Stochastic Modelling to Assess External Environmental Drivers of Atlantic Chub Mackerel Population Dynamics
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Area
2.2. Biomass Trend Estimation
2.2.1. Model Description
- Biomass equation
- Fishing mortality equation
- Index equation
- Catch equation
2.2.2. Model Data Input
2.2.3. Model Output Consistency Analysis and Model Implementation
2.3. Comparison of Model Outputs with Physical and Biogeochemical Variables
2.3.1. Sources of Environmental data
- Bottom salinity;
- 3D temperature (C);
- 3D salinity;
- 3D zonal velocity (m/s);
- 3D meridional velocity (m/s).
- 3D chlorophyll (mg/m);
- 3D net primary production (NPP) (mgC/m);
- 3D oxygen () (mmol/m);
- 3D nitrate () (mmol/m);
- 3D phosphate () (mmol/m).
2.3.2. Comparison Procedure
3. Results
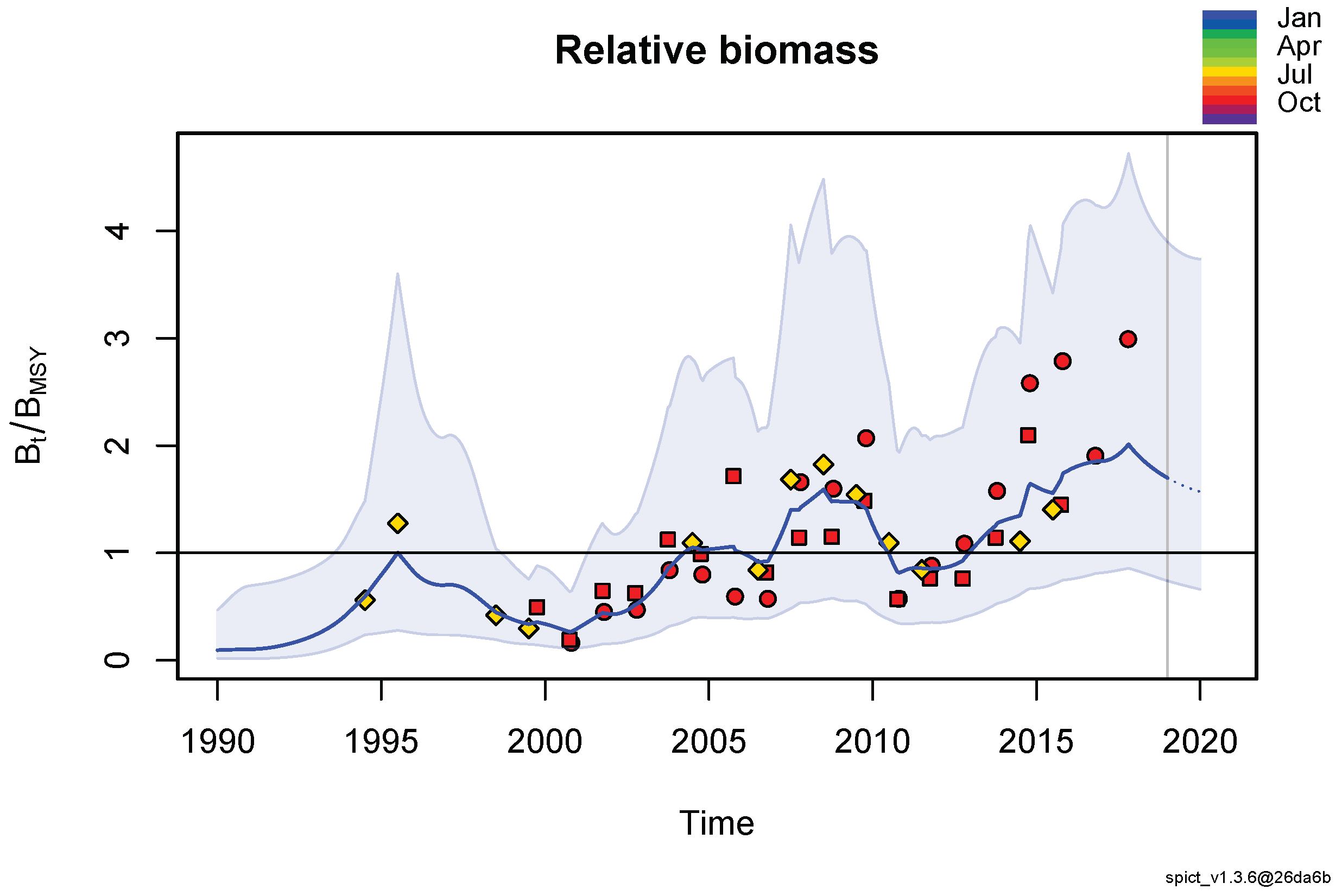
3.1. Model Results
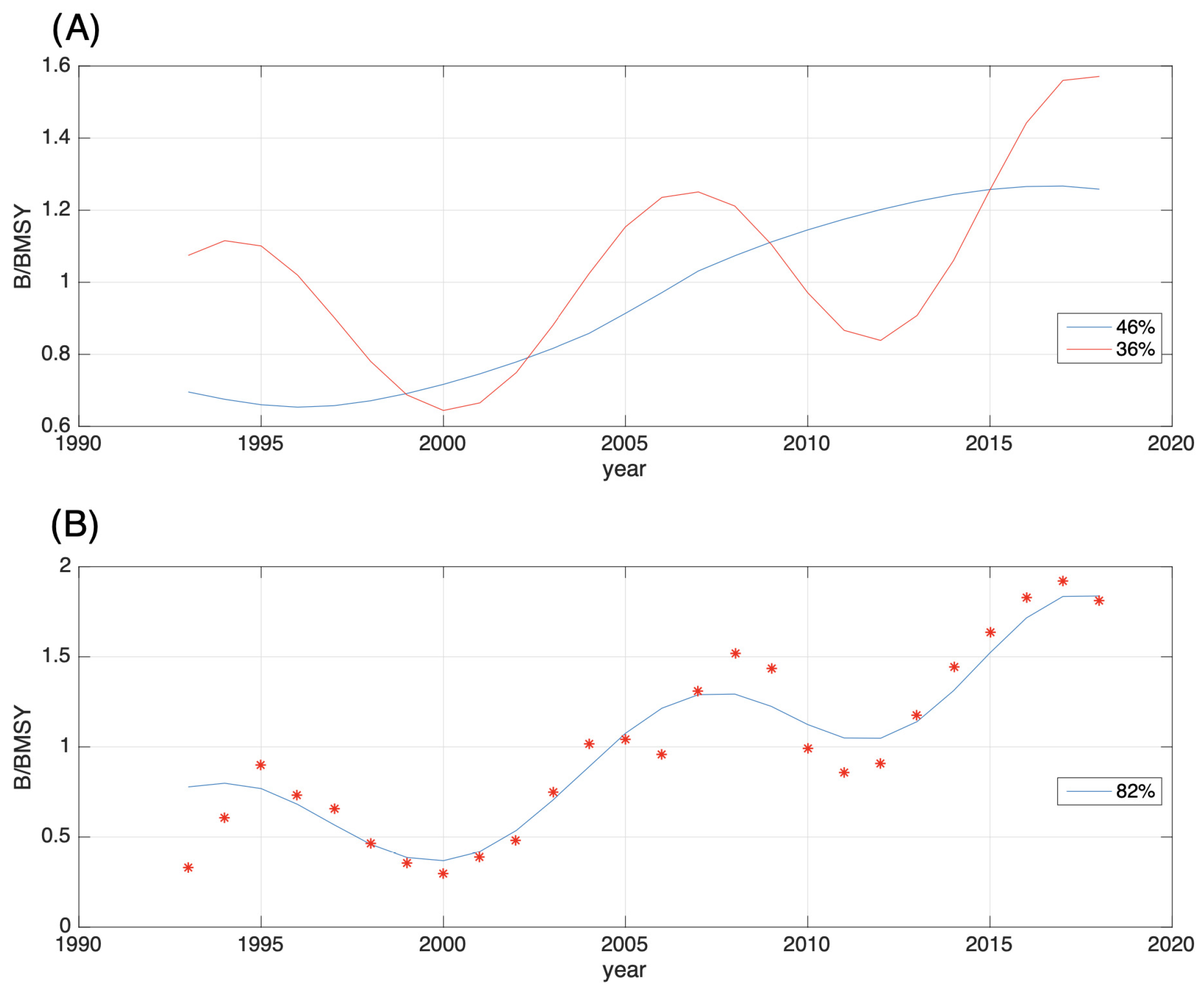
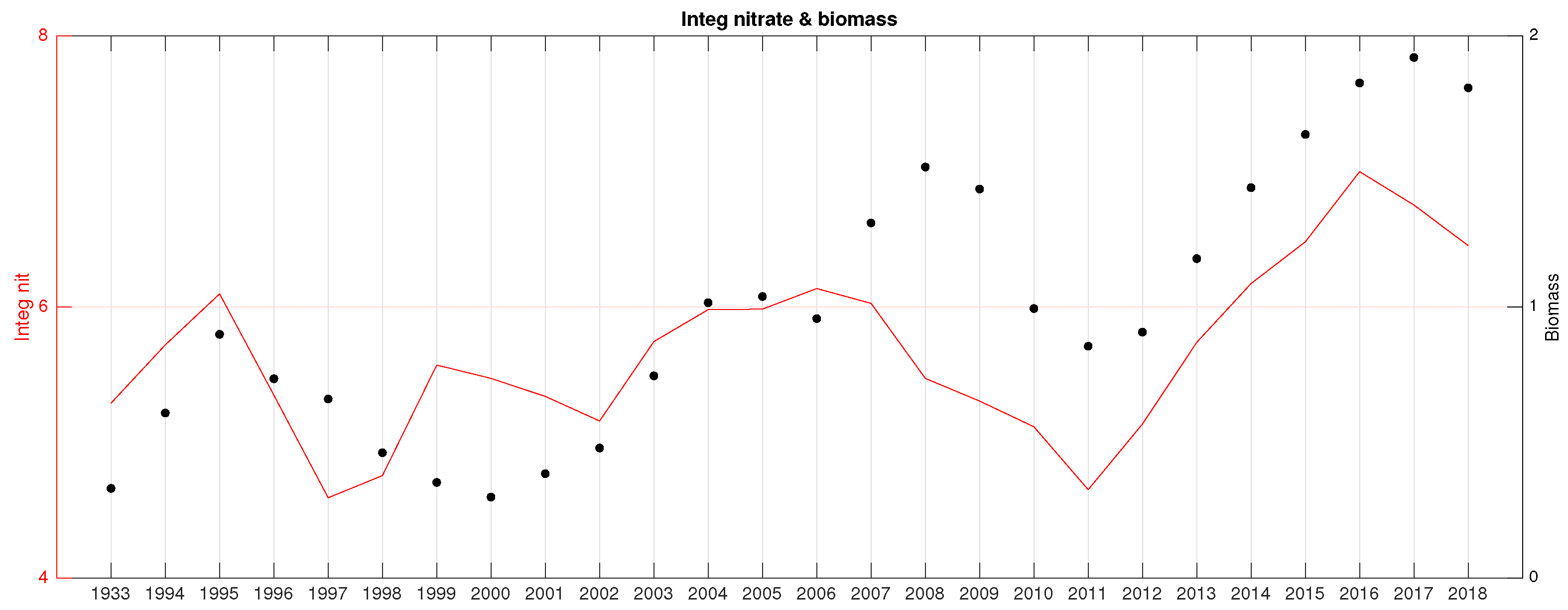
3.2. Spectral Analysis of the Relative Biomass Time Series
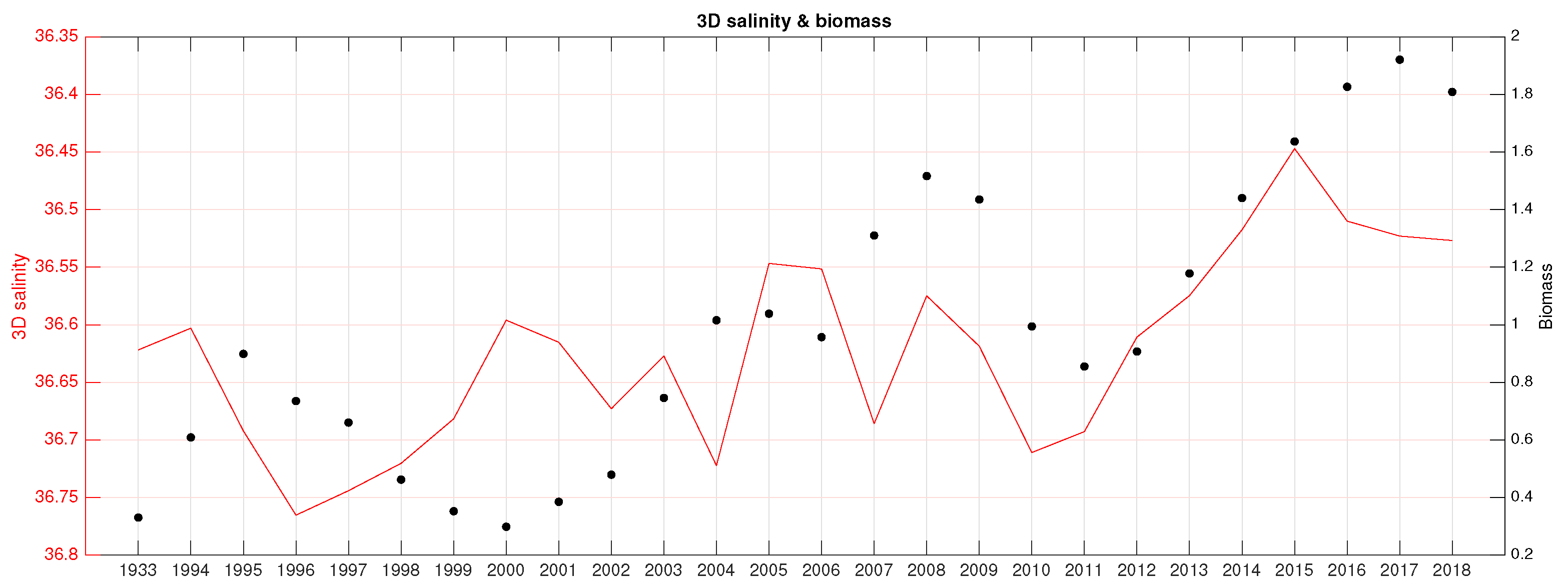
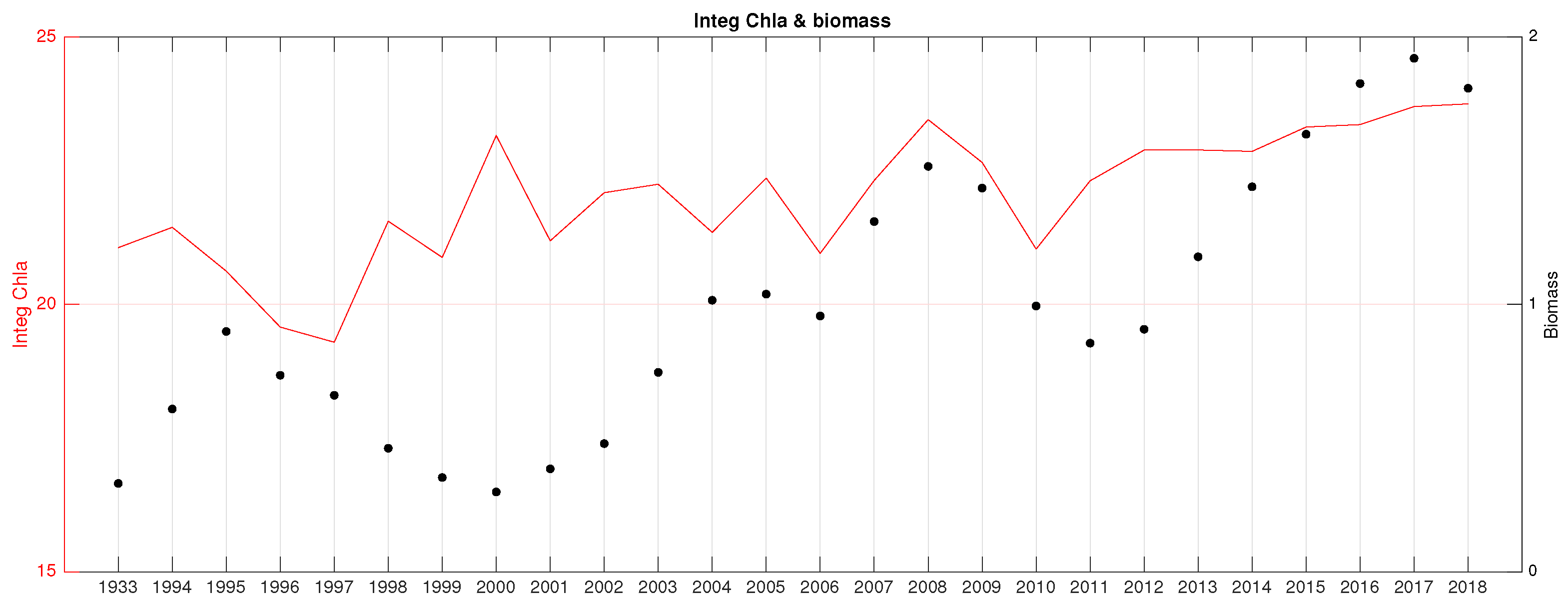
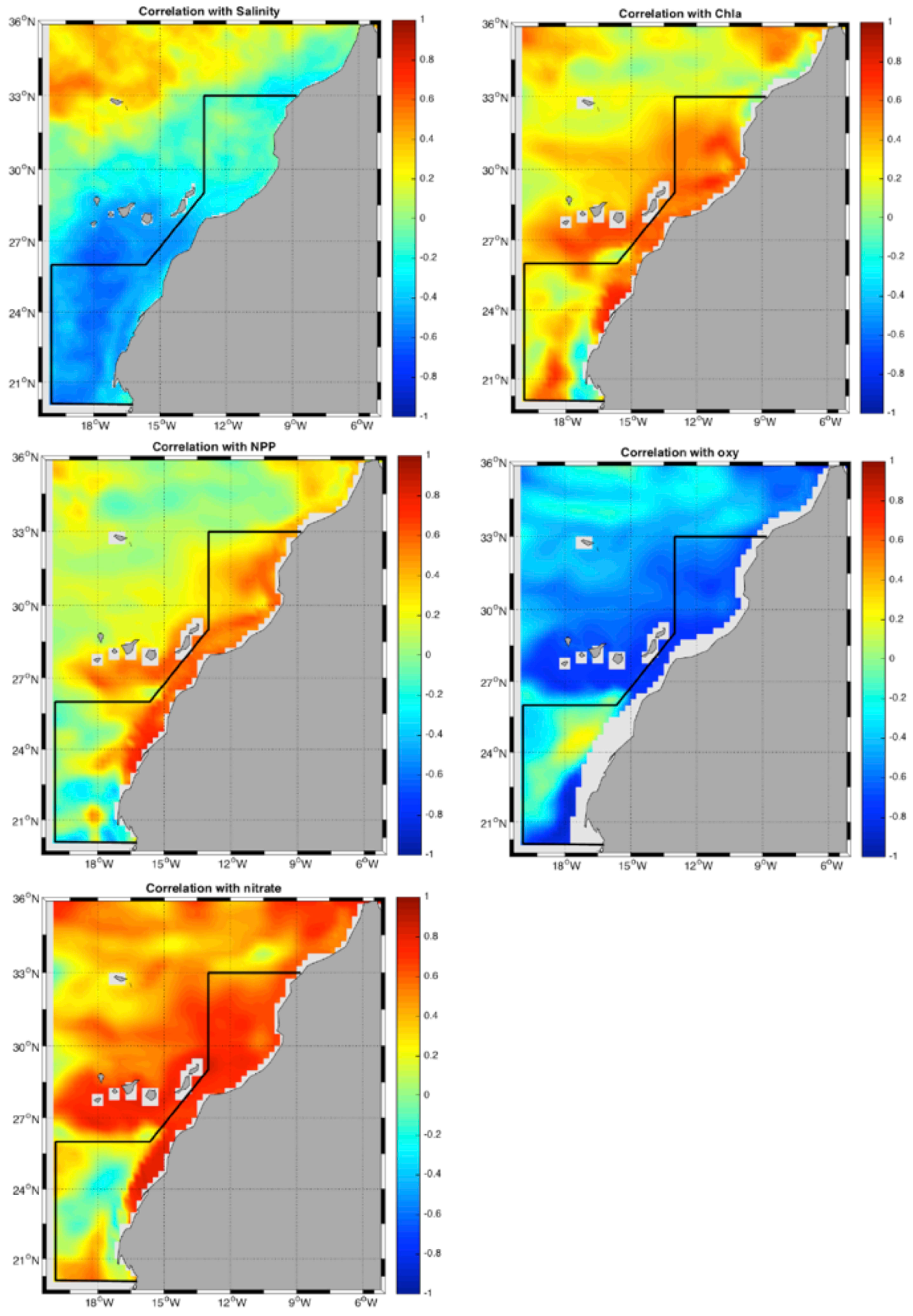
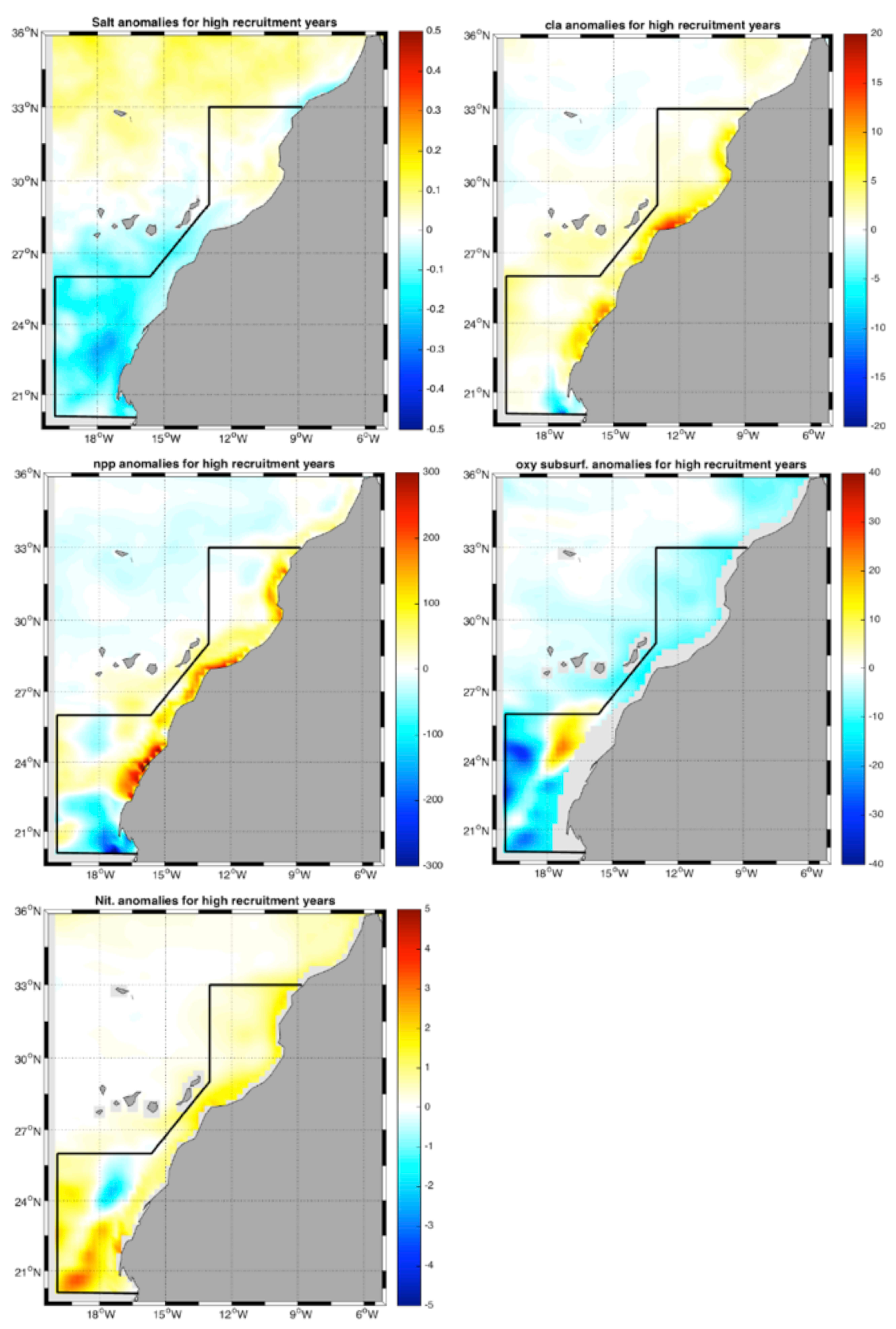
3.3. Correlation Analysis Biomass Trend–Environment
- Mean 3D salinity (3D salt) in the upper 50 m (3D salt) in fall and winter (R∼);
- Mean integrated chlorophyll (chlo) in the upper 150 m in all seasons except winter (R∼);
- Mean integrated net primary production (NPP) in the upper 150 m in all seasons except winter (R∼);
- Subsurface oxygen concentration (Oxy) (100–200 m) in all seasons (R∼);
- Mean integrated nitrate (Nit) in the upper 150 m in all seasons (R∼).
4. Discussion
4.1. Methodological Considerations
4.2. Moroccan Chub Mackerel Fishery
4.3. Environmental Effect on Chub Mackerel Abundance Variability
4.4. Summary
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Cury, P.; Bakun, A.; Crawford, R.J.M.; Jarre, A.; Quiñones, R.A.; Shannon, L.J.; Verheye, H.M. Small pelagics in upwelling systems: Patterns of interaction and structural changes in ’wasp-waist’ ecosystems. ICES J. Mar. Sci. 2000, 57, 603–618. [Google Scholar] [CrossRef]
- FAO. The State of World Fisheries and Aquaculture 2016; Technical Report; FAO: Rome, Italy, 2016. [Google Scholar]
- Fréon, P.; Arístegui, J.; Bertrand, A.; Crawford, R.J.; Field, J.C.; Gibbons, M.J.; Tam, J.; Hutchings, L.; Masski, H.; Mullon, C.; et al. Functional group biodiversity in Eastern Boundary Upwelling Ecosystems questions the wasp-waist trophic structure. Prog. Oceanogr. 2009, 83, 97–106. [Google Scholar] [CrossRef]
- Jeyid, M.A.A. Relations environnement et évolution spatio temporelle des petits poissons pélagiques dans le système d’ Upwelling de la zone NW africaine. Ph.D. Thesis, Université du Littoral Côte d’Opale, Dunkirk, France, 2016. [Google Scholar]
- Crawford, R.J.M.; Shannon, L.J.; Nelson, G. Environmental change_regimes and middle sized pelagic fish in the South East Atlantic Ocean. Sci. Mar. 1995, 3–4, 417–426. [Google Scholar]
- Bas, C. Ecological structures expansion and replacement. In Scientia Marina. Intenational Symposium on Middle-Sized Pelagic Fish; Instituto de Ciencias del Mar: Barcelona, Spain, 1995; Volume 59. [Google Scholar]
- Cury, P.; Roy, C. Optimal environmental window and pelagic fish recruitment success in upwelling areas. Can. J. Fish. Aquat. Sci. 1989, 46, 670–680. [Google Scholar] [CrossRef]
- Checkley, D.M.; Alheit, J.; Oozeki, Y.; Roy, C. Climate Change and Small Pelagic Fish; Cambridge University Press: Cambridge, UK, January 2009; pp. 1–372. [Google Scholar] [CrossRef]
- Fréon, P.; Cury, P.; Shannon, L.; Roy, C. Sustainable exploitation of small pelagic fish stocks challenged by environmental and ecosystem changes: A review. Bull. Mar. Sci. 2005, 76, 385–462. [Google Scholar]
- Porter, S.; Zhisheng, A. Correlation between climate events in the North Atlantic and China during the last glaciation. Nature 1995, 375, 305–308. [Google Scholar] [CrossRef]
- Tilman, D.; Kilham, S.S.; Kilham, P. Phytoplankton community ecology: The role of limiting nutrients. Annu. Rev. Ecol. Syst. 1982, 13, 349–372. [Google Scholar] [CrossRef]
- Arístegui, J.; Barton, E.D.; Álvarez-Salgado, X.A.; Santos, A.M.P.; Figueiras, F.G.; Kifani, S.; Hernández-León, S.; Mason, E.; Machú, E.; Demarcq, H. Sub-regional ecosystem variability in the Canary Current upwelling. Prog. Oceanogr. 2009, 83, 33–48. [Google Scholar] [CrossRef] [Green Version]
- Binet, D. Dynamique du plancton dans les eaux côtières ouest-africaines: écosystènles équilibrés et déséquilibrés. In Pêcheries Ouest—Africaines, Variabilité, Instabilité et Changement; Cury, P., Roy, C., Eds.; IRD Editions: Montpellier, France, 1991; pp. 117–136. [Google Scholar]
- Nieto, K.; Demarcq, H.; McClatchie, S. Mesoscale frontal structures in the Canary Upwelling System: New front and filament detection algorithms applied to spatial and temporal patterns. Remote Sens. Environ. 2012, 123, 339–346. [Google Scholar] [CrossRef]
- INRH/DP. Rapport Annuel de l’Etat des Stocks et des Pêcheries Marocaines 2017; Technical Report; Institut National de Recherche Halieutique: Casablanca, Morocco, 2017; ISSN 2509-1727. [Google Scholar]
- INRH/DP. Rapport Annuel de l’état des Stocks et des Pêcheries Marocaines au Maroc 2019; Technical Report; Département des pêches, Institut National de recherche Halieutique, INRH: Casblanca, Morocco, 2019; ISSN 2509-1727. [Google Scholar]
- Binet, D. Climate and pelagic fisheries in the Canary and Guinea Currents 1964–1993: The role of trade winds and the Southern Oscillation. Oceanol. Acta 1997, 20, 177–190. [Google Scholar]
- Zeeberg, J.J.; Corten, A.; Tjoe-Awie, P.; Coca, J.; Hamady, B. Climate modulates the effects of Sardinella aurita fisheries off Northwest Africa. Fish. Res. 2008, 89, 65–75. [Google Scholar] [CrossRef]
- Houssa, R.; Kifani, S.; Charouki, N. Spatial dynamics of sardine (Sardina pilchardus) and sardinella (Sardinella aurita), two small pelagic fishes in the Canary Current System. Spat. Anal. Fish. Aquat. Sci. 2013, 5, 119–132. [Google Scholar]
- Kifani, S. Climate Dependent Fluctuations of the Moroccan Sardine and Their Impact on Fisheries; Institut Scientifique des Pêches Maritimes: Laayoune, Morocco, 2014; pp. 235–248. [Google Scholar]
- Baali, A.; Charouki, N.; Manchih, K.; Bessa, I.; Elqoraychy, I.; Elqendouci, M.; Amenzoui, K.; Yahyaoui, A. The relationship between Sardinella aurita landings and the environmental factors in Moroccan waters (21°–26° N). Cybium 2019, 43, 51–59. [Google Scholar] [CrossRef]
- Pedersen, M.W.; Berg, C.W. A stochastic surplus production model in continuous time. Fish Fish. 2017, 18, 226–243. [Google Scholar] [CrossRef] [Green Version]
- ICES. Workshop on Atlantic Chub Mackerel (Scomber colias) (WKCOLIAS). ICES Sci. Rep. 2020, 2, 20. [Google Scholar] [CrossRef]
- Al-Eraky, M.M.; Mohamed, N.; Kamel, F.; Al-Qahtani, A.; Madini, M.A.; Hussin, A.; Kamel, N.M.F. Teaching Professionalism By Vignettes In Psychiatry For Nursing Students. Int. J. Adv. Res. 2016, 4, 625–634. [Google Scholar] [CrossRef] [Green Version]
- Bouzzammit, N.; El Ouizgani, H. Morphometric and meristic variation in the atlantic chub mackerel scomber colias Gmelin, 1789 from the Moroccan coast. Indian J. Fish. 2019, 66, 8–15. [Google Scholar] [CrossRef] [Green Version]
- Benazzouz, A.; Mordane, S.; Orbi, A.; Chagdali, M.; Hilmi, K.; Atillah, A.; Lluís Pelegrí, J.; Hervé, D. An improved coastal upwelling index from sea surface temperature using satellite-based approach - The case of the Canary Current upwelling system. Cont. Shelf Res. 2014, 81, 38–54. [Google Scholar] [CrossRef]
- INRH/DRH. Rapport Annuel de l’Etat des Stocks et des Pêcheries Marocaines 2015; Institut National de Recherche Halieutique: Casablanca, Morocco, 2015; p. 295. [Google Scholar]
- Pella, J.J.; Tomlinson, P. A generalized stock production model. Bull. Inter-Am. Trop. Tuna Comm. 1969, 13, 421–458. [Google Scholar]
- Øksendal, B. Stochastic Differential Equations. In Stochastic Differential Equations: An Introduction with Applications; Springer: Berlin/Heidelberg, Germany, 2003; pp. 65–84. [Google Scholar] [CrossRef]
- Ito, K. Lectures on stochastic processes. In Notes by K. Muralidhara Rao. Second edition. Tata Institute of Fundamental Research Lectures on Mathematics and Physics, 24. Distributed for the Tata Institute of Fundamental Research, Bombay; Springer: Berlin/Heidelberg, Germany, 1984; p. iii+233. [Google Scholar]
- Stewart, P. Copemed A Review of Studies of Fishing Gear Selectivity in the Mediterranean; FAO: Rome, Italy, 2001. [Google Scholar]
- FAO. Report of the Working Group on the Assessment of Small Pelagic Fish of Northwest Africa Casablanca, Morocco; Technical Report; FAO: Rome, Italy, July 2020. [Google Scholar] [CrossRef]
- Mohn, R. The retrospective problem in sequential population analysis: An investigation using cod fishery and simulated data. ICES J. Mar. Sci. 1999, 56, 473–488. [Google Scholar] [CrossRef] [Green Version]
- Handbook for the Stochastic Production Model in Continuous Time (SPiCT). Available online: https://github.com/DTUAqua/spict (accessed on 20 May 2021).
- Ghil, M.; Allen, M.R.; Dettinger, M.D.; Ide, K.; Kondrashov, D.; Mann, M.E.; Robertson, A.W.; Saunders, A.; Tian, Y.; Varadi, F.; et al. Advanced spectral methods for climatic time series. Rev. Geophys. 2002, 40, 3-1–3-41. [Google Scholar] [CrossRef] [Green Version]
- Hurtado-ferro, F.; Szuwalski, C.S.; Valero, J.L.; Anderson, S.C.; Cunningham, C.J.; Johnson, K.F.; Licandeo, R.; Mcgilliard, C.R.; Monnahan, C.C.; Muradian, M.L. Looking in the rear-view mirror: Bias and retrospective patterns in integrated, age-structured stock assessment models. ICES J. Mar. Sci. 2015, 72, 99–110. [Google Scholar] [CrossRef]
- Parrish, R.H.; Nelson, C.S.; Bakun, A. Transport Mechanisms and Reproductive Success of Fishes in the California Current. Biol. Oceanogr. 1981, 1, 175–203. [Google Scholar] [CrossRef]
- Belvèze, H. Hydroclimatiques Sur La Pêcherie Marocaine De Petits. In Pêcheries Ouest Africaines: Variabilité, Instabilité et Changement; Philippe, C., Claude, R., Eds.; IRD Editions: Montpellier, France, 1991. [Google Scholar]
- ICES. Working Group on Biological Parameters (WGBIOP); ICES Scientific Reports; ICES: Copenhagen, Denmark, 2019. [Google Scholar] [CrossRef]
- ICES. ICES Reference Points for Stocks in Categories 3 and 4; ICES Technical Guidelines; ICES: Copenhagen, Denmark, 2018. [Google Scholar] [CrossRef]
- Fitzgerald, C.J.; Delanty, K.; Shephard, S. Inland fish stock assessment: Applying data-poor methods from marine systems. Fish. Manag. Ecol. 2018, 25, 240–252. [Google Scholar] [CrossRef]
- Rocha, M.C.; Pennino, M.G.; Cerviño, S.; Izquierdo, F.; Lojo, D.; Paz, A.A.; Tifoura, A.M.; Zanni, M.Y. A Review of Surplus Production Models. Reviews in Fish Biology and Fisheries. Under review.
- Ralston, S.; Punt, A.E.; Hamel, O.S.; Devore, J.D.; Conser, R.J. A meta-analytic approach to quantifying scientific uncertainty in stock assessments. Fish. Bull. 2011, 109, 217–231. [Google Scholar]
- Jensen, A.L. Harvest in a fluctuating environment and conservative harvest for the Fox surplus production model. Ecol. Model. 2005, 182, 1–9. [Google Scholar] [CrossRef]
- Thiaw, M.; Gascuel, D.; Jouffre, D.; Thiaw, O.T. A surplus production model including environmental effects: Application to the Senegalese white shrimp stocks. Prog. Oceanogr. 2009, 83, 351–360. [Google Scholar] [CrossRef]
- Hjort, J. Fluctuations in the great fisheries of Norther Europe. In Rapports et Proce’s-Verbaux des Re´unions du Conseil Permanent International pour l’Exploration de la Mer, En Commission chez ANDR. FRED; H0ST and FILS: Copenhagen, Denmark, 1914; Volume 19, pp. 1–228. [Google Scholar]
- De Oliveira, J.A.; Butterworth, D.S. Limits to the use of environmental indices to reduce risk and/or increase yield in the South African anchovy fishery. Afr. J. Mar. Sci. 2005, 27, 191–203. [Google Scholar] [CrossRef]
- Andre, E. Punt, TzuChuan Huang, M.N.M. Review of integrated size-structured models for stock assessment of hard-to-age crustacean and mollusc species. ICES J. Mar. Sci. 2013, 70, 16–33. [Google Scholar] [CrossRef] [Green Version]
- Rincón, M.M.; Corti, R.; Elvarsson, B.T.; Ramos, F.; Ruiz, J. Granger-causality analysis of integrated-model outputs, a tool to assess external drivers in fishery. Fish. Res. 2019, 213, 42–55. [Google Scholar] [CrossRef] [Green Version]
- Hofmann, E.E.; Powell, T.M. rEnvironmental variability effects on marine fisheries: Four case histories. Ecol. Appl. 1998, 8, S23–S32. [Google Scholar] [CrossRef] [Green Version]
- ICES. Second Workshop on Atlantic chub mackerel (Scomber colias) (WKCOLIAS2). ICES Sci. Rep. 2021, 3, 18. [Google Scholar] [CrossRef]
- Carruthers, T.R.; Walters, C.J.; McAllister, M.K. Evaluating methods that classify fisheries stock status using only fisheries catch data. Fish. Res. 2012, 119–120, 66–79. [Google Scholar] [CrossRef]
- Branch, T.A.; Jensen, O.P.; Ricard, D.; Ye, Y.; Hilborn, R. Contrasting Global Trends in Marine Fishery Status Obtained from Catches and from Stock Assessments. Conserv. Biol. 2011, 25, 777–786. [Google Scholar] [CrossRef]
- Alheit, J.; Roy, C.; Kifani, S. Decadal-Scale Variability in Populations. In Climate Change and Small Pelagic Fish; Cambridge University Press: Cambridge, UK, 2009; pp. 285–299. [Google Scholar] [CrossRef]
- Zorica, B.; Vilibić, I.; Keč, V.Č.; Šepić, J. Environmental conditions conducive to anchovy (Engraulis encrasicolus) spawning in the Adriatic Sea. Fish. Oceanogr. 2013, 22, 32–40. [Google Scholar] [CrossRef]
- Chassot, E.; Bonhommeau, S.; Dulvy, N.K.; Mélin, F.; Watson, R.; Gascuel, D.; Le Pape, O. Global marine primary production constrains fisheries catches. Ecol. Lett. 2010, 13, 495–505. [Google Scholar] [CrossRef]
- Lloret, J.; Palomera, I.; Salat, J.; Sole, I. Impact of freshwater input and wind on landings of anchovy (Engraulis encrasicolus) and sardine (Sardina pilchardus) in shelf waters surrounding the Ebre (Ebro) River delta (north-western Mediterranean). Fish. Oceanogr. 2004, 13, 102–110. [Google Scholar] [CrossRef]
- Gordó-Vilaseca, C.; Pennino, M.G.; Albo-Puigserver, M.; Wolff, M.; Coll, M. Modelling the spatial distribution of Sardina pilchardus and Engraulis encrasicolus spawning habitat in the NW Mediterranean Sea. Mar. Environ. Res. 2021, 169, 105381. [Google Scholar] [CrossRef]
- Franchy, G.; Ojeda, A. Plankton community response to Saharan dust fertilization in subtropical waters off the Canary Islands. Biogeosci. Discuss. 2013, 10, 17275–17307. [Google Scholar] [CrossRef] [Green Version]
- El Mghazli, H.; Mounir, A.; Znari, M.; Naimi, M.; El Ouizgani, H.; Aourir, M. A comparison of spring body condition, maturation status, and diet in the European sardine, Sardina pilchardus (Teleostei: Clupeidae) from contrasting environments off the Moroccan Atlantic coast. Mar. Biol. Res. 2020, 16, 431–445. [Google Scholar] [CrossRef]
- Santander, H. Relationship between anchoveta egg standing stock and parent biomass off Peru. In The Peruvian Anchoveta and Its Upwelling Ecosystem: Three Decades of Change. ICLARM Studies and Reviews; Pauly, D., Tsu-kayama, I., Eds.; IMARPE: Callao, Peru; GTZ: Eschborn, Germany; ICLARM: Manila, Philippines, 1987; pp. 4–143, 179–207. [Google Scholar]
- Lluch-Belda, D.; Elorduy-Garay, J.; Lluch-Cota, S.E.; Ponce-Díaz, G. Centros de Actividad Biológica del Pacífico Mexicano; IRD Edition: Montpellier, France, 1999; pp. 49–64. [Google Scholar]
- Le Fur, J.; Simon, P. A new hypothesis concerning the nature of small pelagic fish clusters. An individual-based modelling study of Sardinella aurita dynamics off West Africa. Ecol. Model. 2009, 220, 1291–1304. [Google Scholar] [CrossRef]
- Ndoye, S.; Capet, X.; Estrade, P.; Sow, B.; Machu, E.; Brochier, T.; Döring, J.; Brehmer, P. Dynamics of a “low-enrichment high-retention” upwelling center over the southern Senegal shelf. Geophys. Res. Lett. 2017, 44, 5034–5043. [Google Scholar] [CrossRef]
- FAO. Report of the FAO Working Group on the Assessment of Small Pelagic Fish off Northwest Africa. Banjul, the Gambia, 18–22 May 2010; Technical Report; FAO Fisheries and Aquaculture Report/FAO Rapport sur les pêches et l’aquaculture. No. 975 Rome; FAO: Rome, Italy, 2011. [Google Scholar]
- Martins, M.M.; Skagen, D.; Marques, V.; Zwolinski, J.; Silva, A. Changes in the abundance and spatial distribution of the Atlantic chub mackerel (Scomber colias) in the pelagic ecosystem and fsheries off Portugal. Sci. Mar. 2013, 77, 551–563. [Google Scholar] [CrossRef] [Green Version]
- INRH/DRH. Rapport Annuel de l’Etat des Stocks et des Pêcheries Marocaines 2016; Technical Report; Institut National de Recherche Halieutique: Casablanca, Morocco, 2013. [Google Scholar]
- INRH/DRH. Etat des Stocks et des Pêcheries Marocaines Synthèse 2018; Technical Report; Institut National de Recherche Halieutique: Casablanca, Morocco, 2014. [Google Scholar]







| Environmental Variables | SST | 3D Temp | 3D Salt | Chlo | NPP | Oxy | Nit |
|---|---|---|---|---|---|---|---|
| For 50 m integration depth | |||||||
| Winter | −0.0001 | 0.009 | −0.6 | 0.43 | 0.43 | −0.83 | 0.38 |
| Spring | −0.05 | −0.10 | −0.38 | 0.61 | 0.57 | −0.80 | 0.45 |
| Summer | 0.26 | 0.17 | −0.36 | 0.54 | 0.48 | −0.65 | 0.43 |
| Fall | 0.31 | 0.22 | −0.51 | 0.58 | 0.47 | −0.73 | 0.43 |
| For 150 m integration depth (except for oxygen with 100–200 m) | |||||||
| Winter | −0.00014 | −0.05 | −0.49 | 0.36 | 0.34 | −0.83 | 0.69 |
| Spring | −0.06 | −0.08 | −0.32 | 0.64 | 0.56 | −0.80 | 0.68 |
| Summer | 0.27 | 0.06 | −0.16 | 0.58 | 0.48 | −0.65 | 0.56 |
| Fall | 0.31 | −0.07 | −0.42 | 0.64 | 0.44 | −0.73 | 0.69 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Derhy, G.; Macías, D.; Elkalay, K.; Khalil, K.; Rincón, M.M. Stochastic Modelling to Assess External Environmental Drivers of Atlantic Chub Mackerel Population Dynamics. Sustainability 2022, 14, 9211. https://doi.org/10.3390/su14159211
Derhy G, Macías D, Elkalay K, Khalil K, Rincón MM. Stochastic Modelling to Assess External Environmental Drivers of Atlantic Chub Mackerel Population Dynamics. Sustainability. 2022; 14(15):9211. https://doi.org/10.3390/su14159211
Chicago/Turabian StyleDerhy, Ghoufrane, Diego Macías, Khalid Elkalay, Karima Khalil, and Margarita María Rincón. 2022. "Stochastic Modelling to Assess External Environmental Drivers of Atlantic Chub Mackerel Population Dynamics" Sustainability 14, no. 15: 9211. https://doi.org/10.3390/su14159211
APA StyleDerhy, G., Macías, D., Elkalay, K., Khalil, K., & Rincón, M. M. (2022). Stochastic Modelling to Assess External Environmental Drivers of Atlantic Chub Mackerel Population Dynamics. Sustainability, 14(15), 9211. https://doi.org/10.3390/su14159211







