Microbial Diversity and Adaptation under Salt-Affected Soils: A Review
Abstract
1. Introduction
2. Ecology of Saline Soil Microorganisms
3. Interaction of Plants and Microbes in Salt-Affected Soils
4. Application Strategy of Halophilic Microbes
4.1. Halophilic Bacteria
4.2. Taxonomy of Halophilic Bacteria
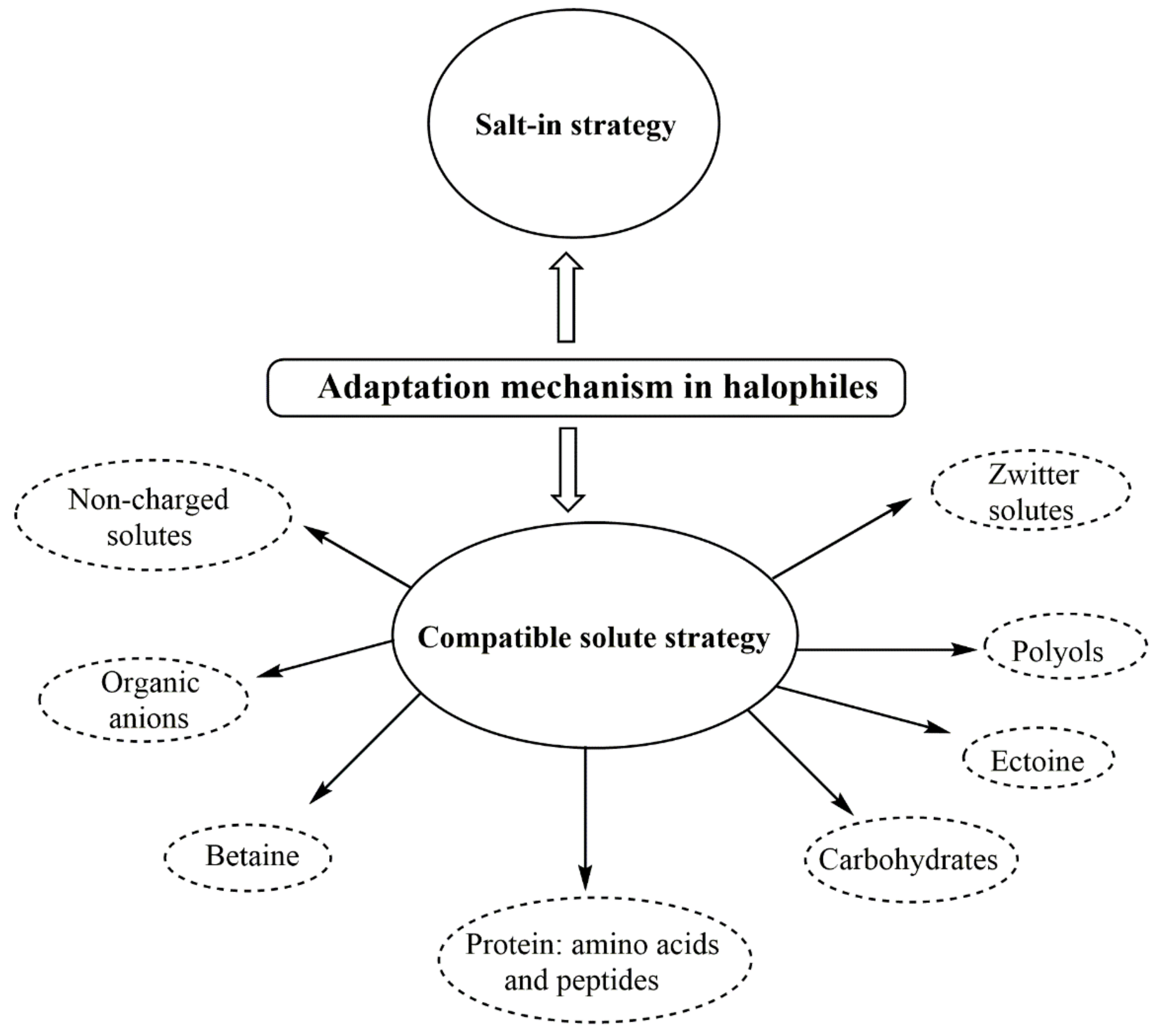
4.3. Adaptability Mechanisms of Halophilic Bacteria for Saline Environments
5. Halophilic Bacteria: Role of Halophilic Bacteria in Plant Growth Promotion under Salt Stress
5.1. The Role of Bacterial Phytohormones
5.2. Aminocyclopropane-1-Carboxylate (ACC) Deaminase
5.3. Phosphate Solubilization
5.4. Antioxidative Activity
5.5. Siderophore Producers
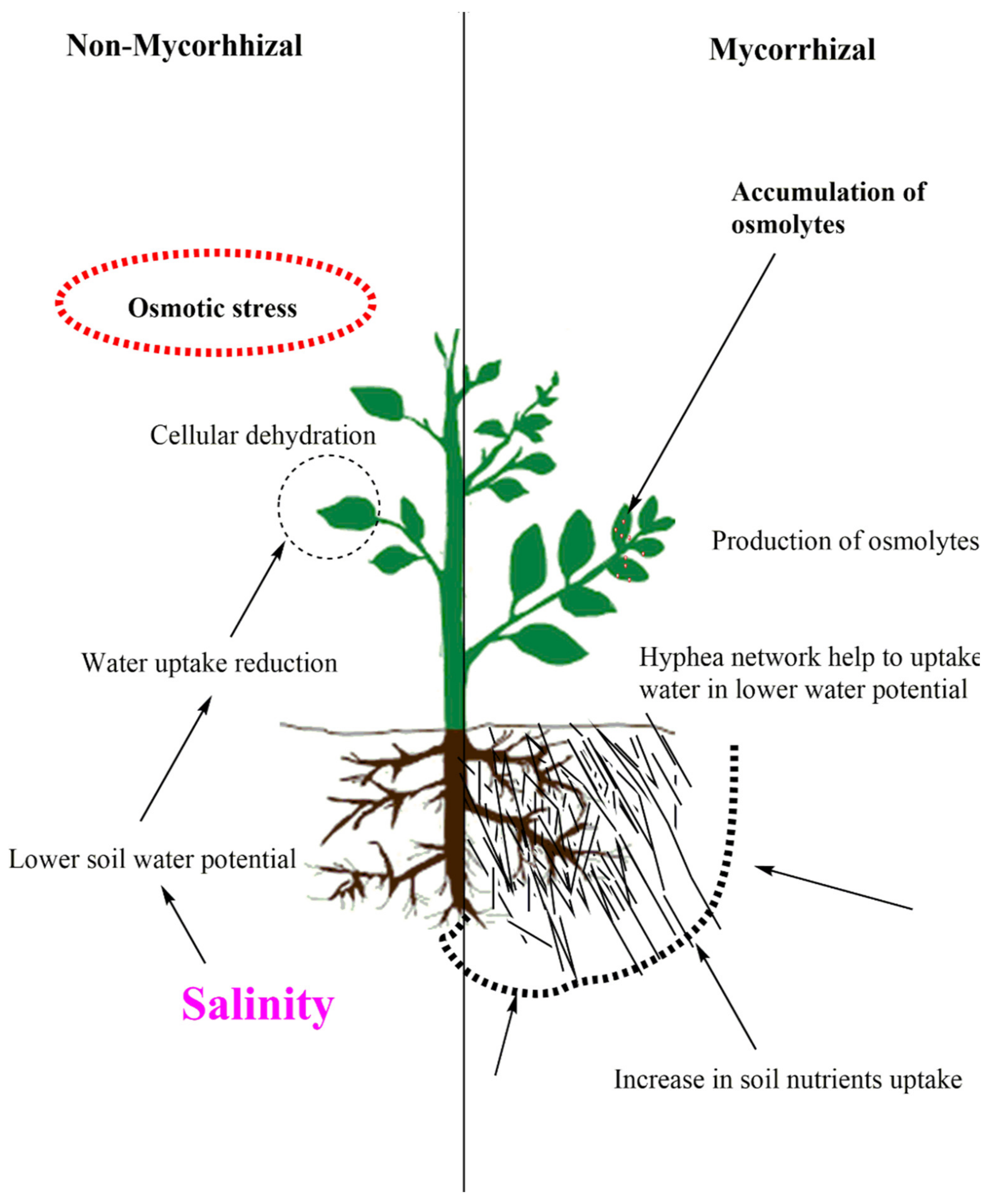
6. Arbuscular Mycorrhizal Fungi
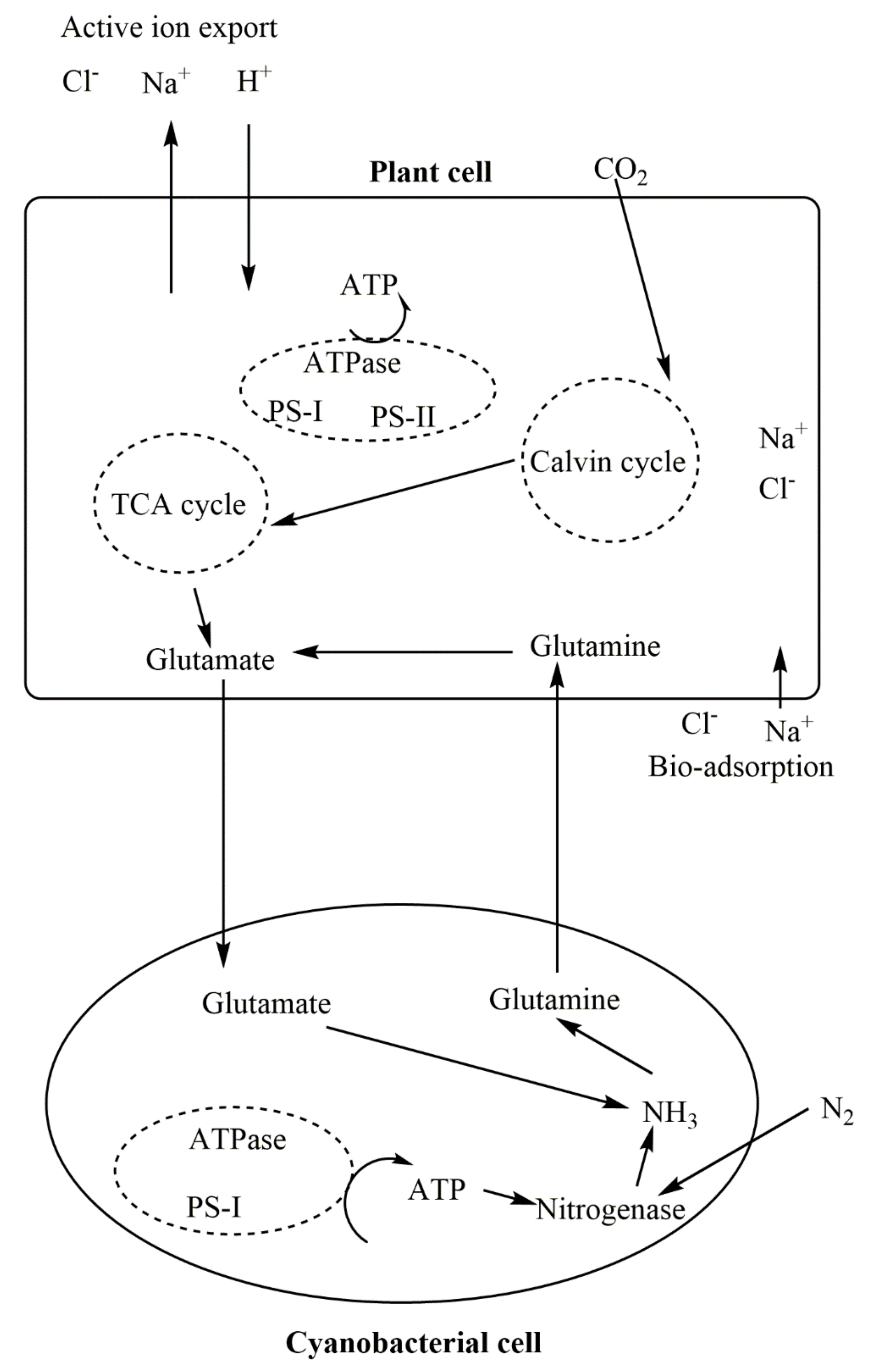
7. Cyanobacteria
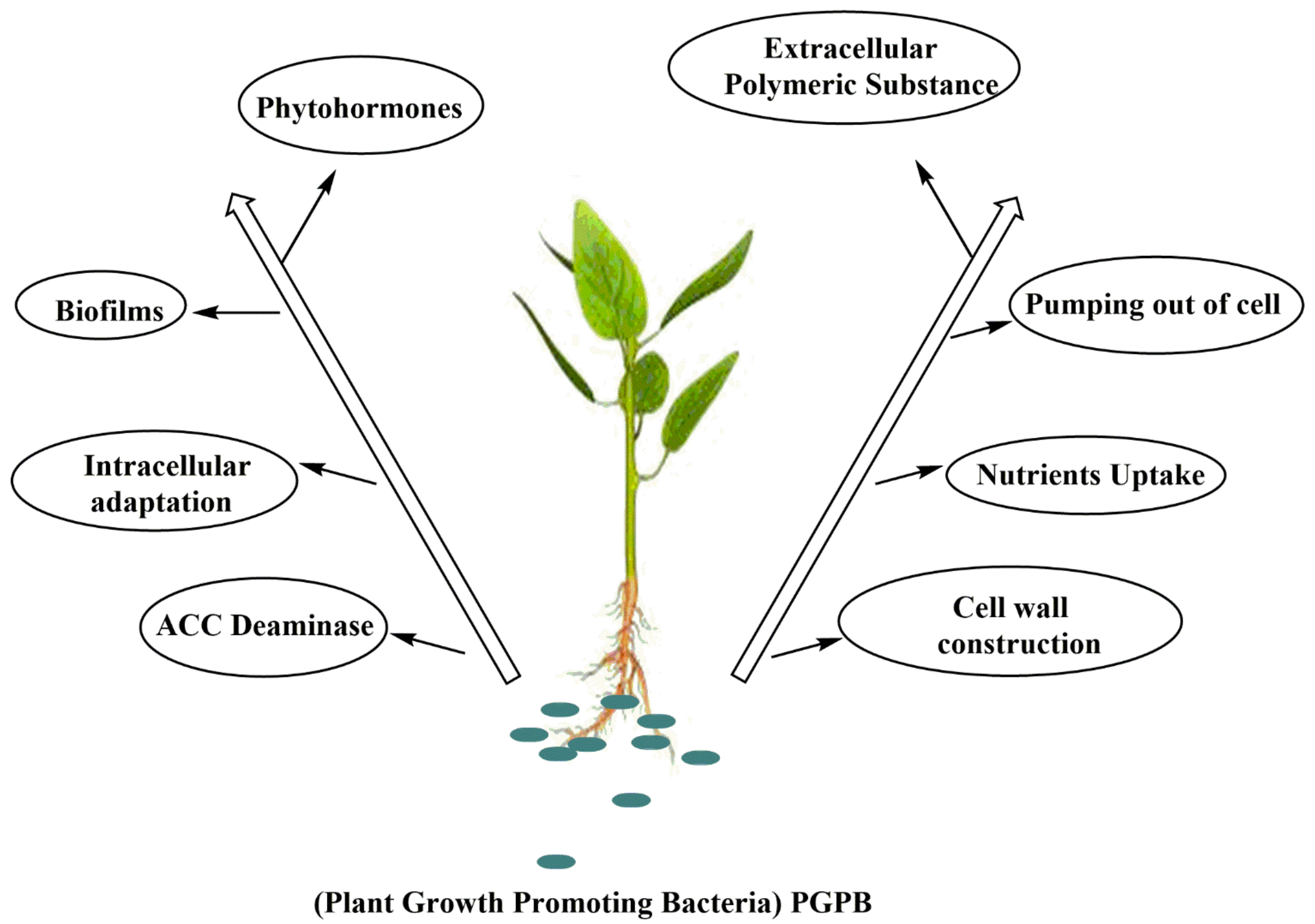
8. Plant Growth-Promoting Bacteria
8.1. Production of Phytohormone and ACC-Deaminase Activity
8.2. Production of Extracellular Polymeric Substance
8.3. Production of Plant Osmolytes and Antioxidant Activity
8.4. Siderophore Production
8.5. Induced Systemic Resistance
8.6. Essential Nutrient Uptake
9. Microbial Inoculation Influencing Soil Properties
10. Future Challenges for Salt Stress Mitigation through Halophilic Microbes
11. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- GAP Report. Global Agricultural Productivity Report (GAP Report); Global Harvest Initiative: Washington, DC, USA, 2018. [Google Scholar]
- Ahmad, P. Oxidative Damage to Plants: Antioxidant Networks and Signaling; Academic Press: Cambridge, MA, USA, 2014. [Google Scholar]
- Kumar, P.; Sharma, P.K. Soil salinity and food security in India. Front. Sustain. Food Syst. 2020, 4, 533781. [Google Scholar] [CrossRef]
- Abedin, M.A.; Habiba, U.; Shaw, R. Salinity Scenario in Mekong, Ganges, and Indus River Deltas Water Insecurity: A Social Dilemma; Emerald Group Publishing Limited: Bingley, UK, 2014; pp. 115–138. [Google Scholar]
- Szabo, S.; Hossain, M.S.; Adger, W.N.; Matthews, Z.; Ahmed, S.; Lázár, A.N. Soil salinity, household wealth and food insecurity in tropical deltas: Evidence from south-west coast of Bangladesh. Sustain. Sci. 2016, 11, 411–421. [Google Scholar] [CrossRef] [PubMed]
- Ghassemi, F.; Jakeman, A.J.; Nix, H.A. Salinisation of Land and Water Resources: Human Causes, Extent, Management and Case Studies; CAB International: Wallingford, UK, 1995. [Google Scholar]
- Schirawski, J.; Perlin, M.H. Plant-microbe interaction 2017-the good, the bad and the diverse. Int. J. Mol. Sci. 2018, 19, 1374. [Google Scholar] [CrossRef]
- Rütting, T.; Aronsson, H.; Elin, S. Efficient use of nitrogen in agriculture. Nutr. Cycl. Agroecosys. 2018, 110, 1–5. [Google Scholar] [CrossRef]
- Richards, L.A. Diagnosis and Improvement of Saline and Alkali Soils, U.S.D.A. Agriculture Handbook No. 60; US Department of Agriculture: Washington, DC, USA, 1954; p. 158.
- Ayyam, V.; Palanivel, S.; Chandrakasan, S. Approaches in land degradation management for productivity enhancement. In Coastal Ecosystems of the Tropics—Adaptive Management; Springer: Singapore, 2019. [Google Scholar]
- Fita, A.; Rodriíguez-Burruezo, A.; Boscaiu, M.; Prohens, J.; Vicente, O. Breeding and domesticating crops adapted to drought and salinity: A new paradigm for increasing food production. Front. Plant Sci. 2015, 6, 978. [Google Scholar] [CrossRef] [PubMed]
- Morton, M.J.; Awlia, M.; Al-Tamimi, N.; Saade, S.; Pailles, Y.; Negrão, S. Salt stress under the scalpel-dissecting the genetics of salt tolerance. Plant J. 2019, 97, 148–163. [Google Scholar] [CrossRef]
- Nina, K.; Thomas, W.K.; Prem, S.B. Beneficial Organisms for Nutrient Uptake. VFRC Report 2014/1, Virtual Fertilizer Research Center; Wageningen Academic Publishers: Washington, DC, USA, 2014; p. 63. [Google Scholar]
- Sahoo, R.K.; Ansari, M.W.; Dangar, T.K.; Mohanty, S.; Tuteja, N. Phenotypic and molecular characterization of efficient nitrogen fixing Azotobacter strains of the rice fields. Protoplasma 2013, 251, 511–523. [Google Scholar] [CrossRef]
- Grover, M.; Ali, S.Z.; Sandhya, V.; Rasul, A.; Venkateswarlu, B. Role of microorganisms in adaptation of agriculture crops to abiotic stresses. World J. Microbiol. Biotechnol. 2011, 27, 1231–1240. [Google Scholar] [CrossRef]
- Schimel, J.P.; Balser, T.C.; Wallenstein, M. Microbial stress response physiology and its implications for ecosystem function. Ecology 2007, 88, 1386–1394. [Google Scholar] [CrossRef]
- Yuan, B.C.; Li, Z.Z.; Liu, H.; Gao, M.; Zhang, Y.Y. Microbial biomass and activity in salt affected soils under arid conditions. Appl. Soil Ecol. 2007, 35, 319–328. [Google Scholar] [CrossRef]
- Ibekwe, A.M.; Poss, J.A.; Grattan, S.R.; Grieve, C.M.; Suarez, D. Bacterial diversity in cucumber (Cucumis sativus) rhizosphere in response to salinity, soil pH, and boron. Soil Biol. Biochem. 2010, 42, 567–575. [Google Scholar] [CrossRef]
- Chowdhury, N.; Marschner, P.; Burns, R.G. Soil microbial activity and community composition: Impact of changes in matric and osmotic potential. Soil Biol. Biochem. 2011, 43, 1229–1236. [Google Scholar] [CrossRef]
- Oren, A. Molecular ecology of extremely halophilic archaea and bacteria. FEMS Microbiol. Ecol. 2002, 39, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Jiang, H.; Dong, H.; Yu, B.; Liu, X.; Li, Y.; Ji, S.; Zhang, C.L. Microbial response to salinity change in Lake Chaka, a hypersaline lake on Tibetan plateau. Environ. Microbiol. 2007, 9, 2603–2621. [Google Scholar] [CrossRef] [PubMed]
- Wood, J.M. Bacterial Osmoregulation: A Paradigm for the Study of Cellular Homeostasis. Annu. Rev. Microbiol. 2011, 65, 215–238. [Google Scholar] [CrossRef]
- Tester, M.; Davenport, R. Na+ tolerance and Na+ transport in higher plants. Ann. Bot. 2003, 91, 503–527. [Google Scholar] [CrossRef]
- Tavakkoli, E.; Rengasamy, P.; McDonald, G.K. High concentrations of Na+ and Clions in soil solution have simultaneous detrimental effects on growth of faba bean under salinity stress. J. Exp. Bot. 2010, 61, 4449–4459. [Google Scholar] [CrossRef]
- Kushner, D.J. Growth and nutrition of halophilic bacteria. In The Biology of Halophilic Bacteria; Vreeland, R.H., Hochstein, L.I., Eds.; CRC Press: Boca Raton, FL, USA, 1993; pp. 87–89. [Google Scholar]
- Arahal, D.R.; Ventosa, A. Moderately halophilic and halotolerant species of Bacillus and related genera. In Applications and Systematic of Bacillus and Relatives; Berkeley, R., Heyndrickx, M., Logan, N., De Vos, P., Eds.; Wiley: Hoboken, NJ, USA, 2002; pp. 83–99. [Google Scholar]
- Ventosa, A.; Nieto, J.J.; Oren, A. Biology of moderately halophilic aerobic bacteria. Microbiol. Mol. Biol. Rev. 1998, 62, 504–544. [Google Scholar] [CrossRef]
- Yoon, J.H.; Kim, I.G.; Kang, K.H.; Oh, T.K.; Park, Y.H. Bacillus marisflavi sp. nov. and Bacillus aquimaris sp. nov. isolated from sea water of a tidal flat of the yellow sea in Korea. Int. J. Syst. Evol. Microbiol. 2003, 53, 1297–1303. [Google Scholar] [CrossRef]
- Rodriguez-Valera, F. Characteristics and microbial ecology of hypersaline environments. In Halophilic Bacteria; Rodriguez-Valera, F., Ed.; CRC Press: Boca Raton, FL, USA, 1988; Volume 1, pp. 3–30. [Google Scholar]
- Garabito, M.J.; Marquez, M.C.; Ventosa, A. Halotolerant Bacillus diversity in hypersaline environments. Can. J. Microbiol. 1998, 44, 95–102. [Google Scholar] [CrossRef]
- Pankhurst, C.E.; Yu, S.; Hawke, B.G.; Harch, B.D. Capacity of fatty acid profiles and substrate utilization patters to describe differences in soil microbial communities associated with increased salinity or alkalinity at three locations in South Australia. Biol. Fertil. Soils 2001, 33, 204–217. [Google Scholar] [CrossRef]
- Gros, R.; Poly, F.; Jocteur-Monrozier, L.; Faivre, P. Plant and soil microbial community responses to solid waste leachates diffusion on grassland. Plant Soil 2003, 255, 445–455. [Google Scholar] [CrossRef]
- Gennari, M.; Abbate, C.; La Porta, V.; Baglieri, A.; Cignetti, A. Microbial response to Na2SO4 additions in a volcanic soil. Arid Land Res. Manag. 2007, 21, 211–227. [Google Scholar] [CrossRef]
- Llamas, D.P.; Gonzales, M.D.; Gonzales, C.I.; Lopez, G.R.; Marquina, J.C. Effects of water potential on spore germination and viability of Fusarium species. J. Ind. Microbiol. Biotechnol. 2008, 35, 1411–1418. [Google Scholar] [CrossRef]
- Mandeel, Q.A. Biodiversity of the genus Fusarium in saline soil habitats. J. Basic Microbiol. 2006, 46, 480–494. [Google Scholar] [CrossRef]
- Juniper, S.; Abbott, L.K. Soil salinity delays germination and limits growth of hyphae from propagules of arbuscular mycorrhizal fungi. Mycorrhiza 2006, 16, 371–379. [Google Scholar] [CrossRef]
- Liang, Y.; Chen, H.; Tang, M.J.; Shen, S.H. Proteome analysis of an ectomycorrhizal fungus Boletus edulis under salt shock. Mycol. Res. 2007, 111, 939–946. [Google Scholar] [CrossRef]
- Sardinha, M.; Muller, T.; Schmeisky, H.; Joergensen, R.G. Microbial performance in soils along a salinity gradient under acidic conditions. Appl. Soil Ecol. 2003, 23, 237–244. [Google Scholar] [CrossRef]
- Wichern, J.; Wichern, F.; Joergensen, R.G. Impact of salinity on soil microbial communities and the decomposition of maize in acidic soils. Geoderma 2006, 137, 100–108. [Google Scholar] [CrossRef]
- Omar, S.A.; Abdel-Sater, M.A.; Khallil, A.M.; Abdalla, M.H. Growth and enzyme activities of fungi and bacteria in soil salinized with sodium chloride. Folia Microbiol. 1994, 39, 23–28. [Google Scholar] [CrossRef]
- Herrmann, A.; Witter, E.; Katterer, T. A method to assess whether ‘preferential use’ occurs after 15N ammonium addition: Implication for the 15N isotope dilution technique. Soil Biol. Biochem. 2005, 37, 183–186. [Google Scholar] [CrossRef]
- Azam, F.; Ifzal, M. Microbial populations immobilizing NH4+-N and NO3−-N differ in their sensitivity to sodium chloride salinity in soil. Soil Biol. Biochem. 2006, 38, 2491–2494. [Google Scholar] [CrossRef]
- Whatmore, A.M.; Chudek, J.A.; Reed, R.H. The effects of osmotic cupshock on the intracellular solute pools of Bacillus subtilis. J. Gen. Microbiol. 1990, 136, 2527–2535. [Google Scholar] [CrossRef] [PubMed]
- Bekki, A.; Trinchant, J.C.; Rigaud, J. Nitrogen fixation (C2H4 reduction) by Medicago nodules and bacteroids under sodium chloride stress. Physiol. Plant. 1987, 71, 61–67. [Google Scholar] [CrossRef]
- Yousef, A.N.; Sprent, J.I. Effect of NaCl on growth, nitrogen incorporation and chemical composition of inoculated and NH4NO3 fertilized Vicia faba L. plants. J. Exp. Bot. 1983, 143, 941–950. [Google Scholar] [CrossRef]
- Wignarajah, K. Growth response of Phaseolus vulgaris to varying salinity regimes. Environ. Exp. Bot. 1990, 2, 141–147. [Google Scholar] [CrossRef]
- Cordovilla, M.P.; Ocana, A.; Ligero, F.; Liuch, C. Salinity effects on grouth analysis and nutrient composition in four grain legumes-Rhyzobium symbiosis. J. Plant Nutr. 1995, 8, 1595–1609. [Google Scholar] [CrossRef]
- Tu, J.C. Effect of salinity on Rhizobium-root hair interaction, nodulation and growth of soybean. Can. J. Plant Sci. 1981, 61, 231–239. [Google Scholar] [CrossRef]
- Bernard, T.; Pocard, J.; Perroud, B.; Le Redulier, P. Variation in the response of salt stressed Rhizobium strains to betaine. Arch. Microbiol. 1986, 143, 359–364. [Google Scholar] [CrossRef]
- Singleton, P.W.; Swaify, S.A.; Bohlool, B.B. Effect of salinity on Rhizobium growth and survival. Appl. Environ. Microbiol. 1982, 44, 884–890. [Google Scholar] [CrossRef]
- Yelton, M.M.; Yang, S.S.; Edie, S.A.; Lim, S.T. Characterzation of an effective salt tolerant fast-growing strain of Rhizobium japonicum. J. Gen. Microbiol. 1983, 129, 1537–1547. [Google Scholar]
- Zhang, X.; Harper, R.; Karsisto, M.; Lindström, K. Diversity of Rhizobium bacteria isolated from the root nodules of leguminous trees. Int. J. Syst. Bacteriol. 1991, 41, 104–113. [Google Scholar] [CrossRef]
- Graham, P.H.; Parker, C.A. Diagnostic features in the characterization of the root nodule bacteria of legumes. Plant Soil 1964, 20, 383–396. [Google Scholar] [CrossRef]
- Sauvage, D.; Hamelin, J.; Larher, F. Glycine betaine and other structurally related compounds improve the salt tolerance of Rhizobium meliloti. Plant Sci. Lett. 1983, 31, 291–302. [Google Scholar] [CrossRef]
- Graham, P.H. Stress tolerance in Rhizobium and Brady Rhizobium and nodulation under adverse soil conditions. Can. J. Microbiol. 1992, 38, 475–484. [Google Scholar] [CrossRef]
- Woese, C. The Archaea: Their history and significance. In The Biochemistry of Archaea (Archaebacteria); Kates, M., Kushner, D., Matheson, A., Eds.; Elsevier: Amsterdam, The Netherlands, 1993; pp. 7–29. [Google Scholar]
- Choudhary, D.K.; Varma, A.; Tuteja, N. Plant-Microbe Interaction: An Approach to Sustainable Agriculture; Springer: Singapore, 2016. [Google Scholar]
- Larsen, H. Halophilic and halotolerant microorganisms—An overview and historical perspective. FEMS Microbiol. Rev. 1986, 39, 3–7. [Google Scholar] [CrossRef]
- Oren, A. Microbial life at high salt concentrations: Phylogenetic and metabolic diversity. Saline Syst. 2008, 4, 2. [Google Scholar] [CrossRef]
- Shakirova, F.M.; Sakhabutdinova, A.R.; Bezrukova, M.V.; Fatkhutdinova, R.A.; Fatkhutdinova, D.R. Changes in the hormonal status of wheat seedlings induced by salicylic acid and salinity. Plant Sci. 2003, 164, 317–322. [Google Scholar] [CrossRef]
- Zeikus, J.G.; Hegge, P.W.; Thompson, T.E.; Phelps, T.J.; Langworthy, T.A. Isolation and descriptionof Haloanaerobium praevalens gen. nov. and sp. nov. J. Biotechnol. 1983, 152, 114–124. [Google Scholar]
- Ahn, J.; Park, J.W.; Mc Connell, J.A.; Ahn, Y.B.; Häggblom, M.M. Kangiella spongicola sp. nov. a halophilic marine bacterium isolated from the sponge Chondrilla nucula. Int. J. Syst. Evol. Microbiol. 2011, 61, 961–964. [Google Scholar] [CrossRef][Green Version]
- Gales, G.; Chehider, N.; Joulian, C.; Battaglia-Brunet, F.; Cayol, J.L.; Postec, A.; Borgomano, J.; Neria-Gonzalez, I.; Lomans, B.; Ollivier, B. Characterization of Halanaerocella petrolearia gen. nov. sp. nov. a new anaerobic moderately halophilic fermentative bacterium isolated from a deep subsurface hypersaline oil reservoir. Extremophiles 2011, 15, 565–571. [Google Scholar] [CrossRef] [PubMed]
- Jiang, F.; Cao, S.-J.; Li, Z.H.; Fan, H.; Li, H.F.; Liu, W.J.; Yuan, H.L. Salisediminibacterium halotolerans gen. nov. sp. nov. a halophilic bacterium isolated from Xiarinaoer soda lake sediment in Inner Mongolia, China. Int. J. Syst. Evol. Microbiol. 2011, 62, 2127–2132. [Google Scholar] [CrossRef] [PubMed]
- Pugin, B.; Blamey, J.M.; Baxter, B.K.; Wiegel, J. Amphibacillus cookii sp. nov. a facultatively aerobic, spore-forming, moderately halophilic, alkalithermotolerant bacterium. Int. J. Syst. Evol. Microbiol. 2012, 62, 2090–2096. [Google Scholar] [CrossRef] [PubMed]
- Blum, J.S.; Kulp, T.R.; Han, S.; Lanoil, B.; Saltikov, C.W.; Stolz, J.F.; Miller, L.G.; Oremland, R.S. Desulfohalophilus alkaliarsenatis gen. nov. sp. nov. an extremely halophilic sulfate-and arsenate-respiring bacterium from Searles Lake California. Extremophiles 2012, 16, 727–742. [Google Scholar] [CrossRef]
- Mezghani, M.; Alazard, D.; Karray, F.; Cayol, J.L.; Joseph, M.; Postec, A.; Fardeau, M.L.; Tholozan, J.L.; Sayadi, S. Halanaerobacter jeridensis sp. nov. isolated from a hypersaline lake. Int. J. Syst. Evol. Microbiol. 2012, 62, 1970–1973. [Google Scholar] [CrossRef]
- Echigo, A.; Minegishi, H.; Shimane, Y.; Kamekura, M.; Usami, R. Natribacillus halophilus gen. nov. sp. nov. a moderately halophilic and alkalitolerant bacterium isolated from soil. Int. J. Syst. Evol. Microbiol. 2012, 62, 289–294. [Google Scholar] [CrossRef]
- Wang, Y.X.; Liu, J.H.; Xiao, W.; Zhang, X.X.; Li, Y.Q.; Lai, Y.H.; Ji, K.Y.; Wen, M.L.; Cui, X.L. Fodinibius salinus gen. nov. sp. nov. a moderately halophilic bacterium isolated from a salt mine. Int. J. Syst. Evol. Microbiol. 2012, 62, 390–396. [Google Scholar] [CrossRef]
- Ishikawa, M.; Yamasato, K.; Kodama, K.; Yasuda, H.; Matsuyama, M.; Okamoto-Kainuma, A.; Koizumi, Y. Alkalibacterium gilvum sp. nov. slightly halophilic and alkaliphilic lactic acid bacterium isolated from soft and semi-hard cheeses. Int. J. Syst. Evol. Microbiol. 2013, 63, 1471–1478. [Google Scholar] [CrossRef]
- Echigo, A.; Minegishi, H.; Shimane, Y.; Kamekura, M.; Itoh, T.; Usami, R. Halomicroarcula pellucidagen nov. sp. nov. a non-pigmented, transparent-colony-forming, halophilic archaeonisolated from solar salt. Int. J. Syst. Evol. Microbiol. 2013, 63, 3556–3562. [Google Scholar] [CrossRef]
- Makhdoumi-Kakhki, A.; Amoozegar, M.A.; Ventosa, A. Salinibacter iranicus sp. nov. and Salinibacter luteus sp. nov. isolated from a salt lake, and emended descriptions of the genus Salinibacter and of Salinibacter ruber. Int. J. Syst. Evol. Microbiol. 2012, 62, 1521–1527. [Google Scholar] [CrossRef]
- Abdeljabbar, H.; Cayol, J.L.; Hania, W.B.; Boudabous, A.; Sadfi, N.; Fardeau, M.L. Halanaerobium sehlinense sp. nov. an extremely halophilic, fermentative, strictly anaerobic bacterium fromsediments of the hypersaline lake Sehline Sebkha. Int. J. Syst. Evol. Microbiol. 2013, 63, 2069–2074. [Google Scholar] [CrossRef] [PubMed]
- Amoozegar, M.A.; Bagheri, M.; Didari, M.; Fazeli, S.A.S.; Schumann, P.; Sanchez-Porro, C.; Ventosa, A. Saliterribacillus persicus gen. nov. sp. nov. a moderately halophilic bacterium isolatedfrom a hypersaline lake. Int. J. Syst. Evol. Microbiol. 2013, 63, 345–351. [Google Scholar] [CrossRef] [PubMed]
- Amoozegar, M.A.; Makhdoumi-Kakhki, A.; Ramezani, M.; Nikou, M.M.; Fazeli, S.A.S.; Schumann, P.; Ventosa, A. Limimonas halophila gen. nov. sp. nov. an extremely halophilic bacterium in the family Rhodospirillaceae. Int. J. Syst. Evol. Microbiol. 2013, 63, 1562–1567. [Google Scholar] [CrossRef] [PubMed]
- Amoozegar, M.A.; Bagheri, M.; Makhdoumi-Kakhki, A.; Didar, M.; Schumann, P.; Nikou, M.M.; Sánchez-Porro, C.; Ventosa, A. Aliicoccus persicus gen. nov. sp. nov. a halophilic member of the Firmicutes isolated from a hypersaline lake. Int. J. Syst. Evol. Microbiol. 2014, 64, 1964–1969. [Google Scholar] [CrossRef]
- Cui, H.L.; Yang, X.; Zhou, Y.G.; Liu, H.C.; Zhou, P.J.; Dyall-Smith, M.L. Halobellus limi sp. nov. and Halobellus salinus sp. nov. isolated from two marine solar salterns. Int. J. Syst. Evol. Microbiol. 2012, 62, 1307–1313. [Google Scholar] [CrossRef]
- Wang, S.; Sun, L.; Wei, D.; Zhou, B.; Zhang, J.; Gu, X.; Jiang, S. Bacillus daqingensis sp. nov. a halophilic, alkaliphilic bacterium isolated from Saline-sodic soil in Daqing, China. J. Microbiol. 2014, 52, 548–553. [Google Scholar] [CrossRef]
- Liu, W.; Yang, S.S. Oceanobacillus aidingensis sp. nov. a moderately halophilic bacterium. Antonie Van Leeuwenhoek 2014, 105, 801–808. [Google Scholar] [CrossRef][Green Version]
- León, M.J.; Fernández, A.B.; Ghai, R.; Sánchez-Porro, C.; Rodriguez-Valera, F.; Ventosa, A. Frommetagenomics to pure culture: Isolation and characterization of the moderately Halophilic bacterium Spiribacter salinus gen. nov. sp. nov. Appl. Environ. Microbiol. 2014, 80, 3850–3857. [Google Scholar] [CrossRef]
- Miao, C.; Jia, F.; Wan, Y.; Zhang, W.; Lin, M.; Jin, W. Halomonas huangheensis sp. nov. a moderatelyhalophilic bacterium isolated from a saline–alkali soil. Int. J. Syst. Evol. Microbiol. 2014, 64, 915–920. [Google Scholar] [CrossRef]
- Parada-Pinilla, M.P.; Díaz-Cárdenas, C.; López, G.; Díaz-Riaño, J.I.; Gonzalez, L.N.; Restrepo, S.; Baena, S. Salifodinibacter halophilus gen. nov. sp. nov. a halophilic gammaproteobacterium in the family Salinisphaeraceae isolated from a salt mine in the Colombian Andes. Int. J. Syst. Evol. Microbiol. 2020, 70, 004490. [Google Scholar] [CrossRef]
- Kushwaha, B.; Jadhav, I.; Jadhav, K. Halomonas sambharensis sp. nov. a Moderately Halophilic Bacterium Isolated from the Saltern Crystallizer Ponds of the Sambhar Salt Lake in India. Curr. Microbiol. 2020, 77, 1125–1134. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Jiang, G.Q.; Lin, H.P.; Sun, P.; Zhang, H.Y.; Lu, D.M.; Tang, S.K. Lentibacillus saliphilus. sp. nov. a moderately halophilic bacterium isolated from a saltern in Korea. Arch. Microbiol. 2020, 203, 621–627. [Google Scholar] [CrossRef] [PubMed]
- Khan, S.A.; Zununi Vahed, S.; Forouhandeh, H.; Tarhriz, V.; Chaparzadeh, N.; Hejazi, M.A.; Hejazi, M.S. Halomonas urmiana sp. nov. a moderately halophilic bacterium isolated from Urmia Lake in Iran. Int. J. Syst. Evol. Microbiol. 2020, 70, 2254–2260. [Google Scholar] [CrossRef] [PubMed]
- Yoo, Y.; Lee, H.; Kwon, B.O.; Khim, J.S.; Baek, S.; Pathiraja, D.; Kim, J.J. Marinobacter halodurans sp. nov. a halophilic bacterium isolated from sediment of a salt flat. Int. J. Syst. Evol. Microbiol. 2020, 70, 6294–6300. [Google Scholar] [CrossRef] [PubMed]
- Cho, G.Y.; Whang, K.S. Aliifodinibius saliphilus sp. nov. a moderately halophilic bacterium isolated from sediment of a crystallizing pond of a saltern. Int. J. Syst. Evol. Microbiol. 2020, 70, 358–363. [Google Scholar] [CrossRef]
- Saralov, A.; Kuznetsov, B.; Reutskikh, E.; Baslerov, R.; Panteleeva, A.; Suzina, N. Arhodomonasrecens sp. nov. a halophilic alkane-utilizing hydrogen-oxidizing bacterium from the brines offlotation enrichment of potassium minerals. Microbiology 2012, 81, 582–588. [Google Scholar] [CrossRef]
- Mukhtar, S.; Zareen, M.; Khaliq, Z.; Mehnaz, S.; Malik, K.A. Phylogenetic analysis of halophyte- associated rhizobacteria and effect of halotolerant and halophilic phosphate-solubilizing biofertilizers on maize growth under salinity stress conditions. J. Appl. Microbiol. 2020, 128, 556–573. [Google Scholar] [CrossRef]
- Edbeib, M.F.; Wahab, R.A.; Huyop, F. Halophiles: Biology, adaptation, and their role in decontamination of hypersaline environments. World J. Microbiol. Biotechnol. 2016, 32, 135. [Google Scholar] [CrossRef]
- Nath, A. Insights into the sequence parameters for halophilicadaptation. Amino Acids 2016, 48, 751–762. [Google Scholar] [CrossRef]
- Anbu, P.; Hur, B.K. Isolation of an organic solvent-tolerantbacterium Bacillus licheniformis PAL05 that is able to secretesolvent-stable lipase. Biotechnol. Appl. Biochem. 2014, 61, 528–534. [Google Scholar] [CrossRef]
- Petrovic, U.; Cimerman, N.; Plemenitas, A. Cellular responses to environmental salinity in the halophilic black yeast Hortaea werneckii. Mol. Microbiol. 2004, 45, 665–672. [Google Scholar] [CrossRef] [PubMed]
- Moghaddam, J.A.; Boehringer, N.; Burdziak, A.; Kunte, H.J.; Galinski, E.A.; Schäberle, T.F. Different strategies ofosmoadaptation in the closely related marine myxobacteria Enhygromyxa salina SWB007 and Plesiocystis pacifica SIR-1. Microbiology 2016, 162, 651–661. [Google Scholar] [CrossRef] [PubMed]
- Tanimura, K.; Matsumoto, T.; Nakayama, H.; Tanaka, T.; Kondo, A. Improvement of ectoine productivity by using sugar transporter-overexpressing Halomonas elongate. Enzyme Microb. Technol. 2016, 89, 63–68. [Google Scholar] [CrossRef] [PubMed]
- Youssef, N.H.; Savage-Ashlock, K.N.; McCully, A.L.; Luedtke, B.; Shaw, E.I.; Hoff, W.D.; Elshahed, M.S. Trehalose/2-sulfotrehalose biosynthesis and glycine-betaine uptake are widely spread mechanisms for osmoadaptation in the Halobacteriales. ISME J. 2014, 8, 636–649. [Google Scholar] [CrossRef]
- Knief, C.; Delmotte, N.; Chaffron, S.; Stark, M.; Innerebner, G.; Wassmann, R.; von Mering, C.; Varholt, J.A. Metaproteogenomic analysis of microbial communities in the phyllosphere and rhizosphere of rice. ISME J. 2012, 6, 1378–1390. [Google Scholar] [CrossRef]
- Karan, R.; Capes, M.D.; Das Sarma, S. Function andbiotechnology of extremophilic enzymes in low water activity. Aquat. Biosyst. 2012, 8, 4–10. [Google Scholar] [CrossRef]
- Ates, O.; Toksoy, E.; Arga, K.Y. Genome-scalereconstruction of metabolic network for a halophilic extremophile Chromohalobacter salexigens DSM 3043. BMC Syst. Biol. 2011, 5, 12. [Google Scholar] [CrossRef]
- Ying, X.; Liu, Y.; Xu, B.; Wang, D.; Jiang, W. Characterization and application of Halomonas shantousis SWA25, a halotolerant bacterium with multiple biogenic aminedegradation activity. Food Add. Cont. 2016, 33, 674–682. [Google Scholar]
- Ciulla, R.A.; Diaz, M.R.; Taylor, B.F.; Roberts, M.F. Organic osmolytes in aerobic bacteria from mono lake, an alkaline, moderately hypersaline environment. Appl. Environ. Microbiol. 1999, 63, 220–226. [Google Scholar] [CrossRef]
- Rajan, A.L.; Joseph, T.C.; Thampuran, N.; James, R.; Ashok, K.K.; Viswanathan, C.; Bansal, K.C. Cloning and heterologous expression of ectoine biosynthesis genes from Bacillus halodurans in Escherichia coli. Biotechnol. Lett. 2008, 30, 1403–1407. [Google Scholar] [CrossRef]
- Attar, N. A new phylum for methanogens. Nat. Rev. Microbiol. 2015, 13, 739–745. [Google Scholar] [CrossRef]
- Sorokin, D.Y.; Abbas, B.; Erik, V.Z.; Muyzer, G. Isolation and characterization of an obligately chemolithoautotrophic Halothiobacillus strain capable of growth on thiocyanate as anenergy source. FEMS Microbiol. Lett. 2014, 354, 69–74. [Google Scholar] [CrossRef] [PubMed]
- Jha, M.; Chourasia, S.; Sinha, S. Microbial consortium for sustainable rice production. Agroecol. Sustain. Food Syst. 2013, 37, 340–362. [Google Scholar] [CrossRef]
- Davis, P.J. The plant hormones: Their nature, Occurrence and functions. In Plant Hormones; Davis, P.J., Ed.; Springer: Dordrecht, The Netherlands, 2004. [Google Scholar]
- Selvakumar, G.; Panneerselvam, P.; Ganeshamurthy, A.N. Bacterial mediated alleviation of abiotic stress in crops. In Bacteria in Agrobiology: Stress Management; Maheshwari, D.K., Ed.; Springer: Berlin/Heidelberg, Germany, 2012; pp. 205–223. [Google Scholar]
- Sagar, A.; Sayyed, R.Z.; Ramteke, P.W.; Sharma, S.; Marraiki, N.; Elgorban, A.M.; Syed, A. ACC deaminase and antioxidant enzymes producing halophilic Enterobacter sp. PR14 promotes the growth of rice and millets under salinity stress. Physiol. Mol. Biol. Plants 2020, 26, 1847–1854. [Google Scholar] [CrossRef]
- Parida, A.K.; Das, A.B. Salt tolerance and salinity effects on plants: A review. Ecotoxicol. Environ. Saf. 2005, 60, 324–349. [Google Scholar] [CrossRef]
- Hayat, R.; Ali, S.; Amara, U.; Khalid, R.; Ahmed, I. Soil beneficial bacteria and their role in plantgrowth promotion: A review. Ann. Microbiol. 2010, 60, 579–598. [Google Scholar] [CrossRef]
- Diby, P.; Sarma, Y.R.; Srinivasan, V.; Anandaraj, M. Pseudomonas fluorescense mediated vigourin black pepper (Piper nigrum L.) under green house cultivation. Ann. Microbiol. 2005, 55, 171–174. [Google Scholar]
- Upadhyay, S.K.; Singh, J.S.; Singh, D.P. Exo-polysaccharide-producing plant growth-promotingrhizobacteria salinity condition. Pedosphere 2011, 21, 214–222. [Google Scholar] [CrossRef]
- Bano, A.; Fatima, M. Salt tolerance in Zea mays L. following inoculation with Rhizobium and Pseudomonas. Biol. Fertil. Soils 2009, 45, 405–413. [Google Scholar] [CrossRef]
- Baldani, J.L.; Reis, V.M.; Baldani, V.L.D.; Dobereiner, J. A brief story of nitrogen fixation in sugarcane—Reasons for success in Brazil. Funct. Plant Biol. 2000, 29, 417–423. [Google Scholar] [CrossRef]
- Del Rio, L.A.; Corpas, F.J.; Sandalio, L.M.; Palma, J.M.; Barroso, J.B. Plant peroxisomes, reactiveoxygen metabolism and nitric oxide. IUBMB Life 2003, 55, 71–81. [Google Scholar] [CrossRef] [PubMed]
- Bor, M.; Ozdemir, F.; Turkan, I. The effect of salt stress on lipid peroxidation and antioxidantsin leaves of sugar beet Beta vulgaris L. and wild beet Beta maritime L. Plant Sci. 2003, 164, 77–84. [Google Scholar] [CrossRef]
- Miller, G.; Susuki, N.; Ciftci-Yilmaz, S.; Mittler, R. Reactive oxygen species homeostasis andsignalling during drought and salinity stresses. Plant Cell Environ. 2010, 33, 453–467. [Google Scholar] [CrossRef] [PubMed]
- Vaishnav, A.; Kumari, S.; Jain, S.; Varma, A.; Tuteja, N.; Choudhary, D.K. PGPR-mediated expressionof salt tolerance gene in soybean through volatiles under sodium nitroprusside. J. Basic Microbiol. 2016, 56, 1274–1288. [Google Scholar] [CrossRef]
- Santoyo, G.; Urtis-Flores, C.A.; Loeza-Lara, P.D.; Orozco-Mosqueda, M.; Glick, B.R. Rhizosphere colonization determinants by plant growth-promoting rhizobacteria (PGPR). Biology 2021, 10, 475. [Google Scholar] [CrossRef]
- Neilands, J.B. Siderophores: Structure and function of microbial iron transport compounds. J. Biol. Chem. 1995, 270, 26723–26726. [Google Scholar] [CrossRef]
- Jha, B.; Gontia, I.; Hartmann, A. The roots of the halophyte Salicornia brachiata are a source of new halotolerant diazotrophic bacteria with plant growth-promoting potential. Plant Soil 2012, 356, 265–277. [Google Scholar] [CrossRef]
- Mukhtar, S.; Malik, K.A.; Mehnaz, S. Osmoadaptation in halophilic bacteria and archaea. Res. J. Biotechnol. 2020, 15, 154–161. [Google Scholar]
- Razzaghi Komaresofla, B.; Alikhani, H.A.; Etesami, H.; Khoshkholgh-Sima, N.A. Improved growth and salinity tolerance of the halophyte Salicornia sp. by co–inoculation with endophytic and rhizosphere bacteria. Appl. Soil Ecol. 2019, 138, 160–170. [Google Scholar] [CrossRef]
- Kharangate-Lad, A.; Bhosle, S. Studies on siderophore and pigment produced by an adhered bacterial strain Halobacillus trueperi MXM-16 from the mangrove ecosystem of Goa, India. Indian J. Microbiol. 2016, 56, 461–466. [Google Scholar] [CrossRef]
- Kuzyk, S.B.; Hughes, E.; Yurkov, V. Discovery of Siderophore and Metallophore Production in the Aerobic Anoxygenic Phototrophs. Microorganisms 2021, 9, 959. [Google Scholar] [CrossRef] [PubMed]
- Borde, M.; Dudhane, M.; Jite, P. Growth photosynthetic activity and antioxidant responses of mycorrhizal and non-mycorrhizal bajra (Pennisetum glaucum) crop under salinity stress condition. Crop Prot. 2011, 30, 265–271. [Google Scholar] [CrossRef]
- Gopal, S.; Chandrasekaran, M.; Shagol, C.; Kim, K.; Sa, T. Spore associated bacteria (SAB) of arbuscular mycorrhizal fungi (AMF) and plant growth promoting rhizobacteria (PGPR) increase nutrient uptake and plant growth under stress conditions. Korean J. Soil Sci. Fertil. 2012, 45, 582–592. [Google Scholar] [CrossRef]
- Smith, S.E.; Jakobsen, I.; Grønlund, M.; Smith, F.A. Roles of arbuscular mycorrhizas in plant phosphorus nutrition: Interactions between pathways of phosphorus uptake in arbuscular mycorrhizal roots have important implications for understanding and manipulating plant phosphorus acquisition. Plant Physiol. 2011, 156, 1050–1057. [Google Scholar] [CrossRef]
- Bharti, N.; Barnawal, D.; Awasthi, A.; Yadav, A.; Kalra, A. Plant growth promoting rhizobacteria alleviate salinity induced negative effects on growth, oil content and physiological status in Mentha arvensis. Acta Physiol. Plant. 2014, 36, 45–60. [Google Scholar] [CrossRef]
- Miransari, M.; Bahrami, H.A.; Rejali, F.; Malakouti, M.J. Using arbuscular mycorrhiza to alleviate the stress of soil compaction on wheat (Triticum aestivum L.) growth. Soil Biol. Biochem. 2008, 40, 1197–1206. [Google Scholar] [CrossRef]
- Miransari, M. Contribution of arbuscular mycorrhizal symbiosis to plant growth under different types of soil stress. Plant Biol. 2010, 12, 563–569. [Google Scholar] [CrossRef]
- Kang, S.M.; Khan, A.L.; Waqas, M.; You, Y.H.; Kim, J.H.; Kim, J.G.; Lee, I.J. Plant growth-promoting rhizobacteria reduce adverse effects of salinity and osmotic stress by regulating phytohormones and antioxidants in Cucumis sativus. J. Plant Interact. 2014, 9, 673–682. [Google Scholar] [CrossRef]
- Liu, Y.; Johnson, N.C.; Mao, L.; Shi, G.; Jiang, S.; Ma, X.; Feng, H. Phylogenetic structure of arbuscular mycorrhizal community shifts in response to increasing soil fertility. Soil Biol. Biochem. 2015, 89, 196–205. [Google Scholar] [CrossRef]
- Mardukhi, B.; Rejali, F.; Daei, G.; Ardakani, M.R.; Malakouti, M.J.; Miransari, M. Mineral uptake of mycorrhizal wheat (Triticum aestivum L.) under salinity stress. Commun. Soil Sci. Plant Anal. 2015, 46, 343–357. [Google Scholar] [CrossRef]
- Ramos, A.C.; Façanha, A.R.; Palma, L.M.; Okorokov, L.A.; Cruz, Z.; Silva, A.G.; Santos, W.O. An outlook on ion signaling and ionome of mycorrhizal symbiosis. Braz. J. Plant Physiol. 2011, 23, 79–89. [Google Scholar] [CrossRef]
- Hajiboland, R.; Aliasgharzadeh, N.; Laiegh, S.F.; Poschenrieder, C. Colonization with arbuscular mycorrhizal fungi improves salinity tolerance of tomato (Solanum lycopersicum L.) plants. Plant Soil 2010, 331, 313–327. [Google Scholar] [CrossRef]
- Aroca, R.; Ruiz-Lozano, J.M.; Zamarreño, Á.M.; Paz, J.A.; García-Mina, J.M.; Pozo, M.J.; López-Ráez, J.A. Arbuscular mycorrhizal symbiosis influences strigolactone production under salinity and alleviates salt stress in lettuce plants. J. Plant Physiol. 2013, 170, 47–55. [Google Scholar] [CrossRef] [PubMed]
- Rai, M.K.; Kalia, R.K.; Singh, R.; Gangola, M.P.; Dhawan, A.K. Developing stress tolerant plants through in vitro selection—An overview of the recent progress. Environ. Exp. Bot. 2011, 71, 89–98. [Google Scholar] [CrossRef]
- Jiang, M.; Zhang, J. Water stress-induced abscisic acid accumulation triggers the increased generation of reactive oxygen species and up-regulates the activities of antioxidant enzymes in maize leaves. J. Exp. Bot. 2002, 53, 2401–2410. [Google Scholar] [CrossRef] [PubMed]
- Nunez, M.; Mazzafera, P.; Mazorra, L.M.; Siqueira, W.J.; Zullo, M.A.T. Influence of a brassinosteroid analogue on antioxidant enzymes in rice grown in culture medium with NaCl. Biol. Plant. 2003, 47, 67–70. [Google Scholar] [CrossRef]
- Huang, Z.; He, C.X.; He, Z.Q.; Zou, Z.R.; Zhang, Z.B. The effects of arbuscular mycorrhizal fungi on reactive oxyradical scavenging system of tomato under salt tolerance. Agric. Sci. China 2010, 9, 1150–1159. [Google Scholar] [CrossRef]
- Al-Khaliel, A.S. Effect of salinity stress on mycorrhizal association and growth response of peanut infected by Glomus mosseae. Plant Soil Environ. 2010, 56, 318–324. [Google Scholar] [CrossRef]
- Sannazzaro, A.I.; Echeverría, M.; Albertó, E.O.; Ruiz, O.A.; Menéndez, A.B. Modulation of polyamine balance in Lotus glaber by salinity and arbuscular mycorrhiza. Plant Physiol. Biochem. 2007, 45, 39–46. [Google Scholar] [CrossRef]
- Datta, P.; Kulkarnni, M.V. Arbuscular mycorrhizal colonization enhances biochemical status and mitigates adverse salt effect on two legumes. Not. Sci. Biol. 2014, 6, 381–393. [Google Scholar] [CrossRef]
- Rillig, M.C. Arbuscular mycorrhizae, glomalin, and soil aggregation. Can. J. Soil Sci. 2004, 84, 355–363. [Google Scholar] [CrossRef]
- Rillig, M.C.; Wright, S.F.; Eviner, V.T. The role of arbuscular mycorrhizal fungi and glomalin in soil aggregation: Comparing effects of five plant species. Plant Soil 2002, 238, 325–333. [Google Scholar] [CrossRef]
- Borie, F.; Rubio, R.; Morales, A. Arbuscular mycorrhizal fungi and soil aggregation. Rev. Cienc. Suelo Nutr. Veg. 2008, 8, 9–18. [Google Scholar] [CrossRef]
- Liu, S.; Guo, X.; Feng, G.; Maimaitiaili, B.; Fan, J.; He, X. Indigenous arbuscular mycorrhizal fungi can alleviate salt stress and promote growth of cotton and maize in saline fields. Plant Soil 2016, 398, 195–206. [Google Scholar] [CrossRef]
- Hashem, A.; Alqarawi, A.A.; Radhakrishnan, R.; Al-Arjani, A.B.F.; Aldehaish, H.A.; Egamberdieva, D.; Abd-Allah, E.F. Arbuscular mycorrhizal fungi regulate the oxidative system, hormones and ionic equilibrium to trigger salt stress tolerance in Cucumis sativus L. Saudi J. Biol. Sci. 2018, 25, 1102–1114. [Google Scholar] [CrossRef]
- Khalloufi, M.; Martínez-Andújar, C.; Lachaâl, M.; Karray-Bouraoui, N.; Pérez-Alfocea, F.; Albacete, A. The interaction between foliar GA3 application and arbuscular mycorrhizal fungi inoculation improves growth in salinized tomato (Solanum lycopersicum L.) plants by modifying the hormonal balance. J. Plant Physiol. 2017, 214, 134–144. [Google Scholar] [CrossRef]
- Porcel, R.; Redondo-Gómez, S.; Mateos-Naranjo, E.; Aroca, R.; Garcia, R.; Ruiz-Lozano, J.M. Arbuscular mycorrhizal symbiosis ameliorates the optimum quantum yield of photosystem II and reduces non-photochemical quenching in rice plants subjected to salt stress. J. Plant Physiol. 2015, 185, 75–83. [Google Scholar] [CrossRef]
- Hajiboland, R.; Dashtebani, F.; Aliasgharzad, N. Physiological responses of halophytic C4 grass Aeluropus littoralis to salinity and arbuscular mycorrhizal fungi colonization. Photosynthetica 2015, 53, 572–584. [Google Scholar] [CrossRef]
- Giri, B.; Kapoor, R.; Mukerji, K.G. Improved tolerance of Acacia nilotica to salt stress by arbuscular mycorrhiza, Glomus fasciculatum may be partly related to elevated K/Na ratios in root and shoot tissues. Microb. Ecol. 2007, 54, 753–760. [Google Scholar] [CrossRef]
- Jixiang, L.; Wang, Y.; Sun, S.; Mu, C.; Yan, X. Effects of arbuscular mycorrhizal fungi on the growth, photosynthesis and photosynthetic pigments of Leymus chinensis seedlings under salt-alkali stress and nitrogen deposition. Sci. Total Environ. 2017, 576, 234–241. [Google Scholar]
- Venkataraman, G.S. Blue-Green Algae for Rice Production: A Manual for Its Promotion (No. 46); Food and Agriculture Org.: Rome, Italy, 1981. [Google Scholar]
- Kaushik, B.D.; Subhashinim, D. Amelioration of salt-affected soils with blue-green algae. II. Improvement in soil properties. Proc. Ind. Natl. Sci. Acad. 1985, 51, 380–389. [Google Scholar]
- Singh, V.; Singh, D.V. Cyanobacteria modulated changes and its impact on bioremediation of saline-alkaline soils. Bangladesh J. Bot. 2015, 44, 653–658. [Google Scholar] [CrossRef]
- Weralupitiya, C.; Wanigatunge, R.P.; Gunawardana, D.; Vithanage, M.; Magana-Arachchi, D. Cyanotoxins uptake and accumulation in crops: Phytotoxicity and implications on human health. Toxicon 2022, 211, 21–35. [Google Scholar] [CrossRef] [PubMed]
- Ladha, J.K.; Reddy, P.M. Extension of nitrogen fixation to rice: Necessity and possibilities. Geol. J. 1995, 35, 363–372. [Google Scholar] [CrossRef]
- Singh, R.N. Role of Blue-Green Algae in Nitrogen Economy of Indian Agriculture; Indian Council of Agricultural Research: New Delhi, India, 1961. [Google Scholar]
- Pandey, K.D.; Shukla, P.N.; Giri, D.D.; Kashyap, A.K. Cyanobacteria in alkaline soil and the effect of cyanobacteria inoculation with pyrite amendments on their reclamation. Biol. Fertil. Soils 2005, 41, 451–457. [Google Scholar] [CrossRef]
- Jaiswal, P.; Kashyap, A.K.; Prasanna, R.; Singh, P.K. Evaluating the potential of N. calcicola and its biocarbonate resistant mutant as bioameleorating agents for ‘usar’ soil. Ind. J. Microbiol. 2010, 50, 12–18. [Google Scholar] [CrossRef] [PubMed]
- Murtaza, B.; Murtaza, G.; Zia-ar-Rehman, M.; Ghafoor, A.; Abubakar, S.; Sabir, M. Reclamation of salt affected soils using amendments and growing wheat crop. Soil Environ. 2011, 30, 130–136. [Google Scholar]
- Hussain, S.; Shaukat, M.; Ashraf, M.; Zhu, C.; Jin, Q.; Zhang, J. Salinity stress in arid and semi-arid climates: Effects and management in field crops. In Climate Change and Agriculture; Hussain, S., Ed.; IntechOpen: London, UK, 2009; pp. 1–26. [Google Scholar]
- Valencia-Cantero, E.; Hernández-Calderón, E.; Velázquez-Becerra, C.; López-Meza, J.E.; Alfaro-Cuevas, R.; López-Bucio, J. Role of dissimilatory fermentative iron-reducing bacteria in Fe uptake by common bean (Phaseolus vulgaris L.) plants grown in alkaline soil. Plant Soil. 2007, 291, 263–273. [Google Scholar] [CrossRef]
- Sarkar, A.; Ghosh, P.K.; Pramanik, K.; Mitra, S.; Soren, T.; Pandey, S.; Mondal, M.H.; Maiti, T.K. A halotolerant Enterobacter sp. displaying ACC deaminase activity promotes rice seedling growth under salt stress. Res. Microbiol. 2018, 169, 20–32. [Google Scholar] [CrossRef]
- Han, D.; Wang, L.; Luo, Y. Isolation, identification, and the growth promoting effects of two antagonistic actinomycete strains from the rhizosphere of Mikania micrantha Kunth. Microbiol. Res. 2018, 208, 1–11. [Google Scholar] [CrossRef]
- Schoenborn, L.; Yates, P.S.; Grinton, B.E.; Hugenholtz, P.; Janssen, P.H. Liquid serial dilution is inferior to solid media for isolation of cultures representative of the phylum-level diversity of soil bacteria. Appl. Environ. Microbiol. 2004, 70, 4363–4366. [Google Scholar] [CrossRef] [PubMed]
- Whipps, J.M. Microbial interactions and biocontrol in the rhizosphere. J. Exp. Bot. 2001, 52, 487–511. [Google Scholar] [CrossRef] [PubMed]
- Ilangumaran, G.; Smith, D.L. Plant Growth Promoting Rhizobacteria in Amelioration of Salinity Stress: A Systems Biology Perspective. Front. Plant Sci. 2017, 8, 1768. [Google Scholar] [CrossRef] [PubMed]
- Obledo, E.N.; Barraga’n-Barraga’n, L.B.; Gutie’rrez-Gonza’lez, P.; Ramı’rez-Herna’ndez, B.C.; Ramı’rez, J.J.; Rodrı’guez-Garay, B. Increased photosyntethic efficiency generated by fungal symbiosis in Agave victoria-reginae. Plant Cell Tissue Organ Cult. 2003, 74, 237–241. [Google Scholar] [CrossRef]
- Kumar, A.; Verma, J.P. The role of microbes to improve crop productivity and soil health. In Ecological Wisdom Inspired Restoration Engineering; Achal, V., Mukherjee, A., Eds.; Springer: Singapore, 2019; pp. 249–265. [Google Scholar]
- Kumar, A.; Singh, S.; Gaurav, A.K.; Srivastava, S.; Verma, J.P. Plant Growth-Promoting Bacteria: Biological Tools for the Mitigation of Salinity Stress in Plants. Front. Microbiol. 2020, 11, 12–16. [Google Scholar] [CrossRef]
- Ahmad, M.; Zahir, Z.A.; Asghar, H.N.; Asghar, M. Inducing salt tolerance in mung bean through co-inoculation with rhizobia and plant-growth-promoting rhizobacteria containing 1-aminocyclopropane-1-carboxylate deaminase. Can. J. Microbiol. 2011, 57, 578–589. [Google Scholar] [CrossRef]
- Erice, G.; Ruíz-Lozano, J.M.; Zamarreñ, Á.M.; García-Mina, J.M.; Aroca, R. Transcriptomic analysis reveals the importance of JA-Ile turnover in the response of Arabidopsis plants to plant growth promoting rhizobacteria and salinity. Environ. Exp. Bot. 2017, 143, 10–19. [Google Scholar] [CrossRef]
- Ansari, F.A.; Ahmad, I.; Pichtel, J. Growth stimulation and alleviation of salinity stress to wheat by the biofilm forming Bacillus pumilus strain FAB10. Appl. Soil Ecol. 2019, 143, 45–54. [Google Scholar] [CrossRef]
- Bhise, K.K.; Bhagwat, P.K.; Dandge, P.B. Synergistic effect of Chryseobacteriumgleum sp. SUK with ACC deaminase activity in alleviation of salt stress and plant growth promotion in Triticum aestivum L. Biotechnology 2017, 7, 101–113. [Google Scholar]
- Gupta, J.; Rathour, R.; Singh, R.; Thakur, I.S. Production and characterization of extracellular polymeric substances (EPS) generated by a carbofuran degrading strain Cupriavidus sp. ISTL7. Bioresour. Technol. 2019, 282, 417–424. [Google Scholar] [CrossRef]
- Vurukonda, S.S.K.P.; Vardharajula, S.; Shrivastava, M. Enhancement of drought stress tolerance in crops by plant growth promoting rhizobacteria. Microbiol. Res. 2016, 184, 13–24. [Google Scholar] [CrossRef] [PubMed]
- Suprasanna, P.; Nikalje, G.C.; Rai, A.N. Osmolyte accumulation and implications in plant abiotic stress tolerance. In Osmolytes and Plants Acclimation to Changing Environment: Emerging Omics Technologies; Springer: New Delhi, India, 2016; pp. 1–12. [Google Scholar]
- Norwood, M.; Truesdale, M.R.; Richter, A.; Scott, P. Photosynthetic carbohydrate metabolism in the resurrection plant Craterostigma plantagineum. J. Exp. Bot. 2000, 51, 159–165. [Google Scholar] [CrossRef] [PubMed]
- Volker, U.; Engelmann, S.; Maul, B.; Riethdorf, S.; Völker, A.; Schmid, R.; Mach, H.; Hecker, M. Analysis of the induction of general stress proteins of Bacillus subtilis. Microbiology 1994, 140, 741–752. [Google Scholar] [CrossRef] [PubMed]
- Bartels, D.; Sunkar, R. Drought and salt tolerance in plants. Crit. Rev. Plant Sci. 2005, 24, 23–58. [Google Scholar] [CrossRef]
- Masalha, J.; Kosegarten, H.; ElmaciÖd Mengel, K. The central role of microbial activity for iron acquisition in maize and sunflower. Biol. Fert. Soils 2000, 30, 433–439. [Google Scholar] [CrossRef]
- Rungin, S.; Indananda, C.; Suttiviriya, P.; Kruasuwan, W.; Jaemsaeng, R.; Thamchaipenet, A. Plant growth enhancing effects by a siderophoreproducing endophytic streptomycete isolated from a Thai jasmine rice plant (Oryza sativa L. cv. KDML105). Antonie Van Leeuwenhoek 2012, 102, 463–472. [Google Scholar] [CrossRef]
- Ramadoss, D.; Lakkineni, V.K.; Bose, P.; Ali, S.; Annapurna, K. Mitigation of salt stress in wheat seedlings by halotolerant bacteria isolated from saline habitats. Springer Plus 2013, 2, 6. [Google Scholar] [CrossRef]
- Barriuso, J.; Solano, B.R.; Gutiérrez Mañero, F.J. Protection against pathogen and salt stress by four plant growth-promoting rhizobacteria isolated from Pinus sp. on Arabidopsis thaliana. Phytopathology 2008, 98, 666–672. [Google Scholar] [CrossRef]
- Glick, B.R. Plant growth-promoting bacteria: Mechanisms and applications. Scientifica 2012, 2012, 963401. [Google Scholar] [CrossRef]
- Olanrewaju, O.S.; Glick, B.R.; Babalola, O.O. Mechanisms of action of plant growth promoting bacteria. World J. Microbiol. Biotechnol. 2017, 33, 197. [Google Scholar] [CrossRef]
- Sharma, R.K.; Archana, G. Cadmium minimization in food crops by cadmium resistant plant growth promoting rhizobacteria. Appl. Soil Ecol. 2016, 107, 66–78. [Google Scholar] [CrossRef]
- Shi-Ying, Z.; Cong, F.; Yong-xia, W.; Yun-sheng, X.; Wei, X.; Xiao-Long, C. Salt-tolerant and plant growth-promoting bacteria isolated from high-yield paddy soil. Can. J. Microbiol. 2018, 64, 968–978. [Google Scholar]
- Jaiswal, D.K.; Verma, J.P.; Prakash, S.; Meena, V.S.; Meena, R.S. Potassium as an important plant nutrient in sustainable agriculture: A state of the art. In Potassium Solubilizing Microorganisms for Sustainable Agriculture; Springer: New Delhi, India, 2016; pp. 21–29. [Google Scholar]
- Santi, C.; Bogusz, D.; Franche, C. Biological nitrogen fixation in non-legume plants. Ann. Bot. 2013, 111, 743–767. [Google Scholar] [CrossRef] [PubMed]
- Etesami, H.; Mirseyed Hosseini, H.; Alikhani, H.A. In planta selection of plant growth promoting endophytic bacteria for rice (Oryza sativa L.). J. Soil Sci. Plant Nutr. 2014, 14, 491–503. [Google Scholar] [CrossRef]
- Mukherjee, A.; Gaurav, A.K.; Singh, S.; Chouhan, G.K.; Kumar, A.; Das, S. Role of Potassium (K) Solubilising Microbes (KSM) in growth and induction of resistance against biotic and abiotic stress in plant: A book review. Clim. Change Environ. Sustain. 2019, 7, 212–214. [Google Scholar]
- Vasanthi, N.; Saleena, L.M.; Raj, S.A. Silica solubilization potential of certain bacterial species in the presence of different silicate minerals. Silicon 2018, 10, 267–275. [Google Scholar] [CrossRef]
- Stewart, J.; Sharpley, A. Controls on dynamics of soil and fertilizer phosphorus and sulfur. In Soil Fertility and Organic Matter as Critical Components of Production Systems (Soil Fertility); Wiley: Hoboken, NJ, USA, 1987; pp. 101–121. [Google Scholar]
- Qadir, M.; Schubert, S. Degradation processes and nutrient constraints in sodic soils. Land Degrad. Dev. 2002, 13, 275–294. [Google Scholar] [CrossRef]
- Troxler, T.G.; Ikenaga, M.; Scinto, L.; Boyer, J.N.; Condit, R.; Perez, R.; Gann, G.D.; Childers, D.L. Patterns of Soil bacteria and Canopy Community structure related to tropical peatland development. Wetlands 2012, 32, 769–782. [Google Scholar] [CrossRef]
- Muhammad, S.; Müller, T.; Joergensen, R.G. Decomposition of pea and maize straw in Pakistani soils along a gradient in salinity. Biol. Fertil. Soils 2006, 43, 93–101. [Google Scholar] [CrossRef]
- Wieder, W.R.; Boehnert, J.; Bonan, G.B. Evaluating soil biogeochemistry parameterizations in Earth system models with observations. Glob. Biogeochem. Cycles 2014, 28, 211–222. [Google Scholar] [CrossRef]
- Powell, J.R.; Karunaratne, S.; Campbell, C.D.; Yao, H.; Robinson, L.; Singh, B.K. Deterministic processes vary during community assembly for ecologically dissimilar taxa. Nat. Commun. 2015, 6, 8444. [Google Scholar] [CrossRef] [PubMed]
- Hong, B.H.; Joe, M.M.; Selvakumar, G.; Kim, K.Y.; Choi, J.H.; Sa, T.M. Influence of salinity variations on exocellular polysaccharide production, biofilm formation and flocculation in halotolerant bacteria. J. Environ. Biol. 2017, 38, 657. [Google Scholar] [CrossRef]
| Halophilic Bacterial Species | Salinity Range for the Growth and Development (%) | References |
|---|---|---|
| Kangiella spongicola | 2–15 | [62] |
| Halanaerocella petrolearia | 6–26 | [63] |
| Salisediminibacterium cookie | 3–30 | [64] |
| Amphibacillus cookie | 6–26 | [65] |
| Desulfohalophilus alkaliarsenatis | 12.5–33 | [66] |
| Halanaerobacter jeridensis | 6–30 | [67] |
| Natribacillus halophilus | 7–23 | [68] |
| Fodinibius salinus | 10–15 | [69] |
| Alkalibacterium gilvum | 0–17.5 | [70] |
| Halomicroarcula pellucida | 20–30 | [71] |
| Salinibacter iranicus | 12–30 | [72] |
| Halanaerobium sehlinen | 5–30 | [73] |
| Saliterribacillus perciscus | 0.5–22.5 | [74] |
| Limimonas halopajila | 15–30 | [75] |
| Aquibacillus halophilus | 0.5–20 | [76] |
| Halobellus salinus | 15–30 | [77] |
| Bacillus daqingensis | 0–16 | [78] |
| Oceanicola flagellatus | 0–21 | [79] |
| Spiribacter salinus | 10–25 | [80] |
| Halomonas huangheensis | 1–20 | [81] |
| Salifodinibacter halophilus | 25 | [82] |
| Halomonas sambharensis | 5–8 | [83] |
| Lentibacillus saliphilus sp. nov. (type strain YIM 93176T) | 0–22 | [84] |
| Halomonas urmiana sp. | 0.5–20 | [85] |
| Marinobacter halodurans sp. nov. | 1–18 | [86] |
| Aliifodinibius saliphilus sp. nov. | 3–25 | [87] |
| Arhodomonas recens | 2–25 | [88] |
| Host Plant | Fungal Species (AMF) | Response by Plant | References |
|---|---|---|---|
| Cucumis sativus L. | Glomus etunicatum, Glomum, intraradices, Glomus mosseae | Biomass increased, photosynthesis pigments synthesis, antioxidants enzymes increased | [149] |
| Solanum lycopersicum L. | Rhizophagus irregularis | Enhanced leaf area, leaf number, root and shoot dry weight and growth harmones | [150] |
| Oryza sativa L. | Claroideoglomus etunicatum | Quantum yield of PSII and photosynthetic rate increased | [151] |
| Aeluropus littoralis | Claroideoglomus etunicatum | Enhanced root, shoot dry mass, soluble sugars, free amino acids | [152] |
| Solanum lycopersicum L. | Glomus intraradices | Improved dry matter, growth parameters, chlorophyll content and ions uptake | [136] |
| Acacia nilotica | Glomus fasciculate | Enhanced root, shoot dry mass, P, Zn and Cu content | [153] |
| Leymus chinensis | Glomus mosseae | Increase in the colonization rate, seedling weight, water content, P and N | [154] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kumawat, C.; Kumar, A.; Parshad, J.; Sharma, S.S.; Patra, A.; Dogra, P.; Yadav, G.K.; Dadhich, S.K.; Verma, R.; Kumawat, G.L. Microbial Diversity and Adaptation under Salt-Affected Soils: A Review. Sustainability 2022, 14, 9280. https://doi.org/10.3390/su14159280
Kumawat C, Kumar A, Parshad J, Sharma SS, Patra A, Dogra P, Yadav GK, Dadhich SK, Verma R, Kumawat GL. Microbial Diversity and Adaptation under Salt-Affected Soils: A Review. Sustainability. 2022; 14(15):9280. https://doi.org/10.3390/su14159280
Chicago/Turabian StyleKumawat, Chiranjeev, Ajay Kumar, Jagdish Parshad, Shyam Sunder Sharma, Abhik Patra, Prerna Dogra, Govind Kumar Yadav, Sunil Kumar Dadhich, Rajhans Verma, and Girdhari Lal Kumawat. 2022. "Microbial Diversity and Adaptation under Salt-Affected Soils: A Review" Sustainability 14, no. 15: 9280. https://doi.org/10.3390/su14159280
APA StyleKumawat, C., Kumar, A., Parshad, J., Sharma, S. S., Patra, A., Dogra, P., Yadav, G. K., Dadhich, S. K., Verma, R., & Kumawat, G. L. (2022). Microbial Diversity and Adaptation under Salt-Affected Soils: A Review. Sustainability, 14(15), 9280. https://doi.org/10.3390/su14159280






