Green Synthesis of Immobilized CuO Photocatalyst for Disinfection of Water
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
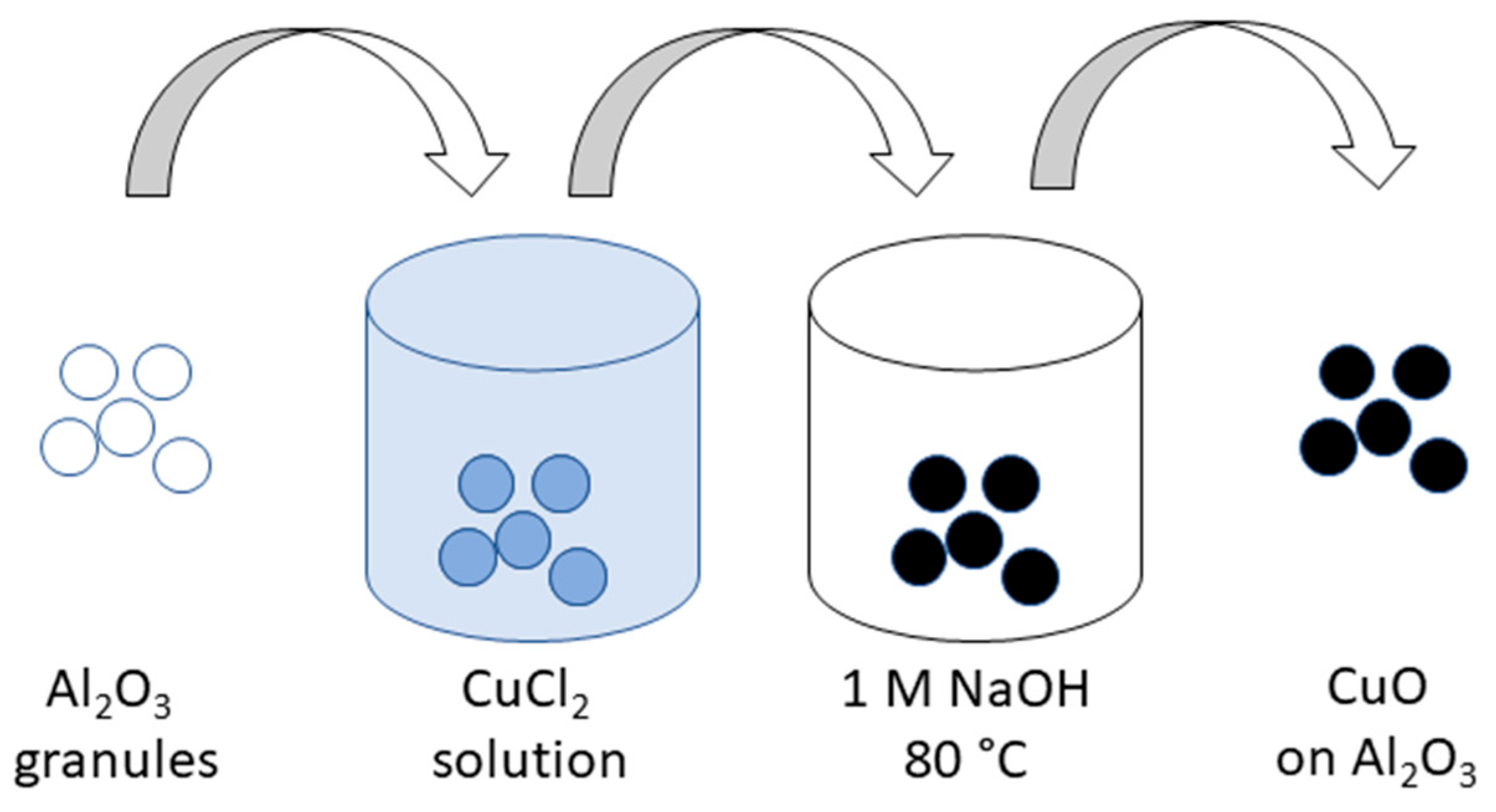
2.2. Synthesis
2.3. Characterization
3. Results and Discussion
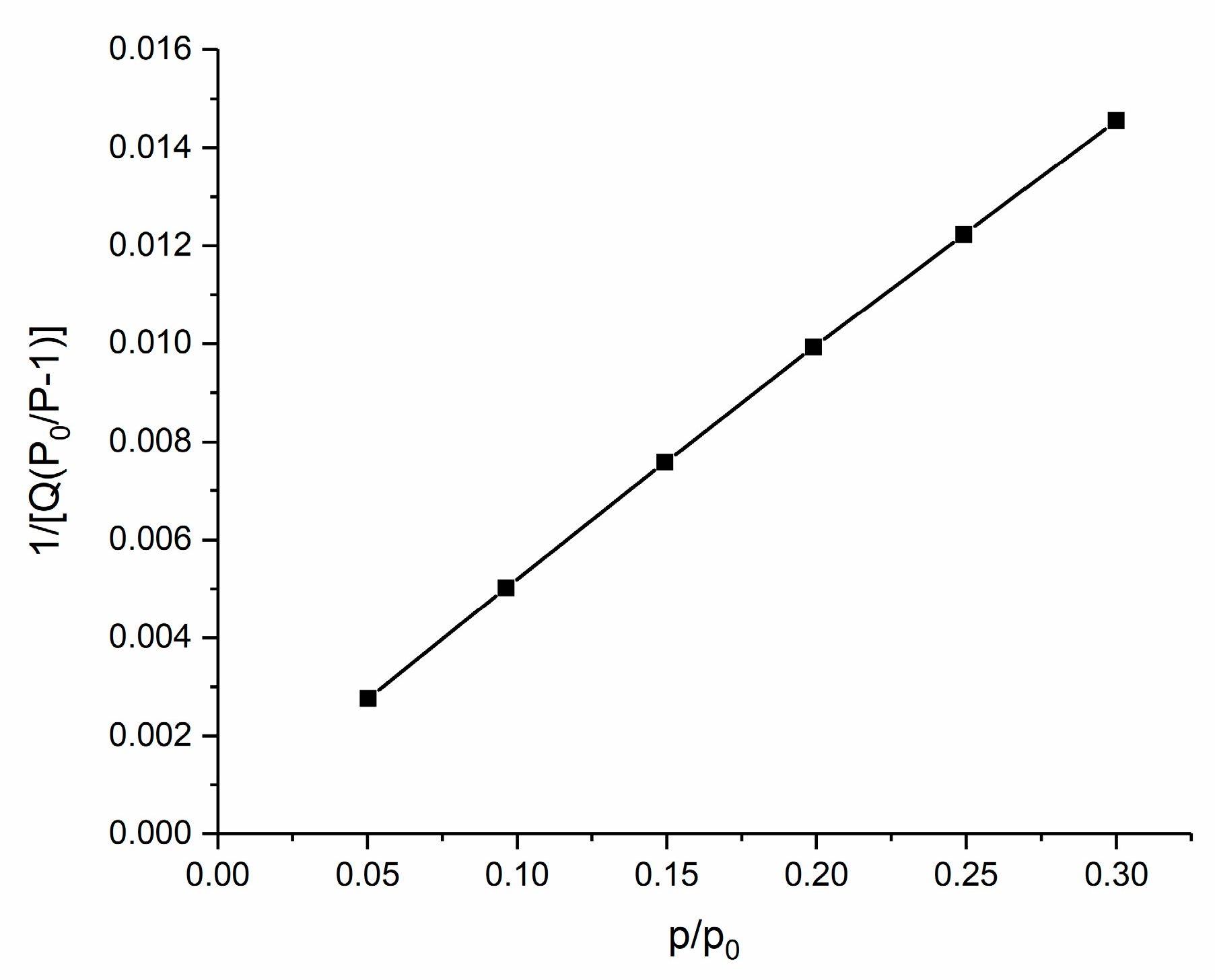
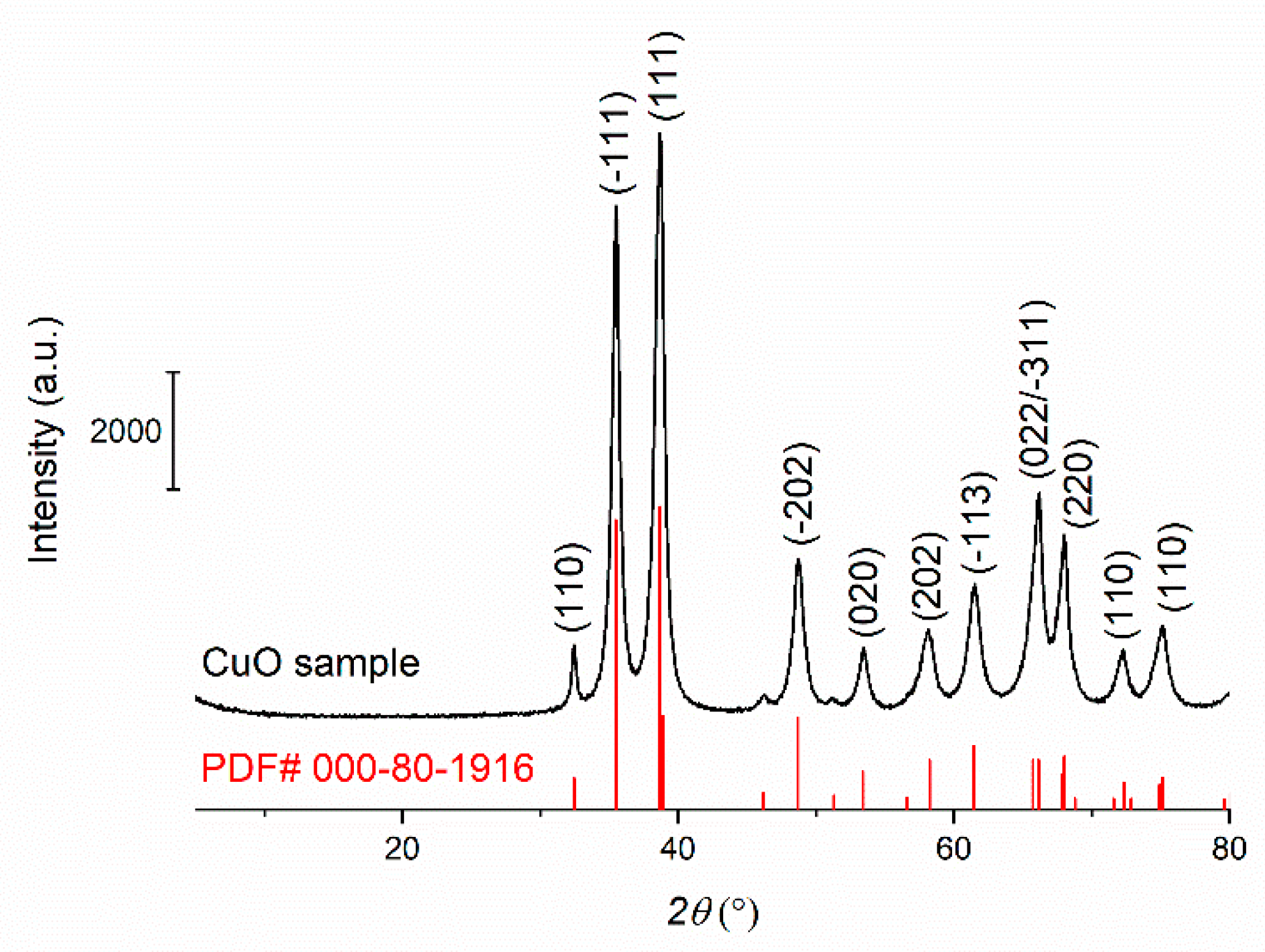
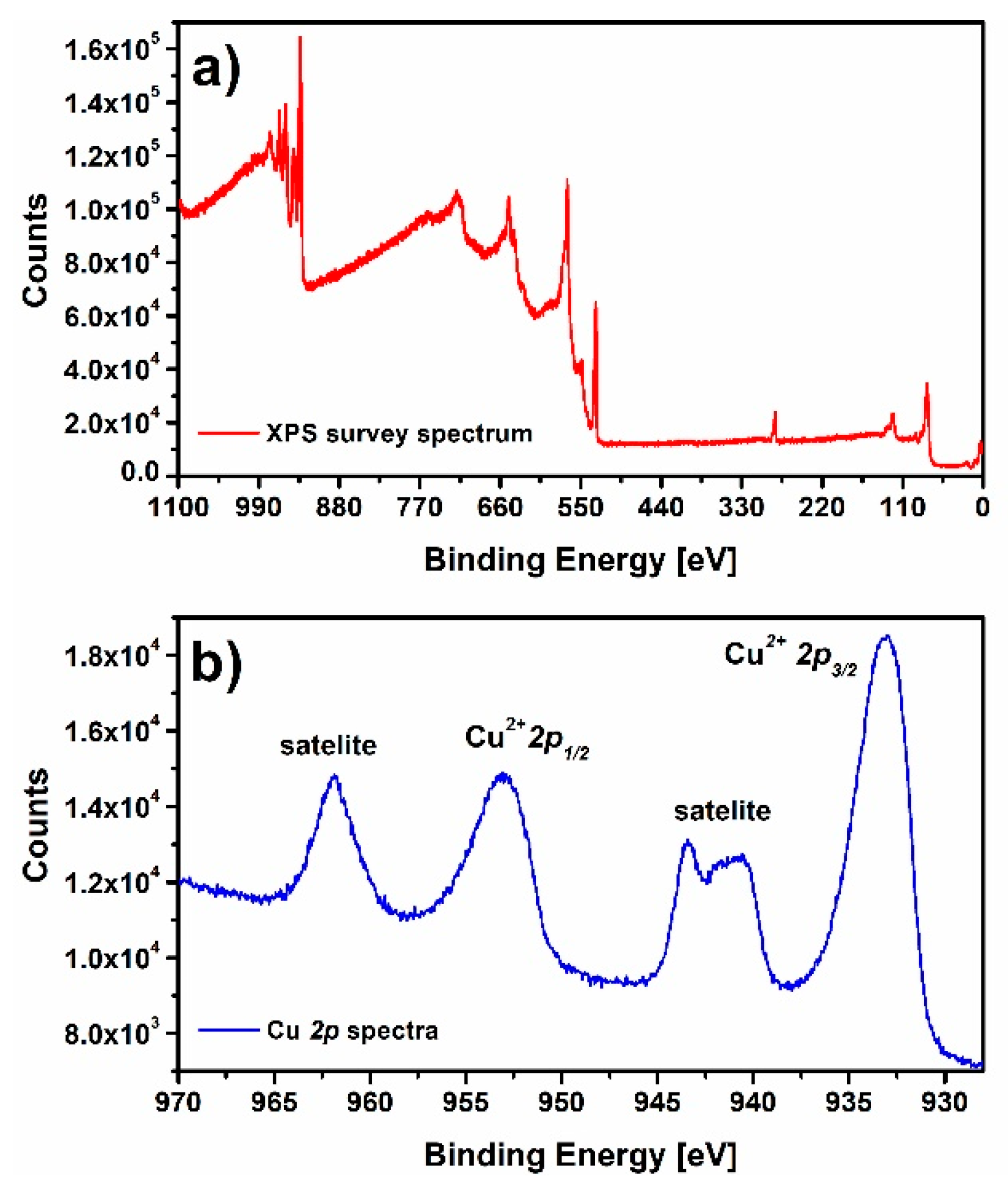
3.1. Material Characterization
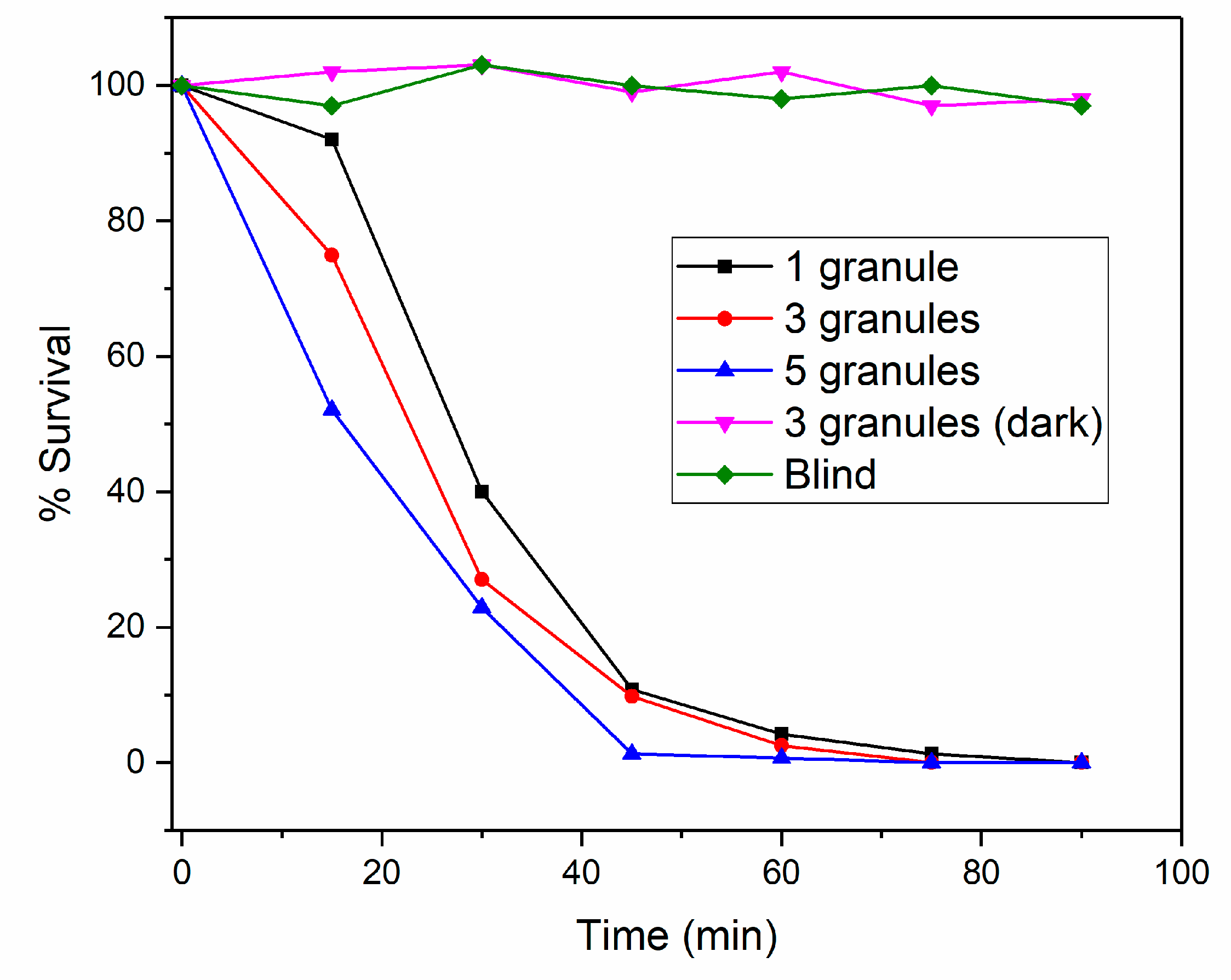
3.2. Disinfection Properties
3.3. ICP-MS Measurement
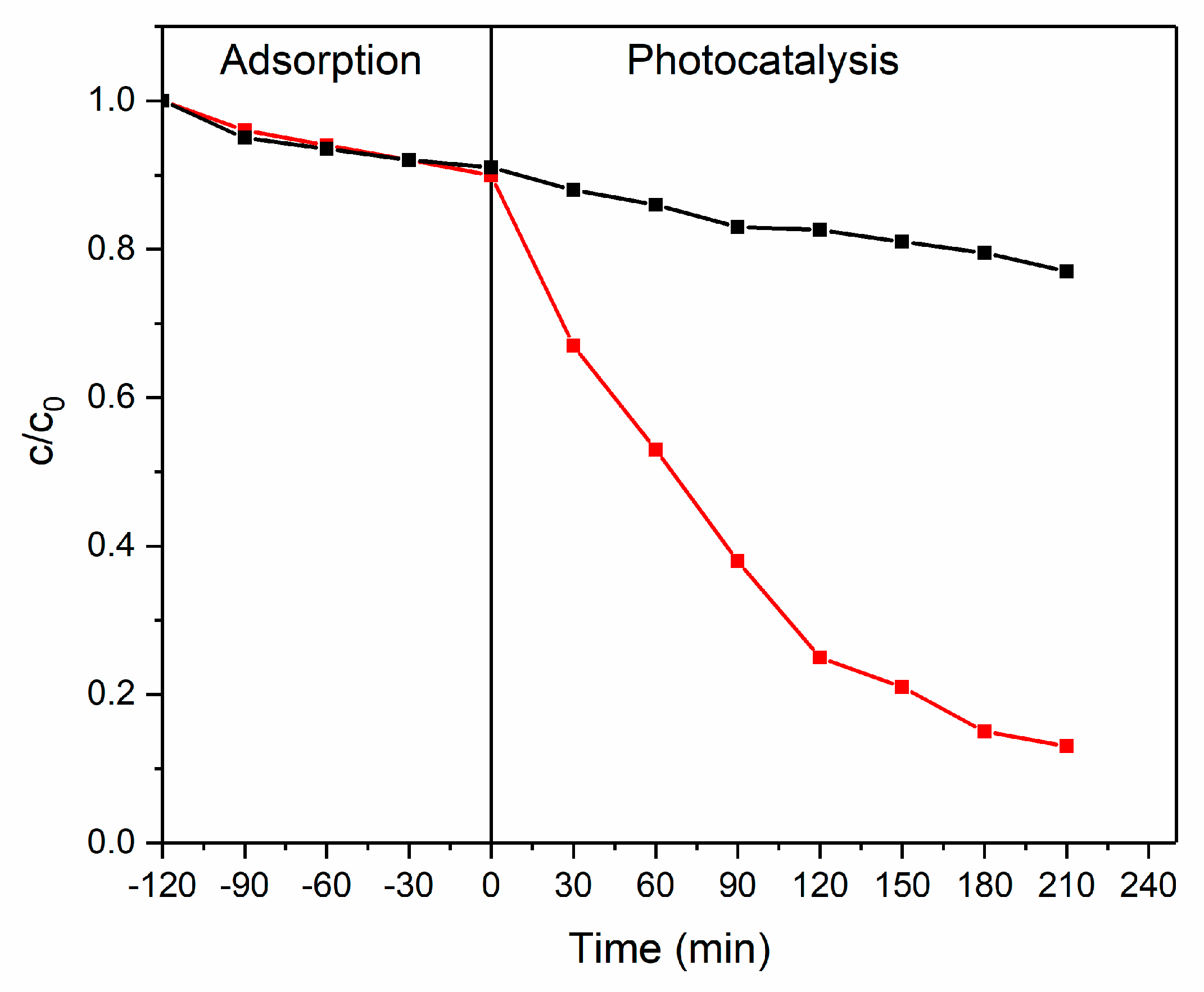
3.4. Photocatalytic Degradation
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Säve-Söderbergh, M.; Toljander, J.; Donat-Vargas, C.; Berglund, M.; Åkesson, A. Exposure to Drinking Water Chlorination By-Products and Fetal Growth and Prematurity: A Nation Wide Register-Based Prospective Study. Environ. Health Perspect. 2020, 128, 057006. [Google Scholar] [CrossRef] [PubMed]
- Boorman, G.A.; Dellarco, V.; Dunnick, J.K.; Chapin, R.E.; Hunter, S.; Hauchman, F.; Gardner, H.; Mike, C.; Sills, R.C. Drinking Water Disinfection Byproducts: Review and Approach to Toxicity Evaluation. Environ. Health Perspect. 1999, 107, 207–217. [Google Scholar] [CrossRef] [PubMed]
- Richardson, S.D.; Postigo, C. Drinking Water Disinfection By-Products. Handb. Environ. Chem. 2012, 20, 93–137. [Google Scholar] [CrossRef]
- You, J.; Guo, Y.; Guo, R.; Liu, X. A Review of Visible Light-Active Photocatalysts for Water Disinfection: Features and Prospects. Chem. Eng. J. 2019, 373, 624–641. [Google Scholar] [CrossRef]
- Sichel, C.; Blanco, J.; Malato, S.; Fernández-Ibáñez, P. Effects of Experimental Conditions on E.Coli Survival during Solar Photocatalytic Water Disinfection. J. Photochem. Photobiol. A Chem. 2007, 189, 239–246. [Google Scholar] [CrossRef]
- Liu, Y.; Zeng, X.; Hu, X.; Hu, J.; Zhang, X. Two-Dimensional Nanomaterials for Photocatalytic Water Disinfection: Recent Progress and Future Challenges. J. Chem. Technol. Biotechnol. 2019, 94, 22–37. [Google Scholar] [CrossRef]
- Zhang, D.; Li, G.; Yu, J.C. Inorganic Materials for Photocatalytic Water Disinfection. J. Mater. Chem. 2010, 20, 4529–4536. [Google Scholar] [CrossRef]
- Ahmed, S.N.; Haider, W. Heterogeneous Photocatalysis and Its Potential Applications in Water and Wastewater Treatment: A Review. Nanotechnology 2018, 29, 342001. [Google Scholar] [CrossRef]
- Djurišić, A.B.; He, Y.; Ng, A.M.C. Visible-Light Photocatalysts: Prospects and Challenges. APL Mater. 2020, 8, 030903. [Google Scholar] [CrossRef]
- Žener, B.; Matoh, L.; Carraro, G.; Miljević, B.; Korošec, R.C. Sulfur-, Nitrogen- and Platinum-Doped Titania Thin Films with High Catalytic Efficiency under Visible-Light Illumination. Beilstein J. Nanotechnol. 2018, 9, 1629–1640. [Google Scholar] [CrossRef] [Green Version]
- Anandan, S.; Ikuma, Y.; Niwa, K. An Overview of Semi-Conductor Photocatalysis: Modification of TiO2 Nanomaterials. Solid State Phenom. 2010, 162, 239–260. [Google Scholar] [CrossRef]
- Chen, D.; Cheng, Y.; Zhou, N.; Chen, P.; Wang, Y.; Li, K.; Huo, S.; Cheng, P.; Peng, P.; Zhang, R.; et al. Photocatalytic Degradation of Organic Pollutants Using TiO2-Based Photocatalysts: A Review. J. Clean. Prod. 2020, 268, 121725. [Google Scholar] [CrossRef]
- Matoh, L.; Žener, B.; Korošec, R.C.; Štangar, U.L. Photocatalytic Water Treatment. In Nanotechnology in Eco-Efficient Construction; Pacheco-Torgal, F., Vittoria Diamanti, M., Nazari, A., Goran Granqvist, C., Pruna, A., Amirkhanian, S., Eds.; Elsevier: Duxford, UK, 2019; Volume 4, pp. 675–702. ISBN 9780081026410. [Google Scholar]
- Šuligoj, A.; Arčon, I.; Mazaj, M.; Dražić, G.; Arčon, D.; Cool, P.; Štangar, U.L.; Tušar, N.N. Surface Modified Titanium Dioxide Using Transition Metals: Nickel as a Winning Transition Metal for Solar Light Photocatalysis. J. Mater. Chem. A 2018, 6, 9882–9892. [Google Scholar] [CrossRef]
- Chiam, S.L.; Pung, S.Y.; Yeoh, F.Y. Recent Developments in MnO2-Based Photocatalysts for Organic Dye Removal: A Review. Environ. Sci. Pollut. Res. 2020, 27, 5759–5778. [Google Scholar] [CrossRef]
- Rao, M.P.; Sathishkumar, P.; Mangalaraja, R.V.; Asiri, A.M.; Sivashanmugam, P.; Anandan, S. Simple and Low-Cost Synthesis of CuO Nanosheets for Visible-Light-Driven Photocatalytic Degradation of Textile Dyes. J. Environ. Chem. Eng. 2018, 6, 2003–2010. [Google Scholar] [CrossRef]
- Katal, R.; Masudy-panah, S.; Kong, E.Y.J.; Dasineh Khiavi, N.; Abadi Farahani, M.H.D.; Gong, X. Nanocrystal-Engineered Thin CuO Film Photocatalyst for Visible-Light-Driven Photocatalytic Degradation of Organic Pollutant in Aqueous Solution. Catal. Today 2020, 340, 236–244. [Google Scholar] [CrossRef]
- Zhang, Q.; Zhang, K.; Xu, D.; Yang, G.; Huang, H.; Nie, F.; Liu, C.; Yang, S. CuO Nanostructures: Synthesis, Characterization, Growth Mechanisms, Fundamental Properties, and Applications. Prog. Mater. Sci. 2014, 60, 208–337. [Google Scholar] [CrossRef]
- Raizada, P.; Sudhaik, A.; Patial, S.; Hasija, V.; Parwaz Khan, A.A.; Singh, P.; Gautam, S.; Kaur, M.; Nguyen, V.H. Engineering Nanostructures of CuO-Based Photocatalysts for Water Treatment: Current Progress and Future Challenges. Arab. J. Chem. 2020, 13, 8424–8457. [Google Scholar] [CrossRef]
- Sahu, K.; Choudhary, S.; Khan, S.A.; Pandey, A.; Mohapatra, S. Thermal Evolution of Morphological, Structural, Optical and Photocatalytic Properties of CuO Thin Films. Nano-Struct. Nano-Objects 2019, 17, 92–102. [Google Scholar] [CrossRef]
- Kumar, K.; Priya, A.; Arun, A.; Hait, S.; Chowdhury, A. Antibacterial and Natural Room-Light Driven Photocatalytic Activities of CuO Nanorods. Mater. Chem. Phys. 2019, 226, 106–112. [Google Scholar] [CrossRef]
- Momeni, M.M.; Mirhosseini, M.; Nazari, Z.; Kazempour, A.; Hakimiyan, M. Antibacterial and Photocatalytic Activity of CuO Nanostructure Films with Different Morphology. J. Mater. Sci. Mater. Electron. 2016, 27, 8131–8137. [Google Scholar] [CrossRef]
- Rao, M.P.; Wu, J.J.; Asiri, A.M.; Anandan, S.; Ashokkumar, M. Photocatalytic Properties of Hierarchical CuO Nanosheets Synthesized by a Solution Phase Method. J. Environ. Sci. 2018, 69, 115–124. [Google Scholar] [CrossRef] [PubMed]
- Alishah, H.; Pourseyedi, S.; Ebrahimipour, S.Y.; Mahani, S.E.; Rafiei, N. Green Synthesis of Starch-Mediated CuO Nanoparticles: Preparation, Characterization, Antimicrobial Activities and in Vitro MTT Assay against MCF-7 Cell Line. Rend. Lincei 2017, 28, 65–71. [Google Scholar] [CrossRef]
- Mageshwari, K.; Sathyamoorthy, R. Flower-Shaped CuO Nanostructures: Synthesis, Characterization and Antimicrobial Activity. J. Mater. Sci. Technol. 2013, 29, 909–914. [Google Scholar] [CrossRef]
- Devi, A.B.; Moirangthem, D.S.; Talukdar, N.C.; Devi, M.D.; Singh, N.R.; Luwang, M.N. Novel Synthesis and Characterization of CuO Nanomaterials: Biological Applications. Chin. Chem. Lett. 2014, 25, 1615–1619. [Google Scholar] [CrossRef]
- Wang, Y.; Jiang, T.; Meng, D.; Jin, H.; Yu, M. Controllable Fabrication of Nanowire-like CuO Film by Anodization and Its Properties. Appl. Surf. Sci. 2015, 349, 636–643. [Google Scholar] [CrossRef]
- Dar, M.A.; Nam, S.H.; Kim, Y.S.; Kim, W.B. Synthesis, Characterization, and Electrochemical Properties of Self-Assembled Leaf-like CuO Nanostructures. J. Solid State Electrochem. 2010, 14, 1719–1726. [Google Scholar] [CrossRef]
- Arshad, A.; Iqbal, J.; Siddiq, M.; Ali, M.U.; Ali, A.; Shabbir, H.; Nazeer, U.B.; Saleem, M.S. Solar Light Triggered Catalytic Performance of Graphene-CuO Nanocomposite for Waste Water Treatment. Ceram. Int. 2017, 43, 10654–10660. [Google Scholar] [CrossRef]
- Nair, M.T.S.; Guerrero, L.; Arenas, O.L.; Nair, P.K. Chemically Deposited Copper Oxide Thin Films: Structural, Optical and Electrical Characteristics. Appl. Surf. Sci. 1999, 150, 143–151. [Google Scholar] [CrossRef]
- Filipič, G.; Cvelbar, U. Copper Oxide Nanowires: A Review of Growth. Nanotechnology 2012, 23, 194001. [Google Scholar] [CrossRef]
- Christy, A.J.; Nehru, L.C.; Umadevi, M. A Novel Combustion Method to Prepare CuO Nanorods and Its Antimicrobial and Photocatalytic Activities. Powder Technol. 2013, 235, 783–786. [Google Scholar] [CrossRef]
- Biesinger, M.C. Advanced Analysis of Copper X-ray Photoelectron Spectra. Surf. Interface Anal. 2017, 49, 1325–1334. [Google Scholar] [CrossRef]
- Losev, A.; Rostov, K.; Tyuliev, G. Electron Beam Induced Reduction of CuO in the Presence of a Surface Carbonaceous Layer: An XPS/HREELS Study. Surf. Sci. 1989, 213, 564–579. [Google Scholar] [CrossRef]
- Fewtrell, L.; Kay, D.; MacGill, S. A Review of the Science behind Drinking Water Standards for Copper. Int. J. Environ. Health Res. 2001, 11, 161–167. [Google Scholar] [CrossRef]
- World Health Organization & International Programme on Chemical Safety. Guidelines for Drinking-Water Quality, 2nd ed.; Health criteria and other supporting information; World Health Organization: Geneva, Switzerland, 1996; Volume 2. [Google Scholar]



Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Matoh, L.; Žener, B.; Genorio, B. Green Synthesis of Immobilized CuO Photocatalyst for Disinfection of Water. Sustainability 2022, 14, 10581. https://doi.org/10.3390/su141710581
Matoh L, Žener B, Genorio B. Green Synthesis of Immobilized CuO Photocatalyst for Disinfection of Water. Sustainability. 2022; 14(17):10581. https://doi.org/10.3390/su141710581
Chicago/Turabian StyleMatoh, Lev, Boštjan Žener, and Boštjan Genorio. 2022. "Green Synthesis of Immobilized CuO Photocatalyst for Disinfection of Water" Sustainability 14, no. 17: 10581. https://doi.org/10.3390/su141710581
APA StyleMatoh, L., Žener, B., & Genorio, B. (2022). Green Synthesis of Immobilized CuO Photocatalyst for Disinfection of Water. Sustainability, 14(17), 10581. https://doi.org/10.3390/su141710581







