Abstract
Currently, the development of new sustainable technologies to recover raw materials from secondary resources has shown a lack of available data on the processes and supplies involved, as well as their environmental impacts. The present research has conducted a life cycle assessment of electrodialytic (ED) technologies to improve critical raw materials recovery in the Portuguese mining industry. To critically appraise the activities from the mining sector and gather data on technical and environmental issues, three waste management scenarios were considered: (1) ED treatment with a deep eutectic solvent as an adjuvant; (2) ED treatment with simultaneous H2 recovery; and (3) ED treatment with sodium chloride as an enhancement. The data presented were based on global databases, technical reports from official sources, and peer-reviewed published experimental outcomes. The estimated results indicated that one of the constraints in applying ED technologies is energy consumption and thus the impacts are highly dependent on energy source choices. On the other hand, as a consequence of the H2 inherently produced by ED technologies, there is a direct potential for energy recovery. Therefore, considering an upscale approach of the ED reactor based on bench scale experimental results, the H2 could be reused in the ED facility or stored. Additionally, according to experimental data, 22% of the tungsten from the fine mine tailings could be recovered. Finally, the possibility to remove 63% of arsenic from mine tailings could decrease contamination risks while creating additional marketable co-products.
1. Introduction
The European Union 2030 climate action targets aim to reduce 55% of the greenhouse gases emissions compared to the values in 1990 [1] and increase the total renewable energy share in energy consumption by up to 32% by 2030 [2]. Moreover, the Sustainable Development Goals include targets for water and energy consumption, waste and biodiversity management, and adaptation of mining operations to climate changing conditions [3].
The European Commission has recently launched the fourth critical raw materials list, where tungsten (W) continues, together with other 29 elements, to be included as a material of high risk of scarcity and economic relevance [4]. Strategies for sustainable reprocessing of mine tailings should be applied to extract valuable raw materials before their disposal or further reuse options.
Tungsten is a valuable transition metal that can be extracted from tungstate minerals [5], such as scheelite (CaWO4) [6] and wolframite ((Fe,Mn)WO4) [7]. Tungsten is applied to produce hard metal and metal carbide powder that can be further treated by powder metallurgical methods for hard metal tools production [8]. If Europe could take full advantage of its own resources, the dependency on other countries to produce hard metal tools could be overcome.
Panasqueira is an underground mine located in Portugal, on the southern edge of the Estrela mountain near the Zezere river, which has operated for more than 130 years. The Panasqueira ores are composed of a series of subhorizontal, stacked, and hydrothermal quartz veins, promoting the mineralization of wolfram-bearing schists and shales. The mineralized area at the Panasqueira mine is 400–2200 m wide, 2500 m long and 500 m deep. During the mining process two types of mine waste are generated: coarse aggregates derived from rock blasting and fine tailings conveyed into dams, both of which have accumulated for more than 100 years [9].
Storage and/or deposition of mine tailings constitute the main threat to the surrounding environment of Panasqueira, particularly via water contamination due to their physical, chemical and mineralogical characteristics and to the volume/ area occupied by them. These residues, namely, the most ancient, may leach harmful elements from storage sites, designated by acid mine drainage [10]. These residues are more exposed to oxygen and water, increasing the chance of acids being formed due to sulfide minerals (e.g., pyrite). Other problems that may arise are related to high levels of cyanide and nitrogen compounds in waters at mine sites from heap leaching and blasting. Particulate matter can be released by the wind from mining activities, such as excavations, blasting, transportation of materials and wind erosion. Moreover, exhaust emissions of the vehicles from mining sites increase the levels of particulates [11]. To prevent and control the pollution from several streams, the Panasqueira mine has an environmental license that complies with limits and conditions for the management of the environmental pressures [12].
The current decrease in ore grade has prompted the assessment of existing resources, energy needs and environmental impacts of mine tailings in a life cycle overview, concerning a circular economy perspective. These approaches play not only an important role in supporting cleaner production, resource management and decision-making in the mining industry, but also in identifying new business opportunities. The current demand and metal prices have leveraged the interest in secondary mining resources for critical raw materials, where the recycling of W has lower-energy negative impacts (<6000 kWht−1) compared to virgin production (10,000 kWht−1), depending on the grade and cut-off [13].
The impacts from the processing of raw materials should be considered during the selection of the Best Available Techniques (BATs), which are the up to date technologies for preventing and minimizing emissions and impacts on the environment [14]. Generally, the BATs promote the improvement of the output and energy efficiency of the raw material production process through replacement of the old equipment with new apparatus, which is less energy consuming [15].
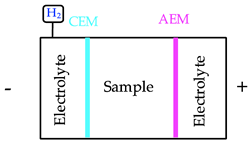
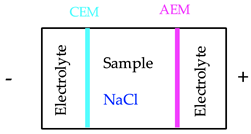
One feasible method to alleviate the impacts of rejected fractions from mining activities is the electrodialytic (ED) process, which consists of the application of a direct low-level current density (mA/cm2) between pairs of electrodes, to remove substances from different environmental substrates. In the ED treatment of mine tailings, anion (AEM) and cation exchange membranes (CEMs) were used to separate the matrix from the electrodes’ compartments [16]. This aimed at controlling the pH conditions of the electrolyte and the matrix, improving the selectivity of the removal of contaminants [17]. The membrane surface attracts dissolved ions with the opposite charge (counter-ions) from the pore water of membranes. Thus, the counter-ions are transported through the membrane due to the electrical current while co-ions, that have the same charge of the membrane, are rejected [17].
Research has been performed to assess the feasibility of applying the ED treatment to W mine tailings to (1) recover W contents and other elements of interest [16], (2) remove harmful compounds [18], (3) to recover H2 that is inherently produced during the treatment [19,20], and (4) to provide a suitable matrix for further reuse in the construction sector as a supplementary cementitious material [21]. Furthermore, the ED process has demonstrated potential to extract W present in fine tailings (approximately 22%) in the presence of biodegradable acid adjuvants—natural deep eutectic solvents (DES) [18].
Life cycle assessment (LCA) is an analysis technique applied to assess potential environmental impacts of a product/service from, e.g., raw material acquisition to waste disposal. LCA provides an estimation of cumulative impacts under environmental categories such as global warming, ozone layer depletion, soil and water acidification, eutrophication, and abiotic depletion of non-fossil and fossil resources [22]. According to ISO 14040 [23] and ISO 14044+A1 [24], the LCA process is systematic and divided into four phases: (1) goal and scope definition; (2) inventory analysis; (3) impact assessment; and (4) interpretation [25]. LCA requires a detailed inventory analysis to ensure a representative assessment of all the inputs and outputs of mass and energy across the whole phases of the product life cycle, designated by life cycle inventory (LCI) [26].
The upgrades on technical processes should, therefore, consider the environmental, social, and economic consequences of managing mine tailings throughout the value chain. For instance, orebody characterization, mine planning, processing, disposal, reprocessing, recycling, and reuse should be integrated. From an economic point of view, decreasing mine tailings is a top priority, followed by reuse, recycling, recovery treatment and disposal. Circular business models supported by public policies could have strategic importance, where economic benefits/incentives could be essential to optimize the recycling management system while increasing recycling rates [27].
The trade-off between raw material extraction from secondary mining resources and the environmental harmfulness of mine tailings after the ED process, as well as the need to critically understand the information that LCA studies can offer, were the base of the present work. Thus, this research shows the potential environmental impacts that should be considered in an LCA when the ED technology is applied to Panasqueira fine tailings. In addition, an upscale hypothetical approach of the ED reactor, based on laboratory experimental results, was developed. The impacts estimated and determined in several scopes are presented in terms of mine tailing management scenarios (direct disposal vs. ED remediation prior to discharge/further reuse options). In this context, three different operational ED conditions were considered: (1) ED treatment with a deep eutectic solvent as an adjuvant; (2) ED treatment with simultaneous H2 recovery; and (3) ED treatment with sodium chloride as an enhancement.
2. Materials and Methods
2.1. Case Description and Production System
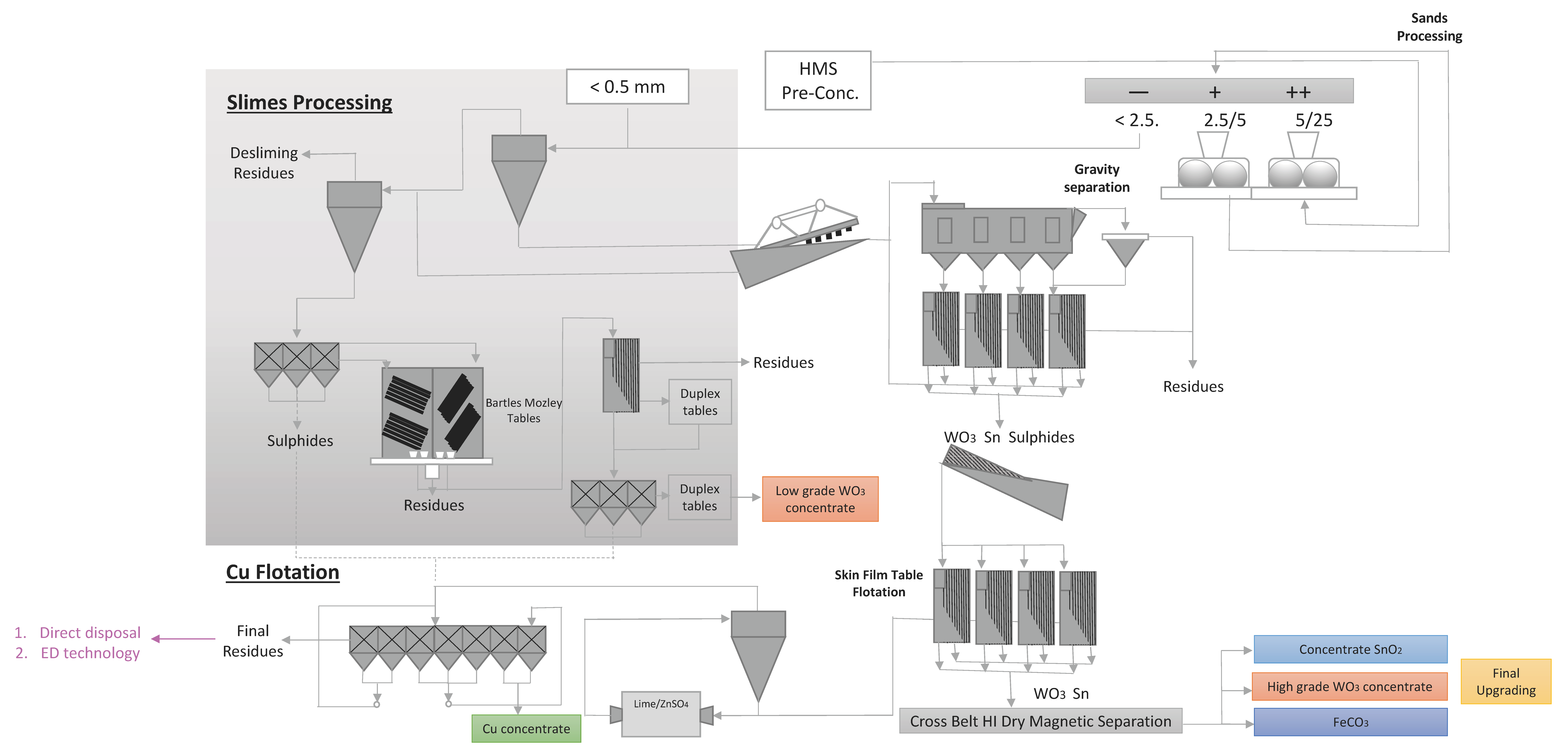
The Panasqueira mine, located in the Centro region of Portugal (Figure 1) and described as the largest Sn–W deposit in Western Europe, has changed its layout over the years due to the W market downturns. In this way, the optimization of mechanical processes and the exploration of alternative ores were considered. The Panasqueira mine process is summarized in Figure 2. The mine has a production plan extending to 30 years in the future [9].
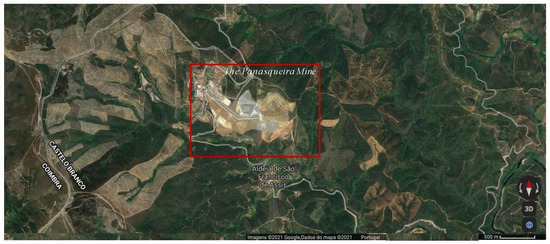
Figure 1.
The Panasqueira mine geografic location—Covilhã, Portugal, 40°10′11.0604″ N, 7°45′23.8752″ W (source: Google maps).
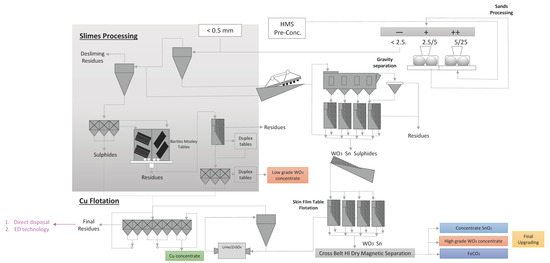
Figure 2.
The Panasqueira mine process and scenarios considered for the management of mine tailings generated (based on [28]). HMS—Heavy media separation.
Wolframite, cassiterite and chalcopyrite are the minerals extracted and used to produce W, copper (Cu) and tin (Sn) concentrates, respectively. The extraction process consists of a room and pillar method, considering geo-mechanical and geological properties of the rock mass. The first stage for the production of W is crushing and milling of the ore, promoting the release of the W mineral from the material. Then, a heavy media separation (HMS) between fine and coarse fractions is performed. This stage promotes the removal of 80% of the ore that does not contain W. Then, the W preconcentrated fraction is subjected to a conventional gravity concentration technique, followed by magnetic flotation in the presence of sulfide and dry magnetic separation [29]. The process is mostly gravitational due to the relative density of the concentrated products in relation to the sterile material [30].
Additionally, there is a wastewater treatment facility, the Mine Water Treatment Station (MWTS), located in Salgueira. The MWTS was projected to treat a maximum of 500 m3/h, where the wastewater comes from the mine, wash activities and heaps [30].
2.2. System Boundaries and LCA Road Map
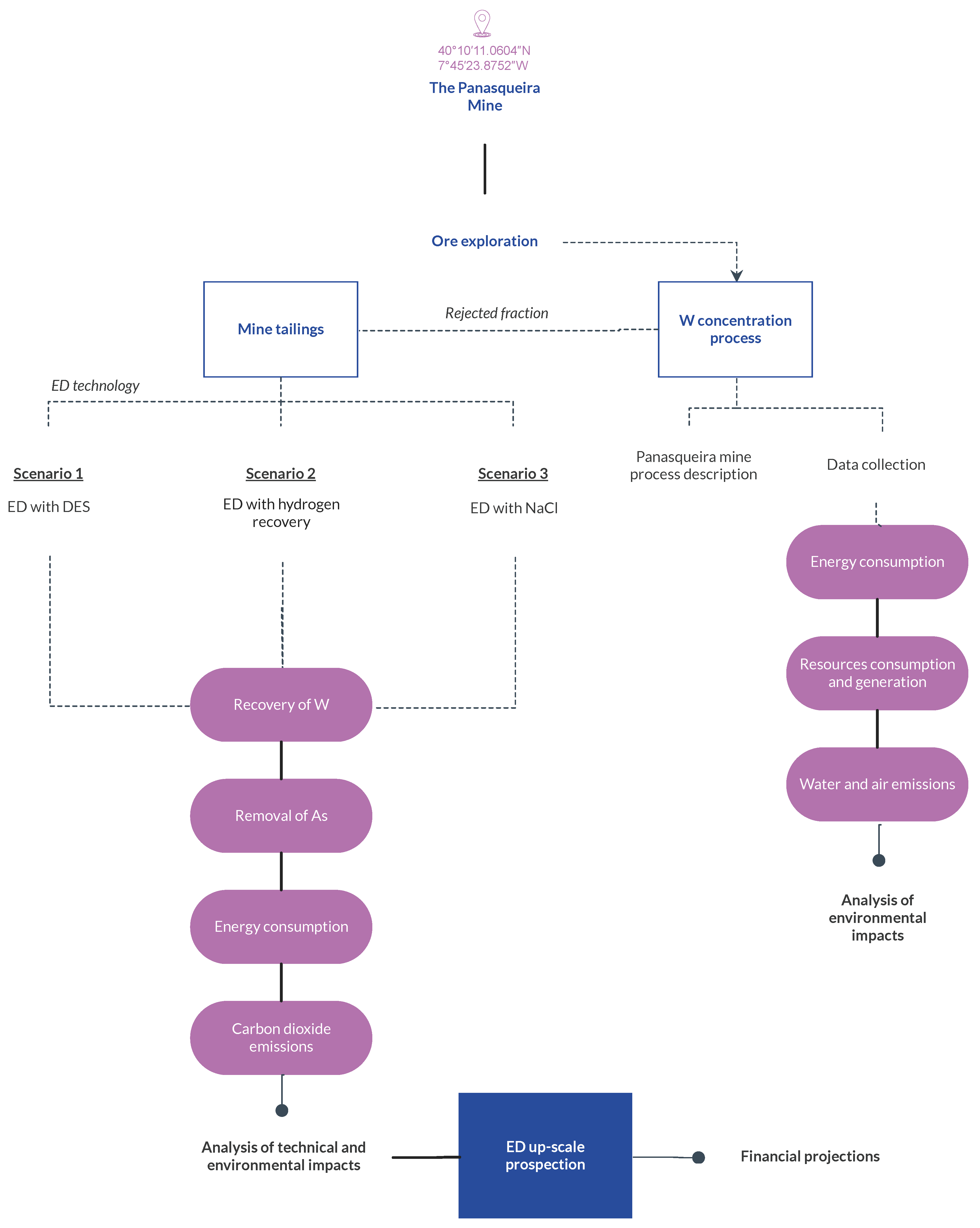
Figure 3 shows the steps considered for the present LCA study. The analysis starts on the exploration of the ore for the W concentrate production. Then, for the rejected fraction of the mining process, three waste management scenarios were considered: (1) ED treatment with DES as an adjuvant; (2) ED treatment with simultaneous H2 recovery; and (3) ED treatment with sodium chloride (NaCl) as an enhancement. Based on the analysis of the ED scenarios, an ED upscaling study was carried out that also coupled financial projections.
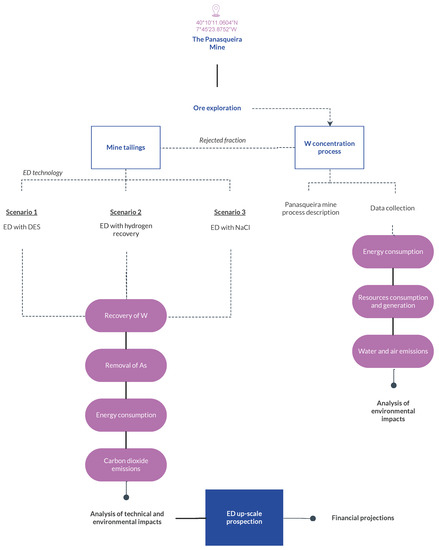
Figure 3.
Flowchart of the life cycle analysis performed for the production of W concentrate and the electrodialytic treatment of mine tailings.
2.3. Data Collection
The data used to build the LCA were selected from three main sources: Ecoinvent database version 3.7.1, technical reports published from the European Commission and results from laboratory works published in international scientific journals (Table 1).

Table 1.
Main sources used for the Life cycle assessment (LCA) data collection.
2.4. Mine Tailings Characterization
Considering the Panasqueira mine plant (Figure 2), the processing of 1000 kg of ore (from excavation activities) was considered as the functional unit for the LCA study. Table 2 presents estimations regarding W concentrate production and mine tailings generation from the processing of 1000 kg of ore. The calculations were based on scientific data available on Panasqueira resource compositions, as referenced in Table 2.

Table 2.
Resources consumed and generated during the production of tungsten concentrate at the Panasqueira mine.
Thus, concerning Table 2, from the processing of 1000 kg of ore, only 2.3 kg of W concentrate is produced, with grades of 75% of WO3. This means that around 997 kg of fine tailings are generated from the processing of 1000 kg of ore. From the mine tailings it is possible to recover 0.8 kg of W/t of ore. The W price (25,500 EUR/t) makes the W recovery attractive from an economic perspective.
2.5. Water and Air Emissions and Resources Consumed
The processing of 1000 kg of ore was selected as the functional unit for the LCA. Water and air emissions during W concentrate production were determined based on Ecoinvent database [31]. In the Ecoinvent platform, the information is presented considering 1 t of W concentrate production. Thus, an extrapolation based on the W concentrate production at the Panasqueira mine was used to estimate its environmental impacts.
The resources consumed and generated during the production of tungsten concentrate at the Panasqueira mine were determined, considering the amount of fine tailings that results from the processing of 1000 kg of ore. In this sense, the amount produced from fine tailings generation was determined by subtracting the initial fraction (1000 kg ore) of the quantities of W, Cu and Sn concentrates produced. The W contents in mine tailings per functional unit were determined based on the concentrate grade of W trioxide (WO3) produced in the plant—75% [29].
The plant also produces Cu and Sn concentrates. However, the amounts of Cu and Sn were only considered to estimate the fine tailings generation, and production impacts were not considered for this study.
2.6. Energy Consumption and CO2 Release
To estimate the energy consumed by the ED system, Equation (1) was applied:
where Vi is the average voltage (Volts) in time i, A is the current intensity (Amperes) and t is the duration (hours) of the experiment. To convert the energy consumed into the quantity of CO2 released for the environment, a conversion factor of 0.23314 kg CO2 per kWh was considered [36].
3. Results and Discussion
3.1. Tungsten Concentrate Production at the Panasqueira Mine: Environmental Impacts
3.1.1. Energy Consumption
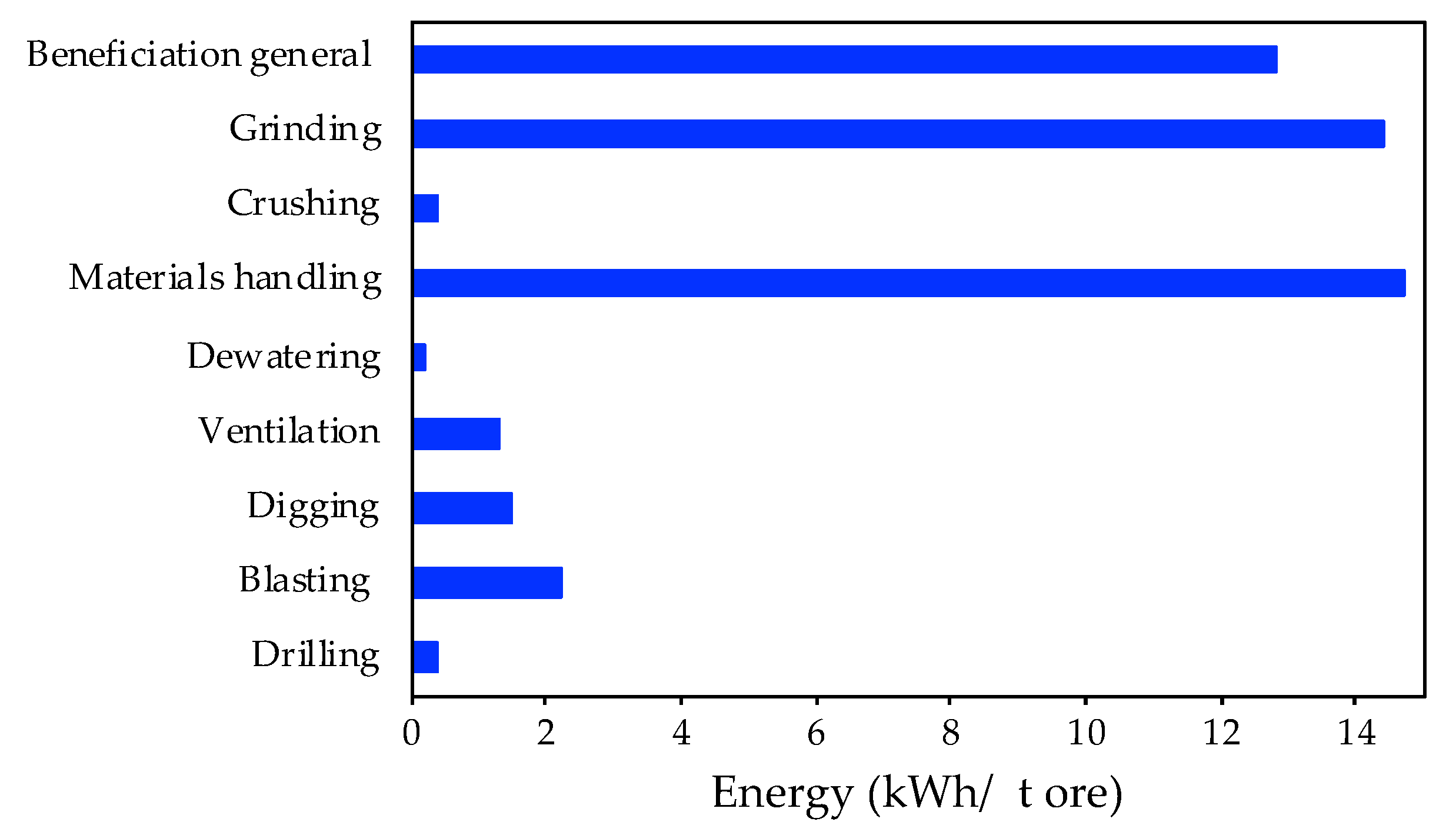
During the W concentrate production at the mining site, there are several high energy consuming phases. The overview presented in Figure 4 is based on data from technical reports. Herein, an average of the energy consumption of each processing step per ton of processed ore during the W production is shown.
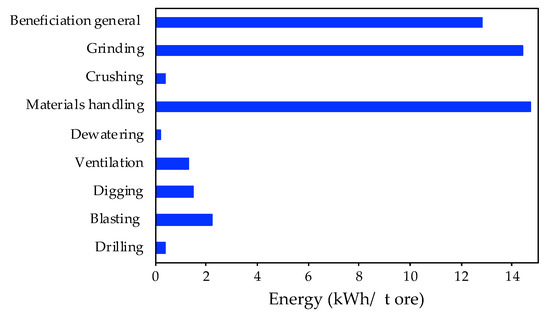
Figure 4.
Energy consumption of mining processing activities (adapted from [37]).
The high energy consuming stages are materials handling (more than 14 kWh/t ore) and grinding (14 kWh/t ore). On the other hand, crushing, dewatering and drilling activities present lower energy consumptions (below 1 kWh/t ore). In addition, the environmental categories which are more affected by those processing steps are global warming potential (12.6 kg CO2 eq/t ore), cumulative energy demand (133 MJ eq/kg), terrestrial acidification (0.29 kg SO2 eq/t ore) and human toxicity (3.4 × 10−5 CTUh/t ore) [37].
3.1.2. Air and Water Emissions
During the processing of 1000 kg of ore, air and water emissions are generated, causing the main environmental pressures. Table 3 presents the emissions that were determined for Panasqueira mine processing based on Ecoinvent data from W concentrate production impacts.

Table 3.
Emissions to the environment during the tungsten concentrate production (determined based on Ecoinvent data for tungsten concentrate production [31]).
Carbon dioxide is the major substance released to air (0.35 kg/t ore) and as observed in Figure 4, the materials handling and grinding are the main operations contributing to its release, in addition to the emission of particles. Regarding water load emissions per ton of ore, the chemical parameters that have higher impacts on aquatic systems are chemical oxygen demand (COD) (4.15 × 10−3 kg), biochemical oxygen demand (BOD5) (2.08 × 10−3 kg) and total organic carbon (TOC) (1.54 × 10−3 kg).
COD is generally used to indirectly determine the amount of organic compounds in aquatic systems and is useful as an indicator of the degree of organic pollution in surface waters [38]. The COD is the most affected parameter by mining activities (4.15 × 10−3 kg/t ore), indicating that not all forms of organic matter (biodegradable and nonbiodegradable) are available.
The BOD5 presents the second highest impacts (2.08 × 10−3 kg/t ore), meaning a decrease in the amount of oxygen consumed, by aerobic biological organisms, to decompose the organic matter in 5 days. An excessive microbial activity causes a reduction in the quantity of oxygen in the water, which may foment the growth of anaerobic organisms and decay the development of other existing communities [39].
The dissolved organic carbon (DOC), which is a potential source of carbon and energy for ecosystem metabolism, plays a central role in many limnological processes, since it is largely derived from terrestrial vegetation, deposited from lake catchments either by streams or by overland flow [40]. The mining impacts in terms of DOC are less pronounced (1.54 × 10−3 kg/t ore), although changes in DOC cycling can result in air–water exchange of CO2 alterations [41].
Mining processes demonstrated the lowest impacts on the total organic carbon (TOC) (1.54 × 10−3 kg/t ore), mainly in the form of DOC. The TOC measures the amount of carbon found in an organic compound. A high organic content means a higher oxygen consumption and, consequently, an increase in the growth of microorganisms that deplete oxygen supplies [42].
The chemical water contamination is reflected in composition changes, while physical contamination results from the presence of fine material, affecting both surface water and aquifers. In the case of metallic ores, chemical contamination can be relevant due to the oxidation of pyrite producing sulfides that may leach heavy metals. In addition, yellowish to red Fe(OH)3 precipitates are formed when acidic water meets neutral water in river basins. These precipitates affect the growth of aquatic plants [30]. Physical contamination by fines occurs when there is a discharge of treatment effluents in water courses. Contamination by suspended solids seriously affects fish communities [30]. In the case of the Panasqueira mine, the Zezere river is the main concern of water pollution.
Mining at levels below the water table will subsequently promote risks of evaporation of aquifers, water degradation by eutrophication and chemical contamination [43]. Acid mining drainage is characterized by a pH below 5 and is related to the sulfides (S2− and S22−), sulfur (S) or thiosulfate (S2O32−) being in contact with water and the atmospheric oxygen (oxidizing conditions). Acid water results from the oxidation of pyrite (FeS2), usually catalyzed by bacteria. Other sulfides such as blends (ZnS), galena (PbS), chalcopyrite (CuFeS2), pyrrhotite (Fe7S8) and arsenopyrite (FeAsS) can contribute to acidifying water resources. Generally, acidic effluents present high concentrations of Fe, manganese (Mn) and aluminum (Al) [30].
To decrease water resources contamination in the area, the Panasqueira mine has an in-house Mine Water Treatment Station (MWTS). The wastewater comes from the surface through infiltration and from the production process, since a significant amount of water is used during drilling and irrigation of the work fronts. The wastewater treatment facility has a volume capacity of 7000 m3. The outlet and the receiving tank were designed to convert relatively soluble ions, such as Fe2+ and Mn2+, into the respective less soluble oxidized forms (Fe3+ and Mn4+). The treatment plant is composed of four tanks with mechanical agitation in which the addition of flocculant and lime is carried out. Lime is added to increase the pH of acidic water and prevent a possible drop in pH when ions such as Fe3+ and Mn4+ precipitate in the form of hydroxides. The solid hydroxides formed and in suspension are deposited at the bottom of the tank, being pumped into the mud dam [30].
3.2. Mine Tailings Management
3.2.1. Electrodialytic Scenarios
Following the assessment of air and water emissions during W concentrate production, the environmental pressures of mine tailings were studied in particular in terms of ED process efficiency for elements extraction and recovery, energy consumption and CO2 emissions. This assessment was based on experimental data from previous works. In fact, the major impacts from waste disposal at mine sites can be divided into two categories [37]:
(1) the loss of productive land following its conversion to a waste storage area;
(2) the introduction of sediment, acidity, and other contaminates into surrounding surface water and groundwater from water running over exposed problematic or chemically reactive wastes, and the consequent soil contamination.
In this sense, alternative ways to direct disposal of mine tailings are desired. Table 4 presents three different scenarios where the ED technology was studied as an alternative to direct disposal. The systems defined have diverse extraction ratios of elements and other features, being important to compare the ED scenarios in terms of achievements and environmental impacts.

Table 4.
Data from the electrodialytic treatment of mine tailings.
Scenario 1 involves the use of a DES in the sample compartment during the ED treatment. DES present advantages in terms of yield, costs and toxicity when compared to conventional ionic liquids [44], which are composed of strong acids and bases [45]. In this way, a two-compartment reactor was operated for 4 days at 0.05 A. The electrolyte and sample sections were separated by an anion exchange membrane [18]. The main outcome of this reactor configuration was the quantity of W extracted from the matrix. The use of choline chloride/oxalic acid (1:1) promoted a higher solubilization of the W and, together with the current applied, a synergetic effect on the recovery of this critical element was observed.
Scenario 2 includes a three-compartment ED reactor and the simultaneous collection of self-produced H2 in an eco-friendlier manner. This configuration presented the lower W recovery, where only 7.5% of W was extracted. However, it should be noted that this system operated only for 1 hour at 0.1 A. Applying this current intensity was possible to access the production of H2, an energy carrier, with 74% H2 purity. Electrokinetics have been coupled with electrodialytic technology for H2 production during the removal of pollutants [20], where the energy requirements for this system are considerably smaller owing to the higher conductivity of the matrix [46]. Additionally, regarding the As removal, a better performance was achieved (48%) in comparison with scenario 1 (16%). Considering the functional unit defined for this study, hypothetically, this system could avoid 1.8 kg of As contamination (Table 4).
Finally, in scenario 3, a three-compartment ED reactor was tested at 0.1 A for 5 days using NaCl to improve the current passage [19]. The main achievement of this system was a 63% As removal from a total of 3.7 kg As/t ore (Table 2), meaning less than 2.4 kg of As contamination per t of ore (Table 4).
Regarding different market segments and environmental concerns, the scenarios considered may show different potentials. In this way, the trade-off for the three scenarios was studied to understand the impacts on resources consumption and its economic feasibility. Table 5 presents an energy assessment for the three bench scale ED treatment scenarios. These tests were performed to treat 39 [18] and 22 g [16,20] of fine tailings in a two- and/or three-compartment ED reactor, respectively. The energy was determined according to Equation (1).

Table 5.
Estimation of the energy consumed and CO2 released during the electrodialytic laboratory treatment of mine tailings.
The highest energy consumption occurred in scenario 3, where a three-compartment reactor and NaCl were used. An average of 5.0 × 10−3 kWh was consumed, with a release of 1.2 g CO2. In fact, this system was operated at a higher current intensity (0.1 A) and thus, it was expected to have a higher energy consumption, and consequently, higher amount of CO2 release. However, the addition of NaCl promoted the control of the power consumption once it led to an increase in media conductivity and therefore lower resistance [47].
On the other hand, scenario 1, performed with natural extractants (DES), demonstrated a decrease in the energy consumption of more than 80%. In this set-up, a current intensity of 0.05 A was applied, which was the main contributor to the energy consumption decrease (1.0 × 10−3 kWh) when compared to the other two scenarios. This means approximately 0.9 g of CO2 emissions to the environment. In fact, this scenario presented the lowest energy consumption per mass of elements extracted (6.0 × 10−3 kWh/g W and 2.5 E × 10−5 kWh/g As).
Scenario 2, which includes H2 recovery, demonstrated an intermediate energy consumption. In scenario 2, as in scenario 3, a current intensity of 0.1 A was applied. However, due to the use of a totally sealed reactor (to ensure no leakage of gases), a decrease in the voltage occurred and thus a decrease in the resistivity inside the reactor was observed. Based on Ohm’s law, if the current intensity is constant, the voltage and the resistivity (or conductivity) are strongly related. The decrease in the ED cell voltage is linked to a conductivity increase in the electrodes’ compartments [20].
This may explain the lower energy consumed (3.0 × 10−3 kWh) compared to scenario 3 (ED with NaCl). Additionally, the possibility to recover the self-hydrogen produced by the ED treatment and reuse to feed the reactor in terms of energetic requirements could provide energy savings of up to 50% [32].
Regarding the water needs for sample suspensions and electrolyte preparation with sodium nitrate (NaNO3), ED configurations that have three compartments require 700 mL (32 times the sample weight) and the two-compartment systems require 600 mL (15 times the sample weight). However, the reuse of secondary water resources during the ED treatment (e.g., secondary effluent) have shown promising results [16] that may contribute to alleviate tap water needs.
Summing up, the laboratory experiments could follow structural designs, such as a central factorial design. However, it is pivotal to properly assess the industrial interest. In a scale-up perspective, the experiments should be carried out sequentially, followed by a process analysis and economic evaluation. Even in the first steps of the research, which can affect the experimental domain of interest, the quality of the information provided could be improved and be key factor for a pilot unit.
3.2.2. Electrodialytic Treatment Upscale Prospection
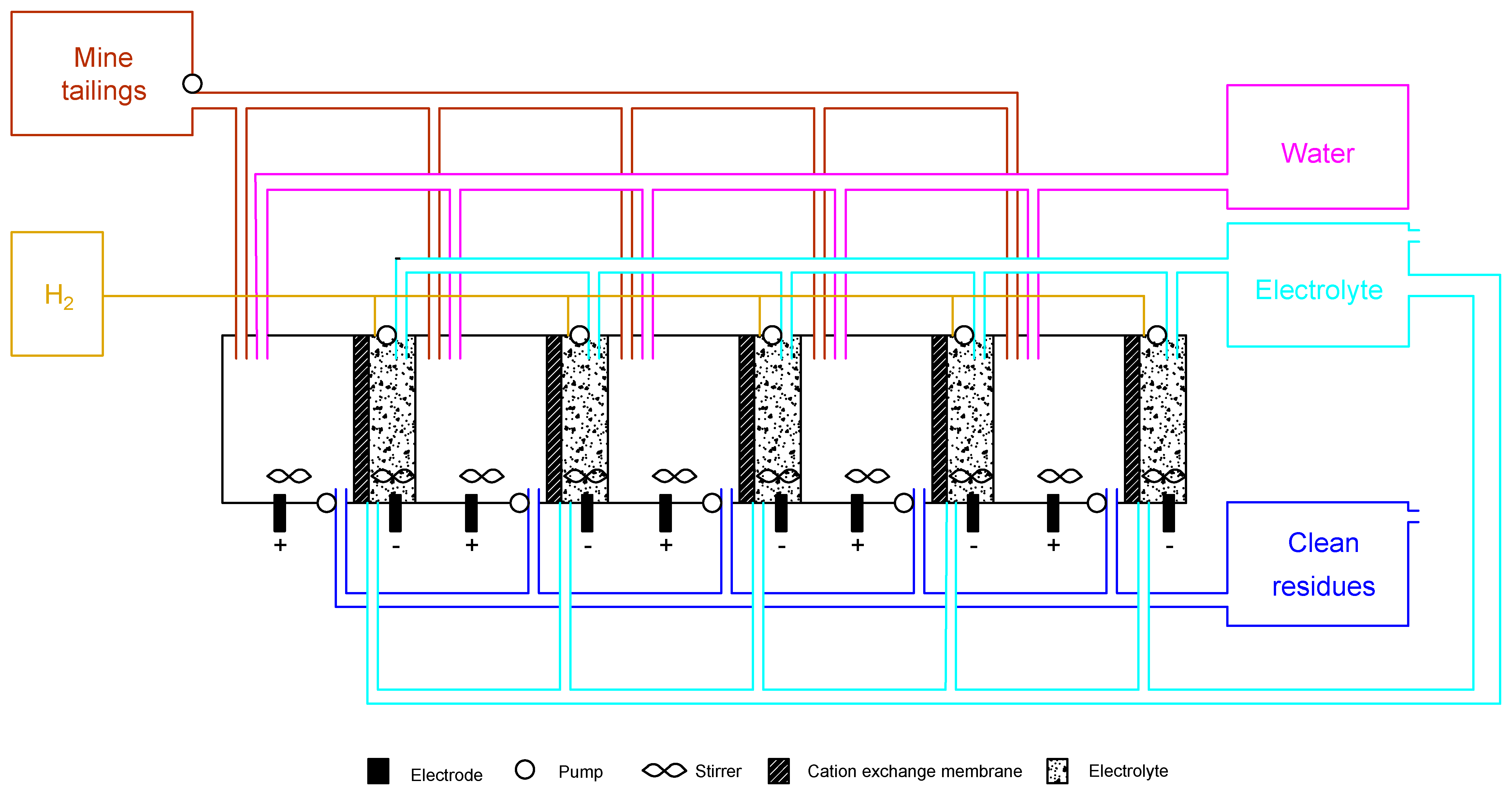
In order to increase the understanding of the scenarios studied, a perspective of a full scalable ED reactor model to support commercial roll-out was carried out. The implementation of the theoretical ED pilot reactor would result in a full running removal and recovery of the target compounds from the fine mine tailings, hypothetically aiming zero liquid or solid discharges. A full closed-loop of residues would change the perspective of mine tailings, which would become a valuable resource instead of a costly waste stream.
The ED plant, which can be either vertical or horizontal, is presented in Figure 5, showing a simplified flow sheet of a loop reactor. It is important to point out that the design of the reactor does not need to be similar to the laboratory scheme. However, it has to be designed to achieve the best data, both in terms of fluid dynamics and transport properties. In Figure 5, the design of the reactor is used as an example of the concept, and merges the best parts of both scenarios 1 and 2:
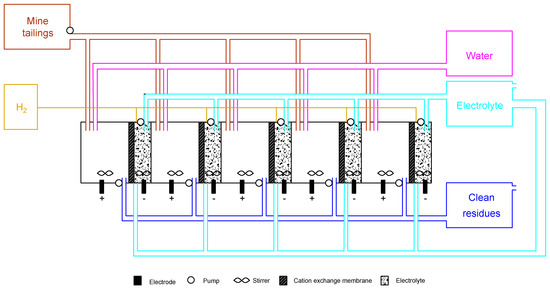
Figure 5.
Theoretical electrodialytic plant facility based on bench scale experiments.
- (1)
- two-compartment reactor design, which is easier to operate;
- (2)
- DES as enhancements, alleviating the consumption of strong acids and bases while incrementing the W recovery;
- (3)
- cation-exchange membrane, which allows H2 recovery for depreciation of implementation and maintenance costs, as well as flexibility in different market segments.
A balance between the ED treatment plant and downstream units needs to be ensured in order to decrease environmental pressures from the disposal of mine tailings. The final product (after the ED treatment) needs to have such a quality that consecutive ED phases will work optimally at minimal operational costs. Since the Panasqueira mine has available land, the installation of solar panels in the south direction with 144 cells at 400 W (2025 × 996 × 40 cm) [48] will promote the use of renewable energy and overcome the environmental pressures and costs regarding ED technology. The use of solar panels decreases the investment cost by avoiding the use of batteries, solar inverters, and power supplies and the maintenance cost since there is no battery waste to manage [49].
Additionally, the scaled-up reactor was dimensioned addressing issues related to a seamless operation with minimal needs for cleaning. The material selected for the reactor was polyethylene due to its chemical and impact resistance, electrical properties and low coefficient of friction. In addition, polyethylene is lightweight, easily processed and offers near-zero moisture absorption [50]. The reactor was dimensioned to treat 10 m3 per day (8 h running time, based on average labor schedules in Portugal) in five conjugate sequential units. The capacity of each block is 2 m3, as reported by other pilot studies [51]. These dimensions promote the treatment of the volume of mine tailings produced per day (0.4 m3), considering: (1) mud’s density of 3031 kg/m3 [52]; (2) 997 kg volume of mine tailings; (3) water needs both for the electrolyte (NaNO3 0.01 M) and sample suspensions. The ED facility was projected to be fully distributed by individual blocks. Each section includes pumps (when gravity transport is not possible), membranes, and sample and electrolyte compartments. Additionally, it includes reservoirs for ED treated and nontreated mine tailings (before the ED process), water, electrolyte and H2, to either reuse or storage. This simplifies the maintenance of the reactor and, consequently, reduces the problems during the treatment.
Scaling in ED occurs due to inorganic species, e.g., PO43−, NH4+, Mg2+, Ca2+ and CO32−, forming low-solubility minerals. The more effective the process is at removing these elements, the less scale will be formed and therefore the maintenance costs for the ED unit will be lower [53].
A constraint of the ED facility is the goal of achieving total reuse of water, since there is a need to treat and/or crystallize the salt from the brine stream (NaNO3) and the effluent suspension produced. There are several technological options for waste brine crystallization. The projection of the ED facility intends to concentrate and crystallize the salts in the brine solution. Crystallization plays an important role in many industries where water recycling is implemented. If it is not possible to perform the purification of brine solutions, the Panasqueira MWTS (capacity = 500 m3/h) [30] could also include the treatment of both brine solution and effluent suspension. In addition, treated water resources from the Panasqueira MWTS could be used to prepare the mine tailings suspensions, aiming for circularity of by-products. This step would promote savings in water consumption by the generation of suitable mixtures to reuse in the ED system.
Moreover, the proposed ED unit combines a reservoir for the collection of the self-produced H2, increasing the value proposition of the ED pilot. Lead-acid batteries are the most useable storage systems, as well as rechargeable batteries, supercapacitors, and redox flow batteries. The most promising systems for renewable energy storage are the lithium-ion batteries and redox flow batteries [49]. Coupling a unit for H2 recovery at the mine can direct the site towards a clean energy transition [54]. In fact, the implementation of H2 as a flexible energy carrier in future energy systems is a top priority in the new EU Green Deal. The smart integration across sectors is encouraged and promotes investments on cutting-edge research and innovation for clean energies [1]. Together with W recovery and As removal, the H2 transfer to be used in the mine ED facility could have socio-economic impacts both on recovery of raw materials and clean energy transition, since it addresses applicable solutions to industries. This stimulates the fostering of synergies in industrial sectors, the creation of new services and the transition and adaptation to eco-innovated systems. The ED treated fine tailings can be further used in construction materials with compatible applications with conventional concrete and fired brick masonry walls [21]. On the other hand, the As can be used in purification processes of zinc leach solutions [55], and residues containing As could be recovered by glass industries, since arsenates can be turned into silicoarsenates during vitrification [56].
Further roll-out may be hampered by the lack on financial commitment to implementation in a declining industry, where investments in utilities are not seen as strategic to the core business. However, low investment solutions with limited capital expenditures (CAPEX) and operating expenditures (OPEX) costs are required. Table 6 presents the financial projections of the ED plant for the initial investment, as well as the first and the fifth years of ED operation.

Table 6.
Financial projections of the expenditures of implementing the theoretical electrodialytic plant facility presented in Figure 5.
It should be noted that Table 6 shows a simplified economic approach of the ED concept, considering only its physical implementation. Through this assessment it is possible to understand the impact of these figures on the broad uptake potential in the mine industry. This section does not cover the fully CAPEX or OPEX estimating procedures. Nevertheless, it provides concepts that can be used in the project evaluation to help the understanding of its application in a full implementation mode. Moreover, Table 6 also details the costs of the components needed for the ED facility. The materials for the ED plant construction (e.g., electrodes, membranes, pumps, tubes) and the reagents needed for the electrolyte and sample suspensions (NaNO3 and DES) are the main contributors of the costs reported in the first- and fifth-year projections. Together with the costs for the manufacturing, and, therefore, the total investment in the first year, a set of other expenses to guarantee the success of the ED process during its lifetime is also foreseen. The total investment before developing the ED facility in a full run mode is approximately 51,000 euros, increasing from around 1 to 7 millions of euros in the first and fifth years, mainly due to NaNO3 and DES consumption.
The investments can be considered high, although further optimization of the processes and research could decrease the values presented. Additionally, the up-scaling theoretical approach should be further optimized, based on a pilot study, to decrease inputs related to energy and resources in a more positive way. This strategy may promote minimization of the negative pressures in the environment and the adaptation of industrial sectors to eco-innovative markets. In particular, the ED plant presented could leverage new market possibilities, the requalification of mining areas after close and the development of new technologies with regard to achieving the Sustainable Development Goals [3].
4. Conclusions
Mining industries have been stimulated to operate in a more sustainable way, reducing their environmental burdens and improving resource management. In this way, eco-efficient processes and alternative scenarios to direct waste disposal of rejected fractions are desired.
This research work evaluated the impacts on the environment that may come from mining processes and three potential scenarios that involve the ED treatment of fine tailings from the Panasqueira mine.
Regarding the impacts of mining processes, materials handling and grinding presented the highest energy consumption and, consequently, CO2 release (0.35 kg/functional unit). A carbon footprint of 12.6 kg CO2 eq/t ore is associated with mining activities. On the other hand, the chemical parameters that have the highest impacts on aquatic systems are COD (4.15 × 10−3 kg), BOD5 (2.08 × 10−3 kg) and TOC (1.54 × 10−3 kg).
The analysis of previous ED data at the bench scale was a key factor to estimate the potential environmental burdens involved in an ED plant conception. In particular, energy consumption is a major concern at an industrial scale. Herein, the ED technology could have a central role in the recovery of metals below 45 microns, a main challenge for the mine. In addition, the mine has a project spanning more than 30 years, which means that new market segments could be explored to keep the development and the requalification of the Panasqueira area after its closure. In this context, aligned with the removal of As contents, the recovery of W and H2 seems to be attractive for the development of the Centro region of Portugal, considering a circular economy perspective, both in terms of raw materials recovery and sustainable energy production. These aspects might decrease the risk associated with mining activities and leverage new business opportunities in the mining sector in the upcoming years.
Thus, concerning the three scenarios studied, different advantages were pointed out. Scenario 1, which involves the use of DES, exhibited a better performance in terms of the quantity of W extracted from the matrix (22%). Scenario 2 considers self-produced H2 recovery during the ED treatment with 74% H2 purity. Scenario 3 presents an approach with conventional reagents and the main achievement of this system was the As removal (63%).
Based on the best features of the scenarios analyzed, a sequential theoretical ED facility was presented. The dimensioning of the reactor was based on the quantity of mine tailings that need to be treated and laboratory data. An investment of approximately 51,000 euros was estimated, increasing from 1 to 7 millions euros in the first and fifth years due to maintenance and operational costs. Nevertheless, the upscaling effect may reduce the inputs—namely, those regarding operational costs and energy consumption. In future works, a pilot study of the ED treatment should be performed to assess the scaling-up influence on technical aspects and to determine the economy of scale’s percentage.
This study provides new insights for the life cycle of mine tailings and a basis for environmental decision-support in the application and roll-out of ED technologies.
Author Contributions
Conceptualization, J.A. and C.M.; Data curation, J.A. and C.M.; Formal analysis, E.P.M. and A.B.R.; Funding acquisition, A.B.R.; Methodology, J.A.; Project administration, A.B.R.; Resources, E.P.M., and A.B.R.; Supervision, A.B.R.; Validation, E.P.M. and A.B.R.; Writing—original draft, J.A.; Writing—review and editing, J.A., C.M., E.P.M., and A.B.R. All authors have read and agreed to the published version of the manuscript.
Funding
This work has received funding from the European Union’s Horizon 2020 research and innovation program under the Marie Skłodowska-Curie grant agreement No. 778045. The authors acknowledge the Portuguese Foundation for Science and Technology (FCT) for the support given to CENSE through the strategic project UIDB/04085/2020 and for J. Almeida PhD fellowship PD/BD/135170/2017 from the EcoCoRe Doctoral program.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Acknowledgments
The authors acknowledge Eng. Manuel Pacheco from Panasqueira mine for providing the sample and Eng. António Galvão for the support in LCA development. This research is anchored by the RESOLUTION LAB, an infrastructure at NOVA School of Science and Technology.
Conflicts of Interest
The authors declare no conflict of interest.
References
- European Commission. Communication from the Commission to the European Parliament, the European Council, the Council, the European Economic and Social Committee and the Committee of the Regions—The European Green Deal. COM/2019/640 final; EC: Brussels, Belgium, 2019. [Google Scholar]
- European Commission. Going Climate-Neutral by 2050—A Strategic Long Term Vision for a Prosperous, Modern, Competitive and Climate-Neutral EU; EC: Brussels, Belgium, 2019. [Google Scholar]
- Mancini, L.; Vidal-Legaz, B.; Vizzarri, M.; Wittmer, D.; Grassi, G.; Pennington, D.W. Mapping the role of raw materials in sustainable development goals. In Sustainable Development Goals. A preliminary Analysis of Links, Monitoring Indicators and Related Policy Initiatives; P O of the European Union: Luxembourg, 2019; p. JRC112892. ISBN 978-92-76-08385. [Google Scholar]
- European Commission. Critical Raw Materials Resilience: Charting a Path towards Greater Security and Sustainability. COM/2020/474 Final; EC: Brussels, Belgium, 2020. [Google Scholar]
- Sharma, P.; Minakshi, M.; Whale, J.; Jean-Fulcrand, A.; Garnweitner, G. Effect of the anionic counterpart: Molybdate vs. tungstate in energy storage for pseudo-capacitor applications. Nanomaterials 2021, 11, 580. [Google Scholar] [CrossRef] [PubMed]
- Minakshi, M.; Mitchell, D.R.G.; Baur, C.; Chable, J.; Barlow, A.J.; Fichtner, M.; Banerjee, A.; Chakraborty, S.; Ahuja, R. Phase evolution in calcium molybdate nanoparticles as a function of synthesis temperature and its electrochemical effect on energy storage. Nanoscale Adv. 2019, 1, 565–580. [Google Scholar] [CrossRef]
- Yang, X. Beneficiation studies of tungsten ores—A review. Miner. Eng. 2018, 125, 111–119. [Google Scholar] [CrossRef]
- European Commission. Integrated Pollution Prevention and Control (IPPC): Reference Document on Best Available Techniques in the Non Ferrous Metals Industries; EC: Belgium, Brussels, 2001. [Google Scholar]
- Ávila, P.F.; Silva, E.F.; Salgueiro, A.R.; Farinha, J.A. Geochemistry and mineralogy of mill tailings impoundments from the Panasqueira Mine (Portugal): Implications for the surrounding environment. Mine Water Environ. 2008, 27, 210–224. [Google Scholar] [CrossRef]
- Candeias, C.; Ávila, P.F.; Silva, E.F.; Ferreira, A.; Salgueiro, A.R.; Teixeira, J.P. Acid mine drainage from the Panasqueira mine and its influence on Zêzere river (Central Portugal). J. Afr. Earth Sci. 2014, 99, 705–712. [Google Scholar] [CrossRef]
- Environmental Law Alliance. Worldwide Guidebook for Evaluating Mining Project EIAs, 1st ed; ELAW: Eugene, OR, USA, 2010. [Google Scholar]
- Agência Portuguesa do Ambiente. Environmental License No 347/0.1/2017—Extraction and Preparation of Non-Ferrous Metal Ores, Comprising Hazardous and Non-Hazardous Waste Disposal Activities; APDO: Lisbon, Portugal, 2017. (In Portuguese) [Google Scholar]
- Okvist, L.; Ye, G.; Hu, X. Innovation Potential in the Recovery of Refractory Metals from Secondary Resources—MSP-REFRAM H2020 project; D3.3; EC: Brussels, Belgium, 2016. [Google Scholar]
- Valderrama, C.; Granados, R.; Cortina, J.L.; Gasol, C.M.; Guillem, M.; Josa, A. Implementation of best available techniques in cement manufacturing: A life-cycle assessment study. J. Clean. Prod. 2012, 25, 60–67. [Google Scholar] [CrossRef]
- Garbarino, E.; Orveillon, G.; Saveyn, H.; Barthe, P.; Eder, P. Best Available Techniques (BAT) Reference Document for the Management of Waste from Extractive Industries in Accordance with Directive 2006/21/EC|EU Science Hub; European Commission: Brussels, Belgium, 2018. [Google Scholar]
- Almeida, J.; Magro, C.; Rosário, A.R.; Mateus, E.P.; Ribeiro, A.B. Electrodialytic treatment of secondary mining resources for raw materials extraction: Reactor design assessment. Sci. Total Environ. 2020, 752, 141822. [Google Scholar] [CrossRef] [PubMed]
- Ribeiro, A.B.; Rodríguez-Maroto, J.M. Electroremediation of heavy metal-contaminated soils -processes and applications. In Trace Elements in the Environment: Biogeochemistry, Biotechnology, and Bioremediation; Prasad, M.N.V., Sajwan, K.S., Naidu, R., Eds.; CRC Press: Boca Raton, FL, USA, 2006; pp. 341–368. ISBN 9781420032048. [Google Scholar]
- Almeida, J.; Craveiro, R.; Faria, P.; Silva, A.S.; Mateus, E.P.; Barreiros, S.; Paiva, A.; Ribeiro, A.B. Electrodialytic removal of tungsten and arsenic from secondary mine resources—deep eutectic solvents enhancement. Sci. Total Environ. 2020, 710, 136364. [Google Scholar] [CrossRef] [PubMed]
- Almeida, J.; Magro, C.; Mateus, E.P.; Ribeiro, A.B. Electrodialytic hydrogen production and critical raw materials recovery from secondary resources. Water 2020, 12, 1262. [Google Scholar] [CrossRef]
- Magro, C.; Almeida, J.; Paz-Garcia, J.M.; Mateus, E.P.; Ribeiro, A.B. Exploring hydrogen production for self-energy generation in electroremediation: A proof of concept. Appl. Energy 2019, 255, 113839. [Google Scholar] [CrossRef]
- Almeida, J.; Faria, P.; Ribeiro, A.B.; Silva, A.S. Effect of mining residues treated with an electrodialytic technology on cement-based mortars. Clean. Eng. Technol. 2020, 1, 100001. [Google Scholar] [CrossRef]
- European Commission. CEN EN 15804:2012+A2:2019—Sustainability of Construction Works. Environmental Product Declarations. Core Rules for the Product Category of Construction Products; EC: Brussels, Belgium, 2012. [Google Scholar]
- International Organization for Standardization. Environmental Management: Life Cycle Assessment. Principles and Framework; ISO: Geneva, Switzerland, 2006. [Google Scholar]
- International Organization for Standardization. Environmental Management: Life Cycle Assessment. Requirements and Guidelines; ISO: Geneva, Switzerland, 2006. [Google Scholar]
- Santos, T.; Almeida, J.; Silvestre, J.; Faria, P. Life cycle assessment of mortars: A review on technical potential and drawbacks. Constr. Build. Mater. 2021, 288, 123069. [Google Scholar] [CrossRef]
- Cuenca-Moyano, G.M.; Zanni, S.; Bonoli, A.; Valverde-Palacios, I. Development of the life cycle inventory of masonry mortar made of natural and recycled aggregates. J. Clean. Prod. 2017, 140, 1272–1286. [Google Scholar] [CrossRef]
- Almeida, J.; Ribeiro, A.B.; Silva, A.S.; Faria, P. Overview of mining residues incorporation in construction materials and barriers for full-scale application. J. Build. Eng. 2020, 29, 101215. [Google Scholar] [CrossRef]
- Leite, M. Reflection contributions on the topic of useful substances formation from extractive production residues. In Proceedings of the Mining Waste: Contributions to the Circular Economy Seminar, Lisbon, Portugal, 26 June 2017. (In Portuguese). [Google Scholar]
- Franco, A.; Vieira, R.; Bunting, R. The Panasqueira Mine at a Glance; International Tungsten Industry Association Newsletter: London, UK, 2014; Volume 3, pp. 1–12. [Google Scholar]
- Gonçalves, D. Recovery of Heavy Metals from Waste Water; Internship Report, The Panasqueira mine; University of Beira Interior: Covilhã, Portugal, 2015. (In Portuguese) [Google Scholar]
- Ecoinvent Tungsten Mine Operation and Beneficiation. Zurich, Switzerland. Available online: https://v371.ecoquery.ecoinvent.org/Details/UPR/c054cf42-f2c0-49ad-a431-a2ef7b4b13fe/8b738ea0-f89e-4627-8679-433616064e82 (accessed on 8 January 2021).
- Magro, C.; Almeida, J.; Paz-Garcia, J.; Mateus, E.; Ribeiro, A. Hydrogen recovery in electrodialytic-based technologies applied to environmental contaminated matrices. In Electrokinetic Remediation for Environmental Security and Sustainability; Ribeiro, A., Prasad, M., Eds.; John Wiley & Sons Ltd: Hoboken, NJ, USA, 2021; pp. 251–270. ISBN 978-1-119-67011-7. [Google Scholar]
- GOV.UK Streamlined Energy and Carbon Reporting (SECR) UK. Available online: https://www.gov.uk/government/publications/academy-trust-financial-management-good-practice-guides/streamlined-energy-and-carbon-reporting (accessed on 26 January 2021).
- Politis, A.; Paspaliaris, I.; Taxiarchou, M. Management of Wastes from Primary Resource Processing: Identification, Environmental Evaluations-MSP-REFRAM H2020 Project; EU: Luxembourg, 2017; Volume D2.4. [Google Scholar]
- Candeias, C.; Melo, R.; Ávila, P.F.; Ferreira da Silva, E.; Salgueiro, A.R.; Teixeira, J.P. Heavy metal pollution in mine-soil-plant system in S. Francisco de Assis—Panasqueira mine (Portugal). Appl. Geochem. 2014, 44, 12–26. [Google Scholar] [CrossRef]
- Metalary Tungsten Price 2020. Available online: https://www.metalary.com/tungsten-price/ (accessed on 11 January 2021).
- International Energy Agency Global Average Levelised Cost of Hydrogen Production by Energy Source and Technology, 2019 and 2050. Available online: https://www.iea.org/data-and-statistics/charts/global-average-levelised-cost-of-hydrogen-production-by-energy-source-and-technology-2019-and-2050 (accessed on 22 March 2021).
- Islam, M.M.M.; Shafi, S.; Bandh, S.A.; Shameem, N. Impact of environmental changes and human activities on bacterial diversity of lakes. In Freshwater Microbiology; Bandh, S.A., Shafi, S., Shameem, N., Eds.; Elsevier: London, UK, 2019; pp. 105–136. ISBN 9780128174951. [Google Scholar]
- Donoso, N.; Gobeyn, S.; Villa-Cox, G.; Boets, P.; Meers, E.; Goethals, P.L.M. Assessing the ecological relevance of organic discharge limits for constructed wetlands by means of a model-based analysis. Water 2018, 10, 63. [Google Scholar] [CrossRef]
- Antoniades, D. Paleolimnology—Lake chemistry. In Encyclopedia of Quaternary Science; Elias, S., Ed.; Elsevier: London, UK, 2007; pp. 2038–2046. ISBN 9780444527479. [Google Scholar]
- Lønborg, C.; Carreira, C.; Jickells, T.; Álvarez-Salgado, X.A. Impacts of global change on ocean dissolved organic carbon (DOC) cycling. Front. Mar. Sci. 2020, 7, 466. [Google Scholar] [CrossRef]
- Roudi, A.M.; Kamyab, H.; Chelliapan, S.; Ashokkumar, V.; Kumar, A.; Yadav, K.K.; Gupta, N. Application of response surface method for total organic carbon reduction in leachate treatment using Fenton process. Environ. Technol. Innov. 2020, 19, 101009. [Google Scholar] [CrossRef]
- Pacheco, M. Panasqueira. Atypical Mining Project. Master’s Thesis, University of Porto, Portugal, 2017. [Google Scholar]
- Schaeffer, N.; Martins, M.A.R.; Neves, C.M.S.S.; Pinho, S.P.; Coutinho, J.A.P. Sustainable hydrophobic terpene-based eutectic solvents for the extraction and separation of metals. Chem. Commun. 2018, 54, 8104–8107. [Google Scholar] [CrossRef]
- Abbott, A.P.; Frisch, G.; Hartley, J.; Ryder, K.S. Processing of metals and metal oxides using ionic liquids. Green Chem. 2011, 13, 471–481. [Google Scholar] [CrossRef]
- Miller de Melo Henrique, J.; Cañizares, P.; Saez, C.; Vieira dos Santos, E.; Rodrigo, M.A. Relevance of gaseous flows in electrochemically assisted soil thermal remediation. Curr. Opin. Electrochem. 2021, 100698. [Google Scholar] [CrossRef]
- Shalaby, A.; Nassef, E.; Mubark, A.; Hussein, M. Phosphate removal from wastewater by electrocoagulation using aluminium electrodes. Am. J. Environ. Eng. Sci. 2014, 1, 90–98. [Google Scholar]
- Greenice. Toledo, Spain. Available online: https://greenice.com/pt/816-economia-de-energia (accessed on 29 January 2021).
- Ganiyu, S.O.; Martínez-Huitle, C.A. The use of renewable energies driving electrochemical technologies for environmental applications. Curr. Opin. Electrochem. 2020, 22, 211–220. [Google Scholar] [CrossRef]
- Chave Vertical Lda. Aveiro, Portugal. Available online: http://www.chavevertical.pt/crbst_192.html (accessed on 29 January 2021).
- Christensen, I.V.; Pedersen, A.J.; Ottosen, L.M.; Ribeiro, A.B. Electrodialytic remediation of CCA-treated waste wood in a 2 m3 pilot plant. Sci. Total Environ. 2006, 364, 45–54. [Google Scholar] [CrossRef] [PubMed]
- Castro-Gomes, J.; Magrinho, M.; Sedira, N.; Beghoura, I.; Humbert, P.; Manso, M.; Fernandes, A.; Silva, R. Alkali-activation of tungsten mining waste mud blended with waste glass: Reactivity, performance and innovative applications. In Proceedings of the ICEUBI 2017—International Conference on Engineering—A Vision for the Future, Covilhã, Portugal, 5–7 December 2017. [Google Scholar]
- Thompson Brewster, E.; Ward, A.J.; Mehta, C.M.; Radjenovic, J.; Batstone, D.J. Predicting scale formation during electrodialytic nutrient recovery. Water Res. 2017, 110, 202–210. [Google Scholar] [CrossRef] [PubMed]
- United Nations. Transforming Our World: The 2030 Agenda for Sustainable Development. A/RES/70/1; UN: New York, NY, USA, 2015. [Google Scholar]
- Liu, W.; Huang, C.; Han, J.; Qin, W. Removal and reuse of arsenic from arsenic-bearing purified residue by alkaline pressure oxidative leaching and reduction of As (V). Hydrometallurgy 2021, 199, 105541. [Google Scholar] [CrossRef]
- U.S. Environmental Protection Agency. Arsenic Treatment Technologies for Soil, Waste, and Water; U.S. Environmental Protection Agency: Washington, DC, USA, 2002.
- Denios online shop. Porto, Portugal. Available online: https://www.denios.pt/shop/ (accessed on 29 January 2020).
- ABC do Acrílico. Almada, Portugal. Available online: http://www.abcdoacrilico.com (accessed on 29 January 2021).
- Changsheng Titanium Co., Ltd. Baoji City, China. Available online: https://www.bjcstytitanium.com/products (accessed on 29 January 2021).
- Xion Water Solutions. London, UK. Available online: http://xionwater.com/products.html (accessed on 29 January 2021).
- Merck KGaA., Darmstadt, Germany. Available online: https://www.merckgroup.com/en/expertise.html (accessed on 29 January 2021).
- La Tienda del Apicultor. Valencia, Spain. Available online: https://www.latiendadelapicultor.com/pt/ (accessed on 29 January 2021).
- Chemical Book. China. Available online: https://www.chemicalbook.com/ProductList_En.aspx?kwd=cholinechloride (accessed on 29 January 2021).
- Ferramentas.pt. Seixal, Portugal. Available online: https://ferramentas.pt (accessed on 29 January 2021).
- Paipu Technology Co., Ltd. Changzhou, China. Available online: http://www.paiputech.com/EN/Productlist.Asp?SortID=48 (accessed on 29 January 2021).
- Expondo. Portugal. Available online: https://www.expondo.pt/artigos-industriais/ (accessed on 29 January 2021).
- PT Robotics. Rio de Mouro, Portugal. Available online: https://www.ptrobotics.com (accessed on 29 January 2021).
- European Commission. Guidelines on the Eligibility of Costs under the Connecting Europe Facility; EC: Brussels, Belgium, 2018. [Google Scholar]
- Andrade, L.H.; Mendes, F.D.S.; Espindola, J.C.; Amaral, M.C.S. Reuse of dairy wastewater treated by membrane bioreactor and nanofiltration: Technical and economic feasibility. Braz. J. Chem. Eng. 2015, 32, 735–747. [Google Scholar] [CrossRef]
- Samhaber, W.M.; Nguyen, M.T. Applicability and costs of nanofiltration in combination with photocatalysis for the treatment of dye house effluents. Beilstein J. Nanotechnol. 2014, 5, 476–484. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).