A Pilot Project to Limit the Human Impacts on the Fragile Antarctic Biota: Mitigation of a Runway through Vegetation Transplantation
Abstract
1. Introduction
2. Materials and Methods
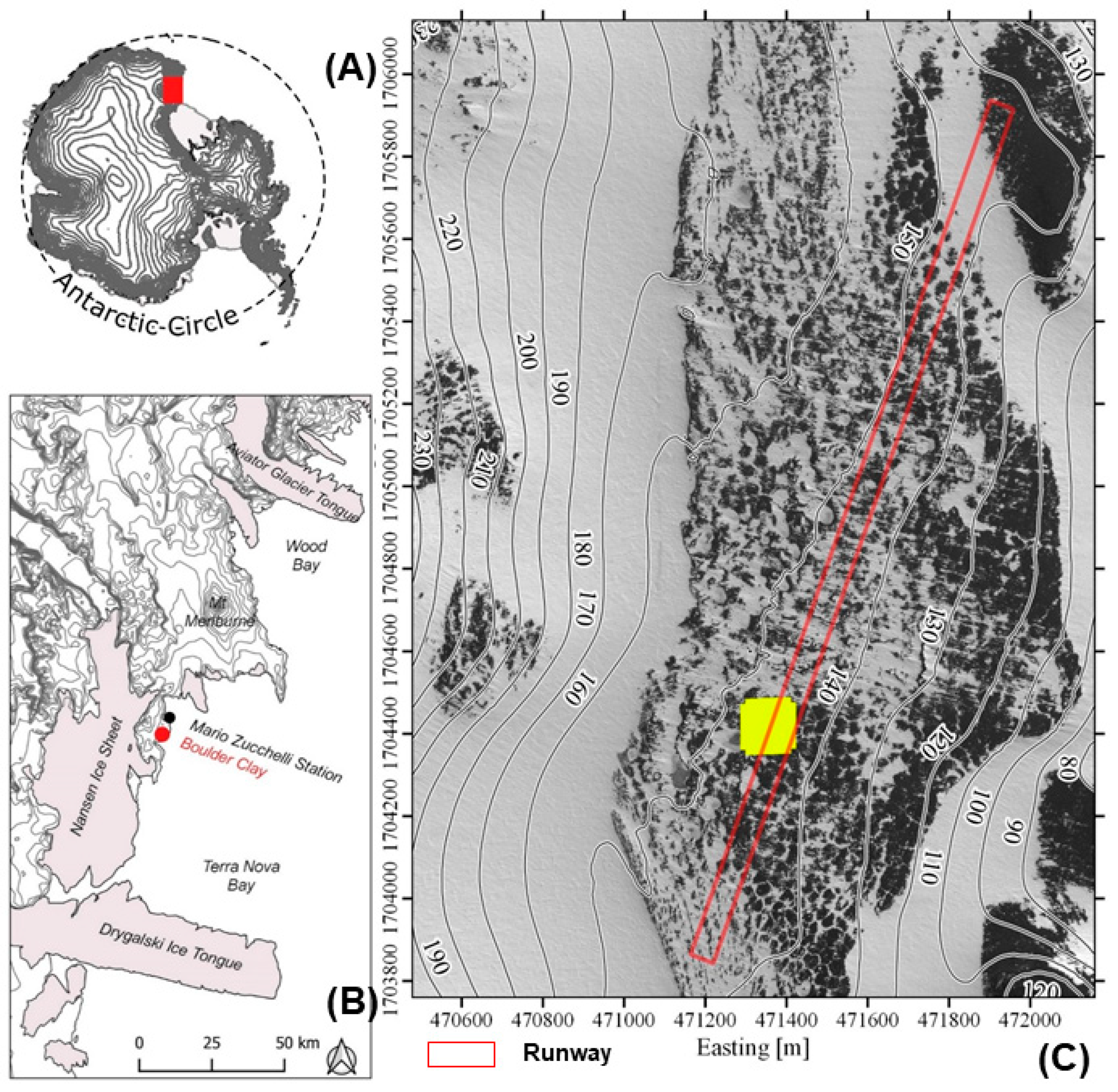
2.1. Study Area
- -
- Buellia frigida dominating (macrolichens and microlichens);
- -
- Lecidella siplei—Bryophytes (mixed lichen and bryophyte communities);
- -
- Epiphytic lichen encrusted Schistidium antarctici (lichen encrusted bryophytes);
- -
- Bryum argenteum and Cyanobacteria (pure bryophytes);
- -
- Cyanobacteria—Bryum-Ceratodon (pure bryophytes).
2.2. Biological Conservation Protocol—Field Investigations
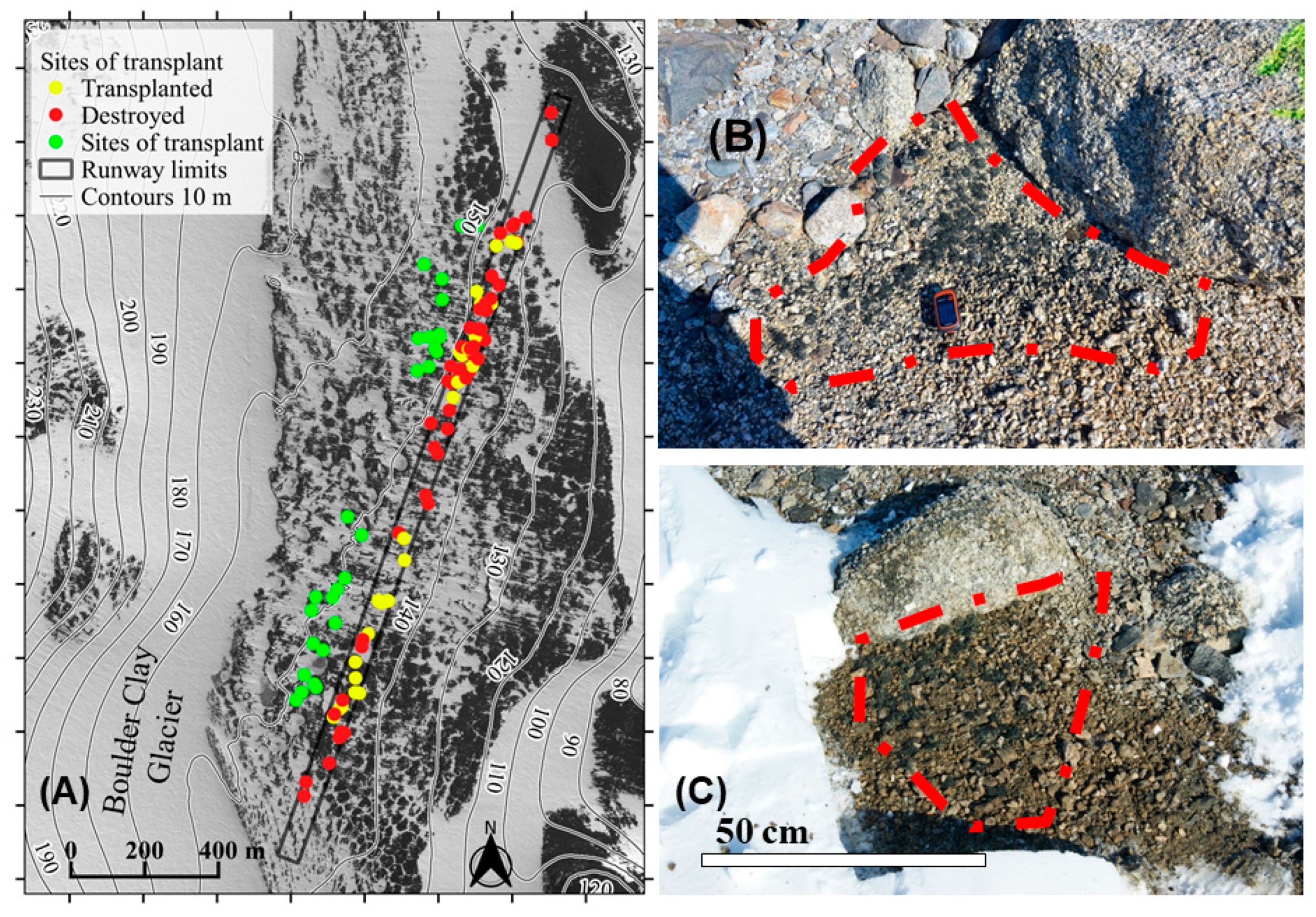
2.3. Pilot Project for Mitigation Actions—Conservation Planning, Transplant and Monitoring
- Areas representative of the vegetation occurring within the runway path;
- Areas with vegetation showing high coverage of the target/dominant species located in a limited area (in most cases ≤ 1 m2), with very healthy individuals;
- Areas with vegetation associations with the characteristics described above and representing community types rare or with limited distribution/occurrence both within the runway path as well as in the surrounding areas;
- Areas with vegetation characterized by the occurrence of rare species and/or with large and/or particularly healthy individuals.
3. Results
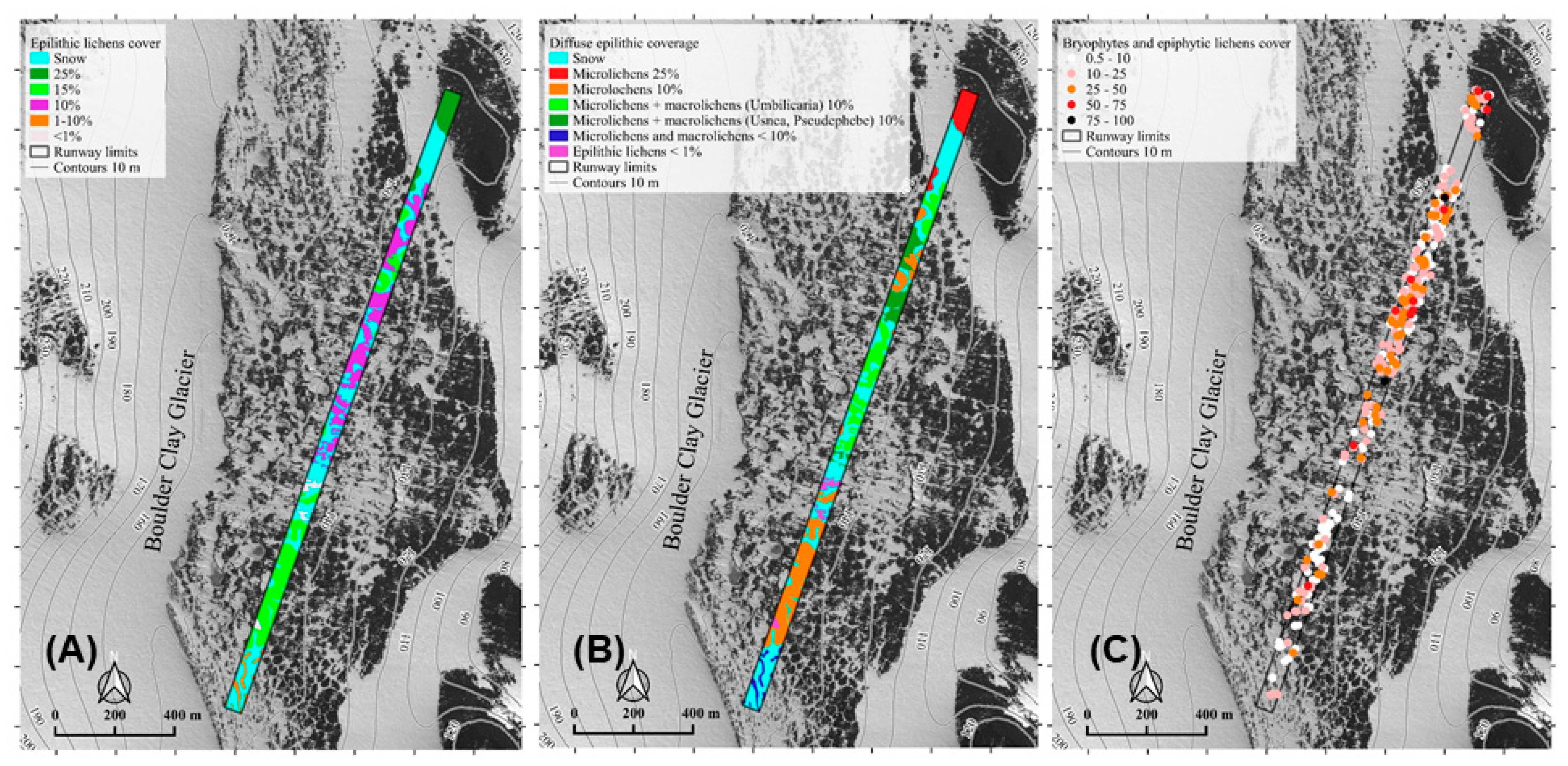
3.1. Flora and Vegetation of the Runway Pathway
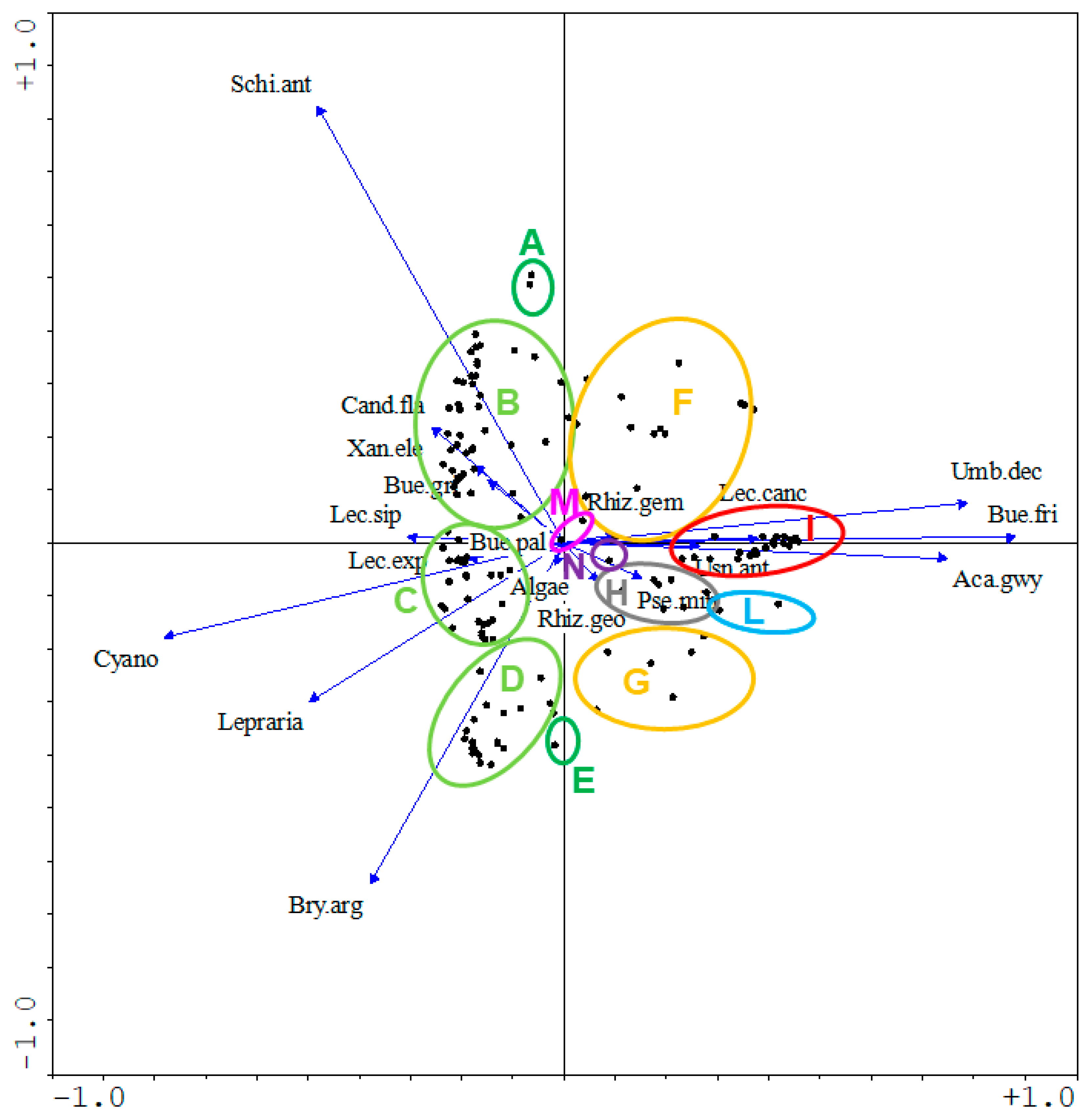
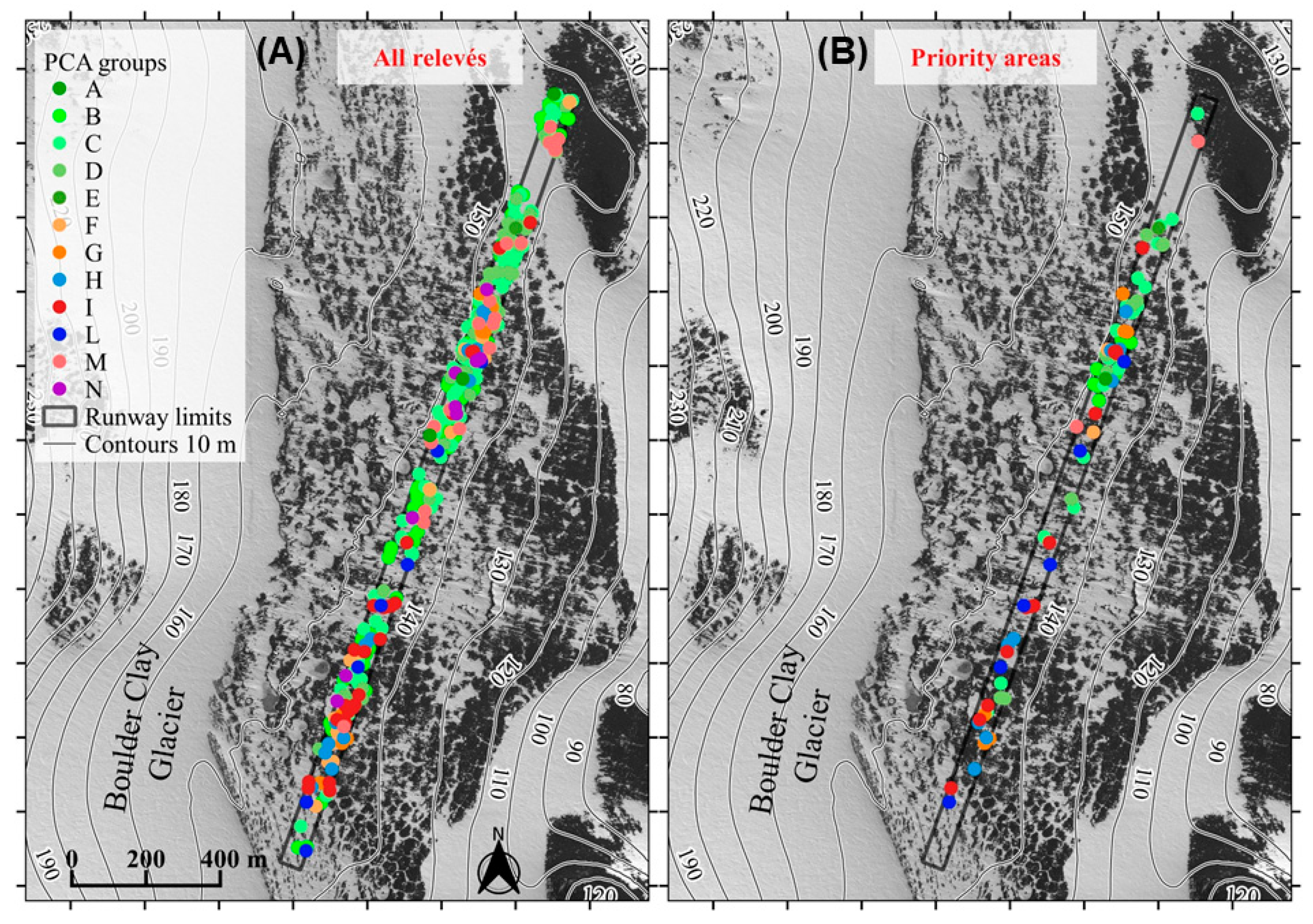
3.2. Active Conservation Planning and Realization through Transplantation
- -
- 13 patches dominated by Usnea antarctica–Umbilicaria decussata (PCA cluster I);
- -
- 6 patches dominated by Buellia frigida (PCA cluster L);
- -
- 9 patches dominated by Pseudephebe minuscula-Lecidella siplei-Bryophytes (PCA cluster H);
- -
- 2 patches dominated by Bryum argenteum and Cyanobacteria (PCA clusters A, E);
- -
- 2 patches with Cyanobacteria pure stands (PCA cluster M);
- -
- 9 patches dominated by the mosaic of Usnea-Umbilicaria and of Bryum-Cyanobacteria (PCA clusters F, G);
- -
- 39 patches dominated by the mosaic of lichen-encrusted bryophytes and of Bryum-Cyanobacteria (PCA clusters B, C, D).
4. Discussion
4.1. Impact of the Runway on the Flora and Vegetation of Boulder Clay
4.2. Pilot Project for the Active Biological Conservation through Vegetation Transplantation
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Green, T.G.A.; Sancho, L.G.; Pintado, A.; Schroeter, B. Functional and spatial pressures on terrestrial vegetation in Antarctica forced by global warming. Polar Biol. 2011, 34, 1643–1656. [Google Scholar] [CrossRef]
- Hughes, K.A.; Fretwell, P.; Rae, J.; Holmes, J.; Fleming, A.H. Untouched Antarctica: Mapping a finite and diminishing environmental re source. Antarct. Sci. 2011, 23, 537–548. [Google Scholar] [CrossRef]
- Terauds, A.; Chown, S.L.; Morgan, F.; Peat, H.J.; Watts, D.J.; Keys, H.; Convey, P.; Bergstrom, D.M. Conservation biogeography of the Antarctic. Divers. Distrib. 2012, 18, 726–741. [Google Scholar] [CrossRef]
- Chown, S.L.; Clarke, A.; Fraser, C.I.; Cary, S.C.; Moon, K.L.; McGeoch, M.A. The changing form of Antarctic biodiversity. Nature 2015, 522, 431–438. [Google Scholar] [CrossRef]
- Terauds, A.; Lee, J.R. Antarctic biogeography revisited: Updating the Antarctic Conservation Biogeographic Regions. Divers. Distrib. 2016, 22, 836–840. [Google Scholar] [CrossRef]
- ATCM. Protocol on Environmental Protection to the Antarctic Treaty; Antarctic Treaty Secretariat: Buenos Aires, Argentina, 1991. [Google Scholar]
- Brooks, S.T.; Jabour, J.; Hoff, J.V.D.; Bergstrom, D.M. Our footprint on Antarctica competes with nature for rare ice-free land. Nat. Sustain. 2019, 2, 185–190. [Google Scholar] [CrossRef]
- Castello, M.; Nimis, P.L. A key to the lichens of Terra Nova Bay (Victoria Land, Continental Antarctica). Ital. J. Zool. 2000, 67, 175–184. [Google Scholar] [CrossRef]
- Cannone, N.; Seppelt, R. A preliminary floristic classification of Northern and Southern Victoria Land vegetation (Continental Antarctica). Antarct. Sci. 2008, 20, 553–562. [Google Scholar] [CrossRef]
- Smykla, J.; Krzewicka, B.; Wilk, K.; Emslie, S.D.; Śliwa, L. Additions to the lichen flora of Victoria Land, Antarctica. Pol. Polar Res. 2011, 32, 123–138. [Google Scholar] [CrossRef]
- Castello, M. Lichens of the Terra Nova Bay area, northern Victoria Land. Studia Geobotanica 2003, 22, 3–59. [Google Scholar]
- Ochyra, R.; Smith, R.I.L.; Bednarek-Ochyra, H. The Illustrated Moss Flora of Antarctica; Cambridge University Press: Cambridge, UK, 2008; p. 685. [Google Scholar]
- Cannone, N.; Convey, P.; Guglielmin, M. Diversity trends of bryophyties in continental Antarctica. Polar Biol. 2013, 36, 259–271. [Google Scholar] [CrossRef]
- Brooks, S.T.; Jabour, J.; Bergstrom, D.M. What is ‘footprint’ in Antarctica: Proposing a set of definitions. Antarct. Sci. 2018, 30, 227–235. [Google Scholar] [CrossRef]
- Cannone, N. A network for monitoring terrestrial ecosystems along a latitudinal gradient in Continental Antarctica. Antarct. Sci. 2006, 18, 549–560. [Google Scholar] [CrossRef]
- Mauro, G.; Fratte, M.D.; Cannone, N. Permafrost warming and vegetation changes in continental Antarctica. Environ. Res. Lett. 2014, 9, 045001. [Google Scholar]
- ATCM. Protocol on Environmental Protection to the Antarctic Treaty; Antarctic Treaty Secretariat: Tromso, Norway, 1998. [Google Scholar]
- ATCM. ATCM XXVIII—CEP VIII. In Guidelines for Environmental Monitoring; Antarctic Treaty Secretariat: Stockholm, Sweden, 2005. [Google Scholar]
- ATCM. ATCM XXXIX—CEP XIX, Annex A. In Comprehensive Environmental Evaluation. Proposed Construction and Operation of a Gravel Runway in the Area of Mario Zucchelli Station, Terra Nova Bay, Victoria Land, Antarctica; Antarctic Treaty Secretariat: Santiago, Chile, 2016. [Google Scholar]
- SERI. The SER International Primer on Ecological Restoration; Society for Ecological Restoration International: Tucson, AZ, USA, 2004. [Google Scholar]
- Menz, M.H.; Dixon, K.W.; Hobbs, R.J. Hurdles and opportunities for landscape-scale restoration. Science 2013, 339, 526–527. [Google Scholar] [CrossRef]
- Perring, M.P.; Standish, R.J.; Price, J.N.; Craig, M.D.; Erickson, T.E.; Ruthrof, K.X.; Whiteley, A.S.; Valentine, L.E.; Hobbs, R.J. Advances in restoration ecology: Rising to the challenges of the coming decades. Ecosphere 2015, 6, 131. [Google Scholar] [CrossRef]
- Bruelheide, H.; Flintrop, T. Evaluating the transplantation of a meadow in the Harz Mountains, Germany. Biol. Conserv. 2000, 92, 109–120. [Google Scholar] [CrossRef]
- Kiehl, K.; Kirmer, A.; Donath, T.W.; Rasran, L.; Hölzel, N. Species introduction in restoration projects—Evaluation of different techniques for the establishment of semi-natural grasslands in Central and Northwestern Europe. Basic Appl. Ecol. 2010, 11, 285–299. [Google Scholar] [CrossRef]
- Mehlhoop, A.C.; Evju, M.; Hagen, D. Transplanting turfs to facilitate recovery in a low-alpine environment—What matters? Appl. Veg. Sci. 2018, 21, 615–625. [Google Scholar] [CrossRef]
- Aradottir, A.L. Turf transplants for restoration of alpine vegetation: Does size matter? J. Appl. Ecol. 2012, 49, 439–446. [Google Scholar] [CrossRef]
- Edwards, J.A.; Greene, D.M. The survival of Falkland Island transplants at South Georgia and Signy Island, South Orkney Islands. Br. Antarct. Surv. Bull. 1973, 33, 33–45. [Google Scholar]
- Edwards, J.A. An experimental introduction of vascular plants from South Georgia to the maritime Antarctic. Br. Antarct. Surv. Bull. 1980, 49, 73–80. [Google Scholar]
- Guglielmin, M.; Biasini, A.; Smiraglia, C. Buried ice landforms in the Northern Foothills (Northern Victoria Land, Antarctica). Some results from electrical soundings. Geographyska Annaler 1997, 79a, 17–24. [Google Scholar] [CrossRef]
- French, H.M.; Guglielmin, M. Observations on the ice-marginal, periglacial geomorphology of Terra Nova Bay, Northern Victoria Land, Antarctica. Permafr. Periglac. Process. 1999, 10, 331–347. [Google Scholar] [CrossRef]
- French, H.; Guglielmin, M. Frozen ground phenomena in the vicinity of Terra Nova Bay, Northern Victoria land, Antarctica: A preliminary report. Geographyska Annaler Phys. Geogr. 2000, 82, 513–526. [Google Scholar] [CrossRef]
- Cannone, N.; Wagner, D.; Hubberten, H.W.; Guglielmin, M. Biotic and abiotic factors influencing soil properties across a latitudinal gradient in Victoria Land, Antarctica. Geoderma 2008, 144, 50–65. [Google Scholar] [CrossRef]
- Rivas-Martínez, S.; Del Río, S.; Penas, Á.; Herrero, L.; Prieto, I.; Álvarez, M.; Díaz, T.E.; Molero, J.; Rivas-Sáenz, S.; Cantó, P.; et al. Biogeographical and bioclimatic outline of Antarctica. Plant Biosyst. 2021, 155, 5–15. [Google Scholar]
- Braun-Blanquet, J. Pflanzensoziologie; Springer: Vienna, Austria, 1964; p. 865. [Google Scholar]
- Cannone, N. Minimum area assessment and different sampling approaches for the study of vegetation communities in Antarctica. Antarct. Sci. 2004, 16, 157–164. [Google Scholar] [CrossRef]
- Zaccara, S.; Patiño, J.; Convey, P.; Vanetti, I.; Cannone, N. Multiple colonization and dispersal events hide the early origin and induce a lack of genetic structure of the moss Bryum argenteum in Antarctica. Ecol. Evol. 2020, 10, 1–17. [Google Scholar] [CrossRef]
- Guglielmin, M. Observations on permafrost ground thermal regimes from Antarctica and the Italian Alps, and their relevance to global climate change. Glob. Planet. Chang. 2004, 40, 159–167. [Google Scholar]
- Guglielmin, M. Ground surface temperature (GST), active layer, and permafrost monitoring in continental Antarctica. Permafr. Periglac. Process. 2006, 17, 133–143. [Google Scholar] [CrossRef]
- Nelson, F.E.; Shiklomanov, N.I.; Hinkel, K.M.; Brown, J. Decadal results from the Circumpolar Active Layer Monitoring (CALM) program. In Proceedings of the 9th International Conference on Permafrost, Fairbanks, AK, USA, 28 June–3 July 2008; Kane, D.L., Hinkel, K.M., Eds.; University of Alaska Press: Fairbanks, AK, USA, 2008; Volume 1, pp. 1273–1780. [Google Scholar]
- Cannone, N.; Convey, P.; Malfasi, M. Antarctic Specially Protected Areas (ASPA): A case study at Rothera Point providing tools and perspectives for the implementation of the ASPA network. Biodivers. Conserv. 2018, 27, 2641–2660. [Google Scholar] [CrossRef]
- Øvstedal, D.O.; Smith, R.I.L. Lichens of Antarctica and South Georgia. A Guide to Their Identification and Ecology; Cambridge University Press: Cambridge, UK, 2001. [Google Scholar]
- Seppelt, R.D.; Türk, R.; Green, A.T.G.; Moser, G.; Pannewitz, S.; Sancho, L.G.; Schroeter, B. Lichen and moss communities of Botany Bay, Granite Harbour, Ross Sea, Antarctica. Antarct. Sci. 2010, 22, 691–702. [Google Scholar] [CrossRef]
- Favero-Longo, S.E.; Cannone, N.; Worland, M.R.; Convey, P.; Piervittori, R.; Guglielmin, M. Changes in lichen diversity and community structure with fur seal population increase on Signy Island, South Orkney Islands. Antarct. Sci. 2010, 23, 65–77. [Google Scholar] [CrossRef]
- Favero-Longo, S.E.; Worland, M.R.; Convey, P.; Smith, R.I.L.; Piervittori, R.; Guglielmin, M.; Cannone, N. Primary succession of lichen and bryophyte communities following glacial recession on Signy Island, South Orkney Islands, Maritime Antarctic. Antarct. Sci. 2012, 24, 323–336. [Google Scholar] [CrossRef]
- Ter Braak, C.J.F.; Šmilauer, P. CANOCO Reference Manual and CanoDraw for Windows User’s Guide: Software for Canonical Community Ordination (ver. 4.5); Microcomputer Power Ed.: Ithaca, NY, USA, 2002. [Google Scholar]
- Kappen, L. Vegetation and ecology of icefree areas of northern Victoria Land, Antarctica. 1. The lichen vegetation of birthday ridge and an inland mountain. Polar Biol. 1985, 4, 213–225. [Google Scholar] [CrossRef]
- Seppelt, R.D.; Green, T.G.A.; Schroeter, B. Lichens and mosses from the Kar Plateau, southern Victoria Land, Antarctica. N. Z. J. Bot. 1995, 33, 203–220. [Google Scholar] [CrossRef]
- Seppelt, R.D.; Green, T.G.A.; Schroeter, B. Addictions and corrections to the lichen flora of the Kar Plateau, southern Victoria Land, Antarctica. N. Z. J. Bot. 1996, 34, 329–331. [Google Scholar] [CrossRef]
- Seppelt, R.D.; Green, T.G.A. A bryophyte flora for southern Victoria Land, Antarctica. N. Z. J. Bot. 1998, 36, 617–635. [Google Scholar] [CrossRef]
- Fernandez, J.A.; Carballeira, A. Differences in the responses of native and transplanted mosses to atmospheric pollution: A possible role of selenium. Environ. Pollut. 2000, 110, 73–78. [Google Scholar] [CrossRef]
- Fernandez, J.A.; Aboal, J.R.; Carballeira, A. Use of native and transplanted mosses as complementary techniques for biomonitoring mercury around an industrial facility. Sci. Total Environ. 2000, 256, 151–161. [Google Scholar] [CrossRef]
- Kosior, G.; Samecka-Cymerman, A.; Kolon, K.; Kempers, A. Bioindication capacity of metal pollution of native and transplanted Pleurozium schreberi under various levels of pollution. Chemosphere 2010, 81, 321–326. [Google Scholar] [CrossRef] [PubMed]
- Kawecki, T.J.; Ebert, D. Conceptual issues in local adaptation. Ecol. Lett. 2004, 7, 1225–1241. [Google Scholar] [CrossRef]
- Hereford, J. A quantitative survey of local adaptation and fitness trade-offs. Am. Nat. 2009, 173, 579–588. [Google Scholar] [CrossRef]
- Merinero, S.; Dahlberg, J.; Ehrl, J.; Hylander, K. Intraspecific variation influences performance of moss transplants along microclimate gradients. Ecology 2020, 2020, e02999. [Google Scholar] [CrossRef]
| Classification from Cannone and Seppelt [9] | 6 | Mosaic 4–6 | Mosaic 4–6 | Mosaic 4–6 | 6 | Mosaic 1–6 | Mosaic 1–6 | 3 | 1 | 2 | |||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| PCA Cluster | A | B | C | D | E | F | G | H | I | L | M | N | |
| Total Vegetation Coverage | 30.8 | 28.4 | 35.9 | 39.4 | 80 | 28.6 | 44.4 | 25.2 | 26.8 | 44.3 | 47.7 | 53.3 | |
| Algae | Algae | 0.001 | 0.6 | 0.012 | 0.3 | 17.7 | 51.7 | ||||||
| Epilithic Lichens | Buellia frigida | 0.19 | 0.4 | 5.1 | 7.1 | 12.5 | 10.9 | 10.4 | |||||
| Epilithic Lichens | Buellia pallida | 0.2 | |||||||||||
| Epilithic Lichens | Acarospora gwynnii | 0.02 | 0.007 | 0.02 | 0.3 | 1.8 | 2.2 | 4 | 1.9 | 0.003 | |||
| Epilithic Lichens | Caloplaca athallina | 0.1 | 0.07 | 0.02 | 0.3 | 0.1 | 0.008 | 0.04 | 1.8 | 0.02 | |||
| Epilithic Lichens | Caloplaca Lewis Smithii | 0.03 | |||||||||||
| Epilithic Lichens | Lecanora fuscobrunnea | 0.04 | 0.02 | ||||||||||
| Epilithic Lichens | Lecidea cancriformis | 0.01 | 0.03 | 0.08 | 0.006 | 0.2 | 0.6 | 0.1 | |||||
| Epilithic Lichens | Rhizocarpon geminatum | 0.06 | 2.9 | ||||||||||
| Epilithic Lichens | Rhizocarpon geographicum | 0.1 | 0.6 | 0.02 | 5.2 | ||||||||
| Epilithic Lichens | Xanthoria elegans | 0.17 | 0.01 | 0.07 | 0.002 | 0.3 | |||||||
| Epilithic Lichens | Pseudephebe minuscula | 0.07 | 0.2 | 0.03 | 1.2 | 7.8 | 2.4 | 0.2 | 2.2 | ||||
| Epilithic Lichens | Lecidella siplei | 1.3 | 1.9 | 0.9 | 1 | 0.01 | 1.4 | 0.3 | 0.3 | 3.2 | 0.7 | ||
| Epilithic Lichens | Umbilicaria decussata | 0.008 | 0.4 | 5.5 | 9.2 | 3.9 | 12.8 | 10.4 | |||||
| Epilithic Lichens | Usnea antarctica | 0.006 | 0.2 | 0.66 | 0.8 | 7 | 0.06 | 2.1 | |||||
| Cyanobacteria | Cyanobacteria | 13.9 | 15.5 | 24 | 6.8 | 18.6 | 3.4 | 1.4 | 10.4 | 31 | 2.1 | ||
| Mosses | Bryum argenteum | 8.6 | 17.6 | 80 | 0.5 | 7.8 | 0.04 | 0.09 | 0.6 | 0.3 | 0.8 | ||
| Mosses | Schistidium antarctici | 31.7 | 16.8 | 14 | 0.01 | 15.6 | 0.3 | 1.9 | 1.1 | ||||
| Epiphytic Lichens | Buellia grimmiae | 0.02 | 0.03 | 0.06 | |||||||||
| Epiphytic Lichens | Candelariella flava | 0.3 | 0.5 | 0.3 | 0.2 | 0.8 | 0.2 | 0.02 | 0.4 | 0.4 | |||
| Epiphytic Lichens | Lecanora expectans | 0.03 | |||||||||||
| Epiphytic Lichens | Lepraria cacuminum | 2.08 | 3.9 | 5.4 | 1.7 | 5.4 | 1.5 | 0.6 | 1.6 | 4.1 |
| Classification (Order According to [9]) | PCA Cluster | Priority Areas Transplanted [n] | Survival | ||
|---|---|---|---|---|---|
| (Formation) | 2017/18 | 2018/19 | [n] | [%] | |
| 1. Usnea–Umbilicaria | I | 3 | 3 | 6 | 100 |
| (Macrolichens) | |||||
| 2. Buellia frigida–Physcia caesia-Xanthoria spp. | L | 2 | 1 | 3 | 100 |
| (Macrolichens and microlichens) | |||||
| 3. Pseudephebe minuscula–Lecidella siplei–Bryophytes | H | 0 | 2 | 2 | 100 |
| (Mixed lichen and bryophyte communities) | |||||
| 1/6. Mosaic of Usnea–Umbilicaria with Bryum argenteum with Cyanobacteria | F; G | 1 | 1 | 2 | 100 |
| 4/6. Mosaic of epiphytic lichens encrusting Bryum argenteum and Schistidium antarctici with B. argenteum with Cyanobacteria | B; C; D | 11 | 4 | 12 | 80 |
| Total | 17 | 11 | 25 | 89.3 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cannone, N.; Ponti, S.; Malfasi, F. A Pilot Project to Limit the Human Impacts on the Fragile Antarctic Biota: Mitigation of a Runway through Vegetation Transplantation. Sustainability 2021, 13, 811. https://doi.org/10.3390/su13020811
Cannone N, Ponti S, Malfasi F. A Pilot Project to Limit the Human Impacts on the Fragile Antarctic Biota: Mitigation of a Runway through Vegetation Transplantation. Sustainability. 2021; 13(2):811. https://doi.org/10.3390/su13020811
Chicago/Turabian StyleCannone, Nicoletta, Stefano Ponti, and Francesco Malfasi. 2021. "A Pilot Project to Limit the Human Impacts on the Fragile Antarctic Biota: Mitigation of a Runway through Vegetation Transplantation" Sustainability 13, no. 2: 811. https://doi.org/10.3390/su13020811
APA StyleCannone, N., Ponti, S., & Malfasi, F. (2021). A Pilot Project to Limit the Human Impacts on the Fragile Antarctic Biota: Mitigation of a Runway through Vegetation Transplantation. Sustainability, 13(2), 811. https://doi.org/10.3390/su13020811





