Sewage Sludge Biorefinery for Circular Economy
Abstract
1. Introduction
2. EBSS Composition
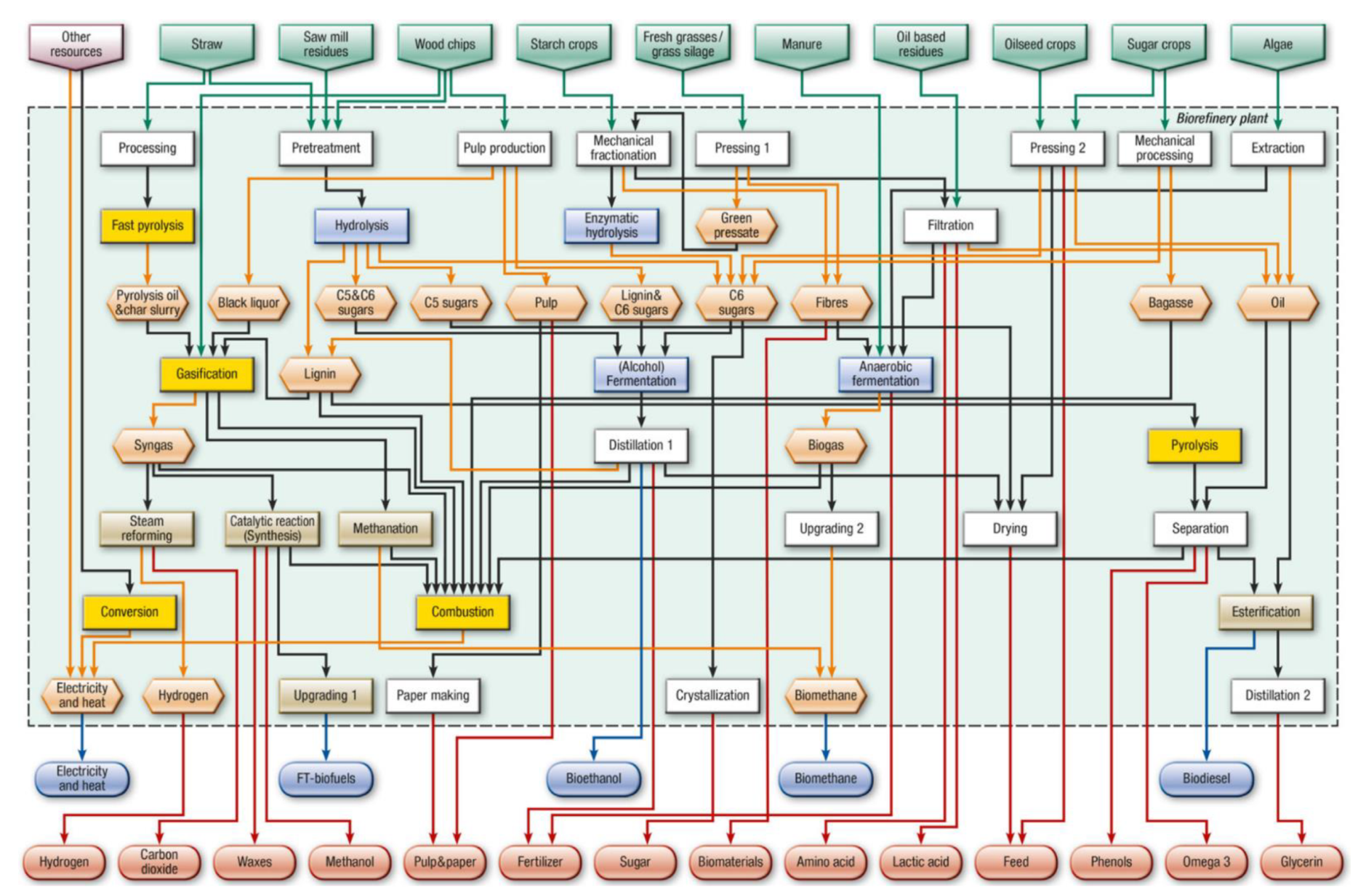
3. Biorefinery
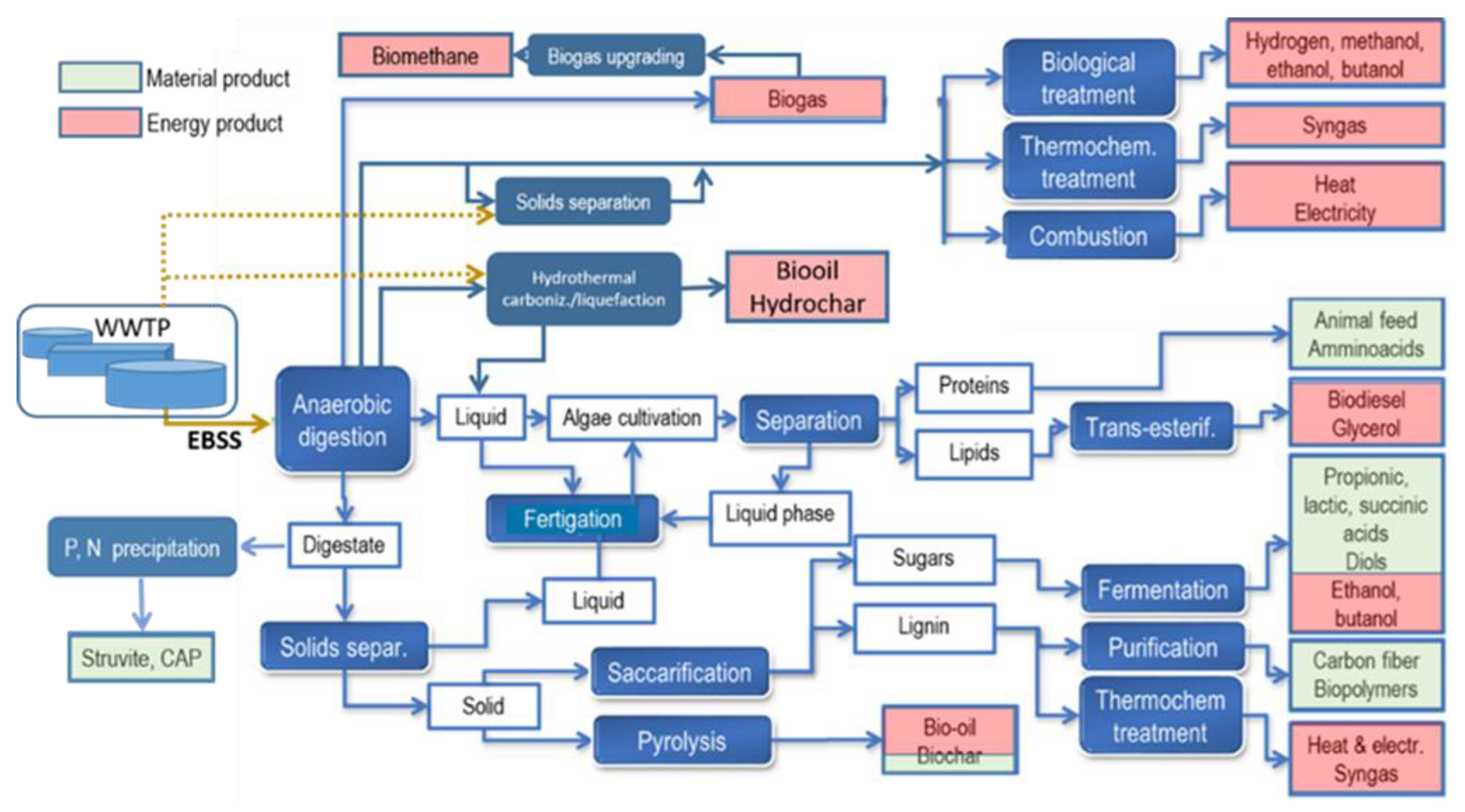
Sewage Sludge Biorefinery
4. Resources Recovery from EBSS Biorefinery
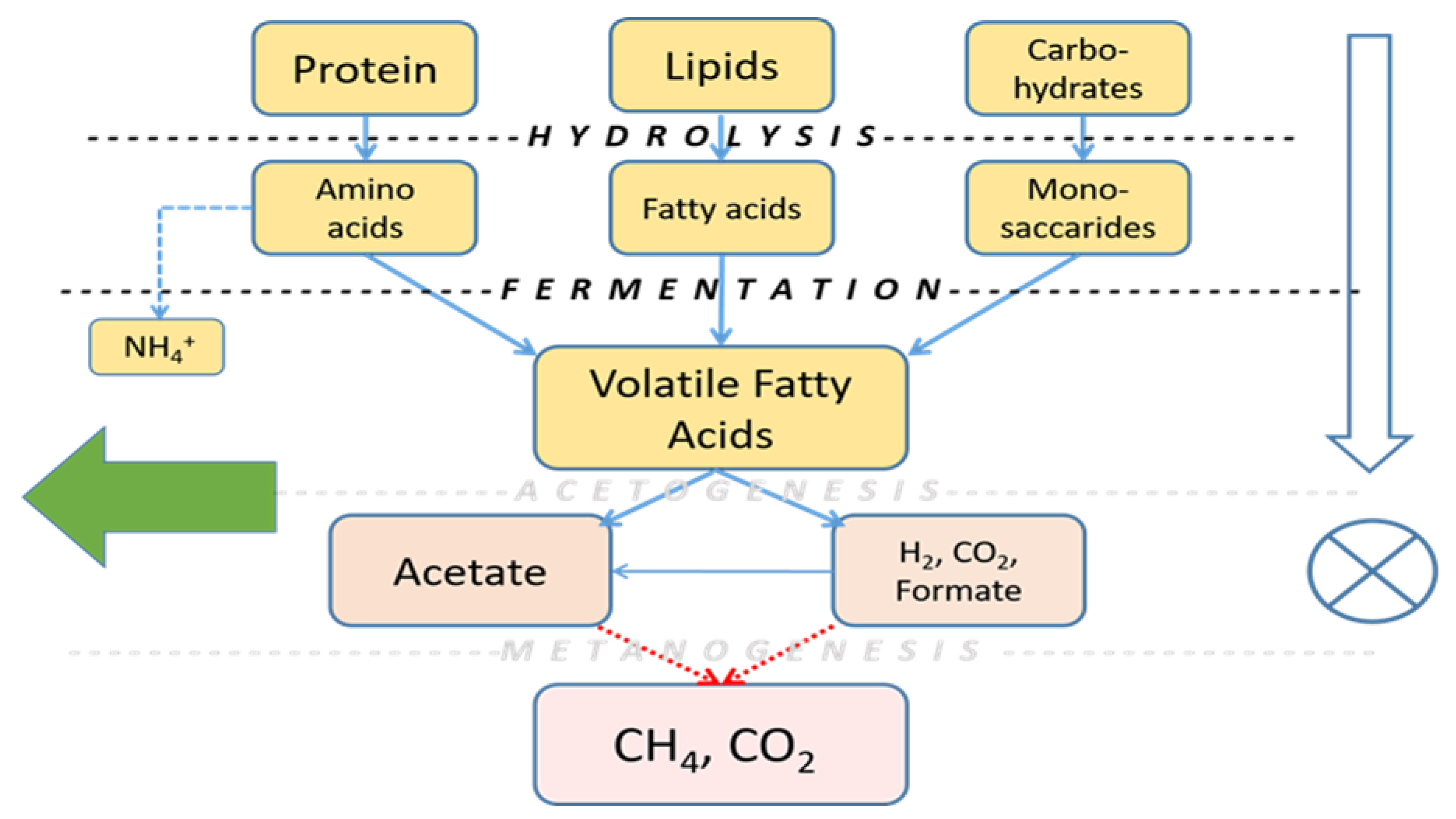
4.1. Anaerobic Fermentation as the First Step in Biomolecule Recovery
4.2. Materials Extraction from Raw Sludge: Resources, Uses and Techniques
4.2.1. Polyhydroxyalkanoates
4.2.2. Extracellular Polymeric Substances
4.2.3. Proteins
4.2.4. Biopesticides
4.2.5. Enzymes
4.2.6. Solvents and Biofuels
5. Biorefinery, Sustainability and Circular Economy
- Technological readiness level (TRL): most of the proposed technologies, although tested and validated at a laboratory or pilot scale, still remain in their infancy from the industrial application aspect, and further operational and cost optimization is needed;
- Biorefinery products such as proteins and enzymes still demand high production costs and raise some concerns in terms of pathogenic and metal toxicity, which effectively stifle technological scaling-up developments; efficient purification procedures for biorefined products (in line with the required purity standards for specific applications) still need further development to improve the overall economics of biorefinery approaches;
- The scale-up of production technology is an important issue since much of the research so far was performed at a laboratory scale. Scaling-up requires the optimization of each biorefinery process step and its integration with the local feedstock supply. The integration of compatible biomass sources (e.g., food and organic fraction of municipal solid waste) would contribute to the overall economics of biorefinery approaches;
- Existing non-technological barriers (i.e., unrelated to technological processes but limiting the subsequent diffusion of the product’s use) should also be taken into account. These can be summarily divided into economic, administrative and market-related barriers [106]. Examples of the first category are: the need for large investments, low profitability (compared to traditional production) and logistic difficulties for feedstock procurement. Administrative barriers may include final products’/materials’ certifications (when needed);
- Public and consumer acceptance: the ultimate success of a CBE approach depends on the consumer acceptance of recovered waste-based products. Consumers’ perception of CBE schemes has been largely overlooked in the scientific literature since generic ethical intentions by citizens do not necessarily translate into virtuous behavior [107].
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- EC. Solving Treatment of Wastewater Sewage Sludge with New HTL Technology to Produce Hydrocarbons, Asphalts and Fertilizers; Life Public Databases Reference: LIFE19 ENV/IT/000165U. 2020. Available online: https://webgate.ec.europa.eu/life/publicWebsite/index.cfm?fuseaction=search.dspPage&n_proj_id=7687 (accessed on 26 September 2022).
- University of Michigan. US Wastewater Treatment—Factsheet Water. 2021. Available online: https://css.umich.edu/sites/default/files/Wastewater%20Treatment_CSS04-14_e2021.pdf (accessed on 26 September 2022).
- Wei, L.; Zhu, F.; Lia, Q.; Xue, C.; Xia, X.; Yu, H.; Zhao, Q.; Jiang, J.; Bai, B. Development, current state and future trends of sludge management in China: Based on exploratory data and CO2-equivaient emissions analysis. Environ. Int. 2020, 144, 106093. [Google Scholar] [CrossRef] [PubMed]
- Shizas, I.; Bagley, D.M. Experimental determination of energy content of unknown organics in municipal wastewater streams. J. Energy Eng. 2004, 130, 45–53. [Google Scholar] [CrossRef]
- Corradini, F.; Meza, P.; Eguiluz, R.; Casado, F.; Huerta-Lwang, E.; Geissen, V. Evidence of microplastic accumulation in agricultural soils from sewage sludge disposal. Sci. Total Environ. 2019, 671, 411–420. [Google Scholar] [CrossRef] [PubMed]
- Hennebert, P.; Anderson, A.; Merdy, P. Mineral Nanoparticles in Waste: Potential Sources, Occurrence in Some Engineered Nanomaterials Leachates, Municipal Sewage Sludges and Municipal Landfill Sludges. J. Biotechnol. Biomater. 2017, 7, 2. [Google Scholar] [CrossRef]
- Mailler, R.; Gasperi, J.; Patureau, D.; Vulliet, E.; Delgenes, N.; Danel, N.; Deshayesa, S.; Eudes, V.; Guerine, S.; Moilleron, R.; et al. Fate of emerging and priority micropollutants during the sewage sludge treatment: Case study of Paris conurbation—Part 1: Contamination of the different types of sewage sludge. Waste Manag. 2017, 59, 379–393. [Google Scholar] [CrossRef]
- Capodaglio, A.G.; Olsson, G. Energy issues in sustainable urban wastewater management: Use, demand reduction and recovery in the urban water cycle. Sustainability 2020, 12, 266. [Google Scholar] [CrossRef]
- Appels, L.; Baeyens, J.; Degreve, J.; Dewil, R. Principles and potential of the anaerobic digestion of waste-activated sludge. Prog. Energy Combust. 2008, 34, 755–781. [Google Scholar] [CrossRef]
- Callegari, A.; Hlavinek, P.; Capodaglio, A.G. Production of energy (biodiesel) and recovery of materials (biochar) from pyrolysis of urban waste sludge. Rev. Ambiente Água 2018, 13, e2128. [Google Scholar] [CrossRef]
- Daneshgar, S.; Buttafava, A.; Callegari, A.; Capodaglio, A.G. Economic and energetic assessment of different phosphorus recovery options from aerobic sludge. J. Clean. Prod. 2019, 223, 729–738. [Google Scholar] [CrossRef]
- Tyagi, V.K.; Lo, S.L. Sludge: A waste or renewable source for energy and resources recovery? Renew. Sustain. Energy Rev. 2013, 25, 708–728. [Google Scholar] [CrossRef]
- Keiding, K.; Wybrandt, L.; Nielsen, P.H. Remember the water: A comment on EPS colligative properties. Water Sci. Technol. 2001, 43, 17–23. [Google Scholar] [CrossRef] [PubMed]
- Ding, Z.; Bourven, I.; Guibaud, G.; van Hullebusch, E.D.; Panico, A.; Pirozzi, F.; Esposito, G. Role of extracellular polymeric substances (EPS) production in bioaggregation: Application to wastewater treatment. Appl. Microbiol. Biotechnol. 2015, 99, 9883–9905. [Google Scholar] [CrossRef] [PubMed]
- Feng, C.; Lotti, T.; Canziani, R.; Lin, Y.; Tagliabue, C.; Malpei, F. Extracellular biopolymers recovered as raw biomaterials from waste granular sludge and potential applications: A critical review. Sci. Total Environ. 2021, 753, 142051. [Google Scholar] [CrossRef] [PubMed]
- Capodaglio, A.G.; Bolognesi, S.; Cecconet, D. Sustainable, decentralized sanitation and reuse with hybrid nature-based systems. Water 2021, 13, 1583. [Google Scholar] [CrossRef]
- Li, K.; Liu, Q.; Fan, F.; Luo, R.; Lu, Q.; Zhou, W.; Huo, S.; Cheng, P.; Liu, J.; Addy, M.; et al. Microalgae-based wastewater treatment for nutrients recovery: A review. Bioresour. Technol. 2019, 291, 121934. [Google Scholar] [CrossRef] [PubMed]
- Cherubini, F.; Jungmeier, G.; Wellisch, M.; Wilke, T.; Skiadas, I.; Ree, V.R.; Jong, D.E. Toward a common classification approach for biorefinery systems. Biofuels Bioprod. Biorefining 2009, 3, 534–546. [Google Scholar] [CrossRef]
- Hingsamer, M.; Jungmeier, G. Biorefineries. In The Role of Bioenergy in the Bioeconomy; Lago, C., Caldés, N., Lechón, Y., Eds.; Academic Press: Cambridge, MA, USA, 2019; Chapter 5; pp. 179–222. [Google Scholar] [CrossRef]
- Callegari, A.; Bolognesi, S.; Cecconet, D.; Capodaglio, A.G. Production technologies, current role and future prospects of biofuels feedstocks: A state-of-the-art review. Crit. Rev. Environ. Sci. Technol. 2020, 50, 384–436. [Google Scholar] [CrossRef]
- Bora, R.R.; Richardson, R.E.; You, F. Resource recovery and waste-to-energy from wastewater sludge via thermochemical conversion technologies in support of circular economy: A comprehensive review. BMC Chem. Eng. 2020, 2, 8. [Google Scholar] [CrossRef]
- Gao, N.; Kamran, K.; Quan, C.; Williams, P.T. Thermochemical conversion of sewage sludge: A critical review. Prog. Energy Combust. Sci. 2020, 79, 100843. [Google Scholar] [CrossRef]
- Hu, M.; Ye, Z.; Zhang, H.; Chen, B.; Pan, Z.; Wang, J. Thermochemical conversion of sewage sludge for energy and resource recovery: Technical challenges and prospects. Environ. Pollut. Bioavailab. 2021, 33, 145–163. [Google Scholar] [CrossRef]
- Bolognesi, S.; Bernardi, G.; Callegari, A.; Dondi, D.; Capodaglio, A.G. Biochar production from sewage sludge and microalgae mixtures: Properties, sustainability and possible role in circular economy. Biomass-Convers. Biorefinery 2021, 11, 289–299. [Google Scholar] [CrossRef]
- Capodaglio, A.G.; Callegari, A.; Dondi, D. Microwave-Induced Pyrolysis for Production of Sustainable Biodiesel from Waste Sludges. Waste Biomass Valoriz. 2016, 7, 703–709. [Google Scholar] [CrossRef]
- Tasca, A.L.; Puccini, M.; Gori, R.; Corsi, I.; Galletti, A.M.R.; Vitolo, S. Hydrothermal carbonization of sewage sludge: A critical analysis of process severity, hydrochar properties and environmental implications. Waste Manag. 2019, 93, 1–13. [Google Scholar] [CrossRef]
- Thomsen, L.B.S.; Carvalho, P.N.; Dos Passos, J.S.; Anastasakis, K.; Bester, K.; Biller, P. Hydrothermal liquefaction of sewage sludge; energy considerations and fate of micropollutants during pilot scale processing. Water Res. 2020, 183, 116101. [Google Scholar] [CrossRef] [PubMed]
- Capodaglio, A.G.; Callegari, A.; Lopez, M.V. European Framework for the Diffusion of Biogas Uses: Emerging Technologies, Acceptance, Incentive Strategies and Institutional-Regulatory Support. Sustainability 2016, 8, 298. [Google Scholar] [CrossRef]
- Badea, G.I.; Radu, G.L. Introductory Chapter: Carboxylic Acids—Key Role in Life Sciences. In Carboxylic Acid—Key Role in Life Sciences; Badea, G.I., Radu, G.L., Eds.; IntechOpen: London, UK, 2018. [Google Scholar] [CrossRef]
- Shen, L.; Hu, H.; Ji, H.; Cai, J.; He, N.; Li, Q.; Wang, Y. Production of poly(hydroxybutyrate-hydroxyvalerate) from waste organics by the two-stage process: Focus on the intermediate volatile fatty acids. Bioresour. Technol. 2014, 166, 194–200. [Google Scholar] [CrossRef]
- Levy, P.F.; Sanderson, J.E.; Kispert, R.G.; Wise, D.L. Biorefining of biomass to liquid fuels and organic chemicals. Enzym. Microb. Technol. 1981, 3, 207–215. [Google Scholar] [CrossRef]
- Cord-Ruwisch, R. Thermodynamics of anaerobic digestion: Mechanism of suppression on biogas production during acidogenesis. INMATEH Agric. Eng. 2019, 57, 287–301. [Google Scholar]
- Li, X.; Zhao, J.; Wang, D.; Yang, Q.; Zeng, G. An efficient and green pretreatment to stimulate short-chain fatty acids production from waste activated sludge anaerobic fermentation using free nitrous acid. Chemosphere 2016, 144, 160–167. [Google Scholar] [CrossRef]
- Zhang, C.; Qin, Y.; Xu, Q.; Liu, X.; Liu, Y.; Ni, B.J.; Yang, Q.; Wang, D.; Li, X.; Wang, Q. Free ammonia-based pretreatment promotes short-chain fatty acid production from waste activated sludge. ACS Sustain. Chem. Eng. 2018, 6, 9120–9129. [Google Scholar] [CrossRef]
- Liu, X.; Du, M.; Yang, J.; Wu, X.; Xu, Q.; Wang, D.; Yang, Q.; Yang, G.; Li, X. Sulfite serving as a pretreatment method for alkaline fermentation to enhance short-chain fatty acid production from waste activated sludge. Chem. Eng. J. 2020, 385, 123991. [Google Scholar] [CrossRef]
- He, Z.W.; Liu, W.Z.; Gao, Q.; Tang, C.C.; Wang, L.; Guo, Z.C.; Zhou, A.J.; Wang, A.J. Potassium ferrate addition as an alternative pre-treatment to enhance short-chain fatty acids production from waste activated sludge. Bioresour. Technol. 2018, 247, 174–181. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Huang, Y.; Xu, Q.; Liu, X.; Yang, Q.; Li, X. Free ammonia aids ultrasound pretreatment to enhance short-chain fatty acids production from waste activated sludge. Bioresour. Technol. 2019, 275, 163–171. [Google Scholar] [CrossRef] [PubMed]
- Pang, H.; Li, L.; He, J.; Yan, Z.; Ma, Z.; Nan, J.; Liu, Y. New insight into enhanced production of short-chain fatty acids from waste activated sludge by cation exchange resin-induced hydrolysis. Chem. Eng. J. 2020, 388, 124235. [Google Scholar] [CrossRef]
- Wu, Y.; Song, K. Effect of thermal activated peroxydisulfate pretreatment on short-chain fatty acids production from waste activated sludge anaerobic fermentation. Bioresour. Technol. 2019, 292, 121977. [Google Scholar] [CrossRef]
- Frison, N.; Katsou, E.; Malamis, S.; Oehmen, A.; Fatone, F. Development of a novel process integrating the treatment of sludge reject water and the production of polyhydroxyalkanoates (PHAs). Environ. Sci. Technol. 2015, 49, 10877–10885. [Google Scholar] [CrossRef]
- Zacharof, M.P.; Lovitt, R.W. Complex effluent streams as a potential source of volatile fatty acids. Waste Biomass Valoriz. 2013, 4, 557–581. [Google Scholar] [CrossRef]
- Wu, S.L.; Wei, W.; Sun, J.; Xu, Q.; Dai, X.; Ni, B.J. Medium-Chain fatty acids and long-chain alcohols production from waste activated sludge via two-stage anaerobic fermentation. Water. Res. 2020, 186, 116381. [Google Scholar] [CrossRef]
- Yishai, O.; Lindner, S.N.; de la Cruz, J.G.; Tenenboim, H.; Bar-Even, A. The formate bio-economy. Curr. Opin. Chem. Biol. 2016, 35, 1–9. [Google Scholar] [CrossRef]
- Raboni, M.; Viotti, P.; Capodaglio, A.G. A comprehensive analysis of the current and future role of biofuels for transport in the European Union (EU). Rev. Ambiente Agua 2015, 10, 9. [Google Scholar] [CrossRef]
- Mühlemeier, I.M.; Speight, R.; Strong, P.J. Biogas, bioreactors and bacterial methane oxidation. In Methane Biocatalysis: Paving the Way to Sustainability; Kalyuzhnaya, M.G., Xing, X., Eds.; Springer International Publishing AG: Cham, Switzerland, 2018; pp. 213–235. [Google Scholar]
- Zhang, T.; Zhou, J.; Wang, X.; Zhang, Y. Coupled effects of methane monooxygenase and nitrogen source on growth and poly-β-hydroxybutyrate (PHB) production of Methylosinus trichosporium OB3b. J. Env. Sci. 2017, 52, 49–57. [Google Scholar] [CrossRef] [PubMed]
- Sharma, V.; Sehgal, R.; Gupta, R. Polyhydroxyalkanoate (PHA): Properties and Modifications. Polymer 2021, 212, 123161. [Google Scholar] [CrossRef]
- Gao, X.; Chen, J.C.; Wu, Q.; Chen, W. Polyhydroxyalkanoates as a source of chemicals, polymers and biofuels. Curr. Opin. Biotechnol. 2011, 22, 768–774. [Google Scholar] [CrossRef]
- Spekreijse, J.; Le Nôtre, J.; van Haveren, J.; Scott, E.L.; Sanders, J.P.M. Simultaneous production of biobased styrene and acrylates using ethenolysis. Green Chem. 2012, 14, 2747–2751. [Google Scholar] [CrossRef]
- Yadav, B.; Talan, A.; Tyagi, R.D.; Drogui, P. Concomitant production of value-added products with polyhydroxyalkanoate (PHA) synthesis: A review. Biores. Technol. 2021, 337, 125419. [Google Scholar] [CrossRef] [PubMed]
- Morgan-Sagastume, F.; Hjort, M.; Cirne, D.; Gérardin, F.; Lacroix, S.; Gaval, G.; Karabegovic, L.; Alexandersson, T.; Johansson, P.; Karlsson, A.; et al. Integrated production of polyhydroxyalkanoates (PHAs) with municipal wastewater and sludge treatment at pilot scale. Bioresour. Technol. 2015, 181, 78–89. [Google Scholar] [CrossRef]
- Battista, F.; Frison, N.; Pavan, P.; Cavinato, C.; Gottardo, M.; Fatone, F.; Eusebi, A.L.; Majone, M.; Zeppilli, M.; Valentino, F.; et al. Food wastes and sewage sludge as feedstock for an urban biorefinery producing biofuels and added-value bioproducts. J. Chem. Technol. Biotechnol. 2020, 95, 328–338. [Google Scholar] [CrossRef]
- Morgan-Sagastume, F.; Karlsson, A.; Johansson, P.; Pratt, S.; Boon, N.; Lant, P.; Werker, A. Production of polyhydroxyalkanoates in open, mixed cultures from a waste sludge stream containing high levels of soluble organics, nitrogen and phosphorus. Water Res. 2010, 44, 5196–5211. [Google Scholar] [CrossRef]
- Bluemink, E.; van Nieuwenhuijzen, A.; Wypkema, E.; Uijterlinde, C. Bio-plastic (poly-hydroxy-alkanoate) production from municipal sewage sludge in the Netherlands: A technology push or a demand driven process? Water Sci. Technol. 2016, 74, 353–358. [Google Scholar] [CrossRef]
- Zhang, X.Q.; Bishop, P.L. Biodegradability of biofilm extracellular polymeric substances. Chemosphere 2003, 50, 63–69. [Google Scholar] [CrossRef]
- Andreoli, C.V.; Von Sperling, M.; Fernandes, F.; Ronteltap, M. Sludge Treatment and Disposal; IWA Publishing: London, UK, 2007. [Google Scholar]
- Lackner, S.; Gilbert, E.M.; Vlaeminck, S.E.; Joss, A.; Horn, H.; van Loosdrecht, M.C.M. Full-scale partial nitritation/anammox experiences–an application survey. Water Res. 2014, 55, 292–303. [Google Scholar] [CrossRef] [PubMed]
- Flemming, H.C.; Wingender, J. Relevance of microbial extracellular polymeric substances (EPSs)—Part II: Technical aspects. Water Sci. Technol. 2001, 43, 9–16. [Google Scholar] [CrossRef] [PubMed]
- Hohapatra, S.P.; Siebel, M.A.; Alaerts, G.J. Effect of Bacillus megaterium on removal of copper from aqueous solutions by activated carbon. J. Environ. Sci. Health Part A 1993, 28, 615–629. [Google Scholar]
- Schlekat, C.E.; Decho, A.W.; Chandler, G.T. Sorption of cadmium to bacterial extracellular polymeric sediment coatings under estuarine conditions. Environ. Toxicol. Chem. Int. J. 1998, 17, 1867–1874. [Google Scholar] [CrossRef]
- Spath, R.; Flemming, H.C.; Wuertz, S. Sorption properties of biofilms. Water Sci. Technol. 1998, 37, 207–210. [Google Scholar] [CrossRef]
- Liu, A.B.; Ahn, I.S.; Mansfield, C.; Lion, L.W.; Shuler, M.L.; Ghiorse, W.C. Phenanthrene desorption from soil in the presence of bacterial extracellular polymer: Observations and model predictions of dynamic behaviour. Water Res. 2000, 35, 835–843. [Google Scholar] [CrossRef]
- Esparza-Soto, M.; Westerhoff, P. Biosorption of humic and fulvic acids to live activated sludge biomass. Water Res. 2003, 37, 2301–2310. [Google Scholar] [CrossRef]
- Sheng, G.P.; Zhang, M.L.; Yu, H.Q. Characterization of adsorption properties of extracellular polymeric substances (EPS) extracted from sludge. Colloids Surf. B Biointerface 2008, 62, 83–90. [Google Scholar] [CrossRef]
- Kim, N.K.; Mao, N.; Lin, R.; Bhattacharyya, D.; van Loosdrecht, M.C.M.; Lin, Y. Flame retardant property of flax fabrics coated by extracellular polymeric substances recovered from both activated sludge and aerobic granular sludge. Water Res. 2020, 170, 115344. [Google Scholar] [CrossRef]
- Liu, H.; Fang, H.H. Extraction of extracellular polymeric substances (EPS) of sludges. J. Biotechnol. 2002, 95, 249–256. [Google Scholar] [CrossRef]
- Dubé, C.D.; Guiot, S.R. Characterization of the protein fraction of the extracellular polymeric substances of three anaerobic granular sludges. AMB Express 2019, 9, 23. [Google Scholar] [CrossRef] [PubMed]
- Felz, S.; Al-Zuhairy, S.; Aarstad, O.A.; van Loosdrecht, M.C.M.; Lin, Y.M. Extraction of structural extracellular polymeric substances from aerobic granular sludge. J. Vis. Exp. 2016, 115, e54534. [Google Scholar] [CrossRef] [PubMed]
- Boleij, M.; Seviour, T.; Wong, L.L.; van Loosdrecht, M.C.M.; Lin, Y. Solubilization and characterization of extracellular proteins from anammox granular sludge. Water Res. 2019, 164, 114952. [Google Scholar] [CrossRef] [PubMed]
- Frølund, B.; Palmgren, R.; Keiding, K.; Nielsen, P.H. Extraction of extracellular polymers from activated sludge using a cation exchange resin. Water Res. 1996, 30, 1749–1758. [Google Scholar] [CrossRef]
- Sheng, G.; Yu, H.; Li, X. Extracellular polymeric substances (EPS) of microbial aggregates in biological wastewater treatment systems: A review. Biotechnol. Adv. 2010, 28, 882–894. [Google Scholar] [CrossRef] [PubMed]
- Sesay, M.L.; Özcengiz, G.; Sanin, F.D. Enzymatic extraction of activated sludge extracellular polymers and implications on bioflocculation. Water Res. 2006, 40, 1359–1366. [Google Scholar] [CrossRef] [PubMed]
- Gehr, R.; Henry, J. Removal of extracellular material techniques and pitfalls. Water Res. 1983, 17, 1743–1748. [Google Scholar] [CrossRef]
- Seviour, T.; Donose, B.C.; Pijuan, M.; Yuan, Z. Purification and conformational analysis of a key exopolysaccharide component of mixed culture aerobic sludge granules. Environ. Sci. Technol. 2010, 44, 4729–4734. [Google Scholar] [CrossRef]
- Capodaglio, A.G. Pulse electric field technology for wastewater and biomass residues’ improved valorization. Processes 2021, 9, 736. [Google Scholar] [CrossRef]
- Hwang, J.; Zhang, L.; Seo, S.; Lee, Y.W.; Jahng, D. Protein recovery from excess sludge for its use as animal feed. Bioresour. Technol. 2008, 99, 8949–8954. [Google Scholar] [CrossRef]
- Su, R.; Hussain, A.; Guo, J.; Guan, J.; He, Q.; Yan, X.; Li, D.; Guo, Z. Animal feeds extracted from excess sludge by enzyme, acid and base hydrolysis processes. ACS Sust. Chem. Eng. 2015, 3, 2084–2091. [Google Scholar]
- Zhuang, L.; Zhou, S.; Wang, Y.; Liu, Z.; Xu, R. Cost-effective production of Bacillus thuringiensis biopesticides by solid-state fermentation using wastewater sludge: Effects of heavy metals. Bioresour. Technol. 2011, 102, 4820–4826. [Google Scholar] [CrossRef] [PubMed]
- Montiel, M.T.; Tyagi, R.; Valero, J.; Surampalli, R. Production of biopesticides using wastewater sludge as a raw material-effect of process parameters. Water Sci. Technol. 2003, 48, 239–246. [Google Scholar] [CrossRef]
- Liu, Z.; Smith, S.R. Enzyme Recovery from Biological Wastewater Treatment. Waste Biomass Valoriz. 2021, 12, 4185–4211. [Google Scholar] [CrossRef]
- Balasubramanian, S.; Tyagi, R.D. Value-Added Bio-Products from Sewage Sludge. In Current Developments in Biotechnology and Bioengineering; Wong, J.W.-C., Tyagi, R.D., Pandey, A., Eds.; Elsevier: Amsterdam, The Netherlands, 2017; pp. 27–42. [Google Scholar]
- Frølund, B.; Palmgren, R.; Keiding, K.; Nielsen, P.H. Enzymatic activity in the activated sludge floc matrix. Appl. Microbiol. Biotechnol. 1995, 43, 708–716. [Google Scholar] [CrossRef]
- Drouin, M.; Lai, C.K.; Tyagi, R.D.; Surampalli, R.Y. Bacillus licheniformis proteases as high value added products from fermentation of wastewater sludge: Pre-treatment of sludge to increase the performance of the process. In Proceedings of the IWA Specialist Conference on Moving Forward Wastewater Biosolids Sustainability: Technical, Managerial and Public Synergy, Moncton, NB, Canada, 24–27 June 2007; pp. 599–605. [Google Scholar]
- Subramanian, S.B.; Yan, S.; Tyagi, R.D.; Surampalli, R.Y. Enzymes: Production and Extraction. In Sustainable Sludge Management: Production of Value Added Products; Tyagi, R.D., Surampalli, R.Y., Yan, S., Zhang, T.C., Eds.; American Society of Civil Engineers: Reston, VA, USA, 2013. [Google Scholar]
- Guanghui, Y.; Pinjing, H.; Liming, S.; Yishu, Z. Enzyme extraction by ultrasound from sludge flocs. J. Environ. Sci. 2009, 21, 204–210. [Google Scholar]
- Jung, J.; Xing, X.H.; Matsumoto, K. Recoverability of protease released from disrupted excess sludge and its potential application to enhanced hydrolysis of proteins in wastewater. Biochem. Eng. J. 2002, 10, 67–72. [Google Scholar] [CrossRef]
- Nabarlatz, D.; Vondrysova, J.; Jenicek, P.; Stüber, F.; Font, J.; Fortuny, A.; Fabregat, A.; Bengoa, C. Hydrolytic enzymes in activated sludge: Extraction of protease and lipase by stirring and ultrasonication. Ultrason. Sonochem. 2010, 17, 923–931. [Google Scholar] [CrossRef]
- Gessesse, A.; Dueholm, T. Lipase and protease extraction from activated sludge. Water Res. 2011, 37, 3652–3657. [Google Scholar] [CrossRef]
- Yu, G.H.; He, P.J.; Shao, L.M.; Lee, D.J. Enzyme activities in activated sludge flocs. Appl. Microbiol. Biotechnol. 2007, 77, 605–612. [Google Scholar] [CrossRef]
- Clavijo Rivera, E.; Montalescot, V.; Viau, M.; Drouin, D.; Bourseau, P.; Frappart, M.; Monteux, C.; Couallier, E. Mechanical cell disruption of Parachlorella kessleri microalgae: Impact on lipid fraction composition. Bioresour. Technol. 2018, 256, 77–85. [Google Scholar] [CrossRef] [PubMed]
- Feng, S.; Ngo, H.H.; Guo, W.; Chang, S.W.; Nguyen, D.D.; Cheng, D.; Varjani, S.; Lei, Z.; Liu, Y. Roles and applications of enzymes for resistant pollutants removal in wastewater treatment. Bioresour. Technol. 2021, 335, 125278. [Google Scholar] [CrossRef] [PubMed]
- Capodaglio, A.G.; Callegari, A. Feedstock and process influence on biodiesel produced from waste sewage sludge. J. Environ. Manag. 2018, 216, 176–182. [Google Scholar] [CrossRef] [PubMed]
- Djandja, O.S.; Wang, Z.C.; Wang, F.; Xu, Y.P.; Duan, P.G. Pyrolysis of Municipal Sewage Sludge for Biofuel Production: A Review. Ind. Eng. Chem. Res. 2020, 59, 16939–16956. [Google Scholar] [CrossRef]
- Quan, L.M.; Kamyab, H.; Yuzir, A.; Ashokkumar, V.; Hosseini, S.E.; Balasubramanian, B.; Kirpichnikova, I. Review of the application of gasification and combustion technology and waste-to-energy technologies in sewage sludge treatment. Fuel 2022, 316, 123199. [Google Scholar] [CrossRef]
- Boocock, D.G.B.; Konar, S.K.; Leung, A.; Ly, L.D. Fuels and chemicals from sewage sludge: 1. The solvent extraction and composition of a lipid from a raw sewage sludge. Fuel 1992, 71, 1283–1289. [Google Scholar] [CrossRef]
- Olkiewicz, M.; Caporgno, M.P.; Fortuny, P.; Stüber, F.; Fabregat, A.; Font, J.; Bengoa, C.M. Direct liquid-liquid extraction of lipid from municipal sewage sludge for biodiesel production. Fuel Process. Technol. 2014, 128, 331–338. [Google Scholar] [CrossRef]
- D’Ambrosio, V.; di Bitonto, L.; Angelini, A.; Gallipoli, A.; Braguglia, C.M.; Pastore, C. Lipid extraction from sewage sludge using green biosolvent for sustainable biodiesel production. J. Clean. Prod. 2021, 329, 129643. [Google Scholar] [CrossRef]
- Olkiewicz, M.; Fortuny, P.; Stüber, F.; Fabregat, A.; Font, J.; Bengoa, C. Effects of pre-treatments on the lipid extraction and biodiesel production from municipal WWTP sludge. Fuel 2015, 141, 250–257. [Google Scholar] [CrossRef]
- Siddiquee, M.N.; Rohani, S. Lipid extraction and biodiesel production from municipal sewage sludges: A review. Renew. Sustain. Energy Rev. 2011, 15, 1067–1072. [Google Scholar] [CrossRef]
- Patiño, Y.; Mantecón, L.G.; Polo, S.; Faba, L.; Díaz, E.; Ordóñez, S. Effect of sludge features and extraction-esterification technology on the synthesis of biodiesel from secondary wastewater treatment sludges. Bioresour. Technol. 2018, 247, 209–216. [Google Scholar] [CrossRef]
- Sakaveli, F.; Petala, M.; Tsiridis, V.; Darakas, E. Enhanced mesophilic anaerobic digestion of primary sewage sludge. Water 2021, 13, 348. [Google Scholar] [CrossRef]
- De Jesus, S.S.; Maciel Filho, R. Recent advances in lipid extraction using green solvents. Renew. Sustain. Energy Rev. 2020, 133, 110289. [Google Scholar] [CrossRef]
- Faragò, M.; Damgaard, A.; Madsen, J.A.; Andersen, J.K.; Thornberg, D.; Andersen, M.H.; Rygaard, M. From wastewater treatment to water resource recovery: Environmental and economic impacts of full-scale implementation. Water Res. 2021, 204, 117554. [Google Scholar] [CrossRef]
- Qandil, M.D.; Abbas, A.I.; Abdelhadi, A.I.; Salem, A.R.; Amano, R.S. Energy Analysis: Ways to Save Energy and Reduce the Emissions in Wastewater Treatment Plants. Int. J. Energy Clean Environ. 2021, 22, 91–112. [Google Scholar] [CrossRef]
- Capodaglio, A.G. Fit-for-purpose urban wastewater reuse: Analysis of issues and available technologies for sustainable multiple barrier approaches. Crit. Rev. Environ. Sci. Technol. 2020, 51, 1619–1666. [Google Scholar] [CrossRef]
- McCormick, K.; Kaberger, T. Key barriers for bioenergy in Europe: Economic conditions, know-how and institutional capacity and supply chain co-ordination. Biomass Bioenergy 2007, 31, 443–452. [Google Scholar] [CrossRef]
- Russo, I.; Confente, I.; Scarpi, D.; Hazen, B.T. From trash to treasure: The impact of consumer perception of bio-waste products in closed-loop supply chains. J. Clean. Prod. 2019, 218, 966–974. [Google Scholar] [CrossRef]
| Type of Pretreatment | Effect | Notes | Reference |
|---|---|---|---|
| HNO2 | 270% short-chain VFA yield increase | Addition of 1.8 mg HNO2/L to influent sludge increased SCVFA production to 195.7 mg COD/g VSS with 6 d HRT Pretreatment accelerated cell and EPS disruption, enhanced sludge solubilization, hydrolysis and acidification, inhibiting methanogens activity | [33] |
| NH3 | 220% short-chain VFA yield increase | Addition of NH3 up to 176.5 mg/L increased SCVFA generation up to 267.2 mg COD/g VSS. Free ammonia accelerated breakdown of EPSs and of sludge cell envelope facilitating organics release | [34] |
| Na2SO3 | 1200% short-chain VFA yield increase | 500 mg sulfite-S/L at 4 d HRT, pH = 9.5 fermentation achieved SCVFA production of 324.8 mg COD/g VSS | [35] |
| K2FeO4 | 472% short-chain VFA yield increase | 56 mg Fe(VI)/g TSS and 5 d HRT increased SCVFA production to 343 mg COD/g VSS | [36] |
| NH3 + ultrasound | 171% short-chain VFA yield increase with US only, 421% with US and free ammonia | 2 w/mL US for 15 min followed by 60 mg/L NH3 for 2 d achieved SCVFA yield of 316.7 mg COD/g VSS | [37] |
| CER (cation-exchange resin) | 230% short-chain VFA yield increase | At dosage of 1.75 g CER/g SS sludge hydrolysis and subsequent SCVFAs production were enhanced by removing cations (Ca2+, Mg2+, Fe3+, Zn2+, etc.) from sludge, leading to EPS disruption and cell lysis. A total of 334.5 mg COD/g VSS SCFAs was produced with first 4 d HRT | [38] |
| S2O2− (Peroxydisulfate) thermally activated | 110% short-chain VFA yield increase | 0.3 mM PDS/g TSS, 9 d HRT at 55 °C achieved maximal SCFAs yield of 3183.51 mg COD/L | [39] |
| PHA Type | Description | Most Common Generating Organism |
|---|---|---|
| Short-chain (SCL-PHA) | Monomeric building blocks of 3–5 carbons (e.g., poly (3-hydroxyvalerate), PHV; poly (3-hydroxybutyrate), PHB). SCL-PHAs are rigid and brittle and lack the mechanical properties required for biomedical and packaging film applications | Cupriavidus necator |
| Medium-chain (MCL-PHA) | Monomeric units of 6–14 carbons (e.g., poly (3-hydroxyoctanoate), PHO). MCL-PHAs are elastomeric but with low mechanical strength, which limits their applications | Pseudomonas mendocina |
| Long-chain (LCL-PHA) | Monomer building blocks of 15 carbons or more (e.g., poly (3-hydroxypentadecanoate)) | Pseudomonas aeruginosa |
| Application Field | Description |
|---|---|
| Medical | Drug delivery Used as raw material for producing tablets, nanoparticles, etc. (e.g., PHB, PHVB and their copolymers used for delivery of antibiotics) Tissue engineering Used to build high mechanical strength scaffolds to promote cell growth through supplying nutrition (e.g., 3D scaffolds for cartilage repair developed on PHA nanofibers) other applications include soft tissue regeneration, nerve repair, cardiovascular treatments Wound dressings Volatile solutions of PHA are used as dressing to form a thin protective film over the wound to prevent contamination. |
| Agricultural | PHAs improve nutritional properties and shelf life of plants under stressful environments. Specialized applications of P(3HB) in agriculture include controlled release of pesticides and insecticides. Potential applications include biodegradable plastic films for crop protection, seeds and fertilizer encapsulation |
| Packaging | Due to their degradable nature, non-solubility, nonpermeable nature and flexibility, PHAs are developed as packaging films, mainly for use as shopping bags, paper coatings (e.g., PHA-coated boxes, paper, sheets, boards) |
| Consumer products | Manufacturing of paper cups, diapers, carpets, etc. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cecconet, D.; Capodaglio, A.G. Sewage Sludge Biorefinery for Circular Economy. Sustainability 2022, 14, 14841. https://doi.org/10.3390/su142214841
Cecconet D, Capodaglio AG. Sewage Sludge Biorefinery for Circular Economy. Sustainability. 2022; 14(22):14841. https://doi.org/10.3390/su142214841
Chicago/Turabian StyleCecconet, Daniele, and Andrea G. Capodaglio. 2022. "Sewage Sludge Biorefinery for Circular Economy" Sustainability 14, no. 22: 14841. https://doi.org/10.3390/su142214841
APA StyleCecconet, D., & Capodaglio, A. G. (2022). Sewage Sludge Biorefinery for Circular Economy. Sustainability, 14(22), 14841. https://doi.org/10.3390/su142214841







