Safety Evaluation of Polyethylene Terephthalate Chemical Recycling Processes
Abstract
:1. Introduction
- Contamination level of the input of the recycling process;
- Cleaning efficiency of the applied mechanical recycling process;
- Exposure scenario to the consumer.
2. Materials and Methods
2.1. Artificially Contaminated PET
2.2. Depolimerisation of Contaminated PET Flakes
2.3. Repolimerisation of BHET
2.4. Determination of the Concentrations of the Model Substance in PET and BHET
2.5. Diffusion Modelling
3. Results and Discussion
3.1. Inut Contamination Levels
3.2. Cleaning Efficiency of the Chemical Process
3.3. Safety Evaluation of a PET Chemical Recycling Process
4. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Lugal, L.; Grant, A.; Cordle, M.; Fletcher, D. PET Market in Europe: State of Play. Eunomia Reports 2020. Available online: https://www.eunomia.co.uk/reports-tools/pet-market-in-europe-state-of-play/ (accessed on 14 October 2021).
- Platt, P. The Future of PET Packaging to 2025; Smithers Information Ltd.: Akron, OH, USA, 2020. [Google Scholar]
- Shojaei, B.; Abtahi, M.; Najafi, M. Chemical recycling of PET: A stepping-stone toward sustainability. Polym. Adv. Technol. 2020, 31, 2912–2938. [Google Scholar] [CrossRef]
- Welle, F. Twenty Years of PET Bottle to Bottle Recycling—An Overview. Res. Conserv. Recycl. 2011, 55, 865–875. [Google Scholar] [CrossRef]
- Van Velzen, E.U.T.; Brouwer, M.T.; Stärker, C.; Welle, F. The effect of recycled content and rPET quality on the properties of PET bottles, Part II: Migration. Pack. Techn. Sci. 2020, 33, 359–371. [Google Scholar] [CrossRef]
- Lechleitner, A.; Schwabl, D.; Schubert, T.; Bauer, M.; Lehner, M. Chemical recycling of mixed waste plastics as complementary process pathway to increase recycling rates. Österr Wasser- Abfallw. 2020, 72, 47–60. [Google Scholar] [CrossRef] [Green Version]
- Chaudhari, U.S.; Lin, Y.; Thompson, V.S.; Handler, R.M.; Pearce, J.M.; Caneba, G.; Muhuri, P.; Watkins, D.; Shonnard, D.R. Systems analysis approach to polyethylene terephthalate and olefin plastics supply chains in the circular economy: A review of data sets and models. ACS Sustain. Chem. Eng. 2021, 9, 7403–7421. [Google Scholar] [CrossRef]
- Park, S.H.; Kim, S.H. Poly (ethylene terephthalate) recycling for high value added textiles. Fash. Text. 2014, 1, 1. [Google Scholar] [CrossRef] [Green Version]
- Sinha, V.; Patel, M.R.; Patel, J.V. PET waste management by chemical recycling: A review. J. Polym. Environ. 2010, 18, 8–25. [Google Scholar] [CrossRef]
- Geyer, B.; Röhner, S.; Lorenz, G.; Kandelbauer, A. Designing oligomeric ethylene terephthalate building blocks by chemical recycling of polyethylene terephtalate. J. Appl. Polym. Sci. 2014, 131, 1–12. [Google Scholar] [CrossRef]
- Payne, J.; Jones, M.D. The chemical recycling of polyesters for a circular plastics economy: Challenges and emerging opportunities. ChemSusChem 2021, 14, 4041–4070. [Google Scholar] [CrossRef] [PubMed]
- Welle, F. Polyethylene terephthalate (PET)—Prime material for a circular economy. Kunstst. Int. 2019, 109, 51–54. [Google Scholar]
- EFSA Panel on Food Contact Materials, Enzymes, Flavourings and Processing Aids (CEF). Scientific Opinion on the criteria to be used for safety evaluation of a mechanical recycling process to produce recycled PET intended to be used for manufacture of materials and articles in contact with food. EFSA J. 2011, 9, 2184. [Google Scholar]
- Franz, R.; Mauer, A.; Welle, F. European survey on post-consumer poly(ethylene terephthalate) materials to determine contamination levels and maximum consumer exposure from food packages made from recycled PET. Food Add. Contam. 2004, 21, 265–286. [Google Scholar] [CrossRef] [PubMed]
- Kroes, R.; Renwick, A.G.; Feron, V.; Galli, C.L.; Gibney, M.; Greim, H.; Guy, R.H.; Lhuguenot, J.C.; van de Sandt, J.J.M. Application of the threshold of toxicological concern (TTC) to the safety evaluation of cosmetic ingredients. Food Chem. Toxicol. 2007, 45, 2533–2562. [Google Scholar] [CrossRef] [PubMed]
- Official Journal of the European Union. Commission Regulation (EC) No 282/2008 of 27 March 2008 on recycled plastic materials and articles intended to come into contact with foods and amending Regulation (EC) No 2023/2006. Off. J. Eur. Union 2008, L86/9. Available online: https://eur-lex.europa.eu/LexUriServ/LexUriServ.do?uri=OJ:L:2008:086:0009:0018:EN:PDF (accessed on 16 November 2021).
- ASTM D4603-18. Standard Test Method for Determining Inherent Viscosity of Poly(Ethylene Terephthalate) (PET) by Glass Capillary Viscometer. Developed by Subcommittee: D20.70. Available online: https://www.astm.org/Standards/D4603.htm (accessed on 16 November 2021).
- DIN 32645:2008-11. Chemical Analysis—Decision Limit, Detection Limit and Determination Limit under Repeatability Conditions—Terms, Methods, Evaluation; Beuth Verlag: Berlin, Germany, 2011; Available online: https://www.beuth.de/de/norm/din-32645/110729574 (accessed on 16 November 2021).
- Roduit, B.; Borgeat, C.H.; Cavin, S.; Fragniere, C.; Dudler, V. Application of finite element analysis (FEA) for the simulation of release of additives from multilayer polymeric packaging structures. Food Addit. Contam. Part A 2005, 22, 945–955. [Google Scholar] [CrossRef] [PubMed]
- Brandsch, R.; Dequatre, C.; Mercea, P.; Milana, M.-R.; Störmer, A.; Trier, X.; Vitrac, O.; Schäfer, A.; Simoneau, C. Practical Guidelines on the Application of Migration Modelling for the Estimation of Specific Migration; EU Report 27529 EN; 2015; ISBN 978-92-79-52790-6. Available online: https://op.europa.eu/de/publication-detail/-/publication/1b79bc61-97f6-11e5-983e-01aa75ed71a1 (accessed on 16 November 2021).
- Franz, R.; Welle, F. Contamination levels in re-collected PET bottles from non-food applications and their impact on the safety of recycled PET for food contact. Molecules 2020, 25, 4998. [Google Scholar] [CrossRef] [PubMed]
- Welle, F. Decontamination efficiency of a new post-consumer poly(ethylene terephthalate) (PET) recycling concept. Food Addit. Contam. 2008, 25, 123–131. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- FDA. Use of Recycled Plastics in Food Packaging (Chemistry Considerations): Guidance for Industry. 2021. Available online: https://www.fda.gov/media/150792/download (accessed on 24 October 2021).
| Surrogate | MW 1 | Functional Group | Physical Properties |
|---|---|---|---|
| Toluene | 92.1 | aromatic hydrocarbon | volatile, non-polar |
| Chlorobenzene | 112.6 | halogenated aromatic hydrocarbon | volatile, medium-polar, aggressive to PET |
| Chloroform | 119.4 | halogenated hydrocarbon | volatile, polar, aggressive towards PET |
| Methyl salicylate | 152.2 | aromatic ester | non-volatile, polar |
| Phenyl cyclohexane | 160.3 | aromatic hydrocarbon | non-volatile, non-polar |
| Benzophenone | 182.2 | aromatic ketone | non-volatile, polar |
| Methyl stearate | 298.5 | aliphatic ester | non-volatile, polar |
| Concentration (mg/kg) (Cleaning Efficiency) | ||||
|---|---|---|---|---|
| Contaminated PET Flakes before Depolymerisation | BHET after Depolymerisation | Amorphous PET Pellets after Repolymerisation | Crystallised PET Pellets after Solid-Stating | |
| Toluene | 157.7 ± 8.7 | <0.10 (>99.94%) | <0.10 | <0.10 |
| Chlorobenzene | 733.9 ± 313.0 | <0.03 (>99.99%) | <0.03 | <0.03 |
| Chloroform | 485.1 ± 233.2 | <0.10 (>99.98%) | <0.10 | <0.10 |
| Methyl salicylate | 852.6 ± 297.2 | <0.04 (>99.99%) | <0.04 | <0.04 |
| Phenyl cyclohexane | 720.6 ± 507.2 | <0.05 (>99.99%) | <0.05 | <0.05 |
| Benzophenone | 712.5 ± 352.6 | <0.06 (>99.99%) | <0.06 | <0.06 |
| Methyl stearate | 865.6 ± 507.5 | <0.04 (>99.99%) | <0.04 | <0.04 |
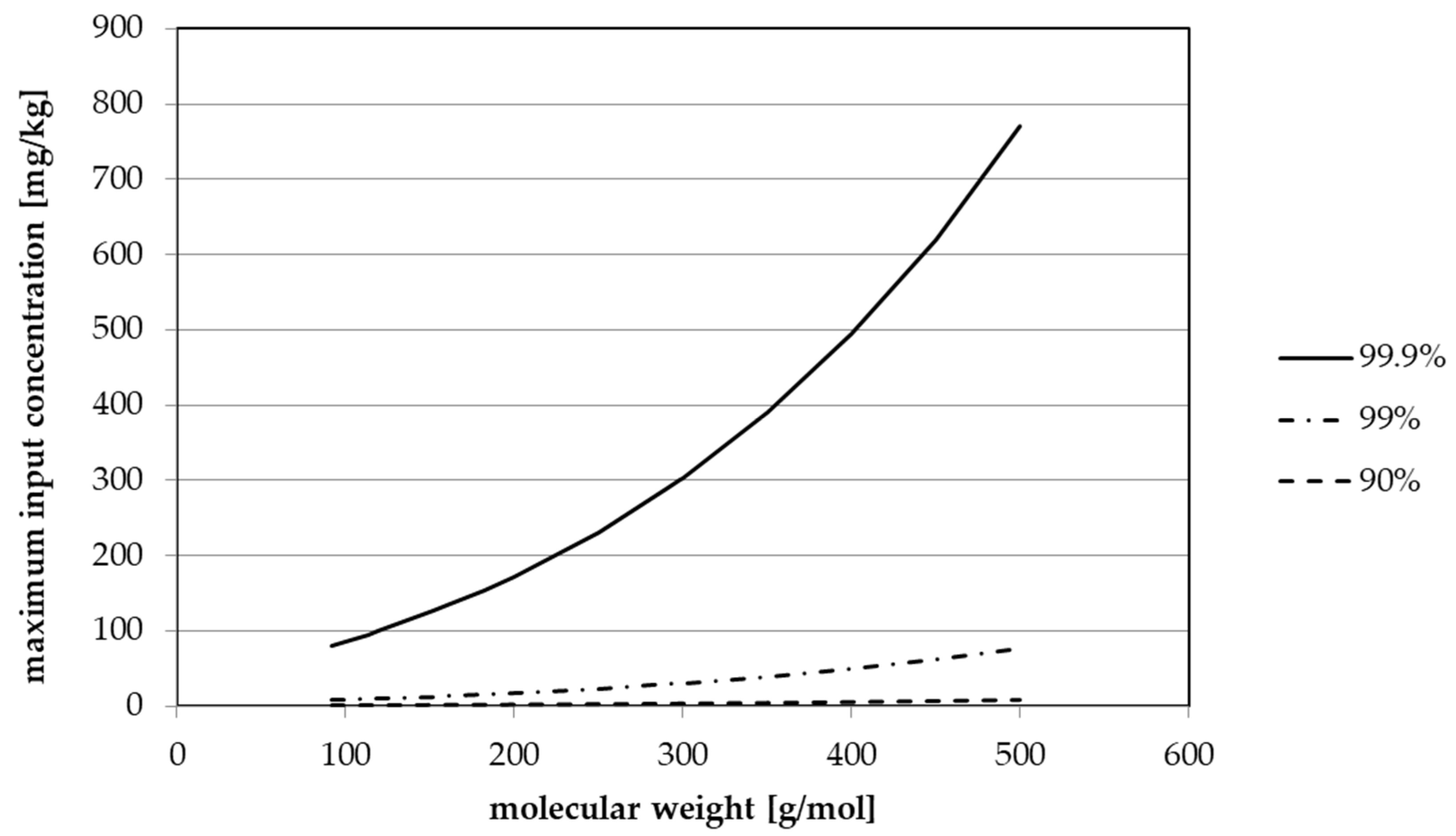
| Surrogate | MW 1 | Maximum Bottle Wall Concentration (mg/kg) Considering the Cleaning Efficiency | |||
|---|---|---|---|---|---|
| 0% | 90% | 99% | 99.9% | ||
| Toluene | 92.1 | 0.0797 | 0.797 | 7.97 | 79.7 |
| Chlorobenzene | 112.6 | 0.0945 | 0.945 | 9.45 | 94.5 |
| Chloroform | 119.4 | 0.0990 | 0.990 | 9.90 | 99.0 |
| Methyl salicylate | 152.2 | 0.126 | 1.26 | 12.6 | 126 |
| Phenyl cyclohexane | 160.3 | 0.133 | 1.33 | 13.3 | 133 |
| Benzophenone | 182.2 | 0.154 | 1.54 | 15.4 | 154 |
| Methyl stearate | 298.5 | 0.300 | 3.00 | 30.0 | 300 |
| Fictive | 400 | 0.495 | 4.95 | 49.5 | 495 |
| Fictive | 500 | 0.770 | 7.70 | 77.0 | 770 |
| Fictive | 750 | 1.98 | 19.8 | 198 | 1980 |
| Fictive | 1000 | 4.41 | 44.1 | 441 | 4410 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Welle, F. Safety Evaluation of Polyethylene Terephthalate Chemical Recycling Processes. Sustainability 2021, 13, 12854. https://doi.org/10.3390/su132212854
Welle F. Safety Evaluation of Polyethylene Terephthalate Chemical Recycling Processes. Sustainability. 2021; 13(22):12854. https://doi.org/10.3390/su132212854
Chicago/Turabian StyleWelle, Frank. 2021. "Safety Evaluation of Polyethylene Terephthalate Chemical Recycling Processes" Sustainability 13, no. 22: 12854. https://doi.org/10.3390/su132212854
APA StyleWelle, F. (2021). Safety Evaluation of Polyethylene Terephthalate Chemical Recycling Processes. Sustainability, 13(22), 12854. https://doi.org/10.3390/su132212854






