellulosic material | 48.7/44.2 | 0.95/0.92 | 1.01/5.7 | [153] |
| Cellbiose and celltriose | 90 | 0.9 | 2.3 | [147] | |
| α-cellulose | 67 | 0.83 | 0.93 | [148] | |
| Lactobacillus delbrueckii HG 106 | Unpolished rice | 90 | 0.73 | 1.5 | [154] |
| L. delbrueckii sub sp. Lactis | Starch | – | 1.0 | – | [155] |
| L. delbrueckii NRRL B-445 | Molasses | – | 0.81 | – | [156] |
| L. delbrueckii sp. lactis NCDC290/L. delbrueckii sp. Delbrueckii NBRC3202 | Kodo millet (Paspalum scrobiculatum) bran residue | 10.53 | – | 0.44 | [157] |
| Engineered and adapted Pediococcus Acidilactici | Yeast extract, peptone, and glucose | 130.8 | – | 1.82 | [158] |
| S. inulinus YBS1-5 | Wheat bran, corn steep liquor, and yeast extract | 70.7 | – | 0.65 | [159] |
| Process | Advantage | Disadvantage | Reference |
|---|---|---|---|
| Precipitation | Can be applied in industrial plants | Sulfuric acid consumption is high | [170,173,174,175] |
| Easy to operate | Generation of gypsum, which requires landfill disposal | ||
| Elevates product yield | Product purity is low | ||
| Liquid extraction | Gypsum is not produced | Regeneration by distillation or back-extraction (stripping) of the extractants is needed | [170,175,176,177] |
| Thermal decomposition risk decreased | Purity is low | ||
| Disadvantageous distribution coefficients of the extraction agents | |||
| Membrane processes | Scale of production flexibility | High membrane cost | [170,178,179] |
| High differentiation | Membrane fouling | ||
| Good performance at purification | Polarization problems | ||
| Possibly to integrate with current fermenters | Retention of lactic acid | ||
| Difficult to increase production | |||
| Molecular distillation | Low thermal decomposition risk | Difficult to increase production | [171,179,180] |
| High differentiation | High vacuum needed | ||
| No solvents needed | |||
| No need for purification | |||
| Reactive distillation | Reaction and separation realized together | Mechanism is complex | [170] |
| Elevates purification values | Reversible chemical reactions in the liquid phase applicability | ||
| Energy consumption is low | Applications are restricted to systems with reasonably fast reaction rates and no mismatch between the temperatures suitable for reaction and separation | ||
| Homogeneous catalyst usage could cause corrosion and separation issues | |||
| Ion exchange adsorption | No waste generated | Large liquor waste as a result of widespread fluent use | [181,182,183,184,185,186,187,188] |
| Easy and simple to operate | Only applicable with low temperatures and short-/mid-term production | ||
| Elevate operational stability and selectivity | Co-extraction of other compounds is difficult | ||
| Accelerated product recovery | |||
| Low maintenance expenses and energy usage | |||
| Not toxic to microorganisms | |||
| Possibility to be integrated in an heterogeneous system | |||
| Reusable resin |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Garrido, R.; Cabeza, L.F.; Falguera, V.; Pérez Navarro, O. Potential Use of Cow Manure for Poly(Lactic Acid) Production. Sustainability 2022, 14, 16753. https://doi.org/10.3390/su142416753
Garrido R, Cabeza LF, Falguera V, Pérez Navarro O. Potential Use of Cow Manure for Poly(Lactic Acid) Production. Sustainability. 2022; 14(24):16753. https://doi.org/10.3390/su142416753
Chicago/Turabian StyleGarrido, Ricard, Luisa F. Cabeza, Víctor Falguera, and Omar Pérez Navarro. 2022. "Potential Use of Cow Manure for Poly(Lactic Acid) Production" Sustainability 14, no. 24: 16753. https://doi.org/10.3390/su142416753
APA StyleGarrido, R., Cabeza, L. F., Falguera, V., & Pérez Navarro, O. (2022). Potential Use of Cow Manure for Poly(Lactic Acid) Production. Sustainability, 14(24), 16753. https://doi.org/10.3390/su142416753









