Circularity Study on PET Bottle-To-Bottle Recycling
Abstract
1. Introduction
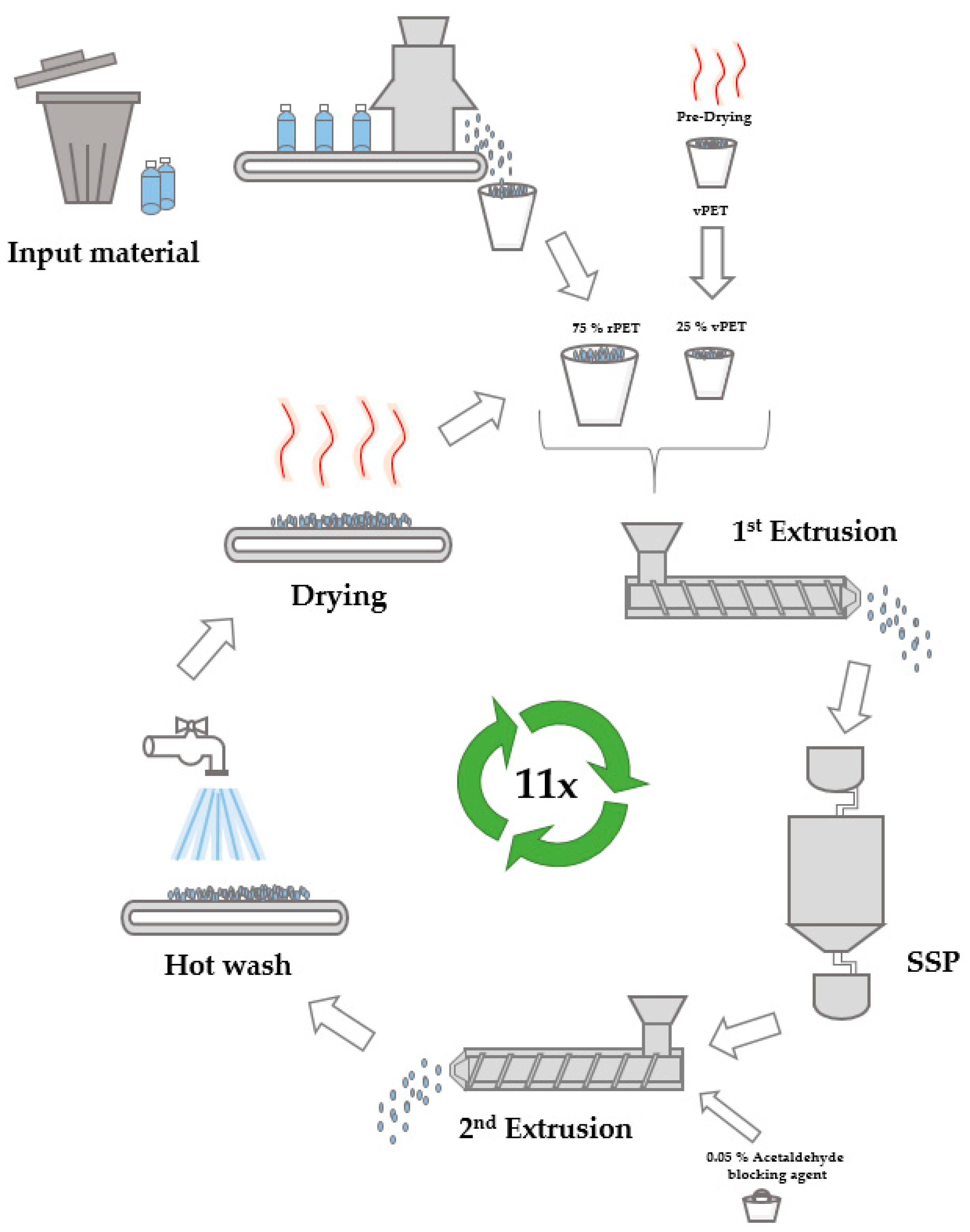
2. Materials and Methods
3. Results
3.1. Intrinsic Viscosity and Optical Quality
3.2. Chemical Quality
3.3. Biological Quality
3.4. Bottle Production
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Kirchherr, J.; Reike, D.; Hekkert, M. Conceptualizing the circular economy: An analysis of 114 definitions. Resour. Conserv. Recycl. 2017, 127, 221–232. [Google Scholar] [CrossRef]
- Eriksen, M.K.; Pivnenko, K.; Olsson, M.E.; Astrup, T.F. Contamination in plastic recycling: Influence of metals on the quality of reprocessed plastic. Waste Manag. 2018, 79, 595–606. [Google Scholar] [CrossRef]
- Welle, F. Twenty years of PET bottle to bottle recycling—An overview. Resour. Conserv. Recycl. 2011, 55, 865–875. [Google Scholar] [CrossRef]
- Awaja, F.; Pavel, D. Recycling of PET. Eur. Polym. J. 2005, 41, 1453–1477. [Google Scholar] [CrossRef]
- Palkopoulou, S.; Joly, C.; Feigenbaum, A.; Papaspyrides, C.D.; Dole, P. Critical review on challenge tests to demonstrate decontamination of polyolefins intended for food contact applications. Trends Food Sci. Technol. 2016, 49, 110–120. [Google Scholar] [CrossRef]
- EFSA. Scientific Opinion on the safety evaluation of the following processes based on Starlinger IV+ ® technology used to recycle post-consumer PET into food contact materials “Preformia, STF, MPTS, PET to PET and Eco Plastic”. EFSA J. 2012, 10, 2828. [Google Scholar] [CrossRef]
- EFSA. Scientific Opinion on the safety evaluation of the following processes based on VACUREMA Prime ® technology used to recycle post-consumer PET into food contact materials “Lux PET”, “Jayplas”, “PolyQuest” and “CIER”. EFSA J. 2012, 10, 2827. [Google Scholar] [CrossRef][Green Version]
- EFSA. Scientific Opinion on the safety evaluation of the process “PETUK SSP” for production of recycled post-consumer PET for use in food contact materials. EFSA J. 2012, 10, 2826. [Google Scholar] [CrossRef][Green Version]
- Eunomia. PET Market in Europe: State of Play; Production, Collection and Recycling Data; London, UK, 2020. Available online: https://www.eunomia.co.uk/reports-tools/pet-market-in-europe-state-of-play/ (accessed on 6 June 2021).
- Brouwer, M.T.; Chacon, F.A.; van Velzen, E.U.T. Effect of recycled content and rPET quality on the properties of PET bottles, part III: Modelling of repetitive recycling. Packag. Technol. Sci. 2020, 33, 373–383. [Google Scholar] [CrossRef]
- Ellen MacArthur Foundation. The Global Commitment: 2020 Progress Report. 2020. Available online: https://www.ellenmacarthurfoundation.org/assets/downloads/Global-Commitment-2020-Progress-Report.pdf (accessed on 15 January 2021).
- Petcore Europe. Pledge for the PET Value Chain to Increase Circularity and Recycling, Brussels. 2019. Available online: https://circulareconomy.europa.eu/platform/sites/default/files/plastic_pledge_statement_petcore_europe_.pdf (accessed on 14 January 2020).
- Ellen MacArthur Foundation. New Plastics Economy Global Commitment: Commitments, Vision and Definitions. 2020. Available online: https://www.newplasticseconomy.org/assets/doc/Global-Commitment_Definitions_2020-1.pdf (accessed on 15 January 2021).
- Petcore Europe. Profile, Activities and Benefits. 2019. Available online: https://www.petcore-europe.org/images/news/pdf/Petcore-Europe_General-presentation_2019.pdf (accessed on 14 January 2020).
- Schneider, M. Unternehmen in Bad Neuenahr Lebt von der Plastikflasche: Firmen-Porträt Petcycle. 2020. Available online: https://ga.de/news/wirtschaft/regional/die-firma-petcycle-ist-abhaengig-von-der-zukunft-der-plastikflaschen_aid-50460293 (accessed on 2 March 2021).
- Petcycle. PETCYCLE Beschließt Steigerung des Recyclateinsatzes auf 75 Prozent. 2020. Available online: https://www.petcycle.de/2020/12/petcycle-beschliesst-steigerung-des-recyclateinsatzes-auf-75-prozent/ (accessed on 2 March 2021).
- European Parliament. Directive (EU) 2019/904 of the European Parliament and of the Council of 5 June 2019 on the reduction of the impact of certain plastic products on the environment: EU 2019/904. Off. J. Eur. Union 2019, 155, 1–19. [Google Scholar]
- Schilter, B.; Burnett, K.; Eskes, C.; Geurts, L.; Jacquet, M.; Kirchnawy, C.; Oldring, P.; Pieper, G.; Pinter, E.; Tacker, M.; et al. Value and limitation of in vitro bioassays to support the application of the threshold of toxicological concern to prioritise unidentified chemicals in food contact materials. Food Addit. Contam. Part A 2019, 36, 1903–1936. [Google Scholar] [CrossRef]
- Franz, R.; Welle, F. Contamination Levels in Recollected PET Bottles from Non-Food Applications and their Impact on the Safety of Recycled PET for Food Contact. Molecules 2020, 25, 4998. [Google Scholar] [CrossRef]
- EFSA. Scientific Opinion on the criteria to be used for safety evaluation of a mechanical recycling process to produce recycled PET intended to be used for manufacture of materials and articles in contact with food. EFSA J. 2011, 9, 893. [Google Scholar] [CrossRef]
- Geueke, B.; Groh, K.; Muncke, J. Food packaging in the circular economy: Overview of chemical safety aspects for commonly used materials. J. Clean. Prod. 2018, 193, 491–505. [Google Scholar] [CrossRef]
- Franz, R.; Mauer, A.; Welle, F. European survey on post-consumer poly (ethylene terephthalate) (PET) materials to determine contamination levels and maximum consumer exposure from food packages made from recycled PET. Food Addit. Contam. 2004, 21, 265–286. [Google Scholar] [CrossRef] [PubMed]
- Welle, F. Migration of Acetaldehyde from PET Bottles into Natural Mineral Water. In Reference Module in Food Science; Elsevier: Amsterdam, The Netherlands, 2018; p. 149. [Google Scholar]
- Mutsuga, M.; Kawamura, Y.; Sugita-Konishi, Y.; Hara-Kudo, Y.; Takatori, K.; Tanamoto, K. Migration of formaldehyde and acetaldehyde into mineral water in polyethylene terephthalate (PET) bottles. Food Addit. Contam. 2006, 23, 212–218. [Google Scholar] [CrossRef] [PubMed]
- van Velzen, E.U.T.; Brouwer, M.T.; Stärker, C.; Welle, F. Effect of recycled content and rPET quality on the properties of PET bottles, part II: Migration. Packag. Technol. Sci. 2020, 33, 359–371. [Google Scholar] [CrossRef]
- Kassouf, A.; Maalouly, J.; Chebib, H.; Rutledge, D.N.; Ducruet, V. Chemometric tools to highlight non-intentionally added substances (NIAS) in polyethylene terephthalate (PET). Talanta 2013, 115, 928–937. [Google Scholar] [CrossRef]
- Tawfik, M.S.; Devlieghere, F.; Huyghebaert, A. Influence of D-limonene absorption on the physical properties of refillable PET. Food Chem. 1998, 61, 157–162. [Google Scholar] [CrossRef]
- Alaerts, L.; Augustinus, M.; van Acker, K. Impact of Bio-Based Plastics on Current Recycling of Plastics. Sustainability 2018, 10, 1487. [Google Scholar] [CrossRef]
- Chacon, F.A.; Brouwer, M.T.; van Velzen, E.U.T. Effect of recycled content and rPET quality on the properties of PET bottles, part I: Optical and mechanical properties. Packag. Technol. Sci. 2020, 33, 347–357. [Google Scholar] [CrossRef]
- Berg, D.; Schaefer, K.; Koerner, A.; Kaufmann, R.; Tillmann, W.; Moeller, M. Reasons for the Discoloration of Postconsumer Poly (ethylene terephthalate) during Reprocessing. Macromol. Mater. Eng. 2016, 301, 1454–1467. [Google Scholar] [CrossRef]
- Luijsterburg, B.; Goossens, H. Assessment of plastic packaging waste: Material origin, methods, properties. Resour. Conserv. Recycl. 2014, 85, 88–97. [Google Scholar] [CrossRef]
- Welle, F.; Bayer, F.; Franz, R. Quantification of the Sorption Behavior of Polyethylene Terephthalate Polymer versus PET/PA Polymer Blends towards Organic Compounds. Packag. Technol. Sci. 2012, 25, 341–349. [Google Scholar] [CrossRef]
- Cruz, S.A.; Zanin, M. PET recycling: Evaluation of the solid state polymerization process. J. Appl. Polym. Sci. 2006, 99, 2117–2123. [Google Scholar] [CrossRef]
- Reloop. Global Deposit Book 2020: An Overview of Deposit Systems for One-Way Beverage Containers. 2020. Available online: https://www.reloopplatform.org/wp-content/uploads/2020/12/2020-Global-Deposit-Book-WEB-version-1DEC2020.pdf (accessed on 19 May 2021).
- Rainer, B.; Mayrhofer, E.; Redl, M.; Dolak, I.; Mislivececk, D.; Czerny, T.; Kirchnawy, C.; Marin-Kuan, M.; Schilter, B.; Tacker, M. Mutagenicity Assessment of Food Contact Material Migrates with the Ames MPF Assay. Food Addit. Contam. Part A 2019, 36, 1419–1432. [Google Scholar] [CrossRef]
- Rieckmann, T.; Besse, K.; Frei, F.; Völker, S. Quantification of Colour Formation in PET Depending on SSP Residence Time, Temperature, and Oxygen Concentration. Macromol. Symp. 2013, 333, 162–171. [Google Scholar] [CrossRef]
- Commission Regulation (EU) No 10/2011 on Plastic Materials and Articles Intended to Come into Contact with Food. Off. J. Eur. Union 2011. Available online: https://eur-lex.europa.eu/legal-content/EN/ALL/?uri=CELEX%3A32011R0010 (accessed on 2 March 2021).
- Barthélémy, E.; Spyropoulos, D.; Milana, M.-R.; Pfaff, K.; Gontard, N.; Lampi, E.; Castle, L. Safety evaluation of mechanical recycling processes used to produce polyethylene terephthalate (PET) intended for food contact applications. Food Addit. Contam. Part A 2014, 31, 490–497. [Google Scholar] [CrossRef]
- Mrozinski, B.A.; Kim, Y.-W.; Lofgren, E.A.; Jabarin, S.A. Chemistry of the interactions of acetaldehyde scavengers for poly (ethylene terephthalate). J. Appl. Polym. Sci. 2013, 130, 4191–4200. [Google Scholar] [CrossRef]
- Franz, R.; Gmeiner, M.; Gruner, A.; Kemmer, D.; Welle, F. Diffusion behaviour of the acetaldehyde scavenger 2-aminobenzamide in polyethylene terephthalate for beverage bottles. Food Addit. Contam. Part A 2016, 33, 364–372. [Google Scholar] [CrossRef]
- Communication from the Commission: The European Green Deal; European Commission: Brussels, Belgium, 2019.
- Pauer, E.; Wohner, B.; Heinrich, V.; Tacker, M. Assessing the Environmental Sustainability of Food Packaging: An Extended Life Cycle Assessment including Packaging-Related Food Losses and Waste and Circularity Assessment. Sustainability 2019, 11, 925. [Google Scholar] [CrossRef]
- Masmoudi, F.; Fenouillot, F.; Mehri, A.; Jaziri, M.; Ammar, E. Characterization and quality assessment of recycled post-consumption poly (ethylene terephthalate) (PET). Environ. Sci. Pollut. Res. Int. 2018, 25, 23307–23314. [Google Scholar] [CrossRef]
- Demirel, B. Optimisation of mould surface temperature and bottle residence time in mould for the carbonated soft drink PET containers. Polym. Test. 2017, 60, 220–228. [Google Scholar] [CrossRef]
- Nijssen, B.; Kamperman, T.; Jetten, J. Acetaldehyde in Mineral Water Stored in Polyethylene Terephthalate (PET) Bottles: Odour Threshold and Quantification. Packag. Technol. Sci. 1996, 9, 175–185. [Google Scholar] [CrossRef]
- Strube, A.; Buettner, A.; Groetzinger, C. Characterization and identification of a plastic-like off-odor in mineral water. Water Supply 2009, 9, 299–309. [Google Scholar] [CrossRef]
- Commission Regulation (EC) No 282/2008 on Recycled Plastic Materials and Articles Intended to Come into Contact with Foods. Off. J. Eur. Union 2008. Available online: https://eur-lex.europa.eu/legal-content/EN/ALL/?uri=CELEX%3A32008R0282 (accessed on 2 March 2021).
- Pinter, E.; Rainer, B.; Czerny, T.; Riegel, E.; Schilter, B.; Marin-Kuan, M.; Tacker, M. Evaluation of the Suitability of Mammalian In Vitro Assays to Assess the Genotoxic Potential of Food Contact Materials. Foods 2020, 9, 237. [Google Scholar] [CrossRef]
- Rainer, B.; Pinter, E.; Czerny, T.; Riegel, E.; Kirchnawy, C.; Marin-Kuan, M.; Schilter, B.; Tacker, M. Suitability of the Ames test to characterise genotoxicity of food contact material migrates. Food Addit. Contam. Part A Chem. Anal. Control Expo. Risk Assess. 2018, 35, 2230–2243. [Google Scholar] [CrossRef]
- Eriksen, M.K.; Christiansen, J.D.; Daugaard, A.E.; Astrup, T.F. Closing the loop for PET, PE and PP waste from households: Influence of material properties and product design for plastic recycling. Waste Manag. 2019, 96, 75–85. [Google Scholar] [CrossRef]
- McDonough, W.; Braungart, M. Cradle to Cradle: Remaking the Way We Make Things, 1st ed.; North Point Press: New York, NY, USA, 2002. [Google Scholar]


| Sample Code | IV [dL/g] | CIE L*a*b* Color Value | |||
|---|---|---|---|---|---|
| L* | a* | b* | ∆E | ||
| vPET | 0.820 | 84.44 | −2.02 | −1.51 | |
| rPET Flakes | 0.778 | - | - | - | |
| Cycle 1 | 0.870 | 69.76 | −2.59 | 0.92 | 14.89 |
| Cycle 2 | 0.854 | 69.74 | −2.80 | 0.47 | 14.85 |
| Cycle 3 | 0.830 | 66.23 | −2.73 | 0.14 | 18.30 |
| Cycle 4 | 0.845 | 69.24 | −2.87 | 0.59 | 15.37 |
| Cycle 5 | 0.835 | 68.67 | −2.86 | 1.35 | 16.05 |
| Cycle 6 | 0.804 | 69.55 | −2.82 | 1.05 | 15.13 |
| Cycle 7 | 0.859 | 69.72 | −2.90 | 0.40 | 14.87 |
| Cycle 8 | 0.801 | 69.88 | −3.00 | 0.59 | 14.74 |
| Cycle 9 | 0.781 | 70.06 | −2.75 | 0.76 | 14.58 |
| Cycle 10 | 0.797 | 68.53 | −2.70 | 0.54 | 16.06 |
| Cycle 11 | 0.809 | 71.02 | −2.68 | 0.30 | 13.56 |
| Sample | Acetaldehyde [mg/kg] | 2-Methyl-1,3-dioxolane [mg/kg] | Benzene [mg/kg] | Ethylene Glycol [mg/kg] | Limonene [mg/kg] |
|---|---|---|---|---|---|
| rPET flakes | 8.2 ± 4.71 | 2.8 ± 1.0 | 0.25 ± 0.25 | 6.7 ± 1.5 | 3.2 ± 1.0 |
| Cycle 1 | 1.0 ± 0.1 | 0.12 ± 0.1 | <0.03 | 0.77 ± 0.02 | <0.03 |
| Cycle 2 | 1.2 ± 0.1 | 0.12 ± 0.1 | <0.03 | 0.66 ± 0.03 | <0.03 |
| Cycle 3 | 1.5 ± 0.1 | 0.14 ± 0.1 | <0.03 | 0.70 ± 0.01 | <0.03 |
| Cycle 4 | 1.3 ± 0.1 | 0.13 ± 0.1 | <0.03 | 0.61 ± 0.01 | <0.03 |
| Cycle 5 | 1.7 ± 0.1 | 0.14 ± 0.1 | <0.03 | 0.88 ± 0.01 | <0.03 |
| Cycle 6 | 1.7 ± 0.1 | 0.16 ± 0.1 | <0.03 | 0.59 ± 0.04 | <0.03 |
| Cycle 7 | 1.4 ± 0.1 | 0.14 ± 0.1 | <0.03 | 0.54 ± 0.01 | <0.03 |
| Cycle 8 | 2.1 ± 0.1 | 0.14 ± 0.1 | <0.03 | 1.12 ± 0.01 | <0.03 |
| Cycle 9 | 1.6 ± 0.1 | 0.13 ± 0.1 | <0.03 | 0.92 ± 0.04 | <0.03 |
| Cycle 10 | 1.4 ± 0.1 | 0.12 ± 0.1 | <0.03 | 0.75 ± 0.01 | <0.03 |
| Cycle 11 | 1.4 ± 0.1 | 0.12 ± 0.1 | <0.03 | 0.73 ± 0.03 | <0.03 |
| Sample | Anthranil Amide [mg/kg] | PET Dimer [mg/kg] | PET Dimer [mg/kg] | PET Dimer [mg/kg] | PET Substance [mg/kg] | PET Trimer [mg/kg] | PET Trimer [mg/kg] | PET Trimer [mg/kg] |
|---|---|---|---|---|---|---|---|---|
| rPET flakes | 10.0 ± 0.4 | 51.9 ± 4.7 | 26.8 ± 0.7 | 130.7 ± 7.5 | 11.4 ± 0.5 | 119.7 ± 9.2 | 4310 ± 138 | 81.6 ± 11.9 |
| Cycle 1 | <5 | 15.5 ± 1.3 | 10.5 ± 0.5 | 53.4 ± 2.6 | 4.0 ± 0.7 | 40.6 ± 1.9 | 1370 ± 58 | 20.9 ± 0.9 |
| Cycle 2 | <5 | 16.5 ± 2.3 | 11.3 ± 0.6 | 56.0 ± 0.4 | 3.9 ± 0.2 | 43.7 ± 0.8 | 1490 ± 12 | 24.1 ± 2.2 |
| Cycle 3 | <5 | 15.2 ± 1.2 | 9.0 ± 0.4 | 47.7 ± 0.7 | 3.2 ± 0.1 | 38.2 ± 0.9 | 1340 ± 25 | 19.6 ± 2.0 |
| Cycle 4 | <5 | 16.2 ± 3.6 | 11.1 ± 0.5 | 57.9 ± 2.3 | 4.0 ± 0.2 | 45.4 ± 1.8 | 1600 ± 17 | 24.3 ± 1.4 |
| Cycle 5 | <5 | 19.6 ± 1.3 | 12.5 ± 0.7 | 66.2 ± 3.2 | 4.2 ± 0.5 | 50.0 ± 0.7 | 1800 ± 38 | 29.2 ± 1.8 |
| Cycle 6 | <5 | 17.7 ± 1.9 | 11.0 ± 0.5 | 59.3 ± 3.3 | 3.7 ± 0.2 | 44.5 ± 1.6 | 1620 ± 55 | 26.1 ± 1.0 |
| Cycle 7 | <5 | 16.8 ± 1.5 | 10.8 ± 0.3 | 59.5 ± 1.3 | 4.1 ± 0.5 | 44.4 ± 0.5 | 1650 ± 15 | 27.3 ± 2.2 |
| Cycle 8 | <5 | 16.1 ± 2.3 | 11.2 ± 0.6 | 61.7 ± 2.9 | 4.0 ± 0.2 | 46.2 ± 1.1 | 1710 ± 35 | 28.1 ± 1.7 |
| Cycle 9 | <5 | 16.5 ± 1.4 | 10.4 ± 0.7 | 55.3 ± 0.4 | 3.6 ± 0.4 | 41.9 ± 1.1 | 1570 ± 38 | 24.1 ± 2.5 |
| Cycle 10 | <5 | 14.9 ± 2.3 | 9.8 ± 0.5 | 52.5 ± 2.2 | 3.8 ± 0.2 | 40.2 ± 1.0 | 1520 ± 34 | 25.1 ± 3.0 |
| Cycle 11 | <5 | 13.2 ± 1.8 | 8.0 ± 0.7 | 42.1 ± 2.2 | 2.9 ± 0.2 | 34.2 ± 1.3 | 1280 ± 36 | 21.4 ± 2.7 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pinter, E.; Welle, F.; Mayrhofer, E.; Pechhacker, A.; Motloch, L.; Lahme, V.; Grant, A.; Tacker, M. Circularity Study on PET Bottle-To-Bottle Recycling. Sustainability 2021, 13, 7370. https://doi.org/10.3390/su13137370
Pinter E, Welle F, Mayrhofer E, Pechhacker A, Motloch L, Lahme V, Grant A, Tacker M. Circularity Study on PET Bottle-To-Bottle Recycling. Sustainability. 2021; 13(13):7370. https://doi.org/10.3390/su13137370
Chicago/Turabian StylePinter, Elisabeth, Frank Welle, Elisa Mayrhofer, Andreas Pechhacker, Lukas Motloch, Vera Lahme, Andy Grant, and Manfred Tacker. 2021. "Circularity Study on PET Bottle-To-Bottle Recycling" Sustainability 13, no. 13: 7370. https://doi.org/10.3390/su13137370
APA StylePinter, E., Welle, F., Mayrhofer, E., Pechhacker, A., Motloch, L., Lahme, V., Grant, A., & Tacker, M. (2021). Circularity Study on PET Bottle-To-Bottle Recycling. Sustainability, 13(13), 7370. https://doi.org/10.3390/su13137370









