Sustainability in ElectroKinetic Remediation Processes: A Critical Analysis
Abstract
1. Introduction
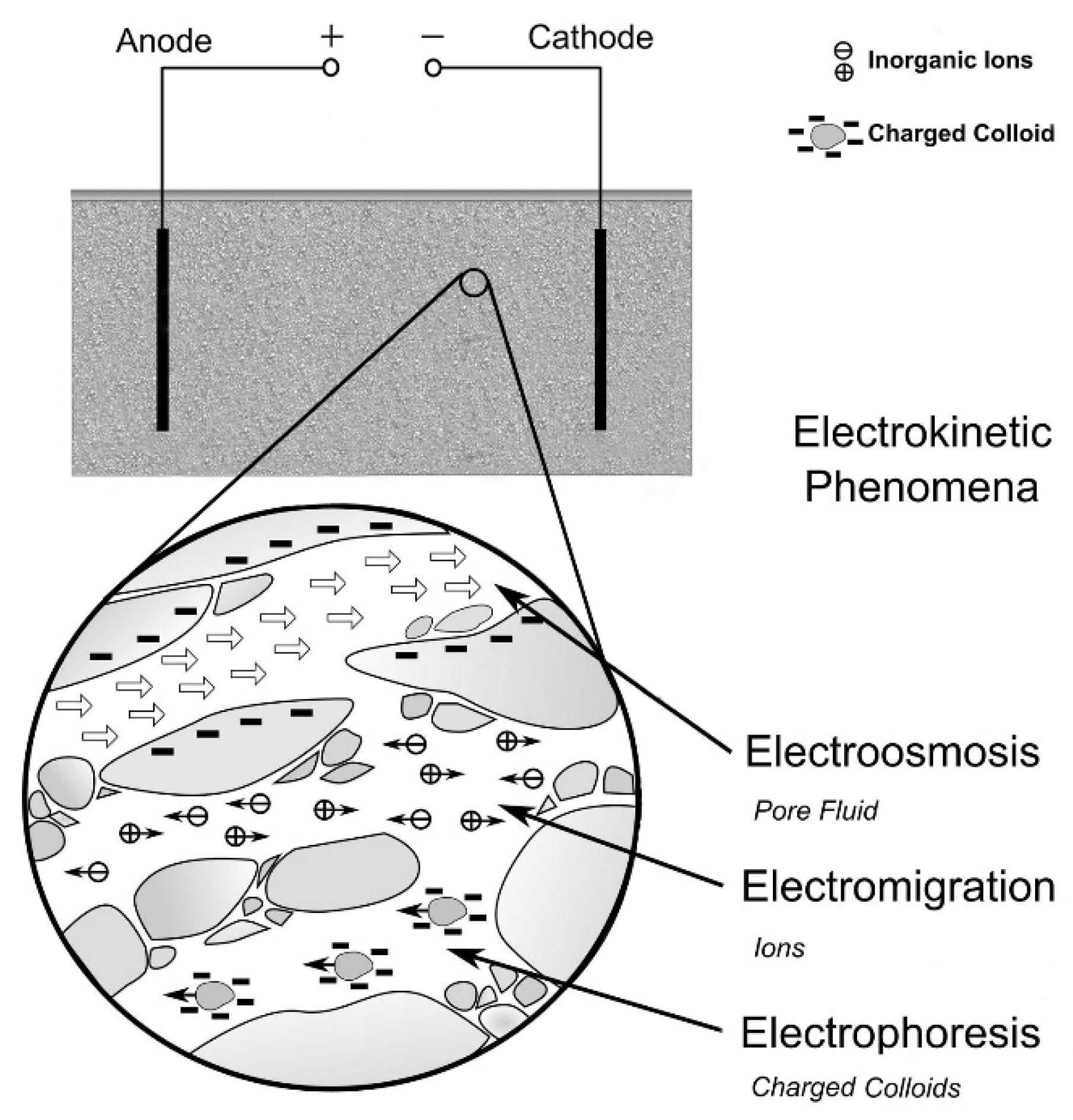
2. Fundamentals of Electrokinetic Remediation
- electroosmosis, i.e., the displacement of the solution naturally present in the soil;
- (electro)migration of electrically charged species;
- electrolysis, a process that occurs on the surfaces of electrodes, generally at the expense of water (decomposition reactions);
- electrophoresis, that is the transport of charged particles of colloidal size present within a stationary fluid, due to the application of an electric gradient.
3. Evolution of the Technology and Its Applications
- the optimization of the pH of electrolytic solutions [37];
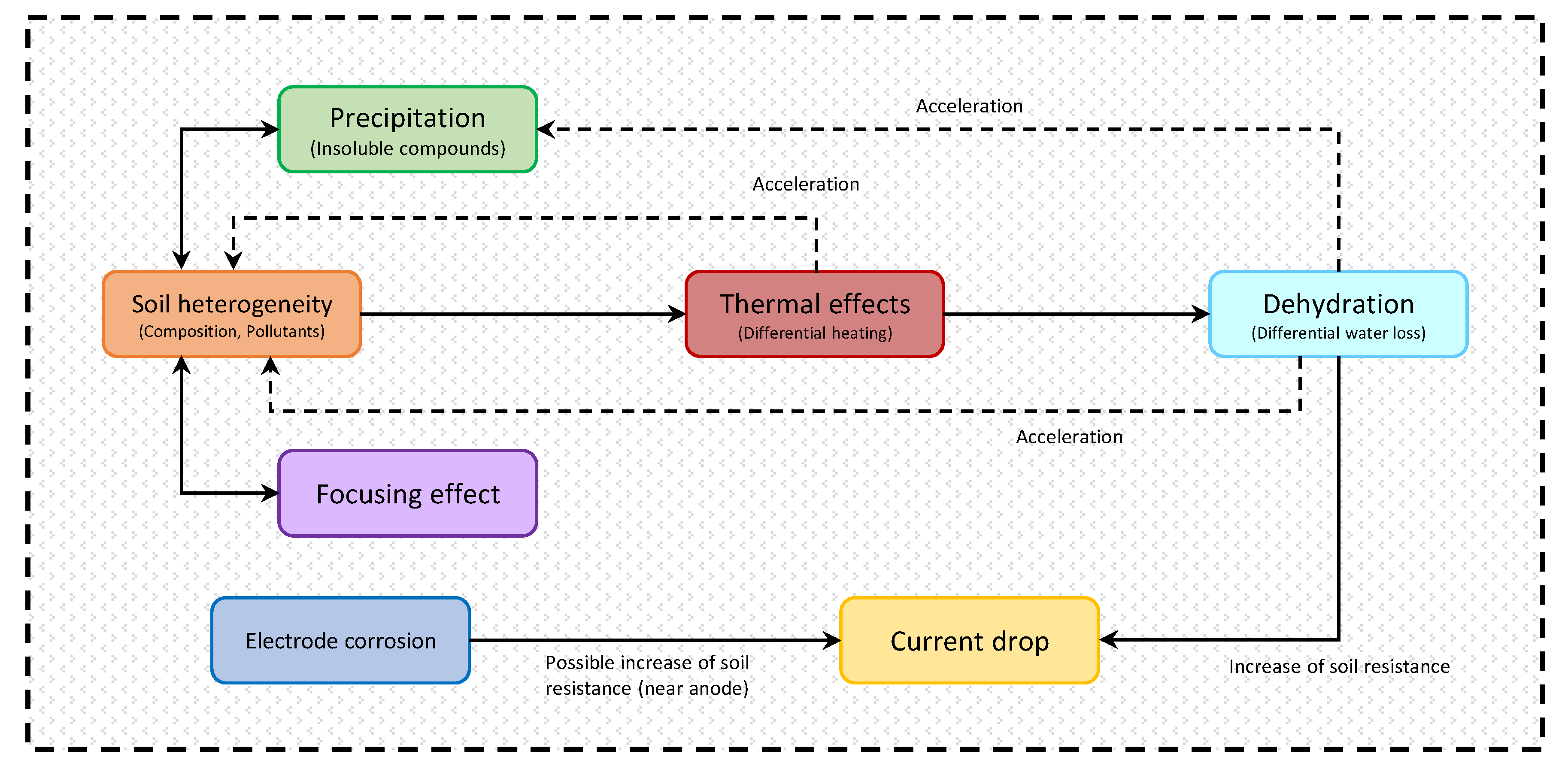
4. Side Effects during EKR
4.1. Thermal Effects
4.2. Precipitation of Insoluble Compounds
4.3. Electrode Corrosion
4.4. Focusing Effect
4.5. Dehydration
5. Energy-Related Aspects
5.1. Energy Consumption
5.2. Energy Saving
5.3. Use of Renewable Energies
5.3.1. Photovoltaic Solar Cells and Wind Turbines
5.3.2. Costs and Social Impacts of Solar and Wind Energy
5.3.3. Self-Powered Technologies
6. Conclusions
Author Contributions
Funding
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Farhadian, M.; Vachelard, C.; Duchez, D.; Larroche, C. In situ bioremediation of monoaromatic pollutants in groundwater: A review. Biores. Technol. 2008, 99, 5296–5308. [Google Scholar] [CrossRef] [PubMed]
- Varjani, S.J. Microbial degradation of petroleum hydrocarbons. Biores. Technol. 2017, 223, 277–286. [Google Scholar] [CrossRef] [PubMed]
- Vocciante, M.; Finocchi, A.; De Folly D’Auris, A.; Conte, A.; Tonziello, J.; Pola, A.; Reverberi, A. Enhanced oil spill remediation by adsorption with interlinked multilayered graphene. Materials 2019, 12, 2231. [Google Scholar] [CrossRef] [PubMed]
- Pietrelli, L.; Francolini, I.; Piozzi, A.; Sighicelli, M.; Vocciante, M. Chromium (III) removal from wastewater by chitosan flakes. Appl. Sci. 2020, 10, 1925. [Google Scholar] [CrossRef]
- Velimirovic, M.; Bianco, C.; Ferrantello, N.; Tosco, T.; Casasso, A.; Sethi, R.; Schmid, D.; Wagner, S.; Miyajima, K.; Klaas, N.; et al. A Large-Scale 3D Study on Transport of Humic Acid-Coated Goethite Nanoparticles for Aquifer Remediation. Water 2020, 12, 1207. [Google Scholar] [CrossRef]
- Khan, F.I.; Husain, T.; Hejazi, R. An overview and analysis of site remediation technologies. J. Environ. Manag. 2004, 71, 95–122. [Google Scholar] [CrossRef]
- Pavel, L.V.; Gavrilescu, M. Overview of ex situ decontamination techniques for soil cleanup. Environ. Eng. Manag. J. 2008, 7, 815–834. [Google Scholar] [CrossRef]
- Chowdhury, A.I.; Gerhard, J.I.; Reynolds, D.; Sleep, B.E.; O’Carroll, D.M. Electrokinetic-enhanced permanganate delivery and remediation of contaminated low permeability porous media. Water Res. 2017, 113, 215–222. [Google Scholar] [CrossRef]
- Franchi, E.; Cosmina, P.; Pedron, F.; Rosellini, I.; Barbafieri, M.; Petruzzelli, G.; Vocciante, M. Improved Arsenic phytoextraction by combined use of mobilizing chemicals and autochtonous soil bacteria. Sci. Total Environ. 2019, 655, 328–336. [Google Scholar] [CrossRef]
- Vocciante, M.; Caretta, A.; Bua, L.; Bagatin, R.; Franchi, E.; Petruzzelli, G.; Ferro, S. Enhancements in phytoremediation technology: Environmental assessment of different biomass disposal solutions in comparison with a consolidated approach. J. Environ. Manag. 2019, 237, 560–568. [Google Scholar] [CrossRef]
- Vocciante, M.; Reverberi, A.P.; Pietrelli, L.; Dovì, V.G. Improved remediation processes through cost-effective estimation of soil properties from surface measurements. J. Clean. Prod. 2017, 167, 680–686. [Google Scholar] [CrossRef]
- da Silva, B.M.; Maranho, L.T. Petroleum-contaminated sites: Decision framework for selecting remediation technologies. J. Hazard. Mater. 2019, 378, 120722. [Google Scholar] [CrossRef] [PubMed]
- Rosestolato, D.; Bagatin, R.; Ferro, S. Electrokinetic remediation of soils polluted by heavy metals (mercury in particular). Chem. Eng. J. 2015, 264, 16–23. [Google Scholar] [CrossRef]
- Ferrucci, A.; Vocciante, M.; Bagatin, R.; Ferro, S. Electrokinetic remediation of soils contaminated by potentially toxic metals: Dedicated analytical tools for assessing the contamination baseline in a complex scenario. J. Environ. Manag. 2017, 203, 1163–1168. [Google Scholar] [CrossRef] [PubMed]
- Vocciante, M.; Caretta, A.; Bua, L.; Bagatin, R.; Ferro, S. Enhancements in ElectroKinetic Remediation Technology: Environmental assessment in comparison with other configurations and consolidated solutions. Chem. Eng. J. 2016, 289, 123–134. [Google Scholar] [CrossRef]
- Millán, M.; Bucio-Rodríguez, P.Y.; Lobato, J.; Fernández-Marchante, C.M.; Roa-Morales, G.; Barrera-Díaz, C.; Rodrigo, M.A. Strategies for powering electrokinetic soil remediation: A way to optimize performance of the environmental technology. J. Environ. Manag. 2020, 267, 110665. [Google Scholar] [CrossRef]
- Reddy, K.R.; Cameselle, C. Electrochemical Remediation Technologies for Polluted Soils, Sediments and Groundwater; John Wiley & Sons: Hoboken, NJ, USA, 2009. [Google Scholar]
- Yeung, A.T.; Gu, Y.Y. A review on techniques to enhance electrochemical remediation of contaminated soils. J. Hazard. Mater. 2011, 195, 11–29. [Google Scholar] [CrossRef]
- Ganiyu, S.O.; Brito, L.R.D.; de Araújo Costa, E.C.T.; dos Santos, E.V.; Martínez-Huitle, C.A. Solar photovoltaic-battery system as a green energy for driven electrochemical wastewater treatment technologies: Application to elimination of Brilliant Blue FCF dye solution. J. Environ. Chem. Eng. 2019, 7, 102924. [Google Scholar] [CrossRef]
- Ganiyu, S.O.; Martínez-Huitle, C.A.; Rodrigo, M.A. Renewable energies driven electrochemical wastewater/soil decontamination technologies: A critical review of fundamental concepts and applications. Appl. Catal. B Environ. 2020, 270, 118857. [Google Scholar] [CrossRef]
- Virkutyte, J.; Sillanpaa, M.; Latostenmaa, P. Electrokinetic soil remediation—Critical overview. Sci. Total Environ. 2002, 289, 97–121. [Google Scholar] [CrossRef]
- Reuss, F.F. Notice sur un nouvel effet de l’électricité galvanique. Mem. Soc. Imp. Nat. Moscou 1809, 2, 327–337. [Google Scholar]
- Acar, Y.B.; Alshawabkeh, A.N. Principles of electrokinetic remediation. Environ. Sci. Technol. 1993, 27, 2638–2647. [Google Scholar] [CrossRef]
- Risco, C.; Rodrigo, S.; López-Vizcaíno, R.; Yustres, A.; Sáez, C.; Cañizares, P.; Navarro, V.; Rodrigo, M.A. Electrochemically assisted fences for the electroremediation of soils polluted with 2,4-D: A case study in a pilot plant. Sep. Purif. Technol. 2015, 156, 234–241. [Google Scholar] [CrossRef]
- Schnarr, M.; Truax, C.; Farquhar, G.; Hood, E.; Gonullu, T.; Stickney, B. Laboratory and controlled field experiments using potassium permanganate to remediate trichloroethylene and perchloroethylene DNAPLs in porous media. J. Contam. Hydrol. 1998, 29, 205–224. [Google Scholar] [CrossRef]
- Reddy, K.R.; Saichek, R.E. Effect of soil type on electrokinetic removal of phenanthrene using surfactants and cosolvents. J. Environ. Eng. 2003, 129, 336–346. [Google Scholar] [CrossRef]
- Reddy, K.R.; Danda, S.; Saichek, R.E. Complicating factors of using ethylenediamine tetraacetic acid to enhance electrokinetic remediation of multiple heavy metals in clayey soils. J. Environ. Eng. 2004, 130, 1357–1366. [Google Scholar] [CrossRef]
- McBratney, A.; Field, D.J.; Koch, A. The dimensions of soil security. Geoderma 2014, 213, 203–213. [Google Scholar] [CrossRef]
- Vocciante, M.; Bagatin, R.; Ferro, S. Enhancements in electrokinetic remediation technology: Focus on water management and wastewater recovery. Chem. Eng. J. 2017, 309, 708–716. [Google Scholar] [CrossRef]
- Acar, Y.B.; Gale, R.J.; Alshawabkeh, A.N.; Marks, R.E.; Puppala, S.; Bricka, M.; Parker, R. Electrokinetic remediation: Basics and technology status. J. Hazard. Mater. 1995, 40, 117–137. [Google Scholar] [CrossRef]
- Gill, R.T.; Harbottle, M.J.; Smith, J.W.N.; Thornton, S.F. Electrokinetic-enhanced bioremediation of organic contaminants: A review of processes and environmental applications. Chemosphere 2014, 107, 31–42. [Google Scholar] [CrossRef]
- Rocha, I.M.V.; Silva, K.N.O.; Silva, D.R.; Martínez-Huitle, C.A.; dos Santos, E.V. Coupling electrokinetic remediation with phytoremediation for depolluting soil with petroleum and the use of electrochemical technologies for treating the effluent generated. Sep. Purif. Technol. 2019, 208, 194–200. [Google Scholar] [CrossRef]
- Porrett, R., Jr. Curious galvanic experiments. Ann. Philos. 1816, 8, 74–76. [Google Scholar]
- Casagrande, L. Electro-osmosis in soils. Géotechnique 1949, 1, 159–177. [Google Scholar] [CrossRef]
- Hamnett, R. A study of the Processes Involved in the Electro-Reclamation of Contaminated Soils. Master’s Thesis, University of Manchester, Manchester, UK, 1980. [Google Scholar]
- Segall, B.A.; Matthias, J.A.; O’Bannon, C.E. Electro-osmosis chemistry and water quality. J. Geotech. Eng. Div. 1980, 106, 1148–1152. [Google Scholar]
- Puppala, S.K.; Alshawabkeh, A.N.; Acar, Y.B.; Gale, R.J.; Bricka, M. Enhanced electrokinetic remediation of high sorption capacity soil. J. Hazard. Mater. 1997, 55, 203–220. [Google Scholar] [CrossRef]
- Ottosen, L.M.; Hansen, H.K.; Laursen, S.; Villumsen, A. Electrodialytic remediation of soil polluted with copper from wood preservation industry. Environ. Sci. Technol. 1997, 31, 1711–1715. [Google Scholar] [CrossRef]
- Li, Z.; Yu, J.W.; Neretnieks, I. Electroremediation: Removal of heavy metals from soils by using cation selective membrane. Environ. Sci. Technol. 1998, 32, 394–397. [Google Scholar] [CrossRef]
- Wong, J.S.; Hicks, R.E.; Probstein, R.F. EDTA-enhanced electroremediation of metal-contaminated soils. J. Hazard. Mater. 1997, 55, 61–79. [Google Scholar] [CrossRef]
- Ko, S.O.; Schlautman, M.A.; Carraway, E.R. Cyclodextrin-enhanced electrokinetic removal of phenanthrene from a model clay soil. Environ. Sci. Technol. 2000, 34, 1535–1541. [Google Scholar] [CrossRef]
- Alshawabkeh, A.N.; Yeung, A.T.; Bricka, M.R. Practical aspects of in-situ electrokinetic extraction. J. Environ. Eng. 1999, 125, 27–35. [Google Scholar] [CrossRef]
- Alshawabkeh, A.N.; Gale, R.J.; Ozsu-Acar, E.; Bricka, M.R. Optimization of 2-D electrode configuration for electrokinetic remediation. J. Soil Contam. 1999, 8, 617–635. [Google Scholar] [CrossRef]
- Kim, K.-J.; Kim, D.-H.; Yoo, J.-C.; Baek, K. Electrokinetic extraction of heavy metals from dredged marine sediment. Sep. Purif. Technol. 2011, 79, 164–169. [Google Scholar] [CrossRef]
- Rozas, F.; Castellote, M. Electrokinetic remediation of dredged sediments polluted with heavy metals with different enhancing electrolytes. Electrochim. Acta 2012, 86, 102–109. [Google Scholar] [CrossRef]
- Iannelli, R.; Masi, M.; Ceccarini, A.; Ostuni, M.B.; Lageman, R.; Muntoni, A.; Pomi, R. Electrokinetic remediation of metal-polluted marine sediments: Experimental investigation for plant design. Electrochim. Acta 2015, 181, 146–159. [Google Scholar] [CrossRef]
- Moussa, D.T.; El-Naas, M.H.; Nasser, M.; Al-Marri, M.J. A comprehensive review of electrocoagulation for water treatment: Potentials and challenges. J. Environ. Manag. 2017, 186, 24–41. [Google Scholar] [CrossRef]
- Mukimin, A.; Zen, N.; Purwanto, A.; Wicaksono, K.A.; Vistanty, H.; Alfauzi, A.S. Application of a full-scale electrocatalytic reactor as real batik printing wastewater treatment by indirect oxidation process. J. Environ. Chem. Eng. 2017, 5, 5222–5232. [Google Scholar] [CrossRef]
- Munoz-Morales, M.; Sáez, C.; Cañizares, P.; Rodrigo, M.A. A new strategy for the electrolytic removal of organics based on adsorption onto granular activated carbon. Electrochem. Commun. 2018, 90, 47–50. [Google Scholar] [CrossRef]
- Warner, S.D.; Ferro, S.; De Battisti, A. Barriere permeabili reattive—Considerazioni generali e stato dell’arte. La Chimica e l’Industria (Milan Italy) 2006, 88, 44–46. [Google Scholar]
- Araújo, R.; Castro, A.C.M.; Baptista, J.S.; Fiúza, A. Nanosized iron based permeable reactive barriers for nitrate removal–Systematic review. Phys. Chem. Earth 2016, 94, 29–34. [Google Scholar] [CrossRef]
- Naje, A.S.; Chelliapan, S.; Zakaria, Z.; Ajeel, M.A.; Alaba, P.A. A review of electrocoagulation technology for the treatment of textile wastewater. Rev. Chem. Eng. 2017, 33, 263–292. [Google Scholar] [CrossRef]
- Nidheesh, P.V.; Zhou, M.; Oturan, M.A. An overview on the removal of synthetic dyes from water by electrochemical advanced oxidation processes. Chemosphere 2018, 197, 210–227. [Google Scholar] [CrossRef] [PubMed]
- Cappello, S.; Cruz Viggi, C.; Yakimov, M.; Rossetti, S.; Matturro, B.; Molina, L.; Segura, A.; Marqués, S.; Yuste, L.; Sevilla, E.; et al. Combining electrokinetic transport and bioremediation for enhanced removal of crude oil from contaminated marine sediments: Results of a long-term, mesocosm-scale experiment. Water Res. 2019, 157, 381–395. [Google Scholar] [CrossRef] [PubMed]
- Aulenta, F.; Canosa, A.; Reale, P.; Rossetti, S.; Panero, S.; Majone, M. Microbial reductive dechlorination of trichloroethene to ethene with electrodes serving as electron donors without the external addition of redox mediators. Biotechnol. Bioeng. 2009, 103, 85–91. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Luo, H.; Fallgren, P.H.; Jin, S.; Ren, Z.J. Bioelectrochemical system platform for sustainable environmental remediation and energy generation. Biotechnol. Adv. 2015, 33, 317–334. [Google Scholar] [CrossRef] [PubMed]
- Domínguez-Garay, A.; Quejigo, J.R.; Dörfler, U.; Schroll, R.; Esteve-Núñez, A. Bioelectroventing: An electrochemical-assisted bioremediation strategy for cleaning-up atrazine-polluted soils. Microb. Biotechnol. 2018, 11, 50–62. [Google Scholar] [CrossRef] [PubMed]
- Yan, F.; Chen, W.; Reible, D. Electrochemical stimulation of PAH biodegradation in sediment. Soil Sediment Contam. 2015, 24, 143–156. [Google Scholar] [CrossRef]
- Barba, S.; López-Vizcaíno, R.; Saez, C.; Villaseñor, J.; Cañizares, P.; Navarro, V.; Rodrigo, M.A. Electro-bioremediation at the prototype scale: What it should be learned for the scale-up. Chem. Eng. J. 2018, 334, 2030–2038. [Google Scholar] [CrossRef]
- Kim, S.H.; Han, H.Y.; Lee, Y.J.; Kim, C.W.; Yang, J.W. Effect of electrokinetic remediation on indigenous microbial activity and community within diesel contaminated soil. Sci. Total Environ. 2010, 408, 3162–3168. [Google Scholar] [CrossRef]
- Velasco-Alvarez, N.; González, I.; Damian-Matsumura, P.; Gutiérrez-Rojas, M. Enhanced hexadecane degradation and low biomass production by Aspergillus niger exposed to an electric current in a model system. Biores. Technol. 2011, 102, 1509–1515. [Google Scholar] [CrossRef]
- Aćimović, D.D.; Karić, S.D.; Nikolić, Ž.M.; Brdarić, T.P.; Tasić, G.S.; Kaninski, M.P.M.; Nikolić, V.M. Electrochemical oxidation of the polycyclic aromatic hydrocarbons in polluted concrete of the residential buildings. Environ. Pollut. 2017, 220, 393–399. [Google Scholar] [CrossRef]
- Huang, H.; Tang, J.; Niu, Z.; Giesy, J.P. Interactions between electrokinetics and rhizoremediation on the remediation of crude oil-contaminated soil. Chemosphere 2019, 229, 418–425. [Google Scholar] [CrossRef] [PubMed]
- Bi, R.; Schlaak, M.; Siefert, E.; Lord, R.; Connolly, H. Influence of electrical fields (AC and DC) on phytoremediation of metal polluted soils with rapeseed (Brassica napus) and tobacco (Nicotiana tabacum). Chemosphere 2011, 83, 318–326. [Google Scholar] [CrossRef] [PubMed]
- Denvir, A.; Hodko, D.; Van Hyfte, J.; Magnuson, J.W. Methods for Enhancing Phytoextraction of Contaminants from Porous Media Using Electrokinetic Phenomena. U.S. Patent No. 6,145,244, 14 November 2000. [Google Scholar]
- Aboughalma, H.; Bi, R.; Schlaak, M. Electrokinetic enhancement on phytoremediation in Zn, Pb, Cu and Cd contaminated soil using potato plants. J. Environ. Sci. Health A 2008, 43, 926–933. [Google Scholar] [CrossRef]
- Cameselle, C.; Gouveia, S. Electrokinetic remediation for the removal of organic contaminants in soils. Curr. Opin. Electrochem. 2018, 11, 41–47. [Google Scholar] [CrossRef]
- O’Connor, C.S.; Lepp, N.W.; Edwards, R.; Sunderland, G. The combined use of electrokinetic remediation and phytoremediation to decontaminate metal-polluted soils: A laboratory-scale feasibility study. Environ. Monit. Assess. 2003, 84, 141–158. [Google Scholar] [CrossRef]
- Reverberi, A.P.; Vocciante, M.; Salerno, M.; Ferretti, M.; Fabiano, B. Green synthesis of silver nanoparticles by low-energy wet bead milling of metal spheres. Materials 2020, 13, 63. [Google Scholar] [CrossRef]
- Trofa, M.; D’Avino, G.; Fabiano, B.; Vocciante, M. Nanoparticles synthesis in wet-operating stirred media: Investigation on the grinding efficiency. Materials 2020, 13, 4281. [Google Scholar] [CrossRef]
- Hosseini, A.; Haeri, S.M.; Mahvelati, S.; Fathi, A. Feasibility of using electrokinetics and nanomaterials to stabilize and improve collapsible soils. J. Rock Mech. Geotech. 2020, 11, 1055–1065. [Google Scholar] [CrossRef]
- Czinnerová, M.; Vološčuková, O.; Marková, K.; Ševců, A.; Černík, M.; Nosek, J. Combining nanoscale zero-valent iron with electrokinetic treatment for remediation of chlorinated ethenes and promoting biodegradation: A long-term field study. Water Res. 2020, 175, 115692. [Google Scholar] [CrossRef]
- Saleem, M.; Chakrabarti, M.H.; Irfan, M.F.; Hajimolana, S.A.; Hussain, M.A.; Diya’uddeen, B.H.; Daud, W.M.A.W. Electrokinetic remediation of nickel from low permeability soil. Int. J. Electrochem. Sci. 2011, 96, 4264–4275. [Google Scholar]
- Wu, C.D.; Fan, C.P.; Xie, Q.J. Study on electrokinetic remediation of PBDEs contaminated soil. Adv. Mater. Res. 2012, 518–523, 2829–2833. [Google Scholar] [CrossRef]
- Fu, R.; Wen, D.; Xia, X.; Zhang, W.; Gu, Y. Electrokinetic remediation of chromium (Cr)-contaminated soil with citric acid (CA) and polyaspartic acid (PASP) as electrolytes. Chem. Eng. J. 2017, 316, 601–608. [Google Scholar] [CrossRef]
- Choi, J.H.; Lee, Y.J.; Lee, H.G.; Ha, T.H.; Bae, J.H. Removal characteristics of salts of greenhouse in field test by in situ electrokinetic process. Electrochim. Acta 2012, 86, 63–71. [Google Scholar] [CrossRef]
- Wen, D.; Fu, R.; Li, Q. Removal of inorganic contaminants in soil by electrokinetic remediation technologies: A review. J. Hazard. Mater. 2020, 401, 123345. [Google Scholar] [CrossRef] [PubMed]
- Wen, D.D.; Fu, R.B.; Zhang, W.; Gu, Y.Y. Enhanced electrokinetic remediation of heavy metals contaminated soils by stainless steel electrodes as well as the phenomenon and mechanism of electrode corrosion and crystallization. Huanjing Kexue 2017, 38, 1209–1217. [Google Scholar] [PubMed]
- Méndez, E.; Pérez, M.; Romero, O.; Beltrán, E.D.; Castro, S.; Corona, J.L.; Corona, A.; Cuevas, M.C.; Bustos, E. Effects of electrode material on the efficiency of hydrocarbon removal by an electrokinetic remediation process. Electrochim. Acta 2012, 86, 148–156. [Google Scholar] [CrossRef]
- Xu, L.; Dai, H.; Skuza, L.; Wei, S. The effects of different electric fields and electrodes on solanum nigrum L. Cd hyperaccumulation in soil. Chemosphere 2020, 246, 125666. [Google Scholar] [CrossRef] [PubMed]
- Hu, J.; Zhu, H.; Ma, Y.; Yi, T.; Mao, X.; Lin, A.; Gan, F. Corrosion protection of stainless steel by separate polypyrrole electrode in acid solutions. Mater. Corros. 2011, 62, 68–73. [Google Scholar] [CrossRef]
- Kim, D.-H.; Jo, S.-U.; Yoo, J.-C.; Baek, K. Ex situ pilot scale electrokinetic restoration of saline soil using pulsed current. Sep. Purif. Technol. 2013, 120, 282–288. [Google Scholar] [CrossRef]
- Ping, L.; Qiyan, F.; Qingjun, M.; Tao, Y. Electrokinetic remediation of chromium and cadmium-contaminated soil from abandoned industrial site. Sep. Purif. Technol. 2012, 98, 216–220. [Google Scholar]
- Cai, Z.-P.; Van Doren, J.; Fang, Z.-Q.; Li, W.-S. Improvement in electrokinetic remediation of Pb-contaminated soil near lead acid battery factory. Trans. Nonferrous Met. Soc. China 2015, 25, 3088–3095. [Google Scholar] [CrossRef]
- Cheng, F.; Guo, S.; Li, G.; Wang, S.; Li, F.; Wu, B. The loss of mobile ions and the aggregation of soil colloid: Results of the electrokinetic effect and the cause of process termination. Electrochim. Acta 2017, 258, 1016–1024. [Google Scholar] [CrossRef]
- Zhou, D.M.; Deng, C.F.; Cang, L. Electrokinetic remediation of a Cu contaminated red soil by conditioning catholyte pH with different enhancing chemical reagents. Chemosphere 2004, 56, 265–273. [Google Scholar] [CrossRef] [PubMed]
- Pazos, M.; Sanroman, M.A.; Cameselle, C. Improvement in electrokinetic remediation of heavy metal spiked kaolin with the polarity exchange technique. Chemosphere 2006, 62, 817–822. [Google Scholar] [CrossRef] [PubMed]
- Ottosen, L.M.; Kristensen, I.V.; Pedersen, A.J.; Hansen, H.K.; Villumsen, A.; Ribeiro, A.B. Electrodialytic removal of heavy metals from different solid waste products. Sep. Sci. Technol. 2003, 38, 1269–1289. [Google Scholar] [CrossRef]
- Sun, Z.; Wu, B.; Guo, P.; Wang, S.; Guo, S. Enhanced electrokinetic remediation and simulation of cadmium-contaminated soil by superimposed electric field. Chemosphere 2019, 233, 17–24. [Google Scholar] [CrossRef] [PubMed]
- Li, G.; Guo, S.; Li, S.; Zhang, L.; Wang, S. Comparison of approaching and fixed anodes for avoiding the ‘focusing’ effect during electrokinetic remediation of chromium-contaminated soil. Chem. Eng. J. 2012, 203, 231–238. [Google Scholar] [CrossRef]
- Amrate, S.; Akretche, D.E.; Innocent, C.; Seta, P. Removal of Pb from a calcareous soil during EDTA-enhanced electrokinetic extraction. Sci. Total Environ. 2005, 349, 56–66. [Google Scholar] [CrossRef]
- Shin, S.Y.; Park, S.M.; Baek, K. Soil moisture could enhance electrokinetic remediation of arsenic-contaminated soil. Environ. Sci. Pollut. Res. 2017, 24, 9820–9825. [Google Scholar] [CrossRef]
- Mohamed Johar, S.; Embong, Z. The optimization of electrokinetic remediation for heavy metals and radioactivity contamination on holyrood-lunas soil (acrisol species) in Sri gading industrial Area, Batu pahat, Johor, Malaysia. Radiat. Prot. Dosimetry 2015, 167, 160–164. [Google Scholar] [CrossRef]
- Jo, S.U.; Kim, D.H.; Yang, J.S.; Baek, K. Pulse-enhanced electrokinetic restoration of sulfate-containing saline greenhouse soil. Electrochim. Acta 2012, 86, 57–62. [Google Scholar] [CrossRef]
- Ryu, B.-G.; Yang, J.-S.; Kim, D.-H.; Baek, K. Pulsed electrokinetic removal of Cd and Zn from fine-grained soil. J. Appl. Electrochem. 2009, 40, 1039–1047. [Google Scholar] [CrossRef]
- Hristova, D.; Betova, I.; Tzvetkoff, T. An electrochemical and analytical characterization of surface films on AISI 316 as electrode material for pulse electrolysis of water. Int. J. Hydrogen Energy 2013, 38, 8232–8243. [Google Scholar] [CrossRef]
- Cucchiella, F.; D’Adamo, I. Feasibility study of developing photovoltaic power projects in Italy: An integrated approach. Renew Sustain. Energy Rev. 2012, 16, 1562–1576. [Google Scholar] [CrossRef]
- Gao, S.; Chen, Y.; Su, J.; Wang, M.; Wei, X.; Jiang, T.; Wang, Z.L. Triboelectric nanogenerator powered electrochemical degradation of organic pollutant using Pt-free carbon materials. ACS Nano 2017, 11, 3965–3972. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Jian, Z.; Cheng, X.; Mei, Y.; Hu, C.; Mei, W.; Li, J.F. Electrokinetic remediation of soil containing Cr(VI) by photovoltaic solar panels and a DC-DC converter. J. Chem. Technol. Biotechnol. 2015, 90, 693–700. [Google Scholar] [CrossRef]
- Jeon, E.K.; Ryu, S.R.; Baek, K. Application of solar-cells in the electrokinetic remediation of As-contaminated soil. Electrochim. Acta 2015, 181, 160–166. [Google Scholar] [CrossRef]
- Hassan, I.; Mohamedelhassan, E.; Yanful, E.K. Solar powered electrokinetic remediation of Cu polluted soil using a novel anode configuration. Electrochim. Acta 2015, 181, 58–67. [Google Scholar] [CrossRef]
- Souza, F.L.; Saéz, C.; Llanos, J.; Lanza, M.R.V.; Cañizares, P.; Rodrigo, M.A. Solar-powered electrokinetic remediation for the treatment of soil polluted with the herbicide 2,4-D. Electrochim. Acta 2016, 190, 371–377. [Google Scholar] [CrossRef]
- Yuan, S.; Zheng, Z.; Chen, J.; Lu, X. Use of solar cell in electrokinetic remediation of cadmium-contaminated soil. J. Hazard. Mater. 2009, 162, 1583–1587. [Google Scholar] [CrossRef]
- Zhou, M.; Xu, J.; Zhu, S.; Wang, Y.; Gao, H. Exchange electrode-electrokinetic remediation of Cr-contaminated soil using solar energy. Separ. Purif. Technol. 2018, 190, 297–306. [Google Scholar] [CrossRef]
- Hansen, H.K.; Rojo, A. Testing pulsed electric fields in electroremediation of copper mine tailings. Electrochim. Acta 2007, 52, 3399–3405. [Google Scholar] [CrossRef]
- Hussein, A.A.; Alatabe, M.J.A. Remediation of lead-contaminated soil, using clean energy in combination with electro-kinetic methods. Pollution 2019, 5, 859–869. [Google Scholar]
- Ganiyu, S.O.; Martínez-Huitle, C.A. The use of renewable energies driving electrochemical technologies for environmental applications. Curr. Opin. Electrochem. 2020, 22, 211–220. [Google Scholar] [CrossRef]
- Millán, M.; Lobato, J.; Cañizares, P.; Rodrigo, M.A. Prediction and management of solar energy to power electrochemical processes for the treatment of wastewater effluents. Electrochim. Acta 2020, 335, 135594. [Google Scholar] [CrossRef]
- Souza, F.L.; Lanza, M.R.V.; Llanos, J.; Sáez, C.; Rodrigo, M.A.; Cañizares, P. A wind-powered BDD electrochemical oxidation process for the removal of herbicides. J. Environ. Manag. 2015, 158, 36–39. [Google Scholar] [CrossRef] [PubMed]
- Malek, P.; Ortiz, J.M.; Schulte-Herbrüggen, H.M.A. Decentralized desalination of brackish water using an electrodialysis system directly powered by wind energy. Desalination 2016, 377, 54–64. [Google Scholar] [CrossRef]
- Souza, F.L.; Llanos, J.; Sáez, C.; Lanza, M.R.V.; Rodrigo, M.A.; Cañizares, P. Performance of wind-powered soil electroremediation process for the removal of 2,4-D from soil. J. Environ. Manag. 2016, 171, 128–132. [Google Scholar] [CrossRef]
- Wei, H.; Cui, D.; Ma, J.; Chu, L.; Zhao, X.; Song, H.; Liu, H.; Liu, T.; Wang, N.; Guo, Z. Energy conversion technologies towards self-powered electrochemical energy storage systems: The state of the art and perspectives. J. Mater. Chem. A 2017, 5, 1873–1894. [Google Scholar] [CrossRef]
- Ould Amrouche, S.; Rekioua, D.; Rekioua, T.; Bacha, S. Overview of energy storage in renewable energy systems. Int. J. Hydrogen Energy 2016, 41, 20914–20927. [Google Scholar] [CrossRef]
- Fernandez-Marchante, C.M.; Millán, M.; Medina-Santos, J.I.; Lobato, J. Environmental and preliminary cost assessments of redox flow batteries for renewable energy storage. Energy Technol. 2019, 8, 1900914. [Google Scholar] [CrossRef]
- Alotto, P.; Guarnieri, M.; Moro, F. Redox flow batteries for the storage of renewable energy: A review. Renew. Sustain. Energy Rev. 2014, 29, 325–335. [Google Scholar] [CrossRef]
- Gonçalves, S.; Rodrigues, T.P.; Chagas, A.L.S. The impact of wind power on the Brazilian labor market. Renew. Sustain. Energy Rev. 2020, 128, 109887. [Google Scholar] [CrossRef]
- Shubbak Mahmood, H. Advances in solar photovoltaics: Technology review and patent trends. Renew. Sustain. Energy Rev. 2019, 115, 109383. [Google Scholar] [CrossRef]
- Logan, B.E.; Regan, J.M. Electricity-producing bacterial communities in microbial fuel cells. Trends Microbiol. 2006, 14, 512–518. [Google Scholar] [CrossRef] [PubMed]
- Habibul, N.; Hu, Y.; Sheng, G.P. Microbial fuel cell driving electrokinetic remediation of toxic metal contaminated soils. J. Hazard. Mater. 2016, 318, 9–14. [Google Scholar] [CrossRef]
- Song, T.S.; Zhang, J.; Hou, S.; Wang, H.; Zhang, D.; Li, S.; Xie, J. In situ electrokinetic remediation of toxic metal-contaminated soil driven by solid phase microbial fuel cells with a wheat straw addition. J. Chem. Technol. Biotechnol. 2018, 93, 2860–2867. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vocciante, M.; Dovì, V.G.; Ferro, S. Sustainability in ElectroKinetic Remediation Processes: A Critical Analysis. Sustainability 2021, 13, 770. https://doi.org/10.3390/su13020770
Vocciante M, Dovì VG, Ferro S. Sustainability in ElectroKinetic Remediation Processes: A Critical Analysis. Sustainability. 2021; 13(2):770. https://doi.org/10.3390/su13020770
Chicago/Turabian StyleVocciante, Marco, Vincenzo G. Dovì, and Sergio Ferro. 2021. "Sustainability in ElectroKinetic Remediation Processes: A Critical Analysis" Sustainability 13, no. 2: 770. https://doi.org/10.3390/su13020770
APA StyleVocciante, M., Dovì, V. G., & Ferro, S. (2021). Sustainability in ElectroKinetic Remediation Processes: A Critical Analysis. Sustainability, 13(2), 770. https://doi.org/10.3390/su13020770





