The Thames: Arresting Ecosystem Decline and Building Back Better
Abstract
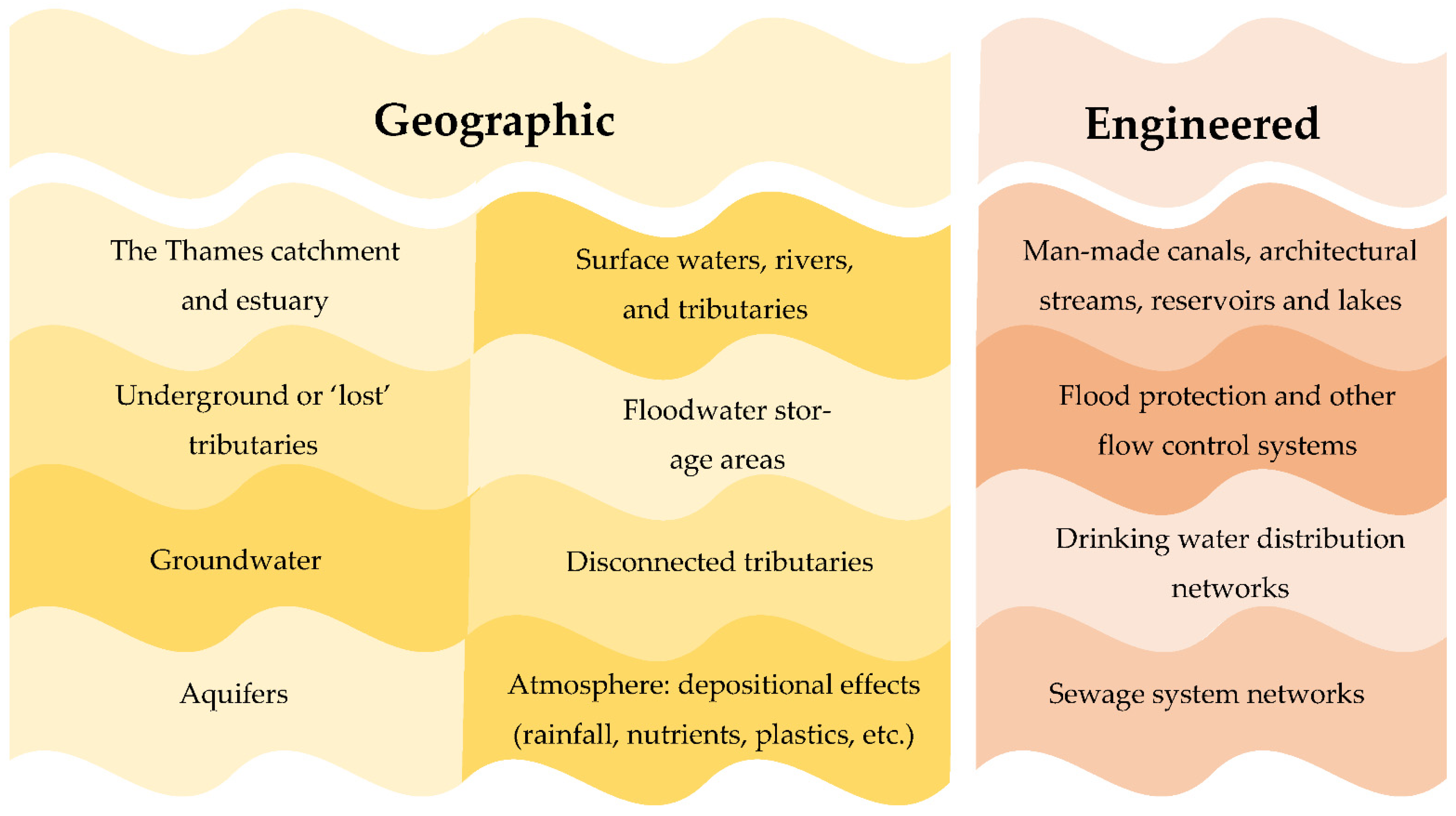
1. Introduction
2. Ecosystem Services
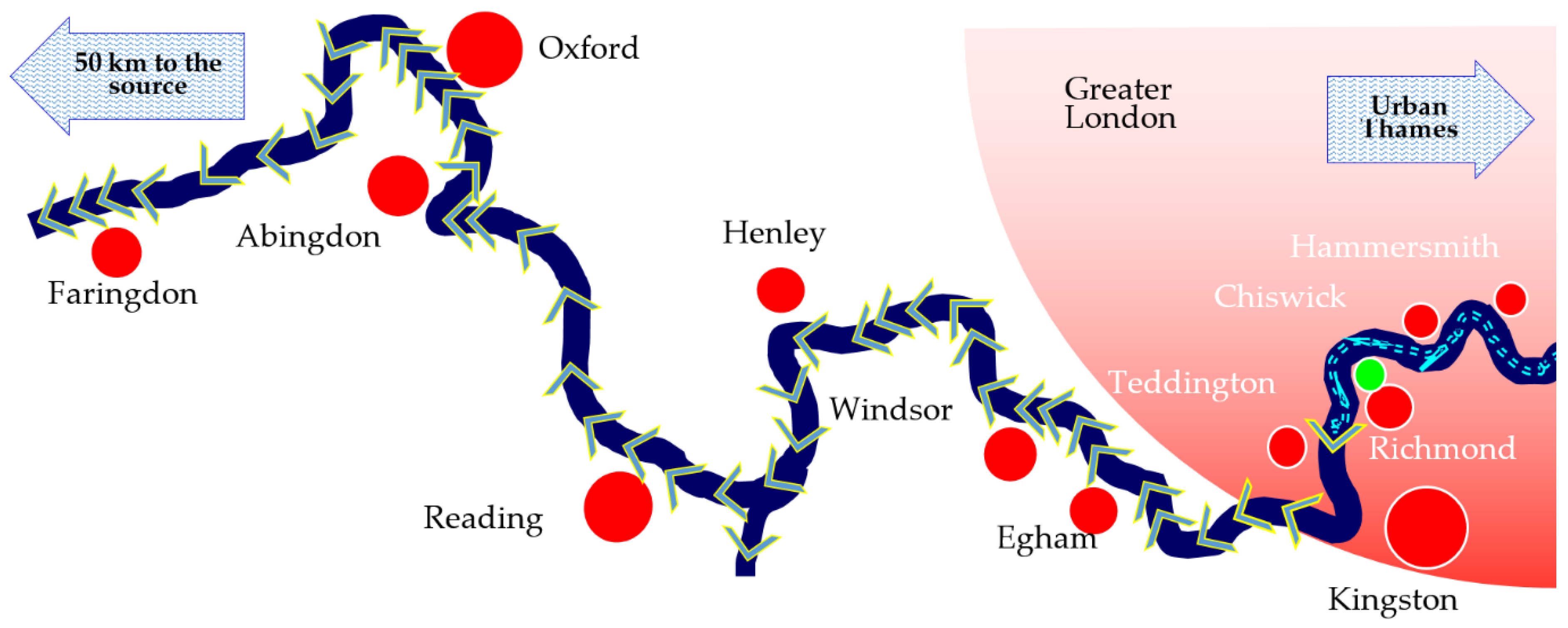
2.1. Recreation, Transport and Water
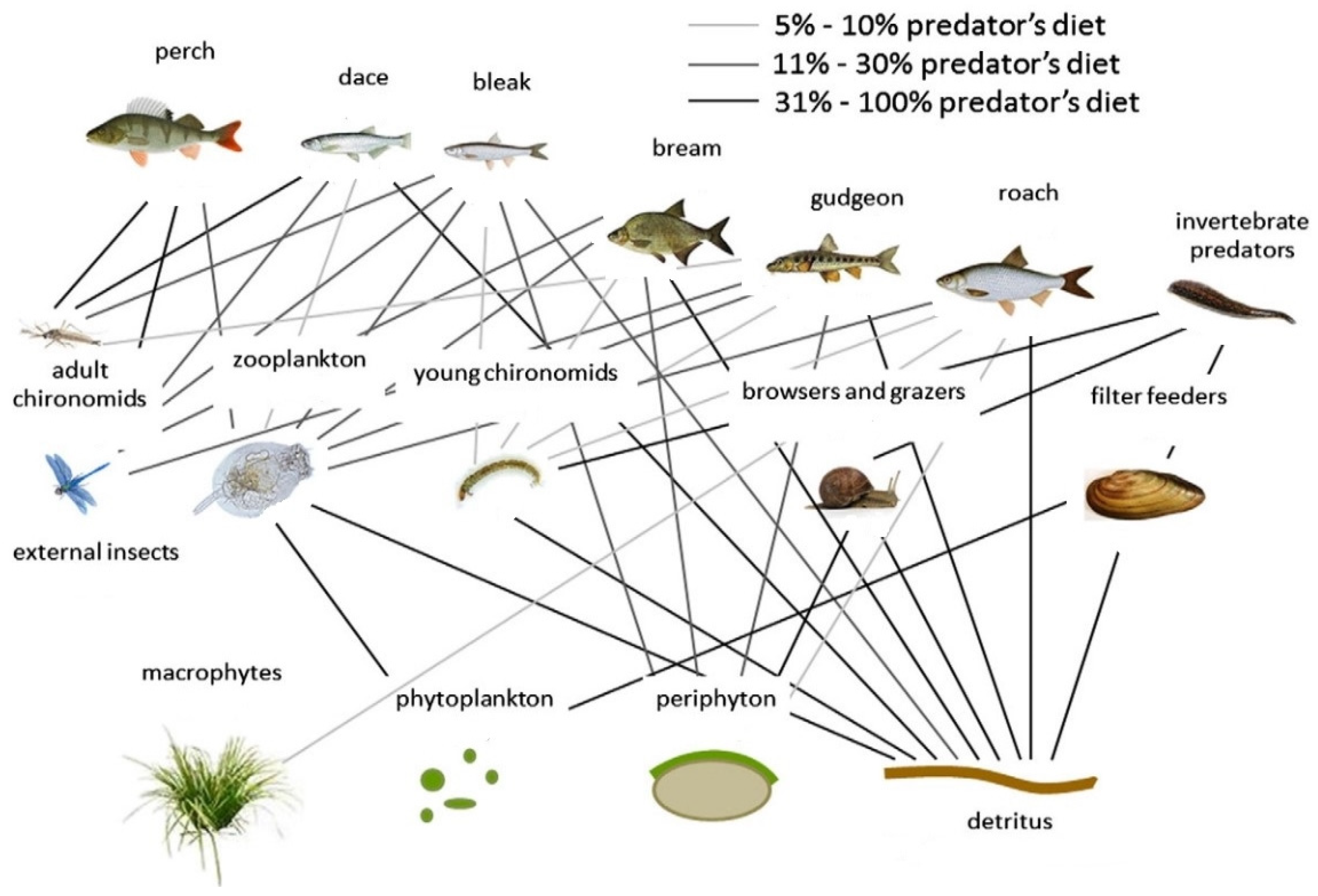
2.2. Fisheries and Ecosystem Decline
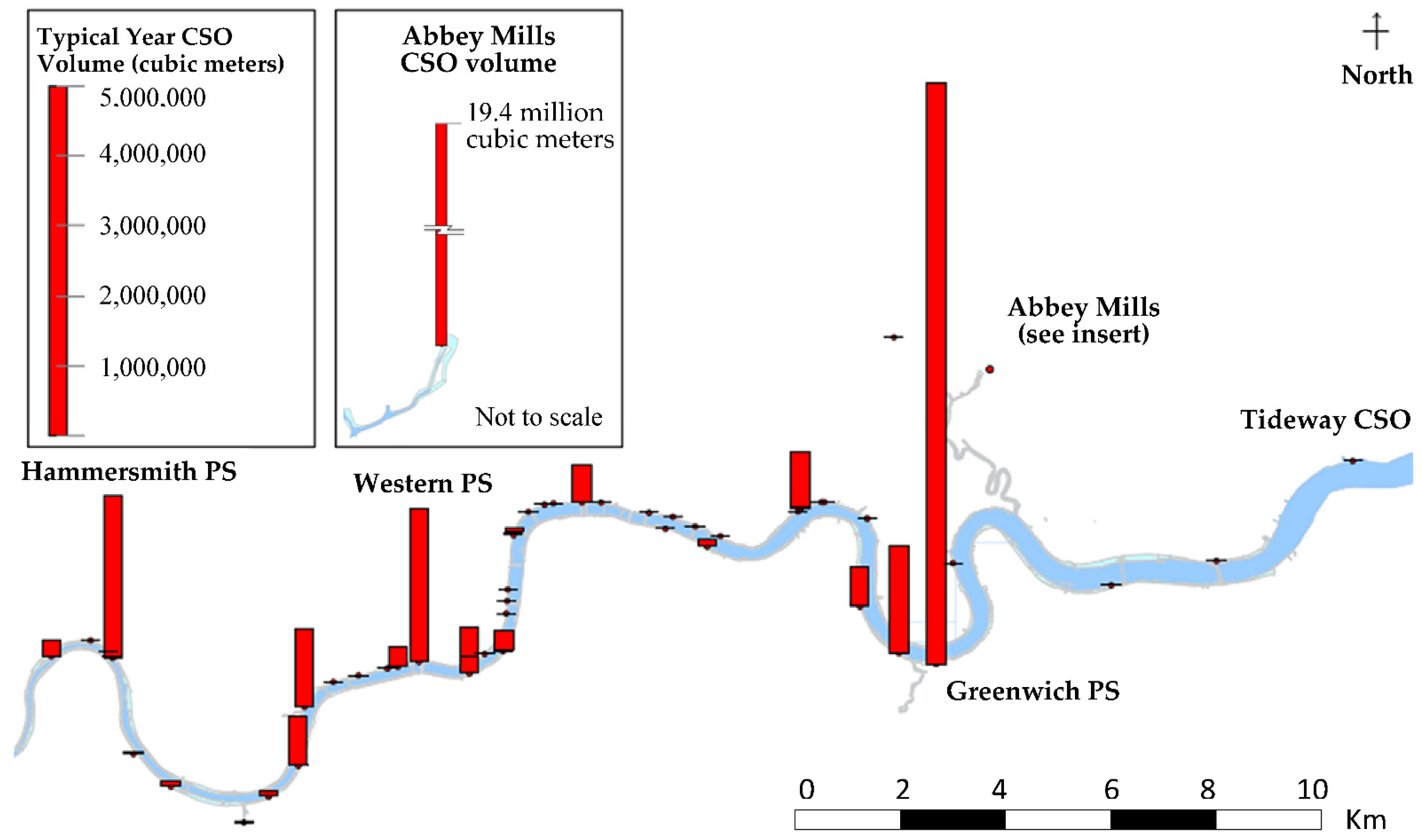
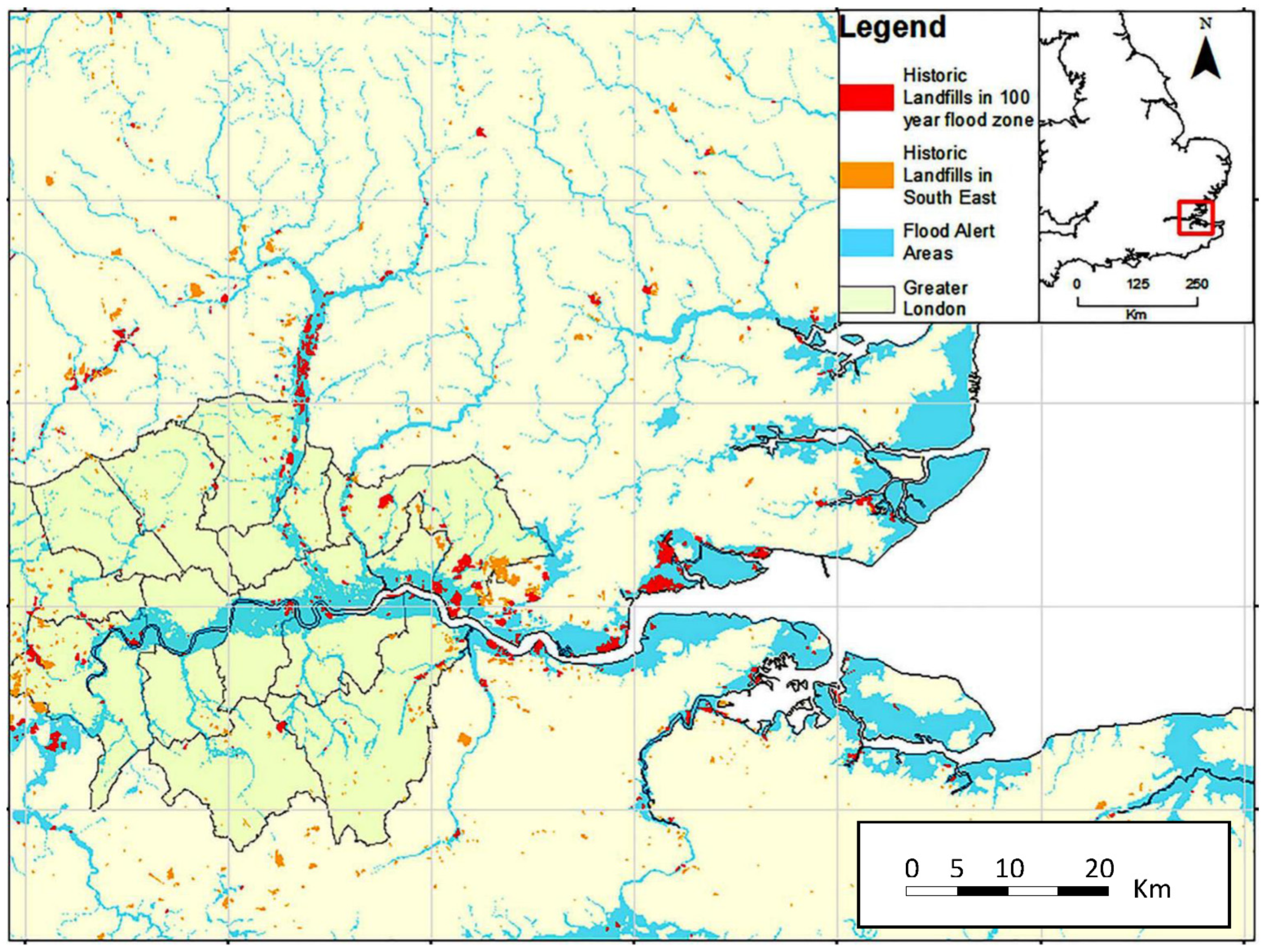
3. Pollution
4. Biodiversity Loss
5. Non-Native Species
6. Past Surveys, Current Management, Sampling and Assessments
7. Remediation, Restoration and Monitoring
8. Conclusions
Supplementary Materials
Funding
Conflicts of Interest
References
- Lavery, S.; Donovan, B. Flood Risk Management in the Thames Estuary Looking Ahead 100 Years. Philos. Trans. R. Soc. A Math. Phys. Eng. Sci. 2005, 363, 1455–1474. [Google Scholar] [CrossRef] [PubMed]
- Dawson, R.J.; Hall, J.W.; Bates, P.D.; Nicholls, R.J. Quantified Analysis of the Probability of Flooding in the Thames Estuary under Imaginable Worst-Case Sea Level Rise Scenarios. Int. J. Water Resour. Dev. 2005, 21, 577–591. [Google Scholar] [CrossRef]
- Canal & Rivers Trust: Fish Species. Available online: https://canalrivertrust.org.uk/enjoy-the-waterways/fishing/fish-species (accessed on 15 May 2021).
- Deaker, D.J.; Agüera, A.; Lin, H.A.; Lawson, C.; Budden, C.; Dworjanyn, S.A.; Mos, B.; Byrne, M. The Hidden Army: Corallivorous Crown-of-Thorns Seastars Can Spend Years as Herbivorous Juveniles. Biol. Lett. 2020, 16, 20190849. [Google Scholar] [CrossRef] [PubMed]
- Attrill, M.J. A Rehabilitated Estuarine Ecosystem: The Environment and Ecology of the Thames Estuary; Kluwer Academic Publishers: London, UK, 1998; pp. 116–121. [Google Scholar]
- Zhang, H.; Han, J.; Chen, F.; Yuan, Q. Review on Plants Selection and Application Effects in Ecological Floating Beds Based on Water Purification. IOP Conf. Ser. Earth Environ. Sci. 2021, 647, 012186. [Google Scholar] [CrossRef]
- Wei, Z.; Van Le, Q.; Peng, W.; Yang, Y.; Yang, H.; Gu, H.; Lam, S.S.; Sonne, C. A Review on Phytoremediation of Contaminants in Air, Water and Soil. J. Hazard. Mater. 2021, 403, 123658. [Google Scholar] [CrossRef]
- Li, J.; Ianaiev, V.; Huff, A.; Zalusky, J.; Ozersky, T.; Katsev, S. Benthic Invaders Control the Phosphorus Cycle in the World’s Largest Freshwater Ecosystem. Proc. Natl. Acad. Sci. USA 2021, 118, e2008223118. [Google Scholar] [CrossRef]
- Vanderploeg, H.A.; Nalepa, T.F.; Jude, D.J.; Mills, E.L.; Holeck, K.T.; Liebig, J.R.; Grigorovich, I.A.; Ojaveer, H. Dispersal and Emerging Ecological Impacts of Ponto-Caspian Species in the Laurentian Great Lakes. Can. J. Fish. Aquat. Sci. 2002, 59, 1209–1228. [Google Scholar] [CrossRef]
- Montgomery, D.R. Soil Erosion and Agricultural Sustainability. Proc. Natl. Acad. Sci. USA 2007, 104, 13268–13272. [Google Scholar] [CrossRef] [PubMed]
- Matrosov, E.S.; Harou, J.J.; Loucks, D.P. A Computationally Efficient Open-Source Water Resource System Simulator-Application to London and the Thames Basin. Environ. Model. Softw. 2011, 26, 1599–1610. [Google Scholar] [CrossRef]
- Department for Environment Food & Rural Affairs (DEFRA). Water Abstraction Plan. Available online: https://www.gov.uk/government/publications/water-abstraction-plan-2017/water-abstraction-plan (accessed on 15 May 2021).
- Griffiths, R. An Essay to Prove That the Jurisdiction and Conservacy of the River of Thames. Printed for T. Longman in Pater-Noster-Row. 1758. Available online: https://thames.me.uk/PDF/1758RiverThamesGriffiths.pdf (accessed on 18 March 2020).
- Bolton, D.K.; Croot, P.E.; Hicks, M.A. A History of the County of Middlesex: Volume 7, Acton, Chiswick, Ealing and Brentford, West Twyford, Willesden. Available online: https://www.british-history.ac.uk/vch/middx/vol7 (accessed on 15 May 2021).
- Wood, L.B. The Restoration of the Tidal Thames.; Hilger: Bristol, UK, 1982; pp. 32–52. [Google Scholar]
- Lombardo, A.; Franco, A.; Pivato, A.; Barausse, A. Food Web Modeling of a River Ecosystem for Risk Assessment of Down-the-Drain Chemicals: A Case Study with AQUATOX. Sci. Total Environ. 2015, 508, 214–227. [Google Scholar] [CrossRef]
- National Biodiversity Network (NBN) Atlas. Available online: https://nbnatlas.org/ (accessed on 10 April 2021).
- Jackson, M.C.; Grey, J. Electronic Supplementary Material Accelerating Rates of Freshwater Invasions in the Catchment of the River Thames List of Freshwater Non-Indigenous Species Recorded as Established in the Thames Catchment. Available online: https://www.academia.edu/2768923/Electronic_Supplementary_Material_for_Jackson_and_Grey_2013_Accelerating_rates_of_freshwater_invasions_in_the_catchment_of_the_River_Thames (accessed on 5 April 2021).
- ZSL (Zoological Society London) Guidance Document Conservation of Tidal Thames Fish through the Planning Process. 2016. Available online: http://www.lbhf.gov.uk/sites/default/files/section_attachments/guidance_document_conservation_of_tidal_thames_fish_through_the_planning_process_october_2016.pdf (accessed on 15 May 2021).
- The Zoological Society London. Available online: https://www.zsl.org/ (accessed on 15 May 2021).
- Taylor, K.G.; Owens, P.N. Sediments in Urban River Basins: A Review of Sediment-Contaminant Dynamics in an Environmental System Conditioned by Human Activities. J. Soils Sediments 2009, 9, 281–303. [Google Scholar] [CrossRef]
- Green, A.E.; Unsworth, R.K.F.; Chadwick, M.A.; Jones, P.J.S. Historical Analysis Exposes Catastrophic Seagrass Loss for the United Kingdom. Front. Plant Sci. 2021, 12, 629962. [Google Scholar] [CrossRef] [PubMed]
- Tideway Tideway-Reconnecting London with the Tidal Thames. Thames Tideway. 2019. Available online: https://www.tideway.london/ (accessed on 18 March 2020).
- Department for Environment Food & Rural Affairs (DEFRA). Creating a River Thames Fit for Our Future: A Strategic and Economic Case for the Thames Tunnel. Available online: https://assets.publishing.service.gov.uk/government/uploads/system/uploads/attachment_data/file/471847/thames-tideway-tunnel-strategic-economic-case.pdf (accessed on 15 May 2021).
- Talling, P. London’s Lost Rivers; Random House Books: London, UK, 2011; pp. 10–50. [Google Scholar]
- Haas, A.F.; Fairoz, M.F.M.; Kelly, L.W.; Nelson, C.E.; Dinsdale, E.A.; Edwards, R.A.; Giles, S.; Hatay, M.; Hisakawa, N.; Knowles, B.; et al. Global Microbialization of Coral Reefs. Nature Microbiol. 2016, 1, 16042. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Armisen, T.; Vercammen, K.; Passerat, J.; Triest, D.; Servais, P.; Cornelis, P. Antimicrobial Resistance of Heterotrophic Bacteria in Sewage-Contaminated Rivers. Water Res. 2011, 45, 788–796. [Google Scholar] [CrossRef]
- Rosi, E.J.; Bechtold, H.A.; Snow, D.; Rojas, M.; Reisinger, A.J.; Kelly, J.J. Urban Stream Microbial Communities Show Resistance to Pharmaceutical Exposure. Ecosphere 2018, 9, e02041. [Google Scholar] [CrossRef]
- Plaas, H.E.; Paerl, H.W. Toxic Cyanobacteria: A Growing Threat to Water and Air Quality. Environ. Sci. Technol. 2021, 55, 44–64. [Google Scholar] [CrossRef]
- Aguilera, A.; Klemenčič, M.; Sueldo, D.J.; Rzymski, P.; Giannuzzi, L.; Martin, M.V. Cell Death in Cyanobacteria: Current Understanding and Recommendations for a Consensus on Its Nomenclature. Front. Microbiol. 2021, 12, 631654. [Google Scholar] [CrossRef] [PubMed]
- Pan, M.; Lyu, T.; Zhan, L.; Matamoros, V.; Angelidaki, I.; Cooper, M.; Pan, G. Mitigating Antibiotic Pollution Using Cyanobacteria: Removal Efficiency, Pathways and Metabolism. Water Res. 2021, 190, 116735. [Google Scholar] [CrossRef]
- Thames Tideway Tunnel Application for Development Consent: Resilience to Change; Thames Water: London, UK, 2013; p. 6.
- Wurtsbaugh, W.A.; Paerl, H.W.; Dodds, W.K. Nutrients, Eutrophication and Harmful Algal Blooms along the Freshwater to Marine Continuum. Wiley Interdiscip. Rev. Water 2019, 6, e1373. [Google Scholar]
- Diaz, R.J.; Rosenberg, R. Spreading Dead Zones and Consequences for Marine Ecosystems. Science 2008, 321, 926–929. [Google Scholar] [CrossRef]
- Brand, J.H.; Spencer, K.L.; O’shea, F.T.; Lindsay, J.E. Potential Pollution Risks of Historic Landfills on Low-Lying Coasts and Estuaries. Wiley Interdiscip. Rev. Water 2018, 5, e1264. [Google Scholar] [CrossRef]
- O’Shea, F.T.; Cundy, A.B.; Spencer, K.L. The Contaminant Legacy from Historic Coastal Landfills and Their Potential as Sources of Diffuse Pollution. Mar. Pollut. Bull. 2018, 128, 446–455. [Google Scholar] [CrossRef] [PubMed]
- Thomas, K.V.; Hurst, M.R.; Matthiessen, P.; McHugh, M.; Smith, A.; Waldock, M.J. An Assessment of in Vitro Androgenic Activity and the Identification of Environmental Androgens in United Kingdom Estuaries. Environ. Toxicol. Chem. 2002, 21, 1456–1461. [Google Scholar] [CrossRef] [PubMed]
- Letsinger, S.; Kay, P.; Rodríguez-Mozaz, S.; Villagrassa, M.; Barceló, D.; Rotchell, J.M. Spatial and Temporal Occurrence of Pharmaceuticals in UK Estuaries. Sci. Total Environ. 2019, 678, 74–84. [Google Scholar] [CrossRef]
- Nielsen, S.V.; Frausing, M.; Henriksen, P.G.; Beedholm, K.; Baatrup, E. The Psychoactive Drug Escitalopram Affects Foraging Behavior in Zebrafish (Danio Rerio). Environ. Toxicol. Chem. 2019, 38, 1902–1910. [Google Scholar] [CrossRef] [PubMed]
- Polverino, G.; Martin, J.M.; Bertram, M.G.; Soman, V.R.; Tan, H.; Brand, J.A.; Mason, R.T.; Wong, B.B.M. Psychoactive Pollution Suppresses Individual Differences in Fish Behaviour. Proc. R. Soc. B Biol. Sci. 2021, 288, 20202294. [Google Scholar] [CrossRef]
- Yin, L.; Wang, B.; Yuan, H.; Deng, S.; Huang, J.; Wang, Y.; Yu, G. Pay Special Attention to the Transformation Products of PPCPs in Environment. Emerg. Contam. 2017, 3, 69–75. [Google Scholar] [CrossRef]
- Fatta-Kassinos, D.; Vasquez, M.I.I.; Kümmerer, K. Transformation Products of Pharmaceuticals in Surface Waters and Wastewater Formed during Photolysis and Advanced Oxidation Processes-Degradation, Elucidation of Byproducts and Assessment of Their Biological Potency. Chemosphere 2011, 85, 693–709. [Google Scholar] [CrossRef]
- Vane, C.H.; Turner, G.H.; Chenery, S.R.; Richardson, M.; Cave, M.C.; Terrington, R.; Gowing, C.J.B.B.; Moss-Hayes, V. Trends in Heavy Metals, Polychlorinated Biphenyls and Toxicity from Sediment Cores of the Inner River Thames Estuary, London, UK. Environ. Sci. Process. Impacts 2020, 22, 364–380. [Google Scholar] [CrossRef]
- Lu, Q.; Jürgens, M.D.; Johnson, A.C.; Graf, C.; Sweetman, A.; Crosse, J.; Whitehead, P. Persistent Organic Pollutants in Sediment and Fish in the River Thames Catchment (UK). Sci. Total Environ. 2017, 576, 78–84. [Google Scholar] [CrossRef]
- Michels, J.; Stippkugel, A.; Lenz, M.; Wirtz, K.; Engel, A. Rapid Aggregation of Biofilm-Covered Microplastics with Marine Biogenic Particles. Proc. R. Soc. B Biol. Sci. 2018, 285, 20181203. [Google Scholar] [CrossRef]
- Kole, P.J.; Löhr, A.J.; Van Belleghem, F.G.A.J.; Ragas, A.M.J. Wear and Tear of Tyres: A Stealthy Source of Microplastics in the Environment. Int. J. Environ. Res. Public Health 2017, 14, 1265. [Google Scholar] [CrossRef] [PubMed]
- Brahney, J.; Mahowald, N.; Prank, M.; Cornwell, G.; Klimont, Z.; Matsui, H.; Prather, K.A. Constraining the Atmospheric Limb of the Plastic Cycle. Proc. Natl. Acad. Sci. USA 2021, 118, e2020719118. [Google Scholar] [CrossRef] [PubMed]
- Wallingford, H.R. Thames 2D Base Model 2012. Available online: https://www.pla.co.uk/assets/NA012_0609_MAL_Thames_2D_base_model.pdf (accessed on 15 May 2021).
- Nogales, B.; Lanfranconi, M.P.; Piña-Villalonga, J.M.; Bosch, R. Anthropogenic Perturbations in Marine Microbial Communities. FEMS Microbiol. Rev. 2010, 35, 275–298. [Google Scholar] [CrossRef]
- Andrews, M.J.; Rickard, D.G. Rehabilitation of the Inner Thames Estuary. Mar. Pollut. Bull. 1980, 11, 327–332. [Google Scholar] [CrossRef]
- Andrews, M.J. Thames estuary: Pollution and recovery. In Effects of Pollutants at the Ecosystem Level; Sheehan, P.J., Miller, D.R., Butler, G.C., Bourdeau, P.H., Eds.; John Wiley & Sons: New York, NY, USA, 1984; pp. 195–227. [Google Scholar]
- Worden, L.; Botsford, L.W.; Hastings, A.; Holland, M.D. Frequency Responses of Age-Structured Populations: Pacific Salmon as an Example. Theor. Popul. Biol. 2010, 78, 239–249. [Google Scholar] [CrossRef]
- Measham, N. Riverfly Census 2015. Salmon Trout Conserv. UK. 2015. Available online: https://salmon-trout.org/wp-content/uploads/2017/08/2015-Report_Riverfly-Census.pdf (accessed on 15 May 2021).
- Ainscough, J.; Barker, J.; Pecorelli, J. Freshwater Bivalve Survey in the Upper Tidal Thames. UK Eur. Conserv. Program. 2015. Available online: https://www.zsl.org/sites/default/files/media/2015-03/Report on ZSLs Freshwater Bivalve Survey in the Upper Tidal Thames 2015.pdf (accessed on 15 May 2021).
- Pecorelli, J. Freshwater Mussel Survey in the Upper Tidal Thames. 2018. Available online: https://www.zsl.org/sites/default/files/media/2018-06/INNS Survey Report_ZSL 2017_Final_07.02.18_0.pdf (accessed on 15 May 2021).
- Cohen, A.N.; Carlton, J.T. Accelerating Invasion Rate in a Highly Invaded Estuary. Science 1998, 279, 555–558. [Google Scholar] [CrossRef] [PubMed]
- Jackson, M.C.; Grey, J. Accelerating Rates of Freshwater Invasions in the Catchment of the River Thames. Biological Invasions 2013, 15, 945–951. [Google Scholar] [CrossRef]
- Pecl, G.T.; Araújo, M.B.; Bell, J.D.; Blanchard, J.; Bonebrake, T.C.; Chen, I.-C.; Clark, T.D.; Colwell, R.K.; Danielsen, F.; Evengård, B.; et al. Biodiversity Redistribution under Climate Change: Impacts on Ecosystems and Human Well-Being. Science 2017, 355, eaai9214. [Google Scholar] [CrossRef] [PubMed]
- Etterson, J.R.; Cornett, M.W.; White, M.A.; Kavajecz, L.C. Assisted Migration across Fixed Seed Zones Detects Adaptation Lags in Two Major North American Tree Species. Ecol. Appl. 2020, 30, e02092. [Google Scholar] [CrossRef]
- Sáenz-Romero, C.; Mendoza-Maya, E.; Gómez-Pineda, E.; Blanco-García, A.; Endara-Agramont, A.R.; Lindig-Cisneros, R.; López-Upton, J.; Trejo-Ramírez, O.; Wehenkel, C.; Cibrián-Tovar, D.; et al. Recent Evidence of Mexican Temperate Forest Decline and the Need for Ex Situ Conservation, Assisted Mi-gration, and Translocation of Species Ensembles as Adaptive Management to Face Projected Climatic Change Impacts in a Megadiverse Country. Can. J. For. Res. 2020, 50, 843–854. [Google Scholar] [CrossRef]
- Sáenz-Romero, C.; O’neill, G.; Aitken, S.N.; Lindig-Cisneros, R. Assisted Migration Field Tests in Canada and Mexico: Lessons, Limitations, and Challenges. Forests 2021, 12, 9. [Google Scholar] [CrossRef]
- Bladon, A.; Donald, P.; Collar, N.; Deng, J.; Dadacha, G.; Wondafrash, M.; Green, R. Climatic change and extinction risk of two globally threatened Ethiopian endemic bird species. PLoS ONE 2021, 16, e0249633. [Google Scholar] [CrossRef]
- Simberloff, D.; Gibbons, L. Now You See Them, Now You Don’t!-Population Crashes of Established Introduced Species. Biol. Invasions 2004, 6, 161–172. [Google Scholar] [CrossRef]
- Weihrauch, D.; Morris, S.; Towle, D.W. Ammonia Excretion in Aquatic and Terrestrial Crabs. J. Exp. Biol. 2004, 207, 4491–4504. [Google Scholar] [CrossRef] [PubMed]
- Briggs, J.C. Rise of Invasive Species Denialism? A Response to Russell and Blackburn. Trends Ecol. Evol. 2017, 32, 231–232. [Google Scholar] [CrossRef]
- Crowley, S.L.; Hinchliffe, S.; Redpath, S.M.; McDonald, R.A. Disagreement About Invasive Species Does Not Equate to Denialism: A Response to Russell and Blackburn. Trends Ecol. Evol. 2017, 32, 228–229. [Google Scholar] [CrossRef] [PubMed]
- Gallardo, B.; Aldridge, D.C. Is Great Britain Heading for a Ponto–Caspian Invasional Meltdown? J. Appl. Ecol. 2015, 52, 41–49. [Google Scholar] [CrossRef]
- Fitzgerald, A. Slipper Limpet Utilisation and Management 2007. Available online: http://www.shellfish.org.uk/files/Literature/Projects-Reports/0701-Slipper_Limpet_Report_Final_Small.pdf (accessed on 15 May 2021).
- Griffiths, A.M.; Ellis, J.S.; Clifton-Dey, D.; Machado-Schiaffino, G.; Bright, D.; Garcia-Vazquez, E.; Stevens, J.R. Restoration versus Recolonisation: The Origin of Atlantic Salmon (Salmo Salar L.) Currently in the River Thames. Biol. Conserv. 2011, 144, 2733–2738. [Google Scholar] [CrossRef]
- Wheeler, A. The Tidal Thames: The History of a River and Its Fishes; Routledge & Kegan Paul: London, UK, 1980; pp. 89–108. [Google Scholar]
- Clark, P.F.; Rainbow, P.S.; Robbins, R.S.; Smith, B.; Yeomanst, W.E.; Thomast, M.; Dobson, G. The Alien Chinese Mitten Crab, Eriocheir Sinensis (Crustacea: Decapoda: Brachyura), in the Thames Catchment. J. Mar. Biol. Ass. UK 1998, 78, 1215–1221. [Google Scholar] [CrossRef]
- Smith, S.R. Organic Contaminants in Sewage Sludge (Biosolids) and Their Significance for Agricultural Recycling. Philos. Trans. R. Soc. A Math. Phys. Eng. Sci. 2009, 367, 4005–4041. [Google Scholar] [CrossRef]
- Richardson, S.D.; Ternes, T.A. Water Analysis: Emerging Contaminants and Current Issues. Anal. Chem. 2018, 90, 398–428. [Google Scholar] [CrossRef]
- Munro, M.; Whitfield, P.; Lee, S. Host-parasite interactions: Case studies of parasitic infections in migratory fish. In A Rehabilitated Estuarine Ecosystem: The Environment and Ecology of the Thames Estuary; Attrill, M.J., Ed.; Springer US: Boston, MA, USA, 1998; pp. 141–167. [Google Scholar]
- Breitburg, D.; Levin, L.A.; Oschlies, A.; Grégoire, M.; Chavez, F.P.; Conley, D.J.; Garçon, V.; Gilbert, D.; Gutiérrez, D.; Isensee, K.; et al. Declining Oxygen in the Global Ocean and Coastal Waters. Science 2018, 359, eaam7240. [Google Scholar] [CrossRef] [PubMed]
- Levin, S.A.; Lubchenco, J. Resilience, Robustness, and Marine Ecosystem-Based Management. Bioscience 2008, 58, 27–32. [Google Scholar] [CrossRef]
- Paniw, M.; James, T.D.; Archer, C.R.; Römer, G.; Levin, S.; Compagnoni, A.; Che, J.; Bennett, C.J.M.; Mooney, A.; Childs, D.Z.; et al. The Myriad of Complex Demographic Responses of Terrestrial Mammals to Climate Change and Gaps of Knowledge: A Global Analysis. J. Anim. Ecol. 2021, Online ahead of print. [CrossRef]
- Chaudhary, C.; Richardson, A.J.; Schoeman, D.S.; Costello, M.J. Global Warming Is Causing a More Pronounced Dip in Marine Species Richness around the Equator. Proc. Natl. Acad. Sci. USA 2021, 118, e2015094118. [Google Scholar] [CrossRef] [PubMed]
- Mastrángelo, M.E.; Pérez-harguindeguy, N.; Enrico, L.; Bennett, E.; Lavorel, S.; Cumming, G.S.; Abeygunawardane, D.; Amarilla, L.D.; Burkhard, B.; Egoh, B.N.; et al. Key Knowledge Gaps to Achieve Global Sustainability Goals. Nat. Sustain. 2019, 2, 1115–1121. [Google Scholar] [CrossRef]
- Department for Environment Food & Rural Affairs (DEFRA). The National Adaptation Programme and the Third Strategy for Climate Adaptation Reporting. 2018. Available online: https://assets.publishing.service.gov.uk/government/uploads/system/uploads/attachment_data/file/727252/national-adaptation-programme-2018.pdf (accessed on 15 May 2021).

| Common Name | Species | Source | Suspected Origin 2 |
|---|---|---|---|
| Wel’s catfish | Silurus glanis (Linnaeus, 1758) | [17] | Stocking |
| Siberian sturgeon | Acipenser baerii (Brandt, 1869) | [3] | Pet trade |
| Sterlet | Acipenser ruthenus (Linnaeus, 1758) | [3] | Pet trade |
| Short-snouted seahorse | Hippocampus hippocampus (Linnaeus, 1758) | [17] | Unknown |
| Bitterling | Rhodeus sericeus (Pallas, 1776) | [17] | Ornamental |
| Koi carp | Cyprinus carpio (Linnaeus, 1758) | [3] | Ornamental |
| Grass carp | Ctenopharyngodon idella (Valenciennes, 1844) | [3] | Stocking |
| Pumpkinseed | Lepomis gibbosus (Linnaeus, 1758) | [3] | Stocking |
| Sunbleak | Leucaspius delineatus (Heckel, 1843) | [17] | Pet trade |
| Topmouth gudgeon | Pseudorasbora parva (Temminck and Schlegel, 1846) | [3] | Pet trade |
| Zander | Sander lucioperca (Linnaeus, 1758) | [3] | Stocking |
| Goldfish | Carassius auratus (Linnaeus, 1758) | [17] | Aquaculture |
| Goldfish × carp hybrid | [18] | ||
| Orfe | Leuciscus idus (Linnaeus, 1758) | [17] | Ornamental |
| Bream × Orfe hybrid | [18] | ||
| Rainbow trout | Oncorhynchus mykiss (Walbaum, 1792) | [17] | Stocking |
| Brook trout | Salvelinus fontinalis (Mitchill, 1814) | [17] | Stocking |
| Fathead minnow | Pimephales promelas (Rafinesque, 1820) | [18] | Ornamental |
| Guppy | Poecilia reticulata (Peters, 1859) | [17] | Ornamental |
| European catfish | Silurus glanis (Linnaeus, 1758) | [17] | Stocking |
| Common Name | Species |
|---|---|
| European smelt | Osmerus eperlanus (Linnaeus, 1758) |
| European eel | Anguilla anguilla (Linnaeus, 1758) |
| Common dace | Leuciscus leuciscus (Linnaeus, 1758) |
| Common goby | Pomatoschistus microps (Krøyer, 1838) |
| Dover sole | Solea solea (Linnaeus, 1758) |
| European seabass | Dicentrarchus labrax (Linnaeus, 1758) |
| European sprat | Sprattus sprattus (Linnaeus, 1758) |
| Flounder | Platichthys flesus (Linnaeus, 1758) |
| Herring | Clupea harengus (Linnaeus, 1758) |
| Stickleback | Gasterosteus aculeatus (Linnaeus, 1758) |
| Pouting | Trisopterus luscus (Linnaeus, 1758) |
| Roach | Rutilus rutilus (Linnaeus, 1758) |
| Sand goby | Pomatoschistus minutus (Pallas, 1770) |
| Whiting | Merlangus merlangus (Linnaeus, 1758) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Richardson, M.; Soloviev, M. The Thames: Arresting Ecosystem Decline and Building Back Better. Sustainability 2021, 13, 6045. https://doi.org/10.3390/su13116045
Richardson M, Soloviev M. The Thames: Arresting Ecosystem Decline and Building Back Better. Sustainability. 2021; 13(11):6045. https://doi.org/10.3390/su13116045
Chicago/Turabian StyleRichardson, Martin, and Mikhail Soloviev. 2021. "The Thames: Arresting Ecosystem Decline and Building Back Better" Sustainability 13, no. 11: 6045. https://doi.org/10.3390/su13116045
APA StyleRichardson, M., & Soloviev, M. (2021). The Thames: Arresting Ecosystem Decline and Building Back Better. Sustainability, 13(11), 6045. https://doi.org/10.3390/su13116045







